 Found 802 hits of ki for UniProtKB: P08482
Found 802 hits of ki for UniProtKB: P08482 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50055978
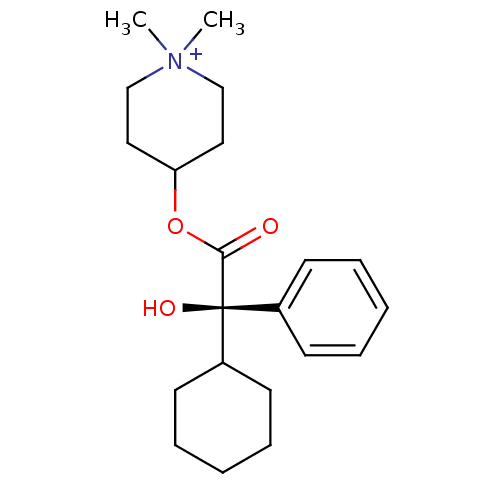
(4-((R)-2-Cyclohexyl-2-hydroxy-2-phenyl-acetoxy)-1,...)Show SMILES C[N+]1(C)CCC(CC1)OC(=O)[C@@](O)(C1CCCCC1)c1ccccc1 Show InChI InChI=1S/C21H32NO3/c1-22(2)15-13-19(14-16-22)25-20(23)21(24,17-9-5-3-6-10-17)18-11-7-4-8-12-18/h3,5-6,9-10,18-19,24H,4,7-8,11-16H2,1-2H3/q+1/t21-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Groningen University Hospital
Curated by ChEMBL
| Assay Description
Binding affinity towards rat Muscarinic acetylcholine receptor M1 was determined |
J Med Chem 40: 117-24 (1997)
Article DOI: 10.1021/jm960374w
BindingDB Entry DOI: 10.7270/Q2VH5PG0 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021919
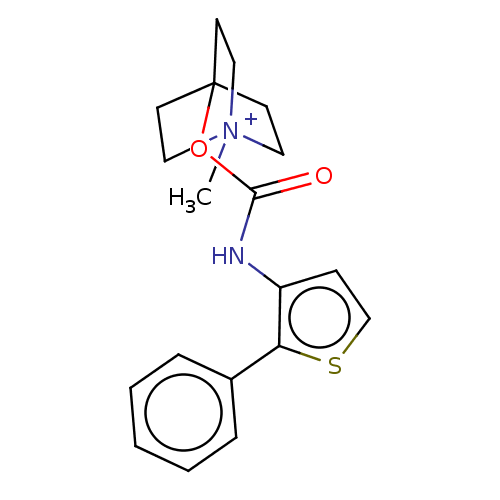
(CHEMBL3298595)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ccsc1-c1ccccc1 Show InChI InChI=1S/C19H22N2O2S.HI/c1-21-11-8-19(9-12-21,10-13-21)23-18(22)20-16-7-14-24-17(16)15-5-3-2-4-6-15;/h2-7,14H,8-13H2,1H3;1H | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50055976
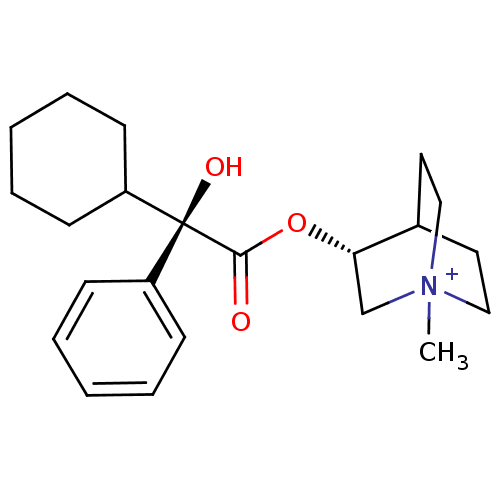
((R)-3-((R)-2-Cyclohexyl-2-hydroxy-2-phenyl-acetoxy...)Show SMILES C[N+]12CCC(CC1)[C@H](C2)OC(=O)[C@@](O)(C1CCCCC1)c1ccccc1 |wU:12.14,wD:12.13,7.10,TLB:9:7:2.3:6.5,(13.94,-4.53,;13.94,-2.99,;14.71,-1.66,;13.24,-1.26,;14.01,.06,;15.34,-.72,;15.34,-2.26,;12.68,-.71,;12.68,-2.25,;11.35,.06,;10.02,-.71,;10.02,-2.26,;8.66,.09,;9.46,1.42,;7.33,.86,;6,.09,;4.67,.86,;4.67,2.4,;6,3.17,;7.33,2.4,;7.89,-1.27,;6.35,-1.27,;5.58,-2.6,;6.35,-3.94,;7.89,-3.95,;8.66,-2.62,)| Show InChI InChI=1S/C22H32NO3/c1-23-14-12-17(13-15-23)20(16-23)26-21(24)22(25,18-8-4-2-5-9-18)19-10-6-3-7-11-19/h2,4-5,8-9,17,19-20,25H,3,6-7,10-16H2,1H3/q+1/t17?,20-,22-,23?/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Groningen University Hospital
Curated by ChEMBL
| Assay Description
Binding affinity towards rat Muscarinic acetylcholine receptor M1 was determined |
J Med Chem 40: 117-24 (1997)
Article DOI: 10.1021/jm960374w
BindingDB Entry DOI: 10.7270/Q2VH5PG0 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50011851
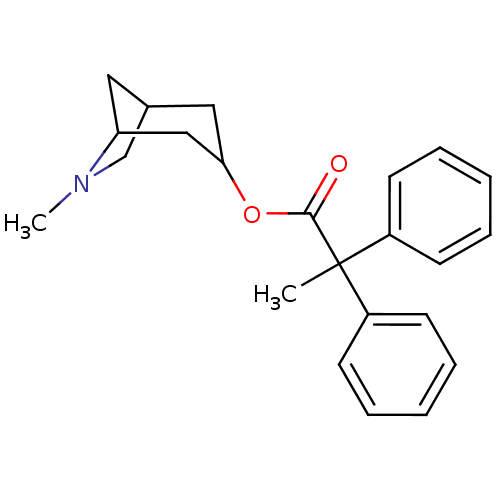
(2,2-Diphenyl-propionic acid 6-methyl-6-aza-bicyclo...)Show SMILES CN1CC2CC1CC(C2)OC(=O)C(C)(c1ccccc1)c1ccccc1 |TLB:9:7:1.2:4,THB:0:1:4:6.7.8| Show InChI InChI=1S/C23H27NO2/c1-23(18-9-5-3-6-10-18,19-11-7-4-8-12-19)22(25)26-21-14-17-13-20(15-21)24(2)16-17/h3-12,17,20-21H,13-16H2,1-2H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0383 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
The compound was tested for binding activity against muscarinic acetylcholine receptor M1, using [3H]QNB as the radioligand. |
J Med Chem 34: 3164-71 (1991)
BindingDB Entry DOI: 10.7270/Q2V988Q7 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021928
(CHEMBL3298599)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ncsc1-c1cccc(Cl)c1 Show InChI InChI=1S/C18H20ClN3O2S.HI/c1-22-8-5-18(6-9-22,7-10-22)24-17(23)21-16-15(25-12-20-16)13-3-2-4-14(19)11-13;/h2-4,11-12H,5-10H2,1H3;1H | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021922
