 Found 212 hits Enz. Inhib. hit(s) with Target = 'Complement C1r subcomponent'
Found 212 hits Enz. Inhib. hit(s) with Target = 'Complement C1r subcomponent' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063701

(2-(2-Iodo-phenylamino)-naphtho[2,3-d][1,3]oxazin-4...)Show InChI InChI=1S/C18H11IN2O2/c19-14-7-3-4-8-15(14)20-18-21-16-10-12-6-2-1-5-11(12)9-13(16)17(22)23-18/h1-10H,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063745
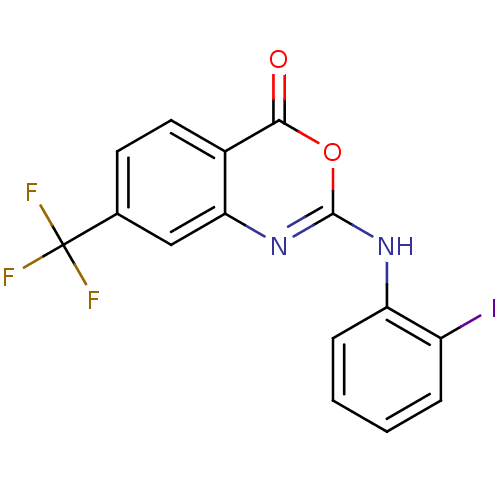
(2-(2-Iodo-phenylamino)-7-trifluoromethyl-benzo[d][...)Show InChI InChI=1S/C15H8F3IN2O2/c16-15(17,18)8-5-6-9-12(7-8)21-14(23-13(9)22)20-11-4-2-1-3-10(11)19/h1-7H,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063726

(7-Chloro-2-(2,6-dichloro-phenylamino)-benzo[d][1,3...)Show InChI InChI=1S/C14H7Cl3N2O2/c15-7-4-5-8-11(6-7)18-14(21-13(8)20)19-12-9(16)2-1-3-10(12)17/h1-6H,(H,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063745

(2-(2-Iodo-phenylamino)-7-trifluoromethyl-benzo[d][...)Show InChI InChI=1S/C15H8F3IN2O2/c16-15(17,18)8-5-6-9-12(7-8)21-14(23-13(9)22)20-11-4-2-1-3-10(11)19/h1-7H,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Compound was evaluated in vitro for inhibitory activity against purified human C1r protease protease |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063722
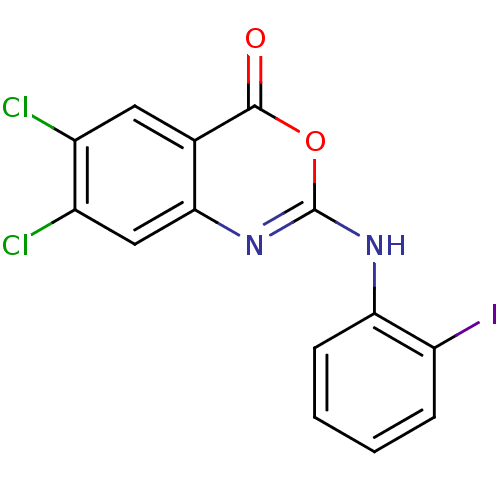
(6,7-Dichloro-2-(2-iodo-phenylamino)-benzo[d][1,3]o...)Show InChI InChI=1S/C14H7Cl2IN2O2/c15-8-5-7-12(6-9(8)16)19-14(21-13(7)20)18-11-4-2-1-3-10(11)17/h1-6H,(H,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50462100
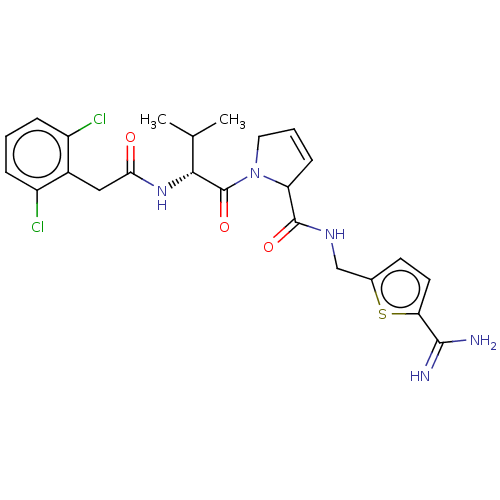
(CHEMBL4247936)Show SMILES CC(C)[C@@H](NC(=O)Cc1c(Cl)cccc1Cl)C(=O)N1CC=CC1C(=O)NCc1ccc(s1)C(N)=N |r,c:21| Show InChI InChI=1S/C24H27Cl2N5O3S/c1-13(2)21(30-20(32)11-15-16(25)5-3-6-17(15)26)24(34)31-10-4-7-18(31)23(33)29-12-14-8-9-19(35-14)22(27)28/h3-9,13,18,21H,10-12H2,1-2H3,(H3,27,28)(H,29,33)(H,30,32)/t18?,21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 600 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of human plasma C1r using Cbz-Gly-Arg-S-Bzl as substrate preincubated for 10 mins followed by substrate addition and measured after 30 min... |
J Med Chem 61: 3253-3276 (2018)
Article DOI: 10.1021/acs.jmedchem.7b00882
BindingDB Entry DOI: 10.7270/Q2MK6GJ9 |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075982

