 Found 326 hits with Last Name = 'hamley' and Initial = 'p'
Found 326 hits with Last Name = 'hamley' and Initial = 'p' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124519

(5,8-Difluoro-2-furan-2-yl-1,2-dihydro-quinazolin-4...)Show InChI InChI=1S/C12H9F2N3O/c13-6-3-4-7(14)10-9(6)11(15)17-12(16-10)8-2-1-5-18-8/h1-5,12,16H,(H2,15,17) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R& D Charnwood
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against human inducible nitiric oxide synthase |
J Med Chem 46: 913-6 (2003)
Article DOI: 10.1021/jm0255926
BindingDB Entry DOI: 10.7270/Q2S75H3M |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50339997
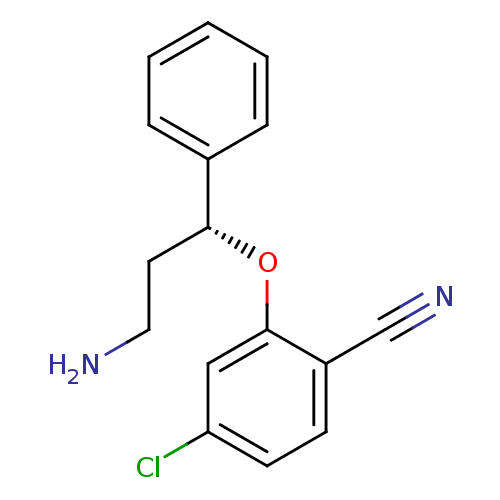
((R)-2-(3-amino-1-phenylpropoxy)-4-chlorobenzonitri...)Show InChI InChI=1S/C16H15ClN2O/c17-14-7-6-13(11-19)16(10-14)20-15(8-9-18)12-4-2-1-3-5-12/h1-7,10,15H,8-9,18H2/t15-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Charnwood
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant iNOS |
Bioorg Med Chem Lett 21: 2468-71 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.061
BindingDB Entry DOI: 10.7270/Q29G5N4S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50339998
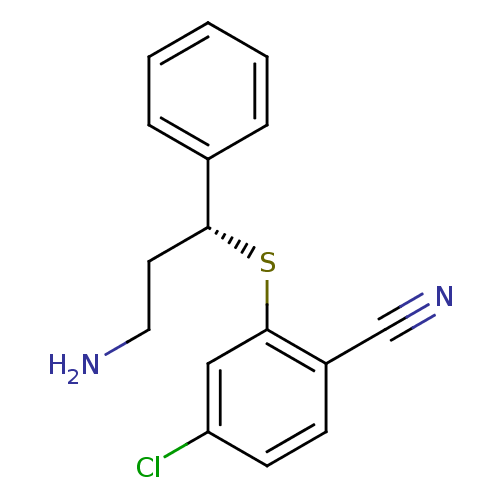
((R)-2-(3-amino-1-phenylpropylthio)-4-chlorobenzoni...)Show InChI InChI=1S/C16H15ClN2S/c17-14-7-6-13(11-19)16(10-14)20-15(8-9-18)12-4-2-1-3-5-12/h1-7,10,15H,8-9,18H2/t15-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Charnwood
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant iNOS |
Bioorg Med Chem Lett 21: 2468-71 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.061
BindingDB Entry DOI: 10.7270/Q29G5N4S |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50340003

((R)-2-(3-amino-1-phenylpropylthio)-6-methylnicotin...)Show InChI InChI=1S/C16H17N3S/c1-12-7-8-14(11-18)16(19-12)20-15(9-10-17)13-5-3-2-4-6-13/h2-8,15H,9-10,17H2,1H3/t15-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Charnwood
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant iNOS |
Bioorg Med Chem Lett 21: 2468-71 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.061
BindingDB Entry DOI: 10.7270/Q29G5N4S |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50340004
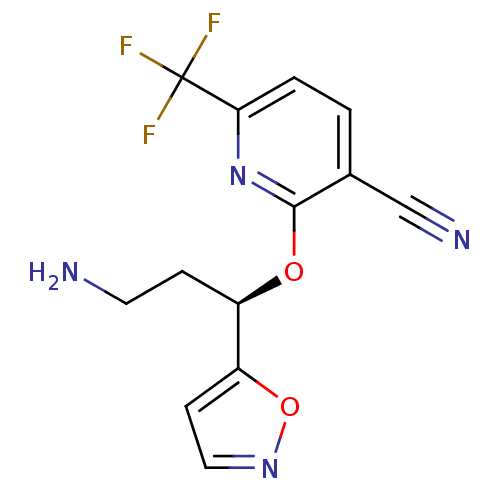
((R)-2-(3-amino-1-(isoxazol-5-yl)propoxy)-6-(triflu...)Show SMILES NCC[C@@H](Oc1nc(ccc1C#N)C(F)(F)F)c1ccno1 |r| Show InChI InChI=1S/C13H11F3N4O2/c14-13(15,16)11-2-1-8(7-18)12(20-11)21-9(3-5-17)10-4-6-19-22-10/h1-2,4,6,9H,3,5,17H2/t9-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.40 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Charnwood
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant iNOS |
Bioorg Med Chem Lett 21: 2468-71 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.061
BindingDB Entry DOI: 10.7270/Q29G5N4S |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Rattus norvegicus (rat)) | BDBM50098959

(5-Ethynyl-4,5-dihydro-thieno[2,3-c]pyridin-7-ylami...)Show InChI InChI=1S/C9H8N2S/c1-2-7-5-6-3-4-12-8(6)9(10)11-7/h1,3-4,7H,5H2,(H2,10,11) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
Ability to inhibit the conversion of [3H]-L-Arg to [3H]-L-citrulline catalyzed by n-NOS from rat cerebellum |
Bioorg Med Chem Lett 11: 1027-30 (2001)
BindingDB Entry DOI: 10.7270/Q2F76BT7 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50340002

(2-[[(1R)-3-Amino-1-phenylpropyl]oxy]-4-chloro-5-fl...)Show InChI InChI=1S/C16H14ClFN2O/c17-13-9-16(12(10-20)8-14(13)18)21-15(6-7-19)11-4-2-1-3-5-11/h1-5,8-9,15H,6-7,19H2/t15-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Charnwood
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant iNOS |
Bioorg Med Chem Lett 21: 2468-71 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.061
BindingDB Entry DOI: 10.7270/Q29G5N4S |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50339996

