 Found 75 hits with Last Name = 'hesek' and Initial = 'd'
Found 75 hits with Last Name = 'hesek' and Initial = 'd' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM82122

(Vinylsulfone, 7)Show InChI InChI=1S/C15H14O3S2/c16-20(17,12-4-11-19)15-9-7-14(8-10-15)18-13-5-2-1-3-6-13/h1-10,12,19H,11H2/b12-4+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
| Assay Description
Inhibition assay using matrix metalloproteinase (MMP-2). |
Chem Biol Drug Des 74: 527-34 (2009)
Checked by Author
Article DOI: 10.1111/j.1747-0285.2009.00881.x
BindingDB Entry DOI: 10.7270/Q20P0XJV |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50335495
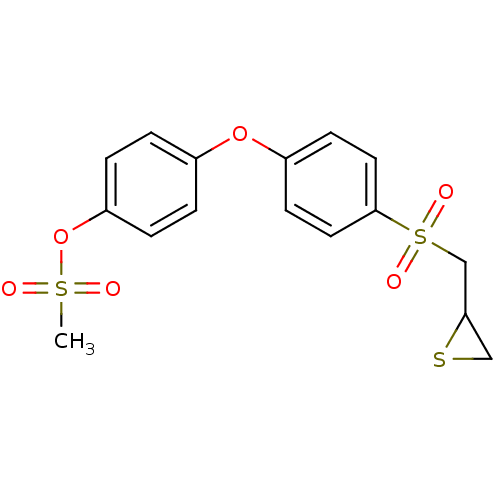
(4-(4-(thiiran-2-ylmethylsulfonyl)phenoxy)phenyl me...)Show SMILES CS(=O)(=O)Oc1ccc(Oc2ccc(cc2)S(=O)(=O)CC2CS2)cc1 Show InChI InChI=1S/C16H16O6S3/c1-24(17,18)22-14-4-2-12(3-5-14)21-13-6-8-16(9-7-13)25(19,20)11-15-10-23-15/h2-9,15H,10-11H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP9 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50388914
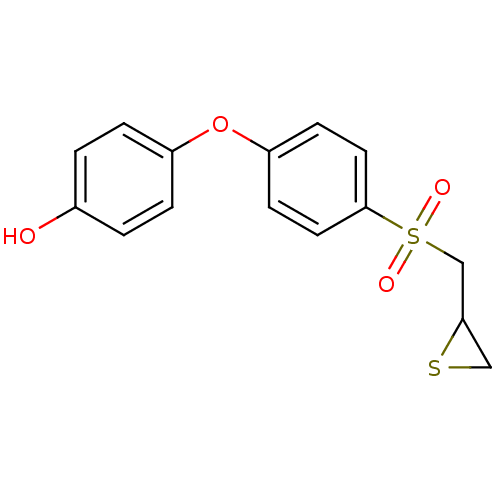
(CHEMBL2063274 | US10357546, p-OH SB-3CT)Show InChI InChI=1S/C15H14O4S2/c16-11-1-3-12(4-2-11)19-13-5-7-15(8-6-13)21(17,18)10-14-9-20-14/h1-8,14,16H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP2 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50388914

(CHEMBL2063274 | US10357546, p-OH SB-3CT)Show InChI InChI=1S/C15H14O4S2/c16-11-1-3-12(4-2-11)19-13-5-7-15(8-6-13)21(17,18)10-14-9-20-14/h1-8,14,16H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human MMP2 expressed in human HeLa cells |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50264809
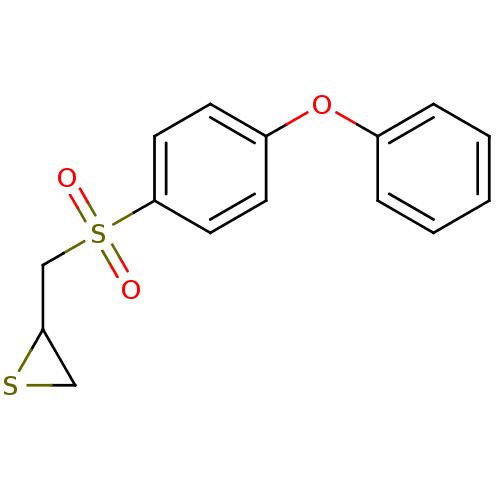
(2-((4-phenoxyphenylsulfonyl)methyl)thiirane | CHEM...)Show InChI InChI=1S/C15H14O3S2/c16-20(17,11-14-10-19-14)15-8-6-13(7-9-15)18-12-4-2-1-3-5-12/h1-9,14H,10-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 13.9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP2 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50459505

