 Found 16 hits with Last Name = 'meininger' and Initial = 'd'
Found 16 hits with Last Name = 'meininger' and Initial = 'd' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
11-beta-hydroxysteroid dehydrogenase 1
(Homo sapiens (Human)) | BDBM50248569

((1-(((R)-3-methyl-4-(4-((S)-1,1,1-trifluoro-2-hydr...)Show SMILES C[C@@H]1CN(CC2(CC2)C(N)=O)CCN1S(=O)(=O)c1ccc(cc1)[C@](C)(O)C(F)(F)F |r| Show InChI InChI=1S/C19H26F3N3O4S/c1-13-11-24(12-18(7-8-18)16(23)26)9-10-25(13)30(28,29)15-5-3-14(4-6-15)17(2,27)19(20,21)22/h3-6,13,27H,7-12H2,1-2H3,(H2,23,26)/t13-,17+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human 11beta-HSD1 using variable cofactor NADPH concentration by Lineweaver burk plot |
Bioorg Med Chem 16: 8922-31 (2008)
Article DOI: 10.1016/j.bmc.2008.08.065
BindingDB Entry DOI: 10.7270/Q22F7N8T |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
11-beta-hydroxysteroid dehydrogenase 1
(Homo sapiens (Human)) | BDBM50248569

