

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Rattus norvegicus (Rat)) | BDBM50143314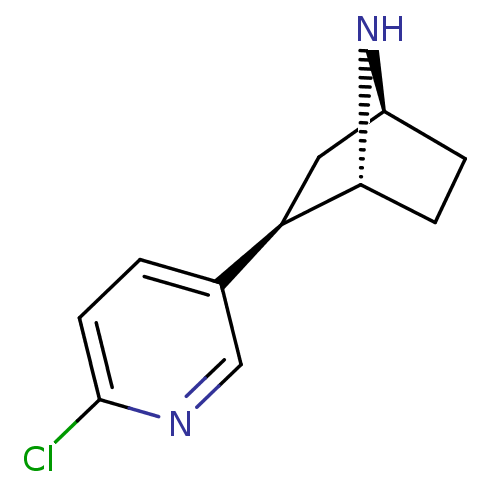 ((+)-Epibatidine | (-)-epibatidine | (1R,2R,4S)-2-(...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Sassari Curated by ChEMBL | Assay Description Displacement of [3H]epibatidine from alpha4beta2 nAChR in rat cerebral cortex membrane | Eur J Med Chem 180: 51-61 (2019) Article DOI: 10.1016/j.ejmech.2019.06.079 BindingDB Entry DOI: 10.7270/Q24Q7ZBT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,R541K,D544E,Q553K,L573M] (Human immunodeficiency virus) | BDBM112660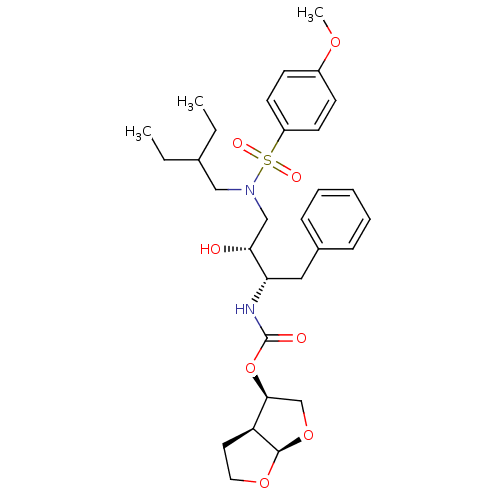 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.000200 | -72.5 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,R541K,D544E,Q553K,L573M] (Human immunodeficiency virus) | BDBM112661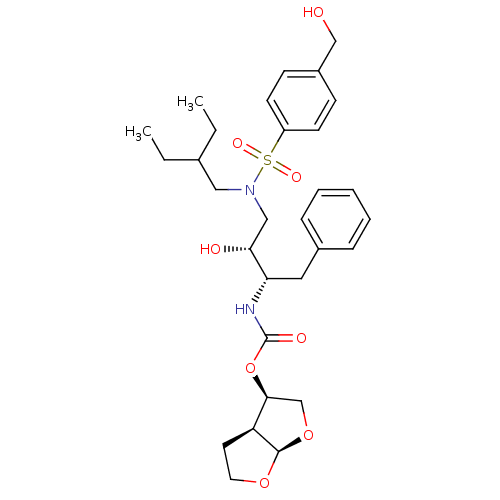 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.000500 | -70.2 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,R541K,D544E,Q553K,L573M] (Human immunodeficiency virus) | BDBM112656 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.000500 | -70.2 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM50423789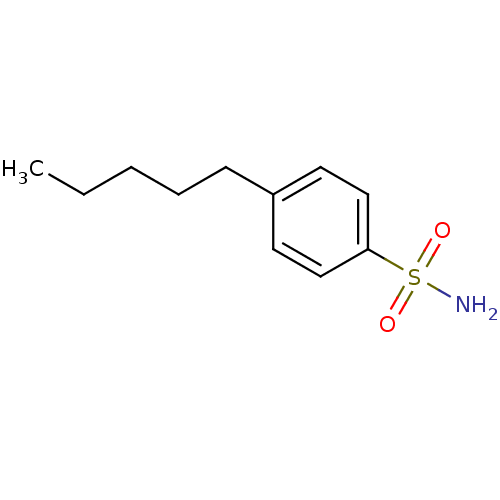 (4-Pentyl-Benzenesulfonamide | 4-Pentylbenzenesulfo...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | 0.000800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Tokushima Graduate School Curated by ChEMBL | Assay Description Binding affinity to human carbonic anhydrase 2 | Bioorg Med Chem Lett 21: 141-4 (2010) Article DOI: 10.1016/j.bmcl.2010.11.050 BindingDB Entry DOI: 10.7270/Q2K938T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM50423789 (4-Pentyl-Benzenesulfonamide | 4-Pentylbenzenesulfo...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | 0.000800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Tokushima Graduate School Curated by ChEMBL | Assay Description Binding affinity to human carbonic anhydrase 2 | Bioorg Med Chem Lett 21: 141-4 (2010) Article DOI: 10.1016/j.bmcl.2010.11.050 BindingDB Entry DOI: 10.7270/Q2K938T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,R541K,D544E,Q553K,L573M] (Human immunodeficiency virus) | BDBM112662 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.000800 | -69.0 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,R541K,D544E,Q553K,L573M] (Human immunodeficiency virus) | BDBM112657 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.000900 | -68.8 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I497V,V499I,E519D,G532V,I538V,R541K,D544E,L547P,Q553K,V566A,L573M] (Human immunodeficiency virus) | BDBM112660 