 Found 6917 hits with Last Name = 'cheng' and Initial = 'c'
Found 6917 hits with Last Name = 'cheng' and Initial = 'c' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514220
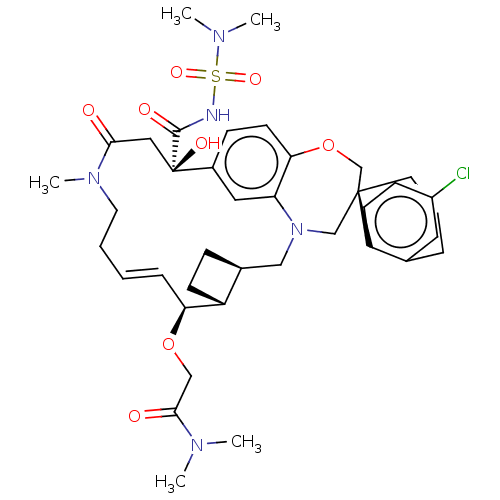
(CHEMBL4535151 | US11274105, Example 188)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OCC(=O)N(C)C)\C=C\CCN(C)C(=O)C[C@](O)(C(=O)NS(=O)(=O)N(C)C)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:15| Show InChI InChI=1S/C39H52ClN5O8S/c1-42(2)36(47)23-52-33-10-6-7-18-44(5)35(46)21-39(49,37(48)41-54(50,51)43(3)4)28-12-16-34-32(20-28)45(22-27-11-14-30(27)33)24-38(25-53-34)17-8-9-26-19-29(40)13-15-31(26)38/h6,10,12-13,15-16,19-20,27,30,33,49H,7-9,11,14,17-18,21-25H2,1-5H3,(H,41,48)/b10-6+/t27-,30+,33-,38-,39+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514203
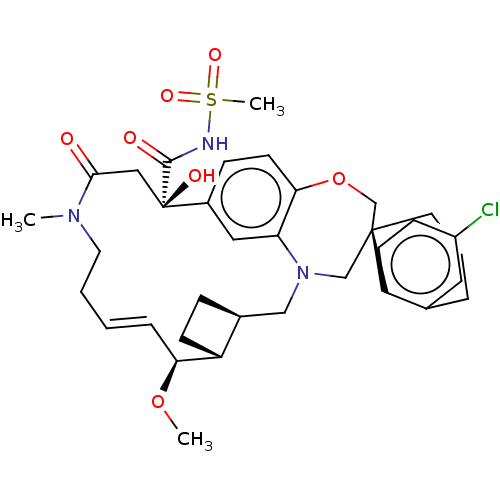
(CHEMBL4593361 | US11274105, Example 6)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OC)\C=C\CCN(C)C(=O)C[C@](O)(C(=O)NS(C)(=O)=O)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:10| Show InChI InChI=1S/C35H44ClN3O7S/c1-38-16-5-4-8-30(45-2)27-12-9-24(27)20-39-21-34(15-6-7-23-17-26(36)11-13-28(23)34)22-46-31-14-10-25(18-29(31)39)35(42,19-32(38)40)33(41)37-47(3,43)44/h4,8,10-11,13-14,17-18,24,27,30,42H,5-7,9,12,15-16,19-22H2,1-3H3,(H,37,41)/b8-4+/t24-,27+,30-,34-,35+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514222
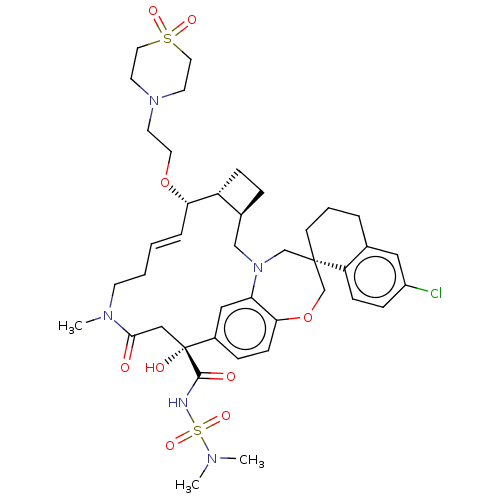
(CHEMBL4580244 | US11274105, Example 193)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OCCN1CCS(=O)(=O)CC1)\C=C\CCN(C)C(=O)C[C@](O)(C(=O)NS(=O)(=O)N(C)C)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:20| Show InChI InChI=1S/C41H56ClN5O9S2/c1-44(2)58(53,54)43-39(49)41(50)25-38(48)45(3)16-5-4-8-36(55-20-17-46-18-21-57(51,52)22-19-46)33-12-9-30(33)26-47-27-40(28-56-37-14-10-31(41)24-35(37)47)15-6-7-29-23-32(42)11-13-34(29)40/h4,8,10-11,13-14,23-24,30,33,36,50H,5-7,9,12,15-22,25-28H2,1-3H3,(H,43,49)/b8-4+/t30-,33+,36-,40-,41+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514196
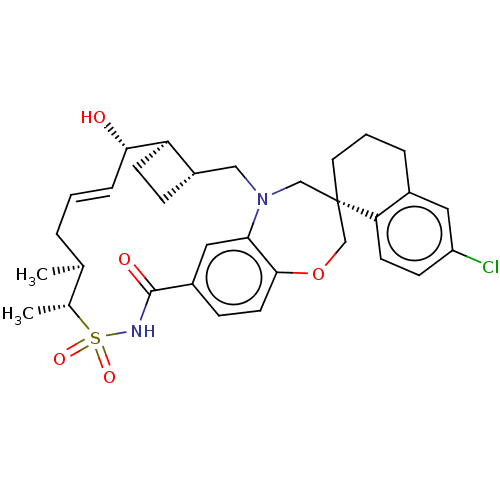
(CHEMBL4476472)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](O)\C=C\C[C@H](C)[C@@H](C)S(=O)(=O)NC(=O)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:9| Show InChI InChI=1S/C32H39ClN2O5S/c1-20-5-3-7-29(36)26-11-8-24(26)17-35-18-32(14-4-6-22-15-25(33)10-12-27(22)32)19-40-30-13-9-23(16-28(30)35)31(37)34-41(38,39)21(20)2/h3,7,9-10,12-13,15-16,20-21,24,26,29,36H,4-6,8,11,14,17-19H2,1-2H3,(H,34,37)/b7-3+/t20-,21+,24-,26+,29-,32-/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 0.0510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514202

