| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50122318 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1519125 (CHEMBL3624318) |
|---|
| IC50 | 3000±n/a nM |
|---|
| Citation |  Wan, H; Schroeder, GM; Hart, AC; Inghrim, J; Grebinski, J; Tokarski, JS; Lorenzi, MV; You, D; Mcdevitt, T; Penhallow, B; Vuppugalla, R; Zhang, Y; Gu, X; Iyer, R; Lombardo, LJ; Trainor, GL; Ruepp, S; Lippy, J; Blat, Y; Sack, JS; Khan, JA; Stefanski, K; Sleczka, B; Mathur, A; Sun, JH; Wong, MK; Wu, DR; Li, P; Gupta, A; Arunachalam, PN; Pragalathan, B; Narayanan, S; K C, N; Kuppusamy, P; Purandare, AV Discovery of a Highly Selective JAK2 Inhibitor, BMS-911543, for the Treatment of Myeloproliferative Neoplasms. ACS Med Chem Lett6:850-5 (2015) [PubMed] Article Wan, H; Schroeder, GM; Hart, AC; Inghrim, J; Grebinski, J; Tokarski, JS; Lorenzi, MV; You, D; Mcdevitt, T; Penhallow, B; Vuppugalla, R; Zhang, Y; Gu, X; Iyer, R; Lombardo, LJ; Trainor, GL; Ruepp, S; Lippy, J; Blat, Y; Sack, JS; Khan, JA; Stefanski, K; Sleczka, B; Mathur, A; Sun, JH; Wong, MK; Wu, DR; Li, P; Gupta, A; Arunachalam, PN; Pragalathan, B; Narayanan, S; K C, N; Kuppusamy, P; Purandare, AV Discovery of a Highly Selective JAK2 Inhibitor, BMS-911543, for the Treatment of Myeloproliferative Neoplasms. ACS Med Chem Lett6:850-5 (2015) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50122318 |
|---|
| n/a |
|---|
| Name | BDBM50122318 |
|---|
| Synonyms: | BMS-911543 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H28N8O |
|---|
| Mol. Mass. | 432.5214 |
|---|
| SMILES | CCn1c(cc2c1nc(Nc1cc(C)n(C)n1)c1ncn(C)c21)C(=O)N(C1CC1)C1CC1 |
|---|
| Structure | 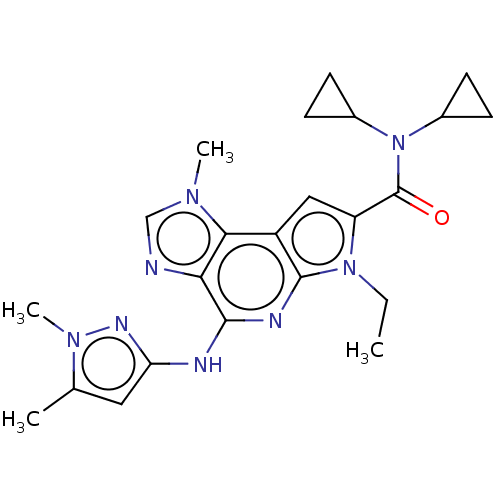
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wan, H; Schroeder, GM; Hart, AC; Inghrim, J; Grebinski, J; Tokarski, JS; Lorenzi, MV; You, D; Mcdevitt, T; Penhallow, B; Vuppugalla, R; Zhang, Y; Gu, X; Iyer, R; Lombardo, LJ; Trainor, GL; Ruepp, S; Lippy, J; Blat, Y; Sack, JS; Khan, JA; Stefanski, K; Sleczka, B; Mathur, A; Sun, JH; Wong, MK; Wu, DR; Li, P; Gupta, A; Arunachalam, PN; Pragalathan, B; Narayanan, S; K C, N; Kuppusamy, P; Purandare, AV Discovery of a Highly Selective JAK2 Inhibitor, BMS-911543, for the Treatment of Myeloproliferative Neoplasms. ACS Med Chem Lett6:850-5 (2015) [PubMed] Article
Wan, H; Schroeder, GM; Hart, AC; Inghrim, J; Grebinski, J; Tokarski, JS; Lorenzi, MV; You, D; Mcdevitt, T; Penhallow, B; Vuppugalla, R; Zhang, Y; Gu, X; Iyer, R; Lombardo, LJ; Trainor, GL; Ruepp, S; Lippy, J; Blat, Y; Sack, JS; Khan, JA; Stefanski, K; Sleczka, B; Mathur, A; Sun, JH; Wong, MK; Wu, DR; Li, P; Gupta, A; Arunachalam, PN; Pragalathan, B; Narayanan, S; K C, N; Kuppusamy, P; Purandare, AV Discovery of a Highly Selective JAK2 Inhibitor, BMS-911543, for the Treatment of Myeloproliferative Neoplasms. ACS Med Chem Lett6:850-5 (2015) [PubMed] Article