

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM22566 (5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 977 | -34.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Abbott Laboratories | Assay Description Competition radioligand binding assays were performed with increasing concentrations of test compound in the presence of [3H]ligand. All binding reac... | J Med Chem 51: 7094-8 (2008) Article DOI: 10.1021/jm8007618 BindingDB Entry DOI: 10.7270/Q2862DS6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM26226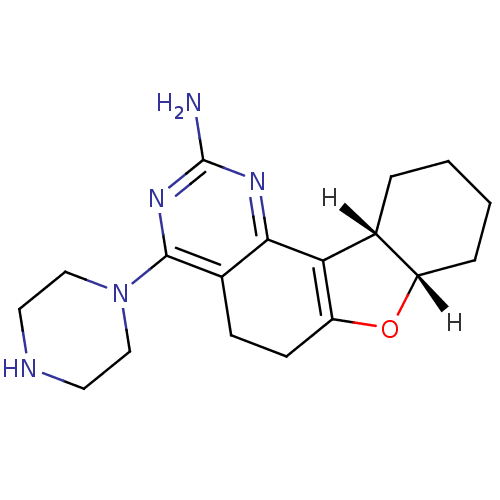 ((12S,17S)-6-(piperazin-1-yl)-11-oxa-3,5-diazatetra...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem Similars | Article PubMed | 3.55E+3 | -31.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Abbott Laboratories | Assay Description Competition radioligand binding assays were performed with increasing concentrations of test compound in the presence of [3H]ligand. All binding reac... | J Med Chem 51: 7094-8 (2008) Article DOI: 10.1021/jm8007618 BindingDB Entry DOI: 10.7270/Q2862DS6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM22914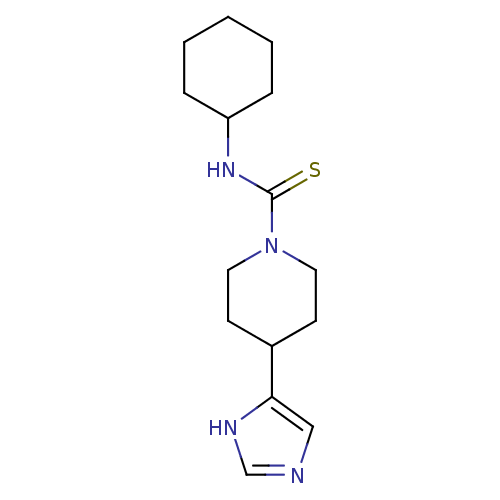 (CHEMBL260374 | N-cyclohexyl-4-(1H-imidazol-5-yl)pi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | >1.00E+4 | >-28.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Abbott Laboratories | Assay Description Competition radioligand binding assays were performed with increasing concentrations of test compound in the presence of [3H]ligand. All binding reac... | J Med Chem 51: 5423-30 (2008) Article DOI: 10.1021/jm8003625 BindingDB Entry DOI: 10.7270/Q21G0JK0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM27213 (4-[3-(4-cyclopropanecarbonylphenoxy)propyl]-1H-imi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+4 | >-28.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Abbott Laboratories | Assay Description Competition radioligand binding assays were performed with increasing concentrations of test compound in the presence of [3H]ligand. All binding reac... | J Med Chem 51: 5423-30 (2008) Article DOI: 10.1021/jm8003625 BindingDB Entry DOI: 10.7270/Q21G0JK0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM27193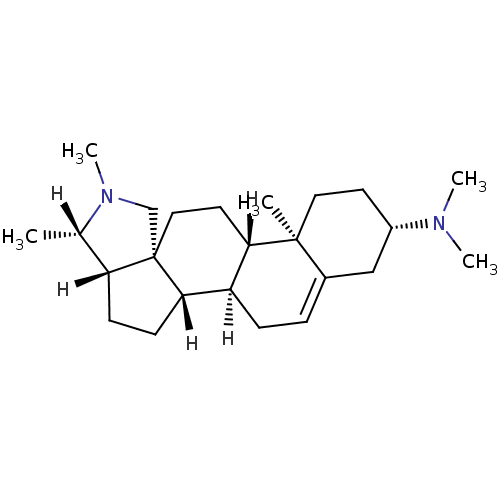 ((1R,2S,5S,6S,9R,12S,13R,16S)-N,N,6,7,13-pentamethy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+4 | >-28.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Abbott Laboratories | Assay Description Competition radioligand binding assays were performed with increasing concentrations of test compound in the presence of [3H]ligand. All binding reac... | J Med Chem 51: 5423-30 (2008) Article DOI: 10.1021/jm8003625 BindingDB Entry DOI: 10.7270/Q21G0JK0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM293397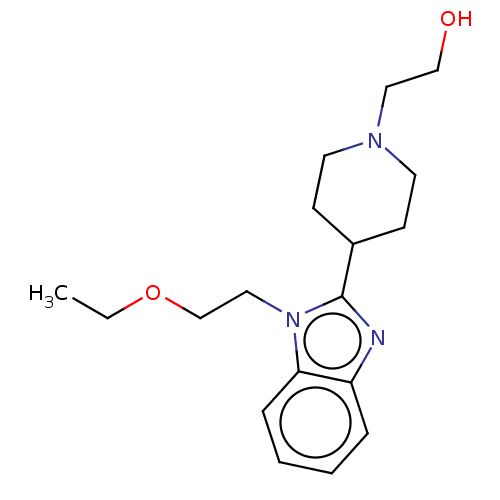 (US10106522, Example 1) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 25 