| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Acetylcholinesterase |
|---|
| Ligand | BDBM50556952 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2057667 (CHEMBL4712668) |
|---|
| Kd | 3.6±n/a nM |
|---|
| Citation |  Viayna, E; Coquelle, N; Cieslikiewicz-Bouet, M; Cisternas, P; Oliva, CA; S�nchez-L�pez, E; Ettcheto, M; Bartolini, M; De Simone, A; Ricchini, M; Rendina, M; Pons, M; Firuzi, O; P�rez, B; Saso, L; Andrisano, V; Nachon, F; Brazzolotto, X; Garc�a, ML; Camins, A; Silman, I; Jean, L; Inestrosa, NC; Colletier, JP; Renard, PY; Mu�oz-Torrero, D Discovery of a Potent Dual Inhibitor of Acetylcholinesterase and Butyrylcholinesterase with Antioxidant Activity that Alleviates Alzheimer-like Pathology in Old APP/PS1 Mice. J Med Chem64:812-839 (2021) [PubMed] Article Viayna, E; Coquelle, N; Cieslikiewicz-Bouet, M; Cisternas, P; Oliva, CA; S�nchez-L�pez, E; Ettcheto, M; Bartolini, M; De Simone, A; Ricchini, M; Rendina, M; Pons, M; Firuzi, O; P�rez, B; Saso, L; Andrisano, V; Nachon, F; Brazzolotto, X; Garc�a, ML; Camins, A; Silman, I; Jean, L; Inestrosa, NC; Colletier, JP; Renard, PY; Mu�oz-Torrero, D Discovery of a Potent Dual Inhibitor of Acetylcholinesterase and Butyrylcholinesterase with Antioxidant Activity that Alleviates Alzheimer-like Pathology in Old APP/PS1 Mice. J Med Chem64:812-839 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Acetylcholinesterase |
|---|
| Name: | Acetylcholinesterase |
|---|
| Synonyms: | ACES_ELEEL | Acetylcholinesterase (AChE) | Acetylcholinesterase (EeAChE) | ache |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 71812.79 |
|---|
| Organism: | Electrophorus electricus (Electric eel) |
|---|
| Description: | n/a |
|---|
| Residue: | 633 |
|---|
| Sequence: | MKILDALLFPVIFIMFFIHLSIAQTDPELTIMTRLGQVQGTRLPVPDRSHVIAFLGIPFA
EPPLGKMRFKPPEPKKPWNDVFDARDYPSACYQYVDTSYPGFSGTEMWNPNRMMSEDCLY
LNVWVPATPRPHNLTVMVWIYGGGFYSGSSSLDVYDGRYLAHSEKVVVVSMNYRVSAFGF
LALNGSAEAPGNVGLLDQRLALQWVQDNIHFFGGNPKQVTIFGESAGAASVGMHLLSPDS
RPKFTRAILQSGVPNGPWRTVSFDEARRRAIKLGRLVGCPDGNDTDLIDCLRSKQPQDLI
DQEWLVLPFSGLFRFSFVPVIDGVVFPDTPEAMLNSGNFKDTQILLGVNQNEGSYFLIYG
APGFSKDNESLITREDFLQGVKMSVPHANEIGLEAVILQYTDWMDEDNPIKNREAMDDIV
GDHNVVCPLQHFAKMYAQYSILQGQTGTASQGNLGWGNSGSASNSGNSQVSVYLYMFDHR
ASNLVWPEWMGVIHGYEIEFVFGLPLEKRLNYTLEEEKLSRRMMKYWANFARTGNPNINV
DGSIDSRRRWPVFTSTEQKHVGLNTDSLKVHKGLKSQFCALWNRFLPRLLNVTENIDDAE
RQWKAEFHRWSSYMMHWKNQFDHYSKQERCTNL
|
|
|
|---|
| BDBM50556952 |
|---|
| n/a |
|---|
| Name | BDBM50556952 |
|---|
| Synonyms: | CHEMBL4754487 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H38ClN3O3 |
|---|
| Mol. Mass. | 548.115 |
|---|
| SMILES | COc1cc(CNC(=O)CCCCCCNc2c3C4CC(Cc3nc3cc(Cl)ccc23)C=C(C)C4)ccc1O |t:34,TLB:23:22:19:34.31.32,THB:16:17:19:34.31.32| |
|---|
| Structure | 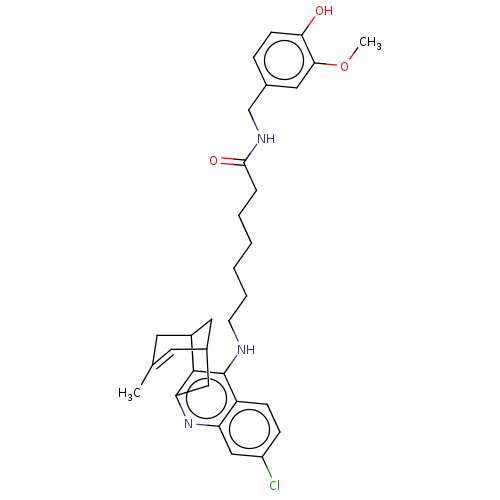
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Viayna, E; Coquelle, N; Cieslikiewicz-Bouet, M; Cisternas, P; Oliva, CA; S�nchez-L�pez, E; Ettcheto, M; Bartolini, M; De Simone, A; Ricchini, M; Rendina, M; Pons, M; Firuzi, O; P�rez, B; Saso, L; Andrisano, V; Nachon, F; Brazzolotto, X; Garc�a, ML; Camins, A; Silman, I; Jean, L; Inestrosa, NC; Colletier, JP; Renard, PY; Mu�oz-Torrero, D Discovery of a Potent Dual Inhibitor of Acetylcholinesterase and Butyrylcholinesterase with Antioxidant Activity that Alleviates Alzheimer-like Pathology in Old APP/PS1 Mice. J Med Chem64:812-839 (2021) [PubMed] Article
Viayna, E; Coquelle, N; Cieslikiewicz-Bouet, M; Cisternas, P; Oliva, CA; S�nchez-L�pez, E; Ettcheto, M; Bartolini, M; De Simone, A; Ricchini, M; Rendina, M; Pons, M; Firuzi, O; P�rez, B; Saso, L; Andrisano, V; Nachon, F; Brazzolotto, X; Garc�a, ML; Camins, A; Silman, I; Jean, L; Inestrosa, NC; Colletier, JP; Renard, PY; Mu�oz-Torrero, D Discovery of a Potent Dual Inhibitor of Acetylcholinesterase and Butyrylcholinesterase with Antioxidant Activity that Alleviates Alzheimer-like Pathology in Old APP/PS1 Mice. J Med Chem64:812-839 (2021) [PubMed] Article