 Found 35 hits of ki for UniProtKB: P08575
Found 35 hits of ki for UniProtKB: P08575 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM199180
(US9217012, 10)Show SMILES CCc1ccc(cc1)C(=O)NCCCCC(NC(=O)C(Cc1ccc(cc1)C(F)(F)P(O)(O)=O)NC(=O)C(Cc1ccccc1)NC(=O)COC1CC(C)CCC1C(C)C)C(N)=O Show InChI InChI=1S/C46H62F2N5O9P/c1-5-31-15-19-34(20-16-31)43(56)50-24-10-9-13-37(42(49)55)52-45(58)39(27-33-17-21-35(22-18-33)46(47,48)63(59,60)61)53-44(57)38(26-32-11-7-6-8-12-32)51-41(54)28-62-40-25-30(4)14-23-36(40)29(2)3/h6-8,11-12,15-22,29-30,36-40H,5,9-10,13-14,23-28H2,1-4H3,(H2,49,55)(H,50,56)(H,51,54)(H,52,58)(H,53,57)(H2,59,60,61) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| >1.00E+3 | >-8.18 | n/a | n/a | n/a | n/a | n/a | 7.0 | 25 |
Indiana University Research and Technology Corporation
US Patent
| Assay Description
PTP activity was assayed using p-nitrophenyl phosphate (pNPP) as a substrate in DMG buffer (50 mM DMG, pH 7.0, 1 mM EDTA, 150 mM NaCl, 2 mM DTT, 0.1 ... |
US Patent US9217012 (2015)
BindingDB Entry DOI: 10.7270/Q2FX788H |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50131547

(2-(N-(4-(2-acetamido-3-(4-(5-chloro-3-hydroxy-2-(m...)Show SMILES CCc1cc(CC(NC(C)=O)C(=O)NCCCCOc2cc(Cl)cc(O)c2C(=O)OC)ccc1N(C(=O)C(O)=O)c1ccccc1C(O)=O Show InChI InChI=1S/C34H36ClN3O11/c1-4-21-15-20(11-12-25(21)38(31(42)33(45)46)26-10-6-5-9-23(26)32(43)44)16-24(37-19(2)39)30(41)36-13-7-8-14-49-28-18-22(35)17-27(40)29(28)34(47)48-3/h5-6,9-12,15,17-18,24,40H,4,7-8,13-14,16H2,1-3H3,(H,36,41)(H,37,39)(H,43,44)(H,45,46) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory activity against CD45 tyrosine phosphatase was determined |
J Med Chem 46: 3437-40 (2003)
Article DOI: 10.1021/jm034088d
BindingDB Entry DOI: 10.7270/Q2WW7H1Z |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM26104
(5-{2-cyclohexyl-4-[4-fluoro-3-(trifluoromethyl)phe...)Show SMILES OC(=O)c1cc(on1)-c1ccc(cc1C1CCCCC1)-c1ccc(F)c(c1)C(F)(F)F Show InChI InChI=1S/C23H19F4NO3/c24-19-9-7-15(11-18(19)23(25,26)27)14-6-8-16(21-12-20(22(29)30)28-31-21)17(10-14)13-4-2-1-3-5-13/h6-13H,1-5H2,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.70E+3 | -6.90 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