((1-(((R)-3-methyl-4-(4-((S)-1,1,1-trifluoro-2-hydr...)Show SMILES C[C@@H]1CN(CC2(CC2)C(N)=O)CCN1S(=O)(=O)c1ccc(cc1)[C@](C)(O)C(F)(F)F |r| Show InChI InChI=1S/C19H26F3N3O4S/c1-13-11-24(12-18(7-8-18)16(23)26)9-10-25(13)30(28,29)15-5-3-14(4-6-15)17(2,27)19(20,21)22/h3-6,13,27H,7-12H2,1-2H3,(H2,23,26)/t13-,17+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human 11beta-HSD1 using variable substrate cortisol concentration by Lineweaver burk plot |
Bioorg Med Chem 16: 8922-31 (2008)
Article DOI: 10.1016/j.bmc.2008.08.065
BindingDB Entry DOI: 10.7270/Q22F7N8T |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
4-hydroxy-tetrahydrodipicolinate reductase
(Escherichia coli) | BDBM59098
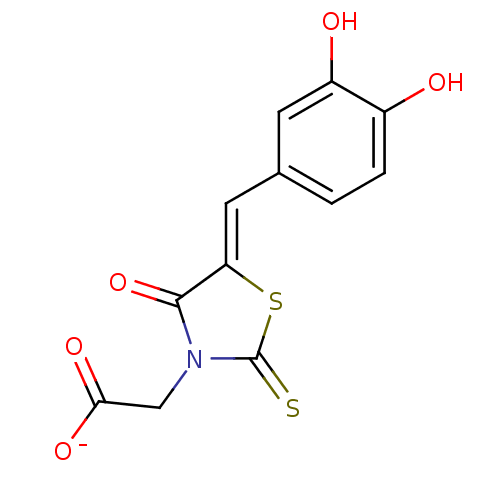
(Bi-ligand, 1)Show InChI InChI=1S/C12H9NO5S2/c14-7-2-1-6(3-8(7)15)4-9-11(18)13(5-10(16)17)12(19)20-9/h1-4,14-15H,5H2,(H,16,17)/p-1/b9-4- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 26 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc
| Assay Description
All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. |
Chem Biol 11: 185-94 (2004)
Article DOI: 10.1016/j.chembiol.2004.02.012
BindingDB Entry DOI: 10.7270/Q2K9360M |
More data for this
Ligand-Target Pair | |
Quinone-dependent D-lactate dehydrogenase
(Escherichia coli) | BDBM59099
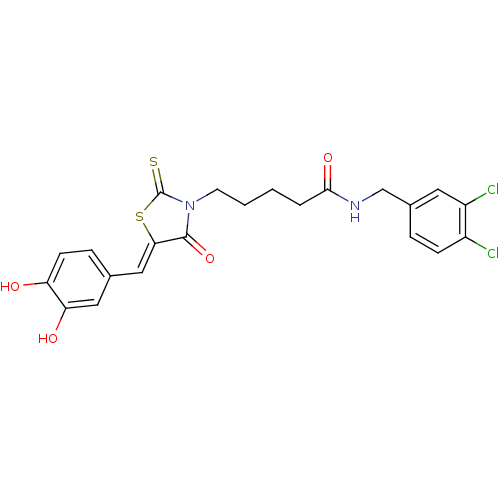
(Bi-ligand, 2)Show SMILES Oc1ccc(\C=C2/SC(=S)N(CCCCC(=O)NCc3ccc(Cl)c(Cl)c3)C2=O)cc1O Show InChI InChI=1S/C22H20Cl2N2O4S2/c23-15-6-4-14(9-16(15)24)12-25-20(29)3-1-2-8-26-21(30)19(32-22(26)31)11-13-5-7-17(27)18(28)10-13/h4-7,9-11,27-28H,1-3,8,12H2,(H,25,29)/b19-11- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 42 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc
| Assay Description
All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. |
Chem Biol 11: 185-94 (2004)
Article DOI: 10.1016/j.chembiol.2004.02.012
BindingDB Entry DOI: 10.7270/Q2K9360M |
More data for this
Ligand-Target Pair | |
4-hydroxy-tetrahydrodipicolinate reductase
(Escherichia coli) | BDBM59101
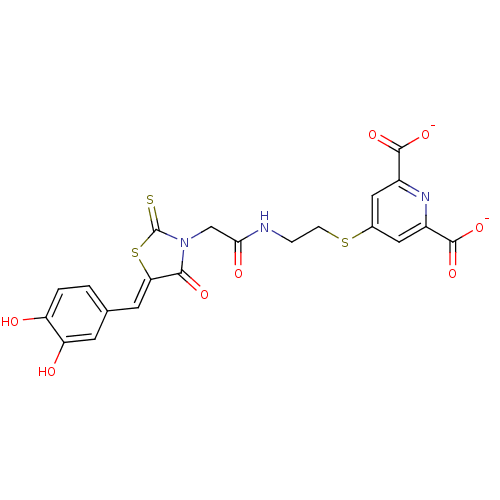
(Bi-ligand, 4)Show SMILES Oc1ccc(\C=C2/SC(=S)N(CC(=O)NCCSc3cc(nc(c3)C([O-])=O)C([O-])=O)C2=O)cc1O Show InChI InChI=1S/C21H17N3O8S3/c25-14-2-1-10(5-15(14)26)6-16-18(28)24(21(33)35-16)9-17(27)22-3-4-34-11-7-12(19(29)30)23-13(8-11)20(31)32/h1-2,5-8,25-26H,3-4,9H2,(H,22,27)(H,29,30)(H,31,32)/p-2/b16-6- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 100 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc
| Assay Description
All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. |
Chem Biol 11: 185-94 (2004)
Article DOI: 10.1016/j.chembiol.2004.02.012
BindingDB Entry DOI: 10.7270/Q2K9360M |
More data for this
Ligand-Target Pair | |
1-deoxy-D-xylulose 5-phosphate reductoisomerase
(Escherichia coli) | BDBM59100
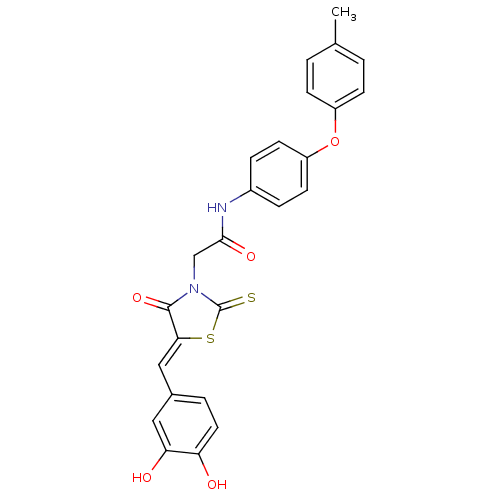
(Bi-ligand, 3)Show SMILES Cc1ccc(Oc2ccc(NC(=O)CN3C(=S)S\C(=C/c4ccc(O)c(O)c4)C3=O)cc2)cc1 Show InChI InChI=1S/C25H20N2O5S2/c1-15-2-7-18(8-3-15)32-19-9-5-17(6-10-19)26-23(30)14-27-24(31)22(34-25(27)33)13-16-4-11-20(28)21(29)12-16/h2-13,28-29H,14H2,1H3,(H,26,30)/b22-13- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 202 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc
| Assay Description
All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. |
Chem Biol 11: 185-94 (2004)
Article DOI: 10.1016/j.chembiol.2004.02.012
BindingDB Entry DOI: 10.7270/Q2K9360M |
More data for this
Ligand-Target Pair | |
Quinone-dependent D-lactate dehydrogenase
(Escherichia coli) | BDBM59101