(CHEMBL3298596)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ncsc1-c1ccccc1 Show InChI InChI=1S/C18H21N3O2S.HI/c1-21-10-7-18(8-11-21,9-12-21)23-17(22)20-16-15(24-13-19-16)14-5-3-2-4-6-14;/h2-6,13H,7-12H2,1H3;1H | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021935
(CHEMBL3298600)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ncsc1-c1ccc(F)cc1 Show InChI InChI=1S/C18H20FN3O2S.HI/c1-22-9-6-18(7-10-22,8-11-22)24-17(23)21-16-15(25-12-20-16)13-2-4-14(19)5-3-13;/h2-5,12H,6-11H2,1H3;1H | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021904
(CHEMBL3298588)Show InChI InChI=1S/C18H20N2O2S.ClH/c21-17(22-18-7-10-20(11-8-18)12-9-18)19-15-6-13-23-16(15)14-4-2-1-3-5-14;/h1-6,13H,7-12H2,(H,19,21);1H | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0950 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021938
(CHEMBL3298763)Show SMILES [I-].C[N+]12CCC(CC1)(CC2)OC(=O)Nc1ncsc1-c1cc(Cl)cc(Cl)c1 Show InChI InChI=1S/C18H19Cl2N3O2S.HI/c1-23-5-2-18(3-6-23,4-7-23)25-17(24)22-16-15(26-11-21-16)12-8-13(19)10-14(20)9-12;/h8-11H,2-7H2,1H3;1H | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50369230
(CHEMBL1398637 | CLIDINIUM)Show SMILES C[N+]12CCC(CC1)[C@H](C2)OC(=O)C(O)(c1ccccc1)c1ccccc1 |wU:7.10,THB:9:7:2.3:6.5,(-1.62,-6.55,;-1.6,-5.01,;-3.17,-5.68,;-3.37,-4.27,;-1.87,-3.61,;-1.8,-1.94,;-1.34,-3.07,;-.49,-4.23,;-.2,-5.66,;.72,-3.27,;.49,-1.75,;-.95,-1.19,;1.69,-.79,;.73,.42,;2.9,.17,;2.67,1.69,;3.87,2.65,;5.3,2.09,;5.53,.57,;4.33,-.39,;2.65,-1.99,;4.17,-1.76,;5.13,-2.97,;4.57,-4.4,;3.05,-4.63,;2.09,-3.43,)| Show InChI InChI=1S/C22H26NO3/c1-23-14-12-17(13-15-23)20(16-23)26-21(24)22(25,18-8-4-2-5-9-18)19-10-6-3-7-11-19/h2-11,17,20,25H,12-16H2,1H3/q+1/t17?,20-,23?/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Groningen University Hospital
Curated by ChEMBL
| Assay Description
Binding affinity towards rat Muscarinic acetylcholine receptor M1 was determined |
J Med Chem 40: 117-24 (1997)
Article DOI: 10.1021/jm960374w
BindingDB Entry DOI: 10.7270/Q2VH5PG0 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50450592
(CHEMBL558910)Show SMILES OC(C(=O)O[C@H]1CN2CCC1CC2)(c1ccccc1)c1ccccc1 |r,wD:5.4,(8.38,-12.97,;7.3,-14.07,;8.63,-14.83,;8.64,-16.37,;9.97,-14.06,;11.3,-14.82,;11.3,-16.36,;12.63,-17.13,;13.96,-16.36,;13.96,-14.82,;12.63,-14.05,;13.05,-15.29,;12,-15.64,;5.97,-14.84,;4.63,-14.07,;3.3,-14.84,;3.31,-16.38,;4.65,-17.15,;5.98,-16.37,;7.29,-12.53,;8.62,-11.76,;8.62,-10.22,;7.28,-9.45,;5.95,-10.23,;5.96,-11.77,)| Show InChI InChI=1S/C21H23NO3/c23-20(25-19-15-22-13-11-16(19)12-14-22)21(24,17-7-3-1-4-8-17)18-9-5-2-6-10-18/h1-10,16,19,24H,11-15H2/t19-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University
Curated by ChEMBL
| Assay Description
Displacement of [3H]pirenzepine from muscarinic acetylcholine receptor M1 in rat cortex homogenates |
J Med Chem 34: 2984-9 (1991)
BindingDB Entry DOI: 10.7270/Q27H1K5M |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50450592

