 Found 175 hits with Last Name = 'hai' and Initial = 'tt'
Found 175 hits with Last Name = 'hai' and Initial = 'tt' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Adenylate kinase 2, mitochondrial
(Rattus norvegicus) | BDBM50367049
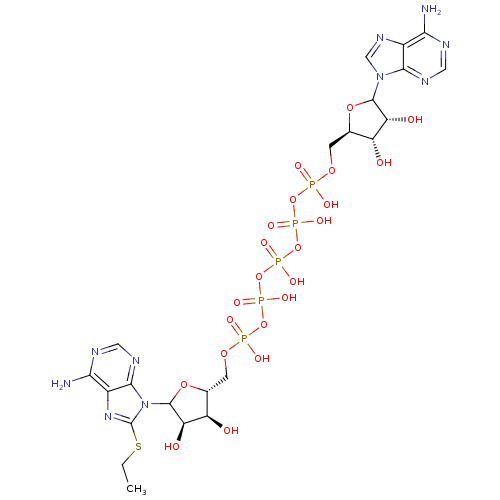
(CHEMBL604399)Show SMILES CCSc1nc2c(N)ncnc2n1C1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OC[C@H]2OC([C@H](O)[C@@H]2O)n2cnc3c(N)ncnc23)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C22H33N10O22P5S/c1-2-60-22-30-11-17(24)26-6-28-19(11)32(22)21-15(36)13(34)9(50-21)4-48-56(39,40)52-58(43,44)54-59(45,46)53-57(41,42)51-55(37,38)47-3-8-12(33)14(35)20(49-8)31-7-29-10-16(23)25-5-27-18(10)31/h5-9,12-15,20-21,33-36H,2-4H2,1H3,(H,37,38)(H,39,40)(H,41,42)(H,43,44)(H,45,46)(H2,23,25,27)(H2,24,26,28)/t8-,9-,12-,13-,14-,15-,20?,21?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against rat adenylate kinase II was determined in the presence of AMP, competitive inhibition |
J Med Chem 25: 1179-84 (1983)
BindingDB Entry DOI: 10.7270/Q25D8SD7 |
More data for this
Ligand-Target Pair | |
Adenylate kinase 2, mitochondrial
(Rattus norvegicus) | BDBM50367049

(CHEMBL604399)Show SMILES CCSc1nc2c(N)ncnc2n1C1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OC[C@H]2OC([C@H](O)[C@@H]2O)n2cnc3c(N)ncnc23)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C22H33N10O22P5S/c1-2-60-22-30-11-17(24)26-6-28-19(11)32(22)21-15(36)13(34)9(50-21)4-48-56(39,40)52-58(43,44)54-59(45,46)53-57(41,42)51-55(37,38)47-3-8-12(33)14(35)20(49-8)31-7-29-10-16(23)25-5-27-18(10)31/h5-9,12-15,20-21,33-36H,2-4H2,1H3,(H,37,38)(H,39,40)(H,41,42)(H,43,44)(H,45,46)(H2,23,25,27)(H2,24,26,28)/t8-,9-,12-,13-,14-,15-,20?,21?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against rat adenylate kinase II was determined in the presence of AMP, competitive inhibition |
J Med Chem 25: 1179-84 (1983)
BindingDB Entry DOI: 10.7270/Q25D8SD7 |
More data for this
Ligand-Target Pair | |
Adenylate kinase 2, mitochondrial
(Rattus norvegicus) | BDBM50367044
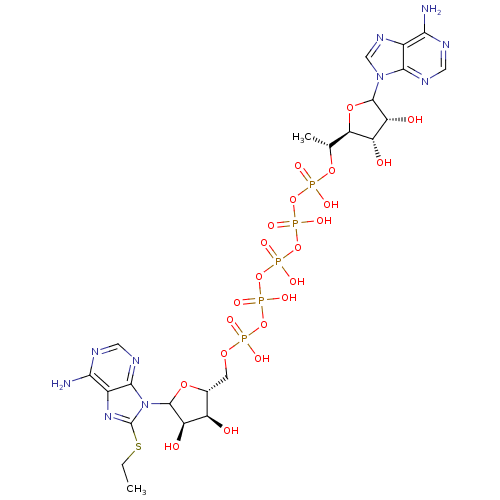
(CHEMBL605639)Show SMILES CCSc1nc2c(N)ncnc2n1C1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)O[C@H](C)[C@H]2OC([C@H](O)[C@@H]2O)n2cnc3c(N)ncnc23)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C23H35N10O22P5S/c1-3-61-23-31-11-18(25)27-6-29-20(11)33(23)22-14(36)12(34)9(49-22)4-48-56(38,39)52-58(42,43)54-60(46,47)55-59(44,45)53-57(40,41)51-8(2)16-13(35)15(37)21(50-16)32-7-30-10-17(24)26-5-28-19(10)32/h5-9,12-16,21-22,34-37H,3-4H2,1-2H3,(H,38,39)(H,40,41)(H,42,43)(H,44,45)(H,46,47)(H2,24,26,28)(H2,25,27,29)/t8-,9-,12-,13+,14-,15-,16-,21?,22?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against rat Adenylate kinase M isoenzyme in the presence of AMP non competitive inhibition |
J Med Chem 25: 1179-84 (1983)
BindingDB Entry DOI: 10.7270/Q25D8SD7 |
More data for this
Ligand-Target Pair | |
Adenylate kinase 2, mitochondrial
(Rattus norvegicus) | BDBM50367044