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | Article PubMed | 0.00100 | -68.5 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,R541K,D544E,Q553K,L573M] (Human immunodeficiency virus) | BDBM112663 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.00150 | -67.5 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Enoyl-acyl-carrier protein reductase (Plasmodium falciparum) | BDBM8726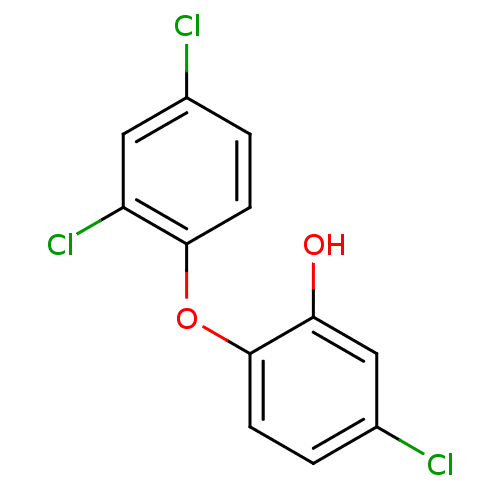 (5-chloro-2-(2,4-dichlorophenoxy)phenol | CHEMBL849...) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 0.00190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Immunology Curated by ChEMBL | Assay Description Inhibition of Plasmodium falciparum ENR in presence of EGCG by dilution assay | J Med Chem 50: 765-75 (2007) Article DOI: 10.1021/jm061154d BindingDB Entry DOI: 10.7270/Q2QJ7J4Q | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| 5'-methylthioadenosine/S-adenosylhomocysteine nucleosidase (Escherichia coli) | BDBM22113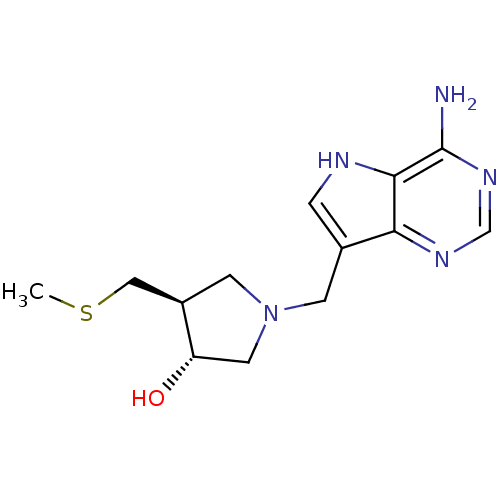 ((3R,4S)-1-({4-amino-5H-pyrrolo[3,2-d]pyrimidin-7-y...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | Purchase DrugBank MCE MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.00200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Victoria University of Wellington Curated by ChEMBL | Assay Description Inhibition of recombinant Escherichia coli MTAN expressed in Escherichia coli BL-21 DE3 using methylthioadenosine as substrate assessed as inhibition... | Bioorg Med Chem 23: 5326-33 (2015) Article DOI: 10.1016/j.bmc.2015.07.059 BindingDB Entry DOI: 10.7270/Q2HX1FF5 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I497V,V499I,E519D,R541K,D544E,L547P,Q553K,A555V,G557S,I568V,L573M,L574M] (Human immunodeficiency virus) | BDBM112656 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.00400 | -65.1 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Endothelin-1 receptor (Homo sapiens (Human)) | BDBM50105033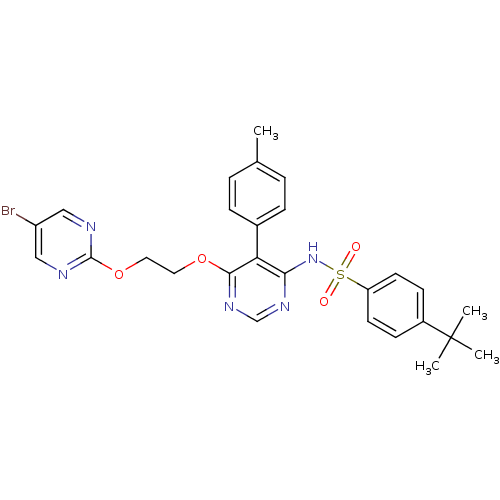 (CHEMBL112531 | N-{6-[2-(5-Bromo-pyrimidin-2-yloxy)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.00420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tanabe Seiyaku Co., Ltd. Curated by ChEMBL | Assay Description Ability to inhibit [125I]ET1 binding to human cloned endothelin A receptor expressed on CHO cells | J Med Chem 44: 3369-77 (2001) BindingDB Entry DOI: 10.7270/Q27M08P8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dimer of Gag-Pol polyprotein [501-599,Q508K,L534I,L564I,C568A,C596A] (Human immunodeficiency virus type 1) | BDBM13925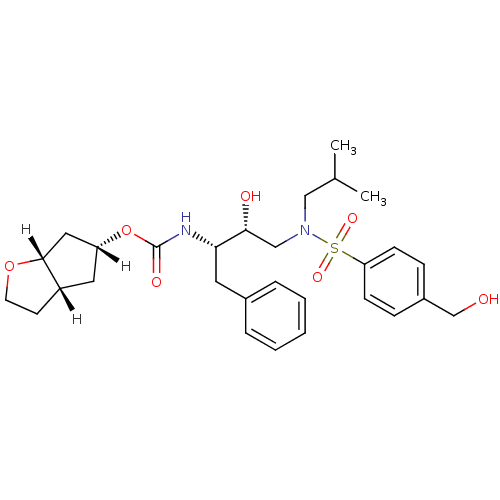 ((3aS,5R,6aR)-hexahydro-2H-cyclopenta[b]furan-5-yl ...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 0.00450 | -64.8 | n/a | n/a | n/a | n/a | n/a | 6.4 | 25 |