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Faes Farma, S.A. US Patent | Assay Description Affinity for histamine H1-receptor was determined by binding studies to H1 receptors. The sample used is a suspension of membranes of CHO cells trans... | US Patent US10106522 (2018) BindingDB Entry DOI: 10.7270/Q2TF00DX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM293413 (US10106522, Compound I-3 in CN 103896915) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | US Patent | n/a | n/a | 271 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Faes Farma, S.A. US Patent | Assay Description Affinity for histamine H1-receptor was determined by binding studies to H1 receptors. The sample used is a suspension of membranes of CHO cells trans... | US Patent US10106522 (2018) BindingDB Entry DOI: 10.7270/Q2TF00DX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM293412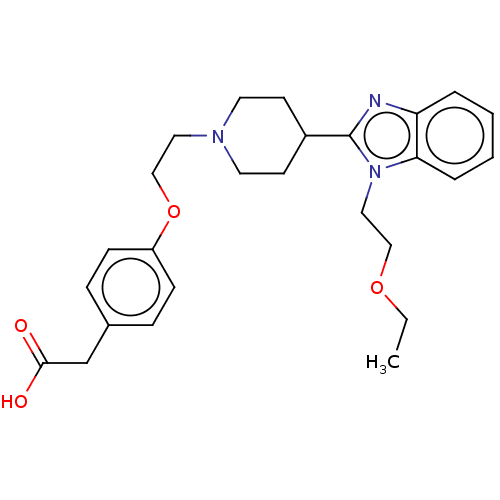 (US10106522, Compound I-8 in CN 103896915) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem | US Patent | n/a | n/a | 1.99E+3 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Faes Farma, S.A. US Patent | Assay Description Affinity for histamine H1-receptor was determined by binding studies to H1 receptors. The sample used is a suspension of membranes of CHO cells trans... | US Patent US10106522 (2018) BindingDB Entry DOI: 10.7270/Q2TF00DX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM293411 (US10106522, Example 5 in EP 0580541) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 424 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Faes Farma, S.A. US Patent | Assay Description Affinity for histamine H1-receptor was determined by binding studies to H1 receptors. The sample used is a suspension of membranes of CHO cells trans... | US Patent US10106522 (2018) BindingDB Entry DOI: 10.7270/Q2TF00DX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM293410 (US10106522, Example 9) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 195 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Faes Farma, S.A. US Patent | Assay Description Affinity for histamine H1-receptor was determined by binding studies to H1 receptors. The sample used is a suspension of membranes of CHO cells trans... | US Patent US10106522 (2018) BindingDB Entry DOI: 10.7270/Q2TF00DX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM293414 (US10106522, Compound I-11 in CN 103896915) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 260 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Faes Farma, S.A. US Patent | Assay Description Affinity for histamine H1-receptor was determined by binding studies to H1 receptors. The sample used is a suspension of membranes of CHO cells trans... | US Patent US10106522 (2018) BindingDB Entry DOI: 10.7270/Q2TF00DX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM293400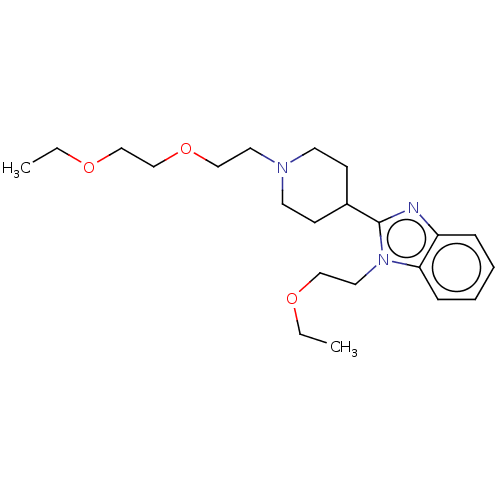 (US10106522, Example 2) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 115 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Faes Farma, S.A. US Patent | Assay Description Affinity for histamine H1-receptor was determined by binding studies to H1 receptors. The sample used is a suspension of membranes of CHO cells trans... | US Patent US10106522 (2018) BindingDB Entry DOI: 10.7270/Q2TF00DX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM293403 (US10106522, Example 3) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 28 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Faes Farma, S.A. US Patent | Assay Description Affinity for histamine H1-receptor was determined by binding studies to H1 receptors. The sample used is a suspension of membranes of CHO cells trans... | US Patent US10106522 (2018) BindingDB Entry DOI: 10.7270/Q2TF00DX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM293406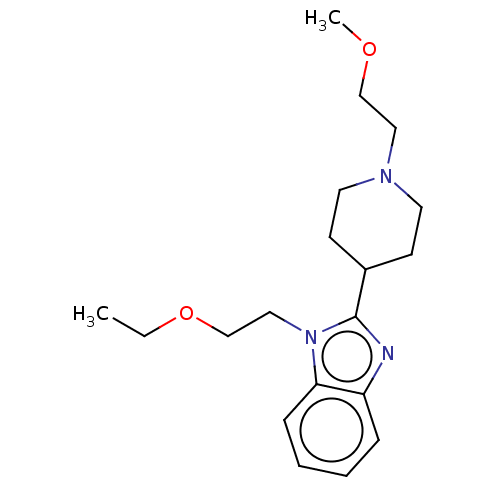 (US10106522, Example 4) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 93 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Faes Farma, S.A. US Patent | Assay Description Affinity for histamine H1-receptor was determined by binding studies to H1 receptors. The sample used is a suspension of membranes of CHO cells trans... | US Patent US10106522 (2018) BindingDB Entry DOI: 10.7270/Q2TF00DX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM293407 (US10106522, Example 5) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 54 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Faes Farma, S.A. US Patent | Assay Description Affinity for histamine H1-receptor was determined by binding studies to H1 receptors. The sample used is a suspension of membranes of CHO cells trans... | US Patent US10106522 (2018) BindingDB Entry DOI: 10.7270/Q2TF00DX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM293408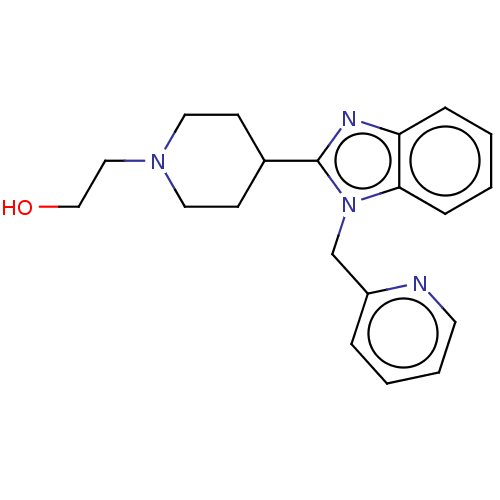 (US10106522, Example 7) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 175 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Faes Farma, S.A. US Patent | Assay Description Affinity for histamine H1-receptor was determined by binding studies to H1 receptors. The sample used is a suspension of membranes of CHO cells trans... | US Patent US10106522 (2018) BindingDB Entry DOI: 10.7270/Q2TF00DX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM293409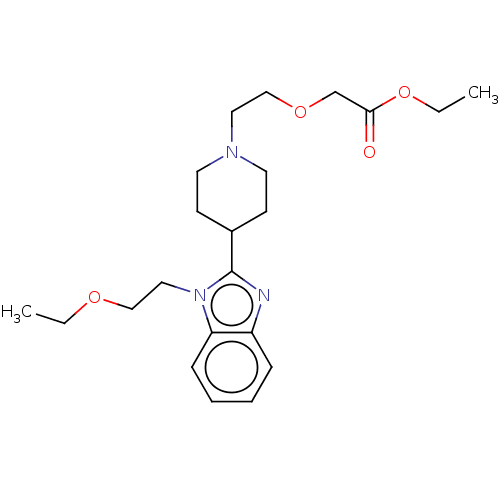 (US10106522, Example 8) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 169 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Faes Farma, S.A. US Patent | Assay Description Affinity for histamine H1-receptor was determined by binding studies to H1 receptors. The sample used is a suspension of membranes of CHO cells trans... | US Patent US10106522 (2018) BindingDB Entry DOI: 10.7270/Q2TF00DX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Cavia porcellus (domestic guinea pig)) | BDBM22566 (5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+3 | >-34.2 | n/a | n/a | n/a | n/a | n/a | 7.5 | 25 |