((R)-4-chloro-2-(3-(methylamino)-1-phenylpropoxy)be...)Show InChI InChI=1S/C17H17ClN2O/c1-20-10-9-16(13-5-3-2-4-6-13)21-17-11-15(18)8-7-14(17)12-19/h2-8,11,16,20H,9-10H2,1H3/t16-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Charnwood
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant iNOS |
Bioorg Med Chem Lett 21: 2468-71 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.061
BindingDB Entry DOI: 10.7270/Q29G5N4S |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50091817
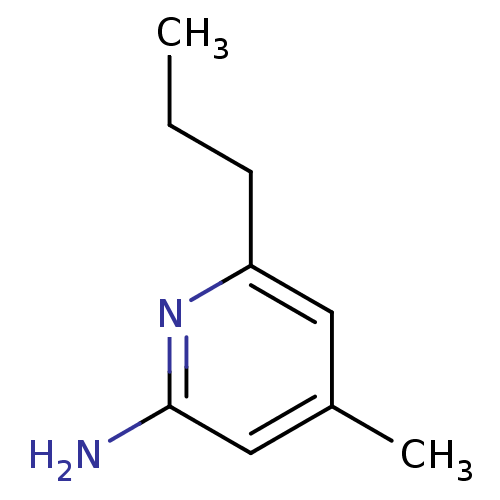
(4-Methyl-6-propyl-pyridin-2-ylamine | 4-Methyl-6-p...)Show InChI InChI=1S/C9H14N2/c1-3-4-8-5-7(2)6-9(10)11-8/h5-6H,3-4H2,1-2H3,(H2,10,11) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Inducible nitric oxide synthase. |
J Med Chem 47: 3320-3 (2004)
Article DOI: 10.1021/jm031035n
BindingDB Entry DOI: 10.7270/Q25M656D |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM36395

(5-Fluoro-2-thiophen-2-yl-1,2-dihydroquinazolin-4-a...)Show InChI InChI=1S/C12H10FN3S/c13-7-3-1-4-8-10(7)11(14)16-12(15-8)9-5-2-6-17-9/h1-6,12,15H,(H2,14,16) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | 7.0 | 37 |
The Scripps Research Institute
| Assay Description
Nitric oxide formation from NOS was monitored by the hemoglobin capture assay. The assay was initiated by addition of enzyme and was monitored at 401... |
Nat Chem Biol 4: 700-7 (2008)
Article DOI: 10.1038/nchembio.115
BindingDB Entry DOI: 10.7270/Q2SB443Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50091817

(4-Methyl-6-propyl-pyridin-2-ylamine | 4-Methyl-6-p...)Show InChI InChI=1S/C9H14N2/c1-3-4-8-5-7(2)6-9(10)11-8/h5-6H,3-4H2,1-2H3,(H2,10,11) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
In vitro inhibition of human neuronal nitric oxide synthase. |
J Med Chem 47: 3320-3 (2004)
Article DOI: 10.1021/jm031035n
BindingDB Entry DOI: 10.7270/Q25M656D |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Rattus norvegicus (rat)) | BDBM50098962
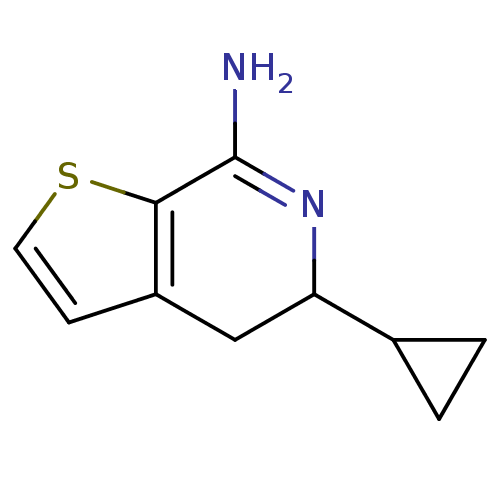
(5-Cyclopropyl-4,5-dihydro-thieno[2,3-c]pyridin-7-y...)Show InChI InChI=1S/C10H12N2S/c11-10-9-7(3-4-13-9)5-8(12-10)6-1-2-6/h3-4,6,8H,1-2,5H2,(H2,11,12) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
Ability to inhibit the conversion of [3H]-L-Arg to [3H]-L-citrulline catalyzed by n-NOS from rat cerebellum |
Bioorg Med Chem Lett 11: 1027-30 (2001)
BindingDB Entry DOI: 10.7270/Q2F76BT7 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50091817