(4-Methoxy-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxazin...)Show SMILES COc1ccc(cc1)S(=O)(=O)N(C)c1nc2ccccc2c(=O)o1 Show InChI InChI=1S/C16H14N2O5S/c1-18(24(20,21)12-9-7-11(22-2)8-10-12)16-17-14-6-4-3-5-13(14)15(19)23-16/h3-10H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063711
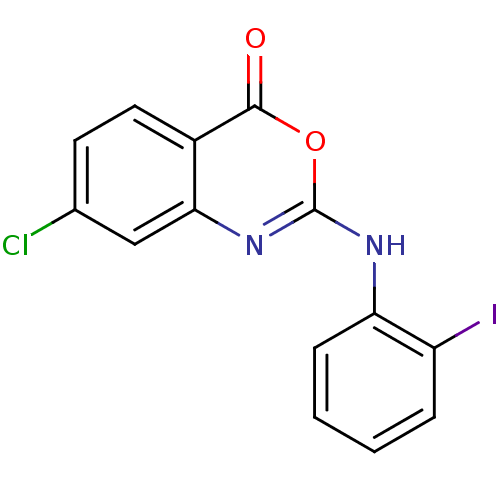
(7-Chloro-2-(2-iodo-phenylamino)-benzo[d][1,3]oxazi...)Show InChI InChI=1S/C14H8ClIN2O2/c15-8-5-6-9-12(7-8)18-14(20-13(9)19)17-11-4-2-1-3-10(11)16/h1-7H,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50289007
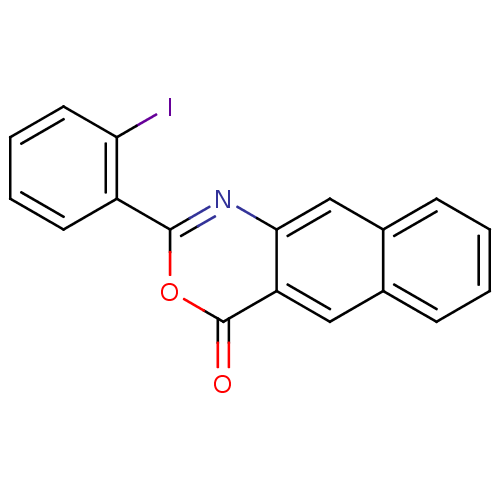
(2-(2-Iodo-phenyl)-naphtho[2,3-d][1,3]oxazin-4-one ...)Show InChI InChI=1S/C18H10INO2/c19-15-8-4-3-7-13(15)17-20-16-10-12-6-2-1-5-11(12)9-14(16)18(21)22-17/h1-10H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of C1r serine protease |
Bioorg Med Chem Lett 6: 679-682 (1996)
Article DOI: 10.1016/0960-894X(96)00094-7
BindingDB Entry DOI: 10.7270/Q2P84BVF |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063717

(7-Chloro-2-(2-chloro-phenylamino)-benzo[d][1,3]oxa...)Show InChI InChI=1S/C14H8Cl2N2O2/c15-8-5-6-9-12(7-8)18-14(20-13(9)19)17-11-4-2-1-3-10(11)16/h1-7H,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
50% inhibition of human C1r serine protease after 60 mins using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063717

(7-Chloro-2-(2-chloro-phenylamino)-benzo[d][1,3]oxa...)Show InChI InChI=1S/C14H8Cl2N2O2/c15-8-5-6-9-12(7-8)18-14(20-13(9)19)17-11-4-2-1-3-10(11)16/h1-7H,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063723
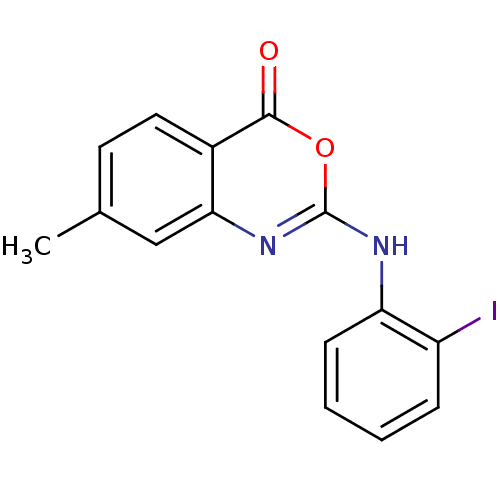
(2-(2-Iodo-phenylamino)-7-methyl-benzo[d][1,3]oxazi...)Show InChI InChI=1S/C15H11IN2O2/c1-9-6-7-10-13(8-9)18-15(20-14(10)19)17-12-5-3-2-4-11(12)16/h2-8H,1H3,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063721
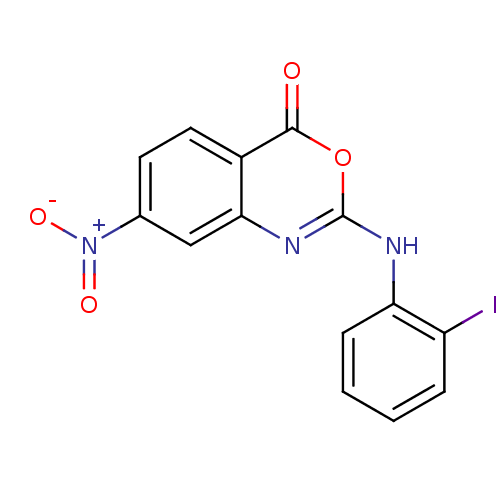
(2-(2-Iodo-phenylamino)-7-nitro-benzo[d][1,3]oxazin...)Show InChI InChI=1S/C14H8IN3O4/c15-10-3-1-2-4-11(10)16-14-17-12-7-8(18(20)21)5-6-9(12)13(19)22-14/h1-7H,(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063698