(CHEMBL558910)Show SMILES OC(C(=O)O[C@H]1CN2CCC1CC2)(c1ccccc1)c1ccccc1 |r,wD:5.4,(8.38,-12.97,;7.3,-14.07,;8.63,-14.83,;8.64,-16.37,;9.97,-14.06,;11.3,-14.82,;11.3,-16.36,;12.63,-17.13,;13.96,-16.36,;13.96,-14.82,;12.63,-14.05,;13.05,-15.29,;12,-15.64,;5.97,-14.84,;4.63,-14.07,;3.3,-14.84,;3.31,-16.38,;4.65,-17.15,;5.98,-16.37,;7.29,-12.53,;8.62,-11.76,;8.62,-10.22,;7.28,-9.45,;5.95,-10.23,;5.96,-11.77,)| Show InChI InChI=1S/C21H23NO3/c23-20(25-19-15-22-13-11-16(19)12-14-22)21(24,17-7-3-1-4-8-17)18-9-5-2-6-10-18/h1-10,16,19,24H,11-15H2/t19-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oak Ridge National Laboratory
Curated by ChEMBL
| Assay Description
Ability to displace [3H]pirenzepine (PZ) from M1 receptor in rat cortex homogenate |
J Med Chem 36: 848-54 (1993)
BindingDB Entry DOI: 10.7270/Q2S46SM0 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50450592

(CHEMBL558910)Show SMILES OC(C(=O)O[C@H]1CN2CCC1CC2)(c1ccccc1)c1ccccc1 |r,wD:5.4,(8.38,-12.97,;7.3,-14.07,;8.63,-14.83,;8.64,-16.37,;9.97,-14.06,;11.3,-14.82,;11.3,-16.36,;12.63,-17.13,;13.96,-16.36,;13.96,-14.82,;12.63,-14.05,;13.05,-15.29,;12,-15.64,;5.97,-14.84,;4.63,-14.07,;3.3,-14.84,;3.31,-16.38,;4.65,-17.15,;5.98,-16.37,;7.29,-12.53,;8.62,-11.76,;8.62,-10.22,;7.28,-9.45,;5.95,-10.23,;5.96,-11.77,)| Show InChI InChI=1S/C21H23NO3/c23-20(25-19-15-22-13-11-16(19)12-14-22)21(24,17-7-3-1-4-8-17)18-9-5-2-6-10-18/h1-10,16,19,24H,11-15H2/t19-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]pirenzepine from muscarinic M1 receptor of rat cortex homogenates. |
Bioorg Med Chem Lett 7: 979-984 (1997)
Article DOI: 10.1016/S0960-894X(97)00143-1
BindingDB Entry DOI: 10.7270/Q2N29XFM |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50296345