(CHEMBL4215451)Show SMILES Cl.NCc1ccc(Oc2ccc(cc2)S(=O)(=O)C[C@H]2CS2)cc1 |r| Show InChI InChI=1S/C16H17NO3S2.ClH/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15;/h1-8,15H,9-11,17H2;1H/t15-;/m1./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Slow binding inhibition of human recombinant MMP9 catalytic domain using MOCAc-Pro-Leu-Gly-Leu-A2pr(Dnp)-Ala-Arg-NH2 substrate |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50335495

(4-(4-(thiiran-2-ylmethylsulfonyl)phenoxy)phenyl me...)Show SMILES CS(=O)(=O)Oc1ccc(Oc2ccc(cc2)S(=O)(=O)CC2CS2)cc1 Show InChI InChI=1S/C16H16O6S3/c1-24(17,18)22-14-4-2-12(3-5-14)21-13-6-8-16(9-7-13)25(19,20)11-15-10-23-15/h2-9,15H,10-11H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP2 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50264809

(2-((4-phenoxyphenylsulfonyl)methyl)thiirane | CHEM...)Show InChI InChI=1S/C15H14O3S2/c16-20(17,11-14-10-19-14)15-8-6-13(7-9-15)18-12-4-2-1-3-5-12/h1-9,14H,10-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 28 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human MMP2 |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM50264809

(2-((4-phenoxyphenylsulfonyl)methyl)thiirane | CHEM...)Show InChI InChI=1S/C15H14O3S2/c16-20(17,11-14-10-19-14)15-8-6-13(7-9-15)18-12-4-2-1-3-5-12/h1-9,14H,10-11H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP14 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50459506
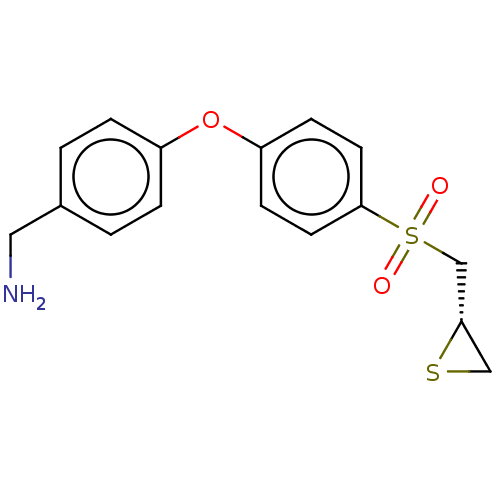
(CHEMBL4214929)Show SMILES Cl.NCc1ccc(Oc2ccc(cc2)S(=O)(=O)C[C@@H]2CS2)cc1 |r| Show InChI InChI=1S/C16H17NO3S2.ClH/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15;/h1-8,15H,9-11,17H2;1H/t15-;/m0./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Slow binding inhibition of human recombinant MMP2 using MOCAc-Pro-Leu-Gly-Leu-A2pr(Dnp)-Ala-Arg-NH2 substrate |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50388911
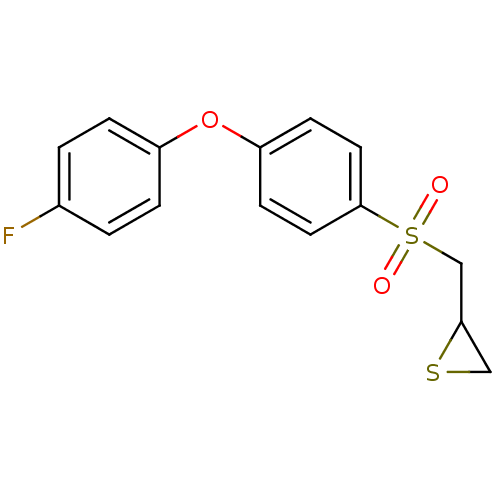
(CHEMBL2063270)Show InChI InChI=1S/C15H13FO3S2/c16-11-1-3-12(4-2-11)19-13-5-7-15(8-6-13)21(17,18)10-14-9-20-14/h1-8,14H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 44 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP9 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM50459506

