| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuraminidase |
|---|
| Ligand | BDBM4994 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1735128 (CHEMBL4150664) |
|---|
| IC50 | 4.2±n/a nM |
|---|
| Citation |  Zhang, J; Poongavanam, V; Kang, D; Bertagnin, C; Lu, H; Kong, X; Ju, H; Lu, X; Gao, P; Tian, Y; Jia, H; Desta, S; Ding, X; Sun, L; Fang, Z; Huang, B; Liang, X; Jia, R; Ma, X; Xu, W; Murugan, NA; Loregian, A; Huang, B; Zhan, P; Liu, X Optimization of N-Substituted Oseltamivir Derivatives as Potent Inhibitors of Group-1 and -2 Influenza A Neuraminidases, Including a Drug-Resistant Variant. J Med Chem61:6379-6397 (2018) [PubMed] Article Zhang, J; Poongavanam, V; Kang, D; Bertagnin, C; Lu, H; Kong, X; Ju, H; Lu, X; Gao, P; Tian, Y; Jia, H; Desta, S; Ding, X; Sun, L; Fang, Z; Huang, B; Liang, X; Jia, R; Ma, X; Xu, W; Murugan, NA; Loregian, A; Huang, B; Zhan, P; Liu, X Optimization of N-Substituted Oseltamivir Derivatives as Potent Inhibitors of Group-1 and -2 Influenza A Neuraminidases, Including a Drug-Resistant Variant. J Med Chem61:6379-6397 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuraminidase |
|---|
| Name: | Neuraminidase |
|---|
| Synonyms: | NA | NRAM_I33A0 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 49687.56 |
|---|
| Organism: | Influenza A virus |
|---|
| Description: | ChEMBL_1347422 |
|---|
| Residue: | 453 |
|---|
| Sequence: | MNPNQKIITIGSICMVVGIISLILQIGNIISIWISHSIQTGNQNHTGICNQGIITYNVVA
GQDSTSVILTGNSSLCPIRGWAIHSKDNGIRIGSKGDVFVIREPFISCSHLECRTFFLTQ
GALLNDKHSNGTVKDRSPYRALMSCPVGEAPSPYNSRFESVAWSASACHDGMGWLTIGIS
GPDNGAVAVLKYNGIITETIKSWRKKILRTQESECTCVNGSCFTIMTDGPSNGLASYKIF
KIEKGKVTKSIELNAPNSHYEECSCYPDTGKVMCVCRDNWHGSNRPWVSFDQNLDYQIGY
ICSGVFGDNPRPKDGPGSCGPVSADGANGVKGFSYRYGNGVWIGRTKSDSSRHGFEMIWD
PNGWTETDSRFSVRQDVVAMTDRSGYSGSFVQHPELTGLDCMRPCFWVELIRGRPEEETI
WTSGSIISFCGVNSDTVDWSWPDGAELPFTIDK
|
|
|
|---|
| BDBM4994 |
|---|
| n/a |
|---|
| Name | BDBM4994 |
|---|
| Synonyms: | (3R,4R,5S)-5-amino-4-acetamido-3-(pentan-3-yloxy)cyclohex-1-ene-1-carboxylic acid | CHEMBL674 | GS4071 | Oseltamivir carboxylate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C14H24N2O4 |
|---|
| Mol. Mass. | 284.3514 |
|---|
| SMILES | CCC(CC)O[C@@H]1C=C(C[C@H](N)[C@H]1NC(C)=O)C(O)=O |r,c:7| |
|---|
| Structure | 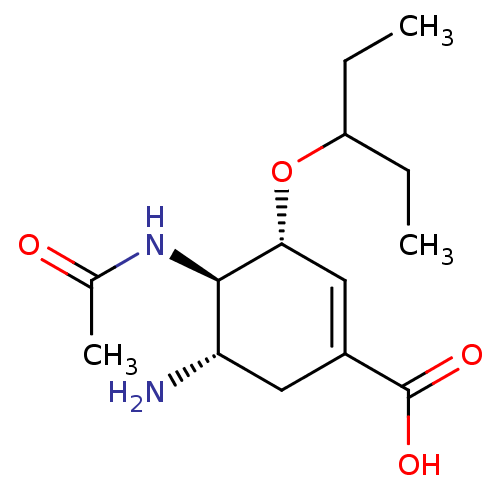
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhang, J; Poongavanam, V; Kang, D; Bertagnin, C; Lu, H; Kong, X; Ju, H; Lu, X; Gao, P; Tian, Y; Jia, H; Desta, S; Ding, X; Sun, L; Fang, Z; Huang, B; Liang, X; Jia, R; Ma, X; Xu, W; Murugan, NA; Loregian, A; Huang, B; Zhan, P; Liu, X Optimization of N-Substituted Oseltamivir Derivatives as Potent Inhibitors of Group-1 and -2 Influenza A Neuraminidases, Including a Drug-Resistant Variant. J Med Chem61:6379-6397 (2018) [PubMed] Article
Zhang, J; Poongavanam, V; Kang, D; Bertagnin, C; Lu, H; Kong, X; Ju, H; Lu, X; Gao, P; Tian, Y; Jia, H; Desta, S; Ding, X; Sun, L; Fang, Z; Huang, B; Liang, X; Jia, R; Ma, X; Xu, W; Murugan, NA; Loregian, A; Huang, B; Zhan, P; Liu, X Optimization of N-Substituted Oseltamivir Derivatives as Potent Inhibitors of Group-1 and -2 Influenza A Neuraminidases, Including a Drug-Resistant Variant. J Med Chem61:6379-6397 (2018) [PubMed] Article