((1R,2R,4S,5R,7S)-7-(2-hydroxy-2,2-di-thiophen-2-yl...)Show SMILES C[N+]1(C)[C@@H]2C[C@H](C[C@@H]1[C@@H]1O[C@H]21)OC(=O)C(O)(c1cccs1)c1cccs1 |TLB:11:5:1:8.10| Show InChI InChI=1S/C19H22NO4S2/c1-20(2)12-9-11(10-13(20)17-16(12)24-17)23-18(21)19(22,14-5-3-7-25-14)15-6-4-8-26-15/h3-8,11-13,16-17,22H,9-10H2,1-2H3/q+1/t11-,12-,13-,16-,17+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021905
(CHEMBL3298589)Show SMILES Cl.O=C(Nc1ccsc1-c1ccccc1)O[C@H]1CN2CCC1CC2 |r,wD:16.16,(41.69,-11.32,;44.26,-6.71,;42.92,-7.48,;41.59,-6.7,;41.6,-5.16,;42.84,-4.27,;42.37,-2.81,;40.83,-2.8,;40.35,-4.27,;38.89,-4.74,;37.74,-3.7,;36.28,-4.18,;35.96,-5.69,;37.11,-6.72,;38.57,-6.24,;42.92,-9.02,;44.25,-9.79,;44.25,-11.32,;45.58,-12.1,;46.92,-11.33,;46.92,-9.78,;45.61,-9.01,;44.85,-10.35,;46.34,-10.75,)| Show InChI InChI=1S/C18H20N2O2S.ClH/c21-18(22-16-12-20-9-6-13(16)7-10-20)19-15-8-11-23-17(15)14-4-2-1-3-5-14;/h1-5,8,11,13,16H,6-7,9-10,12H2,(H,19,21);1H/t16-;/m0./s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50015720
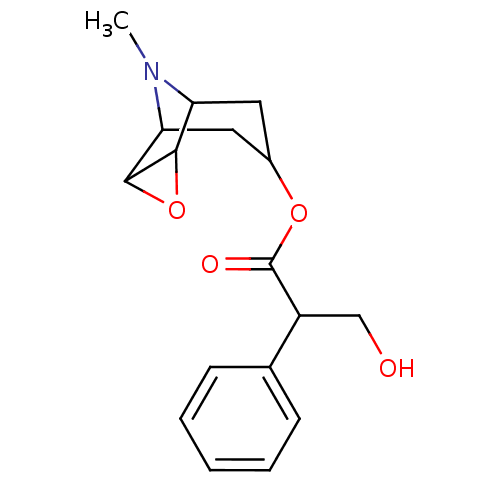
((hyoscine)3-Hydroxy-2-phenyl-propionic acid 9-meth...)Show SMILES CN1C2CC(CC1C1OC21)OC(=O)C(CO)c1ccccc1 |TLB:8:9:1:3.5.4,8:7:1:3.5.4,0:1:9.7:3.5.4,THB:10:4:9.7:1| Show InChI InChI=1S/C17H21NO4/c1-18-13-7-11(8-14(18)16-15(13)22-16)21-17(20)12(9-19)10-5-3-2-4-6-10/h2-6,11-16,19H,7-9H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Group Research Ltd.
Curated by PDSP Ki Database
| |
Mol Pharmacol 38: 805-15 (1990)
BindingDB Entry DOI: 10.7270/Q2H993P0 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50241132
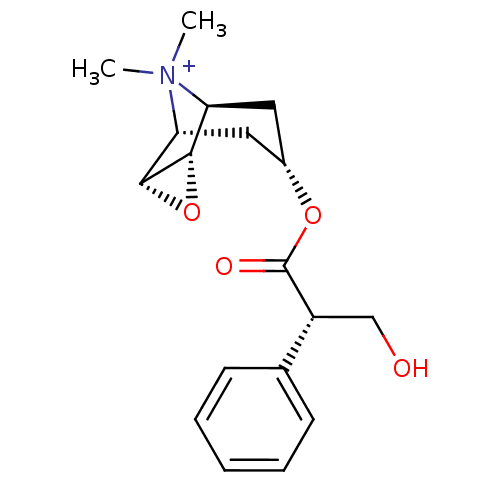
(3-Hydroxy-2-phenyl-propionic acid 9-methyl-3-oxa-9...)Show SMILES C[N+]1(C)[C@H]2C[C@@H](C[C@@H]1[C@H]1O[C@@H]21)OC(=O)[C@H](CO)c1ccccc1 |r,TLB:9:8:4.5.6:1,9:10:4.5.6:1,THB:11:5:1:8.10| Show InChI InChI=1S/C18H24NO4/c1-19(2)14-8-12(9-15(19)17-16(14)23-17)22-18(21)13(10-20)11-6-4-3-5-7-11/h3-7,12-17,20H,8-10H2,1-2H3/q+1/t12-,13-,14-,15+,16-,17+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute for Medical Research
Curated by PDSP Ki Database
| |
Annu Rev Pharmacol Toxicol 30: 633-73 (1990)
Article DOI: 10.1146/annurev.pa.30.040190.003221
BindingDB Entry DOI: 10.7270/Q21Z42WK |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM86231
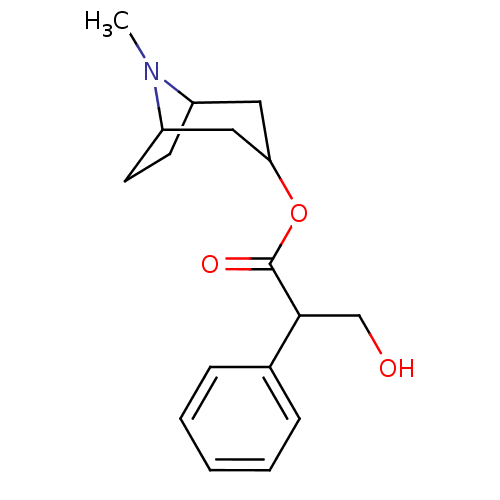
(ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...)Show SMILES CN1C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |THB:9:7:1:3.4| Show InChI InChI=1S/C17H23NO3/c1-18-13-7-8-14(18)10-15(9-13)21-17(20)16(11-19)12-5-3-2-4-6-12/h2-6,13-16,19H,7-11H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Mental Health
Curated by PDSP Ki Database
| |
Mol Pharmacol 35: 469-76 (1989)
BindingDB Entry DOI: 10.7270/Q2V69H2Z |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50368152
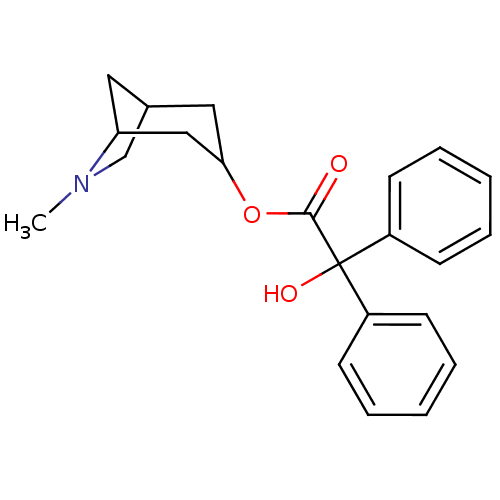
(CHEMBL318812)Show SMILES CN1CC2CC1CC(C2)OC(=O)C(O)(c1ccccc1)c1ccccc1 |THB:9:7:1.2:4,0:1:4:6.7.8| Show InChI InChI=1S/C22H25NO3/c1-23-15-16-12-19(23)14-20(13-16)26-21(24)22(25,17-8-4-2-5-9-17)18-10-6-3-7-11-18/h2-11,16,19-20,25H,12-15H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Binding activity against rat muscarinic acetylcholine receptor M1 using [3H]QNB as the radioligand |
J Med Chem 34: 3164-71 (1991)
BindingDB Entry DOI: 10.7270/Q2V988Q7 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM86698
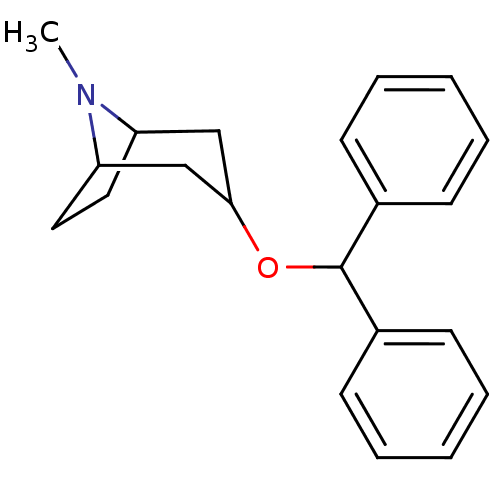
(BENZTROPINE | CAS_132-17-2 | CHEMBL116590 | NSC_23...)Show SMILES CN1C2CCC1CC(C2)OC(c1ccccc1)c1ccccc1 |THB:9:7:1:3.4| Show InChI InChI=1S/C21H25NO/c1-22-18-12-13-19(22)15-20(14-18)23-21(16-8-4-2-5-9-16)17-10-6-3-7-11-17/h2-11,18-21H,12-15H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Group Research Ltd.
Curated by PDSP Ki Database
| |
Mol Pharmacol 38: 805-15 (1990)
BindingDB Entry DOI: 10.7270/Q2H993P0 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50403547
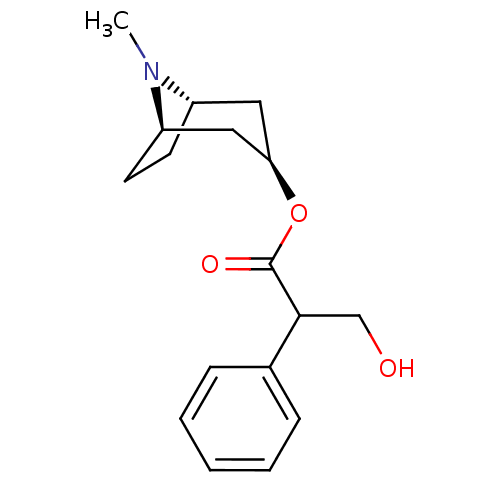
(ATROPEN | ATROPINE)Show SMILES CN1[C@H]2CC[C@@H]1C[C@@H](C2)OC(=O)C(CO)c1ccccc1 |r,THB:9:7:1:3.4| Show InChI InChI=1S/C17H23NO3/c1-18-13-7-8-14(18)10-15(9-13)21-17(20)16(11-19)12-5-3-2-4-6-12/h2-6,13-16,19H,7-11H2,1H3/t13-,14+,15+,16? | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Oak Ridge National Laboratory
Curated by ChEMBL
| Assay Description
Ability to displace [3H]pirenzepine (PZ) from M1 receptor in rat cortex homogenate |
J Med Chem 36: 848-54 (1993)
BindingDB Entry DOI: 10.7270/Q2S46SM0 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50403547