Johnson & Johnson Pharmaceutical | Assay Description The Ki values were calculated based on an experimentally determined appropriate Kd value according to Cheng and Prusoff. | J Pharmacol Exp Ther 309: 404-13 (2004) Article DOI: 10.1124/jpet.103.061754 BindingDB Entry DOI: 10.7270/Q2VX0DTD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM22566 (5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+4 | >-28.5 | n/a | n/a | n/a | n/a | n/a | 7.5 | 25 |
Johnson & Johnson Pharmaceutical | Assay Description The Ki values were calculated based on an experimentally determined appropriate Kd value according to Cheng and Prusoff. | J Pharmacol Exp Ther 309: 404-13 (2004) Article DOI: 10.1124/jpet.103.061754 BindingDB Entry DOI: 10.7270/Q2VX0DTD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Mus musculus (mouse)) | BDBM22566 (5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | >1.00E+4 | >-28.5 | n/a | n/a | n/a | n/a | n/a | 7.5 | 25 |
Johnson & Johnson Pharmaceutical | Assay Description The Ki values were calculated based on an experimentally determined appropriate Kd value according to Cheng and Prusoff. | J Pharmacol Exp Ther 309: 404-13 (2004) Article DOI: 10.1124/jpet.103.061754 BindingDB Entry DOI: 10.7270/Q2VX0DTD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM255474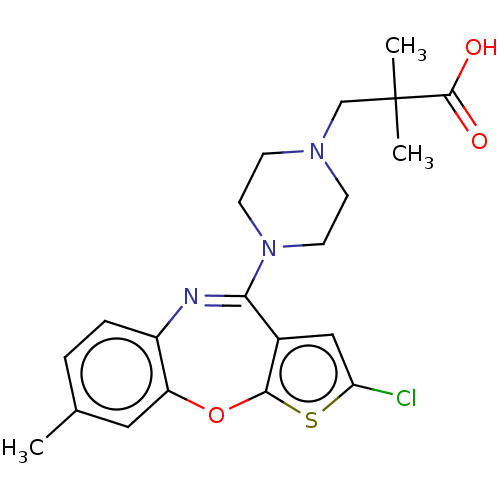 (US9481688, 1 | US9481688, 3) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 20.6 | -43.9 | n/a | n/a | n/a | n/a | n/a | 7.6 | 25 |
Eli Lilly and Company US Patent | Assay Description [3H]-Pyrilamine binding experiments are carried out in SPA (scintillation proximity assay) 96-well format. Membranes used in this assay are prepared ... | US Patent US9481688 (2016) BindingDB Entry DOI: 10.7270/Q25D8QRW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM255474 (US9481688, 1 | US9481688, 3) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 58.8 | -41.3 | n/a | n/a | n/a | n/a | n/a | 7.6 | 25 |
Eli Lilly and Company US Patent | Assay Description [3H]-Pyrilamine binding experiments are carried out in SPA (scintillation proximity assay) 96-well format. Membranes used in this assay are prepared ... | US Patent US9481688 (2016) BindingDB Entry DOI: 10.7270/Q25D8QRW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Cavia porcellus (domestic guinea pig)) | BDBM81541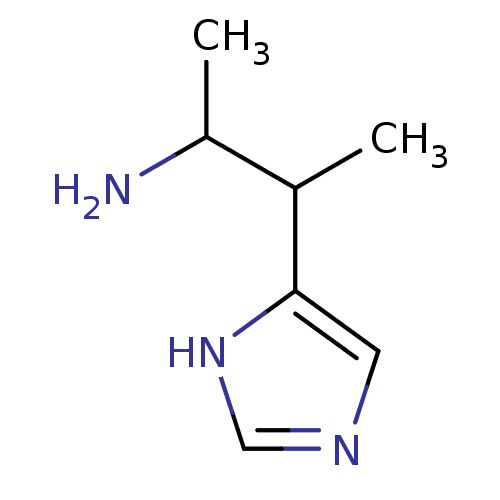 (CAS_195530 | NSC_195530 | S-Beta-Dimethylhistamine...) | UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Queen's Medical Centre Curated by PDSP Ki Database | Biochem Soc Trans 20: 122-5 (1992) Article DOI: 10.1042/bst0200122 BindingDB Entry DOI: 10.7270/Q2RX99KH | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50017721 (1-Methyl-4-(5H-dibenzo(a,d)cycloheptenylidene)pipe...