(4-Guanidino-benzoic acid 6-carbamimidoyl-naphthale...)Show SMILES [#7]\[#6](-[#7])=[#7]/c1ccc(cc1)-[#6](=O)-[#8]-c1ccc2cc(ccc2c1)-[#6](-[#7])=[#7] Show InChI InChI=1S/C19H17N5O2/c20-17(21)14-2-1-13-10-16(8-5-12(13)9-14)26-18(25)11-3-6-15(7-4-11)24-19(22)23/h1-10H,(H3,20,21)(H4,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Queensland
Curated by ChEMBL
| Assay Description
Inhibition of human serum C1r using AAME as substrate after 30 mins |
J Med Chem 61: 3253-3276 (2018)
Article DOI: 10.1021/acs.jmedchem.7b00882
BindingDB Entry DOI: 10.7270/Q2MK6GJ9 |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063724

(2-(2,6-Dichloro-phenylamino)-benzo[d][1,3]oxazin-4...)Show InChI InChI=1S/C14H8Cl2N2O2/c15-9-5-3-6-10(16)12(9)18-14-17-11-7-2-1-4-8(11)13(19)20-14/h1-7H,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
50% inhibition of human C1r serine protease after 60 mins using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063711

(7-Chloro-2-(2-iodo-phenylamino)-benzo[d][1,3]oxazi...)Show InChI InChI=1S/C14H8ClIN2O2/c15-8-5-6-9-12(7-8)18-14(20-13(9)19)17-11-4-2-1-3-10(11)16/h1-7H,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
50% inhibition of human C1r serine protease after 60 mins using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063726

(7-Chloro-2-(2,6-dichloro-phenylamino)-benzo[d][1,3...)Show InChI InChI=1S/C14H7Cl3N2O2/c15-7-4-5-8-11(6-7)18-14(21-13(8)20)19-12-9(16)2-1-3-10(12)17/h1-6H,(H,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
50% inhibition of human C1r serine protease after 60 mins using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063724

(2-(2,6-Dichloro-phenylamino)-benzo[d][1,3]oxazin-4...)Show InChI InChI=1S/C14H8Cl2N2O2/c15-9-5-3-6-10(16)12(9)18-14-17-11-7-2-1-4-8(11)13(19)20-14/h1-7H,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063732

(2-(2-Chloro-phenylamino)-7-methyl-benzo[d][1,3]oxa...)Show InChI InChI=1S/C15H11ClN2O2/c1-9-6-7-10-13(8-9)18-15(20-14(10)19)17-12-5-3-2-4-11(12)16/h2-8H,1H3,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
50% inhibition of human C1r serine protease after 60 mins using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063698

(4-Guanidino-benzoic acid 6-carbamimidoyl-naphthale...)Show SMILES [#7]\[#6](-[#7])=[#7]/c1ccc(cc1)-[#6](=O)-[#8]-c1ccc2cc(ccc2c1)-[#6](-[#7])=[#7] Show InChI InChI=1S/C19H17N5O2/c20-17(21)14-2-1-13-10-16(8-5-12(13)9-14)26-18(25)11-3-6-15(7-4-11)24-19(22)23/h1-10H,(H3,20,21)(H4,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against C1r serine protease |
Bioorg Med Chem Lett 6: 679-682 (1996)
Article DOI: 10.1016/0960-894X(96)00094-7
BindingDB Entry DOI: 10.7270/Q2P84BVF |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063698