(ATROPEN | ATROPINE)Show SMILES CN1[C@H]2CC[C@@H]1C[C@@H](C2)OC(=O)C(CO)c1ccccc1 |r,THB:9:7:1:3.4| Show InChI InChI=1S/C17H23NO3/c1-18-13-7-8-14(18)10-15(9-13)21-17(20)16(11-19)12-5-3-2-4-6-12/h2-6,13-16,19H,7-11H2,1H3/t13-,14+,15+,16? | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Virginia Commonwealth University
Curated by ChEMBL
| Assay Description
Displacement of [3H]pirenzepine from muscarinic acetylcholine receptor M1 in rat cortex homogenates |
J Med Chem 34: 2984-9 (1991)
BindingDB Entry DOI: 10.7270/Q27H1K5M |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM86231

(ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...)Show SMILES CN1C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |THB:9:7:1:3.4| Show InChI InChI=1S/C17H23NO3/c1-18-13-7-8-14(18)10-15(9-13)21-17(20)16(11-19)12-5-3-2-4-6-12/h2-6,13-16,19H,7-11H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Group Research Ltd.
Curated by PDSP Ki Database
| |
Mol Pharmacol 38: 805-15 (1990)
BindingDB Entry DOI: 10.7270/Q2H993P0 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021892
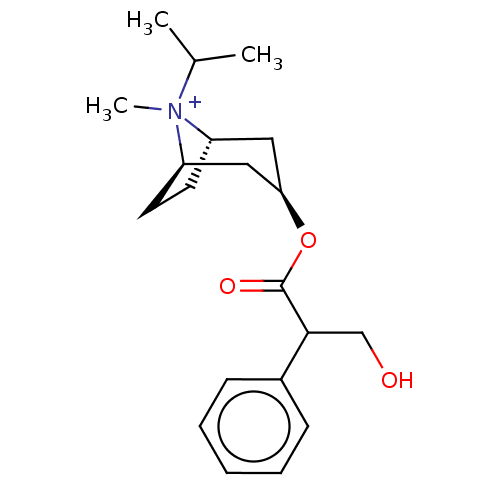
(Ipratropium Bromide)Show SMILES [Br-].[H][C@]12CC[C@]([H])(C[C@@H](C1)OC(=O)C(CO)c1ccccc1)[N+]2(C)C(C)C |TLB:10:8:22:4.3,23:22:8.7.9:4.3,THB:24:22:8.7.9:4.3| Show InChI InChI=1S/C20H30NO3.BrH/c1-14(2)21(3)16-9-10-17(21)12-18(11-16)24-20(23)19(13-22)15-7-5-4-6-8-15;/h4-8,14,16-19,22H,9-13H2,1-3H3;1H/q+1;/p-1/t16-,17+,18+,19?,21?; | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM82379
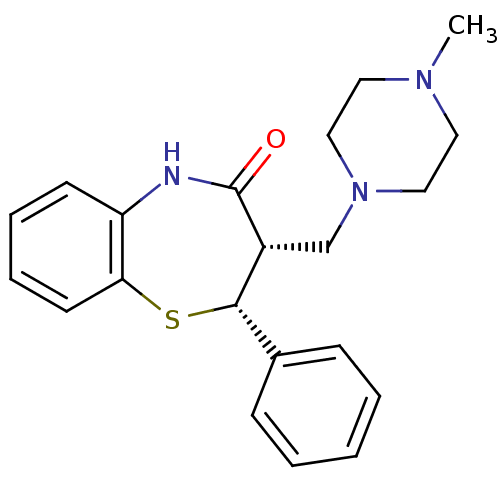
(BTM 1042 hydrochloride | BTM1086 | CAS_87687-13-6)Show SMILES CN1CCN(C[C@@H]2[C@@H](Sc3ccccc3NC2=O)c2ccccc2)CC1 Show InChI InChI=1S/C21H25N3OS/c1-23-11-13-24(14-12-23)15-17-20(16-7-3-2-4-8-16)26-19-10-6-5-9-18(19)22-21(17)25/h2-10,17,20H,11-15H2,1H3,(H,22,25)/t17-,20+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Group Research Ltd.
Curated by PDSP Ki Database
| |
Mol Pharmacol 38: 805-15 (1990)
BindingDB Entry DOI: 10.7270/Q2H993P0 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021926
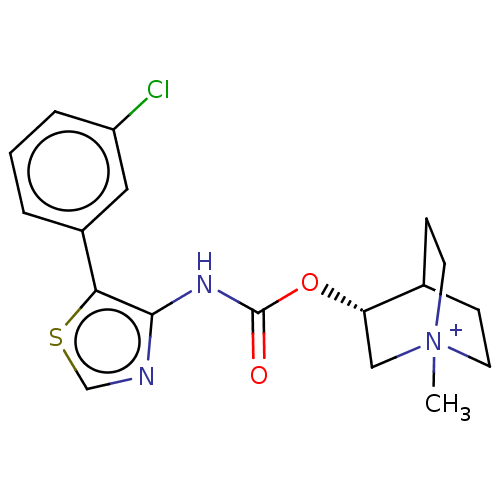
(CHEMBL3298598)Show SMILES C[N+]12CCC(CC1)[C@H](C2)OC(=O)Nc1ncsc1-c1cccc(Cl)c1 |r| Show InChI InChI=1S/C18H20ClN3O2S.HI/c1-22-7-5-12(6-8-22)15(10-22)24-18(23)21-17-16(25-11-20-17)13-3-2-4-14(19)9-13;/h2-4,9,11-12,15H,5-8,10H2,1H3;1H/t12?,15-,22?;/m0./s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50344285
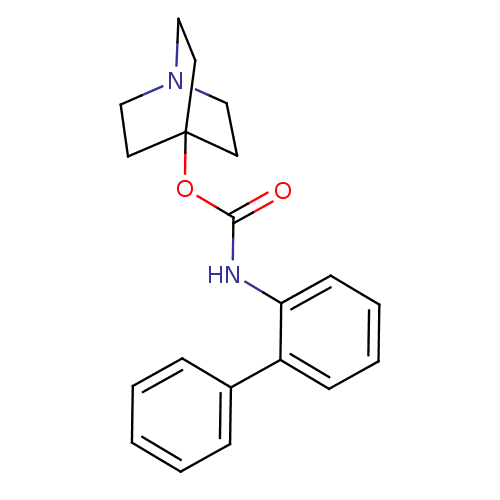
(CHEMBL1779047 | quinuclidin-4-yl biphenyl-2-ylcarb...)Show InChI InChI=1S/C20H22N2O2/c23-19(24-20-10-13-22(14-11-20)15-12-20)21-18-9-5-4-8-17(18)16-6-2-1-3-7-16/h1-9H,10-15H2,(H,21,23) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM86231

(ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...)Show SMILES CN1C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |THB:9:7:1:3.4| Show InChI InChI=1S/C17H23NO3/c1-18-13-7-8-14(18)10-15(9-13)21-17(20)16(11-19)12-5-3-2-4-6-12/h2-6,13-16,19H,7-11H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute for Medical Research
Curated by PDSP Ki Database
| |
Annu Rev Pharmacol Toxicol 30: 633-73 (1990)
Article DOI: 10.1146/annurev.pa.30.040190.003221
BindingDB Entry DOI: 10.7270/Q21Z42WK |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50370683

(CHEMBL1169543)Show SMILES O=C(O[C@H]1CN2CCC1CC2)N1CCc2ccccc2[C@H]1c1ccccc1 |r,wD:20.24,3.2,THB:2:3:7.6:9.10,(-1.33,3.85,;-1.33,2.31,;-2.67,1.54,;-4,2.31,;-4.18,3.72,;-5.76,3.08,;-7.14,3.75,;-6.88,2.32,;-5.51,1.68,;-5.61,0,;-6.05,1.14,;,1.54,;;1.33,-.77,;2.67,,;4,-.77,;5.33,,;5.33,1.54,;4,2.31,;2.67,1.54,;1.33,2.31,;1.33,3.85,;2.67,4.62,;2.67,6.16,;1.33,6.93,;,6.16,;,4.62,)| Show InChI InChI=1S/C23H26N2O2/c26-23(27-21-16-24-13-10-18(21)11-14-24)25-15-12-17-6-4-5-9-20(17)22(25)19-7-2-1-3-8-19/h1-9,18,21-22H,10-16H2/t21-,22+/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for rat cortex muscarinic acetylcholine receptor M1 using [3H]pirenzepine |
J Med Chem 48: 6597-606 (2005)
Article DOI: 10.1021/jm050099q
BindingDB Entry DOI: 10.7270/Q2NP257C |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021893
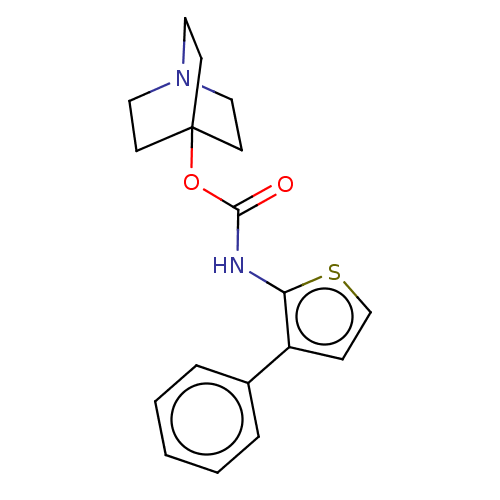
(CHEMBL3298333)Show InChI InChI=1S/C18H20N2O2S/c21-17(22-18-7-10-20(11-8-18)12-9-18)19-16-15(6-13-23-16)14-4-2-1-3-5-14/h1-6,13H,7-12H2,(H,19,21) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021924

