 Found 137 hits with Last Name = 'philpott' and Initial = 'k'
Found 137 hits with Last Name = 'philpott' and Initial = 'k' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50419206
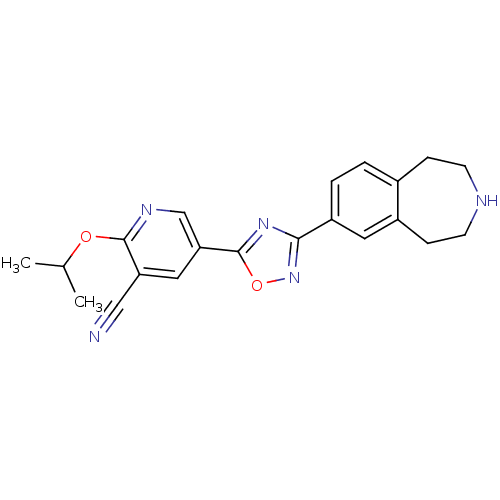
(CHEMBL1836170)Show SMILES CC(C)Oc1ncc(cc1C#N)-c1nc(no1)-c1ccc2CCNCCc2c1 Show InChI InChI=1S/C21H21N5O2/c1-13(2)27-20-17(11-22)10-18(12-24-20)21-25-19(26-28-21)16-4-3-14-5-7-23-8-6-15(14)9-16/h3-4,9-10,12-13,23H,5-8H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.16E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50354138

(CHEMBL1836215)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CN(CCc2c1C)C(CO)CO Show InChI InChI=1S/C25H28N4O4/c1-15(2)32-23-7-5-17(10-19(23)11-26)25-27-24(28-33-25)22-6-4-18-12-29(20(13-30)14-31)9-8-21(18)16(22)3/h4-7,10,15,20,30-31H,8-9,12-14H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50419207
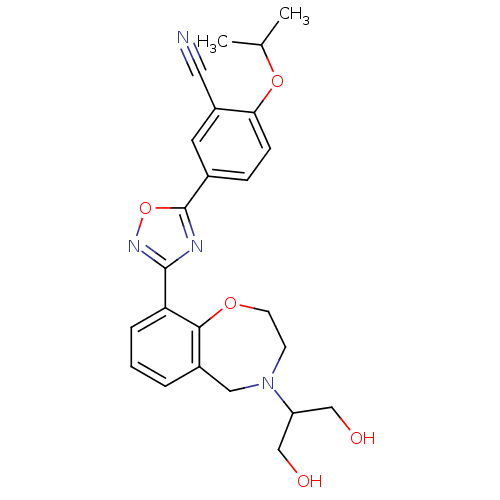
(CHEMBL1836212)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1cccc2CN(CCOc12)C(CO)CO Show InChI InChI=1S/C24H26N4O5/c1-15(2)32-21-7-6-16(10-18(21)11-25)24-26-23(27-33-24)20-5-3-4-17-12-28(19(13-29)14-30)8-9-31-22(17)20/h3-7,10,15,19,29-30H,8-9,12-14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.76E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50034557
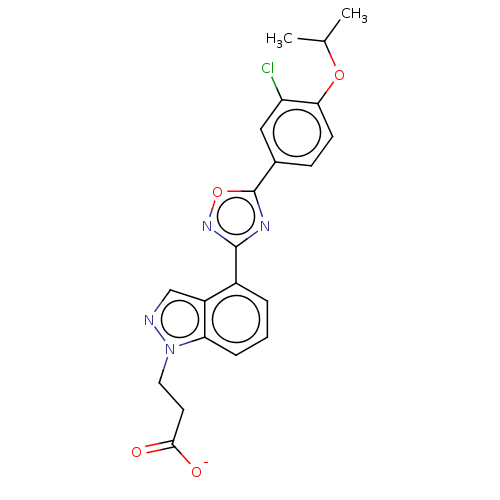
(CHEMBL3360360)Show SMILES [Na+].CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1cccc2n(CCC([O-])=O)ncc12 Show InChI InChI=1S/C21H19ClN4O4.Na/c1-12(2)29-18-7-6-13(10-16(18)22)21-24-20(25-30-21)14-4-3-5-17-15(14)11-23-26(17)9-8-19(27)28;/h3-7,10-12H,8-9H2,1-2H3,(H,27,28);/q;+1/p-1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50354138

(CHEMBL1836215)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CN(CCc2c1C)C(CO)CO Show InChI InChI=1S/C25H28N4O4/c1-15(2)32-23-7-5-17(10-19(23)11-26)25-27-24(28-33-25)22-6-4-18-12-29(20(13-30)14-31)9-8-21(18)16(22)3/h4-7,10,15,20,30-31H,8-9,12-14H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 using vivid green as substrate |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50419201