(CHEMBL4214929)Show SMILES Cl.NCc1ccc(Oc2ccc(cc2)S(=O)(=O)C[C@@H]2CS2)cc1 |r| Show InChI InChI=1S/C16H17NO3S2.ClH/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15;/h1-8,15H,9-11,17H2;1H/t15-;/m0./s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 53 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Slow binding inhibition of human recombinant MMP14 catalytic domain using MOCAc-Pro-Leu-Gly-Leu-A2pr(Dnp)-Ala-Arg-NH2 substrate |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50388911

(CHEMBL2063270)Show InChI InChI=1S/C15H13FO3S2/c16-11-1-3-12(4-2-11)19-13-5-7-15(8-6-13)21(17,18)10-14-9-20-14/h1-8,14H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 61 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP2 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50264809

(2-((4-phenoxyphenylsulfonyl)methyl)thiirane | CHEM...)Show InChI InChI=1S/C15H14O3S2/c16-20(17,11-14-10-19-14)15-8-6-13(7-9-15)18-12-4-2-1-3-5-12/h1-9,14H,10-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 64 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
| Assay Description
Inhibition assay using matrix metalloproteinase (MMP-2). |
Chem Biol Drug Des 74: 527-34 (2009)
Checked by Author
Article DOI: 10.1111/j.1747-0285.2009.00881.x
BindingDB Entry DOI: 10.7270/Q20P0XJV |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50388912
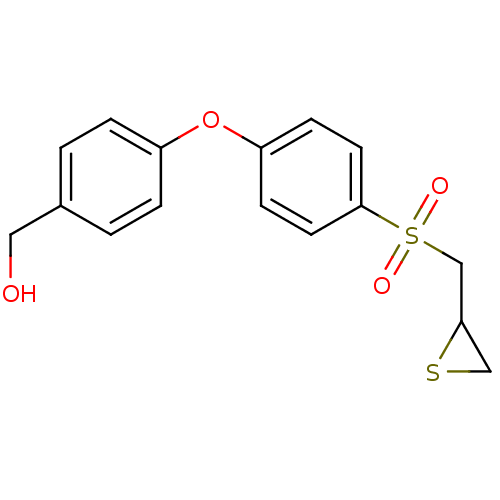
(CHEMBL2063273)Show InChI InChI=1S/C16H16O4S2/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15/h1-8,15,17H,9-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 78 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP2 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM389618
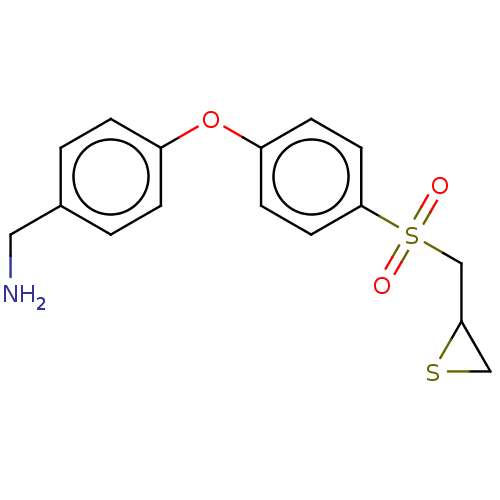
(US10357546, ND-336 | US9951035, ND-336 (5))Show InChI InChI=1S/C16H17NO3S2/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15/h1-8,15H,9-11,17H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 85 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Slow binding inhibition of human recombinant MMP2 using MOCAc-Pro-Leu-Gly-Leu-A2pr(Dnp)-Ala-Arg-NH2 substrate |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM50388914