Purdue University | Assay Description The Ki values were determined by substrate cleavage assay using fluorogenic substrate, 2-(aminobenzoyl)-Thr-Ile-Nle-Phe(p-NO2)-Gln-Arg-NH2. A standar... | J Med Chem 49: 5252-61 (2006) Article DOI: 10.1021/jm060561m BindingDB Entry DOI: 10.7270/Q23R0R41 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I497V,V499I,E519D,G532V,I538V,R541K,D544E,L547P,Q553K,V566A,L573M] (Human immunodeficiency virus) | BDBM112655 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | Article PubMed | 0.00500 | -64.5 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dimer of Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM194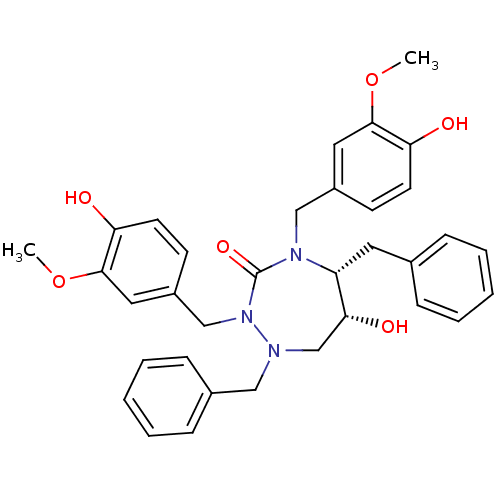 ((5R,6R)-1,5-dibenzyl-6-hydroxy-2,4-bis[(4-hydroxy-...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.00500 | -65.6 | n/a | n/a | n/a | n/a | n/a | 4.7 | 30 |
Abbott Laboratories | Assay Description HIV-1 protease activity was measured by a continuous fluorometric assay using the internally quenched fluorogenic substrate DABCYL-GABA-Ser-Gln-Tyr-P... | J Med Chem 39: 392-7 (1996) Article DOI: 10.1021/jm9507183 BindingDB Entry DOI: 10.7270/Q2V40SC6 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,R541K,D544E,Q553K,L573M] (Human immunodeficiency virus) | BDBM8125 ((3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl N-[(2S...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 0.00500 | -64.5 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,R541K,D544E,Q553K,L573M] (Human immunodeficiency virus) | BDBM112659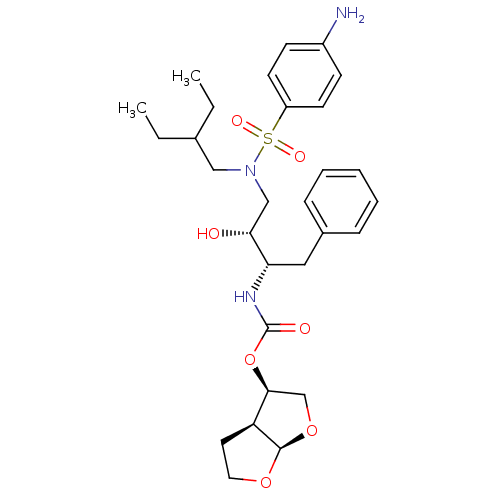 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.00500 | -64.5 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50229232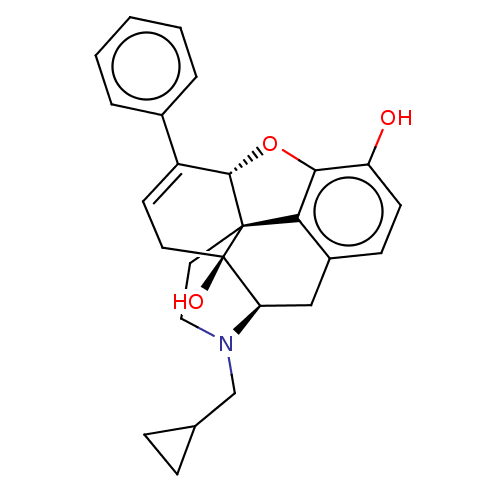 (CHEMBL610527) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota Curated by ChEMBL | Assay Description Compound was evaluated for the binding affinity to Opioid receptor mu using [3H]DAMGO as radioligand in guinea pig brain membrane | J Med Chem 34: 1715-20 (1991) BindingDB Entry DOI: 10.7270/Q2T43TPG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protease (Human immunodeficiency virus 1 (HIV-1)) | BDBM112661 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | Article PubMed | 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Massachusetts Medical School Curated by ChEMBL | Assay Description Inhibition of HIV1 NL4-3 protease I84V mutant expressed in Escherichia coli TAP-106 cells using EDANS/DABCYL-labelled 10-amino acid containing natura... | J Med Chem 62: 8062-8079 (2019) Article DOI: 10.1021/acs.jmedchem.9b00838 BindingDB Entry DOI: 10.7270/Q26H4MQ1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM50423788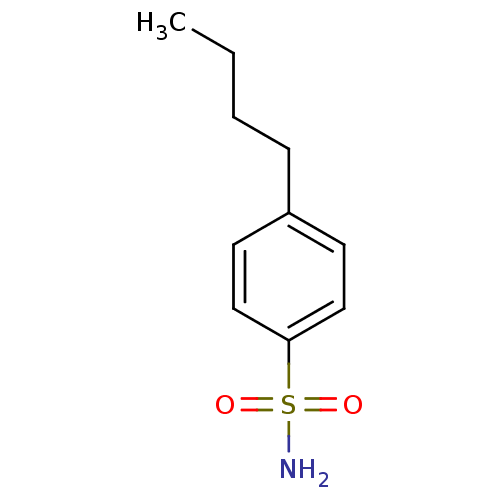 (4-Butyl-Benzenesulfonamide | 4-Butylbenzenesulfona...