 Found 58 hits Enz. Inhib. hit(s) with all data for entry = 50034566
Found 58 hits Enz. Inhib. hit(s) with all data for entry = 50034566 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364330
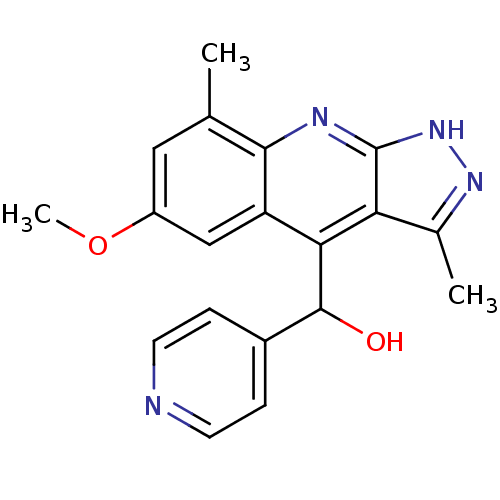
(CHEMBL1949936)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ccncc3)c2c1 Show InChI InChI=1S/C19H18N4O2/c1-10-8-13(25-3)9-14-16(18(24)12-4-6-20-7-5-12)15-11(2)22-23-19(15)21-17(10)14/h4-9,18,24H,1-3H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364327
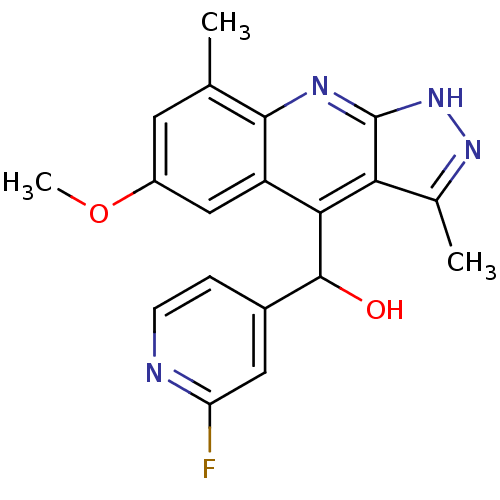
(CHEMBL1949939)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ccnc(F)c3)c2c1 Show InChI InChI=1S/C19H17FN4O2/c1-9-6-12(26-3)8-13-16(18(25)11-4-5-21-14(20)7-11)15-10(2)23-24-19(15)22-17(9)13/h4-8,18,25H,1-3H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364339
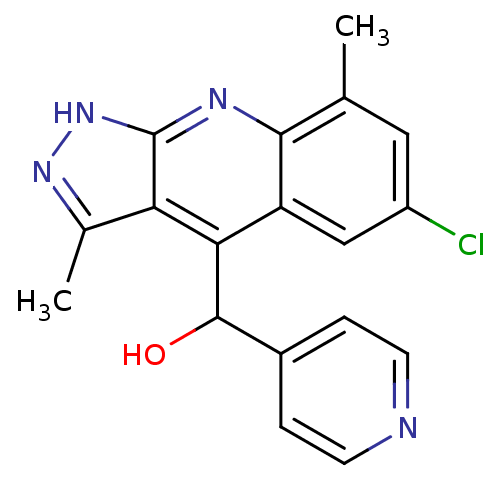