University of California at Berkeley
| Assay Description
The reaction was started by addition of pNPP substrate, and reaction progress was monitored at 405 nm. The initial rate data collected was used for d... |
J Am Chem Soc 129: 9613-5 (2007)
Article DOI: 10.1021/ja0727520
BindingDB Entry DOI: 10.7270/Q2PG1Q2M |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50069998
(Ammonium; [7-(difluoro-phosphono-methyl)-naphthale...)Show SMILES OP(O)(=O)C(F)(F)c1ccc2cccc(c2c1)C(F)(F)P(O)([O-])=O Show InChI InChI=1S/C12H10F4O6P2/c13-11(14,23(17,18)19)8-5-4-7-2-1-3-10(9(7)6-8)12(15,16)24(20,21)22/h1-6H,(H2,17,18,19)(H2,20,21,22)/p-1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 9.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Toronto
Curated by ChEMBL
| Assay Description
Compound was evaluated for inhibitory constant against CD45 |
Bioorg Med Chem Lett 8: 345-50 (1999)
BindingDB Entry DOI: 10.7270/Q2TD9XV0 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50331945

(Asp-BrPmp-Leu | CHEMBL1289231)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccc(cc1)C(Br)P(O)(O)=O)NC(=O)[C@@H](N)CC(O)=O)C(O)=O |r| Show InChI InChI=1S/C20H29BrN3O9P/c1-10(2)7-15(20(29)30)24-19(28)14(23-18(27)13(22)9-16(25)26)8-11-3-5-12(6-4-11)17(21)34(31,32)33/h3-6,10,13-15,17H,7-9,22H2,1-2H3,(H,23,27)(H,24,28)(H,25,26)(H,29,30)(H2,31,32,33)/t13-,14-,15-,17?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alberta
Curated by ChEMBL
| Assay Description
Inhibition of human CD45-cytoplasmic domain at 35 uM after 10 mins by Kitz-Wilson analysis |
Bioorg Med Chem 18: 8679-86 (2010)
Article DOI: 10.1016/j.bmc.2010.09.040
BindingDB Entry DOI: 10.7270/Q2H995FP |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50118751
(6-(Oxalyl-amino)-1H-indole-7-carboxylic acid | 6-(...)Show InChI InChI=1S/C11H8N2O5/c14-9(11(17)18)13-6-2-1-5-3-4-12-8(5)7(6)10(15)16/h1-4,12H,(H,13,14)(H,15,16)(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50132460