(CHEMBL1836171)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CN(CC(=O)N[C@@H](C)CO)CCc2c1C |r| Show InChI InChI=1S/C27H31N5O4/c1-16(2)35-24-8-6-19(11-21(24)12-28)27-30-26(31-36-27)23-7-5-20-13-32(10-9-22(20)18(23)4)14-25(34)29-17(3)15-33/h5-8,11,16-17,33H,9-10,13-15H2,1-4H3,(H,29,34)/t17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50419204

(CHEMBL1836213)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1cnc2CN(CCc2c1C)C(CO)CO Show InChI InChI=1S/C24H27N5O4/c1-14(2)32-22-5-4-16(8-17(22)9-25)24-27-23(28-33-24)20-10-26-21-11-29(18(12-30)13-31)7-6-19(21)15(20)3/h4-5,8,10,14,18,30-31H,6-7,11-13H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.26E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50354138

(CHEMBL1836215)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CN(CCc2c1C)C(CO)CO Show InChI InChI=1S/C25H28N4O4/c1-15(2)32-23-7-5-17(10-19(23)11-26)25-27-24(28-33-25)22-6-4-18-12-29(20(13-30)14-31)9-8-21(18)16(22)3/h4-7,10,15,20,30-31H,8-9,12-14H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.29E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50354138

(CHEMBL1836215)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CN(CCc2c1C)C(CO)CO Show InChI InChI=1S/C25H28N4O4/c1-15(2)32-23-7-5-17(10-19(23)11-26)25-27-24(28-33-25)22-6-4-18-12-29(20(13-30)14-31)9-8-21(18)16(22)3/h4-7,10,15,20,30-31H,8-9,12-14H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.53E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 using vivid red as substrate |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50419205
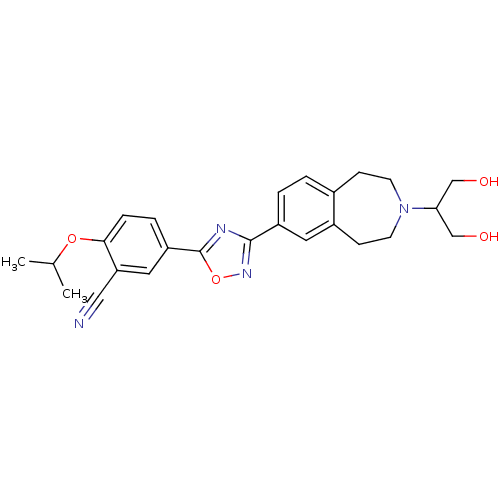
(CHEMBL1836172)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CCN(CCc2c1)C(CO)CO Show InChI InChI=1S/C25H28N4O4/c1-16(2)32-23-6-5-20(12-21(23)13-26)25-27-24(28-33-25)19-4-3-17-7-9-29(22(14-30)15-31)10-8-18(17)11-19/h3-6,11-12,16,22,30-31H,7-10,14-15H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.78E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50419203