(CHEMBL2063274 | US10357546, p-OH SB-3CT)Show InChI InChI=1S/C15H14O4S2/c16-11-1-3-12(4-2-11)19-13-5-7-15(8-6-13)21(17,18)10-14-9-20-14/h1-8,14,16H,9-10H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP14 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50388917
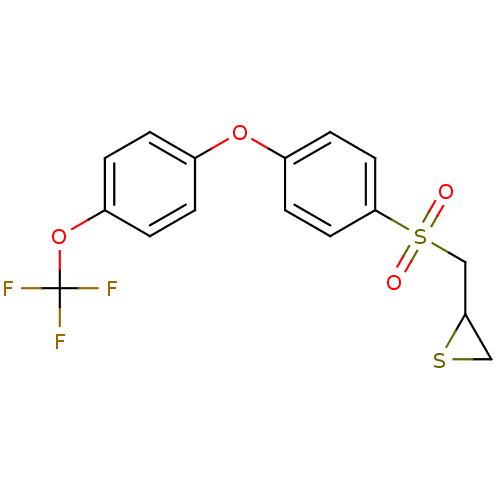
(CHEMBL2063277)Show SMILES FC(F)(F)Oc1ccc(Oc2ccc(cc2)S(=O)(=O)CC2CS2)cc1 Show InChI InChI=1S/C16H13F3O4S2/c17-16(18,19)23-13-3-1-11(2-4-13)22-12-5-7-15(8-6-12)25(20,21)10-14-9-24-14/h1-8,14H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP2 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM50459505

(CHEMBL4215451)Show SMILES Cl.NCc1ccc(Oc2ccc(cc2)S(=O)(=O)C[C@H]2CS2)cc1 |r| Show InChI InChI=1S/C16H17NO3S2.ClH/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15;/h1-8,15H,9-11,17H2;1H/t15-;/m1./s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 119 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Slow binding inhibition of human recombinant MMP14 catalytic domain using MOCAc-Pro-Leu-Gly-Leu-A2pr(Dnp)-Ala-Arg-NH2 substrate |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM389618

(US10357546, ND-336 | US9951035, ND-336 (5))Show InChI InChI=1S/C16H17NO3S2/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15/h1-8,15H,9-11,17H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Slow binding inhibition of human recombinant MMP14 catalytic domain using MOCAc-Pro-Leu-Gly-Leu-A2pr(Dnp)-Ala-Arg-NH2 substrate |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50459505

(CHEMBL4215451)Show SMILES Cl.NCc1ccc(Oc2ccc(cc2)S(=O)(=O)C[C@H]2CS2)cc1 |r| Show InChI InChI=1S/C16H17NO3S2.ClH/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15;/h1-8,15H,9-11,17H2;1H/t15-;/m1./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 127 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Slow binding inhibition of human recombinant MMP2 using MOCAc-Pro-Leu-Gly-Leu-A2pr(Dnp)-Ala-Arg-NH2 substrate |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM50388916
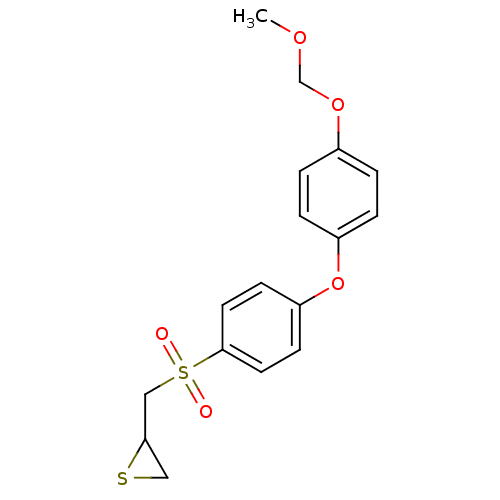
(CHEMBL2063276)Show InChI InChI=1S/C17H18O5S2/c1-20-12-21-13-2-4-14(5-3-13)22-15-6-8-17(9-7-15)24(18,19)11-16-10-23-16/h2-9,16H,10-12H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP14 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM50335495

(4-(4-(thiiran-2-ylmethylsulfonyl)phenoxy)phenyl me...)Show SMILES CS(=O)(=O)Oc1ccc(Oc2ccc(cc2)S(=O)(=O)CC2CS2)cc1 Show InChI InChI=1S/C16H16O6S3/c1-24(17,18)22-14-4-2-12(3-5-14)21-13-6-8-16(9-7-13)25(19,20)11-15-10-23-15/h2-9,15H,10-11H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 145 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP14 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM389618

(US10357546, ND-336 | US9951035, ND-336 (5))Show InChI InChI=1S/C16H17NO3S2/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15/h1-8,15H,9-11,17H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Slow binding inhibition of human recombinant MMP9 catalytic domain using MOCAc-Pro-Leu-Gly-Leu-A2pr(Dnp)-Ala-Arg-NH2 substrate |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50388914