(4-Methyl-6-propyl-pyridin-2-ylamine | 4-Methyl-6-p...)Show InChI InChI=1S/C9H14N2/c1-3-4-8-5-7(2)6-9(10)11-8/h5-6H,3-4H2,1-2H3,(H2,10,11) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | 7.0 | 37 |
The Scripps Research Institute
| Assay Description
Nitric oxide formation from NOS was monitored by the hemoglobin capture assay. The assay was initiated by addition of enzyme and was monitored at 401... |
Nat Chem Biol 4: 700-7 (2008)
Article DOI: 10.1038/nchembio.115
BindingDB Entry DOI: 10.7270/Q2SB443Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, brain
(Rattus norvegicus (rat)) | BDBM50098961
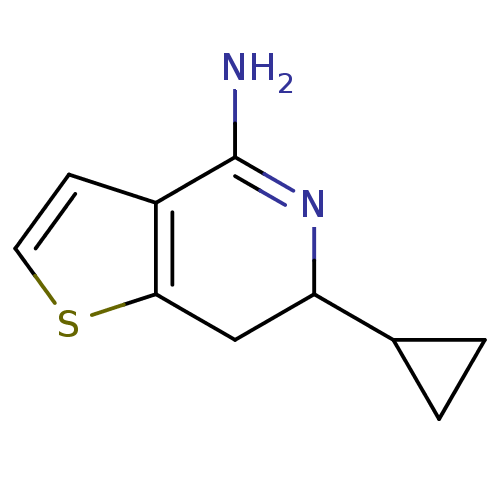
(6-Cyclopropyl-6,7-dihydro-thieno[3,2-c]pyridin-4-y...)Show InChI InChI=1S/C10H12N2S/c11-10-7-3-4-13-9(7)5-8(12-10)6-1-2-6/h3-4,6,8H,1-2,5H2,(H2,11,12) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
Ability to inhibit the conversion of [3H]-L-Arg to [3H]-L-citrulline catalyzed by n-NOS from rat cerebellum |
Bioorg Med Chem Lett 11: 1027-30 (2001)
BindingDB Entry DOI: 10.7270/Q2F76BT7 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124537
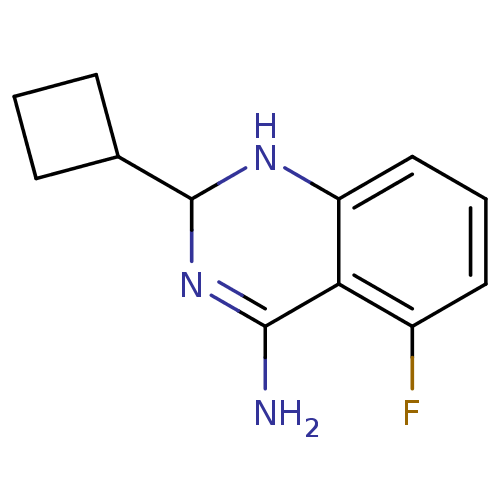
(2-Cyclobutyl-5-fluoro-1,2-dihydro-quinazolin-4-yla...)Show InChI InChI=1S/C12H14FN3/c13-8-5-2-6-9-10(8)11(14)16-12(15-9)7-3-1-4-7/h2,5-7,12,15H,1,3-4H2,(H2,14,16) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R& D Charnwood
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against human inducible nitiric oxide synthase |
J Med Chem 46: 913-6 (2003)
Article DOI: 10.1021/jm0255926
BindingDB Entry DOI: 10.7270/Q2S75H3M |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50339995
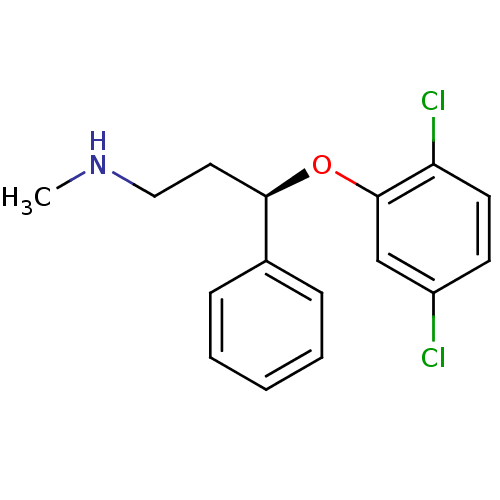
((R)-3-(2,5-dichlorophenoxy)-N-methyl-3-phenylpropa...)Show InChI InChI=1S/C16H17Cl2NO/c1-19-10-9-15(12-5-3-2-4-6-12)20-16-11-13(17)7-8-14(16)18/h2-8,11,15,19H,9-10H2,1H3/t15-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Charnwood
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant iNOS |
Bioorg Med Chem Lett 21: 2468-71 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.061
BindingDB Entry DOI: 10.7270/Q29G5N4S |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50091817

(4-Methyl-6-propyl-pyridin-2-ylamine | 4-Methyl-6-p...)Show InChI InChI=1S/C9H14N2/c1-3-4-8-5-7(2)6-9(10)11-8/h5-6H,3-4H2,1-2H3,(H2,10,11) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | 7.0 | 37 |
The Scripps Research Institute
| Assay Description
Nitric oxide formation from NOS was monitored by the hemoglobin capture assay. The assay was initiated by addition of enzyme and was monitored at 401... |
Nat Chem Biol 4: 700-7 (2008)
Article DOI: 10.1038/nchembio.115
BindingDB Entry DOI: 10.7270/Q2SB443Q |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Rattus norvegicus (rat)) | BDBM50098951
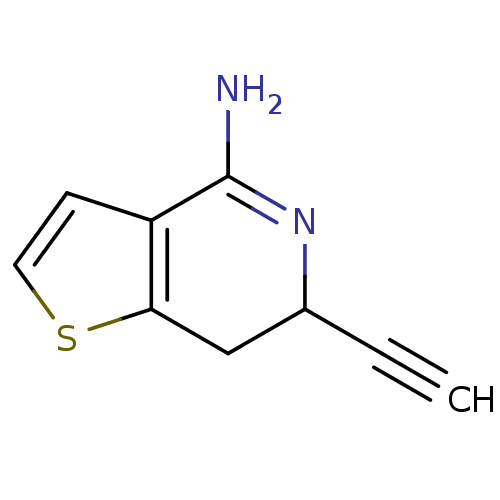
(6-Ethynyl-6,7-dihydro-thieno[3,2-c]pyridin-4-ylami...)Show InChI InChI=1S/C9H8N2S/c1-2-6-5-8-7(3-4-12-8)9(10)11-6/h1,3-4,6H,5H2,(H2,10,11) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
Ability to inhibit the conversion of [3H]-L-Arg to [3H]-L-citrulline catalyzed by n-NOS from rat cerebellum |
Bioorg Med Chem Lett 11: 1027-30 (2001)
BindingDB Entry DOI: 10.7270/Q2F76BT7 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50148168
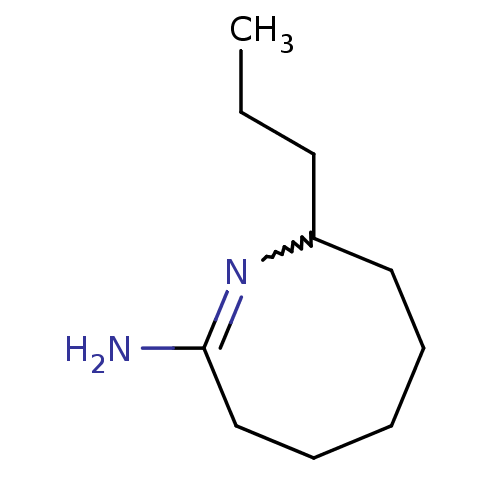
((E)-8-Propyl-3,4,5,6,7,8-hexahydro-azocin-2-ylamin...)Show InChI InChI=1S/C10H20N2/c1-2-6-9-7-4-3-5-8-10(11)12-9/h9H,2-8H2,1H3,(H2,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Inducible nitric oxide synthase. |
J Med Chem 47: 3320-3 (2004)
Article DOI: 10.1021/jm031035n
BindingDB Entry DOI: 10.7270/Q25M656D |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124531

