 Found 262 hits with Last Name = 'rogers' and Initial = 'rs'
Found 262 hits with Last Name = 'rogers' and Initial = 'rs' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057581
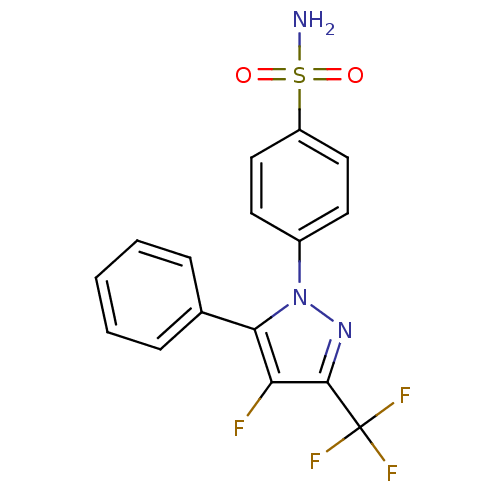
(4-(4-Fluoro-5-phenyl-3-trifluoromethyl-pyrazol-1-y...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(c(F)c1-c1ccccc1)C(F)(F)F Show InChI InChI=1S/C16H11F4N3O2S/c17-13-14(10-4-2-1-3-5-10)23(22-15(13)16(18,19)20)11-6-8-12(9-7-11)26(21,24)25/h1-9H,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057618
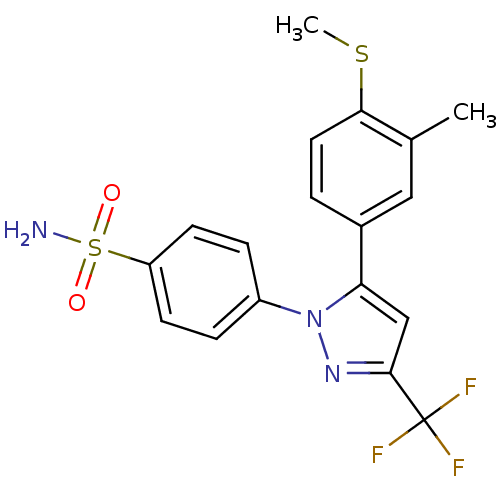
(4-[5-(3-Methyl-4-methylsulfanyl-phenyl)-3-trifluor...)Show SMILES CSc1ccc(cc1C)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C18H16F3N3O2S2/c1-11-9-12(3-8-16(11)27-2)15-10-17(18(19,20)21)23-24(15)13-4-6-14(7-5-13)28(22,25)26/h3-10H,1-2H3,(H2,22,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057551
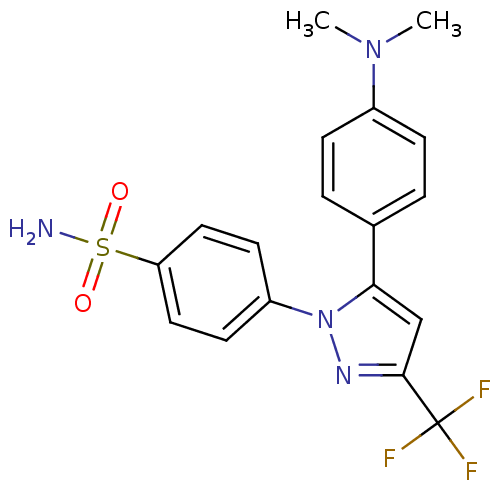
(4-[5-(4-Dimethylamino-phenyl)-3-trifluoromethyl-py...)Show SMILES CN(C)c1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C18H17F3N4O2S/c1-24(2)13-5-3-12(4-6-13)16-11-17(18(19,20)21)23-25(16)14-7-9-15(10-8-14)28(22,26)27/h3-11H,1-2H3,(H2,22,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM13063
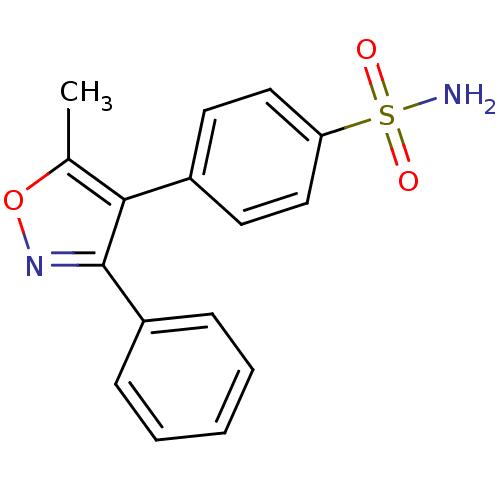
(4-(5-methyl-3-phenyl-1,2-oxazol-4-yl)benzene-1-sul...)Show InChI InChI=1S/C16H14N2O3S/c1-11-15(12-7-9-14(10-8-12)22(17,19)20)16(18-21-11)13-5-3-2-4-6-13/h2-10H,1H3,(H2,17,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In Vitro activity of compound against human recombinant Prostaglandin G/H synthase 2 |
J Med Chem 43: 775-7 (2000)