(2-[4-((S)-2-tert-Butoxycarbonylamino-3-{4-[(2-carb...)Show SMILES CCC(CN(C(=O)C(O)=O)c1ccc(C[C@H](NC(=O)OC(C)(C)C)C(=O)NCCCCOc2cccc(O)c2C(=O)OC)cc1)C(O)=O Show InChI InChI=1S/C33H43N3O12/c1-6-21(29(40)41)19-36(28(39)30(42)43)22-14-12-20(13-15-22)18-23(35-32(45)48-33(2,3)4)27(38)34-16-7-8-17-47-25-11-9-10-24(37)26(25)31(44)46-5/h9-15,21,23,37H,6-8,16-19H2,1-5H3,(H,34,38)(H,35,45)(H,40,41)(H,42,43)/t21?,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory potency against CD45 Phosphatase |
Bioorg Med Chem Lett 13: 3129-32 (2003)
BindingDB Entry DOI: 10.7270/Q24B30QV |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50132461
(2-(4-{2-tert-Butoxycarbonylamino-2-[4-(3-hydroxy-2...)Show SMILES COC(=O)c1c(O)cccc1OCCCCNC(=O)C(Cc1ccc(OC(C(O)=O)C(O)=O)cc1)NC(=O)OC(C)(C)C Show InChI InChI=1S/C29H36N2O12/c1-29(2,3)43-28(39)31-19(16-17-10-12-18(13-11-17)42-23(25(34)35)26(36)37)24(33)30-14-5-6-15-41-21-9-7-8-20(32)22(21)27(38)40-4/h7-13,19,23,32H,5-6,14-16H2,1-4H3,(H,30,33)(H,31,39)(H,34,35)(H,36,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory potency against CD45 Phosphatase |
Bioorg Med Chem Lett 13: 3129-32 (2003)
BindingDB Entry DOI: 10.7270/Q24B30QV |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50132465
(5-{(S)-2-((S)-2-tert-Butoxycarbonylamino-3-phenyl-...)Show SMILES COC(=O)c1c(O)cccc1OCCCCNC(=O)[C@H](Cc1ccc(OC(C)C(O)=O)c(c1)C(O)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C Show InChI InChI=1S/C39H47N3O13/c1-23(35(46)47)54-30-17-16-25(20-26(30)36(48)49)22-27(33(44)40-18-9-10-19-53-31-15-11-14-29(43)32(31)37(50)52-5)41-34(45)28(21-24-12-7-6-8-13-24)42-38(51)55-39(2,3)4/h6-8,11-17,20,23,27-28,43H,9-10,18-19,21-22H2,1-5H3,(H,40,44)(H,41,45)(H,42,51)(H,46,47)(H,48,49)/t23?,27-,28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory potency against CD45 Phosphatase |
Bioorg Med Chem Lett 13: 3129-32 (2003)
BindingDB Entry DOI: 10.7270/Q24B30QV |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM14267
(4-bromo-3-(carboxymethoxy)-5-[4-(2-formamidoacetic...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1ccc(NC(=O)CC(O)=O)cc1 Show InChI InChI=1S/C16H12BrNO8S/c17-12-13(26-6-11(22)23)15(16(24)25)27-14(12)7-1-3-8(4-2-7)18-9(19)5-10(20)21/h1-4H,5-6H2,(H,18,19)(H,20,21)(H,22,23)(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50299462