(4-[4'-amino-5'-fluorospiro[hexahydropyridine-4,2'-...)Show SMILES NC1=NC2(CCN(CC2)C(=O)c2ccc(cc2)C#N)Nc2cccc(F)c12 |t:1| Show InChI InChI=1S/C20H18FN5O/c21-15-2-1-3-16-17(15)18(23)25-20(24-16)8-10-26(11-9-20)19(27)14-6-4-13(12-22)5-7-14/h1-7,24H,8-11H2,(H2,23,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R& D Charnwood
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against human inducible nitiric oxide synthase |
J Med Chem 46: 913-6 (2003)
Article DOI: 10.1021/jm0255926
BindingDB Entry DOI: 10.7270/Q2S75H3M |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM36396

(CD24894148 | N-[2-(4-Amino-5,8-difluoro-1,2-dihydr...)Show SMILES NC1=NC(CCNC(=O)c2ccoc2)Nc2c(F)ccc(F)c12 |t:1| Show InChI InChI=1S/C15H14F2N4O2/c16-9-1-2-10(17)13-12(9)14(18)21-11(20-13)3-5-19-15(22)8-4-6-23-7-8/h1-2,4,6-7,11,20H,3,5H2,(H2,18,21)(H,19,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | 7.0 | 37 |
The Scripps Research Institute
| Assay Description
Nitric oxide formation from NOS was monitored by the hemoglobin capture assay. The assay was initiated by addition of enzyme and was monitored at 401... |
Nat Chem Biol 4: 700-7 (2008)
Article DOI: 10.1038/nchembio.115
BindingDB Entry DOI: 10.7270/Q2SB443Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, endothelial
(Homo sapiens (Human)) | BDBM50091817

(4-Methyl-6-propyl-pyridin-2-ylamine | 4-Methyl-6-p...)Show InChI InChI=1S/C9H14N2/c1-3-4-8-5-7(2)6-9(10)11-8/h5-6H,3-4H2,1-2H3,(H2,10,11) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | 7.0 | 37 |
The Scripps Research Institute
| Assay Description
Nitric oxide formation from NOS was monitored by the hemoglobin capture assay. The assay was initiated by addition of enzyme and was monitored at 401... |
Nat Chem Biol 4: 700-7 (2008)
Article DOI: 10.1038/nchembio.115
BindingDB Entry DOI: 10.7270/Q2SB443Q |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124535

(1-(6-CYANO-3-PYRIDYLCARBONYL)-5',8'-DIFLUOROSPIRO[...)Show SMILES NC1=NC2(CCN(CC2)C(=O)c2ccc(nc2)C#N)Nc2c(F)ccc(F)c12 |t:1| Show InChI InChI=1S/C19H16F2N6O/c20-13-3-4-14(21)16-15(13)17(23)26-19(25-16)5-7-27(8-6-19)18(28)11-1-2-12(9-22)24-10-11/h1-4,10,25H,5-8H2,(H2,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 35 | n/a | n/a | n/a | n/a | 7.0 | 37 |
The Scripps Research Institute
| Assay Description
Nitric oxide formation from NOS was monitored by the hemoglobin capture assay. The assay was initiated by addition of enzyme and was monitored at 401... |
Nat Chem Biol 4: 700-7 (2008)
Article DOI: 10.1038/nchembio.115
BindingDB Entry DOI: 10.7270/Q2SB443Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124521

(CHEMBL174290 | ethyl 4'-amino-5'-fluorospiro[hexah...)Show SMILES CCOC(=O)N1CCC2(CC1)Nc1cccc(F)c1C(N)=N2 |c:22| Show InChI InChI=1S/C15H19FN4O2/c1-2-22-14(21)20-8-6-15(7-9-20)18-11-5-3-4-10(16)12(11)13(17)19-15/h3-5,18H,2,6-9H2,1H3,(H2,17,19) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R& D Charnwood
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against human inducible nitiric oxide synthase |
J Med Chem 46: 913-6 (2003)
Article DOI: 10.1021/jm0255926
BindingDB Entry DOI: 10.7270/Q2S75H3M |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124535

(1-(6-CYANO-3-PYRIDYLCARBONYL)-5',8'-DIFLUOROSPIRO[...)Show SMILES NC1=NC2(CCN(CC2)C(=O)c2ccc(nc2)C#N)Nc2c(F)ccc(F)c12 |t:1| Show InChI InChI=1S/C19H16F2N6O/c20-13-3-4-14(21)16-15(13)17(23)26-19(25-16)5-7-27(8-6-19)18(28)11-1-2-12(9-22)24-10-11/h1-4,10,25H,5-8H2,(H2,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R& D Charnwood
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against human inducible nitiric oxide synthase |
J Med Chem 46: 913-6 (2003)
Article DOI: 10.1021/jm0255926
BindingDB Entry DOI: 10.7270/Q2S75H3M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124535

(1-(6-CYANO-3-PYRIDYLCARBONYL)-5',8'-DIFLUOROSPIRO[...)Show SMILES NC1=NC2(CCN(CC2)C(=O)c2ccc(nc2)C#N)Nc2c(F)ccc(F)c12 |t:1| Show InChI InChI=1S/C19H16F2N6O/c20-13-3-4-14(21)16-15(13)17(23)26-19(25-16)5-7-27(8-6-19)18(28)11-1-2-12(9-22)24-10-11/h1-4,10,25H,5-8H2,(H2,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Inducible nitric oxide synthase. |
J Med Chem 47: 3320-3 (2004)
Article DOI: 10.1021/jm031035n
BindingDB Entry DOI: 10.7270/Q25M656D |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124533