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Tokushima Graduate School Curated by ChEMBL | Assay Description Binding affinity to human carbonic anhydrase 2 | Bioorg Med Chem Lett 21: 141-4 (2010) Article DOI: 10.1016/j.bmcl.2010.11.050 BindingDB Entry DOI: 10.7270/Q2K938T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I497V,V499I,E519D,G532V,I538V,R541K,D544E,L547P,Q553K,V566A,L573M] (Human immunodeficiency virus) | BDBM112661 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | Article PubMed | 0.00500 | -64.5 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM50423788 (4-Butyl-Benzenesulfonamide | 4-Butylbenzenesulfona...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid PDB UniChem Similars | Article PubMed | 0.00500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Tokushima Graduate School Curated by ChEMBL | Assay Description Binding affinity to human carbonic anhydrase 2 | Bioorg Med Chem Lett 21: 141-4 (2010) Article DOI: 10.1016/j.bmcl.2010.11.050 BindingDB Entry DOI: 10.7270/Q2K938T6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,I534V,R541K,D544E,Q553K,A555V,L573M] (Human immunodeficiency virus) | BDBM112654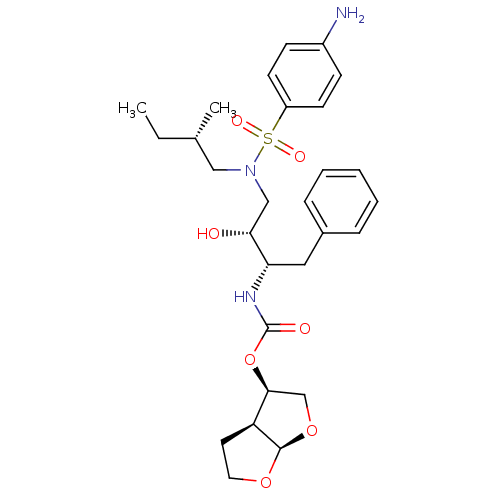 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.00600 | -64.1 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,I534V,R541K,D544E,Q553K,A555V,L573M] (Human immunodeficiency virus) | BDBM112662 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.00600 | -64.1 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| M17 leucyl aminopeptidase (Plasmodium falciparum 3D7) | BDBM50497553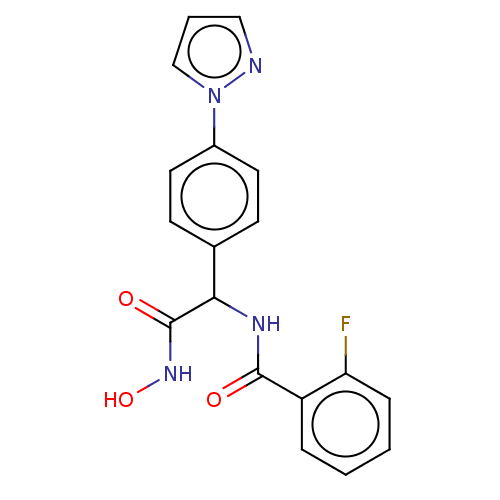 (CHEMBL3359696) | PDB MMDB KEGG UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Monash University Curated by ChEMBL | Assay Description Inhibition of Plasmodium falciparum M17 incubated for 10 mins before H-Leu-NHMec substrate addition by fluorescence assay | J Med Chem 57: 9168-83 (2014) Article DOI: 10.1021/jm501323a BindingDB Entry DOI: 10.7270/Q2QN69RJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| B1 bradykinin receptor (Homo sapiens (Human)) | BDBM50156449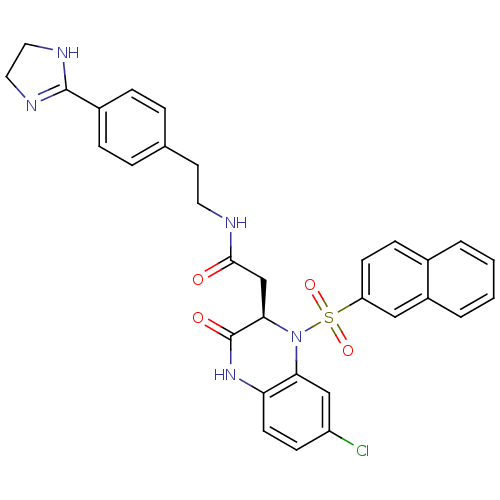 (2-[(R)-7-Chloro-1-(naphthalene-2-sulfonyl)-3-oxo-1...