(CHEMBL1950085)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3ccncc3)c12 Show InChI InChI=1S/C18H15ClN4O/c1-9-7-12(19)8-13-15(17(24)11-3-5-20-6-4-11)14-10(2)22-23-18(14)21-16(9)13/h3-8,17,24H,1-2H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364340
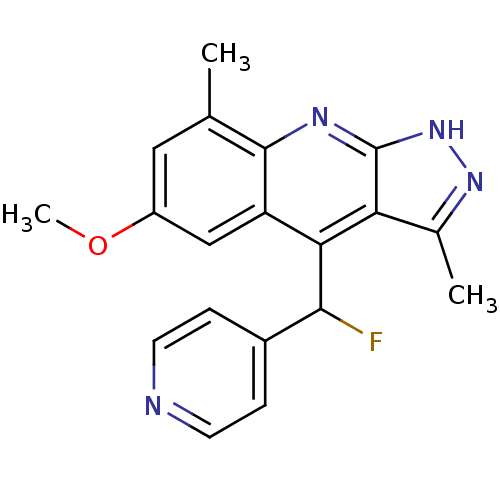
(CHEMBL1950084)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(F)c3ccncc3)c2c1 Show InChI InChI=1S/C19H17FN4O/c1-10-8-13(25-3)9-14-16(17(20)12-4-6-21-7-5-12)15-11(2)23-24-19(15)22-18(10)14/h4-9,17H,1-3H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364335
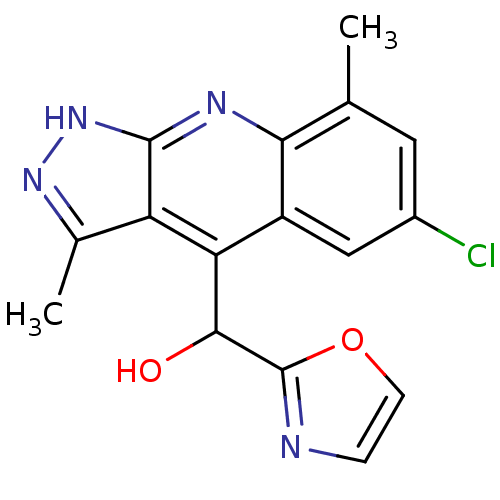
(CHEMBL1950089)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3ncco3)c12 Show InChI InChI=1S/C16H13ClN4O2/c1-7-5-9(17)6-10-12(14(22)16-18-3-4-23-16)11-8(2)20-21-15(11)19-13(7)10/h3-6,14,22H,1-2H3,(H,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364332