(2-(3-(dihydroxymethyl)naphthalen-2-ylamino)-2-oxoa...)Show InChI InChI=1S/C13H11NO5/c15-11(13(18)19)14-10-6-8-4-2-1-3-7(8)5-9(10)12(16)17/h1-6,12,16-17H,(H,14,15)(H,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 3.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50331944

(CHEMBL1288332 | L-BrPmp-OH)Show SMILES N[C@@H](Cc1ccc(cc1)C(Br)P(O)(O)=O)C(O)=O |r| Show InChI InChI=1S/C10H13BrNO5P/c11-9(18(15,16)17)7-3-1-6(2-4-7)5-8(12)10(13)14/h1-4,8-9H,5,12H2,(H,13,14)(H2,15,16,17)/t8-,9?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Alberta
Curated by ChEMBL
| Assay Description
Inhibition of human CD45-cytoplasmic domain at 150 uM after 10 mins by Kitz-Wilson analysis |
Bioorg Med Chem 18: 8679-86 (2010)
Article DOI: 10.1016/j.bmc.2010.09.040
BindingDB Entry DOI: 10.7270/Q2H995FP |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50118762
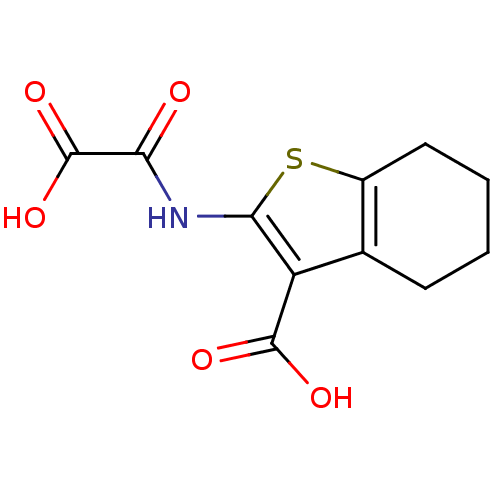
(2-(Oxalyl-amino)-4,5,6,7-tetrahydro-benzo[b]thioph...)Show InChI InChI=1S/C11H11NO5S/c13-8(11(16)17)12-9-7(10(14)15)5-3-1-2-4-6(5)18-9/h1-4H2,(H,12,13)(H,14,15)(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50118778

(5-iodo-2-(oxaloamino)benzoic acid | CHEMBL336908)Show InChI InChI=1S/C9H6INO5/c10-4-1-2-6(5(3-4)8(13)14)11-7(12)9(15)16/h1-3H,(H,11,12)(H,13,14)(H,15,16) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| 4.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50118744
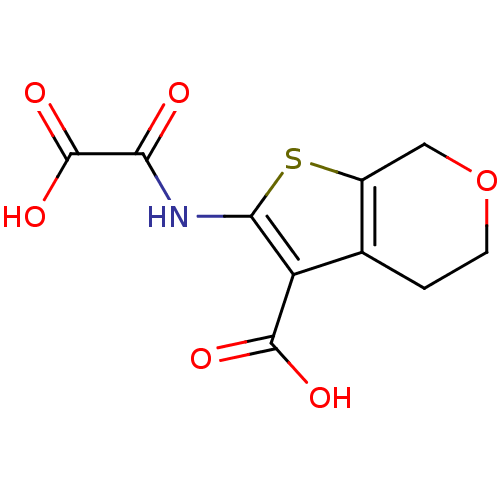
(2-(Oxalyl-amino)-4,7-dihydro-5H-thieno[2,3-c]pyran...)Show InChI InChI=1S/C10H9NO6S/c12-7(10(15)16)11-8-6(9(13)14)4-1-2-17-3-5(4)18-8/h1-3H2,(H,11,12)(H,13,14)(H,15,16) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 5.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM14258

(4-bromo-3-(carboxymethoxy)-5-[4-(pyridine-3-amido)...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1ccc(NC(=O)c2cccnc2)cc1 Show InChI InChI=1S/C19H13BrN2O6S/c20-14-15(28-9-13(23)24)17(19(26)27)29-16(14)10-3-5-12(6-4-10)22-18(25)11-2-1-7-21-8-11/h1-8H,9H2,(H,22,25)(H,23,24)(H,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50118796

(6-(OXALYL-AMINO)-1H-INDOLE-5-CARBOXYLIC ACID | 6-(...)Show InChI InChI=1S/C11H8N2O5/c14-9(11(17)18)13-8-4-7-5(1-2-12-7)3-6(8)10(15)16/h1-4,12H,(H,13,14)(H,15,16)(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 5.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50118792