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Université Libre de Bruxelles Curated by PDSP Ki Database | Eur J Biochem 224: 489-95 (1994) Article DOI: 10.1111/j.1432-1033.1994.00489.x BindingDB Entry DOI: 10.7270/Q2H70DB4 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM82479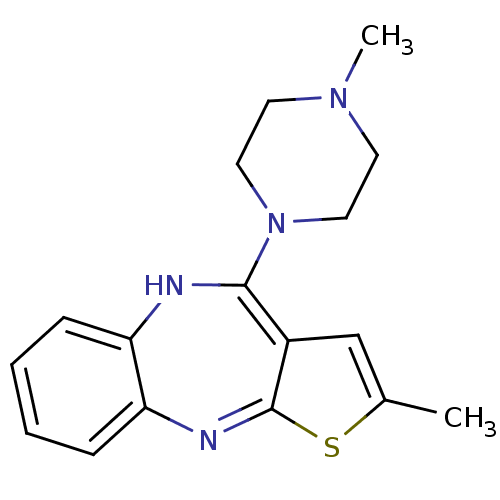 (CAS_132539-06-1 | NSC_4585 | OLANZAPINE | USRE4934...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents | Article PubMed | 0.0870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Foundation for Medical Education and Research and Mayo Clinic Curated by PDSP Ki Database | Life Sci 68: 29-39 (2000) Article DOI: 10.1016/s0024-3205(00)00911-5 BindingDB Entry DOI: 10.7270/Q2057DHP | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50079527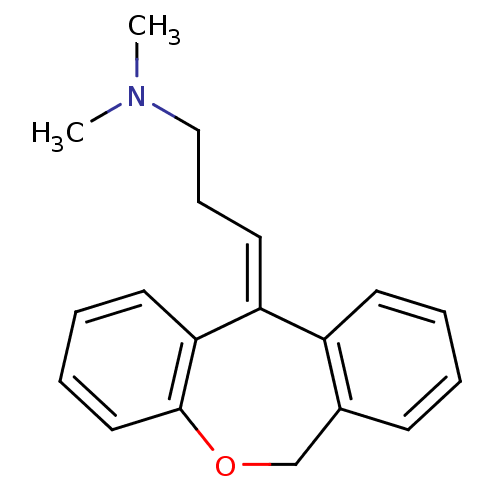 ((3E)-3-dibenzo[b,e]oxepin-11(6H)-ylidene-N,N-dimet...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill Curated by PDSP Ki Database | J Pharmacol Exp Ther 302: 328-36 (2002) Article DOI: 10.1124/jpet.302.1.328 BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50418268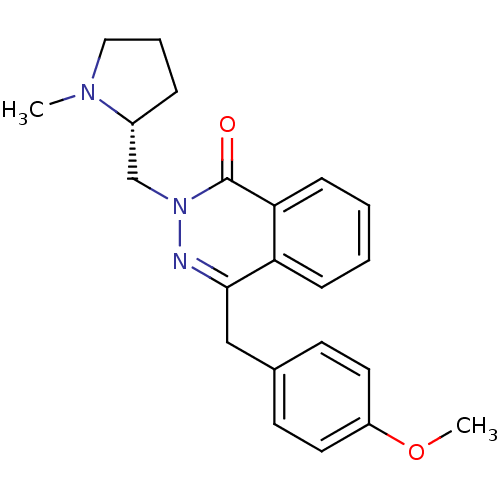 (CHEMBL1767137) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre Curated by ChEMBL | Assay Description Antagonist activity at human recombinant histamine H1 receptor expressed in intact CHO cells assessed as inhibition of histamine-induced cellular cal... | J Med Chem 54: 2183-95 (2011) Article DOI: 10.1021/jm1013874 BindingDB Entry DOI: 10.7270/Q2GQ6Z2Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Cavia porcellus (domestic guinea pig)) | BDBM50017721 (1-Methyl-4-(5H-dibenzo(a,d)cycloheptenylidene)pipe...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Université Libre de Bruxelles Curated by PDSP Ki Database | Eur J Biochem 224: 489-95 (1994) Article DOI: 10.1111/j.1432-1033.1994.00489.x BindingDB Entry DOI: 10.7270/Q2H70DB4 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Cavia porcellus (domestic guinea pig)) | BDBM50002099 (CHEMBL366965 | Dimethyl-[3-(2-methyl-6H-dibenzo[b,...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyowa Hakko Kogyo Co., Ltd. Curated by ChEMBL | Assay Description Binding affinity for histamine H1 receptor from guinea pig cerebellum, using [3H]pyrilamine as radioligand at 0.1 uM | J Med Chem 35: 2074-84 (1992) BindingDB Entry DOI: 10.7270/Q2N87BC0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50002087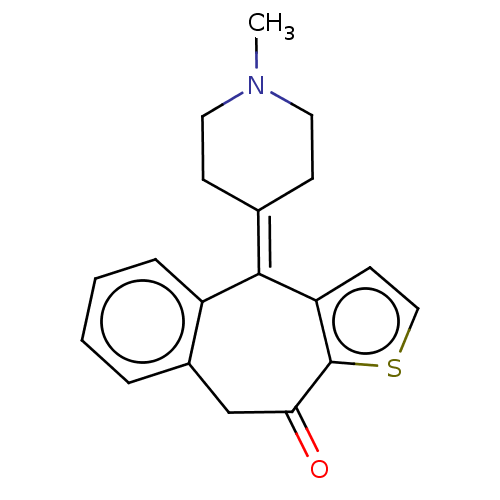 (4-(1-Methyl-piperidin-4-ylidene)-4,9-dihydro-1-thi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | DrugBank Article PubMed | 0.158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Antagonist activity against human histamine H1 receptor expressed in CHO cells by FLIPR assay | J Med Chem 53: 7778-95 (2010) Article DOI: 10.1021/jm100856p BindingDB Entry DOI: 10.7270/Q2FQ9WWQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50418267 (CHEMBL1767138) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre Curated by ChEMBL | Assay Description