(CHEMBL1949940)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ncco3)c2c1 Show InChI InChI=1S/C17H16N4O3/c1-8-6-10(23-3)7-11-13(15(22)17-18-4-5-24-17)12-9(2)20-21-16(12)19-14(8)11/h4-7,15,22H,1-3H3,(H,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364336

(CHEMBL1950088)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3ccnc(F)c3)c12 Show InChI InChI=1S/C18H14ClFN4O/c1-8-5-11(19)7-12-15(17(25)10-3-4-21-13(20)6-10)14-9(2)23-24-18(14)22-16(8)12/h3-7,17,25H,1-2H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364346

(CHEMBL1950083)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(=O)c3ccncc3)c2c1 Show InChI InChI=1S/C19H16N4O2/c1-10-8-13(25-3)9-14-16(18(24)12-4-6-20-7-5-12)15-11(2)22-23-19(15)21-17(10)14/h4-9H,1-3H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364342

(CHEMBL1950081)Show SMILES COc1ccc(cc1)C(C#N)c1c2c(C)n[nH]c2nc2c(C)cc(OC)cc12 Show InChI InChI=1S/C22H20N4O2/c1-12-9-16(28-4)10-17-20(19-13(2)25-26-22(19)24-21(12)17)18(11-23)14-5-7-15(27-3)8-6-14/h5-10,18H,1-4H3,(H,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364345
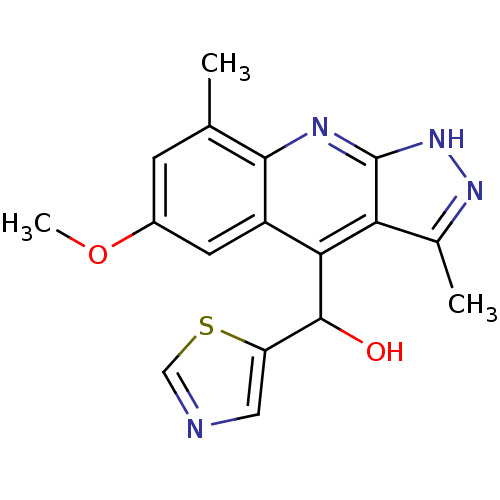
(CHEMBL1949942)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3cncs3)c2c1 Show InChI InChI=1S/C17H16N4O2S/c1-8-4-10(23-3)5-11-14(16(22)12-6-18-7-24-12)13-9(2)20-21-17(13)19-15(8)11/h4-7,16,22H,1-3H3,(H,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364344
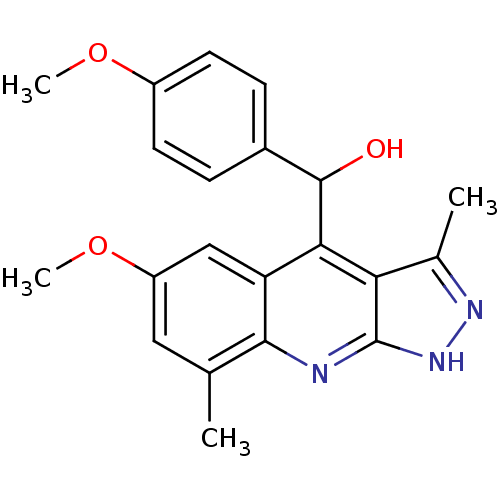
(CHEMBL1950078)Show SMILES COc1ccc(cc1)C(O)c1c2c(C)n[nH]c2nc2c(C)cc(OC)cc12 Show InChI InChI=1S/C21H21N3O3/c1-11-9-15(27-4)10-16-18(17-12(2)23-24-21(17)22-19(11)16)20(25)13-5-7-14(26-3)8-6-13/h5-10,20,25H,1-4H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364333
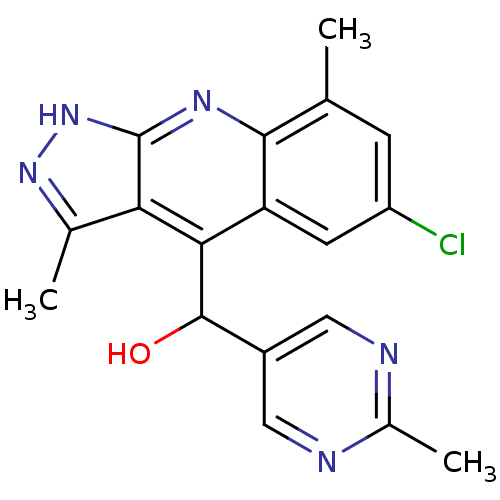
(CHEMBL1950091)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3cnc(C)nc3)c12 Show InChI InChI=1S/C18H16ClN5O/c1-8-4-12(19)5-13-15(17(25)11-6-20-10(3)21-7-11)14-9(2)23-24-18(14)22-16(8)13/h4-7,17,25H,1-3H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364341

(CHEMBL1950082)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(c2c1)C(C)(O)c1ccncc1 Show InChI InChI=1S/C20H20N4O2/c1-11-9-14(26-4)10-15-17(20(3,25)13-5-7-21-8-6-13)16-12(2)23-24-19(16)22-18(11)15/h5-10,25H,1-4H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50362043

(CHEMBL1940053)Show InChI InChI=1S/C19H24N4O2/c1-12-9-14(24-3)10-15-16(11-23-5-4-7-25-8-6-23)17-13(2)21-22-19(17)20-18(12)15/h9-10H,4-8,11H2,1-3H3,(H,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364329