(CHEMBL4446369 | US11274105, Example 179)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OCCN1CC(F)(F)C1)\C=C\CCN(C)C(=O)C[C@](O)(C(=O)NS(=O)(=O)N(C)C)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:18| Show InChI InChI=1S/C40H52ClF2N5O7S/c1-45(2)56(52,53)44-37(50)40(51)21-36(49)46(3)16-5-4-8-34(54-18-17-47-24-39(42,43)25-47)31-12-9-28(31)22-48-23-38(26-55-35-14-10-29(40)20-33(35)48)15-6-7-27-19-30(41)11-13-32(27)38/h4,8,10-11,13-14,19-20,28,31,34,51H,5-7,9,12,15-18,21-26H2,1-3H3,(H,44,50)/b8-4+/t28-,31+,34-,38-,40+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514199
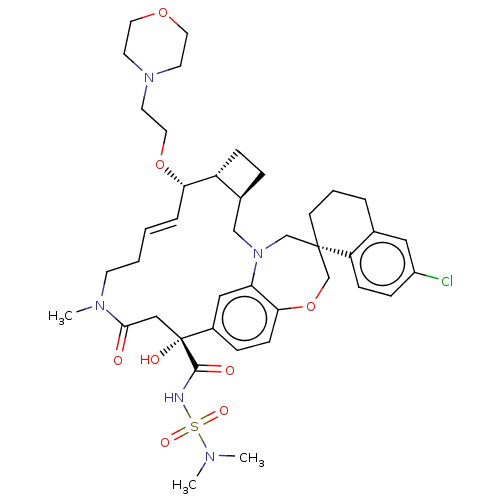
(CHEMBL4553660 | US11274105, Example 182)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OCCN1CCOCC1)\C=C\CCN(C)C(=O)C[C@](O)(C(=O)NS(=O)(=O)N(C)C)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:18| Show InChI InChI=1S/C41H56ClN5O8S/c1-44(2)56(51,52)43-39(49)41(50)25-38(48)45(3)16-5-4-8-36(54-22-19-46-17-20-53-21-18-46)33-12-9-30(33)26-47-27-40(28-55-37-14-10-31(41)24-35(37)47)15-6-7-29-23-32(42)11-13-34(29)40/h4,8,10-11,13-14,23-24,30,33,36,50H,5-7,9,12,15-22,25-28H2,1-3H3,(H,43,49)/b8-4+/t30-,33+,36-,40-,41+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(Homo sapiens (Human)) | BDBM50370582
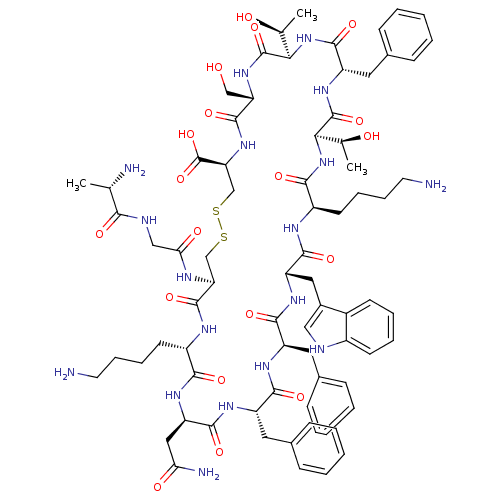
(CHEMBL1791306)Show SMILES C[C@H](O)[C@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](NC(=O)[C@@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CSSC[C@H](NC(=O)[C@H](CO)NC1=O)C(O)=O)NC(=O)CNC(=O)[C@H](C)N)[C@H](C)O Show InChI InChI=1S/C76H104N18O19S2/c1-41(79)64(100)82-37-61(99)83-58-39-114-115-40-59(76(112)113)92-72(108)57(38-95)91-75(111)63(43(3)97)94-71(107)54(33-46-23-11-6-12-24-46)90-74(110)62(42(2)96)93-66(102)51(28-16-18-30-78)84-69(105)55(34-47-36-81-49-26-14-13-25-48(47)49)88-68(104)53(32-45-21-9-5-10-22-45)86-67(103)52(31-44-19-7-4-8-20-44)87-70(106)56(35-60(80)98)89-65(101)50(85-73(58)109)27-15-17-29-77/h4-14,19-26,36,41-43,50-59,62-63,81,95-97H,15-18,27-35,37-40,77-79H2,1-3H3,(H2,80,98)(H,82,100)(H,83,99)(H,84,105)(H,85,109)(H,86,103)(H,87,106)(H,88,104)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t41-,42-,43-,50-,51+,52-,53+,54-,55-,56+,57-,58-,59-,62+,63+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by ChEMBL
| Assay Description
Inhibition of human somatostatin receptor type 2 |
J Med Chem 48: 4025-30 (2005)
Article DOI: 10.1021/jm058184l
BindingDB Entry DOI: 10.7270/Q2736RP5 |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514200
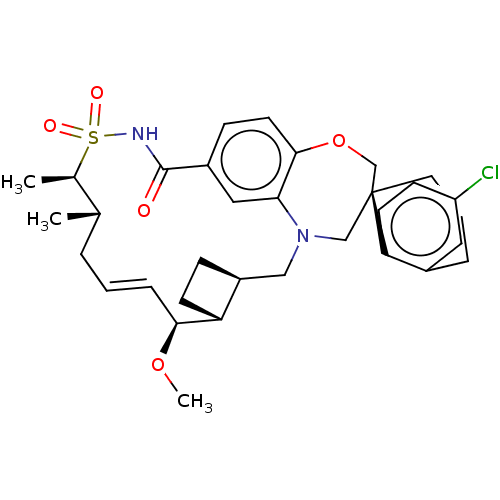
(CHEMBL4446378 | US10703733, Comparative Example 1)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OC)\C=C\C[C@H](C)[C@@H](C)S(=O)(=O)NC(=O)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:10| Show InChI InChI=1S/C33H41ClN2O5S/c1-21-6-4-8-30(40-3)27-12-9-25(27)18-36-19-33(15-5-7-23-16-26(34)11-13-28(23)33)20-41-31-14-10-24(17-29(31)36)32(37)35-42(38,39)22(21)2/h4,8,10-11,13-14,16-17,21-22,25,27,30H,5-7,9,12,15,18-20H2,1-3H3,(H,35,37)/b8-4+/t21-,22+,25-,27+,30-,33-/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 0.0760 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514215
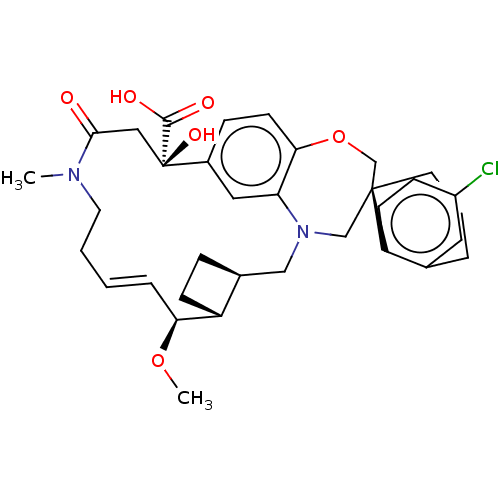
(CHEMBL4577379 | US11274105, Example 4)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OC)\C=C\CCN(C)C(=O)C[C@](O)(C(O)=O)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:10| Show InChI InChI=1S/C34H41ClN2O6/c1-36-15-4-3-7-29(42-2)26-11-8-23(26)19-37-20-33(14-5-6-22-16-25(35)10-12-27(22)33)21-43-30-13-9-24(17-28(30)37)34(41,32(39)40)18-31(36)38/h3,7,9-10,12-13,16-17,23,26,29,41H,4-6,8,11,14-15,18-21H2,1-2H3,(H,39,40)/b7-3+/t23-,26+,29-,33-,34+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514218
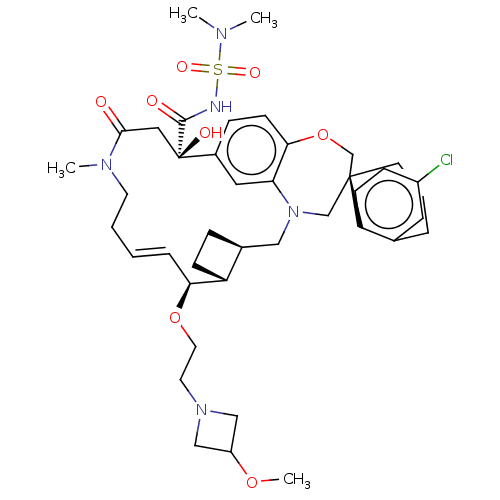
(CHEMBL4539543 | US11274105, Example 197)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OCCN1CC(C1)OC)\C=C\CCN(C)C(=O)C[C@](O)(C(=O)NS(=O)(=O)N(C)C)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:18| Show InChI InChI=1S/C41H56ClN5O8S/c1-44(2)56(51,52)43-39(49)41(50)22-38(48)45(3)17-6-5-9-36(54-19-18-46-24-32(25-46)53-4)33-13-10-29(33)23-47-26-40(27-55-37-15-11-30(41)21-35(37)47)16-7-8-28-20-31(42)12-14-34(28)40/h5,9,11-12,14-15,20-21,29,32-33,36,50H,6-8,10,13,16-19,22-27H2,1-4H3,(H,43,49)/b9-5+/t29-,33+,36-,40-,41+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514206
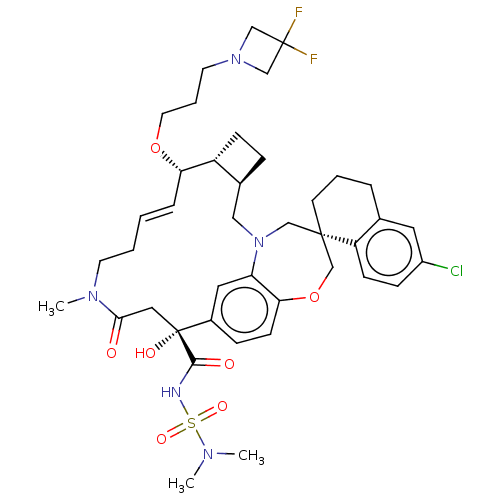
(CHEMBL4588330 | US11274105, Example 187)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OCCCN1CC(F)(F)C1)\C=C\CCN(C)C(=O)C[C@](O)(C(=O)NS(=O)(=O)N(C)C)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:19| Show InChI InChI=1S/C41H54ClF2N5O7S/c1-46(2)57(53,54)45-38(51)41(52)22-37(50)47(3)17-5-4-9-35(55-19-7-18-48-25-40(43,44)26-48)32-13-10-29(32)23-49-24-39(27-56-36-15-11-30(41)21-34(36)49)16-6-8-28-20-31(42)12-14-33(28)39/h4,9,11-12,14-15,20-21,29,32,35,52H,5-8,10,13,16-19,22-27H2,1-3H3,(H,45,51)/b9-4+/t29-,32+,35-,39-,41+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514201

(CHEMBL4547370 | US11274105, Example 191)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OCCOCC(F)F)\C=C\CCN(C)C(=O)C[C@](O)(C(=O)NS(=O)(=O)N(C)C)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:16| Show InChI InChI=1S/C39H51ClF2N4O8S/c1-44(2)55(50,51)43-37(48)39(49)21-36(47)45(3)16-5-4-8-33(53-18-17-52-23-35(41)42)30-12-9-27(30)22-46-24-38(25-54-34-14-10-28(39)20-32(34)46)15-6-7-26-19-29(40)11-13-31(26)38/h4,8,10-11,13-14,19-20,27,30,33,35,49H,5-7,9,12,15-18,21-25H2,1-3H3,(H,43,48)/b8-4+/t27-,30+,33-,38-,39+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514214