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by ChEMBL | Assay Description Binding affinity to human Bradykinin receptor B1 over-expressed in transgenic rats was determined by ex vivo receptor occupancy assay | Bioorg Med Chem Lett 14: 6045-8 (2004) Article DOI: 10.1016/j.bmcl.2004.09.074 BindingDB Entry DOI: 10.7270/Q2V1248Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuropeptide Y receptor type 4 (Homo sapiens (Human)) | BDBM50466554 (CHEMBL4286615) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Displacement of [125I]-PP from human Y4R expressed in CHO-K1 cell membranes after 2 hrs by scintillation proximity assay | J Med Chem 61: 10519-10530 (2018) Article DOI: 10.1021/acs.jmedchem.8b01046 BindingDB Entry DOI: 10.7270/Q2ZC85JC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuropeptide Y receptor type 4 (Homo sapiens (Human)) | BDBM50466543 (CHEMBL4276961) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Displacement of [125I]-PP from human Y4R expressed in CHO-K1 cell membranes after 2 hrs by scintillation proximity assay | J Med Chem 61: 10519-10530 (2018) Article DOI: 10.1021/acs.jmedchem.8b01046 BindingDB Entry DOI: 10.7270/Q2ZC85JC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuropeptide Y receptor type 4 (Homo sapiens (Human)) | BDBM50466554 (CHEMBL4286615) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Displacement of [125I]-PP from human Y4R expressed in CHO-K1 cell membranes after 2 hrs by scintillation proximity assay | J Med Chem 61: 10519-10530 (2018) Article DOI: 10.1021/acs.jmedchem.8b01046 BindingDB Entry DOI: 10.7270/Q2ZC85JC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuropeptide Y receptor type 4 (Homo sapiens (Human)) | BDBM50466543 (CHEMBL4276961) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Displacement of [125I]-PP from human Y4R expressed in CHO-K1 cell membranes after 2 hrs by scintillation proximity assay | J Med Chem 61: 10519-10530 (2018) Article DOI: 10.1021/acs.jmedchem.8b01046 BindingDB Entry DOI: 10.7270/Q2ZC85JC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor subunit alpha-4/beta-2 (Rattus norvegicus (Rat)) | BDBM50515333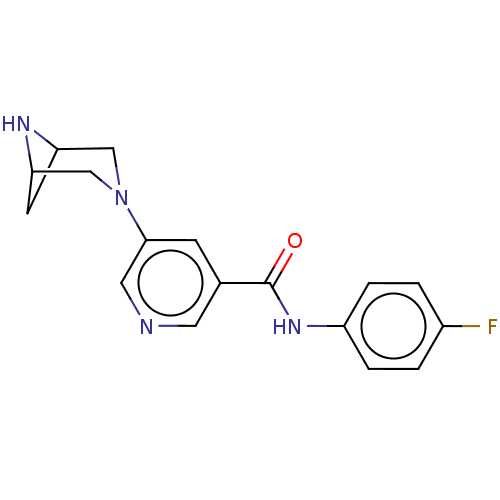 (CHEMBL4460815) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Sassari Curated by ChEMBL | Assay Description Displacement of [3H]epibatidine from alpha4beta2 nAChR in rat cerebral cortex membrane | Eur J Med Chem 180: 51-61 (2019) Article DOI: 10.1016/j.ejmech.2019.06.079 BindingDB Entry DOI: 10.7270/Q24Q7ZBT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Cavia porcellus (domestic guinea pig)) | BDBM50229232 (CHEMBL610527) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota Curated by ChEMBL | Assay Description Compound was evaluated for the binding affinity to opioid receptor kappa using [3H]-EK as radioligand in guinea pig brain membrane | J Med Chem 34: 1715-20 (1991) BindingDB Entry DOI: 10.7270/Q2T43TPG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Calcitonin gene-related peptide type 1 receptor (Homo sapiens (Human)) | BDBM50273292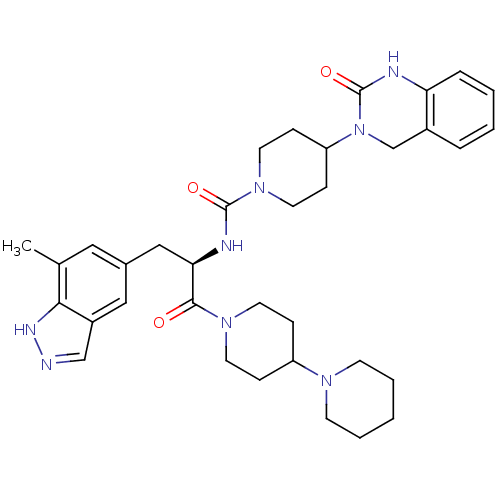 ((R)-N-(1-(1,4'-bipiperidin-1'-yl)-3-(7-methyl-1H-i...