(CHEMBL1949937)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3cccnc3)c2c1 Show InChI InChI=1S/C19H18N4O2/c1-10-7-13(25-3)8-14-16(18(24)12-5-4-6-20-9-12)15-11(2)22-23-19(15)21-17(10)14/h4-9,18,24H,1-3H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364338
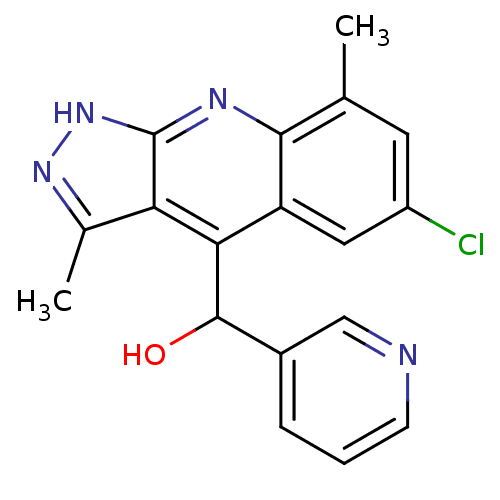
(CHEMBL1950086)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3cccnc3)c12 Show InChI InChI=1S/C18H15ClN4O/c1-9-6-12(19)7-13-15(17(24)11-4-3-5-20-8-11)14-10(2)22-23-18(14)21-16(9)13/h3-8,17,24H,1-2H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364343
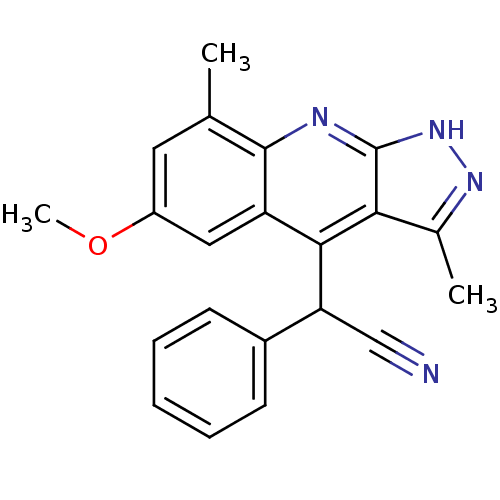
(CHEMBL1950080)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(C#N)c3ccccc3)c2c1 Show InChI InChI=1S/C21H18N4O/c1-12-9-15(26-3)10-16-19(17(11-22)14-7-5-4-6-8-14)18-13(2)24-25-21(18)23-20(12)16/h4-10,17H,1-3H3,(H,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364331

(CHEMBL1949941)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3cncn3C)c2c1 Show InChI InChI=1S/C18H19N5O2/c1-9-5-11(25-4)6-12-15(17(24)13-7-19-8-23(13)3)14-10(2)21-22-18(14)20-16(9)12/h5-8,17,24H,1-4H3,(H,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364334
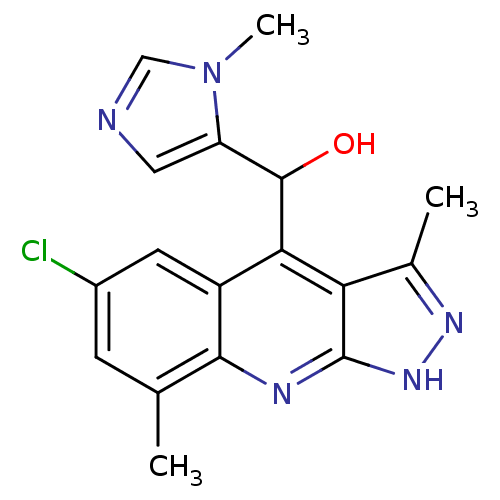
(CHEMBL1950090)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3cncn3C)c12 Show InChI InChI=1S/C17H16ClN5O/c1-8-4-10(18)5-11-14(16(24)12-6-19-7-23(12)3)13-9(2)21-22-17(13)20-15(8)11/h4-7,16,24H,1-3H3,(H,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364337