(CHEMBL4542646 | US11274105, Example 41)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](O)\C=C\CCN(C)C(=O)C[C@](O)(C(O)=O)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:9| Show InChI InChI=1S/C33H39ClN2O6/c1-35-14-3-2-6-28(37)25-10-7-22(25)18-36-19-32(13-4-5-21-15-24(34)9-11-26(21)32)20-42-29-12-8-23(16-27(29)36)33(41,31(39)40)17-30(35)38/h2,6,8-9,11-12,15-16,22,25,28,37,41H,3-5,7,10,13-14,17-20H2,1H3,(H,39,40)/b6-2+/t22-,25+,28-,32-,33+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514216
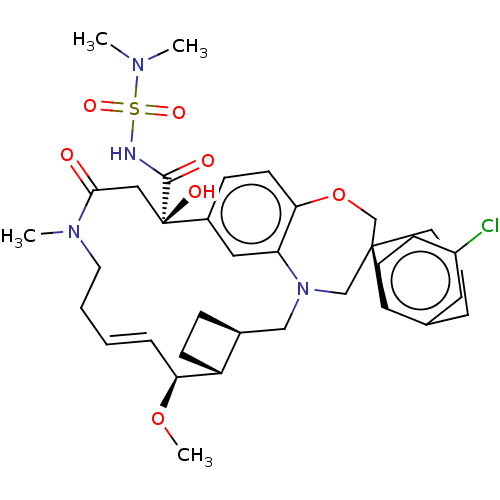
(CHEMBL4528051 | US11274105, Example 5)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OC)\C=C\CCN(C)C(=O)C[C@](O)(C(=O)NS(=O)(=O)N(C)C)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:10| Show InChI InChI=1S/C36H47ClN4O7S/c1-39(2)49(45,46)38-34(43)36(44)20-33(42)40(3)17-6-5-9-31(47-4)28-13-10-25(28)21-41-22-35(23-48-32-15-11-26(36)19-30(32)41)16-7-8-24-18-27(37)12-14-29(24)35/h5,9,11-12,14-15,18-19,25,28,31,44H,6-8,10,13,16-17,20-23H2,1-4H3,(H,38,43)/b9-5+/t25-,28+,31-,35-,36+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514219

(CHEMBL4438074 | US11274105, Example 181)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OCCN1CC(F)C1)\C=C\CCN(C)C(=O)C[C@](O)(C(=O)NS(=O)(=O)N(C)C)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:17| Show InChI InChI=1S/C40H53ClFN5O7S/c1-44(2)55(51,52)43-38(49)40(50)21-37(48)45(3)16-5-4-8-35(53-18-17-46-23-31(42)24-46)32-12-9-28(32)22-47-25-39(26-54-36-14-10-29(40)20-34(36)47)15-6-7-27-19-30(41)11-13-33(27)39/h4,8,10-11,13-14,19-20,28,31-32,35,50H,5-7,9,12,15-18,21-26H2,1-3H3,(H,43,49)/b8-4+/t28-,32+,35-,39-,40+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514204
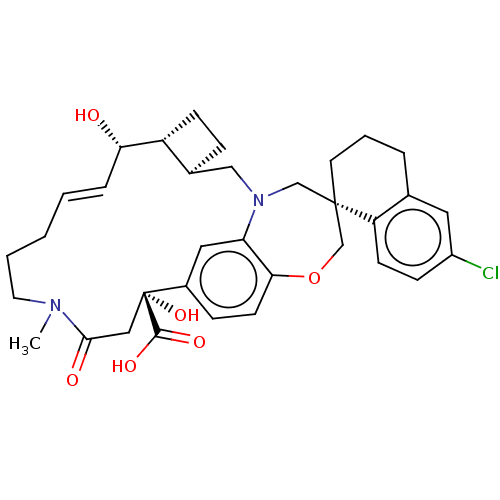
(CHEMBL4437832)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](O)\C=C\CCCN(C)C(=O)C[C@](O)(C(O)=O)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:9| Show InChI InChI=1S/C34H41ClN2O6/c1-36-15-4-2-3-7-29(38)26-11-8-23(26)19-37-20-33(14-5-6-22-16-25(35)10-12-27(22)33)21-43-30-13-9-24(17-28(30)37)34(42,32(40)41)18-31(36)39/h3,7,9-10,12-13,16-17,23,26,29,38,42H,2,4-6,8,11,14-15,18-21H2,1H3,(H,40,41)/b7-3+/t23-,26+,29-,33-,34+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Glucocorticoid receptor
(Homo sapiens (Human)) | BDBM50448167
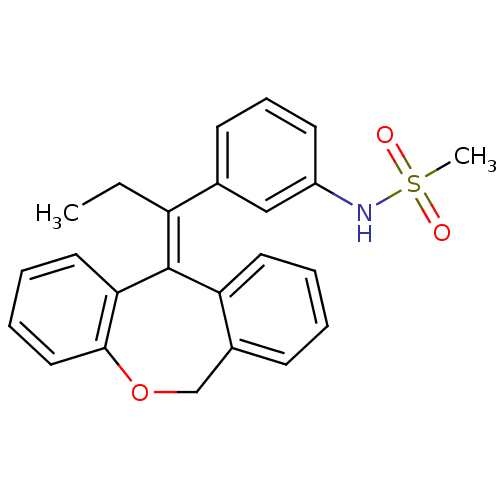
(CHEMBL3120318)Show SMILES CC\C(c1cccc(NS(C)(=O)=O)c1)=C1\c2ccccc2COc2ccccc12 Show InChI InChI=1S/C24H23NO3S/c1-3-20(17-10-8-11-19(15-17)25-29(2,26)27)24-21-12-5-4-9-18(21)16-28-23-14-7-6-13-22(23)24/h4-15,25H,3,16H2,1-2H3/b24-20+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.146 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-dexamethasone from human glucocorticoid receptor expressed in HEK293 cells |
J Med Chem 57: 849-60 (2014)
Article DOI: 10.1021/jm401616g
BindingDB Entry DOI: 10.7270/Q23X885F |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514217