(CHEMBL605639)Show SMILES CCSc1nc2c(N)ncnc2n1C1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)O[C@H](C)[C@H]2OC([C@H](O)[C@@H]2O)n2cnc3c(N)ncnc23)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C23H35N10O22P5S/c1-3-61-23-31-11-18(25)27-6-29-20(11)33(23)22-14(36)12(34)9(49-22)4-48-56(38,39)52-58(42,43)54-60(46,47)55-59(44,45)53-57(40,41)51-8(2)16-13(35)15(37)21(50-16)32-7-30-10-17(24)26-5-28-19(10)32/h5-9,12-16,21-22,34-37H,3-4H2,1-2H3,(H,38,39)(H,40,41)(H,42,43)(H,44,45)(H,46,47)(H2,24,26,28)(H2,25,27,29)/t8-,9-,12-,13+,14-,15-,16-,21?,22?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against rat adenylate kinase II was determined in the presence of AMP, non competitive inhibition |
J Med Chem 25: 1179-84 (1983)
BindingDB Entry DOI: 10.7270/Q25D8SD7 |
More data for this
Ligand-Target Pair | |
Thymidine kinase, cytosolic
(Rattus norvegicus) | BDBM50205415
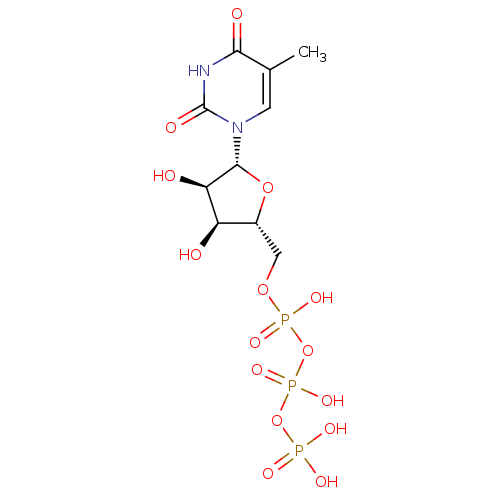
(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 3.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Evaluated for the mixed objective Non-competitive inhibition constant Ki against TdR varied rat mitochondrial thymidine kinase |
J Med Chem 25: 801-5 (1982)
BindingDB Entry DOI: 10.7270/Q2BZ66M6 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM18134
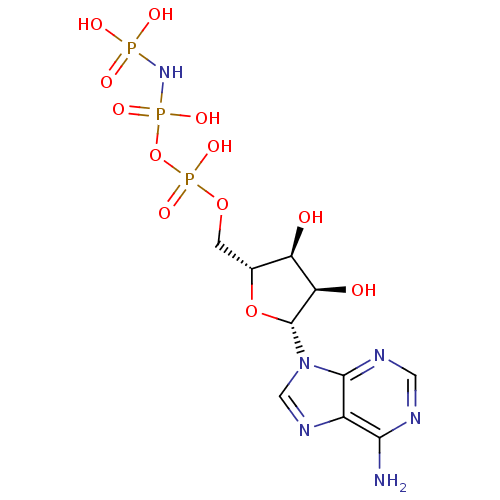
(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)NP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H17N6O12P3/c11-8-5-9(13-2-12-8)16(3-14-5)10-7(18)6(17)4(27-10)1-26-31(24,25)28-30(22,23)15-29(19,20)21/h2-4,6-7,10,17-18H,1H2,(H,24,25)(H2,11,12,13)(H4,15,19,20,21,22,23)/t4-,6-,7-,10-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat Methionine adenosyltransferase was reported |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
Thymidine kinase, cytosolic
(Rattus norvegicus) | BDBM50367095

(CHEMBL607917)Show SMILES Nc1ncnc2n(cnc12)C1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(=O)Cc2cn(C3C[C@H](O)[C@@H](CO)O3)c(=O)[nH]c2=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C20H28N7O17P3/c21-16-13-17(23-6-22-16)27(7-24-13)19-15(31)14(30)11(42-19)4-40-46(36,37)44-47(38,39)43-45(34,35)5-8-2-26(20(33)25-18(8)32)12-1-9(29)10(3-28)41-12/h2,6-7,9-12,14-15,19,28-31H,1,3-5H2,(H,34,35)(H,36,37)(H,38,39)(H2,21,22,23)(H,25,32,33)/t9-,10+,11+,12?,14+,15+,19?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Evaluated for the competitive inhibition constant Ki against ATP varied rat mitochondrial thymidine kinase |
J Med Chem 25: 801-5 (1982)
BindingDB Entry DOI: 10.7270/Q2BZ66M6 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM18134

(({[({[(2R,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-3,4-...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)NP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H17N6O12P3/c11-8-5-9(13-2-12-8)16(3-14-5)10-7(18)6(17)4(27-10)1-26-31(24,25)28-30(22,23)15-29(19,20)21/h2-4,6-7,10,17-18H,1H2,(H,24,25)(H2,11,12,13)(H4,15,19,20,21,22,23)/t4-,6-,7-,10-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 5.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat kidney Methionine adenosyltransferase II |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
Thymidine kinase, cytosolic
(Rattus norvegicus) | BDBM50205415