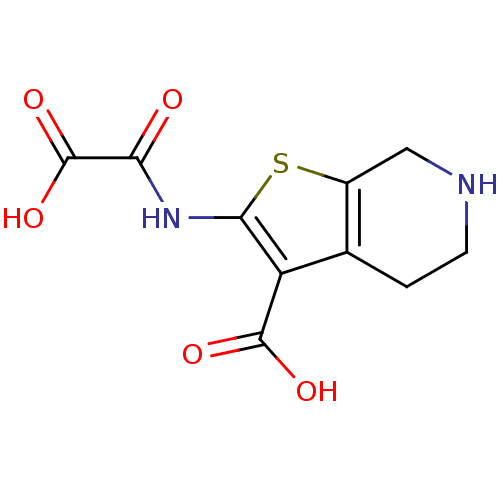
(2-(OXALYL-AMINO)-4,5,6,7-TETRAHYDRO-THIENO[2,3-C]P...)Show InChI InChI=1S/C10H10N2O5S/c13-7(10(16)17)12-8-6(9(14)15)4-1-2-11-3-5(4)18-8/h11H,1-3H2,(H,12,13)(H,14,15)(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 5.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM14255
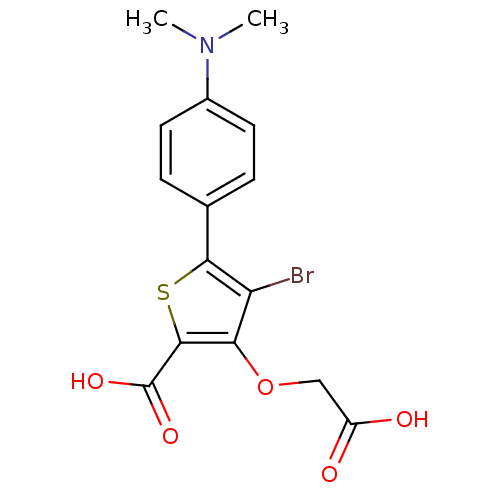
(4-bromo-3-(carboxymethoxy)-5-[4-(dimethylamino)phe...)Show SMILES CN(C)c1ccc(cc1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C15H14BrNO5S/c1-17(2)9-5-3-8(4-6-9)13-11(16)12(22-7-10(18)19)14(23-13)15(20)21/h3-6H,7H2,1-2H3,(H,18,19)(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50219566
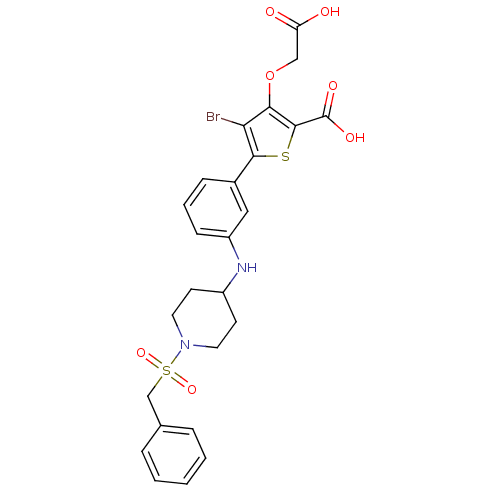
(4-bromo-3-carboxymethoxy-5-[3-(1-phenylmethanesulf...)Show SMILES OC(=O)COc1c(Br)c(sc1C(O)=O)-c1cccc(NC2CCN(CC2)S(=O)(=O)Cc2ccccc2)c1 Show InChI InChI=1S/C25H25BrN2O7S2/c26-21-22(35-14-20(29)30)24(25(31)32)36-23(21)17-7-4-8-19(13-17)27-18-9-11-28(12-10-18)37(33,34)15-16-5-2-1-3-6-16/h1-8,13,18,27H,9-12,14-15H2,(H,29,30)(H,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 7.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CD45 |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50312261
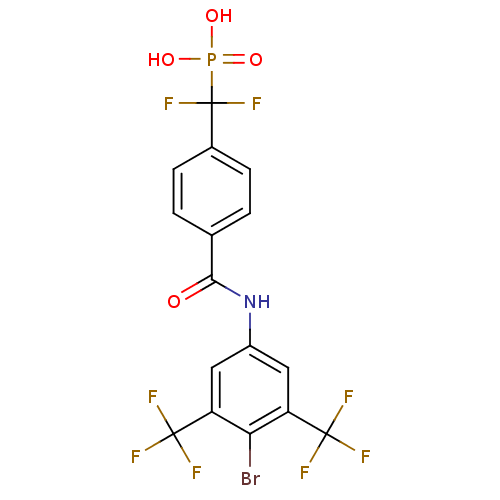
((4-(4-bromo-3,5-bis(trifluoromethyl)phenylcarbamoy...)Show SMILES OP(O)(=O)C(F)(F)c1ccc(cc1)C(=O)Nc1cc(c(Br)c(c1)C(F)(F)F)C(F)(F)F Show InChI InChI=1S/C16H9BrF8NO4P/c17-12-10(14(18,19)20)5-9(6-11(12)15(21,22)23)26-13(27)7-1-3-8(4-2-7)16(24,25)31(28,29)30/h1-6H,(H,26,27)(H2,28,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California
Curated by ChEMBL
| Assay Description
Inhibition of human CD45 |
Bioorg Med Chem Lett 19: 6851-4 (2009)
Article DOI: 10.1016/j.bmcl.2009.10.090
BindingDB Entry DOI: 10.7270/Q21G0MDK |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50219573
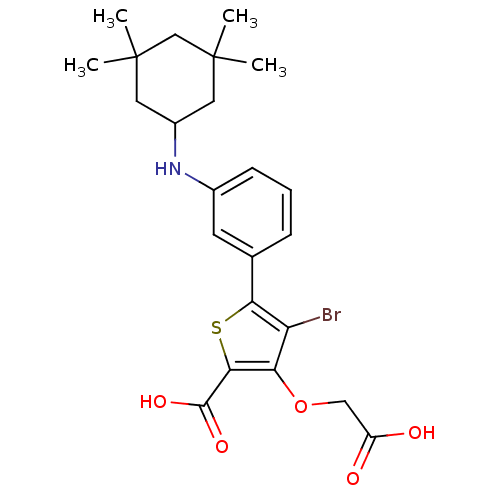
(4-BROMO-3-(CARBOXYMETHOXY)-5-{3-[(3,3,5,5-TETRAMET...)Show SMILES CC1(C)CC(CC(C)(C)C1)Nc1cccc(c1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C23H28BrNO5S/c1-22(2)9-15(10-23(3,4)12-22)25-14-7-5-6-13(8-14)19-17(24)18(30-11-16(26)27)20(31-19)21(28)29/h5-8,15,25H,9-12H2,1-4H3,(H,26,27)(H,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1.51E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CD45 |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50219573