(CHEMBL1836214)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CN(CCc2c1)C(CO)CO Show InChI InChI=1S/C24H26N4O4/c1-15(2)31-22-6-5-18(10-20(22)11-25)24-26-23(27-32-24)17-3-4-19-12-28(21(13-29)14-30)8-7-16(19)9-17/h3-6,9-10,15,21,29-30H,7-8,12-14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50034556
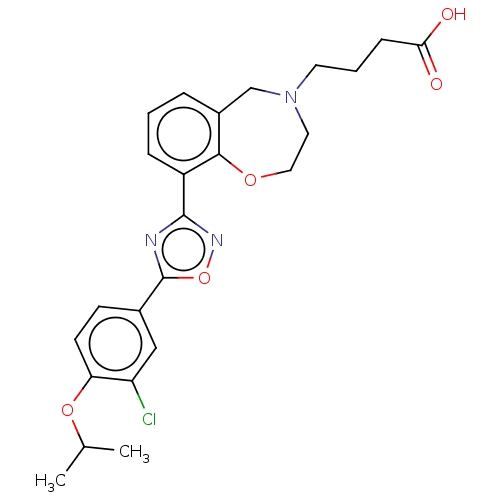
(CHEMBL3359844)Show SMILES CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1cccc2CN(CCCC(O)=O)CCOc12 Show InChI InChI=1S/C24H26ClN3O5/c1-15(2)32-20-9-8-16(13-19(20)25)24-26-23(27-33-24)18-6-3-5-17-14-28(10-4-7-21(29)30)11-12-31-22(17)18/h3,5-6,8-9,13,15H,4,7,10-12,14H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50034554
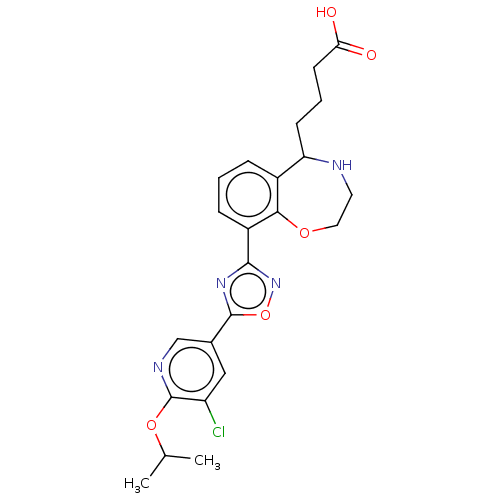
(CHEMBL3359854)Show SMILES CC(C)Oc1ncc(cc1Cl)-c1nc(no1)-c1cccc2C(CCCC(O)=O)NCCOc12 Show InChI InChI=1S/C23H25ClN4O5/c1-13(2)32-23-17(24)11-14(12-26-23)22-27-21(28-33-22)16-6-3-5-15-18(7-4-8-19(29)30)25-9-10-31-20(15)16/h3,5-6,11-13,18,25H,4,7-10H2,1-2H3,(H,29,30) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50034554

(CHEMBL3359854)Show SMILES CC(C)Oc1ncc(cc1Cl)-c1nc(no1)-c1cccc2C(CCCC(O)=O)NCCOc12 Show InChI InChI=1S/C23H25ClN4O5/c1-13(2)32-23-17(24)11-14(12-26-23)22-27-21(28-33-22)16-6-3-5-15-18(7-4-8-19(29)30)25-9-10-31-20(15)16/h3,5-6,11-13,18,25H,4,7-10H2,1-2H3,(H,29,30) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50034555

(CHEMBL3359847)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CCN(CCCC(O)=O)CCc2c1 Show InChI InChI=1S/C26H28N4O4/c1-17(2)33-23-8-7-21(15-22(23)16-27)26-28-25(29-34-26)20-6-5-18-9-12-30(11-3-4-24(31)32)13-10-19(18)14-20/h5-8,14-15,17H,3-4,9-13H2,1-2H3,(H,31,32) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50034556

(CHEMBL3359844)Show SMILES CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1cccc2CN(CCCC(O)=O)CCOc12 Show InChI InChI=1S/C24H26ClN3O5/c1-15(2)32-20-9-8-16(13-19(20)25)24-26-23(27-33-24)18-6-3-5-17-14-28(10-4-7-21(29)30)11-12-31-22(17)18/h3,5-6,8-9,13,15H,4,7,10-12,14H2,1-2H3,(H,29,30) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50034557

(CHEMBL3360360)Show SMILES [Na+].CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1cccc2n(CCC([O-])=O)ncc12 Show InChI InChI=1S/C21H19ClN4O4.Na/c1-12(2)29-18-7-6-13(10-16(18)22)21-24-20(25-30-21)14-4-3-5-17-15(14)11-23-26(17)9-8-19(27)28;/h3-7,10-12H,8-9H2,1-2H3,(H,27,28);/q;+1/p-1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50034554

(CHEMBL3359854)Show SMILES CC(C)Oc1ncc(cc1Cl)-c1nc(no1)-c1cccc2C(CCCC(O)=O)NCCOc12 Show InChI InChI=1S/C23H25ClN4O5/c1-13(2)32-23-17(24)11-14(12-26-23)22-27-21(28-33-22)16-6-3-5-15-18(7-4-8-19(29)30)25-9-10-31-20(15)16/h3,5-6,11-13,18,25H,4,7-10H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50034554

(CHEMBL3359854)Show SMILES CC(C)Oc1ncc(cc1Cl)-c1nc(no1)-c1cccc2C(CCCC(O)=O)NCCOc12 Show InChI InChI=1S/C23H25ClN4O5/c1-13(2)32-23-17(24)11-14(12-26-23)22-27-21(28-33-22)16-6-3-5-15-18(7-4-8-19(29)30)25-9-10-31-20(15)16/h3,5-6,11-13,18,25H,4,7-10H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50034555