(CHEMBL1950087)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3ccccn3)c12 Show InChI InChI=1S/C18H15ClN4O/c1-9-7-11(19)8-12-15(17(24)13-5-3-4-6-20-13)14-10(2)22-23-18(14)21-16(9)12/h3-8,17,24H,1-2H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 39 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364348

(CHEMBL1950077)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ccccc3)c2c1 Show InChI InChI=1S/C20H19N3O2/c1-11-9-14(25-3)10-15-17(19(24)13-7-5-4-6-8-13)16-12(2)22-23-20(16)21-18(11)15/h4-10,19,24H,1-3H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364328
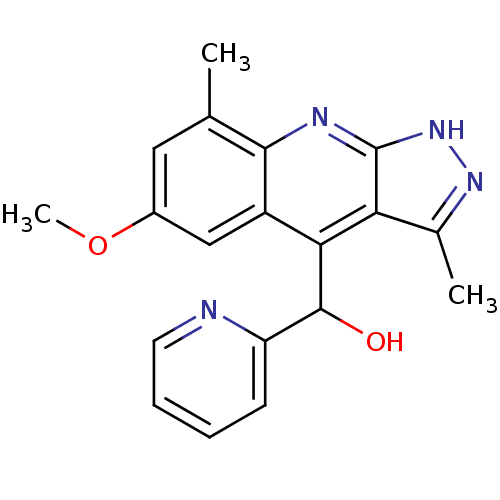
(CHEMBL1949938)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ccccn3)c2c1 Show InChI InChI=1S/C19H18N4O2/c1-10-8-12(25-3)9-13-16(18(24)14-6-4-5-7-20-14)15-11(2)22-23-19(15)21-17(10)13/h4-9,18,24H,1-3H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 54 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
cAMP and cAMP-inhibited cGMP 3',5'-cyclic phosphodiesterase 10A
(Homo sapiens (Human)) | BDBM50364347

(CHEMBL1950079)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ccc(F)cc3)c2c1 Show InChI InChI=1S/C20H18FN3O2/c1-10-8-14(26-3)9-15-17(19(25)12-4-6-13(21)7-5-12)16-11(2)23-24-20(16)22-18(10)15/h4-9,19,25H,1-3H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 83 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant PDE10A assessed as hydrolysis of cAMP to AMP using [3H]cAMP substrate by scintillation proximity assay |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364342

(CHEMBL1950081)Show SMILES COc1ccc(cc1)C(C#N)c1c2c(C)n[nH]c2nc2c(C)cc(OC)cc12 Show InChI InChI=1S/C22H20N4O2/c1-12-9-16(28-4)10-17-20(19-13(2)25-26-22(19)24-21(12)17)18(11-23)14-5-7-15(27-3)8-6-14/h5-10,18H,1-4H3,(H,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364340

(CHEMBL1950084)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(F)c3ccncc3)c2c1 Show InChI InChI=1S/C19H17FN4O/c1-10-8-13(25-3)9-14-16(17(20)12-4-6-21-7-5-12)15-11(2)23-24-19(15)22-18(10)14/h4-9,17H,1-3H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364339

(CHEMBL1950085)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3ccncc3)c12 Show InChI InChI=1S/C18H15ClN4O/c1-9-7-12(19)8-13-15(17(24)11-3-5-20-6-4-11)14-10(2)22-23-18(14)21-16(9)13/h3-8,17,24H,1-2H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364334

(CHEMBL1950090)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3cncn3C)c12 Show InChI InChI=1S/C17H16ClN5O/c1-8-4-10(18)5-11-14(16(24)12-6-19-7-23(12)3)13-9(2)21-22-17(13)20-15(8)11/h4-7,16,24H,1-3H3,(H,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364339

(CHEMBL1950085)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3ccncc3)c12 Show InChI InChI=1S/C18H15ClN4O/c1-9-7-12(19)8-13-15(17(24)11-3-5-20-6-4-11)14-10(2)22-23-18(14)21-16(9)13/h3-8,17,24H,1-2H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Time dependent inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364327