(4-BROMO-3-(CARBOXYMETHOXY)-5-{3-[(3,3,5,5-TETRAMET...)Show SMILES CC1(C)CC(CC(C)(C)C1)Nc1cccc(c1)-c1sc(C(O)=O)c(OCC(O)=O)c1Br Show InChI InChI=1S/C23H28BrNO5S/c1-22(2)9-15(10-23(3,4)12-22)25-14-7-5-6-13(8-14)19-17(24)18(30-11-16(26)27)20(31-19)21(28)29/h5-8,15,25H,9-12H2,1-4H3,(H,26,27)(H,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 1.51E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50118789
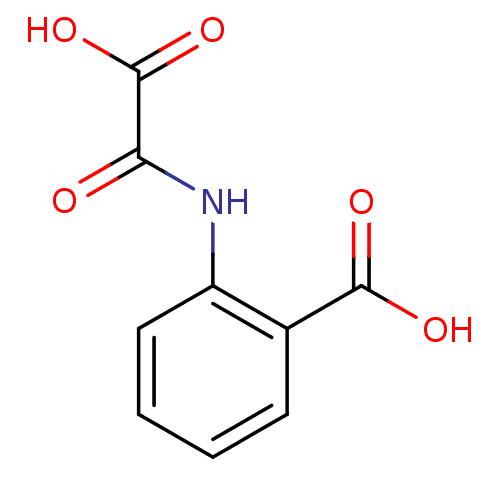
(2-(oxaloamino)benzoic acid | CHEMBL139050)Show InChI InChI=1S/C9H7NO5/c11-7(9(14)15)10-6-4-2-1-3-5(6)8(12)13/h1-4H,(H,10,11)(H,12,13)(H,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 1.60E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50131545
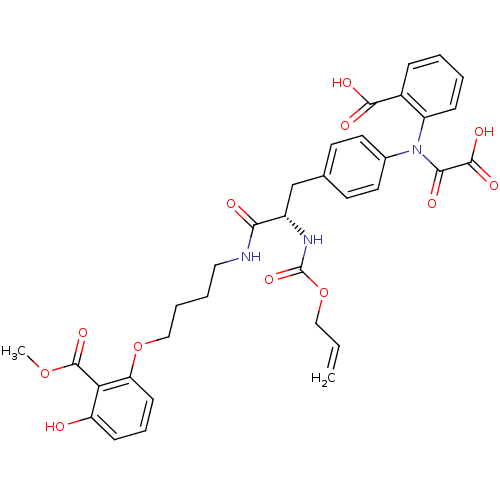
((S)-2-(N-(4-(2-(allyloxycarbonylamino)-3-(4-(3-hyd...)Show SMILES COC(=O)c1c(O)cccc1OCCCCNC(=O)[C@H](Cc1ccc(cc1)N(C(=O)C(O)=O)c1ccccc1C(O)=O)NC(=O)OCC=C Show InChI InChI=1S/C34H35N3O12/c1-3-18-49-34(46)36-24(29(39)35-17-6-7-19-48-27-12-8-11-26(38)28(27)33(45)47-2)20-21-13-15-22(16-14-21)37(30(40)32(43)44)25-10-5-4-9-23(25)31(41)42/h3-5,8-16,24,38H,1,6-7,17-20H2,2H3,(H,35,39)(H,36,46)(H,41,42)(H,43,44)/t24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory activity against CD45 tyrosine phosphatase was determined |
J Med Chem 46: 3437-40 (2003)
Article DOI: 10.1021/jm034088d
BindingDB Entry DOI: 10.7270/Q2WW7H1Z |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50118749
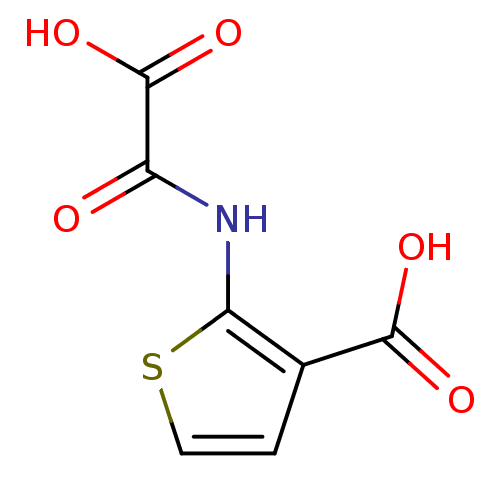
(2-(Oxalyl-amino)-thiophene-3-carboxylic acid | 2-(...)Show InChI InChI=1S/C7H5NO5S/c9-4(7(12)13)8-5-3(6(10)11)1-2-14-5/h1-2H,(H,8,9)(H,10,11)(H,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.10E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50118779
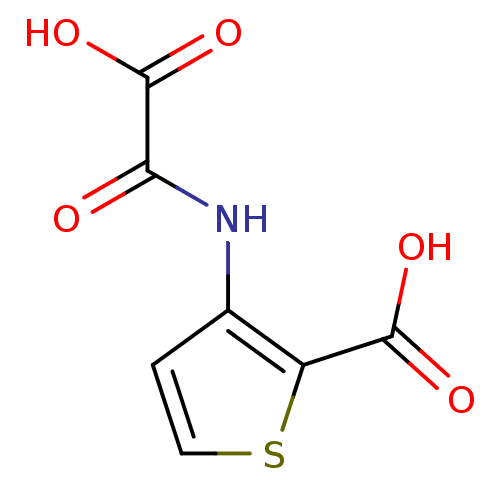
(3-(Oxalyl-amino)-thiophene-2-carboxylic acid | 3-(...)Show InChI InChI=1S/C7H5NO5S/c9-5(7(12)13)8-3-1-2-14-4(3)6(10)11/h1-2H,(H,8,9)(H,10,11)(H,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.60E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50441296