(CHEMBL3359847)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CCN(CCCC(O)=O)CCc2c1 Show InChI InChI=1S/C26H28N4O4/c1-17(2)33-23-8-7-21(15-22(23)16-27)26-28-25(29-34-26)20-6-5-18-9-12-30(11-3-4-24(31)32)13-10-19(18)14-20/h5-8,14-15,17H,3-4,9-13H2,1-2H3,(H,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50034556

(CHEMBL3359844)Show SMILES CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1cccc2CN(CCCC(O)=O)CCOc12 Show InChI InChI=1S/C24H26ClN3O5/c1-15(2)32-20-9-8-16(13-19(20)25)24-26-23(27-33-24)18-6-3-5-17-14-28(10-4-7-21(29)30)11-12-31-22(17)18/h3,5-6,8-9,13,15H,4,7,10-12,14H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50034557

(CHEMBL3360360)Show SMILES [Na+].CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1cccc2n(CCC([O-])=O)ncc12 Show InChI InChI=1S/C21H19ClN4O4.Na/c1-12(2)29-18-7-6-13(10-16(18)22)21-24-20(25-30-21)14-4-3-5-17-15(14)11-23-26(17)9-8-19(27)28;/h3-7,10-12H,8-9H2,1-2H3,(H,27,28);/q;+1/p-1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50034554

(CHEMBL3359854)Show SMILES CC(C)Oc1ncc(cc1Cl)-c1nc(no1)-c1cccc2C(CCCC(O)=O)NCCOc12 Show InChI InChI=1S/C23H25ClN4O5/c1-13(2)32-23-17(24)11-14(12-26-23)22-27-21(28-33-22)16-6-3-5-15-18(7-4-8-19(29)30)25-9-10-31-20(15)16/h3,5-6,11-13,18,25H,4,7-10H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50034554

(CHEMBL3359854)Show SMILES CC(C)Oc1ncc(cc1Cl)-c1nc(no1)-c1cccc2C(CCCC(O)=O)NCCOc12 Show InChI InChI=1S/C23H25ClN4O5/c1-13(2)32-23-17(24)11-14(12-26-23)22-27-21(28-33-22)16-6-3-5-15-18(7-4-8-19(29)30)25-9-10-31-20(15)16/h3,5-6,11-13,18,25H,4,7-10H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50034555

(CHEMBL3359847)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CCN(CCCC(O)=O)CCc2c1 Show InChI InChI=1S/C26H28N4O4/c1-17(2)33-23-8-7-21(15-22(23)16-27)26-28-25(29-34-26)20-6-5-18-9-12-30(11-3-4-24(31)32)13-10-19(18)14-20/h5-8,14-15,17H,3-4,9-13H2,1-2H3,(H,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50034554

(CHEMBL3359854)Show SMILES CC(C)Oc1ncc(cc1Cl)-c1nc(no1)-c1cccc2C(CCCC(O)=O)NCCOc12 Show InChI InChI=1S/C23H25ClN4O5/c1-13(2)32-23-17(24)11-14(12-26-23)22-27-21(28-33-22)16-6-3-5-15-18(7-4-8-19(29)30)25-9-10-31-20(15)16/h3,5-6,11-13,18,25H,4,7-10H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50034554

(CHEMBL3359854)Show SMILES CC(C)Oc1ncc(cc1Cl)-c1nc(no1)-c1cccc2C(CCCC(O)=O)NCCOc12 Show InChI InChI=1S/C23H25ClN4O5/c1-13(2)32-23-17(24)11-14(12-26-23)22-27-21(28-33-22)16-6-3-5-15-18(7-4-8-19(29)30)25-9-10-31-20(15)16/h3,5-6,11-13,18,25H,4,7-10H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50034555

(CHEMBL3359847)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CCN(CCCC(O)=O)CCc2c1 Show InChI InChI=1S/C26H28N4O4/c1-17(2)33-23-8-7-21(15-22(23)16-27)26-28-25(29-34-26)20-6-5-18-9-12-30(11-3-4-24(31)32)13-10-19(18)14-20/h5-8,14-15,17H,3-4,9-13H2,1-2H3,(H,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50034556

(CHEMBL3359844)Show SMILES CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1cccc2CN(CCCC(O)=O)CCOc12 Show InChI InChI=1S/C24H26ClN3O5/c1-15(2)32-20-9-8-16(13-19(20)25)24-26-23(27-33-24)18-6-3-5-17-14-28(10-4-7-21(29)30)11-12-31-22(17)18/h3,5-6,8-9,13,15H,4,7,10-12,14H2,1-2H3,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50034557