(({[({[(2R,3S,4R,5R)-3,4-dihydroxy-5-(5-methyl-2,4-...)Show SMILES Cc1cn([C@@H]2O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]2O)c(=O)[nH]c1=O Show InChI InChI=1S/C10H17N2O15P3/c1-4-2-12(10(16)11-8(4)15)9-7(14)6(13)5(25-9)3-24-29(20,21)27-30(22,23)26-28(17,18)19/h2,5-7,9,13-14H,3H2,1H3,(H,20,21)(H,22,23)(H,11,15,16)(H2,17,18,19)/t5-,6-,7-,9-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 6.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Evaluated for the Non-competitive inhibition constant Ki against TdR varied rat cytoplasmic soluble thymidine kinase |
J Med Chem 25: 801-5 (1982)
BindingDB Entry DOI: 10.7270/Q2BZ66M6 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1
(Rattus norvegicus) | BDBM50367305
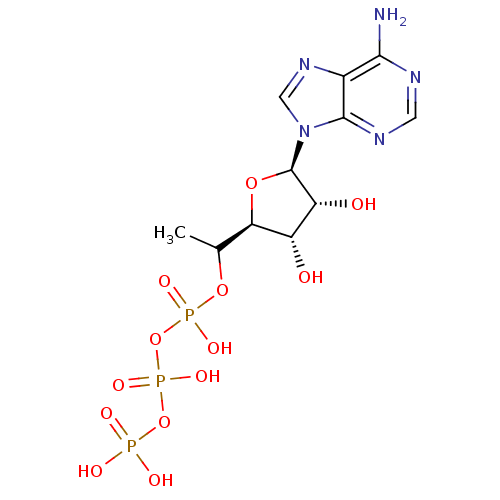
(CHEMBL1791430)Show SMILES CC(OP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C11H18N5O13P3/c1-4(27-31(22,23)29-32(24,25)28-30(19,20)21)8-6(17)7(18)11(26-8)16-3-15-5-9(12)13-2-14-10(5)16/h2-4,6-8,11,17-18H,1H3,(H,22,23)(H,24,25)(H2,12,13,14)(H2,19,20,21)/t4?,6-,7+,8+,11+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 9.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat liver Methionine adenosyltransferase I, activity expressed as Ki |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
Adenylate kinase 2, mitochondrial
(Rattus norvegicus) | BDBM50367040
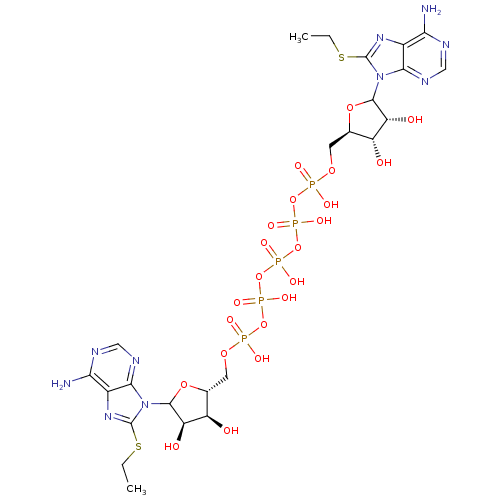
(CHEMBL606017)Show SMILES CCSc1nc2c(N)ncnc2n1C1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OC[C@H]2OC([C@H](O)[C@@H]2O)n2c(SCC)nc3c(N)ncnc23)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C24H37N10O22P5S2/c1-3-62-23-31-11-17(25)27-7-29-19(11)33(23)21-15(37)13(35)9(51-21)5-49-57(39,40)53-59(43,44)55-61(47,48)56-60(45,46)54-58(41,42)50-6-10-14(36)16(38)22(52-10)34-20-12(18(26)28-8-30-20)32-24(34)63-4-2/h7-10,13-16,21-22,35-38H,3-6H2,1-2H3,(H,39,40)(H,41,42)(H,43,44)(H,45,46)(H,47,48)(H2,25,27,29)(H2,26,28,30)/t9-,10-,13-,14-,15-,16-,21?,22?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against rat adenylate kinase II was determined in the presence of AMP, competitive inhibition |
J Med Chem 25: 1179-84 (1983)
BindingDB Entry DOI: 10.7270/Q25D8SD7 |
More data for this
Ligand-Target Pair | |
Adenylate kinase 2, mitochondrial
(Rattus norvegicus) | BDBM50367040

(CHEMBL606017)Show SMILES CCSc1nc2c(N)ncnc2n1C1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OC[C@H]2OC([C@H](O)[C@@H]2O)n2c(SCC)nc3c(N)ncnc23)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C24H37N10O22P5S2/c1-3-62-23-31-11-17(25)27-7-29-19(11)33(23)21-15(37)13(35)9(51-21)5-49-57(39,40)53-59(43,44)55-61(47,48)56-60(45,46)54-58(41,42)50-6-10-14(36)16(38)22(52-10)34-20-12(18(26)28-8-30-20)32-24(34)63-4-2/h7-10,13-16,21-22,35-38H,3-6H2,1-2H3,(H,39,40)(H,41,42)(H,43,44)(H,45,46)(H,47,48)(H2,25,27,29)(H2,26,28,30)/t9-,10-,13-,14-,15-,16-,21?,22?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against rat adenylate kinase II was determined in the presence of AMP, competitive inhibition |
J Med Chem 25: 1179-84 (1983)
BindingDB Entry DOI: 10.7270/Q25D8SD7 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367328
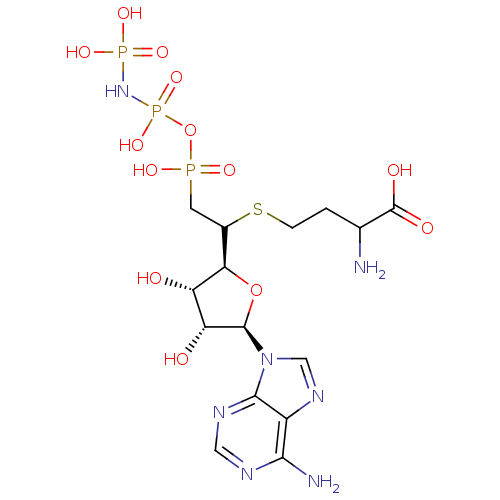
(CHEMBL1791415)Show SMILES NC(CCSC(CP(O)(=O)OP(O)(=O)NP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C15H26N7O13P3S/c16-6(15(25)26)1-2-39-7(3-36(27,28)35-38(32,33)21-37(29,30)31)11-9(23)10(24)14(34-11)22-5-20-8-12(17)18-4-19-13(8)22/h4-7,9-11,14,23-24H,1-3,16H2,(H,25,26)(H,27,28)(H2,17,18,19)(H4,21,29,30,31,32,33)/t6?,7?,9-,10+,11+,14+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated on novikoff hepatoma (MAT-T) form of rat methionine adenosyltransferase, when ATP was the variable substrate (60 uM... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1
(Rattus norvegicus) | BDBM50367303
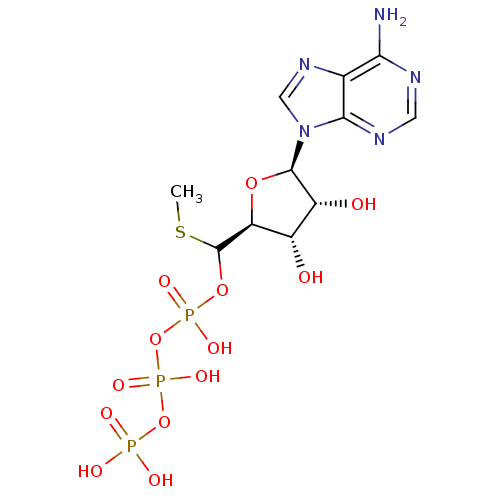
(CHEMBL1791433)Show SMILES CSC(OP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11(27-31(22,23)29-32(24,25)28-30(19,20)21)7-5(17)6(18)10(26-7)16-3-15-4-8(12)13-2-14-9(4)16/h2-3,5-7,10-11,17-18H,1H3,(H,22,23)(H,24,25)(H2,12,13,14)(H2,19,20,21)/t5-,6+,7-,10+,11?/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat liver Methionine adenosyltransferase I, activity expressed as Ki |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1
(Rattus norvegicus) | BDBM50367042
(CHEMBL606221)Show SMILES Nc1ncnc2n(cnc12)C1O[C@H](CCOP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C11H18N5O13P3/c12-9-6-10(14-3-13-9)16(4-15-6)11-8(18)7(17)5(27-11)1-2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-5,7-8,11,17-18H,1-2H2,(H,22,23)(H,24,25)(H2,12,13,14)(H2,19,20,21)/t5-,7-,8-,11?/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat liver Methionine adenosyltransferase I |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50367328