(CHEMBL4452794 | US11274105, Example 196)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](OCCN1CCC(F)(F)CC1)\C=C\CCN(C)C(=O)C[C@](O)(C(=O)NS(=O)(=O)N(C)C)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:20| Show InChI InChI=1S/C42H56ClF2N5O7S/c1-47(2)58(54,55)46-39(52)42(53)25-38(51)48(3)18-5-4-8-36(56-22-21-49-19-16-41(44,45)17-20-49)33-12-9-30(33)26-50-27-40(28-57-37-14-10-31(42)24-35(37)50)15-6-7-29-23-32(43)11-13-34(29)40/h4,8,10-11,13-14,23-24,30,33,36,53H,5-7,9,12,15-22,25-28H2,1-3H3,(H,46,52)/b8-4+/t30-,33+,36-,40-,42+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Glucocorticoid receptor
(Homo sapiens (Human)) | BDBM50448165
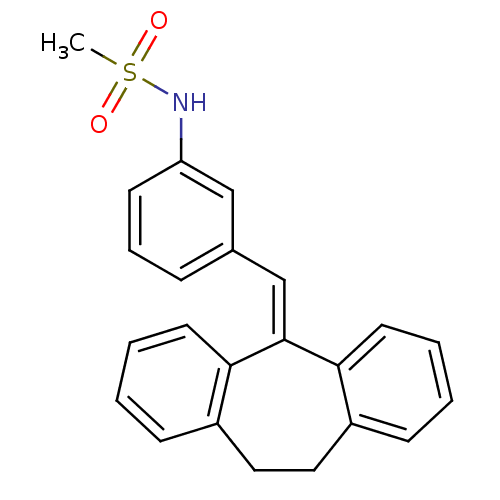
(CHEMBL3120320)Show SMILES [#6]S(=O)(=O)[#7]-c1cccc(\[#6]=[#6]-2/c3ccccc3-[#6]-[#6]-c3ccccc-23)c1 Show InChI InChI=1S/C23H21NO2S/c1-27(25,26)24-20-10-6-7-17(15-20)16-23-21-11-4-2-8-18(21)13-14-19-9-3-5-12-22(19)23/h2-12,15-16,24H,13-14H2,1H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.226 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-dexamethasone from human glucocorticoid receptor expressed in HEK293 cells |
J Med Chem 57: 849-60 (2014)
Article DOI: 10.1021/jm401616g
BindingDB Entry DOI: 10.7270/Q23X885F |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(GUINEA PIG) | BDBM50037135
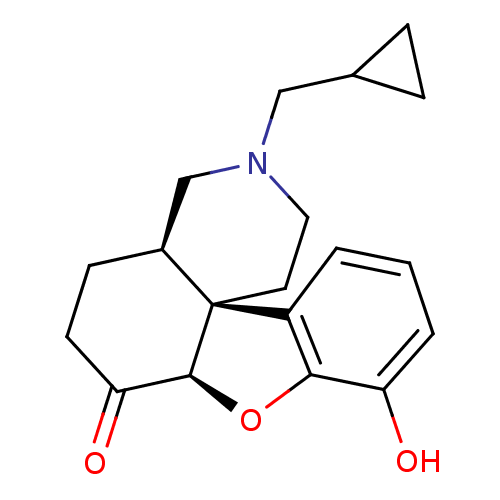
((4aR,7aR,12bS)-3-Cyclopropylmethyl-9-hydroxy-2,3,4...)Show SMILES Oc1cccc2c1O[C@H]1C(=O)CC[C@H]3CN(CC4CC4)CC[C@]213 Show InChI InChI=1S/C19H23NO3/c21-15-3-1-2-14-17(15)23-18-16(22)7-6-13-11-20(10-12-4-5-12)9-8-19(13,14)18/h1-3,12-13,18,21H,4-11H2/t13-,18-,19-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Taiwan University
Curated by ChEMBL
| Assay Description
In vivo binding affinity against mu opioid receptor was measured by using labeled ligand [3H]-Naloxone (0.5 nM) |
J Med Chem 37: 3121-7 (1994)
BindingDB Entry DOI: 10.7270/Q2PK0F6R |
More data for this
Ligand-Target Pair | |
Glucocorticoid receptor
(Homo sapiens (Human)) | BDBM50448166
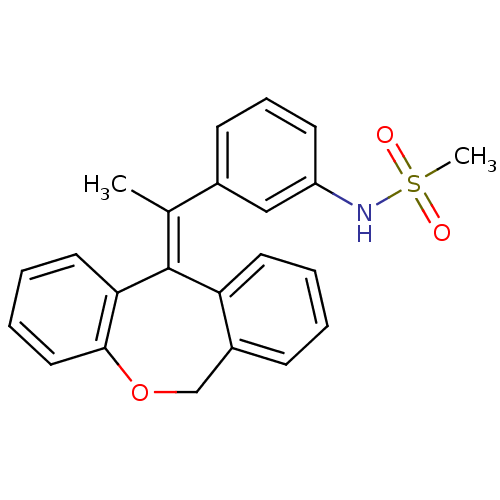
(CHEMBL3120319)Show SMILES C\C(c1cccc(NS(C)(=O)=O)c1)=C1\c2ccccc2COc2ccccc12 Show InChI InChI=1S/C23H21NO3S/c1-16(17-9-7-10-19(14-17)24-28(2,25)26)23-20-11-4-3-8-18(20)15-27-22-13-6-5-12-21(22)23/h3-14,24H,15H2,1-2H3/b23-16+ | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.257 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-dexamethasone from human glucocorticoid receptor expressed in HEK293 cells |
J Med Chem 57: 849-60 (2014)
Article DOI: 10.1021/jm401616g
BindingDB Entry DOI: 10.7270/Q23X885F |
More data for this
Ligand-Target Pair | |
Glucocorticoid receptor
(Homo sapiens (Human)) | BDBM50448168
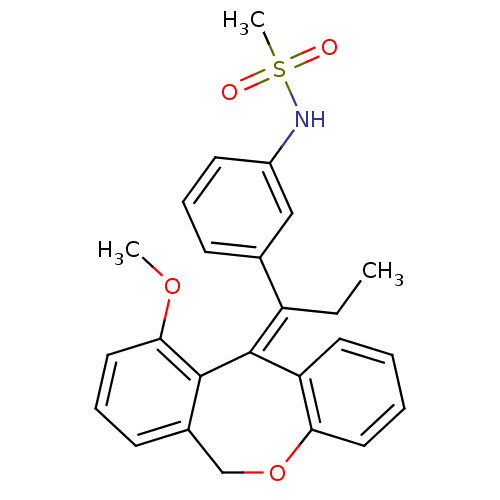
(CHEMBL3120317)Show SMILES CC\C(c1cccc(NS(C)(=O)=O)c1)=C1/c2ccccc2OCc2cccc(OC)c12 Show InChI InChI=1S/C25H25NO4S/c1-4-20(17-9-7-11-19(15-17)26-31(3,27)28)25-21-12-5-6-13-22(21)30-16-18-10-8-14-23(29-2)24(18)25/h5-15,26H,4,16H2,1-3H3/b25-20- | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.268 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-dexamethasone from human glucocorticoid receptor expressed in HEK293 cells |
J Med Chem 57: 849-60 (2014)
Article DOI: 10.1021/jm401616g
BindingDB Entry DOI: 10.7270/Q23X885F |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514208
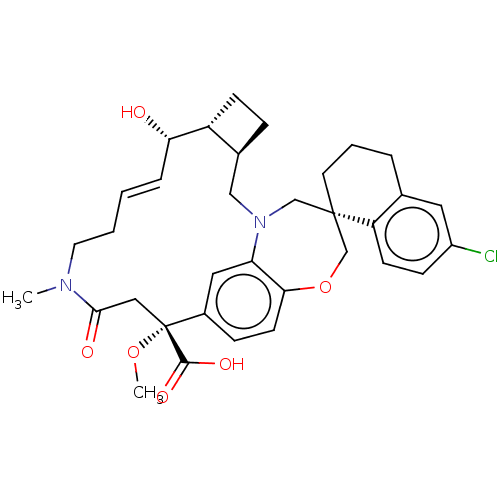
(CHEMBL4469850 | US11274105, Example 61)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](O)\C=C\CCN(C)C(=O)C[C@](OC)(C(O)=O)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:9| Show InChI InChI=1S/C34H41ClN2O6/c1-36-15-4-3-7-29(38)26-11-8-23(26)19-37-20-33(14-5-6-22-16-25(35)10-12-27(22)33)21-43-30-13-9-24(17-28(30)37)34(42-2,32(40)41)18-31(36)39/h3,7,9-10,12-13,16-17,23,26,29,38H,4-6,8,11,14-15,18-21H2,1-2H3,(H,40,41)/b7-3+/t23-,26+,29-,33-,34+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Mineralocorticoid receptor
(Homo sapiens (Human)) | BDBM19190
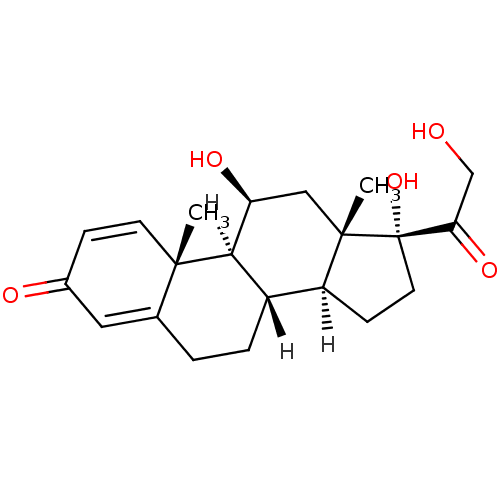
((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...)Show SMILES [H][C@@]12CC[C@](O)(C(=O)CO)[C@@]1(C)C[C@H](O)[C@@]1([H])[C@@]2([H])CCC2=CC(=O)C=C[C@]12C |r,c:27,t:23| Show InChI InChI=1S/C21H28O5/c1-19-7-5-13(23)9-12(19)3-4-14-15-6-8-21(26,17(25)11-22)20(15,2)10-16(24)18(14)19/h5,7,9,14-16,18,22,24,26H,3-4,6,8,10-11H2,1-2H3/t14-,15-,16-,18+,19-,20-,21-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-aldosterone from human mineralocorticoid receptor expressed in HEK293 cells |
J Med Chem 57: 849-60 (2014)
Article DOI: 10.1021/jm401616g
BindingDB Entry DOI: 10.7270/Q23X885F |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50258880
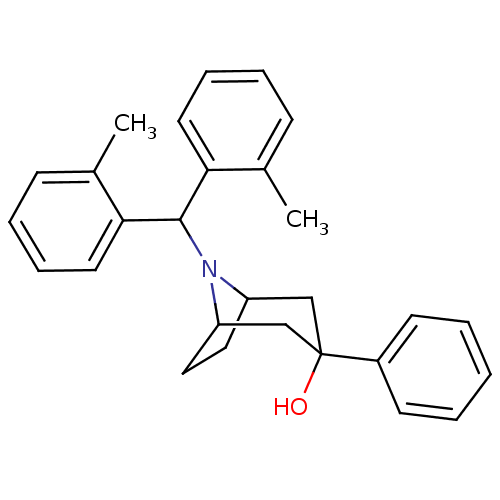
(8-(dio-tolylmethyl)-3-phenyl-8-azabicyclo[3.2.1]oc...)Show SMILES Cc1ccccc1C(N1C2CCC1CC(O)(C2)c1ccccc1)c1ccccc1C |THB:15:14:8:10.11| Show InChI InChI=1S/C28H31NO/c1-20-10-6-8-14-25(20)27(26-15-9-7-11-21(26)2)29-23-16-17-24(29)19-28(30,18-23)22-12-4-3-5-13-22/h3-15,23-24,27,30H,16-19H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Displacement of [125I][Tyr14]nociceptin from human cloned NOP receptor expressed in CHO cells |
Bioorg Med Chem Lett 19: 2519-23 (2009)
Article DOI: 10.1016/j.bmcl.2009.03.031
BindingDB Entry DOI: 10.7270/Q20V8CPS |
More data for this
Ligand-Target Pair | |
Mineralocorticoid receptor
(Homo sapiens (Human)) | BDBM50448166