(4-Guanidino-benzoic acid 6-carbamimidoyl-naphthale...)Show SMILES [#7]\[#6](-[#7])=[#7]/c1ccc(cc1)-[#6](=O)-[#8]-c1ccc2cc(ccc2c1)-[#6](-[#7])=[#7] Show InChI InChI=1S/C19H17N5O2/c20-17(21)14-2-1-13-10-16(8-5-12(13)9-14)26-18(25)11-3-6-15(7-4-11)24-19(22)23/h1-10H,(H3,20,21)(H4,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
| n/a | n/a | 1.04E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against C1r serine protease |
Bioorg Med Chem Lett 6: 679-682 (1996)
Article DOI: 10.1016/0960-894X(96)00094-7
BindingDB Entry DOI: 10.7270/Q2P84BVF |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063703
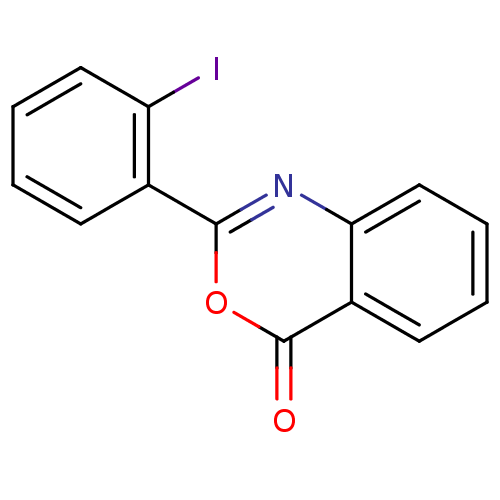
(2-(2-Iodo-phenyl)-benzo[d][1,3]oxazin-4-one | CHEM...)Show InChI InChI=1S/C14H8INO2/c15-11-7-3-1-5-9(11)13-16-12-8-4-2-6-10(12)14(17)18-13/h1-8H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 1.37E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against C1r serine protease |
Bioorg Med Chem Lett 6: 679-682 (1996)
Article DOI: 10.1016/0960-894X(96)00094-7
BindingDB Entry DOI: 10.7270/Q2P84BVF |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063704
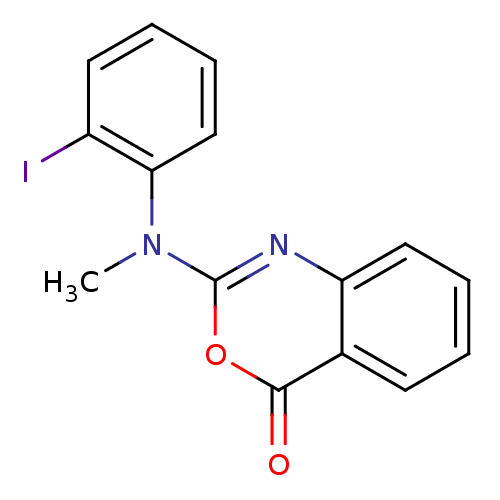
(2-[(2-Iodo-phenyl)-methyl-amino]-benzo[d][1,3]oxaz...)Show InChI InChI=1S/C15H11IN2O2/c1-18(13-9-5-3-7-11(13)16)15-17-12-8-4-2-6-10(12)14(19)20-15/h2-9H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063732

(2-(2-Chloro-phenylamino)-7-methyl-benzo[d][1,3]oxa...)Show InChI InChI=1S/C15H11ClN2O2/c1-9-6-7-10-13(8-9)18-15(20-14(10)19)17-12-5-3-2-4-11(12)16/h2-8H,1H3,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063703

(2-(2-Iodo-phenyl)-benzo[d][1,3]oxazin-4-one | CHEM...)Show InChI InChI=1S/C14H8INO2/c15-11-7-3-1-5-9(11)13-16-12-8-4-2-6-10(12)14(17)18-13/h1-8H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against C1r serine protease |
Bioorg Med Chem Lett 6: 679-682 (1996)
Article DOI: 10.1016/0960-894X(96)00094-7
BindingDB Entry DOI: 10.7270/Q2P84BVF |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063720

(2-(2-Chloro-phenylamino)-7-nitro-benzo[d][1,3]oxaz...)Show SMILES [O-][N+](=O)c1ccc2c(c1)nc(Nc1ccccc1Cl)oc2=O Show InChI InChI=1S/C14H8ClN3O4/c15-10-3-1-2-4-11(10)16-14-17-12-7-8(18(20)21)5-6-9(12)13(19)22-14/h1-7H,(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075992

(4-Guanidino-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxaz...)Show SMILES [#6]-[#7](-c1nc2ccccc2c(=O)o1)S(=O)(=O)c1ccc(cc1)\[#7]=[#6](/[#7])-[#7] Show InChI InChI=1S/C16H15N5O4S/c1-21(16-20-13-5-3-2-4-12(13)14(22)25-16)26(23,24)11-8-6-10(7-9-11)19-15(17)18/h2-9H,1H3,(H4,17,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063723

(2-(2-Iodo-phenylamino)-7-methyl-benzo[d][1,3]oxazi...)Show InChI InChI=1S/C15H11IN2O2/c1-9-6-7-10-13(8-9)18-15(20-14(10)19)17-12-5-3-2-4-11(12)16/h2-8H,1H3,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
50% inhibition of human C1r serine protease after 60 mins using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063740