(CHEMBL1791415)Show SMILES NC(CCSC(CP(O)(=O)OP(O)(=O)NP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C15H26N7O13P3S/c16-6(15(25)26)1-2-39-7(3-36(27,28)35-38(32,33)21-37(29,30)31)11-9(23)10(24)14(34-11)22-5-20-8-12(17)18-4-19-13(8)22/h4-7,9-11,14,23-24H,1-3,16H2,(H,25,26)(H,27,28)(H2,17,18,19)(H4,21,29,30,31,32,33)/t6?,7?,9-,10+,11+,14+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated with kidney Methionine adenosyltransferase II form of rat methionine adenosyltransferase, when ATP was the variable... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367328

(CHEMBL1791415)Show SMILES NC(CCSC(CP(O)(=O)OP(O)(=O)NP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C15H26N7O13P3S/c16-6(15(25)26)1-2-39-7(3-36(27,28)35-38(32,33)21-37(29,30)31)11-9(23)10(24)14(34-11)22-5-20-8-12(17)18-4-19-13(8)22/h4-7,9-11,14,23-24H,1-3,16H2,(H,25,26)(H,27,28)(H2,17,18,19)(H4,21,29,30,31,32,33)/t6?,7?,9-,10+,11+,14+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated with kidney (MAT-2) form of rat methionine adenosyltransferase, when methionine was the variable substrate (2 mM) |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367329
(CHEMBL1791416)Show SMILES NC(CCSC(CP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C15H25N6O14P3S/c16-6(15(24)25)1-2-39-7(3-36(26,27)34-38(31,32)35-37(28,29)30)11-9(22)10(23)14(33-11)21-5-20-8-12(17)18-4-19-13(8)21/h4-7,9-11,14,22-23H,1-3,16H2,(H,24,25)(H,26,27)(H,31,32)(H2,17,18,19)(H2,28,29,30)/t6?,7?,9-,10+,11+,14+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated on novikoff hepatoma (MAT-T) form of rat methionine adenosyltransferase, when ATP was the variable substrate (60 uM... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367329

(CHEMBL1791416)Show SMILES NC(CCSC(CP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C15H25N6O14P3S/c16-6(15(24)25)1-2-39-7(3-36(26,27)34-38(31,32)35-37(28,29)30)11-9(22)10(23)14(33-11)21-5-20-8-12(17)18-4-19-13(8)21/h4-7,9-11,14,22-23H,1-3,16H2,(H,24,25)(H,26,27)(H,31,32)(H2,17,18,19)(H2,28,29,30)/t6?,7?,9-,10+,11+,14+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated on novikoff hepatoma (MAT-T) form of rat methionine adenosyltransferase, when ATP was the variable substrate (60 uM... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50452293
(CHEMBL2092766)Show SMILES CCC[C@]1(COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C13H22N5O13P3/c1-2-3-13(4-28-33(24,25)31-34(26,27)30-32(21,22)23)9(20)8(19)12(29-13)18-6-17-7-10(14)15-5-16-11(7)18/h5-6,8-9,12,19-20H,2-4H2,1H3,(H,24,25)(H,26,27)(H2,14,15,16)(H2,21,22,23)/t8-,9+,12-,13-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated on novikoff hepatoma (MAT-T) form of rat methionine adenosyltransferase, when ATP was the variable substrate (60 uM... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50452293

(CHEMBL2092766)Show SMILES CCC[C@]1(COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C13H22N5O13P3/c1-2-3-13(4-28-33(24,25)31-34(26,27)30-32(21,22)23)9(20)8(19)12(29-13)18-6-17-7-10(14)15-5-16-11(7)18/h5-6,8-9,12,19-20H,2-4H2,1H3,(H,24,25)(H,26,27)(H2,14,15,16)(H2,21,22,23)/t8-,9+,12-,13-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated on novikoff hepatoma (MAT-T) form of rat methionine adenosyltransferase, when ATP was the variable substrate (60 uM... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367301
(CHEMBL1791432)Show SMILES CCCC(OP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C13H22N5O13P3/c1-2-3-6(29-33(24,25)31-34(26,27)30-32(21,22)23)10-8(19)9(20)13(28-10)18-5-17-7-11(14)15-4-16-12(7)18/h4-6,8-10,13,19-20H,2-3H2,1H3,(H,24,25)(H,26,27)(H2,14,15,16)(H2,21,22,23)/t6?,8-,9+,10+,13+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of rat methionine adenosyltransferase, activity expressed as Ki |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367328