(CHEMBL2063274 | US10357546, p-OH SB-3CT)Show InChI InChI=1S/C15H14O4S2/c16-11-1-3-12(4-2-11)19-13-5-7-15(8-6-13)21(17,18)10-14-9-20-14/h1-8,14,16H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP9 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50388916

(CHEMBL2063276)Show InChI InChI=1S/C17H18O5S2/c1-20-12-21-13-2-4-14(5-3-13)22-15-6-8-17(9-7-15)24(18,19)11-16-10-23-16/h2-9,16H,10-12H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP9 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50388914

(CHEMBL2063274 | US10357546, p-OH SB-3CT)Show InChI InChI=1S/C15H14O4S2/c16-11-1-3-12(4-2-11)19-13-5-7-15(8-6-13)21(17,18)10-14-9-20-14/h1-8,14,16H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human MMP9 expressed in human HeLa cells |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50388916

(CHEMBL2063276)Show InChI InChI=1S/C17H18O5S2/c1-20-12-21-13-2-4-14(5-3-13)22-15-6-8-17(9-7-15)24(18,19)11-16-10-23-16/h2-9,16H,10-12H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP2 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50459506

(CHEMBL4214929)Show SMILES Cl.NCc1ccc(Oc2ccc(cc2)S(=O)(=O)C[C@@H]2CS2)cc1 |r| Show InChI InChI=1S/C16H17NO3S2.ClH/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15;/h1-8,15H,9-11,17H2;1H/t15-;/m0./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Slow binding inhibition of human recombinant MMP9 catalytic domain using MOCAc-Pro-Leu-Gly-Leu-A2pr(Dnp)-Ala-Arg-NH2 substrate |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM50388910
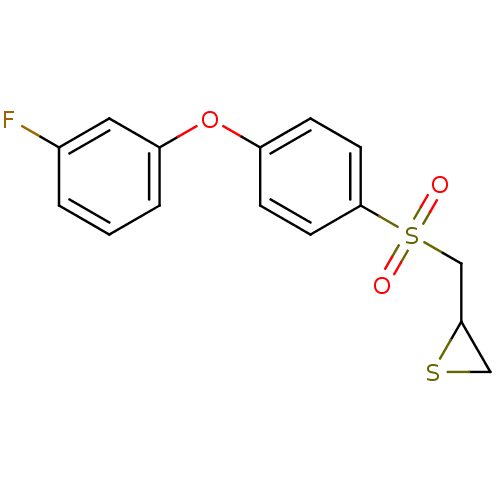
(CHEMBL2063271)Show InChI InChI=1S/C15H13FO3S2/c16-11-2-1-3-13(8-11)19-12-4-6-15(7-5-12)21(17,18)10-14-9-20-14/h1-8,14H,9-10H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP14 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM50388912

(CHEMBL2063273)Show InChI InChI=1S/C16H16O4S2/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15/h1-8,15,17H,9-11H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 215 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP14 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM50388915
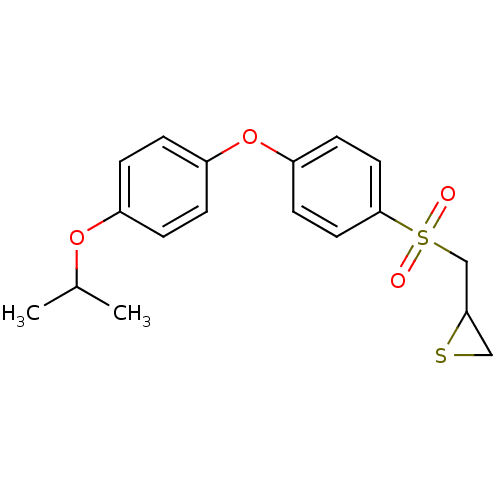
(CHEMBL2063275)Show InChI InChI=1S/C18H20O4S2/c1-13(2)21-14-3-5-15(6-4-14)22-16-7-9-18(10-8-16)24(19,20)12-17-11-23-17/h3-10,13,17H,11-12H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP14 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50388913
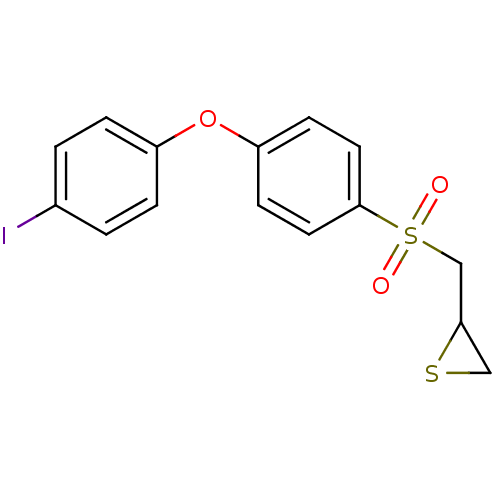
(CHEMBL2063272)Show InChI InChI=1S/C15H13IO3S2/c16-11-1-3-12(4-2-11)19-13-5-7-15(8-6-13)21(17,18)10-14-9-20-14/h1-8,14H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP9 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50388915