Antagonist activity at human recombinant histamine H1 receptor expressed in intact CHO cells assessed as inhibition of histamine-induced cellular cal... | J Med Chem 54: 2183-95 (2011) Article DOI: 10.1021/jm1013874 BindingDB Entry DOI: 10.7270/Q2GQ6Z2Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50391708 (CHEMBL1767136) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre Curated by ChEMBL | Assay Description Antagonist activity at human recombinant histamine H1 receptor expressed in intact CHO cells assessed as inhibition of histamine-induced cellular cal... | J Med Chem 54: 2183-95 (2011) Article DOI: 10.1021/jm1013874 BindingDB Entry DOI: 10.7270/Q2GQ6Z2Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Cavia porcellus (domestic guinea pig)) | BDBM50079527 ((3E)-3-dibenzo[b,e]oxepin-11(6H)-ylidene-N,N-dimet...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill Curated by PDSP Ki Database | J Pharmacol Exp Ther 302: 328-36 (2002) Article DOI: 10.1124/jpet.302.1.328 BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM84994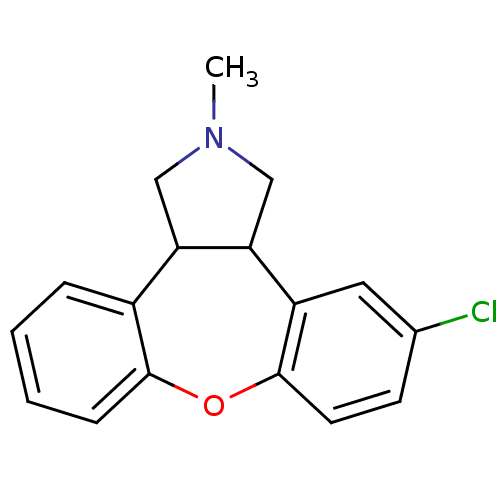 (CAS_163091 | NSC_163091 | ORG-5222) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank PC cid PC sid UniChem Similars | DrugBank Article PubMed | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation Curated by PDSP Ki Database | Psychopharmacology (Berl) 124: 57-73 (1996) Article DOI: 10.1007/bf02245606 BindingDB Entry DOI: 10.7270/Q2610XV6 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50391708 (CHEMBL1767136) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.162 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre Curated by ChEMBL | Assay Description Antagonist activity at human H1 receptor expressed in CHO cells assessed as inhibition of calcium mobilization by FLIPR assay | Bioorg Med Chem 20: 6097-108 (2012) Article DOI: 10.1016/j.bmc.2012.08.032 BindingDB Entry DOI: 10.7270/Q2NG4RQC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Cavia porcellus (domestic guinea pig)) | BDBM50020712 (10,11-dihydro-5-(gamma-dimethylaminopropylidene)-5...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Emory University Curated by PDSP Ki Database | J Pharmacol Exp Ther 283: 1305-22 (1997) BindingDB Entry DOI: 10.7270/Q25Q4TMX | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50079527 ((3E)-3-dibenzo[b,e]oxepin-11(6H)-ylidene-N,N-dimet...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Mayo Clinic Jacksonville Curated by PDSP Ki Database | Psychopharmacology (Berl) 114: 559-65 (1994) Article DOI: 10.1007/BF02244985 BindingDB Entry DOI: 10.7270/Q25X27FZ | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM112780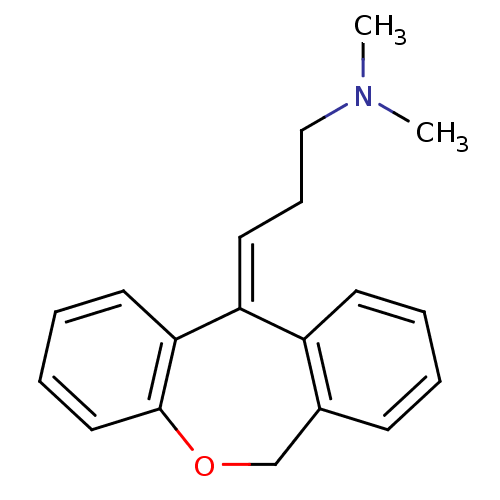 (US8629135, SW-07) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid PDB UniChem Similars | Article PubMed | 0.178 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details Article DOI: 10.1021/acs.jmedchem.2c00125 BindingDB Entry DOI: 10.7270/Q29W0KK1 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50079527 ((3E)-3-dibenzo[b,e]oxepin-11(6H)-ylidene-N,N-dimet...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.178 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