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research & Development Curated by ChEMBL | Assay Description Displacement of [I125]CGRP from human CGRP receptor in SK-N-MC cells | J Med Chem 51: 4858-61 (2008) Article DOI: 10.1021/jm800546t BindingDB Entry DOI: 10.7270/Q2N016BV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protease (Human immunodeficiency virus 1 (HIV-1)) | BDBM112656 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | Article PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Massachusetts Medical School Curated by ChEMBL | Assay Description Inhibition of HIV1 NL4-3 protease I84V mutant expressed in Escherichia coli TAP-106 cells using EDANS/DABCYL-labelled 10-amino acid containing natura... | J Med Chem 62: 8062-8079 (2019) Article DOI: 10.1021/acs.jmedchem.9b00838 BindingDB Entry DOI: 10.7270/Q26H4MQ1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Somatostatin receptor type 2 (Homo sapiens (Human)) | BDBM85357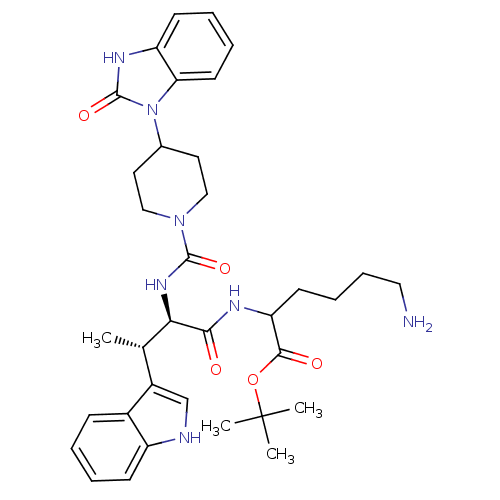 (2-[[(2R,3S)-2-[[4-[(2-Oxo-2,3-dihydro-1H-benzimida...) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories Curated by PDSP Ki Database | Proc Natl Acad Sci U S A 95: 10836-41 (1998) Article DOI: 10.1073/pnas.95.18.10836 BindingDB Entry DOI: 10.7270/Q2XW4HCM | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,I534V,R541K,D544E,Q553K,A555V,L573M] (Human immunodeficiency virus) | BDBM112660 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.0100 | -62.8 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| M17 leucyl aminopeptidase (Plasmodium falciparum 3D7) | BDBM50497551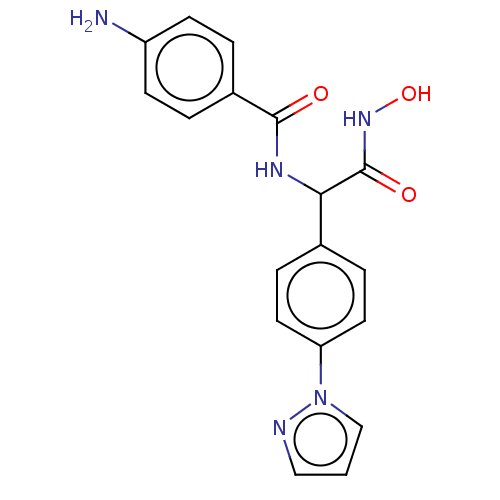 (CHEMBL3359700) | PDB MMDB KEGG UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem | PDB Article PubMed | 0.0110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Monash University Curated by ChEMBL | Assay Description Inhibition of Plasmodium falciparum M17 incubated for 10 mins before H-Leu-NHMec substrate addition by fluorescence assay | J Med Chem 57: 9168-83 (2014) Article DOI: 10.1021/jm501323a BindingDB Entry DOI: 10.7270/Q2QN69RJ | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM476758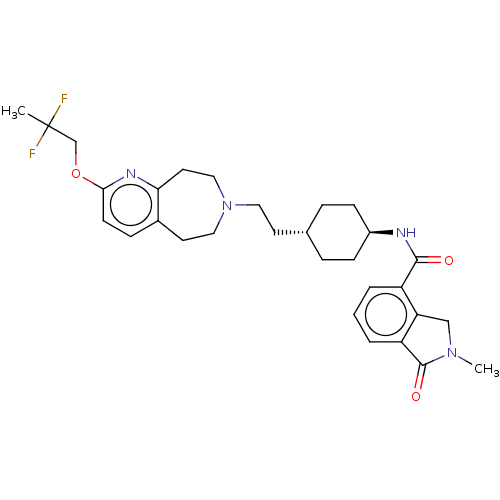 (US10870660, Compound III-024 | US11345716, Compoun...