(CHEMBL1791415)Show SMILES NC(CCSC(CP(O)(=O)OP(O)(=O)NP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C15H26N7O13P3S/c16-6(15(25)26)1-2-39-7(3-36(27,28)35-38(32,33)21-37(29,30)31)11-9(23)10(24)14(34-11)22-5-20-8-12(17)18-4-19-13(8)22/h4-7,9-11,14,23-24H,1-3,16H2,(H,25,26)(H,27,28)(H2,17,18,19)(H4,21,29,30,31,32,33)/t6?,7?,9-,10+,11+,14+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated with kidney Methionine adenosyltransferase II form of rat methionine adenosyltransferase, when ATP was the variable... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50367329

(CHEMBL1791416)Show SMILES NC(CCSC(CP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C15H25N6O14P3S/c16-6(15(24)25)1-2-39-7(3-36(26,27)34-38(31,32)35-37(28,29)30)11-9(22)10(23)14(33-11)21-5-20-8-12(17)18-4-19-13(8)21/h4-7,9-11,14,22-23H,1-3,16H2,(H,24,25)(H,26,27)(H,31,32)(H2,17,18,19)(H2,28,29,30)/t6?,7?,9-,10+,11+,14+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated with kidney Methionine adenosyltransferase II form of rat methionine adenosyltransferase, when ATP was the variable... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50367329

(CHEMBL1791416)Show SMILES NC(CCSC(CP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C15H25N6O14P3S/c16-6(15(24)25)1-2-39-7(3-36(26,27)34-38(31,32)35-37(28,29)30)11-9(22)10(23)14(33-11)21-5-20-8-12(17)18-4-19-13(8)21/h4-7,9-11,14,22-23H,1-3,16H2,(H,24,25)(H,26,27)(H,31,32)(H2,17,18,19)(H2,28,29,30)/t6?,7?,9-,10+,11+,14+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated with kidney Methionine adenosyltransferase II form of rat methionine adenosyltransferase, when ATP was the variable... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50452293

(CHEMBL2092766)Show SMILES CCC[C@]1(COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C13H22N5O13P3/c1-2-3-13(4-28-33(24,25)31-34(26,27)30-32(21,22)23)9(20)8(19)12(29-13)18-6-17-7-10(14)15-5-16-11(7)18/h5-6,8-9,12,19-20H,2-4H2,1H3,(H,24,25)(H,26,27)(H2,14,15,16)(H2,21,22,23)/t8-,9+,12-,13-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated with kidney Methionine adenosyltransferase II form of rat methionine adenosyltransferase, when ATP was the variable... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50452293

(CHEMBL2092766)Show SMILES CCC[C@]1(COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C13H22N5O13P3/c1-2-3-13(4-28-33(24,25)31-34(26,27)30-32(21,22)23)9(20)8(19)12(29-13)18-6-17-7-10(14)15-5-16-11(7)18/h5-6,8-9,12,19-20H,2-4H2,1H3,(H,24,25)(H,26,27)(H2,14,15,16)(H2,21,22,23)/t8-,9+,12-,13-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated with kidney Methionine adenosyltransferase II form of rat methionine adenosyltransferase, when ATP was the variable... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50367301

(CHEMBL1791432)Show SMILES CCCC(OP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C13H22N5O13P3/c1-2-3-6(29-33(24,25)31-34(26,27)30-32(21,22)23)10-8(19)9(20)13(28-10)18-5-17-7-11(14)15-4-16-12(7)18/h4-6,8-10,13,19-20H,2-3H2,1H3,(H,24,25)(H,26,27)(H2,14,15,16)(H2,21,22,23)/t6?,8-,9+,10+,13+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat kidney Methionine adenosyltransferase II, activity expressed as Ki |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
Adenylate kinase 2, mitochondrial
(Rattus norvegicus) | BDBM50367048
(CHEMBL605430)Show SMILES CCC[C@@H](OP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OC[C@H]1OC([C@H](O)[C@@H]1O)n1c(SCC)nc2c(N)ncnc12)[C@H]1OC([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C25H39N10O22P5S/c1-3-5-10(18-15(37)17(39)23(52-18)34-9-32-12-19(26)28-7-30-21(12)34)53-59(42,43)55-61(46,47)57-62(48,49)56-60(44,45)54-58(40,41)50-6-11-14(36)16(38)24(51-11)35-22-13(20(27)29-8-31-22)33-25(35)63-4-2/h7-11,14-18,23-24,36-39H,3-6H2,1-2H3,(H,40,41)(H,42,43)(H,44,45)(H,46,47)(H,48,49)(H2,26,28,30)(H2,27,29,31)/t10-,11-,14-,15+,16-,17-,18-,23?,24?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 5.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against rat adenylate kinase II was determined in the presence of AMP, competitive inhibition |
J Med Chem 25: 1179-84 (1983)
BindingDB Entry DOI: 10.7270/Q25D8SD7 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367329

(CHEMBL1791416)Show SMILES NC(CCSC(CP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C15H25N6O14P3S/c16-6(15(24)25)1-2-39-7(3-36(26,27)34-38(31,32)35-37(28,29)30)11-9(22)10(23)14(33-11)21-5-20-8-12(17)18-4-19-13(8)21/h4-7,9-11,14,22-23H,1-3,16H2,(H,24,25)(H,26,27)(H,31,32)(H2,17,18,19)(H2,28,29,30)/t6?,7?,9-,10+,11+,14+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 6.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated with kidney (MAT-2) form of rat methionine adenosyltransferase, when methionine was the variable substrate (2 mM) |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367329