BindingDB Entry DOI: 10.7270/Q2NV9HG1 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50057554
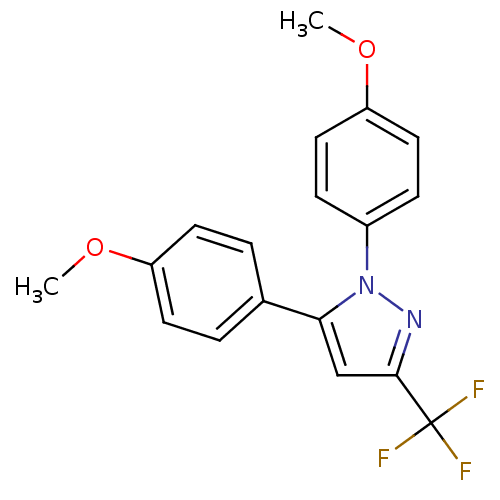
(1,5-Bis-(4-methoxy-phenyl)-3-trifluoromethyl-1H-py...)Show SMILES COc1ccc(cc1)-c1cc(nn1-c1ccc(OC)cc1)C(F)(F)F Show InChI InChI=1S/C18H15F3N2O2/c1-24-14-7-3-12(4-8-14)16-11-17(18(19,20)21)22-23(16)13-5-9-15(25-2)10-6-13/h3-11H,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block recombinant human prostaglandin G/H synthase 1 (COX-1) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM13063

(4-(5-methyl-3-phenyl-1,2-oxazol-4-yl)benzene-1-sul...)Show InChI InChI=1S/C16H14N2O3S/c1-11-15(12-7-9-14(10-8-12)22(17,19)20)16(18-21-11)13-5-3-2-4-6-13/h2-10H,1H3,(H2,17,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In Vitro activity of compound against human recombinant Prostaglandin G/H synthase 2 |
J Med Chem 43: 775-7 (2000)
BindingDB Entry DOI: 10.7270/Q2NV9HG1 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057606
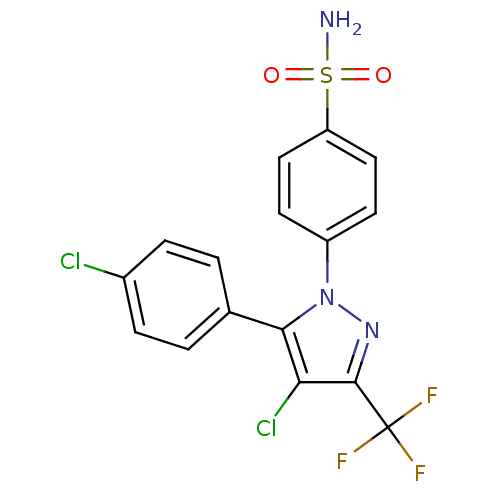
(4-[4-Chloro-5-(4-chloro-phenyl)-3-trifluoromethyl-...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(c(Cl)c1-c1ccc(Cl)cc1)C(F)(F)F Show InChI InChI=1S/C16H10Cl2F3N3O2S/c17-10-3-1-9(2-4-10)14-13(18)15(16(19,20)21)23-24(14)11-5-7-12(8-6-11)27(22,25)26/h1-8H,(H2,22,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057575
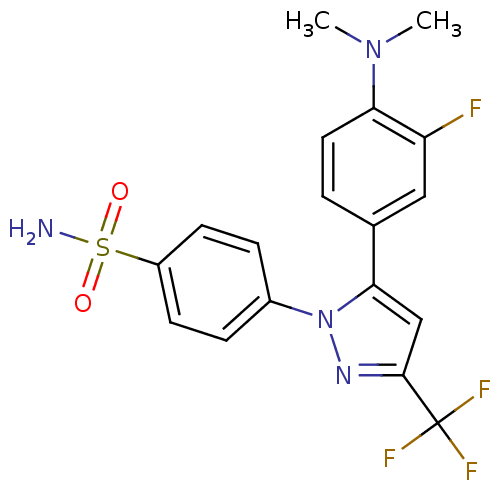
(4-[5-(4-Dimethylamino-3-fluoro-phenyl)-3-trifluoro...)Show SMILES CN(C)c1ccc(cc1F)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C18H16F4N4O2S/c1-25(2)15-8-3-11(9-14(15)19)16-10-17(18(20,21)22)24-26(16)12-4-6-13(7-5-12)29(23,27)28/h3-10H,1-2H3,(H2,23,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057609