(CHEMBL3298597)Show SMILES C[N+]12CCC(CC1)[C@H](C2)OC(=O)Nc1ncsc1-c1ccccc1 |r| Show InChI InChI=1S/C18H21N3O2S.HI/c1-21-9-7-13(8-10-21)15(11-21)23-18(22)20-17-16(24-12-19-17)14-5-3-2-4-6-14;/h2-6,12-13,15H,7-11H2,1H3;1H/t13?,15-,21?;/m0./s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021909
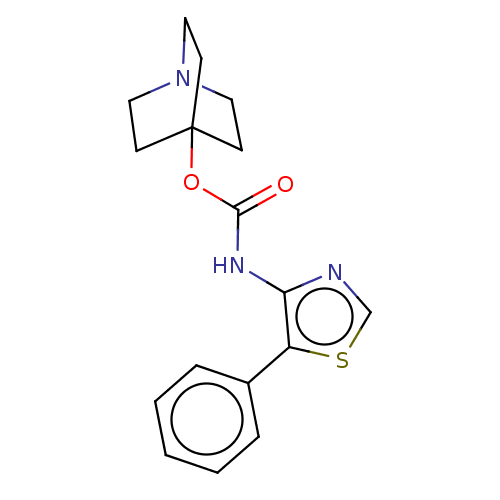
(CHEMBL3298591)Show InChI InChI=1S/C17H19N3O2S/c21-16(22-17-6-9-20(10-7-17)11-8-17)19-15-14(23-12-18-15)13-4-2-1-3-5-13/h1-5,12H,6-11H2,(H,19,21) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50403485

(CHEMBL292857)Show SMILES C[N+]1(C)C2CCC1CC1(C2)OC(=O)C(O1)(c1ccccc1)c1ccccc1 |TLB:14:8:1:4.5,THB:10:8:1:4.5| Show InChI InChI=1S/C23H26NO3/c1-24(2)19-13-14-20(24)16-22(15-19)26-21(25)23(27-22,17-9-5-3-6-10-17)18-11-7-4-8-12-18/h3-12,19-20H,13-16H2,1-2H3/q+1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.398 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was tested for inhibiting [3H]pirenzepine Binding to Muscarinic receptor (M1) receptor in Rat Cortex Homogenates |
Bioorg Med Chem Lett 5: 2325-2330 (1995)
Article DOI: 10.1016/0960-894X(95)00403-G
BindingDB Entry DOI: 10.7270/Q28G8MW2 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50403485

(CHEMBL292857)Show SMILES C[N+]1(C)C2CCC1CC1(C2)OC(=O)C(O1)(c1ccccc1)c1ccccc1 |TLB:14:8:1:4.5,THB:10:8:1:4.5| Show InChI InChI=1S/C23H26NO3/c1-24(2)19-13-14-20(24)16-22(15-19)26-21(25)23(27-22,17-9-5-3-6-10-17)18-11-7-4-8-12-18/h3-12,19-20H,13-16H2,1-2H3/q+1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.398 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was tested for inhibiting [3H]pirenzepine Binding to Muscarinic receptor (M1) receptor in Rat Cortex Homogenates |
Bioorg Med Chem Lett 5: 2325-2330 (1995)
Article DOI: 10.1016/0960-894X(95)00403-G
BindingDB Entry DOI: 10.7270/Q28G8MW2 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50079583
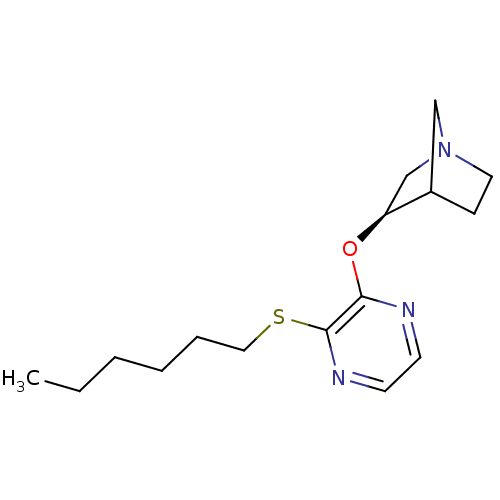
((R)-3-(3-Hexylsulfanyl-pyrazin-2-yloxy)-1-aza-bicy...)Show SMILES CCCCCCSc1nccnc1O[C@H]1CN2CCC1C2 |TLB:13:14:20:18.17| Show InChI InChI=1S/C16H25N3OS/c1-2-3-4-5-10-21-16-15(17-7-8-18-16)20-14-12-19-9-6-13(14)11-19/h7-8,13-14H,2-6,9-12H2,1H3/t13?,14-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth-Ayerst Research
Curated by ChEMBL
| Assay Description
Binding affinity against M1 receptor was determined by displacing [3H]OXO-M in rat cortical homogenate |
Bioorg Med Chem Lett 9: 1895-900 (1999)
BindingDB Entry DOI: 10.7270/Q2G161CX |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50454823
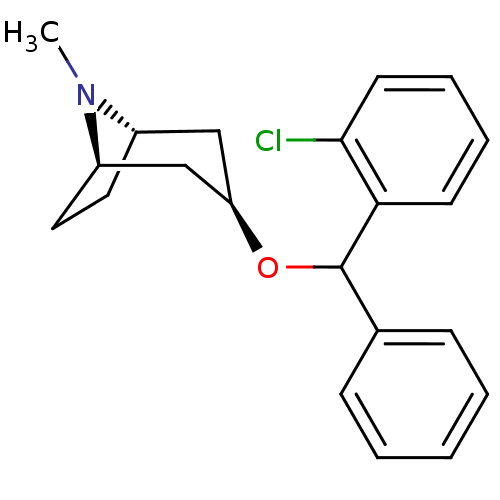
(CHEMBL3084910)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)OC(c1ccccc1)c1ccccc1Cl)N2C |TLB:9:7:24:2.3| Show InChI InChI=1S/C21H24ClNO/c1-23-16-11-12-17(23)14-18(13-16)24-21(15-7-3-2-4-8-15)19-9-5-6-10-20(19)22/h2-10,16-18,21H,11-14H2,1H3/t16-,17+,18+,21? | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institutes of Health
Curated by ChEMBL
| Assay Description
Displacement of [3H]pirenzepine binding at muscarinic Muscarinic acetylcholine receptor M1 in rat brain P2 membranes. |
J Med Chem 40: 851-7 (1997)
Article DOI: 10.1021/jm950782k
BindingDB Entry DOI: 10.7270/Q21R6R54 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50453183
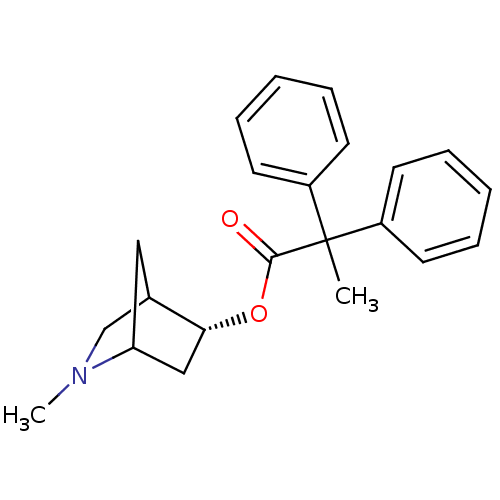
(CHEMBL2114060)Show SMILES CN1CC2CC1C[C@H]2OC(=O)C(C)(c1ccccc1)c1ccccc1 |r,THB:8:7:1.2:4,0:1:7.6:4| Show InChI InChI=1S/C22H25NO2/c1-22(17-9-5-3-6-10-17,18-11-7-4-8-12-18)21(24)25-20-14-19-13-16(20)15-23(19)2/h3-12,16,19-20H,13-15H2,1-2H3/t16?,19?,20-/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.423 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-QNB binding to Muscarinic acetylcholine receptor M1 transfected with CHO cell |
J Med Chem 35: 2184-91 (1992)
BindingDB Entry DOI: 10.7270/Q2B27T74 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50454830
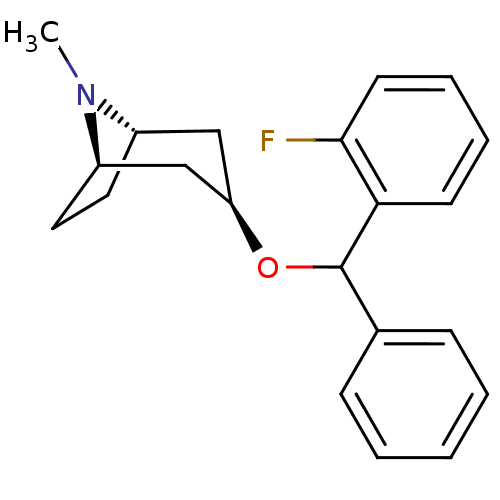
(CHEMBL3084869)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)OC(c1ccccc1)c1ccccc1F)N2C |TLB:9:7:24:2.3| Show InChI InChI=1S/C21H24FNO/c1-23-16-11-12-17(23)14-18(13-16)24-21(15-7-3-2-4-8-15)19-9-5-6-10-20(19)22/h2-10,16-18,21H,11-14H2,1H3/t16-,17+,18+,21? | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institutes of Health
Curated by ChEMBL
| Assay Description
Displacement of [3H]pirenzepine binding at muscarinic Muscarinic acetylcholine receptor M1 in rat brain P2 membranes. |
J Med Chem 40: 851-7 (1997)
Article DOI: 10.1021/jm950782k
BindingDB Entry DOI: 10.7270/Q21R6R54 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50403547