(CHEMBL1791416)Show SMILES NC(CCSC(CP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C15H25N6O14P3S/c16-6(15(24)25)1-2-39-7(3-36(26,27)34-38(31,32)35-37(28,29)30)11-9(22)10(23)14(33-11)21-5-20-8-12(17)18-4-19-13(8)21/h4-7,9-11,14,22-23H,1-3,16H2,(H,24,25)(H,26,27)(H,31,32)(H2,17,18,19)(H2,28,29,30)/t6?,7?,9-,10+,11+,14+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 6.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated with kidney (MAT-2) form of rat methionine adenosyltransferase, when methionine was the variable substrate (2 mM) |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
Adenylate kinase 2, mitochondrial
(Rattus norvegicus) | BDBM50367048

(CHEMBL605430)Show SMILES CCC[C@@H](OP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OC[C@H]1OC([C@H](O)[C@@H]1O)n1c(SCC)nc2c(N)ncnc12)[C@H]1OC([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C25H39N10O22P5S/c1-3-5-10(18-15(37)17(39)23(52-18)34-9-32-12-19(26)28-7-30-21(12)34)53-59(42,43)55-61(46,47)57-62(48,49)56-60(44,45)54-58(40,41)50-6-11-14(36)16(38)24(51-11)35-22-13(20(27)29-8-31-22)33-25(35)63-4-2/h7-11,14-18,23-24,36-39H,3-6H2,1-2H3,(H,40,41)(H,42,43)(H,44,45)(H,46,47)(H,48,49)(H2,26,28,30)(H2,27,29,31)/t10-,11-,14-,15+,16-,17-,18-,23?,24?/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 6.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against rat adenylate kinase II was determined in the presence of AMP,competitive and non competitive inhibition |
J Med Chem 25: 1179-84 (1983)
BindingDB Entry DOI: 10.7270/Q25D8SD7 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50367042

(CHEMBL606221)Show SMILES Nc1ncnc2n(cnc12)C1O[C@H](CCOP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C11H18N5O13P3/c12-9-6-10(14-3-13-9)16(4-15-6)11-8(18)7(17)5(27-11)1-2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-5,7-8,11,17-18H,1-2H2,(H,22,23)(H,24,25)(H2,12,13,14)(H2,19,20,21)/t5-,7-,8-,11?/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 7.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat kidney Methionine adenosyltransferase II |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367305

(CHEMBL1791430)Show SMILES CC(OP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C11H18N5O13P3/c1-4(27-31(22,23)29-32(24,25)28-30(19,20)21)8-6(17)7(18)11(26-8)16-3-15-5-9(12)13-2-14-10(5)16/h2-4,6-8,11,17-18H,1H3,(H,22,23)(H,24,25)(H2,12,13,14)(H2,19,20,21)/t4?,6-,7+,8+,11+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of rat methionine adenosyltransferase, activity expressed as Ki |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
Adenylate kinase 2, mitochondrial
(Rattus norvegicus) | BDBM18137

(AMP | CHEMBL752 | US11185100, TABLE 7.3 | [(2R,3S,...)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](O)[C@H]1O Show InChI InChI=1S/C10H14N5O7P/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(22-10)1-21-23(18,19)20/h2-4,6-7,10,16-17H,1H2,(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against rat adenylate kinase II was determined in the presence of AMP |
J Med Chem 25: 1179-84 (1983)
BindingDB Entry DOI: 10.7270/Q25D8SD7 |
More data for this
Ligand-Target Pair | |
Adenylate kinase 2, mitochondrial
(Rattus norvegicus) | BDBM50010309
(CHEMBL3251358)Show SMILES CCSc1nc2c(N)ncnc2n1[C@@H]1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C12H20N5O13P3S/c1-2-34-12-16-6-9(13)14-4-15-10(6)17(12)11-8(19)7(18)5(28-11)3-27-32(23,24)30-33(25,26)29-31(20,21)22/h4-5,7-8,11,18-19H,2-3H2,1H3,(H,23,24)(H,25,26)(H2,13,14,15)(H2,20,21,22)/t5-,7-,8-,11-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 8.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against rat adenylate kinase II was determined in the presence of ATP |
J Med Chem 25: 1179-84 (1983)
BindingDB Entry DOI: 10.7270/Q25D8SD7 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367331
(CHEMBL1791417)Show SMILES NC(CCS(=O)C(CP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C15H25N6O15P3S/c16-6(15(24)25)1-2-40(33)7(3-37(26,27)35-39(31,32)36-38(28,29)30)11-9(22)10(23)14(34-11)21-5-20-8-12(17)18-4-19-13(8)21/h4-7,9-11,14,22-23H,1-3,16H2,(H,24,25)(H,26,27)(H,31,32)(H2,17,18,19)(H2,28,29,30)/t6?,7?,9-,10+,11+,14+,40?/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 8.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated on novikoff hepatoma (MAT-T) form of rat methionine adenosyltransferase, when ATP was the variable substrate (60 uM... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
Adenylate kinase 2, mitochondrial
(Rattus norvegicus) | BDBM50366480

(ADENOSINE TRIPHOSPHATE | ATP)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](CO[P@](O)(=O)O[P@@](O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H16N5O13P3/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(26-10)1-25-30(21,22)28-31(23,24)27-29(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H,23,24)(H2,11,12,13)(H2,18,19,20)/t4-,6-,7-,10-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 9.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against rat adenylate kinase II was determined in the presence of ATP, non competitive inhibition |
J Med Chem 25: 1179-84 (1983)
BindingDB Entry DOI: 10.7270/Q25D8SD7 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367300
(CHEMBL610977)Show SMILES CCCCSc1ncnc2n(cnc12)C1O[C@H](OP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C13H21N4O13P3S/c1-2-3-4-34-11-7-10(14-5-15-11)17(6-16-7)12-8(18)9(19)13(27-12)28-32(23,24)30-33(25,26)29-31(20,21)22/h5-6,8-9,12-13,18-19H,2-4H2,1H3,(H,23,24)(H,25,26)(H2,20,21,22)/t8-,9+,12?,13-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of rat methionine adenosyltransferase, activity expressed as Ki |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50118232