(2-(2,6-Dichloro-phenylamino)-7-trifluoromethyl-ben...)Show SMILES FC(F)(F)c1ccc2c(c1)nc(Nc1c(Cl)cccc1Cl)oc2=O Show InChI InChI=1S/C15H7Cl2F3N2O2/c16-9-2-1-3-10(17)12(9)22-14-21-11-6-7(15(18,19)20)4-5-8(11)13(23)24-14/h1-6H,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50288998
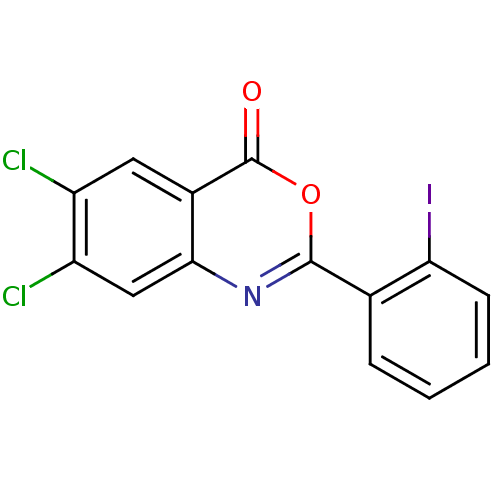
(6,7-Dichloro-2-(2-iodo-phenyl)-benzo[d][1,3]oxazin...)Show InChI InChI=1S/C14H6Cl2INO2/c15-9-5-8-12(6-10(9)16)18-13(20-14(8)19)7-3-1-2-4-11(7)17/h1-6H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against C1r serine protease |
Bioorg Med Chem Lett 6: 679-682 (1996)
Article DOI: 10.1016/0960-894X(96)00094-7
BindingDB Entry DOI: 10.7270/Q2P84BVF |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063708
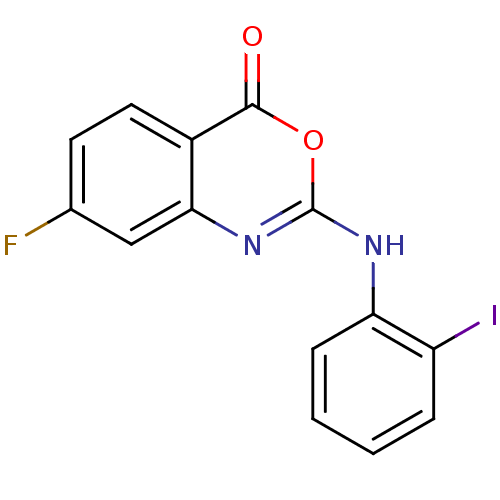
(7-Fluoro-2-(2-iodo-phenylamino)-benzo[d][1,3]oxazi...)Show InChI InChI=1S/C14H8FIN2O2/c15-8-5-6-9-12(7-8)18-14(20-13(9)19)17-11-4-2-1-3-10(11)16/h1-7H,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063697
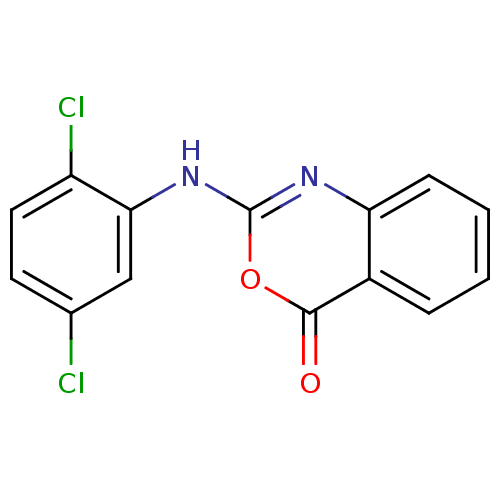
(2-(2,5-Dichloro-phenylamino)-benzo[d][1,3]oxazin-4...)Show InChI InChI=1S/C14H8Cl2N2O2/c15-8-5-6-10(16)12(7-8)18-14-17-11-4-2-1-3-9(11)13(19)20-14/h1-7H,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
50% inhibition of human C1r serine protease after 60 mins using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063697