(CHEMBL3120319)Show SMILES C\C(c1cccc(NS(C)(=O)=O)c1)=C1\c2ccccc2COc2ccccc12 Show InChI InChI=1S/C23H21NO3S/c1-16(17-9-7-10-19(14-17)24-28(2,25)26)23-20-11-4-3-8-18(20)15-27-22-13-6-5-12-21(22)23/h3-14,24H,15H2,1-2H3/b23-16+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.342 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-aldosterone from human mineralocorticoid receptor expressed in HEK293 cells |
J Med Chem 57: 849-60 (2014)
Article DOI: 10.1021/jm401616g
BindingDB Entry DOI: 10.7270/Q23X885F |
More data for this
Ligand-Target Pair | |
Mineralocorticoid receptor
(Homo sapiens (Human)) | BDBM18207

((1R,2S,10S,11S,13R,14R,15S,17S)-1-fluoro-14,17-dih...)Show SMILES [H][C@@]12C[C@@H](C)[C@](O)(C(=O)CO)[C@@]1(C)C[C@H](O)[C@@]1(F)[C@@]2([H])CCC2=CC(=O)C=C[C@]12C |c:28,t:24| Show InChI InChI=1S/C22H29FO5/c1-12-8-16-15-5-4-13-9-14(25)6-7-19(13,2)21(15,23)17(26)10-20(16,3)22(12,28)18(27)11-24/h6-7,9,12,15-17,24,26,28H,4-5,8,10-11H2,1-3H3/t12-,15+,16+,17+,19+,20+,21+,22+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-aldosterone from human mineralocorticoid receptor expressed in HEK293 cells |
J Med Chem 57: 849-60 (2014)
Article DOI: 10.1021/jm401616g
BindingDB Entry DOI: 10.7270/Q23X885F |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514207

(CHEMBL4562159)Show SMILES [H][C@@]12CC[C@@]1([H])[C@H](CCCCN(C)C(=O)C[C@](O)(C(=O)NS(=O)(=O)N(C)C)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1)OC |r| Show InChI InChI=1S/C36H49ClN4O7S/c1-39(2)49(45,46)38-34(43)36(44)20-33(42)40(3)17-6-5-9-31(47-4)28-13-10-25(28)21-41-22-35(23-48-32-15-11-26(36)19-30(32)41)16-7-8-24-18-27(37)12-14-29(24)35/h11-12,14-15,18-19,25,28,31,44H,5-10,13,16-17,20-23H2,1-4H3,(H,38,43)/t25-,28+,31-,35-,36+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50373621
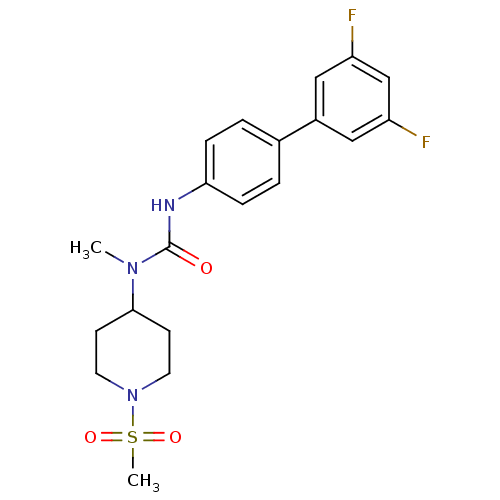
(CHEMBL403414)Show SMILES CN(C1CCN(CC1)S(C)(=O)=O)C(=O)Nc1ccc(cc1)-c1cc(F)cc(F)c1 Show InChI InChI=1S/C20H23F2N3O3S/c1-24(19-7-9-25(10-8-19)29(2,27)28)20(26)23-18-5-3-14(4-6-18)15-11-16(21)13-17(22)12-15/h3-6,11-13,19H,7-10H2,1-2H3,(H,23,26) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Displacement of [125]PYY from human chimeric NPY Y5 receptor expressed in CHOK1 cells |
Bioorg Med Chem Lett 18: 1146-50 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.132
BindingDB Entry DOI: 10.7270/Q27D2W1W |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50202412
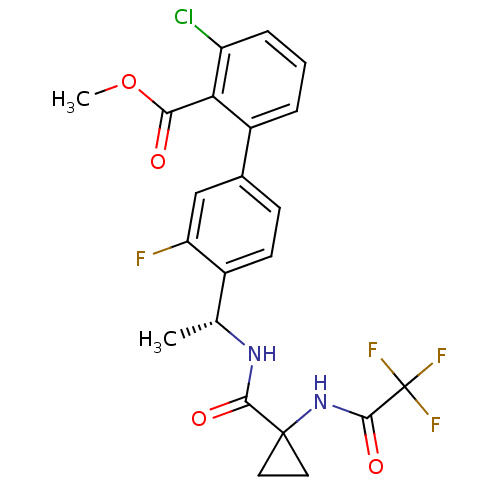
(3-Chloro-3'-fluoro-4'-((R)-1-{[1-(2,2,2-trifluoro-...)Show SMILES COC(=O)c1c(Cl)cccc1-c1ccc([C@@H](C)NC(=O)C2(CC2)NC(=O)C(F)(F)F)c(F)c1 |r| Show InChI InChI=1S/C22H19ClF4N2O4/c1-11(28-19(31)21(8-9-21)29-20(32)22(25,26)27)13-7-6-12(10-16(13)24)14-4-3-5-15(23)17(14)18(30)33-2/h3-7,10-11H,8-9H2,1-2H3,(H,28,31)(H,29,32)/t11-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Inhibition of human bradykinin B1 receptor |
Bioorg Med Chem Lett 18: 5027-31 (2008)
Article DOI: 10.1016/j.bmcl.2008.08.014
BindingDB Entry DOI: 10.7270/Q2NG4QDX |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514197
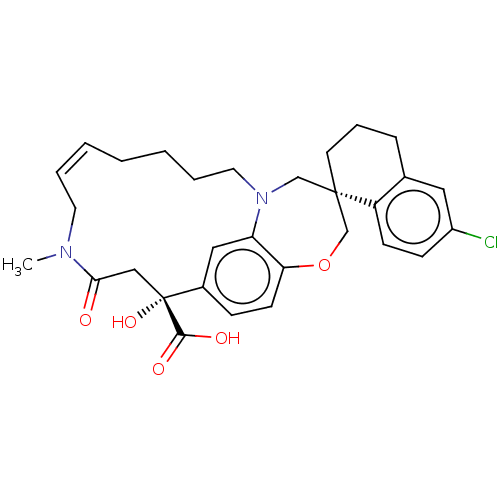
(CHEMBL4561691 | US11274105, Example 63)Show SMILES CN1C\C=C\CCCCN2C[C@@]3(CCCc4cc(Cl)ccc34)COc3ccc(cc23)[C@](O)(CC1=O)C(O)=O |r,t:3| Show InChI InChI=1S/C30H35ClN2O5/c1-32-14-5-3-2-4-6-15-33-19-29(13-7-8-21-16-23(31)10-11-24(21)29)20-38-26-12-9-22(17-25(26)33)30(37,28(35)36)18-27(32)34/h3,5,9-12,16-17,37H,2,4,6-8,13-15,18-20H2,1H3,(H,35,36)/b5-3+/t29-,30+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mu-type opioid receptor
(GUINEA PIG) | BDBM50037134

((+)-trans-3-Cyclopropylmethyl-2,3,4,4aalpha,5,6,7,...)Show SMILES Oc1cccc2c1O[C@H]1CCC[C@H]3CN(CC4CC4)CC[C@]213 Show InChI InChI=1S/C19H25NO2/c21-16-5-2-4-15-18(16)22-17-6-1-3-14-12-20(11-13-7-8-13)10-9-19(14,15)17/h2,4-5,13-14,17,21H,1,3,6-12H2/t14-,17-,19-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.570 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Taiwan University
Curated by ChEMBL
| Assay Description
In vivo binding affinity against mu opioid receptor was measured by using labeled ligand [3H]-Naloxone (0.5 nM) |
J Med Chem 37: 3121-7 (1994)
BindingDB Entry DOI: 10.7270/Q2PK0F6R |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50127438
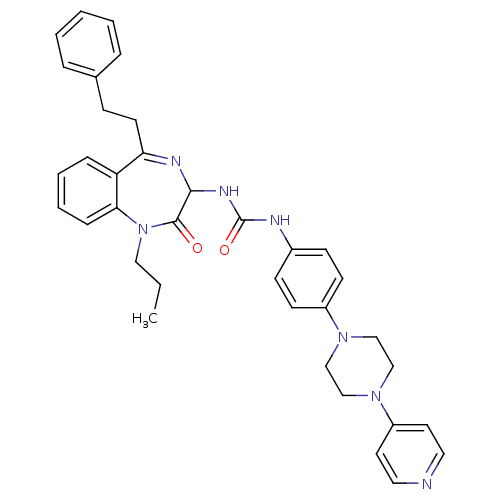
(1-(2-Oxo-5-phenethyl-1-propyl-2,3-dihydro-1H-benzo...)Show SMILES CCCN1c2ccccc2C(CCc2ccccc2)=NC(NC(=O)Nc2ccc(cc2)N2CCN(CC2)c2ccncc2)C1=O |c:20| Show InChI InChI=1S/C36H39N7O2/c1-2-22-43-33-11-7-6-10-31(33)32(17-12-27-8-4-3-5-9-27)39-34(35(43)44)40-36(45)38-28-13-15-29(16-14-28)41-23-25-42(26-24-41)30-18-20-37-21-19-30/h3-11,13-16,18-21,34H,2,12,17,22-26H2,1H3,(H2,38,40,45) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Inhibition of human bradykinin B1 receptor |
Bioorg Med Chem Lett 18: 5027-31 (2008)
Article DOI: 10.1016/j.bmcl.2008.08.014
BindingDB Entry DOI: 10.7270/Q2NG4QDX |
More data for this
Ligand-Target Pair | |
Glucocorticoid receptor
(Homo sapiens (Human)) | BDBM18207