(CHEMBL2063275)Show InChI InChI=1S/C18H20O4S2/c1-13(2)21-14-3-5-15(6-4-14)22-16-7-9-18(10-8-16)24(19,20)12-17-11-23-17/h3-10,13,17H,11-12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP2 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Beta-hexosaminidase
(Pseudomonas aeruginosa) | BDBM50386412
(CHEMBL2047302)Show SMILES CC(=O)N[C@H]1CN[C@H](CO)[C@@H](O)[C@@H]1O |r| Show InChI InChI=1S/C8H16N2O4/c1-4(12)10-5-2-9-6(3-11)8(14)7(5)13/h5-9,11,13-14H,2-3H2,1H3,(H,10,12)/t5-,6+,7+,8+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of Pseudomonas aeruginosa PAO1 NagZ using PNP-GlcNAc as substrate assessed as release of 4-nitrophenolate ion after 4 mins by ... |
ACS Med Chem Lett 3: 238-242 (2012)
Article DOI: 10.1021/ml2002746
BindingDB Entry DOI: 10.7270/Q2KD1ZZJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50264809

(2-((4-phenoxyphenylsulfonyl)methyl)thiirane | CHEM...)Show InChI InChI=1S/C15H14O3S2/c16-20(17,11-14-10-19-14)15-8-6-13(7-9-15)18-12-4-2-1-3-5-12/h1-9,14H,10-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
| Assay Description
Inhibition assay using matrix metalloproteinase (MMP-2). |
Chem Biol Drug Des 74: 527-34 (2009)
Checked by Author
Article DOI: 10.1111/j.1747-0285.2009.00881.x
BindingDB Entry DOI: 10.7270/Q20P0XJV |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50388912

(CHEMBL2063273)Show InChI InChI=1S/C16H16O4S2/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15/h1-8,15,17H,9-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP9 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50264809

(2-((4-phenoxyphenylsulfonyl)methyl)thiirane | CHEM...)Show InChI InChI=1S/C15H14O3S2/c16-20(17,11-14-10-19-14)15-8-6-13(7-9-15)18-12-4-2-1-3-5-12/h1-9,14H,10-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human MMP9 |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50388910

(CHEMBL2063271)Show InChI InChI=1S/C15H13FO3S2/c16-11-2-1-3-13(8-11)19-12-4-6-15(7-5-12)21(17,18)10-14-9-20-14/h1-8,14H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP2 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50388915

(CHEMBL2063275)Show InChI InChI=1S/C18H20O4S2/c1-13(2)21-14-3-5-15(6-4-14)22-16-7-9-18(10-8-16)24(19,20)12-17-11-23-17/h3-10,13,17H,11-12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP9 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50335495

(4-(4-(thiiran-2-ylmethylsulfonyl)phenoxy)phenyl me...)Show SMILES CS(=O)(=O)Oc1ccc(Oc2ccc(cc2)S(=O)(=O)CC2CS2)cc1 Show InChI InChI=1S/C16H16O6S3/c1-24(17,18)22-14-4-2-12(3-5-14)21-13-6-8-16(9-7-13)25(19,20)11-15-10-23-15/h2-9,15H,10-11H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 575 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP3 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-14
(Homo sapiens (Human)) | BDBM50388911