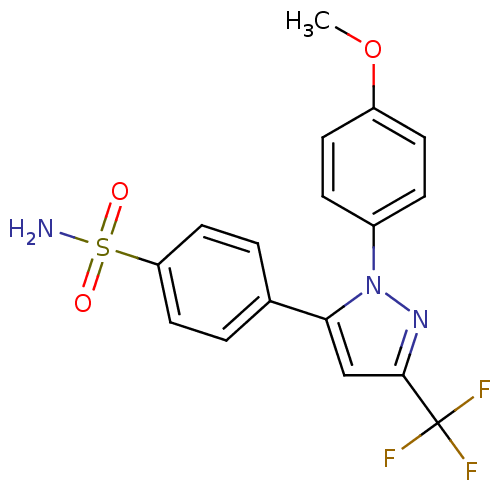
(4-[2-(4-Methoxy-phenyl)-5-trifluoromethyl-2H-pyraz...)Show SMILES COc1ccc(cc1)-n1nc(cc1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O3S/c1-26-13-6-4-12(5-7-13)23-15(10-16(22-23)17(18,19)20)11-2-8-14(9-3-11)27(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM13065

(5-(4-chlorophenyl)-1-(4-methoxyphenyl)-3-(trifluor...)Show SMILES COc1ccc(cc1)-n1nc(cc1-c1ccc(Cl)cc1)C(F)(F)F Show InChI InChI=1S/C17H12ClF3N2O/c1-24-14-8-6-13(7-9-14)23-15(10-16(22-23)17(19,20)21)11-2-4-12(18)5-3-11/h2-10H,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block recombinant human prostaglandin G/H synthase 1 (COX-1) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057589
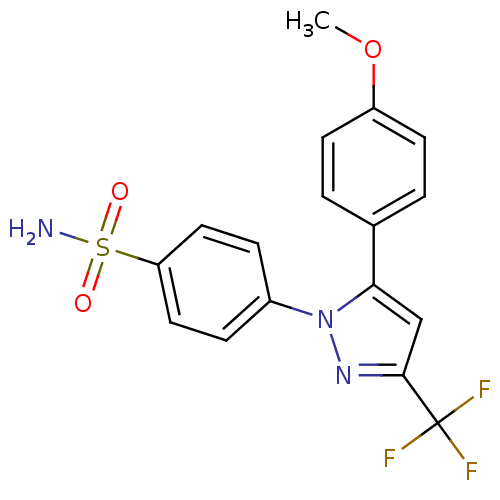
(4-[5-(4-Methoxy-phenyl)-3-trifluoromethyl-pyrazol-...)Show SMILES COc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O3S/c1-26-13-6-2-11(3-7-13)15-10-16(17(18,19)20)22-23(15)12-4-8-14(9-5-12)27(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50286050
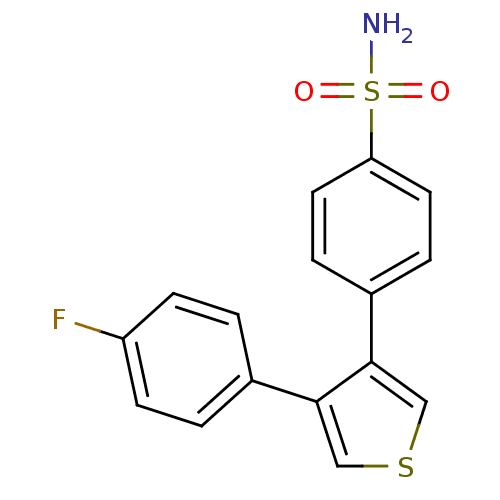
(4-[4-(4-Fluoro-phenyl)-thiophen-3-yl]-benzenesulfo...)Show InChI InChI=1S/C16H12FNO2S2/c17-13-5-1-11(2-6-13)15-9-21-10-16(15)12-3-7-14(8-4-12)22(18,19)20/h1-10H,(H2,18,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against human recombinant Prostaglandin G/H synthase 2 |
Bioorg Med Chem Lett 5: 2919-2922 (1995)
Article DOI: 10.1016/0960-894X(95)00512-R
BindingDB Entry DOI: 10.7270/Q2HM58D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057591
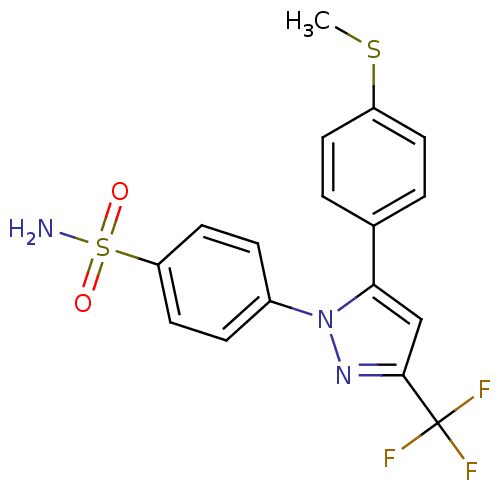