((1R,2S,10S,11S,13R,14R,15S,17S)-1-fluoro-14,17-dih...)Show SMILES [H][C@@]12C[C@@H](C)[C@](O)(C(=O)CO)[C@@]1(C)C[C@H](O)[C@@]1(F)[C@@]2([H])CCC2=CC(=O)C=C[C@]12C |c:28,t:24| Show InChI InChI=1S/C22H29FO5/c1-12-8-16-15-5-4-13-9-14(25)6-7-19(13,2)21(15,23)17(26)10-20(16,3)22(12,28)18(27)11-24/h6-7,9,12,15-17,24,26,28H,4-5,8,10-11H2,1-3H3/t12-,15+,16+,17+,19+,20+,21+,22+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 0.690 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-dexamethasone from human glucocorticoid receptor expressed in HEK293 cells |
J Med Chem 57: 849-60 (2014)
Article DOI: 10.1021/jm401616g
BindingDB Entry DOI: 10.7270/Q23X885F |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
cGMP-specific 3',5'-cyclic phosphodiesterase
(Homo sapiens (Human)) | BDBM50088373
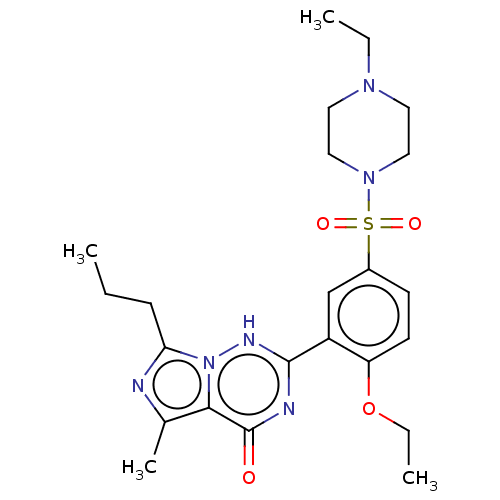
(CHEBI:46295 | Vardenafil | cid_110634)Show SMILES CCCc1nc(C)c2n1[nH]c(nc2=O)-c1cc(ccc1OCC)S(=O)(=O)N1CCN(CC)CC1 Show InChI InChI=1S/C23H32N6O4S/c1-5-8-20-24-16(4)21-23(30)25-22(26-29(20)21)18-15-17(9-10-19(18)33-7-3)34(31,32)28-13-11-27(6-2)12-14-28/h9-10,15H,5-8,11-14H2,1-4H3,(H,25,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| DrugBank
PDB
Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shandong University
Curated by ChEMBL
| Assay Description
Inhibition of PDE5 (unknown origin) |
Eur J Med Chem 158: 767-780 (2018)
Article DOI: 10.1016/j.ejmech.2018.09.028
BindingDB Entry DOI: 10.7270/Q2JS9T4N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50373623
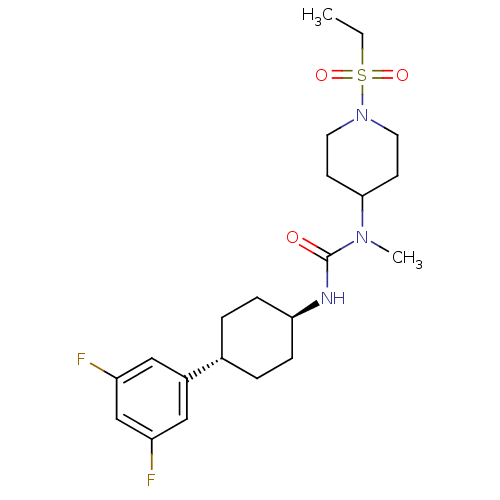
(CHEMBL404461)Show SMILES CCS(=O)(=O)N1CCC(CC1)N(C)C(=O)N[C@H]1CC[C@@H](CC1)c1cc(F)cc(F)c1 |wU:16.16,wD:19.23,(10.08,-2.96,;10.08,-1.42,;8.75,-.65,;7.98,-1.98,;9.53,.68,;7.42,.12,;6.08,-.64,;4.76,.13,;4.77,1.66,;6.09,2.44,;7.42,1.67,;3.43,2.43,;3.43,3.97,;2.1,1.66,;2.1,.12,;.77,2.43,;-.57,1.66,;-.56,.13,;-1.9,-.64,;-3.24,.13,;-3.23,1.68,;-1.9,2.44,;-4.57,-.63,;-5.91,.15,;-7.24,-.61,;-8.57,.16,;-7.25,-2.16,;-5.91,-2.93,;-5.91,-4.47,;-4.58,-2.16,)| Show InChI InChI=1S/C21H31F2N3O3S/c1-3-30(28,29)26-10-8-20(9-11-26)25(2)21(27)24-19-6-4-15(5-7-19)16-12-17(22)14-18(23)13-16/h12-15,19-20H,3-11H2,1-2H3,(H,24,27)/t15-,19- | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Displacement of [125]PYY from human chimeric NPY Y5 receptor expressed in CHOK1 cells |
Bioorg Med Chem Lett 18: 1146-50 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.132
BindingDB Entry DOI: 10.7270/Q27D2W1W |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepatitis C virus (HCV genotype 1a, isolate H)) | BDBM27048
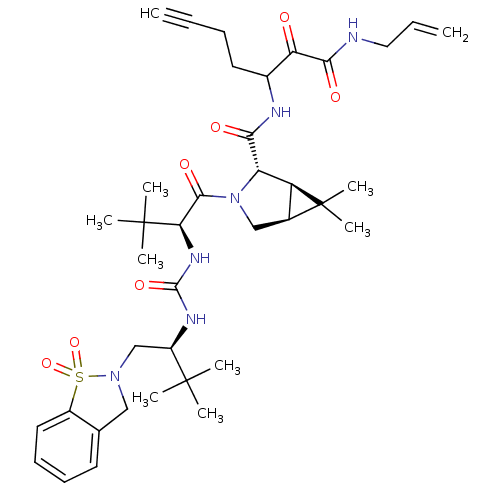
(3-{[(1R,2S,5S)-3-[(2S)-2-({[(2S)-1-(1,1-dioxo-2,3-...)Show SMILES CC(C)(C)[C@@H](CN1Cc2ccccc2S1(=O)=O)NC(=O)N[C@H](C(=O)N1C[C@H]2[C@@H]([C@H]1C(=O)NC(CCC#C)C(=O)C(=O)NCC=C)C2(C)C)C(C)(C)C |r| Show InChI InChI=1S/C38H54N6O7S/c1-11-13-17-25(30(45)33(47)39-19-12-2)40-32(46)29-28-24(38(28,9)10)21-44(29)34(48)31(37(6,7)8)42-35(49)41-27(36(3,4)5)22-43-20-23-16-14-15-18-26(23)52(43,50)51/h1,12,14-16,18,24-25,27-29,31H,2,13,17,19-22H2,3-10H3,(H,39,47)(H,40,46)(H2,41,42,49)/t24-,25?,27+,28-,29-,31+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.900 | -52.5 | n/a | n/a | n/a | n/a | n/a | 6.5 | 30 |
Schering-Plough Research Institute
| Assay Description
Proteolytic cleavage of the ester linkage between the Nva (L-norvaline) and the chromophore (PAP) was monitored for change in absorbance at 370 nm. I... |
Bioorg Med Chem Lett 19: 1105-9 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.111
BindingDB Entry DOI: 10.7270/Q22B8WBZ |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(RAT) | BDBM50127438

(1-(2-Oxo-5-phenethyl-1-propyl-2,3-dihydro-1H-benzo...)Show SMILES CCCN1c2ccccc2C(CCc2ccccc2)=NC(NC(=O)Nc2ccc(cc2)N2CCN(CC2)c2ccncc2)C1=O |c:20| Show InChI InChI=1S/C36H39N7O2/c1-2-22-43-33-11-7-6-10-31(33)32(17-12-27-8-4-3-5-9-27)39-34(35(43)44)40-36(45)38-28-13-15-29(16-14-28)41-23-25-42(26-24-41)30-18-20-37-21-19-30/h3-11,13-16,18-21,34H,2,12,17,22-26H2,1H3,(H2,38,40,45) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.920 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at rat bradykinin B1 receptor |
Bioorg Med Chem Lett 18: 5027-31 (2008)
Article DOI: 10.1016/j.bmcl.2008.08.014
BindingDB Entry DOI: 10.7270/Q2NG4QDX |
More data for this
Ligand-Target Pair | |
Mineralocorticoid receptor
(Homo sapiens (Human)) | BDBM50448167