(2-(2,5-Dichloro-phenylamino)-benzo[d][1,3]oxazin-4...)Show InChI InChI=1S/C14H8Cl2N2O2/c15-8-5-6-10(16)12(7-8)18-14-17-11-4-2-1-3-9(11)13(19)20-14/h1-7H,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063730
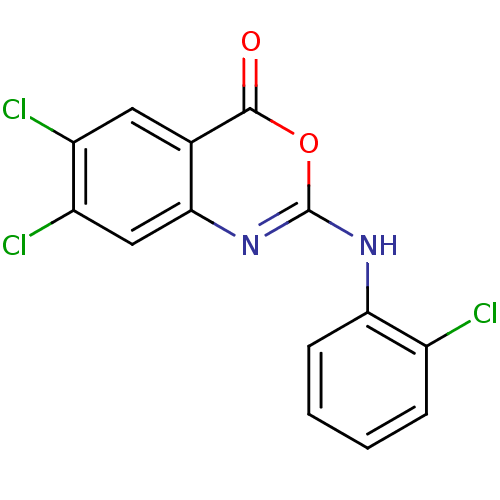
(6,7-Dichloro-2-(2-chloro-phenylamino)-benzo[d][1,3...)Show InChI InChI=1S/C14H7Cl3N2O2/c15-8-3-1-2-4-11(8)18-14-19-12-6-10(17)9(16)5-7(12)13(20)21-14/h1-6H,(H,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063708

(7-Fluoro-2-(2-iodo-phenylamino)-benzo[d][1,3]oxazi...)Show InChI InChI=1S/C14H8FIN2O2/c15-8-5-6-9-12(7-8)18-14(20-13(9)19)17-11-4-2-1-3-10(11)16/h1-7H,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
50% inhibition of human C1r serine protease after 60 mins using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075978
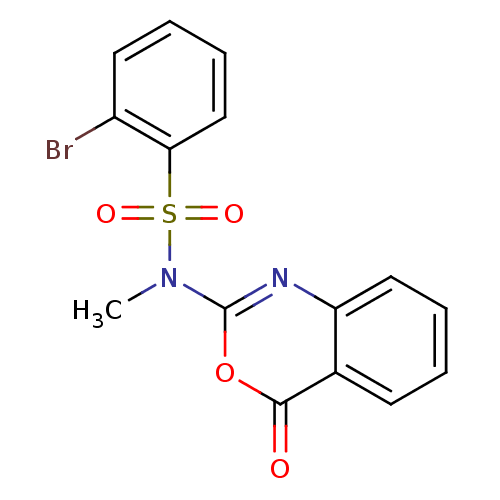
(2-Bromo-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxazin-2...)Show InChI InChI=1S/C15H11BrN2O4S/c1-18(23(20,21)13-9-5-3-7-11(13)16)15-17-12-8-4-2-6-10(12)14(19)22-15/h2-9H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50289016
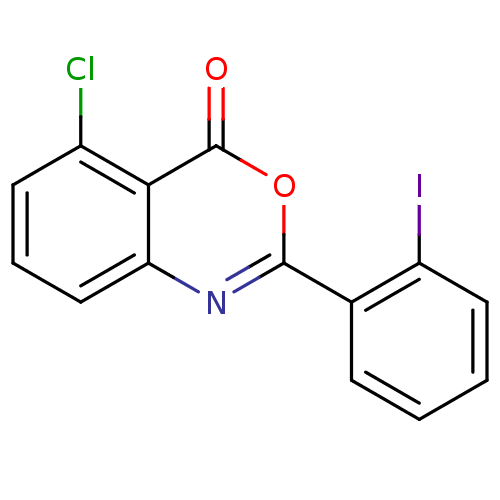
(5-Chloro-2-(2-iodo-phenyl)-benzo[d][1,3]oxazin-4-o...)Show InChI InChI=1S/C14H7ClINO2/c15-9-5-3-7-11-12(9)14(18)19-13(17-11)8-4-1-2-6-10(8)16/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against C1r serine protease |
Bioorg Med Chem Lett 6: 679-682 (1996)
Article DOI: 10.1016/0960-894X(96)00094-7
BindingDB Entry DOI: 10.7270/Q2P84BVF |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075992