(4-[5-(4-Methylsulfanyl-phenyl)-3-trifluoromethyl-p...)Show SMILES CSc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O2S2/c1-26-13-6-2-11(3-7-13)15-10-16(17(18,19)20)22-23(15)12-4-8-14(9-5-12)27(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50286047

(4-[4-(3-Fluoro-4-methoxy-phenyl)-thiophen-3-yl]-be...)Show InChI InChI=1S/C17H14FNO3S2/c1-22-17-7-4-12(8-16(17)18)15-10-23-9-14(15)11-2-5-13(6-3-11)24(19,20)21/h2-10H,1H3,(H2,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against human recombinant Prostaglandin G/H synthase 2 |
Bioorg Med Chem Lett 5: 2919-2922 (1995)
Article DOI: 10.1016/0960-894X(95)00512-R
BindingDB Entry DOI: 10.7270/Q2HM58D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057620
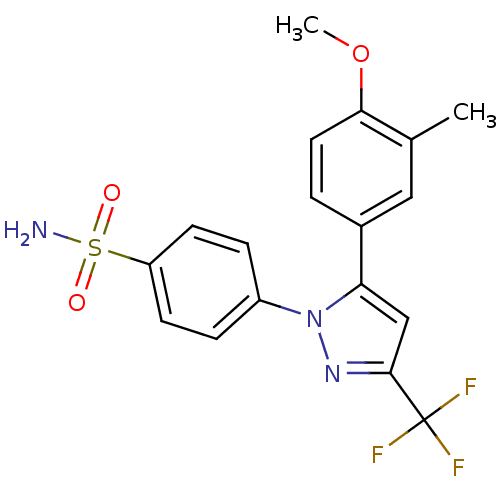
(4-[5-(4-Methoxy-3-methyl-phenyl)-3-trifluoromethyl...)Show SMILES COc1ccc(cc1C)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C18H16F3N3O3S/c1-11-9-12(3-8-16(11)27-2)15-10-17(18(19,20)21)23-24(15)13-4-6-14(7-5-13)28(22,25)26/h3-10H,1-2H3,(H2,22,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057527
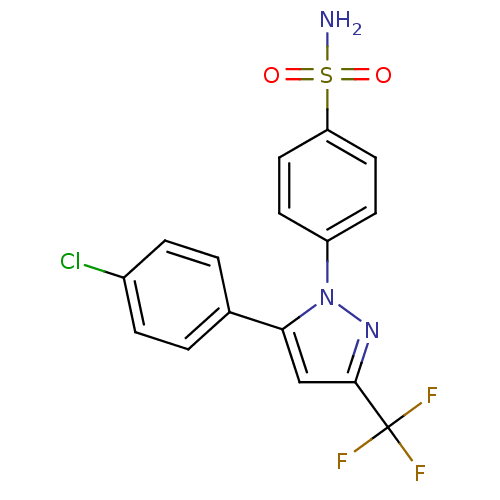
(4-(5-(4-chlorophenyl)-3-(trifluoromethyl)-1H-pyraz...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(cc1-c1ccc(Cl)cc1)C(F)(F)F Show InChI InChI=1S/C16H11ClF3N3O2S/c17-11-3-1-10(2-4-11)14-9-15(16(18,19)20)22-23(14)12-5-7-13(8-6-12)26(21,24)25/h1-9H,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057610

(4-[5-(4-Chloro-phenyl)-3-difluoromethyl-pyrazol-1-...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(cc1-c1ccc(Cl)cc1)C(F)F Show InChI InChI=1S/C16H12ClF2N3O2S/c17-11-3-1-10(2-4-11)15-9-14(16(18)19)21-22(15)12-5-7-13(8-6-12)25(20,23)24/h1-9,16H,(H2,20,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057602