(CHEMBL3120318)Show SMILES CC\C(c1cccc(NS(C)(=O)=O)c1)=C1\c2ccccc2COc2ccccc12 Show InChI InChI=1S/C24H23NO3S/c1-3-20(17-10-8-11-19(15-17)25-29(2,26)27)24-21-12-5-4-9-18(21)16-28-23-14-7-6-13-22(23)24/h4-15,25H,3,16H2,1-2H3/b24-20+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.967 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-aldosterone from human mineralocorticoid receptor expressed in HEK293 cells |
J Med Chem 57: 849-60 (2014)
Article DOI: 10.1021/jm401616g
BindingDB Entry DOI: 10.7270/Q23X885F |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(Homo sapiens (Human)) | BDBM50370577
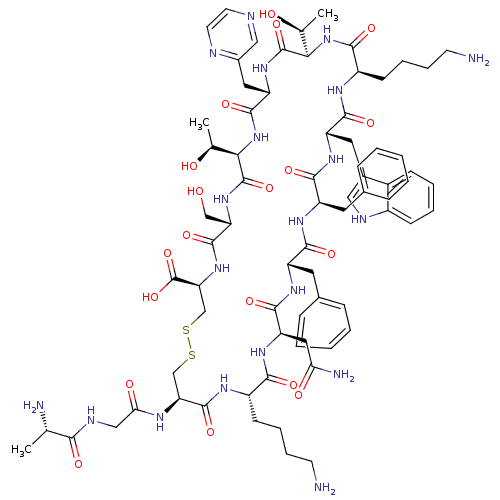
(CHEMBL1791312)Show SMILES C[C@H](O)[C@H]1NC(=O)[C@H](Cc2cnccn2)NC(=O)[C@H](NC(=O)[C@@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CSSC[C@H](NC(=O)[C@H](CO)NC1=O)C(O)=O)NC(=O)CNC(=O)[C@H](C)N)[C@H](C)O Show InChI InChI=1S/C74H102N20O19S2/c1-39(77)62(100)82-35-59(99)83-56-37-114-115-38-57(74(112)113)92-70(108)55(36-95)91-73(111)61(41(3)97)94-69(107)53(31-45-34-79-26-27-80-45)90-72(110)60(40(2)96)93-64(102)49(23-13-15-25-76)84-67(105)52(30-44-33-81-47-21-11-10-20-46(44)47)88-66(104)51(29-43-18-8-5-9-19-43)86-65(103)50(28-42-16-6-4-7-17-42)87-68(106)54(32-58(78)98)89-63(101)48(85-71(56)109)22-12-14-24-75/h4-11,16-21,26-27,33-34,39-41,48-57,60-61,81,95-97H,12-15,22-25,28-32,35-38,75-77H2,1-3H3,(H2,78,98)(H,82,100)(H,83,99)(H,84,105)(H,85,109)(H,86,103)(H,87,106)(H,88,104)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t39-,40-,41-,48-,49+,50-,51+,52-,53-,54+,55-,56-,57-,60+,61+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by ChEMBL
| Assay Description
Inhibition of human somatostatin receptor type 2 |
J Med Chem 48: 4025-30 (2005)
Article DOI: 10.1021/jm058184l
BindingDB Entry DOI: 10.7270/Q2736RP5 |
More data for this
Ligand-Target Pair | |
Somatostatin receptor type 2
(Homo sapiens (Human)) | BDBM50370578
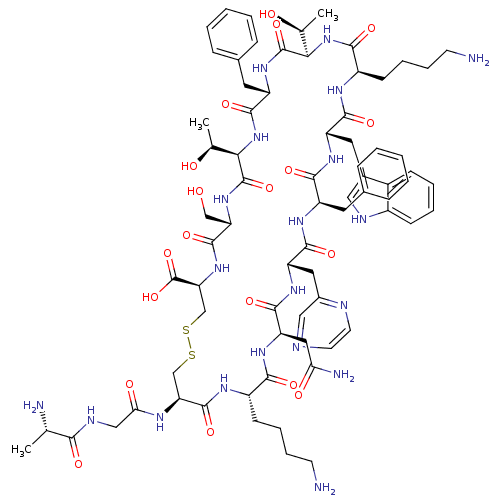
(CHEMBL1791304)Show SMILES C[C@H](O)[C@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](NC(=O)[C@@H](CCCCN)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2cnccn2)NC(=O)[C@@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CSSC[C@H](NC(=O)[C@H](CO)NC1=O)C(O)=O)NC(=O)CNC(=O)[C@H](C)N)[C@H](C)O Show InChI InChI=1S/C74H102N20O19S2/c1-39(77)62(100)82-35-59(99)83-56-37-114-115-38-57(74(112)113)92-70(108)55(36-95)91-73(111)61(41(3)97)94-69(107)51(29-43-18-8-5-9-19-43)90-72(110)60(40(2)96)93-64(102)49(23-13-15-25-76)84-66(104)52(30-44-33-81-47-21-11-10-20-46(44)47)87-65(103)50(28-42-16-6-4-7-17-42)86-67(105)53(31-45-34-79-26-27-80-45)88-68(106)54(32-58(78)98)89-63(101)48(85-71(56)109)22-12-14-24-75/h4-11,16-21,26-27,33-34,39-41,48-57,60-61,81,95-97H,12-15,22-25,28-32,35-38,75-77H2,1-3H3,(H2,78,98)(H,82,100)(H,83,99)(H,84,104)(H,85,109)(H,86,105)(H,87,103)(H,88,106)(H,89,101)(H,90,110)(H,91,111)(H,92,108)(H,93,102)(H,94,107)(H,112,113)/t39-,40-,41-,48-,49+,50+,51-,52-,53-,54+,55-,56-,57-,60+,61+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Pennsylvania
Curated by ChEMBL
| Assay Description
Inhibition of human somatostatin receptor type 2 |
J Med Chem 48: 4025-30 (2005)
Article DOI: 10.1021/jm058184l
BindingDB Entry DOI: 10.7270/Q2736RP5 |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514209

(CHEMBL4460664)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](O)CCCCN(C)C(=O)C[C@@](O)(C(O)=O)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r| Show InChI InChI=1S/C33H41ClN2O6/c1-35-14-3-2-6-28(37)25-10-7-22(25)18-36-19-32(13-4-5-21-15-24(34)9-11-26(21)32)20-42-29-12-8-23(16-27(29)36)33(41,31(39)40)17-30(35)38/h8-9,11-12,15-16,22,25,28,37,41H,2-7,10,13-14,17-20H2,1H3,(H,39,40)/t22-,25+,28-,32-,33-/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50514205

(CHEMBL4452403)Show SMILES [H][C@@]12CC[C@@]1([H])[C@@H](O)\C=C\CN(C)C(=O)C[C@](O)(C(O)=O)c1ccc3OC[C@]4(CCCc5cc(Cl)ccc45)CN(C2)c3c1 |r,t:9| Show InChI InChI=1S/C32H37ClN2O6/c1-34-13-3-5-27(36)24-9-6-21(24)17-35-18-31(12-2-4-20-14-23(33)8-10-25(20)31)19-41-28-11-7-22(15-26(28)35)32(40,30(38)39)16-29(34)37/h3,5,7-8,10-11,14-15,21,24,27,36,40H,2,4,6,9,12-13,16-19H2,1H3,(H,38,39)/b5-3+/t21-,24+,27-,31-,32+/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant C-terminal His6x-tagged human Mcl-1 (171 to 327 residues) interaction with biotinylated human Bim (51 to 76 residues) incub... |
J Med Chem 62: 10258-10271 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01310
BindingDB Entry DOI: 10.7270/Q2TQ64WW |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(GUINEA PIG) | BDBM50000788
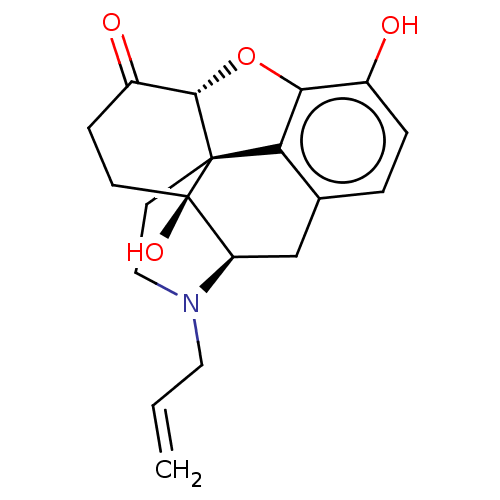
((morphine)4-methyl-(1S,5R,13R,14S,17R)-12-oxa-4-az...)Show SMILES Oc1ccc2C[C@H]3N(CC=C)CC[C@@]45[C@@H](Oc1c24)C(=O)CC[C@@]35O |r| Show InChI InChI=1S/C19H21NO4/c1-2-8-20-9-7-18-15-11-3-4-12(21)16(15)24-17(18)13(22)5-6-19(18,23)14(20)10-11/h2-4,14,17,21,23H,1,5-10H2/t14-,17+,18+,19-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Taiwan University
Curated by ChEMBL
| Assay Description
In vivo binding affinity against mu opioid receptor was measured by using labeled ligand [3H]-Naloxone (0.5 nM) |
J Med Chem 37: 3121-7 (1994)
BindingDB Entry DOI: 10.7270/Q2PK0F6R |
More data for this
Ligand-Target Pair | |
Glucocorticoid receptor
(Homo sapiens (Human)) | BDBM19190