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.0120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SHIONOGI & CO., LTD. US Patent | Assay Description 225 nL of the solutions of the non-specific ligand or the compounds of the present invention at each concentration (in case of vehicle, final concent... | US Patent US10870660 (2020) BindingDB Entry DOI: 10.7270/Q2M61PBD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I497V,V499I,E519D,G532V,I538V,R541K,D544E,L547P,Q553K,V566A,L573M] (Human immunodeficiency virus) | BDBM112658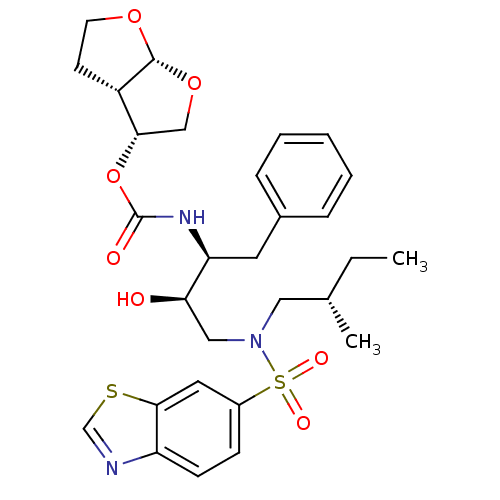 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | Article PubMed | 0.0120 | -62.3 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| M17 leucyl aminopeptidase (Plasmodium falciparum 3D7) | BDBM50497552 (CHEMBL3359698) | PDB MMDB KEGG UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Monash University Curated by ChEMBL | Assay Description Inhibition of Plasmodium falciparum M17 incubated for 10 mins before H-Leu-NHMec substrate addition by fluorescence assay | J Med Chem 57: 9168-83 (2014) Article DOI: 10.1021/jm501323a BindingDB Entry DOI: 10.7270/Q2QN69RJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Calcitonin gene-related peptide type 1 receptor (Homo sapiens (Human)) | BDBM50268484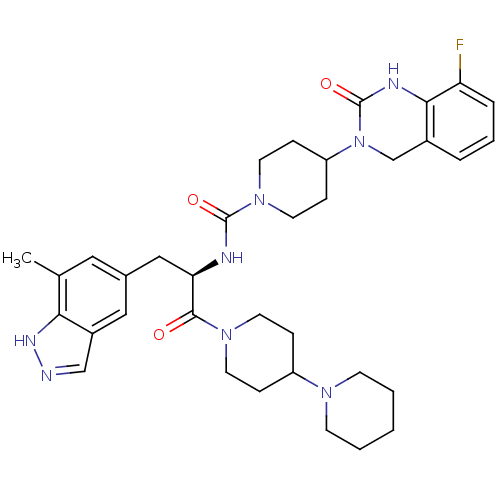 ((R)-4-(8-Fluoro-2-oxo-1,2-dihydroquinazolin-3(4H)-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0128 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research & Development Curated by ChEMBL | Assay Description Displacement of [I125]CGRP from human CGRP receptor in SK-N-MC cells | J Med Chem 51: 4858-61 (2008) Article DOI: 10.1021/jm800546t BindingDB Entry DOI: 10.7270/Q2N016BV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM82552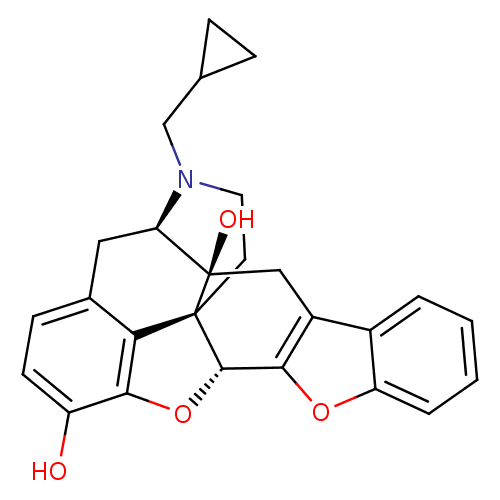 (CAS_111555-58-9 | NTB | naltrindolebenzofuran) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid PDB UniChem Similars | PubMed | 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota Curated by ChEMBL | Assay Description Compound was evaluated for the binding affinity to opioid receptor delta using [3H]DADLE as radioligand in guinea pig brain membrane | J Med Chem 34: 1715-20 (1991) BindingDB Entry DOI: 10.7270/Q2T43TPG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I494L,I497V,V499I,E519D,I534V,R541K,D544E,Q553K,A555V,L573M] (Human immunodeficiency virus) | BDBM112656 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.0130 | -62.1 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Gag-Pol polyprotein [484-582,Q491K,I497V,V499I,E519D,R541K,D544E,L547P,Q553K,A555V,G557S,I568V,L573M,L574M] (Human immunodeficiency virus) | BDBM112654 ((3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-yl ((2S,3...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.0130 | -62.1 | n/a | n/a | n/a | n/a | n/a | 4.7 | 25 |