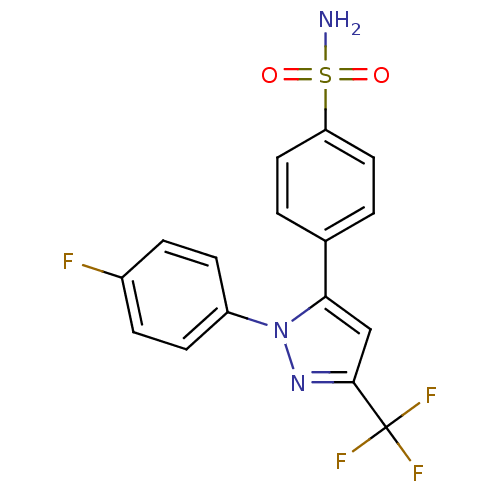
(4-[2-(4-Fluoro-phenyl)-5-trifluoromethyl-2H-pyrazo...)Show SMILES NS(=O)(=O)c1ccc(cc1)-c1cc(nn1-c1ccc(F)cc1)C(F)(F)F Show InChI InChI=1S/C16H11F4N3O2S/c17-11-3-5-12(6-4-11)23-14(9-15(22-23)16(18,19)20)10-1-7-13(8-2-10)26(21,24)25/h1-9H,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057572
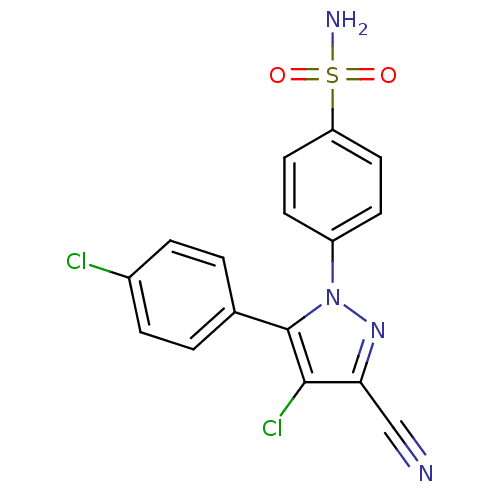
(4-[4-Chloro-5-(4-chloro-phenyl)-3-cyano-pyrazol-1-...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(C#N)c(Cl)c1-c1ccc(Cl)cc1 Show InChI InChI=1S/C16H10Cl2N4O2S/c17-11-3-1-10(2-4-11)16-15(18)14(9-19)21-22(16)12-5-7-13(8-6-12)25(20,23)24/h1-8H,(H2,20,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057552

(4-[5-(5-Bromo-thiophen-2-yl)-3-trifluoromethyl-pyr...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(cc1-c1ccc(Br)s1)C(F)(F)F Show InChI InChI=1S/C14H9BrF3N3O2S2/c15-13-6-5-11(24-13)10-7-12(14(16,17)18)20-21(10)8-1-3-9(4-2-8)25(19,22)23/h1-7H,(H2,19,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057608
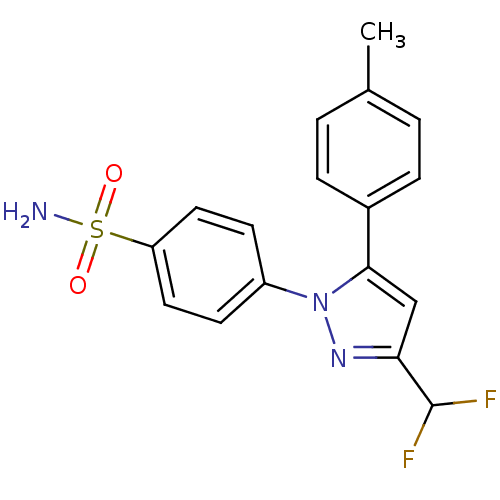
(4-(3-Difluoromethyl-5-p-tolyl-pyrazol-1-yl)-benzen...)Show SMILES Cc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)F Show InChI InChI=1S/C17H15F2N3O2S/c1-11-2-4-12(5-3-11)16-10-15(17(18)19)21-22(16)13-6-8-14(9-7-13)25(20,23)24/h2-10,17H,1H3,(H2,20,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029600
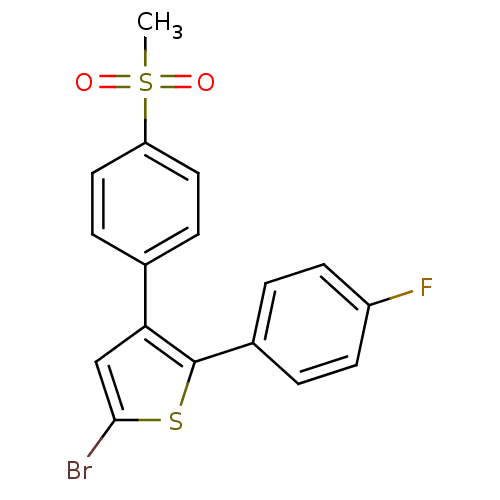
(5-Bromo-2-(4-fluoro-phenyl)-3-(4-methanesulfonyl-p...)Show SMILES CS(=O)(=O)c1ccc(cc1)-c1cc(Br)sc1-c1ccc(F)cc1 Show InChI InChI=1S/C17H12BrFO2S2/c1-23(20,21)14-8-4-11(5-9-14)15-10-16(18)22-17(15)12-2-6-13(19)7-3-12/h2-10H,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against human recombinant Prostaglandin G/H synthase 2 |