(2-methylsulfanylethyl 4'-amino-5'-fluorospiro[hexa...)Show SMILES CSCCOC(=O)N1CCC2(CC1)Nc1cccc(F)c1C(N)=N2 |c:24| Show InChI InChI=1S/C16H21FN4O2S/c1-24-10-9-23-15(22)21-7-5-16(6-8-21)19-12-4-2-3-11(17)13(12)14(18)20-16/h2-4,19H,5-10H2,1H3,(H2,18,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R& D Charnwood
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against human inducible nitiric oxide synthase |
J Med Chem 46: 913-6 (2003)
Article DOI: 10.1021/jm0255926
BindingDB Entry DOI: 10.7270/Q2S75H3M |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Rattus norvegicus (rat)) | BDBM50098950
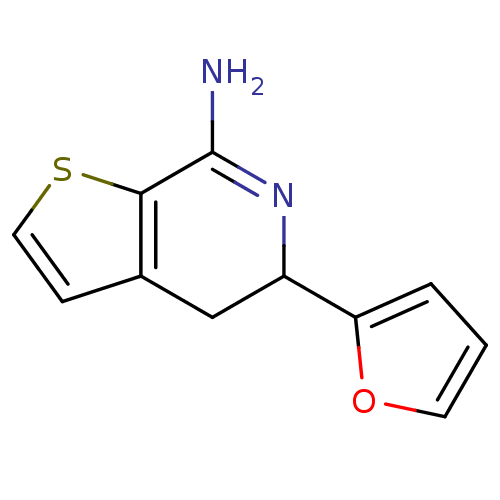
(5-Furan-2-yl-4,5-dihydro-thieno[2,3-c]pyridin-7-yl...)Show InChI InChI=1S/C11H10N2OS/c12-11-10-7(3-5-15-10)6-8(13-11)9-2-1-4-14-9/h1-5,8H,6H2,(H2,12,13) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
Ability to inhibit the conversion of [3H]-L-Arg to [3H]-L-citrulline catalyzed by n-NOS from rat cerebellum |
Bioorg Med Chem Lett 11: 1027-30 (2001)
BindingDB Entry DOI: 10.7270/Q2F76BT7 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50098959

(5-Ethynyl-4,5-dihydro-thieno[2,3-c]pyridin-7-ylami...)Show InChI InChI=1S/C9H8N2S/c1-2-7-5-6-3-4-12-8(6)9(10)11-7/h1,3-4,7H,5H2,(H2,10,11) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
Ability to inhibit the conversion of [3H]-L-Arg to [3H]-L-citrulline catalyzed by i-NOS from human DLD-1 cells |
Bioorg Med Chem Lett 11: 1027-30 (2001)
BindingDB Entry DOI: 10.7270/Q2F76BT7 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM36402

((3R)-3-Propyl-2,3-dihydrothieno[2,3-f][1,4]oxazepi...)Show InChI InChI=1S/C10H14N2OS/c1-2-3-7-6-13-8-4-5-14-9(8)10(11)12-7/h4-5,7H,2-3,6H2,1H3,(H2,11,12)/t7-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | 7.0 | 37 |
The Scripps Research Institute
| Assay Description
Nitric oxide formation from NOS was monitored by the hemoglobin capture assay. The assay was initiated by addition of enzyme and was monitored at 401... |
Nat Chem Biol 4: 700-7 (2008)
Article DOI: 10.1038/nchembio.115
BindingDB Entry DOI: 10.7270/Q2SB443Q |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM36401
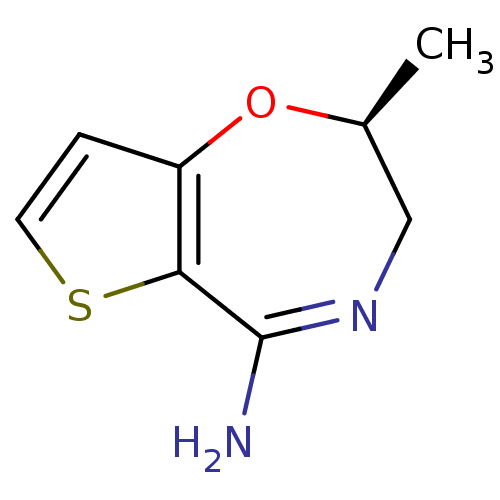
((2S)-2-Methyl-2,3-dihydrothieno[2,3-f][1,4]oxazepi...)Show InChI InChI=1S/C8H10N2OS/c1-5-4-10-8(9)7-6(11-5)2-3-12-7/h2-3,5H,4H2,1H3,(H2,9,10)/t5-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | 7.0 | 37 |
The Scripps Research Institute
| Assay Description
Nitric oxide formation from NOS was monitored by the hemoglobin capture assay. The assay was initiated by addition of enzyme and was monitored at 401... |
Nat Chem Biol 4: 700-7 (2008)
Article DOI: 10.1038/nchembio.115
BindingDB Entry DOI: 10.7270/Q2SB443Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124535

(1-(6-CYANO-3-PYRIDYLCARBONYL)-5',8'-DIFLUOROSPIRO[...)Show SMILES NC1=NC2(CCN(CC2)C(=O)c2ccc(nc2)C#N)Nc2c(F)ccc(F)c12 |t:1| Show InChI InChI=1S/C19H16F2N6O/c20-13-3-4-14(21)16-15(13)17(23)26-19(25-16)5-7-27(8-6-19)18(28)11-1-2-12(9-22)24-10-11/h1-4,10,25H,5-8H2,(H2,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Charnwood
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant iNOS |
Bioorg Med Chem Lett 21: 2468-71 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.061
BindingDB Entry DOI: 10.7270/Q29G5N4S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50091805