VU University Amsterdam Curated by ChEMBL | Assay Description Displacement of [3H]mepyramine from human histamine H1 receptor expressed in HEK293 cells after 1 to 1.5 hrs by scintillation counting | J Med Chem 54: 8195-206 (2011) Article DOI: 10.1021/jm2011589 BindingDB Entry DOI: 10.7270/Q2QF8T85 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM22872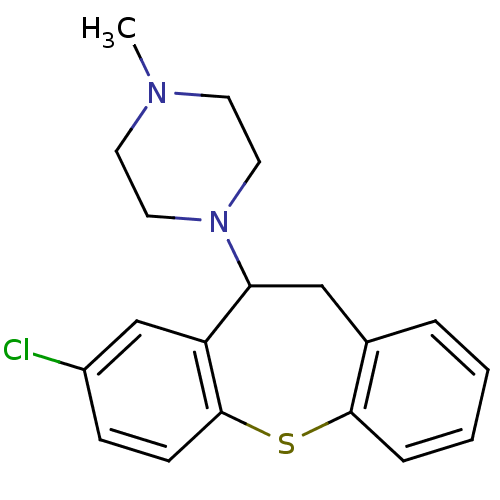 (1-(3-chloro-5,6-dihydrobenzo[b][1]benzothiepin-5-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen Curated by ChEMBL | Assay Description Displacement of [3H]prozosin from human cloned histamine H1 receptor expressed in CHO cells | J Med Chem 53: 7021-34 (2010) Article DOI: 10.1021/jm100652h BindingDB Entry DOI: 10.7270/Q20C4W0P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50418266 (CHEMBL1767141) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre Curated by ChEMBL | Assay Description Antagonist activity at human recombinant histamine H1 receptor expressed in intact CHO cells assessed as inhibition of histamine-induced cellular cal... | J Med Chem 54: 2183-95 (2011) Article DOI: 10.1021/jm1013874 BindingDB Entry DOI: 10.7270/Q2GQ6Z2Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM22869 (6-chloro-10-(4-methylpiperazin-1-yl)-2,9-diazatric...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation Curated by PDSP Ki Database | Psychopharmacology (Berl) 124: 57-73 (1996) Article DOI: 10.1007/bf02245606 BindingDB Entry DOI: 10.7270/Q2610XV6 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50418265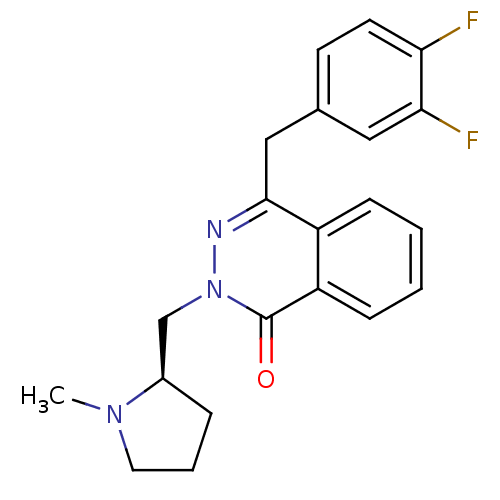 (CHEMBL1767149) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.251 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre Curated by ChEMBL | Assay Description Antagonist activity at human recombinant histamine H1 receptor expressed in intact CHO cells assessed as inhibition of histamine-induced cellular cal... | J Med Chem 54: 2183-95 (2011) Article DOI: 10.1021/jm1013874 BindingDB Entry DOI: 10.7270/Q2GQ6Z2Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50391698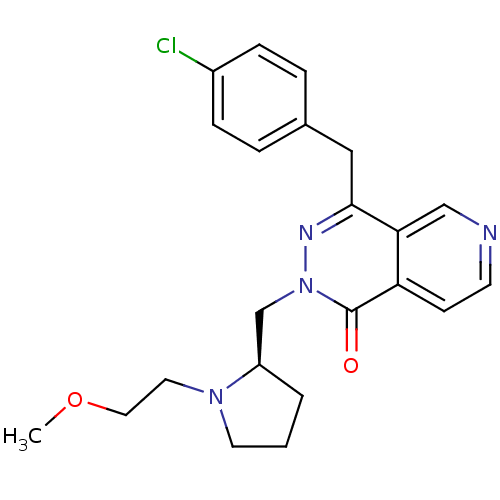 (CHEMBL2146801) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.251 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre Curated by ChEMBL | Assay Description Antagonist activity at human H1 receptor expressed in CHO cells assessed as inhibition of calcium mobilization by FLIPR assay | Bioorg Med Chem 20: 6097-108 (2012) Article DOI: 10.1016/j.bmc.2012.08.032 BindingDB Entry DOI: 10.7270/Q2NG4RQC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50418298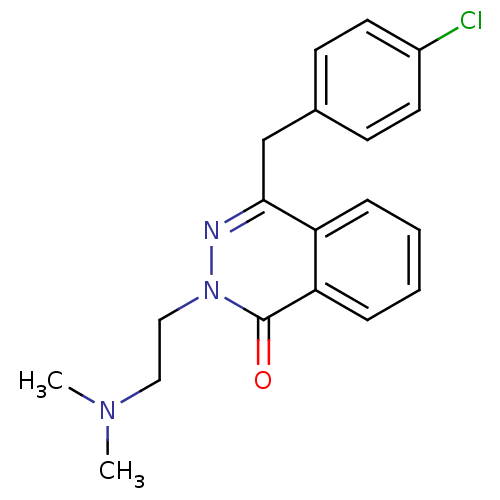 (CHEMBL1767134) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.251 