Bioorg Med Chem Lett 5: 2919-2922 (1995)
Article DOI: 10.1016/0960-894X(95)00512-R
BindingDB Entry DOI: 10.7270/Q2HM58D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057562

(4-[3-Difluoromethyl-5-(4-methoxy-phenyl)-pyrazol-1...)Show SMILES COc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)F Show InChI InChI=1S/C17H15F2N3O3S/c1-25-13-6-2-11(3-7-13)16-10-15(17(18)19)21-22(16)12-4-8-14(9-5-12)26(20,23)24/h2-10,17H,1H3,(H2,20,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057560
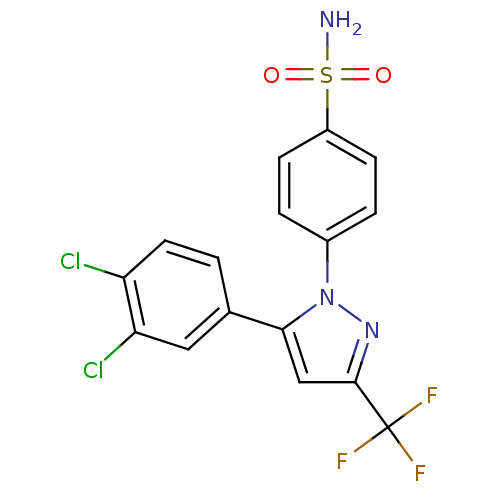
(4-[5-(3,4-Dichloro-phenyl)-3-trifluoromethyl-pyraz...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(cc1-c1ccc(Cl)c(Cl)c1)C(F)(F)F Show InChI InChI=1S/C16H10Cl2F3N3O2S/c17-12-6-1-9(7-13(12)18)14-8-15(16(19,20)21)23-24(14)10-2-4-11(5-3-10)27(22,25)26/h1-8H,(H2,22,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50286048
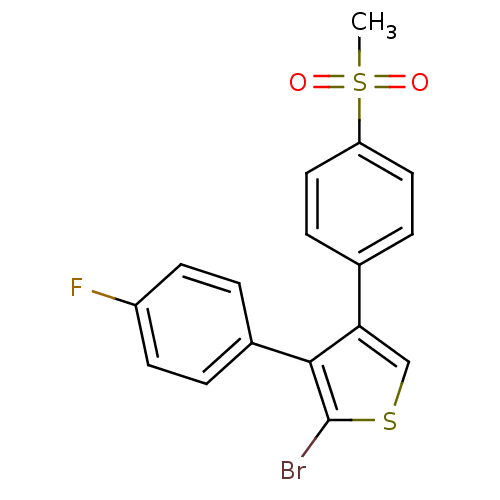
(2-Bromo-3-(4-fluoro-phenyl)-4-(4-methanesulfonyl-p...)Show SMILES CS(=O)(=O)c1ccc(cc1)-c1csc(Br)c1-c1ccc(F)cc1 Show InChI InChI=1S/C17H12BrFO2S2/c1-23(20,21)14-8-4-11(5-9-14)15-10-22-17(18)16(15)12-2-6-13(19)7-3-12/h2-10H,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against human recombinant Prostaglandin G/H synthase 2 |
Bioorg Med Chem Lett 5: 2919-2922 (1995)
Article DOI: 10.1016/0960-894X(95)00512-R
BindingDB Entry DOI: 10.7270/Q2HM58D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057573
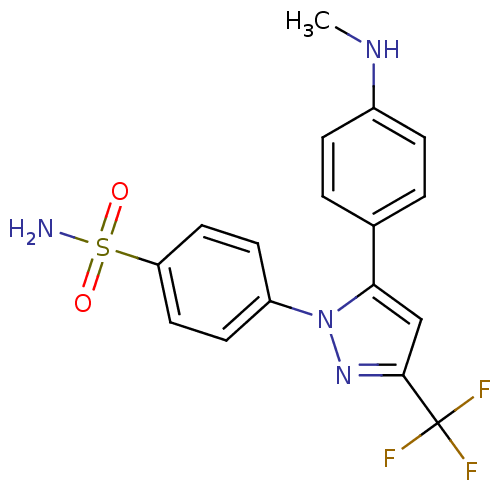