(2-MeSATP | ATP, 2-meS | CHEMBL336208)Show SMILES CSc1nc(N)c2ncn([C@@H]3O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]3O)c2n1 Show InChI InChI=1S/C11H18N5O13P3S/c1-33-11-14-8(12)5-9(15-11)16(3-13-5)10-7(18)6(17)4(27-10)2-26-31(22,23)29-32(24,25)28-30(19,20)21/h3-4,6-7,10,17-18H,2H2,1H3,(H,22,23)(H,24,25)(H2,12,14,15)(H2,19,20,21)/t4-,6-,7-,10-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 1.10E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat Methionine adenosyltransferase was reported |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
Thymidine kinase, cytosolic
(Rattus norvegicus) | BDBM50367095

(CHEMBL607917)Show SMILES Nc1ncnc2n(cnc12)C1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(=O)Cc2cn(C3C[C@H](O)[C@@H](CO)O3)c(=O)[nH]c2=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C20H28N7O17P3/c21-16-13-17(23-6-22-16)27(7-24-13)19-15(31)14(30)11(42-19)4-40-46(36,37)44-47(38,39)43-45(34,35)5-8-2-26(20(33)25-18(8)32)12-1-9(29)10(3-28)41-12/h2,6-7,9-12,14-15,19,28-31H,1,3-5H2,(H,34,35)(H,36,37)(H,38,39)(H2,21,22,23)(H,25,32,33)/t9-,10+,11+,12?,14+,15+,19?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Evaluated for the mixed objective Non-competitive inhibition constant Ki against TdR varied rat mitochondrial thymidine kinase |
J Med Chem 25: 801-5 (1982)
BindingDB Entry DOI: 10.7270/Q2BZ66M6 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367307
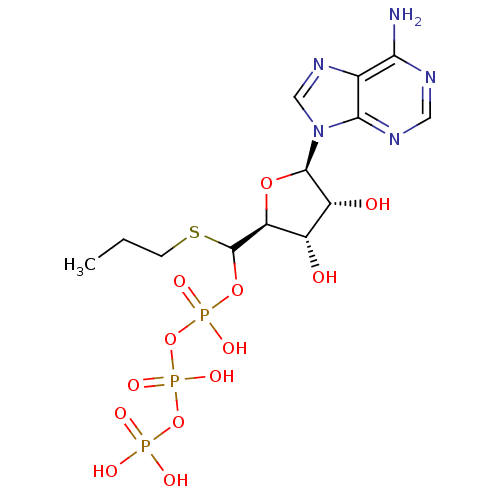
(CHEMBL1791431)Show SMILES CCCSC(OP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C13H22N5O13P3S/c1-2-3-35-13(29-33(24,25)31-34(26,27)30-32(21,22)23)9-7(19)8(20)12(28-9)18-5-17-6-10(14)15-4-16-11(6)18/h4-5,7-9,12-13,19-20H,2-3H2,1H3,(H,24,25)(H,26,27)(H2,14,15,16)(H2,21,22,23)/t7-,8+,9-,12+,13?/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of rat methionine adenosyltransferase, activity expressed as Ki |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50367331

(CHEMBL1791417)Show SMILES NC(CCS(=O)C(CP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C15H25N6O15P3S/c16-6(15(24)25)1-2-40(33)7(3-37(26,27)35-39(31,32)36-38(28,29)30)11-9(22)10(23)14(34-11)21-5-20-8-12(17)18-4-19-13(8)21/h4-7,9-11,14,22-23H,1-3,16H2,(H,24,25)(H,26,27)(H,31,32)(H2,17,18,19)(H2,28,29,30)/t6?,7?,9-,10+,11+,14+,40?/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition constant was evaluated with kidney Methionine adenosyltransferase II form of rat methionine adenosyltransferase, when ATP was the variable... |
J Med Chem 29: 1030-8 (1986)
BindingDB Entry DOI: 10.7270/Q2CF9QN4 |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50026197
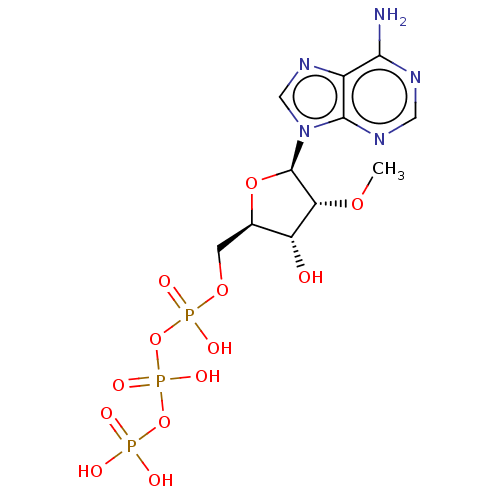
(Adenosine 5'-triphosphate derivative | CHEMBL31428...)Show SMILES CO[C@@H]1[C@H](O)[C@@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O[C@H]1n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C11H18N5O13P3/c1-25-8-7(17)5(2-26-31(21,22)29-32(23,24)28-30(18,19)20)27-11(8)16-4-15-6-9(12)13-3-14-10(6)16/h3-5,7-8,11,17H,2H2,1H3,(H,21,22)(H,23,24)(H2,12,13,14)(H2,18,19,20) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.40E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat kidney Methionine adenosyltransferase II, activity expressed as Ki |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50026197