(4-Guanidino-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxaz...)Show SMILES [#6]-[#7](-c1nc2ccccc2c(=O)o1)S(=O)(=O)c1ccc(cc1)\[#7]=[#6](/[#7])-[#7] Show InChI InChI=1S/C16H15N5O4S/c1-21(16-20-13-5-3-2-4-12(13)14(22)25-16)26(23,24)11-8-6-10(7-9-11)19-15(17)18/h2-9H,1H3,(H4,17,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50289004
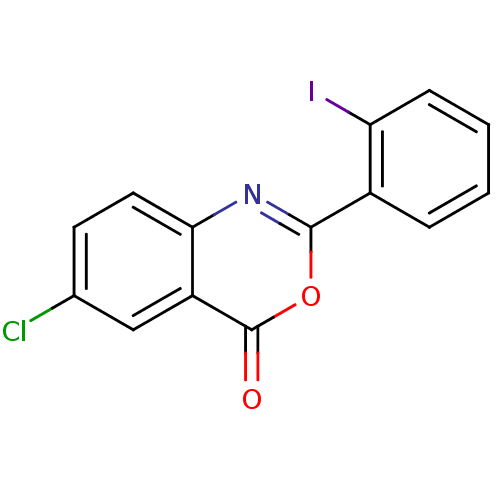
(6-Chloro-2-(2-iodo-phenyl)-benzo[d][1,3]oxazin-4-o...)Show InChI InChI=1S/C14H7ClINO2/c15-8-5-6-12-10(7-8)14(18)19-13(17-12)9-3-1-2-4-11(9)16/h1-7H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of C1r serine protease |
Bioorg Med Chem Lett 6: 679-682 (1996)
Article DOI: 10.1016/0960-894X(96)00094-7
BindingDB Entry DOI: 10.7270/Q2P84BVF |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075986
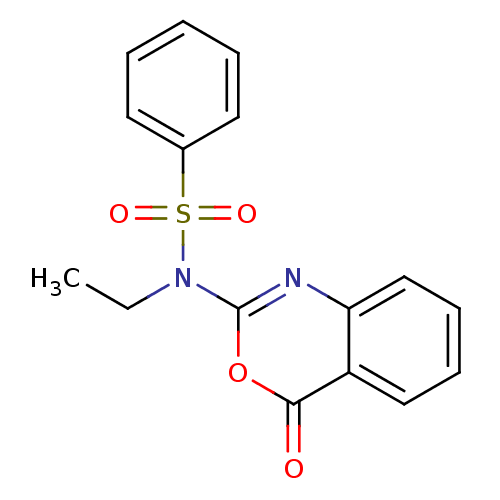
(CHEMBL168038 | N-Ethyl-N-(4-oxo-4H-benzo[d][1,3]ox...)Show InChI InChI=1S/C16H14N2O4S/c1-2-18(23(20,21)12-8-4-3-5-9-12)16-17-14-11-7-6-10-13(14)15(19)22-16/h3-11H,2H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063715

(2-(2-Iodo-phenylamino)-benzo[d][1,3]oxazin-4-one |...)Show InChI InChI=1S/C14H9IN2O2/c15-10-6-2-4-8-12(10)17-14-16-11-7-3-1-5-9(11)13(18)19-14/h1-8H,(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063745

(2-(2-Iodo-phenylamino)-7-trifluoromethyl-benzo[d][...)Show InChI InChI=1S/C15H8F3IN2O2/c16-15(17,18)8-5-6-9-12(7-8)21-14(23-13(9)22)20-11-4-2-1-3-10(11)19/h1-7H,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Evaluated in vitro for inhibitory activity against purified human C1r protease incubated in buffer for 60 minutes |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063745

(2-(2-Iodo-phenylamino)-7-trifluoromethyl-benzo[d][...)Show InChI InChI=1S/C15H8F3IN2O2/c16-15(17,18)8-5-6-9-12(7-8)21-14(23-13(9)22)20-11-4-2-1-3-10(11)19/h1-7H,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
50% inhibition of human C1r serine protease after 60 mins using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075993
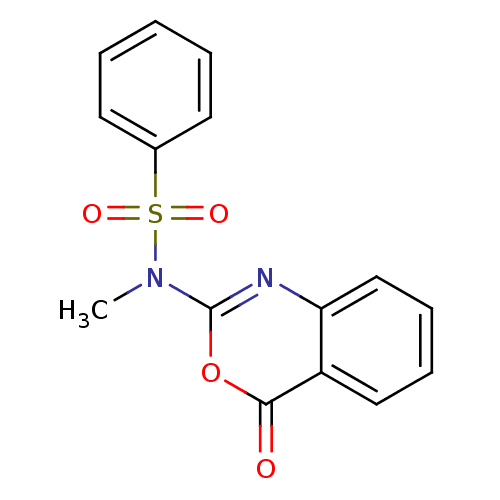
(CHEMBL166632 | N-Methyl-N-(4-oxo-4H-benzo[d][1,3]o...)Show InChI InChI=1S/C15H12N2O4S/c1-17(22(19,20)11-7-3-2-4-8-11)15-16-13-10-6-5-9-12(13)14(18)21-15/h2-10H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063714