((1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-...)Show SMILES [H][C@@]12CC[C@](O)(C(=O)CO)[C@@]1(C)C[C@H](O)[C@@]1([H])[C@@]2([H])CCC2=CC(=O)C=C[C@]12C |r,c:27,t:23| Show InChI InChI=1S/C21H28O5/c1-19-7-5-13(23)9-12(19)3-4-14-15-6-8-21(26,17(25)11-22)20(15,2)10-16(24)18(14)19/h5,7,9,14-16,18,22,24,26H,3-4,6,8,10-11H2,1-2H3/t14-,15-,16-,18+,19-,20-,21-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-dexamethasone from human glucocorticoid receptor expressed in HEK293 cells |
J Med Chem 57: 849-60 (2014)
Article DOI: 10.1021/jm401616g
BindingDB Entry DOI: 10.7270/Q23X885F |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50373639
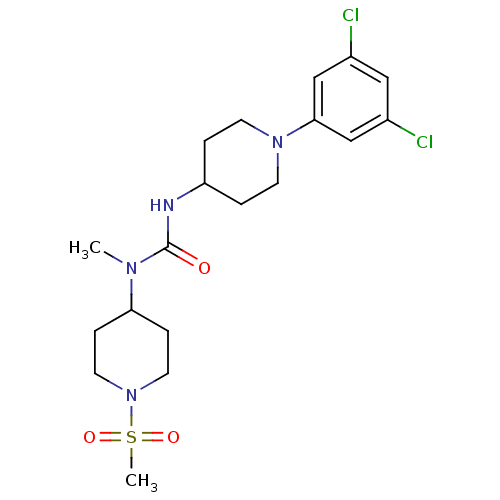
(CHEMBL428423)Show SMILES CN(C1CCN(CC1)S(C)(=O)=O)C(=O)NC1CCN(CC1)c1cc(Cl)cc(Cl)c1 Show InChI InChI=1S/C19H28Cl2N4O3S/c1-23(17-5-9-25(10-6-17)29(2,27)28)19(26)22-16-3-7-24(8-4-16)18-12-14(20)11-15(21)13-18/h11-13,16-17H,3-10H2,1-2H3,(H,22,26) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Displacement of [125]PYY from human chimeric NPY Y5 receptor expressed in CHOK1 cells |
Bioorg Med Chem Lett 18: 1146-50 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.132
BindingDB Entry DOI: 10.7270/Q27D2W1W |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50373624
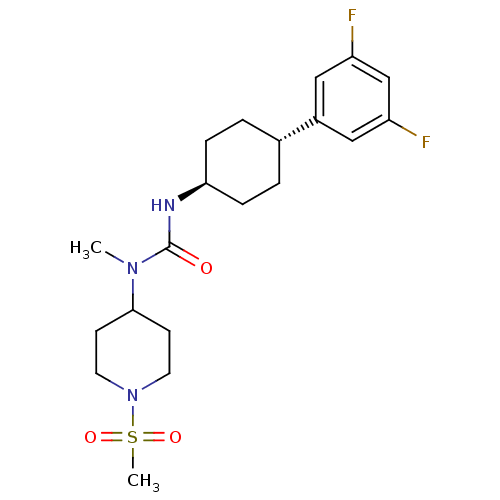
(CHEMBL255353)Show SMILES CN(C1CCN(CC1)S(C)(=O)=O)C(=O)N[C@H]1CC[C@@H](CC1)c1cc(F)cc(F)c1 |wU:15.15,wD:18.22,(28.66,-44.4,;28.66,-45.94,;29.99,-46.71,;29.98,-48.24,;31.3,-49.01,;32.64,-48.25,;32.65,-46.7,;31.31,-45.93,;33.97,-49.02,;35.3,-49.79,;33.2,-50.35,;34.75,-47.69,;27.32,-46.71,;27.32,-48.25,;25.99,-45.94,;24.66,-46.71,;24.66,-48.24,;23.32,-49.01,;21.99,-48.24,;21.99,-46.69,;23.32,-45.93,;20.65,-49,;19.32,-48.22,;17.98,-48.98,;16.65,-48.21,;17.98,-50.53,;19.32,-51.3,;19.32,-52.84,;20.65,-50.53,)| Show InChI InChI=1S/C20H29F2N3O3S/c1-24(19-7-9-25(10-8-19)29(2,27)28)20(26)23-18-5-3-14(4-6-18)15-11-16(21)13-17(22)12-15/h11-14,18-19H,3-10H2,1-2H3,(H,23,26)/t14-,18- | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Displacement of [125]PYY from human chimeric NPY Y5 receptor expressed in CHOK1 cells |
Bioorg Med Chem Lett 18: 1146-50 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.132
BindingDB Entry DOI: 10.7270/Q27D2W1W |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50037134

((+)-trans-3-Cyclopropylmethyl-2,3,4,4aalpha,5,6,7,...)Show SMILES Oc1cccc2c1O[C@H]1CCC[C@H]3CN(CC4CC4)CC[C@]213 Show InChI InChI=1S/C19H25NO2/c21-16-5-2-4-15-18(16)22-17-6-1-3-14-12-20(11-13-7-8-13)10-9-19(14,15)17/h2,4-5,13-14,17,21H,1,3,6-12H2/t14-,17-,19-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Taiwan University
Curated by ChEMBL
| Assay Description
In vivo binding affinity against kappa opioid receptor was measured by using labeled ligand [3H]-ethylketocyclazocine (1 nM) with 500 nM DADLE and 20... |
J Med Chem 37: 3121-7 (1994)
BindingDB Entry DOI: 10.7270/Q2PK0F6R |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(GUINEA PIG) | BDBM50037131

((4aR,7S,7aR,12bS)-3-Cyclopropylmethyl-2,3,4,4a,5,6...)Show SMILES O[C@H]1CC[C@H]2CN(CC3CC3)CC[C@]22[C@H]1Oc1c2cccc1O Show InChI InChI=1S/C19H25NO3/c21-15-3-1-2-14-17(15)23-18-16(22)7-6-13-11-20(10-12-4-5-12)9-8-19(13,14)18/h1-3,12-13,16,18,21-22H,4-11H2/t13-,16-,18-,19-/m0/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Taiwan University
Curated by ChEMBL
| Assay Description
In vivo binding affinity against mu opioid receptor was measured by using labeled ligand [3H]-Naloxone (0.5 nM) |
J Med Chem 37: 3121-7 (1994)
BindingDB Entry DOI: 10.7270/Q2PK0F6R |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50373625
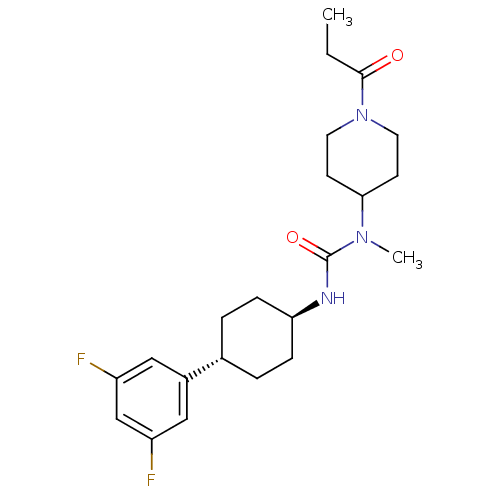
(CHEMBL403918)Show SMILES CCC(=O)N1CCC(CC1)N(C)C(=O)N[C@H]1CC[C@@H](CC1)c1cc(F)cc(F)c1 |wU:15.15,wD:18.22,(36.07,-39.93,;34.73,-39.16,;33.4,-39.92,;33.39,-41.46,;32.07,-39.15,;30.73,-39.91,;29.4,-39.14,;29.42,-37.61,;30.74,-36.83,;32.07,-37.6,;28.08,-36.84,;28.08,-35.3,;26.75,-37.61,;26.75,-39.15,;25.41,-36.84,;24.08,-37.61,;24.08,-39.14,;22.74,-39.91,;21.41,-39.14,;21.42,-37.59,;22.75,-36.83,;20.07,-39.9,;20.07,-41.43,;18.74,-42.2,;18.74,-43.74,;17.4,-41.43,;17.41,-39.88,;16.08,-39.11,;18.74,-39.12,)| Show InChI InChI=1S/C22H31F2N3O2/c1-3-21(28)27-10-8-20(9-11-27)26(2)22(29)25-19-6-4-15(5-7-19)16-12-17(23)14-18(24)13-16/h12-15,19-20H,3-11H2,1-2H3,(H,25,29)/t15-,19- | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Displacement of [125]PYY from human chimeric NPY Y5 receptor expressed in CHOK1 cells |
Bioorg Med Chem Lett 18: 1146-50 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.132
BindingDB Entry DOI: 10.7270/Q27D2W1W |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data