(2-amino-4,6-dimethylpyridine | 4,6-Dimethyl-pyridi...)Show InChI InChI=1S/C7H10N2/c1-5-3-6(2)9-7(8)4-5/h3-4H,1-2H3,(H2,8,9) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 45 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Inducible nitric oxide synthase. |
J Med Chem 47: 3320-3 (2004)
Article DOI: 10.1021/jm031035n
BindingDB Entry DOI: 10.7270/Q25M656D |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124532
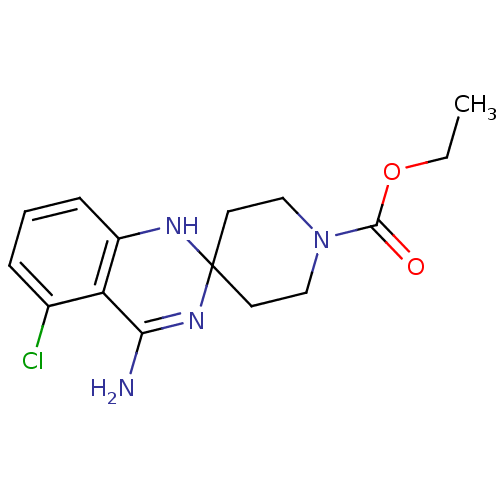
(CHEMBL366978 | ethyl 4'-amino-5'-chlorospiro[hexah...)Show SMILES CCOC(=O)N1CCC2(CC1)Nc1cccc(Cl)c1C(N)=N2 |c:22| Show InChI InChI=1S/C15H19ClN4O2/c1-2-22-14(21)20-8-6-15(7-9-20)18-11-5-3-4-10(16)12(11)13(17)19-15/h3-5,18H,2,6-9H2,1H3,(H2,17,19) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 47 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R& D Charnwood
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against human inducible nitiric oxide synthase |
J Med Chem 46: 913-6 (2003)
Article DOI: 10.1021/jm0255926
BindingDB Entry DOI: 10.7270/Q2S75H3M |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124520
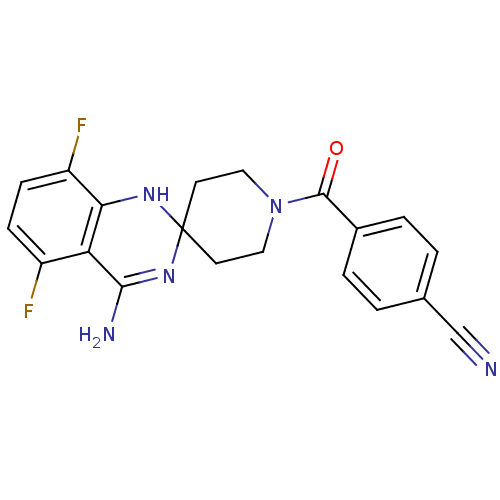
(4-[4'-amino-5',8'-difluorospiro[hexahydropyridine-...)Show SMILES NC1=NC2(CCN(CC2)C(=O)c2ccc(cc2)C#N)Nc2c(F)ccc(F)c12 |t:1| Show InChI InChI=1S/C20H17F2N5O/c21-14-5-6-15(22)17-16(14)18(24)26-20(25-17)7-9-27(10-8-20)19(28)13-3-1-12(11-23)2-4-13/h1-6,25H,7-10H2,(H2,24,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R& D Charnwood
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against human inducible nitiric oxide synthase |
J Med Chem 46: 913-6 (2003)
Article DOI: 10.1021/jm0255926
BindingDB Entry DOI: 10.7270/Q2S75H3M |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, endothelial
(Homo sapiens (Human)) | BDBM50091817

(4-Methyl-6-propyl-pyridin-2-ylamine | 4-Methyl-6-p...)Show InChI InChI=1S/C9H14N2/c1-3-4-8-5-7(2)6-9(10)11-8/h5-6H,3-4H2,1-2H3,(H2,10,11) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
In vitro inhibition of human endothelial nitric oxide synthase. |
J Med Chem 47: 3320-3 (2004)
Article DOI: 10.1021/jm031035n
BindingDB Entry DOI: 10.7270/Q25M656D |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, endothelial
(Homo sapiens (Human)) | BDBM50098962

(5-Cyclopropyl-4,5-dihydro-thieno[2,3-c]pyridin-7-y...)Show InChI InChI=1S/C10H12N2S/c11-10-9-7(3-4-13-9)5-8(12-10)6-1-2-6/h3-4,6,8H,1-2,5H2,(H2,11,12) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
Ability to inhibit the conversion of [3H]-L-Arg to [3H]-L-citrulline catalyzed by e-NOS from HUVECcells |
Bioorg Med Chem Lett 11: 1027-30 (2001)
BindingDB Entry DOI: 10.7270/Q2F76BT7 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124529

(5-Fluoro-2-(4-fluoro-phenyl)-1,2-dihydro-quinazoli...)Show InChI InChI=1S/C14H11F2N3/c15-9-6-4-8(5-7-9)14-18-11-3-1-2-10(16)12(11)13(17)19-14/h1-7,14,18H,(H2,17,19) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R& D Charnwood
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against human inducible nitiric oxide synthase |
J Med Chem 46: 913-6 (2003)
Article DOI: 10.1021/jm0255926
BindingDB Entry DOI: 10.7270/Q2S75H3M |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Rattus norvegicus (rat)) | BDBM50098953
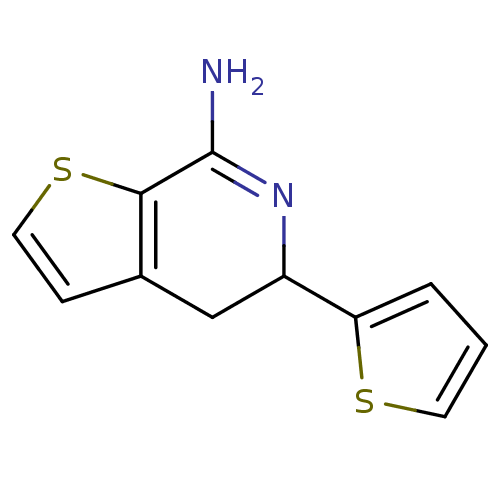
(5-Thiophen-2-yl-4,5-dihydro-thieno[2,3-c]pyridin-7...)Show InChI InChI=1S/C11H10N2S2/c12-11-10-7(3-5-15-10)6-8(13-11)9-2-1-4-14-9/h1-5,8H,6H2,(H2,12,13) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
Ability to inhibit the conversion of [3H]-L-Arg to [3H]-L-citrulline catalyzed by n-NOS from rat cerebellum |
Bioorg Med Chem Lett 11: 1027-30 (2001)
BindingDB Entry DOI: 10.7270/Q2F76BT7 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM36402