University of Massachusetts Medical School, Worcester, MA 01605, USA | Assay Description The reaction mixture contained 2 µL of protease and 2 µL of inhibitor (or DMSO as a control) and was incubated for 20-30 min at room temper... | Chem Biol 20: 1116-24 (2013) Article DOI: 10.1016/j.chembiol.2013.07.014 BindingDB Entry DOI: 10.7270/Q2HQ3XKF | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Melatonin receptor type 1A (Homo sapiens (Human)) | BDBM50118470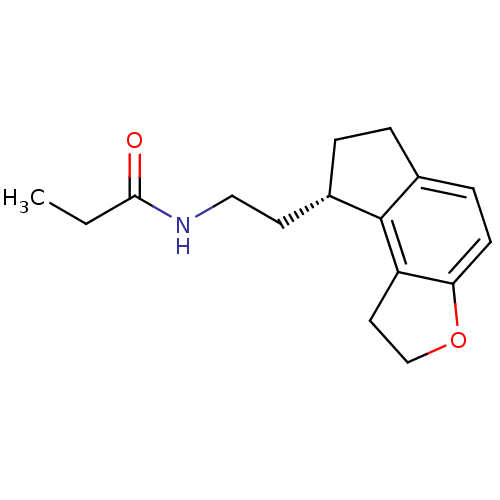 (CHEMBL1218 | N-[2-(1,6,7,8-Tetrahydro-2H-3-oxa-as-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB PubMed | 0.0138 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Chemical Industries, Ltd. Curated by ChEMBL | Assay Description Binding affinity against human Melatonin receptor type 1A (MT1) | J Med Chem 45: 4222-39 (2002) BindingDB Entry DOI: 10.7270/Q2D799S6 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Calcitonin gene-related peptide type 1 receptor (Homo sapiens (Human)) | BDBM50576175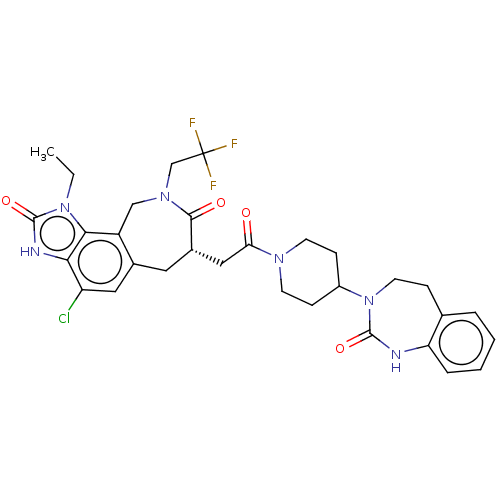 (CHEMBL4859941) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Displacement of [125I]CGRP from human CGRP receptor in human SK-N-MC cells measured after 2 hrs by scintillation counting analysis | Citation and Details Article DOI: 10.1016/j.bmcl.2021.128077 BindingDB Entry DOI: 10.7270/Q23N276F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dimer of Gag-Pol polyprotein [501-599,Q508K,L534I,L564I,C568A,C596A] (Human immunodeficiency virus type 1) | BDBM8125 ((3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl N-[(2S...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 0.0140 | -62.0 | n/a | n/a | n/a | n/a | n/a | 6.4 | 25 |
Purdue University | Assay Description The Ki values were determined by substrate cleavage assay using fluorogenic substrate, 2-(aminobenzoyl)-Thr-Ile-Nle-Phe(p-NO2)-Gln-Arg-NH2. A standar... | J Med Chem 49: 5252-61 (2006) Article DOI: 10.1021/jm060561m BindingDB Entry DOI: 10.7270/Q23R0R41 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| D(3) dopamine receptor (Homo sapiens (Human)) | BDBM476918 (US10870660, Compound II-057) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.0140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SHIONOGI & CO., LTD. US Patent | Assay Description 225 nL of the solutions of the non-specific ligand or the compounds of the present invention at each concentration (in case of vehicle, final concent... | US Patent US10870660 (2020) BindingDB Entry DOI: 10.7270/Q2M61PBD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 150112 total ) | Next | Last >> |