(Bi-ligand, 4)Show SMILES Oc1ccc(\C=C2/SC(=S)N(CC(=O)NCCSc3cc(nc(c3)C([O-])=O)C([O-])=O)C2=O)cc1O Show InChI InChI=1S/C21H17N3O8S3/c25-14-2-1-10(5-15(14)26)6-16-18(28)24(21(33)35-16)9-17(27)22-3-4-34-11-7-12(19(29)30)23-13(8-11)20(31)32/h1-2,5-8,25-26H,3-4,9H2,(H,22,27)(H,29,30)(H,31,32)/p-2/b16-6- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 620 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc
| Assay Description
All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. |
Chem Biol 11: 185-94 (2004)
Article DOI: 10.1016/j.chembiol.2004.02.012
BindingDB Entry DOI: 10.7270/Q2K9360M |
More data for this
Ligand-Target Pair | |
1-deoxy-D-xylulose 5-phosphate reductoisomerase
(Escherichia coli) | BDBM59101

(Bi-ligand, 4)Show SMILES Oc1ccc(\C=C2/SC(=S)N(CC(=O)NCCSc3cc(nc(c3)C([O-])=O)C([O-])=O)C2=O)cc1O Show InChI InChI=1S/C21H17N3O8S3/c25-14-2-1-10(5-15(14)26)6-16-18(28)24(21(33)35-16)9-17(27)22-3-4-34-11-7-12(19(29)30)23-13(8-11)20(31)32/h1-2,5-8,25-26H,3-4,9H2,(H,22,27)(H,29,30)(H,31,32)/p-2/b16-6- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 7.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc
| Assay Description
All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. |
Chem Biol 11: 185-94 (2004)
Article DOI: 10.1016/j.chembiol.2004.02.012
BindingDB Entry DOI: 10.7270/Q2K9360M |
More data for this
Ligand-Target Pair | |
1-deoxy-D-xylulose 5-phosphate reductoisomerase
(Escherichia coli) | BDBM59099

(Bi-ligand, 2)Show SMILES Oc1ccc(\C=C2/SC(=S)N(CCCCC(=O)NCc3ccc(Cl)c(Cl)c3)C2=O)cc1O Show InChI InChI=1S/C22H20Cl2N2O4S2/c23-15-6-4-14(9-16(15)24)12-25-20(29)3-1-2-8-26-21(30)19(32-22(26)31)11-13-5-7-17(27)18(28)10-13/h4-7,9-11,27-28H,1-3,8,12H2,(H,25,29)/b19-11- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc
| Assay Description
All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. |
Chem Biol 11: 185-94 (2004)
Article DOI: 10.1016/j.chembiol.2004.02.012
BindingDB Entry DOI: 10.7270/Q2K9360M |
More data for this
Ligand-Target Pair | |
Quinone-dependent D-lactate dehydrogenase
(Escherichia coli) | BDBM59100

(Bi-ligand, 3)Show SMILES Cc1ccc(Oc2ccc(NC(=O)CN3C(=S)S\C(=C/c4ccc(O)c(O)c4)C3=O)cc2)cc1 Show InChI InChI=1S/C25H20N2O5S2/c1-15-2-7-18(8-3-15)32-19-9-5-17(6-10-19)26-23(30)14-27-24(31)22(34-25(27)33)13-16-4-11-20(28)21(29)12-16/h2-13,28-29H,14H2,1H3,(H,26,30)/b22-13- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc
| Assay Description
All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. |
Chem Biol 11: 185-94 (2004)
Article DOI: 10.1016/j.chembiol.2004.02.012
BindingDB Entry DOI: 10.7270/Q2K9360M |
More data for this
Ligand-Target Pair | |
4-hydroxy-tetrahydrodipicolinate reductase
(Escherichia coli) | BDBM59100

(Bi-ligand, 3)Show SMILES Cc1ccc(Oc2ccc(NC(=O)CN3C(=S)S\C(=C/c4ccc(O)c(O)c4)C3=O)cc2)cc1 Show InChI InChI=1S/C25H20N2O5S2/c1-15-2-7-18(8-3-15)32-19-9-5-17(6-10-19)26-23(30)14-27-24(31)22(34-25(27)33)13-16-4-11-20(28)21(29)12-16/h2-13,28-29H,14H2,1H3,(H,26,30)/b22-13- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >2.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc
| Assay Description
All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. |
Chem Biol 11: 185-94 (2004)
Article DOI: 10.1016/j.chembiol.2004.02.012
BindingDB Entry DOI: 10.7270/Q2K9360M |
More data for this
Ligand-Target Pair | |
4-hydroxy-tetrahydrodipicolinate reductase
(Escherichia coli) | BDBM59099