(4-[5-(4-Methylamino-phenyl)-3-trifluoromethyl-pyra...)Show SMILES CNc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H15F3N4O2S/c1-22-12-4-2-11(3-5-12)15-10-16(17(18,19)20)23-24(15)13-6-8-14(9-7-13)27(21,25)26/h2-10,22H,1H3,(H2,21,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50057532

(1-(4-Chloro-phenyl)-5-(4-methoxy-phenyl)-3-trifluo...)Show SMILES COc1ccc(cc1)-c1cc(nn1-c1ccc(Cl)cc1)C(F)(F)F Show InChI InChI=1S/C17H12ClF3N2O/c1-24-14-8-2-11(3-9-14)15-10-16(17(19,20)21)22-23(15)13-6-4-12(18)5-7-13/h2-10H,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block recombinant human prostaglandin G/H synthase 1 (COX-1) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057544
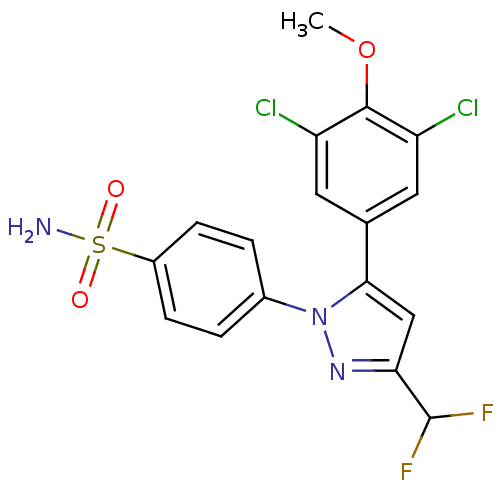
(4-[5-(3,5-Dichloro-4-methoxy-phenyl)-3-difluoromet...)Show SMILES COc1c(Cl)cc(cc1Cl)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)F Show InChI InChI=1S/C17H13Cl2F2N3O3S/c1-27-16-12(18)6-9(7-13(16)19)15-8-14(17(20)21)23-24(15)10-2-4-11(5-3-10)28(22,25)26/h2-8,17H,1H3,(H2,22,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057592
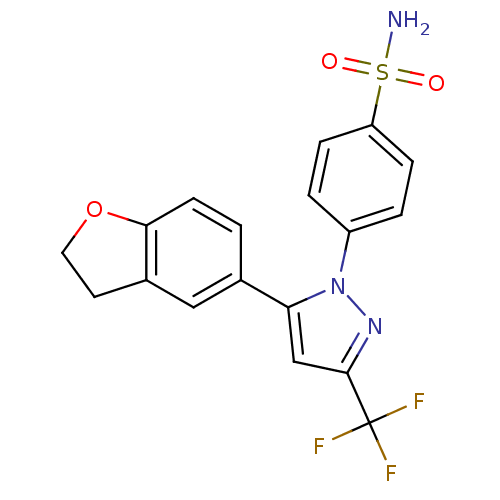
(4-[5-(2,3-Dihydro-benzofuran-5-yl)-3-trifluorometh...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(cc1-c1ccc2OCCc2c1)C(F)(F)F Show InChI InChI=1S/C18H14F3N3O3S/c19-18(20,21)17-10-15(11-1-6-16-12(9-11)7-8-27-16)24(23-17)13-2-4-14(5-3-13)28(22,25)26/h1-6,9-10H,7-8H2,(H2,22,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
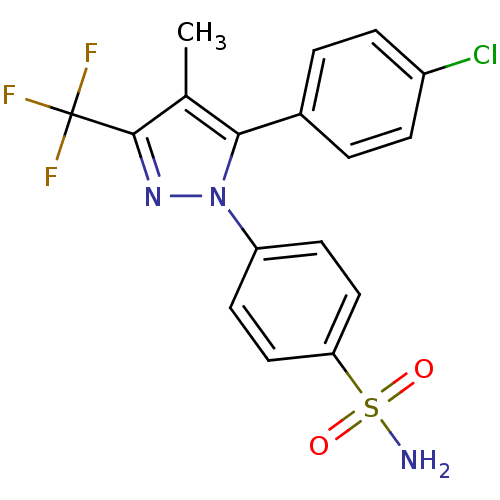
(Homo sapiens (Human)) | BDBM50057553