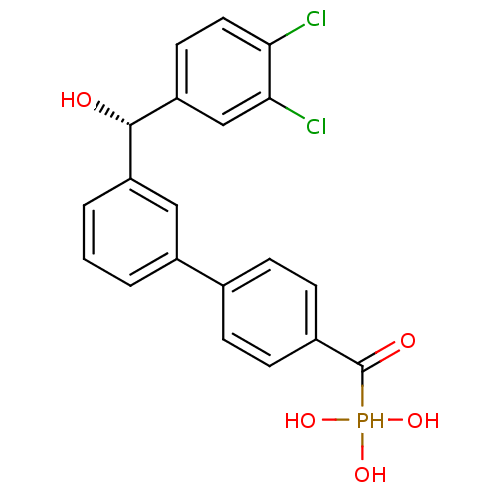
(CHEMBL2431665 | CHEMBL2431667)Show SMILES O[C@@H](c1cccc(c1)-c1ccc(cc1)C(=O)P(O)(O)O)c1ccc(Cl)c(Cl)c1 |r| Show InChI InChI=1S/C20H17Cl2O5P/c21-17-9-8-16(11-18(17)22)19(23)15-3-1-2-14(10-15)12-4-6-13(7-5-12)20(24)28(25,26)27/h1-11,19,23,25-28H/t19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.70E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yale University
Curated by ChEMBL
| Assay Description
Inhibition of human CD45 using pNPP as substrate after 5 mins by spectrophotometric plate reader analysis |
J Med Chem 56: 7636-50 (2013)
Article DOI: 10.1021/jm401037h
BindingDB Entry DOI: 10.7270/Q2HH6MHW |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM14245
(4-bromo-3-(carboxymethoxy)-5-phenylthiophene-2-car...)Show InChI InChI=1S/C13H9BrO5S/c14-9-10(19-6-8(15)16)12(13(17)18)20-11(9)7-4-2-1-3-5-7/h1-5H,6H2,(H,15,16)(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 2.80E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM14245

(4-bromo-3-(carboxymethoxy)-5-phenylthiophene-2-car...)Show InChI InChI=1S/C13H9BrO5S/c14-9-10(19-6-8(15)16)12(13(17)18)20-11(9)7-4-2-1-3-5-7/h1-5H,6H2,(H,15,16)(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 2.80E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant CD45 |
J Med Chem 50: 4681-98 (2007)
Article DOI: 10.1021/jm0702478
BindingDB Entry DOI: 10.7270/Q2TX3F37 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50441297
(CHEMBL2431664 | CHEMBL2431666)Show SMILES O[C@H](c1cccc(c1)-c1ccc(cc1)C(=O)P(O)(O)O)c1ccc(Cl)c(Cl)c1 |r| Show InChI InChI=1S/C20H17Cl2O5P/c21-17-9-8-16(11-18(17)22)19(23)15-3-1-2-14(10-15)12-4-6-13(7-5-12)20(24)28(25,26)27/h1-11,19,23,25-28H/t19-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yale University
Curated by ChEMBL
| Assay Description
Inhibition of human CD45 using pNPP as substrate after 5 mins by spectrophotometric plate reader analysis |
J Med Chem 56: 7636-50 (2013)
Article DOI: 10.1021/jm401037h
BindingDB Entry DOI: 10.7270/Q2HH6MHW |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50299461