(Adenosine 5'-triphosphate derivative | CHEMBL31428...)Show SMILES CO[C@@H]1[C@H](O)[C@@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O[C@H]1n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C11H18N5O13P3/c1-25-8-7(17)5(2-26-31(21,22)29-32(23,24)28-30(18,19)20)27-11(8)16-4-15-6-9(12)13-3-14-10(6)16/h3-5,7-8,11,17H,2H2,1H3,(H,21,22)(H,23,24)(H2,12,13,14)(H2,18,19,20) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.70E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of rat methionine adenosyltransferase, activity expressed as Ki |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50026193
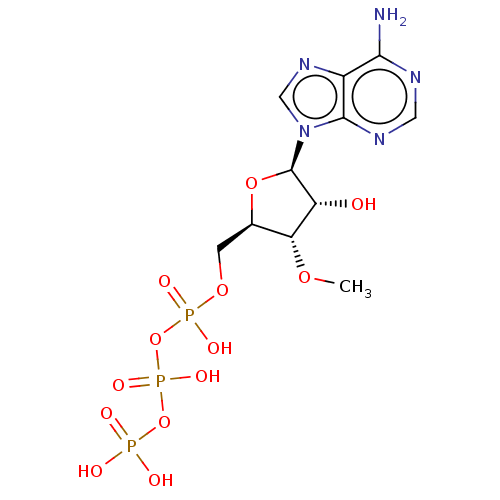
(Adenosine 5'-triphosphate derivative | CHEMBL31428...)Show SMILES CO[C@@H]1[C@@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O[C@H]([C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C11H18N5O13P3/c1-25-8-5(2-26-31(21,22)29-32(23,24)28-30(18,19)20)27-11(7(8)17)16-4-15-6-9(12)13-3-14-10(6)16/h3-5,7-8,11,17H,2H2,1H3,(H,21,22)(H,23,24)(H2,12,13,14)(H2,18,19,20) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.70E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat kidney Methionine adenosyltransferase II, activity expressed as Ki |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50026193

(Adenosine 5'-triphosphate derivative | CHEMBL31428...)Show SMILES CO[C@@H]1[C@@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O[C@H]([C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C11H18N5O13P3/c1-25-8-5(2-26-31(21,22)29-32(23,24)28-30(18,19)20)27-11(7(8)17)16-4-15-6-9(12)13-3-14-10(6)16/h3-5,7-8,11,17H,2H2,1H3,(H,21,22)(H,23,24)(H2,12,13,14)(H2,18,19,20) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.80E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of rat methionine adenosyltransferase, activity expressed as Ki |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-2
(Rattus norvegicus) | BDBM50367305

(CHEMBL1791430)Show SMILES CC(OP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C11H18N5O13P3/c1-4(27-31(22,23)29-32(24,25)28-30(19,20)21)8-6(17)7(18)11(26-8)16-3-15-5-9(12)13-2-14-10(5)16/h2-4,6-8,11,17-18H,1H3,(H,22,23)(H,24,25)(H2,12,13,14)(H2,19,20,21)/t4?,6-,7+,8+,11+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.90E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat kidney Methionine adenosyltransferase II, activity expressed as Ki |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
S-adenosylmethionine synthase isoform type-1/type-2
(Rattus norvegicus) | BDBM50367304
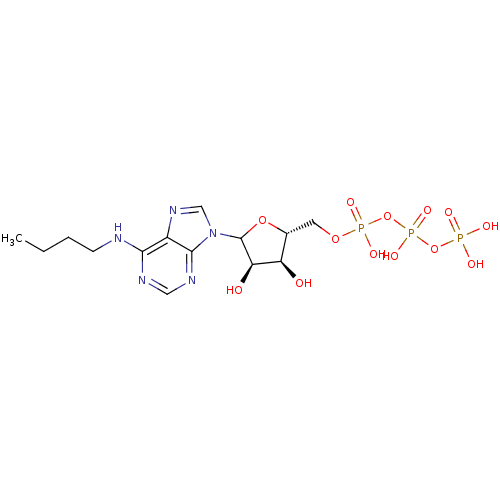
(CHEMBL610404)Show SMILES CCCCNc1ncnc2n(cnc12)C1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(O)=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C14H24N5O13P3/c1-2-3-4-15-12-9-13(17-6-16-12)19(7-18-9)14-11(21)10(20)8(30-14)5-29-34(25,26)32-35(27,28)31-33(22,23)24/h6-8,10-11,14,20-21H,2-5H2,1H3,(H,25,26)(H,27,28)(H,15,16,17)(H2,22,23,24)/t8-,10-,11-,14?/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.10E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant against rat methionine adenosyltransferase was reported |
J Med Chem 29: 318-22 (1986)
BindingDB Entry DOI: 10.7270/Q2DF6RSH |
More data for this
Ligand-Target Pair | |
Thymidine kinase, cytosolic
(Rattus norvegicus) | BDBM50367095

(CHEMBL607917)Show SMILES Nc1ncnc2n(cnc12)C1O[C@H](COP(O)(=O)OP(O)(=O)OP(O)(=O)Cc2cn(C3C[C@H](O)[C@@H](CO)O3)c(=O)[nH]c2=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C20H28N7O17P3/c21-16-13-17(23-6-22-16)27(7-24-13)19-15(31)14(30)11(42-19)4-40-46(36,37)44-47(38,39)43-45(34,35)5-8-2-26(20(33)25-18(8)32)12-1-9(29)10(3-28)41-12/h2,6-7,9-12,14-15,19,28-31H,1,3-5H2,(H,34,35)(H,36,37)(H,38,39)(H2,21,22,23)(H,25,32,33)/t9-,10+,11+,12?,14+,15+,19?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.60E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Evaluated for the mixed objective Non-competitive inhibition constant Ki against ATP varied cytoplasmic soluble thymidine kinase |
J Med Chem 25: 801-5 (1982)
BindingDB Entry DOI: 10.7270/Q2BZ66M6 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data