(ATROPEN | ATROPINE)Show SMILES CN1[C@H]2CC[C@@H]1C[C@@H](C2)OC(=O)C(CO)c1ccccc1 |r,THB:9:7:1:3.4| Show InChI InChI=1S/C17H23NO3/c1-18-13-7-8-14(18)10-15(9-13)21-17(20)16(11-19)12-5-3-2-4-6-12/h2-6,13-16,19H,7-11H2,1H3/t13-,14+,15+,16? | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nova Pharmaceutical Corporation
Curated by ChEMBL
| Assay Description
Evaluated for the phosphatidyl inositol turnover at Muscarinic acetylcholine receptor M1 in rat cortex |
J Med Chem 31: 1463-6 (1988)
BindingDB Entry DOI: 10.7270/Q2DV1KGN |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50408535
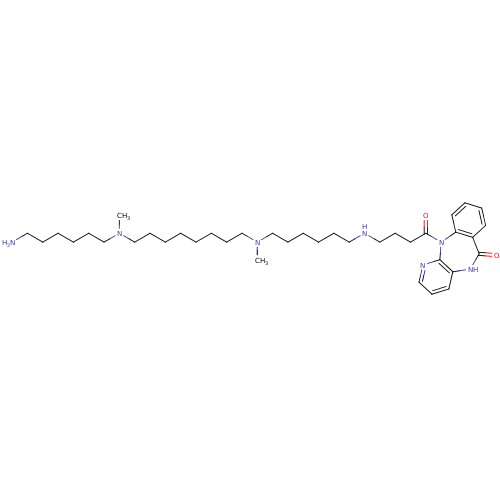
(CHEMBL131865)Show SMILES CN(CCCCCCN)CCCCCCCCN(C)CCCCCCNCCCC(=O)N1c2ccccc2C(=O)Nc2cccnc12 Show InChI InChI=1S/C38H63N7O2/c1-43(31-17-9-5-13-25-39)29-15-7-3-4-8-16-30-44(2)32-18-10-6-14-26-40-27-20-24-36(46)45-35-23-12-11-21-33(35)38(47)42-34-22-19-28-41-37(34)45/h11-12,19,21-23,28,40H,3-10,13-18,20,24-27,29-32,39H2,1-2H3,(H,42,47) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Bologna
Curated by ChEMBL
| Assay Description
Binding affinity for rat cortex Muscarinic acetylcholine receptor M1 |
J Med Chem 41: 4150-60 (1998)
Checked by Author
Article DOI: 10.1021/jm981038d
BindingDB Entry DOI: 10.7270/Q2KH0PJH |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50454825
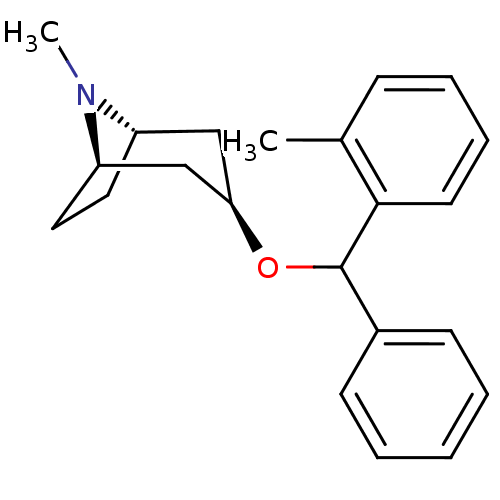
(CHEMBL3084878)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)OC(c1ccccc1)c1ccccc1C)N2C |TLB:9:7:24:3.2| Show InChI InChI=1S/C22H27NO/c1-16-8-6-7-11-21(16)22(17-9-4-3-5-10-17)24-20-14-18-12-13-19(15-20)23(18)2/h3-11,18-20,22H,12-15H2,1-2H3/t18-,19+,20+,22? | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institutes of Health
Curated by ChEMBL
| Assay Description
Displacement of [3H]pirenzepine binding at muscarinic Muscarinic acetylcholine receptor M1 in rat brain P2 membranes. |
J Med Chem 40: 851-7 (1997)
Article DOI: 10.1021/jm950782k
BindingDB Entry DOI: 10.7270/Q21R6R54 |
More data for this
Ligand-Target Pair | |
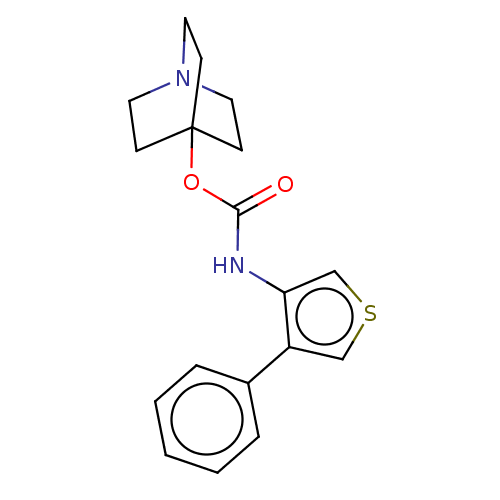
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50021901
(CHEMBL3298334)Show InChI InChI=1S/C18H20N2O2S.ClH/c21-17(22-18-6-9-20(10-7-18)11-8-18)19-16-13-23-12-15(16)14-4-2-1-3-5-14;/h1-5,12-13H,6-11H2,(H,19,21);1H | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]-NMS from Sprague-Dawley rat muscarinic M1 receptor in cortex membrane |
Bioorg Med Chem 22: 3478-87 (2014)
Article DOI: 10.1016/j.bmc.2014.04.031
BindingDB Entry DOI: 10.7270/Q2XS5X00 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM86231

(ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...)Show SMILES CN1C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |THB:9:7:1:3.4| Show InChI InChI=1S/C17H23NO3/c1-18-13-7-8-14(18)10-15(9-13)21-17(20)16(11-19)12-5-3-2-4-6-12/h2-6,13-16,19H,7-11H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic Jacksonville
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 257: 1121-9 (1991)
BindingDB Entry DOI: 10.7270/Q2G73C6R |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50452855

(Isoptpo Hyoscine | Scopolamine)Show SMILES [H][C@@]12O[C@@]1([H])[C@]1([H])C[C@@H](C[C@@]2([H])N1C)OC(=O)[C@H](CO)c1ccccc1 |r,TLB:14:8:12:1.3,2:1:12:8.7.9,2:3:12:8.7.9| Show InChI InChI=1S/C17H21NO4/c1-18-13-7-11(8-14(18)16-15(13)22-16)21-17(20)12(9-19)10-5-3-2-4-6-10/h2-6,11-16,19H,7-9H2,1H3/t11-,12-,13-,14+,15+,16+/m1/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Groningen University Hospital
Curated by ChEMBL
| Assay Description
Binding affinity towards rat Muscarinic acetylcholine receptor M1 was determined |
J Med Chem 40: 117-24 (1997)
Article DOI: 10.1021/jm960374w
BindingDB Entry DOI: 10.7270/Q2VH5PG0 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM86231

(ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...)Show SMILES CN1C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |THB:9:7:1:3.4| Show InChI InChI=1S/C17H23NO3/c1-18-13-7-8-14(18)10-15(9-13)21-17(20)16(11-19)12-5-3-2-4-6-12/h2-6,13-16,19H,7-11H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona Health Science Center
Curated by PDSP Ki Database
| |
Life Sci 50: 355-63 (1992)
Article DOI: 10.1016/0024-3205(92)90437-t
BindingDB Entry DOI: 10.7270/Q2XK8D19 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50240680
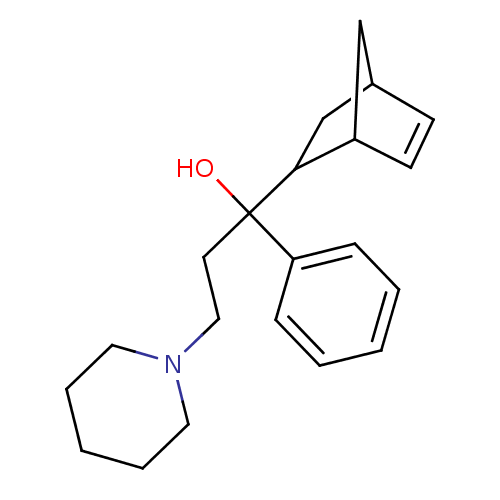
(1-(bicyclo[2.2.1]hept-5-en-2-yl)-1-phenyl-3-(piper...)Show SMILES OC(CCN1CCCCC1)(C1CC2CC1C=C2)c1ccccc1 |c:17| Show InChI InChI=1S/C21H29NO/c23-21(19-7-3-1-4-8-19,11-14-22-12-5-2-6-13-22)20-16-17-9-10-18(20)15-17/h1,3-4,7-10,17-18,20,23H,2,5-6,11-16H2 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
| PubMed
| 0.540 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Group Research Ltd.
Curated by PDSP Ki Database
| |
Mol Pharmacol 38: 805-15 (1990)
BindingDB Entry DOI: 10.7270/Q2H993P0 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM86231

(ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...)Show SMILES CN1C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |THB:9:7:1:3.4| Show InChI InChI=1S/C17H23NO3/c1-18-13-7-8-14(18)10-15(9-13)21-17(20)16(11-19)12-5-3-2-4-6-12/h2-6,13-16,19H,7-11H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 250: 565-72 (1989)
BindingDB Entry DOI: 10.7270/Q2GX492D |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50176065
(4-DAMP | 4-Diphenylacetoxy-1,1-dimethyl-piperidini...)Show InChI InChI=1S/C21H26NO2/c1-22(2)15-13-19(14-16-22)24-21(23)20(17-9-5-3-6-10-17)18-11-7-4-8-12-18/h3-12,19-20H,13-16H2,1-2H3/q+1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| | 0.589 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of binding of [3H]pirenzepine to muscarinic receptor (M1) in rat cortex homogenates |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2ZK5JTF |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50176065

(4-DAMP | 4-Diphenylacetoxy-1,1-dimethyl-piperidini...)Show InChI InChI=1S/C21H26NO2/c1-22(2)15-13-19(14-16-22)24-21(23)20(17-9-5-3-6-10-17)18-11-7-4-8-12-18/h3-12,19-20H,13-16H2,1-2H3/q+1 | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
| 0.589 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was tested for inhibiting [3H]pirenzepine Binding to Muscarinic receptor (M1) receptor in Rat Cortex Homogenates |
Bioorg Med Chem Lett 5: 2325-2330 (1995)
Article DOI: 10.1016/0960-894X(95)00403-G
BindingDB Entry DOI: 10.7270/Q28G8MW2 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(RAT) | BDBM50366775
(BENZTROPINE | Benzatropine)Show SMILES CN1[C@H]2CC[C@@H]1C[C@@H](C2)OC(c1ccccc1)c1ccccc1 |r| Show InChI InChI=1S/C21H25NO/c1-22-18-12-13-19(22)15-20(14-18)23-21(16-8-4-2-5-9-16)17-10-6-3-7-11-17/h2-11,18-21H,12-15H2,1H3/t18-,19+,20+ | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institutes of Health
Curated by ChEMBL
| Assay Description
Displacement of [3H]pirenzepine binding at muscarinic Muscarinic acetylcholine receptor M1 in rat brain P2 membranes. |
J Med Chem 40: 851-7 (1997)
Article DOI: 10.1021/jm950782k
BindingDB Entry DOI: 10.7270/Q21R6R54 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data