(CHEMBL3360360)Show SMILES [Na+].CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1cccc2n(CCC([O-])=O)ncc12 Show InChI InChI=1S/C21H19ClN4O4.Na/c1-12(2)29-18-7-6-13(10-16(18)22)21-24-20(25-30-21)14-4-3-5-17-15(14)11-23-26(17)9-8-19(27)28;/h3-7,10-12H,8-9H2,1-2H3,(H,27,28);/q;+1/p-1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 (unknown origin) |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50354138

(CHEMBL1836215)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CN(CCc2c1C)C(CO)CO Show InChI InChI=1S/C25H28N4O4/c1-15(2)32-23-7-5-17(10-19(23)11-26)25-27-24(28-33-25)22-6-4-18-12-29(20(13-30)14-31)9-8-21(18)16(22)3/h4-7,10,15,20,30-31H,8-9,12-14H2,1-3H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50354138

(CHEMBL1836215)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CN(CCc2c1C)C(CO)CO Show InChI InChI=1S/C25H28N4O4/c1-15(2)32-23-7-5-17(10-19(23)11-26)25-27-24(28-33-25)22-6-4-18-12-29(20(13-30)14-31)9-8-21(18)16(22)3/h4-7,10,15,20,30-31H,8-9,12-14H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C19 |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034617
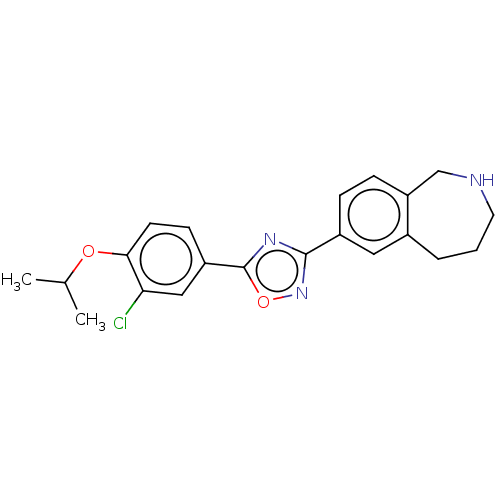
(CHEMBL3360373)Show SMILES CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1ccc2CNCCCc2c1 Show InChI InChI=1S/C21H22ClN3O2/c1-13(2)26-19-8-7-16(11-18(19)22)21-24-20(25-27-21)15-5-6-17-12-23-9-3-4-14(17)10-15/h5-8,10-11,13,23H,3-4,9,12H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 398 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034622