((S)-7-(1,1-Dioxo-1H-1lambda*6*-benzo[d]isothiazol-...)Show SMILES OC(=O)C(=O)Nc1sc2[C@H](COC3=NS(=O)(=O)c4ccccc34)OCCc2c1C(O)=O |r,t:12| Show InChI InChI=1S/C18H14N2O9S2/c21-14(18(24)25)19-16-12(17(22)23)9-5-6-28-10(13(9)30-16)7-29-15-8-3-1-2-4-11(8)31(26,27)20-15/h1-4,10H,5-7H2,(H,19,21)(H,22,23)(H,24,25)/t10-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 4.89E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50441298
(CHEMBL2431708)Show SMILES Oc1c(cccc1-c1cccc(c1)C(F)(F)P(O)(O)=O)C1CCCCC1 Show InChI InChI=1S/C19H21F2O4P/c20-19(21,26(23,24)25)15-9-4-8-14(12-15)17-11-5-10-16(18(17)22)13-6-2-1-3-7-13/h4-5,8-13,22H,1-3,6-7H2,(H2,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| >5.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yale University
Curated by ChEMBL
| Assay Description
Inhibition of human CD45 using pNPP as substrate after 5 mins by spectrophotometric plate reader analysis |
J Med Chem 56: 7636-50 (2013)
Article DOI: 10.1021/jm401037h
BindingDB Entry DOI: 10.7270/Q2HH6MHW |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM14230
(5-(carboxymethoxy)-3,7-dithiatricyclo[6.4.0.0^{2,6...)Show InChI InChI=1S/C13H8O5S2/c14-8(15)5-18-9-11-10(20-12(9)13(16)17)6-3-1-2-4-7(6)19-11/h1-4H,5H2,(H,14,15)(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.25E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by ChEMBL
| Assay Description
Inhibition of PTPRC |
J Med Chem 52: 6649-59 (2009)
Article DOI: 10.1021/jm9008899
BindingDB Entry DOI: 10.7270/Q29023T5 |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein phosphatase C
(Homo sapiens (Human)) | BDBM50118750
(2-(Oxalyl-amino)-6-phenethyl-4,5,6,7-tetrahydro-th...)Show SMILES OC(=O)C(=O)Nc1sc2CN(CCc3ccccc3)CCc2c1C(O)=O Show InChI InChI=1S/C18H18N2O5S/c21-15(18(24)25)19-16-14(17(22)23)12-7-9-20(10-13(12)26-16)8-6-11-4-2-1-3-5-11/h1-5H,6-10H2,(H,19,21)(H,22,23)(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.40E+6 | n/a | n/a | n/a | n/a | n/a | n/a | 7.0 | n/a |
Novo Nordisk A/S
Curated by ChEMBL
| Assay Description
Inhibitory effect against human CD45 tyrosine phosphatase using p-nitrophenyl phosphate substrate at a pH 7.0 |
J Med Chem 45: 4443-59 (2002)
BindingDB Entry DOI: 10.7270/Q2QJ7GM5 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data