(6-Chloro-2-(2-iodo-phenylamino)-benzo[d][1,3]oxazi...)Show InChI InChI=1S/C14H8ClIN2O2/c15-8-5-6-11-9(7-8)13(19)20-14(17-11)18-12-4-2-1-3-10(12)16/h1-7H,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063738
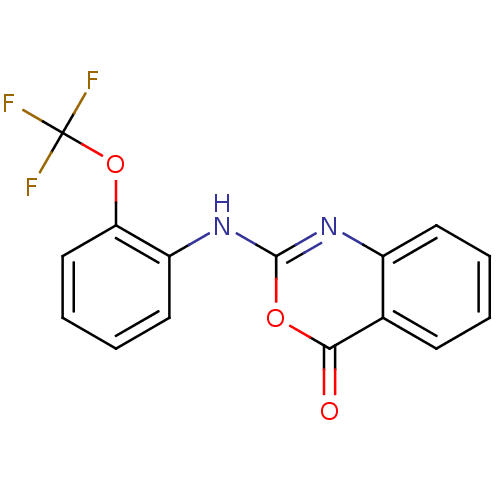
(2-(2-Trifluoromethoxy-phenylamino)-benzo[d][1,3]ox...)Show InChI InChI=1S/C15H9F3N2O3/c16-15(17,18)23-12-8-4-3-7-11(12)20-14-19-10-6-2-1-5-9(10)13(21)22-14/h1-8H,(H,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50288996
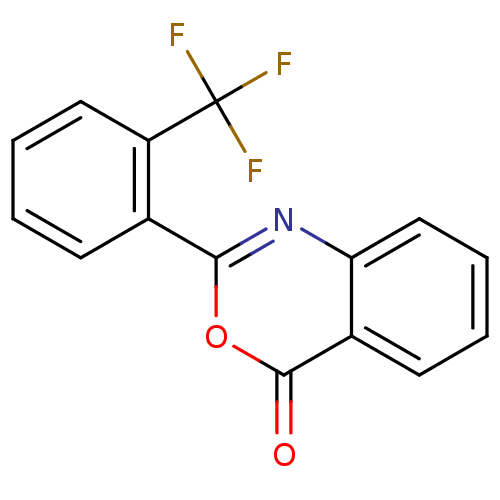
(2-(2-Trifluoromethyl-phenyl)-benzo[d][1,3]oxazin-4...)Show InChI InChI=1S/C15H8F3NO2/c16-15(17,18)11-7-3-1-5-9(11)13-19-12-8-4-2-6-10(12)14(20)21-13/h1-8H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
| n/a | n/a | 4.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of C1r serine protease |
Bioorg Med Chem Lett 6: 679-682 (1996)
Article DOI: 10.1016/0960-894X(96)00094-7
BindingDB Entry DOI: 10.7270/Q2P84BVF |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063740

(2-(2,6-Dichloro-phenylamino)-7-trifluoromethyl-ben...)Show SMILES FC(F)(F)c1ccc2c(c1)nc(Nc1c(Cl)cccc1Cl)oc2=O Show InChI InChI=1S/C15H7Cl2F3N2O2/c16-9-2-1-3-10(17)12(9)22-14-21-11-6-7(15(18,19)20)4-5-8(11)13(23)24-14/h1-6H,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
50% inhibition of human C1r serine protease after 60 mins using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50289011
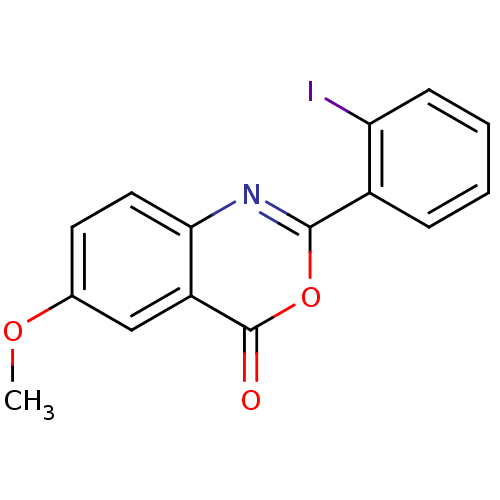
(2-(2-Iodo-phenyl)-6-methoxy-benzo[d][1,3]oxazin-4-...)Show InChI InChI=1S/C15H10INO3/c1-19-9-6-7-13-11(8-9)15(18)20-14(17-13)10-4-2-3-5-12(10)16/h2-8H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against C1r serine protease |
Bioorg Med Chem Lett 6: 679-682 (1996)
Article DOI: 10.1016/0960-894X(96)00094-7
BindingDB Entry DOI: 10.7270/Q2P84BVF |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063718
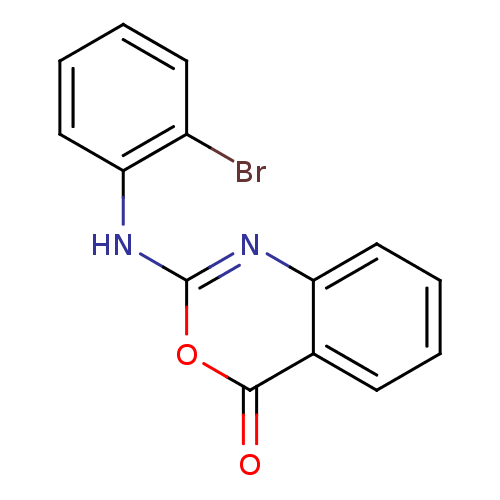
(2-(2-Bromo-phenylamino)-benzo[d][1,3]oxazin-4-one ...)Show InChI InChI=1S/C14H9BrN2O2/c15-10-6-2-4-8-12(10)17-14-16-11-7-3-1-5-9(11)13(18)19-14/h1-8H,(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibition of 50% of human C1r Serine Protease by initially using CbzGly-Arg-S-Bzl as substrate |
J Med Chem 41: 1060-7 (1998)
Article DOI: 10.1021/jm970394d
BindingDB Entry DOI: 10.7270/Q2PZ57ZT |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data