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Medicines Research Centre Curated by ChEMBL | Assay Description Antagonist activity at human recombinant histamine H1 receptor expressed in intact CHO cells assessed as inhibition of histamine-induced cellular cal... | J Med Chem 54: 2183-95 (2011) Article DOI: 10.1021/jm1013874 BindingDB Entry DOI: 10.7270/Q2GQ6Z2Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50079527 ((3E)-3-dibenzo[b,e]oxepin-11(6H)-ylidene-N,N-dimet...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.251 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Amsterdam Curated by ChEMBL | Assay Description Displacement of [3H]mepyramine from human wild type N-terminal hemagglutinin-tagged histamine H1 receptor expressed in HEK293T cells after 4 hrs by m... | J Med Chem 59: 9047-9061 (2016) Article DOI: 10.1021/acs.jmedchem.6b00981 BindingDB Entry DOI: 10.7270/Q2G44S8Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Cavia porcellus (domestic guinea pig)) | BDBM50079527 ((3E)-3-dibenzo[b,e]oxepin-11(6H)-ylidene-N,N-dimet...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill Curated by PDSP Ki Database | J Pharmacol Exp Ther 302: 328-36 (2002) Article DOI: 10.1124/jpet.302.1.328 BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Homo sapiens (Human)) | BDBM50315206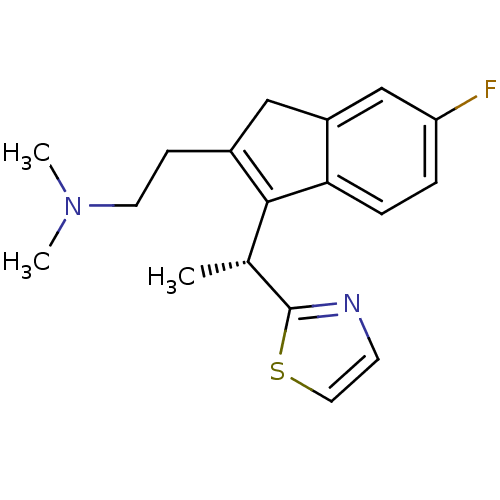 ((R)-2-(6-fluoro-3-(1-(thiazol-2-yl)ethyl)-1H-inden...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences Curated by ChEMBL | Assay Description Binding affinity at histamine H1 receptor | Bioorg Med Chem Lett 20: 2629-33 (2010) Article DOI: 10.1016/j.bmcl.2010.02.055 BindingDB Entry DOI: 10.7270/Q2NG4QSV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (RAT) | BDBM35254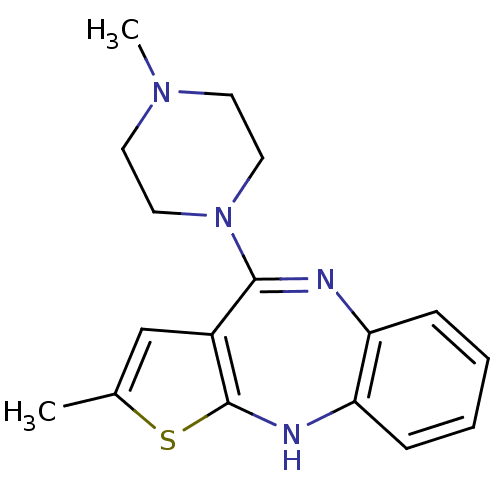 (2-methyl-4-(4-methylpiperazin-1-yl)-10H-thieno[2,3...) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG PC cid PC sid UniChem Patents Similars | PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Siena Curated by ChEMBL | Assay Description Half-maximal inhibition of [3H]7-OH-DPAT binding to Histamine H1 receptor in rat tissue homogenate | J Med Chem 45: 344-59 (2002) BindingDB Entry DOI: 10.7270/Q2TX3G26 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H1 receptor (Cavia porcellus (domestic guinea pig)) | BDBM50002087 (4-(1-Methyl-piperidin-4-ylidene)-4,9-dihydro-1-thi...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid UniChem Patents Similars | PubMed | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Kyowa Hakko Kogyo Co., Ltd. Curated by ChEMBL | Assay Description The compound was tested in vitro for binding affinity against histamine H1 receptor from guinea pig cerebellum, using [3H]pyrilamine as radioligand a... | J Med Chem 35: 2074-84 (1992) BindingDB Entry DOI: 10.7270/Q2N87BC0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 3469 total ) | Next | Last >> |