((3R)-3-Propyl-2,3-dihydrothieno[2,3-f][1,4]oxazepi...)Show InChI InChI=1S/C10H14N2OS/c1-2-3-7-6-13-8-4-5-14-9(8)10(11)12-7/h4-5,7H,2-3,6H2,1H3,(H2,11,12)/t7-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | 7.0 | 37 |
The Scripps Research Institute
| Assay Description
Nitric oxide formation from NOS was monitored by the hemoglobin capture assay. The assay was initiated by addition of enzyme and was monitored at 401... |
Nat Chem Biol 4: 700-7 (2008)
Article DOI: 10.1038/nchembio.115
BindingDB Entry DOI: 10.7270/Q2SB443Q |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50098962

(5-Cyclopropyl-4,5-dihydro-thieno[2,3-c]pyridin-7-y...)Show InChI InChI=1S/C10H12N2S/c11-10-9-7(3-4-13-9)5-8(12-10)6-1-2-6/h3-4,6,8H,1-2,5H2,(H2,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 51 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
Ability to inhibit the conversion of [3H]-L-Arg to [3H]-L-citrulline catalyzed by i-NOS from human DLD-1 cells |
Bioorg Med Chem Lett 11: 1027-30 (2001)
BindingDB Entry DOI: 10.7270/Q2F76BT7 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50340001
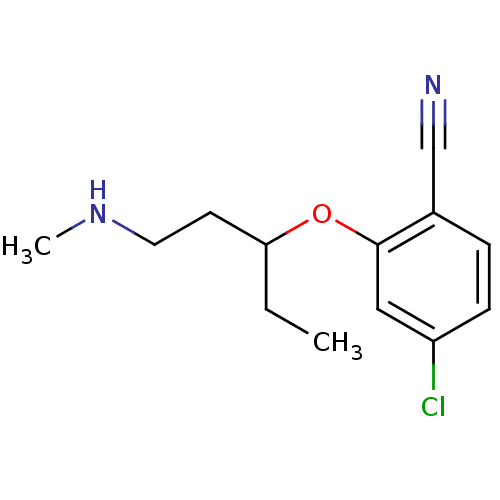
(CHEMBL1762478 | rac-4-chloro-2-(1-(methylamino)pen...)Show InChI InChI=1S/C13H17ClN2O/c1-3-12(6-7-16-2)17-13-8-11(14)5-4-10(13)9-15/h4-5,8,12,16H,3,6-7H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 51 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Charnwood
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant iNOS |
Bioorg Med Chem Lett 21: 2468-71 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.061
BindingDB Entry DOI: 10.7270/Q29G5N4S |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50148169
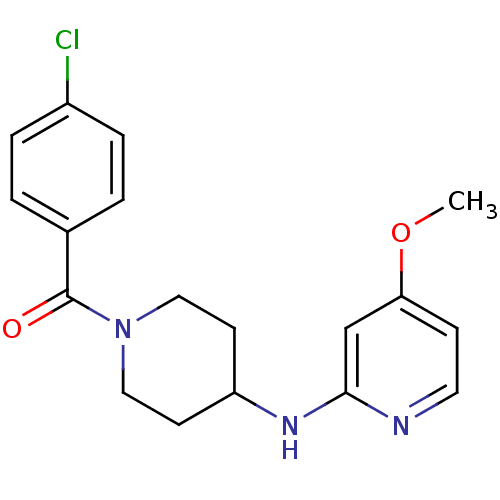
((4-Chloro-phenyl)-[4-(4-methoxy-pyridin-2-ylamino)...)Show InChI InChI=1S/C18H20ClN3O2/c1-24-16-6-9-20-17(12-16)21-15-7-10-22(11-8-15)18(23)13-2-4-14(19)5-3-13/h2-6,9,12,15H,7-8,10-11H2,1H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 53 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Inducible nitric oxide synthase. |
J Med Chem 47: 3320-3 (2004)
Article DOI: 10.1021/jm031035n
BindingDB Entry DOI: 10.7270/Q25M656D |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50124519