(CHEMBL3360372)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CNCCOc2c1 Show InChI InChI=1S/C21H20N4O3/c1-13(2)27-18-6-5-15(9-17(18)11-22)21-24-20(25-28-21)14-3-4-16-12-23-7-8-26-19(16)10-14/h3-6,9-10,13,23H,7-8,12H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 79 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034624
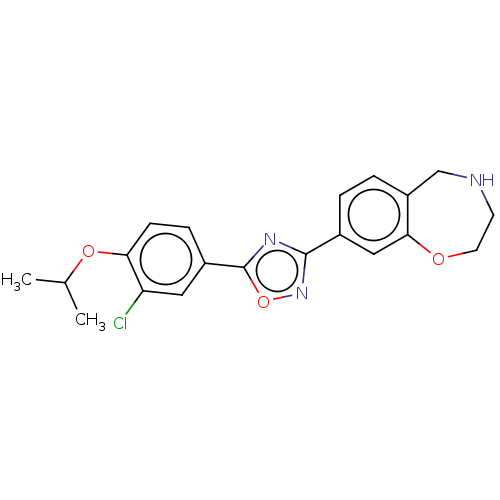
(CHEMBL3360371)Show SMILES CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1ccc2CNCCOc2c1 Show InChI InChI=1S/C20H20ClN3O3/c1-12(2)26-17-6-5-14(9-16(17)21)20-23-19(24-27-20)13-3-4-15-11-22-7-8-25-18(15)10-13/h3-6,9-10,12,22H,7-8,11H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 316 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034626
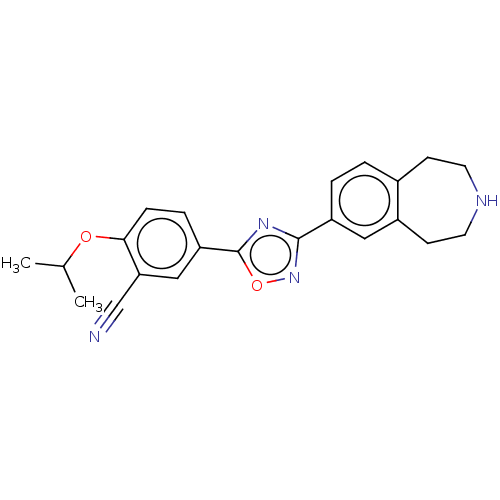
(CHEMBL3360370)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CCNCCc2c1 Show InChI InChI=1S/C22H22N4O2/c1-14(2)27-20-6-5-18(12-19(20)13-23)22-25-21(26-28-22)17-4-3-15-7-9-24-10-8-16(15)11-17/h3-6,11-12,14,24H,7-10H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 25 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034627
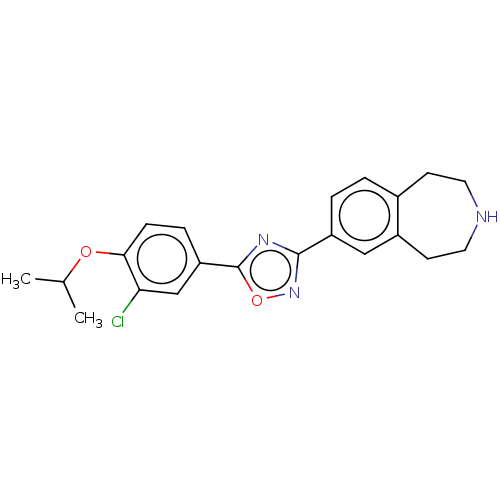
(CHEMBL3360369)Show SMILES CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1ccc2CCNCCc2c1 Show InChI InChI=1S/C21H22ClN3O2/c1-13(2)26-19-6-5-17(12-18(19)22)21-24-20(25-27-21)16-4-3-14-7-9-23-10-8-15(14)11-16/h3-6,11-13,23H,7-10H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 158 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034628
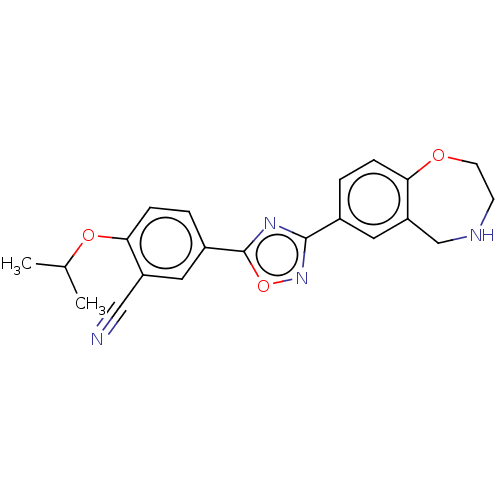
(CHEMBL3360368)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2OCCNCc2c1 Show InChI InChI=1S/C21H20N4O3/c1-13(2)27-19-6-4-15(10-16(19)11-22)21-24-20(25-28-21)14-3-5-18-17(9-14)12-23-7-8-26-18/h3-6,9-10,13,23H,7-8,12H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 25 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034629
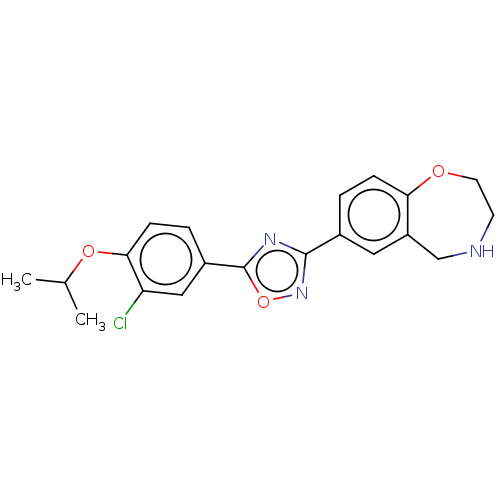
(CHEMBL3360367)Show SMILES CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1ccc2OCCNCc2c1 Show InChI InChI=1S/C20H20ClN3O3/c1-12(2)26-18-6-4-14(10-16(18)21)20-23-19(24-27-20)13-3-5-17-15(9-13)11-22-7-8-25-17/h3-6,9-10,12,22H,7-8,11H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 126 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034630
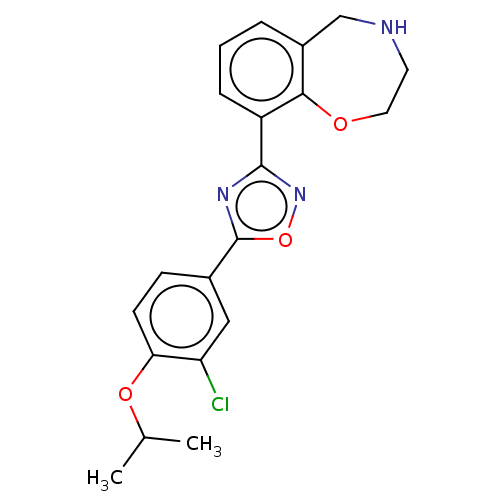
(CHEMBL3360366)Show SMILES CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1cccc2CNCCOc12 Show InChI InChI=1S/C20H20ClN3O3/c1-12(2)26-17-7-6-13(10-16(17)21)20-23-19(24-27-20)15-5-3-4-14-11-22-8-9-25-18(14)15/h3-7,10,12,22H,8-9,11H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 79 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034631
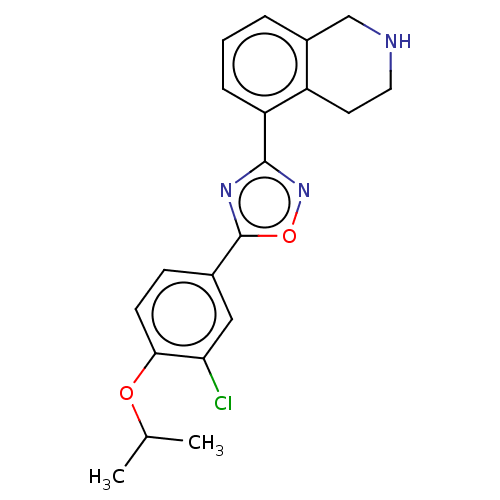
(CHEMBL3360365)Show SMILES CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1cccc2CNCCc12 Show InChI InChI=1S/C20H20ClN3O2/c1-12(2)25-18-7-6-13(10-17(18)21)20-23-19(24-26-20)16-5-3-4-14-11-22-9-8-15(14)16/h3-7,10,12,22H,8-9,11H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 40 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034632