(Bi-ligand, 2)Show SMILES Oc1ccc(\C=C2/SC(=S)N(CCCCC(=O)NCc3ccc(Cl)c(Cl)c3)C2=O)cc1O Show InChI InChI=1S/C22H20Cl2N2O4S2/c23-15-6-4-14(9-16(15)24)12-25-20(29)3-1-2-8-26-21(30)19(32-22(26)31)11-13-5-7-17(27)18(28)10-13/h4-7,9-11,27-28H,1-3,8,12H2,(H,25,29)/b19-11- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc
| Assay Description
All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. |
Chem Biol 11: 185-94 (2004)
Article DOI: 10.1016/j.chembiol.2004.02.012
BindingDB Entry DOI: 10.7270/Q2K9360M |
More data for this
Ligand-Target Pair | |
1-deoxy-D-xylulose 5-phosphate reductoisomerase
(Escherichia coli) | BDBM59098

(Bi-ligand, 1)Show InChI InChI=1S/C12H9NO5S2/c14-7-2-1-6(3-8(7)15)4-9-11(18)13(5-10(16)17)12(19)20-9/h1-4,14-15H,5H2,(H,16,17)/p-1/b9-4- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc
| Assay Description
All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. |
Chem Biol 11: 185-94 (2004)
Article DOI: 10.1016/j.chembiol.2004.02.012
BindingDB Entry DOI: 10.7270/Q2K9360M |
More data for this
Ligand-Target Pair | |
Quinone-dependent D-lactate dehydrogenase
(Escherichia coli) | BDBM59098

(Bi-ligand, 1)Show InChI InChI=1S/C12H9NO5S2/c14-7-2-1-6(3-8(7)15)4-9-11(18)13(5-10(16)17)12(19)20-9/h1-4,14-15H,5H2,(H,16,17)/p-1/b9-4- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | 7.4 | n/a |
Triad Therapeutics, Inc
| Assay Description
All reactions were monitored spectrophotometrically at 340 nm by using initial rates from the first 5% of reaction. |
Chem Biol 11: 185-94 (2004)
Article DOI: 10.1016/j.chembiol.2004.02.012
BindingDB Entry DOI: 10.7270/Q2K9360M |
More data for this
Ligand-Target Pair | |
11-beta-hydroxysteroid dehydrogenase 1
(Homo sapiens (Human)) | BDBM50248569

((1-(((R)-3-methyl-4-(4-((S)-1,1,1-trifluoro-2-hydr...)Show SMILES C[C@@H]1CN(CC2(CC2)C(N)=O)CCN1S(=O)(=O)c1ccc(cc1)[C@](C)(O)C(F)(F)F |r| Show InChI InChI=1S/C19H26F3N3O4S/c1-13-11-24(12-18(7-8-18)16(23)26)9-10-25(13)30(28,29)15-5-3-14(4-6-15)17(2,27)19(20,21)22/h3-6,13,27H,7-12H2,1-2H3,(H2,23,26)/t13-,17+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of FLAG tagged human recombinant 11beta-HSD1 reductase activity expressed in Trichoplusia ni Hi5 cells |
Bioorg Med Chem 16: 8922-31 (2008)
Article DOI: 10.1016/j.bmc.2008.08.065
BindingDB Entry DOI: 10.7270/Q22F7N8T |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
11-beta-hydroxysteroid dehydrogenase 1
(Homo sapiens (Human)) | BDBM50263346
((3-(1-(4-fluorophenyl)cyclopropyl)-4-isopropyl-5-(...)Show SMILES CC(C)n1c(nnc1C1(CC1)c1ccc(F)cc1)-c1ccc(OC(F)(F)F)cc1 Show InChI InChI=1S/C21H19F4N3O/c1-13(2)28-18(14-3-9-17(10-4-14)29-21(23,24)25)26-27-19(28)20(11-12-20)15-5-7-16(22)8-6-15/h3-10,13H,11-12H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
| MMDB
PDB
Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of FLAG tagged human recombinant 11beta-HSD1 reductase activity expressed in Trichoplusia ni Hi5 cells |
Bioorg Med Chem 16: 8922-31 (2008)
Article DOI: 10.1016/j.bmc.2008.08.065
BindingDB Entry DOI: 10.7270/Q22F7N8T |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data