(CHEMBL1949939)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ccnc(F)c3)c2c1 Show InChI InChI=1S/C19H17FN4O2/c1-9-6-12(26-3)8-13-16(18(25)11-4-5-21-14(20)7-11)15-10(2)23-24-19(15)22-17(9)13/h4-8,18,25H,1-3H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364345

(CHEMBL1949942)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3cncs3)c2c1 Show InChI InChI=1S/C17H16N4O2S/c1-8-4-10(23-3)5-11-14(16(22)12-6-18-7-24-12)13-9(2)20-21-17(13)19-15(8)11/h4-7,16,22H,1-3H3,(H,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Time dependent inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364334

(CHEMBL1950090)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3cncn3C)c12 Show InChI InChI=1S/C17H16ClN5O/c1-8-4-10(18)5-11-14(16(24)12-6-19-7-23(12)3)13-9(2)21-22-17(13)20-15(8)11/h4-7,16,24H,1-3H3,(H,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Time dependent inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364343

(CHEMBL1950080)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(C#N)c3ccccc3)c2c1 Show InChI InChI=1S/C21H18N4O/c1-12-9-15(26-3)10-16-19(17(11-22)14-7-5-4-6-8-14)18-13(2)24-25-21(18)23-20(12)16/h4-10,17H,1-3H3,(H,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364335

(CHEMBL1950089)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3ncco3)c12 Show InChI InChI=1S/C16H13ClN4O2/c1-7-5-9(17)6-10-12(14(22)16-18-3-4-23-16)11-8(2)20-21-15(11)19-13(7)10/h3-6,14,22H,1-2H3,(H,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Time dependent inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364338

(CHEMBL1950086)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3cccnc3)c12 Show InChI InChI=1S/C18H15ClN4O/c1-9-6-12(19)7-13-15(17(24)11-4-3-5-20-8-11)14-10(2)22-23-18(14)21-16(9)13/h3-8,17,24H,1-2H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364344

(CHEMBL1950078)Show SMILES COc1ccc(cc1)C(O)c1c2c(C)n[nH]c2nc2c(C)cc(OC)cc12 Show InChI InChI=1S/C21H21N3O3/c1-11-9-15(27-4)10-16-18(17-12(2)23-24-21(17)22-19(11)16)20(25)13-5-7-14(26-3)8-6-13/h5-10,20,25H,1-4H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Time dependent inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364331

(CHEMBL1949941)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3cncn3C)c2c1 Show InChI InChI=1S/C18H19N5O2/c1-9-5-11(25-4)6-12-15(17(24)13-7-19-8-23(13)3)14-10(2)21-22-18(14)20-16(9)12/h5-8,17,24H,1-4H3,(H,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364337

(CHEMBL1950087)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3ccccn3)c12 Show InChI InChI=1S/C18H15ClN4O/c1-9-7-11(19)8-12-15(17(24)13-5-3-4-6-20-13)14-10(2)22-23-18(14)21-16(9)12/h3-8,17,24H,1-2H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364338

(CHEMBL1950086)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3cccnc3)c12 Show InChI InChI=1S/C18H15ClN4O/c1-9-6-12(19)7-13-15(17(24)11-4-3-5-20-8-11)14-10(2)22-23-18(14)21-16(9)13/h3-8,17,24H,1-2H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Time dependent inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364329

(CHEMBL1949937)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3cccnc3)c2c1 Show InChI InChI=1S/C19H18N4O2/c1-10-7-13(25-3)8-14-16(18(24)12-5-4-6-20-9-12)15-11(2)22-23-19(15)21-17(10)14/h4-9,18,24H,1-3H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Time dependent inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364330