(CHEMBL3360364)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CNCCc2c1 Show InChI InChI=1S/C21H20N4O2/c1-13(2)26-19-6-5-16(10-18(19)11-22)21-24-20(25-27-21)15-3-4-17-12-23-8-7-14(17)9-15/h3-6,9-10,13,23H,7-8,12H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 4 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034633
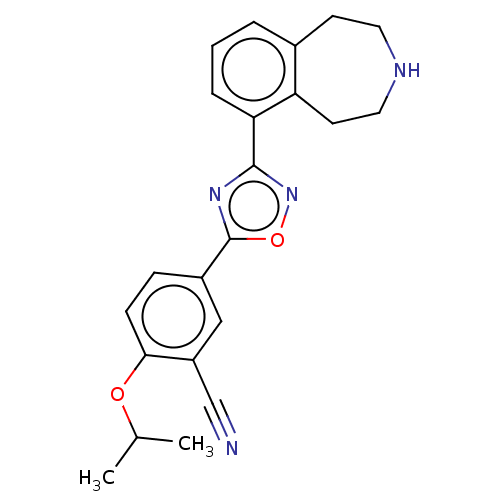
(CHEMBL3360363)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1cccc2CCNCCc12 Show InChI InChI=1S/C22H22N4O2/c1-14(2)27-20-7-6-16(12-17(20)13-23)22-25-21(26-28-22)19-5-3-4-15-8-10-24-11-9-18(15)19/h3-7,12,14,24H,8-11H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034634
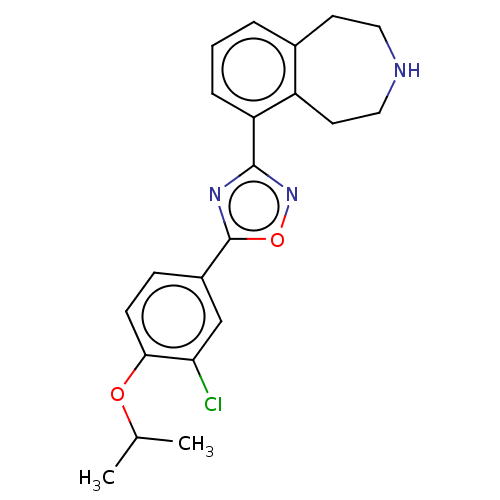
(CHEMBL3360362)Show SMILES CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1cccc2CCNCCc12 Show InChI InChI=1S/C21H22ClN3O2/c1-13(2)26-19-7-6-15(12-18(19)22)21-24-20(25-27-21)17-5-3-4-14-8-10-23-11-9-16(14)17/h3-7,12-13,23H,8-11H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 16 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034635
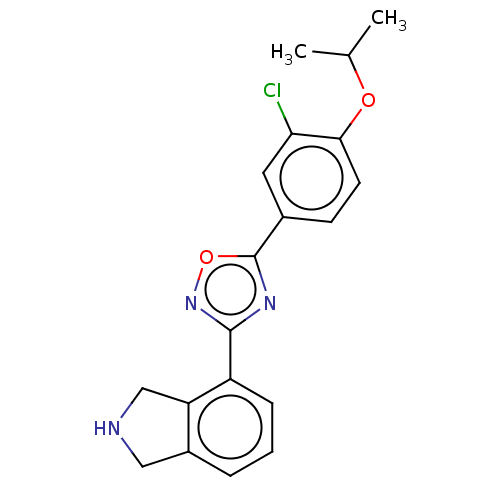
(CHEMBL3360361)Show InChI InChI=1S/C19H18ClN3O2/c1-11(2)24-17-7-6-12(8-16(17)20)19-22-18(23-25-19)14-5-3-4-13-9-21-10-15(13)14/h3-8,11,21H,9-10H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 7.90 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034557