(CHEMBL2063270)Show InChI InChI=1S/C15H13FO3S2/c16-11-1-3-12(4-2-11)19-13-5-7-15(8-6-13)21(17,18)10-14-9-20-14/h1-8,14H,9-10H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP14 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50264809

(2-((4-phenoxyphenylsulfonyl)methyl)thiirane | CHEM...)Show InChI InChI=1S/C15H14O3S2/c16-20(17,11-14-10-19-14)15-8-6-13(7-9-15)18-12-4-2-1-3-5-12/h1-9,14H,10-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP9 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM50388913

(CHEMBL2063272)Show InChI InChI=1S/C15H13IO3S2/c16-11-1-3-12(4-2-11)19-13-5-7-15(8-6-13)21(17,18)10-14-9-20-14/h1-8,14H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP2 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50388917

(CHEMBL2063277)Show SMILES FC(F)(F)Oc1ccc(Oc2ccc(cc2)S(=O)(=O)CC2CS2)cc1 Show InChI InChI=1S/C16H13F3O4S2/c17-16(18,19)23-13-3-1-11(2-4-13)22-12-5-7-15(8-6-12)25(20,21)10-14-9-24-14/h1-8,14H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 930 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP9 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM82121

(Gama, gama-dimethyl derivative, 6)Show InChI InChI=1S/C17H18O3S2/c1-17(2)16(21-17)12-22(18,19)15-10-8-14(9-11-15)20-13-6-4-3-5-7-13/h3-11,16H,12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
| Assay Description
Inhibition assay using matrix metalloproteinase (MMP-2). |
Chem Biol Drug Des 74: 527-34 (2009)
Checked by Author
Article DOI: 10.1111/j.1747-0285.2009.00881.x
BindingDB Entry DOI: 10.7270/Q20P0XJV |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM82119

(Sulfoxide, 4)Show InChI InChI=1S/C15H14O2S2/c16-19(11-14-10-18-14)15-8-6-13(7-9-15)17-12-4-2-1-3-5-12/h1-9,14H,10-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
| Assay Description
Inhibition assay using matrix metalloproteinase (MMP-2). |
Chem Biol Drug Des 74: 527-34 (2009)
Checked by Author
Article DOI: 10.1111/j.1747-0285.2009.00881.x
BindingDB Entry DOI: 10.7270/Q20P0XJV |
More data for this
Ligand-Target Pair | |
Neutrophil collagenase
(Homo sapiens (Human)) | BDBM50264809

(2-((4-phenoxyphenylsulfonyl)methyl)thiirane | CHEM...)Show InChI InChI=1S/C15H14O3S2/c16-20(17,11-14-10-19-14)15-8-6-13(7-9-15)18-12-4-2-1-3-5-12/h1-9,14H,10-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Non-competitive inhibition of MMP8 (unknown origin) |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
Neutrophil collagenase
(Homo sapiens (Human)) | BDBM50459506

(CHEMBL4214929)Show SMILES Cl.NCc1ccc(Oc2ccc(cc2)S(=O)(=O)C[C@@H]2CS2)cc1 |r| Show InChI InChI=1S/C16H17NO3S2.ClH/c17-9-12-1-3-13(4-2-12)20-14-5-7-16(8-6-14)22(18,19)11-15-10-21-15;/h1-8,15H,9-11,17H2;1H/t15-;/m0./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Notre Dame
Curated by ChEMBL
| Assay Description
Non-competitive inhibition of human recombinant MMP8 catalytic domain using Mca-Lys-Pro-Leu-Gly-Leu-Dpa-Ala-Arg-NH2 substrate |
J Med Chem 61: 8825-8837 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01005
BindingDB Entry DOI: 10.7270/Q2BZ68PX |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50388914

(CHEMBL2063274 | US10357546, p-OH SB-3CT)Show InChI InChI=1S/C15H14O4S2/c16-11-1-3-12(4-2-11)19-13-5-7-15(8-6-13)21(17,18)10-14-9-20-14/h1-8,14,16H,9-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Competitive inhibition of human MMP3 using fluorogenic substrate by Dixon plot analysis |
ACS Med Chem Lett 3: 490-495 (2012)
Article DOI: 10.1021/ml300050b
BindingDB Entry DOI: 10.7270/Q2T43V4Q |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data