(CHEMBL1949936)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ccncc3)c2c1 Show InChI InChI=1S/C19H18N4O2/c1-10-8-13(25-3)9-14-16(18(24)12-4-6-20-7-5-12)15-11(2)22-23-19(15)21-17(10)14/h4-9,18,24H,1-3H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364330

(CHEMBL1949936)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ccncc3)c2c1 Show InChI InChI=1S/C19H18N4O2/c1-10-8-13(25-3)9-14-16(18(24)12-4-6-20-7-5-12)15-11(2)22-23-19(15)21-17(10)14/h4-9,18,24H,1-3H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Time dependent inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364327

(CHEMBL1949939)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ccnc(F)c3)c2c1 Show InChI InChI=1S/C19H17FN4O2/c1-9-6-12(26-3)8-13-16(18(25)11-4-5-21-14(20)7-11)15-10(2)23-24-19(15)22-17(9)13/h4-8,18,25H,1-3H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Time dependent inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364332

(CHEMBL1949940)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ncco3)c2c1 Show InChI InChI=1S/C17H16N4O3/c1-8-6-10(23-3)7-11-13(15(22)17-18-4-5-24-17)12-9(2)20-21-16(12)19-14(8)11/h4-7,15,22H,1-3H3,(H,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364328

(CHEMBL1949938)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ccccn3)c2c1 Show InChI InChI=1S/C19H18N4O2/c1-10-8-12(25-3)9-13-16(18(24)14-6-4-5-7-20-14)15-11(2)22-23-19(15)21-17(10)13/h4-9,18,24H,1-3H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364331

(CHEMBL1949941)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3cncn3C)c2c1 Show InChI InChI=1S/C18H19N5O2/c1-9-5-11(25-4)6-12-15(17(24)13-7-19-8-23(13)3)14-10(2)21-22-18(14)20-16(9)12/h5-8,17,24H,1-4H3,(H,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Time dependent inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364341

(CHEMBL1950082)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(c2c1)C(C)(O)c1ccncc1 Show InChI InChI=1S/C20H20N4O2/c1-11-9-14(26-4)10-15-17(20(3,25)13-5-7-21-8-6-13)16-12(2)23-24-19(16)22-18(11)15/h5-10,25H,1-4H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364335

(CHEMBL1950089)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3ncco3)c12 Show InChI InChI=1S/C16H13ClN4O2/c1-7-5-9(17)6-10-12(14(22)16-18-3-4-23-16)11-8(2)20-21-15(11)19-13(7)10/h3-6,14,22H,1-2H3,(H,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364332

(CHEMBL1949940)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ncco3)c2c1 Show InChI InChI=1S/C17H16N4O3/c1-8-6-10(23-3)7-11-13(15(22)17-18-4-5-24-17)12-9(2)20-21-16(12)19-14(8)11/h4-7,15,22H,1-3H3,(H,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Time dependent inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364333

(CHEMBL1950091)Show SMILES Cc1n[nH]c2nc3c(C)cc(Cl)cc3c(C(O)c3cnc(C)nc3)c12 Show InChI InChI=1S/C18H16ClN5O/c1-8-4-12(19)5-13-15(17(25)11-6-20-10(3)21-7-11)14-9(2)23-24-18(14)22-16(8)13/h4-7,17,25H,1-3H3,(H,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Reversible inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50364328

(CHEMBL1949938)Show SMILES COc1cc(C)c2nc3[nH]nc(C)c3c(C(O)c3ccccn3)c2c1 Show InChI InChI=1S/C19H18N4O2/c1-10-8-12(25-3)9-13-16(18(24)14-6-4-5-7-20-14)15-11(2)22-23-19(15)21-17(10)13/h4-9,18,24H,1-3H3,(H,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.06E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Time dependent inhibition of CYP3A4 |
Bioorg Med Chem Lett 22: 1335-9 (2012)
Article DOI: 10.1016/j.bmcl.2011.12.080
BindingDB Entry DOI: 10.7270/Q2RF5VGF |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data