(5,8-Difluoro-2-furan-2-yl-1,2-dihydro-quinazolin-4...)Show InChI InChI=1S/C12H9F2N3O/c13-6-3-4-7(14)10-9(6)11(15)17-12(16-10)8-2-1-5-18-8/h1-5,12,16H,(H2,15,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R& D Charnwood
Curated by ChEMBL
| Assay Description
Inhibitory activity against human neuronal nitiric oxide synthase |
J Med Chem 46: 913-6 (2003)
Article DOI: 10.1021/jm0255926
BindingDB Entry DOI: 10.7270/Q2S75H3M |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50124525
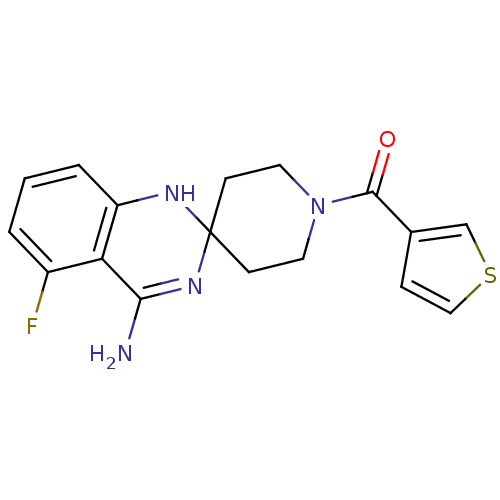
(4'-amino-5'-fluorospiro[hexahydropyridine-4,2'-(1'...)Show SMILES NC1=NC2(CCN(CC2)C(=O)c2ccsc2)Nc2cccc(F)c12 |t:1| Show InChI InChI=1S/C17H17FN4OS/c18-12-2-1-3-13-14(12)15(19)21-17(20-13)5-7-22(8-6-17)16(23)11-4-9-24-10-11/h1-4,9-10,20H,5-8H2,(H2,19,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 67 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R& D Charnwood
Curated by ChEMBL
| Assay Description
Inhibitory activity of compound against human inducible nitiric oxide synthase |
J Med Chem 46: 913-6 (2003)
Article DOI: 10.1021/jm0255926
BindingDB Entry DOI: 10.7270/Q2S75H3M |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50086467
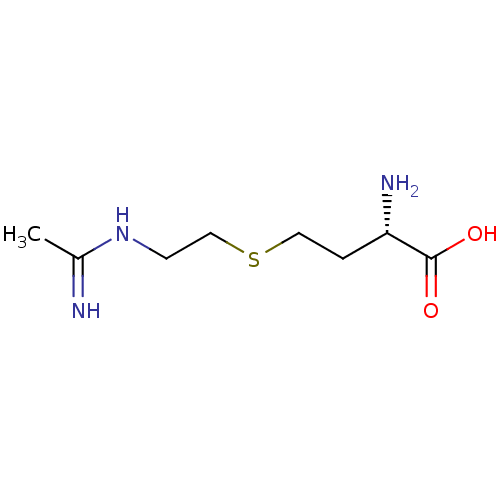
((S)-4-(2-Acetimidoylamino-ethylsulfanyl)-2-amino-b...)Show InChI InChI=1S/C8H17N3O2S/c1-6(9)11-3-5-14-4-2-7(10)8(12)13/h7H,2-5,10H2,1H3,(H2,9,11)(H,12,13)/t7-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Inducible nitric oxide synthase. |
J Med Chem 47: 3320-3 (2004)
Article DOI: 10.1021/jm031035n
BindingDB Entry DOI: 10.7270/Q25M656D |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50148167
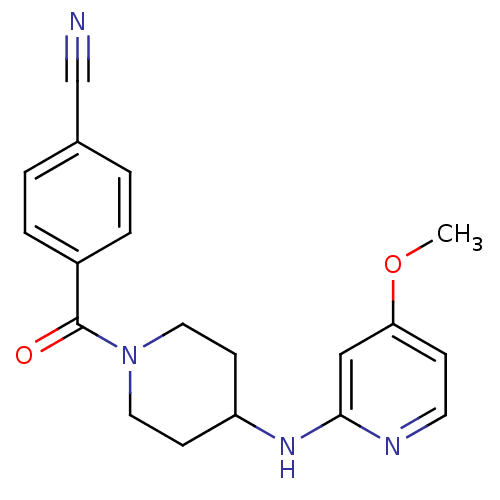
(4-(4-(4-methoxypyridin-2-ylamino)piperidine-1-carb...)Show InChI InChI=1S/C19H20N4O2/c1-25-17-6-9-21-18(12-17)22-16-7-10-23(11-8-16)19(24)15-4-2-14(13-20)3-5-15/h2-6,9,12,16H,7-8,10-11H2,1H3,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 71 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca R&D Charnwood
Curated by ChEMBL
| Assay Description
In vitro inhibition of human Inducible nitric oxide synthase. |
J Med Chem 47: 3320-3 (2004)
Article DOI: 10.1021/jm031035n
BindingDB Entry DOI: 10.7270/Q25M656D |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50339999
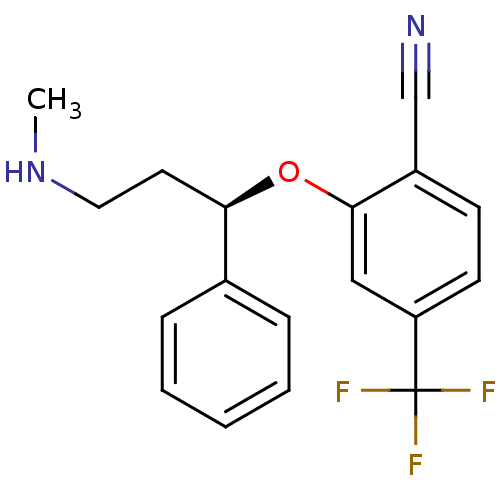
((R)-2-(3-(methylamino)-1-phenylpropoxy)-4-(trifluo...)Show SMILES CNCC[C@@H](Oc1cc(ccc1C#N)C(F)(F)F)c1ccccc1 |r| Show InChI InChI=1S/C18H17F3N2O/c1-23-10-9-16(13-5-3-2-4-6-13)24-17-11-15(18(19,20)21)8-7-14(17)12-22/h2-8,11,16,23H,9-10H2,1H3/t16-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 71 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Charnwood
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant iNOS |
Bioorg Med Chem Lett 21: 2468-71 (2011)
Article DOI: 10.1016/j.bmcl.2011.02.061
BindingDB Entry DOI: 10.7270/Q29G5N4S |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50148167

(4-(4-(4-methoxypyridin-2-ylamino)piperidine-1-carb...)Show InChI InChI=1S/C19H20N4O2/c1-25-17-6-9-21-18(12-17)22-16-7-10-23(11-8-16)19(24)15-4-2-14(13-20)3-5-15/h2-6,9,12,16H,7-8,10-11H2,1H3,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 74 | n/a | n/a | n/a | n/a | 7.0 | 37 |
The Scripps Research Institute
| Assay Description
Nitric oxide formation from NOS was monitored by the hemoglobin capture assay. The assay was initiated by addition of enzyme and was monitored at 401... |
Nat Chem Biol 4: 700-7 (2008)
Article DOI: 10.1038/nchembio.115
BindingDB Entry DOI: 10.7270/Q2SB443Q |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, endothelial
(Homo sapiens (Human)) | BDBM36401

((2S)-2-Methyl-2,3-dihydrothieno[2,3-f][1,4]oxazepi...)Show InChI InChI=1S/C8H10N2OS/c1-5-4-10-8(9)7-6(11-5)2-3-12-7/h2-3,5H,4H2,1H3,(H2,9,10)/t5-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | 7.0 | 37 |
The Scripps Research Institute
| Assay Description
Nitric oxide formation from NOS was monitored by the hemoglobin capture assay. The assay was initiated by addition of enzyme and was monitored at 401... |
Nat Chem Biol 4: 700-7 (2008)
Article DOI: 10.1038/nchembio.115
BindingDB Entry DOI: 10.7270/Q2SB443Q |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data