(CHEMBL3360360)Show SMILES [Na+].CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1cccc2n(CCC([O-])=O)ncc12 Show InChI InChI=1S/C21H19ClN4O4.Na/c1-12(2)29-18-7-6-13(10-16(18)22)21-24-20(25-30-21)14-4-3-5-17-15(14)11-23-26(17)9-8-19(27)28;/h3-7,10-12H,8-9H2,1-2H3,(H,27,28);/q;+1/p-1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 20 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034636

(CHEMBL3360359)Show SMILES [Na+].CC(C)Oc1ccc(cc1Cl)-c1nc(no1)-c1ccc2n(CCC([O-])=O)ncc2c1 Show InChI InChI=1S/C21H19ClN4O4.Na/c1-12(2)29-18-6-4-14(10-16(18)22)21-24-20(25-30-21)13-3-5-17-15(9-13)11-23-26(17)8-7-19(27)28;/h3-6,9-12H,7-8H2,1-2H3,(H,27,28);/q;+1/p-1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 32 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50034637

(CHEMBL3360358)Show SMILES [Na+].[O-]C(=O)c1cnn(Cc2ccc(cc2)-c2noc(n2)-c2cc(c(s2)C(F)(F)F)-c2ccccc2)c1 Show InChI InChI=1S/C24H15F3N4O3S.Na/c25-24(26,27)20-18(15-4-2-1-3-5-15)10-19(35-20)22-29-21(30-34-22)16-8-6-14(7-9-16)12-31-13-17(11-28-31)23(32)33;/h1-11,13H,12H2,(H,32,33);/q;+1/p-1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 251 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1R expressed in RH7777 membranes assessed as [35S]GTPgammaS binding after 30 mins |
J Med Chem 57: 10424-42 (2014)
Article DOI: 10.1021/jm5010336
BindingDB Entry DOI: 10.7270/Q2DF6SSX |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50419201

(CHEMBL1836171)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CN(CC(=O)N[C@@H](C)CO)CCc2c1C |r| Show InChI InChI=1S/C27H31N5O4/c1-16(2)35-24-8-6-19(11-21(24)12-28)27-30-26(31-36-27)23-7-5-20-13-32(10-9-22(20)18(23)4)14-25(34)29-17(3)15-33/h5-8,11,16-17,33H,9-10,13-15H2,1-4H3,(H,29,34)/t17-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.26 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor expressed in CHO-K1 EDG1 cells expressing beta-arrestin 2 after 105 mins by chemi-luminiscence assay |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |
Sphingosine 1-phosphate receptor 1
(Homo sapiens (Human)) | BDBM50354138

(CHEMBL1836215)Show SMILES CC(C)Oc1ccc(cc1C#N)-c1nc(no1)-c1ccc2CN(CCc2c1C)C(CO)CO Show InChI InChI=1S/C25H28N4O4/c1-15(2)32-23-7-5-17(10-19(23)11-26)25-27-24(28-33-25)22-6-4-18-12-29(20(13-30)14-31)9-8-21(18)16(22)3/h4-7,10,15,20,30-31H,8-9,12-14H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.16 | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Agonist activity at human S1P1 receptor expressed in CHO-K1 EDG1 cells expressing beta-arrestin 2 after 105 mins by chemi-luminiscence assay |
J Med Chem 54: 6724-33 (2011)
Article DOI: 10.1021/jm200609t
BindingDB Entry DOI: 10.7270/Q27P8ZSM |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data