(4-[5-(4-Chloro-phenyl)-4-methyl-3-trifluoromethyl-...)Show SMILES Cc1c(nn(c1-c1ccc(Cl)cc1)-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H13ClF3N3O2S/c1-10-15(11-2-4-12(18)5-3-11)24(23-16(10)17(19,20)21)13-6-8-14(9-7-13)27(22,25)26/h2-9H,1H3,(H2,22,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
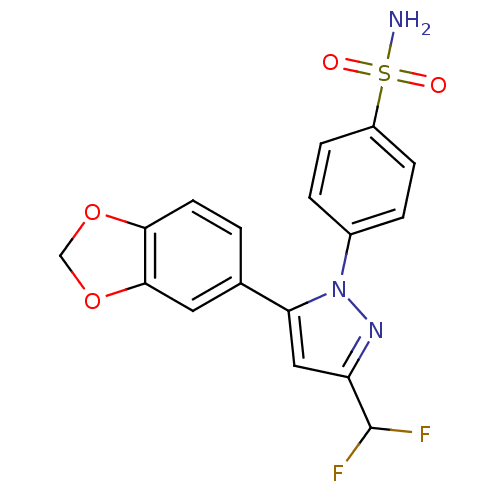
(Homo sapiens (Human)) | BDBM50057600
(4-(5-Benzo[1,3]dioxol-5-yl-3-difluoromethyl-pyrazo...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(cc1-c1ccc2OCOc2c1)C(F)F Show InChI InChI=1S/C17H13F2N3O4S/c18-17(19)13-8-14(10-1-6-15-16(7-10)26-9-25-15)22(21-13)11-2-4-12(5-3-11)27(20,23)24/h1-8,17H,9H2,(H2,20,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057605
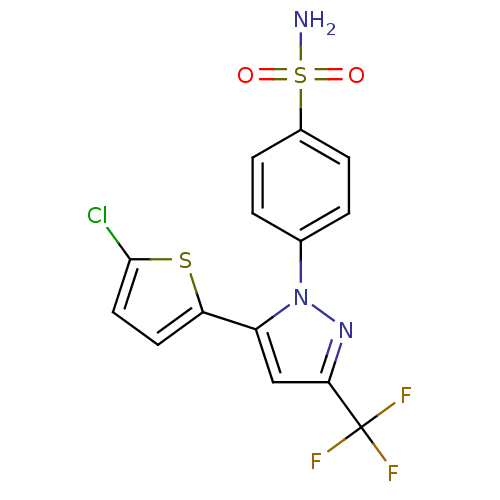
(4-[5-(5-Chloro-thiophen-2-yl)-3-trifluoromethyl-py...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(cc1-c1ccc(Cl)s1)C(F)(F)F Show InChI InChI=1S/C14H9ClF3N3O2S2/c15-13-6-5-11(24-13)10-7-12(14(16,17)18)20-21(10)8-1-3-9(4-2-8)25(19,22)23/h1-7H,(H2,19,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057596
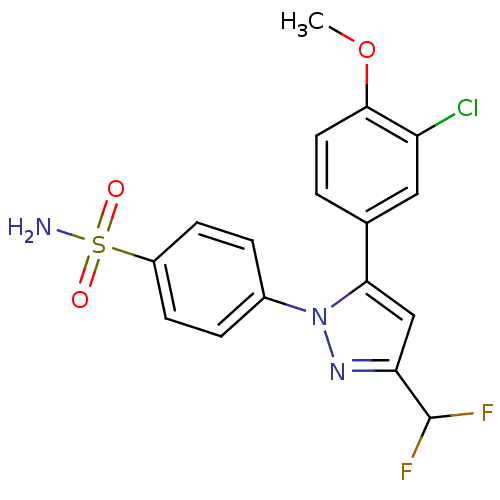
(4-[5-(3-Chloro-4-methoxy-phenyl)-3-difluoromethyl-...)Show SMILES COc1ccc(cc1Cl)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)F Show InChI InChI=1S/C17H14ClF2N3O3S/c1-26-16-7-2-10(8-13(16)18)15-9-14(17(19)20)22-23(15)11-3-5-12(6-4-11)27(21,24)25/h2-9,17H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057541
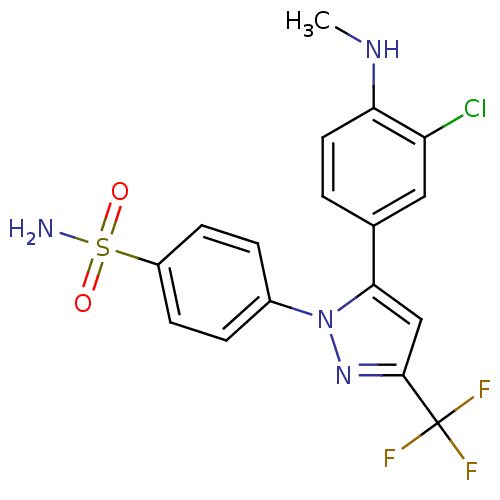
(4-[5-(3-Chloro-4-methylamino-phenyl)-3-trifluorome...)Show SMILES CNc1ccc(cc1Cl)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14ClF3N4O2S/c1-23-14-7-2-10(8-13(14)18)15-9-16(17(19,20)21)24-25(15)11-3-5-12(6-4-11)28(22,26)27/h2-9,23H,1H3,(H2,22,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057528
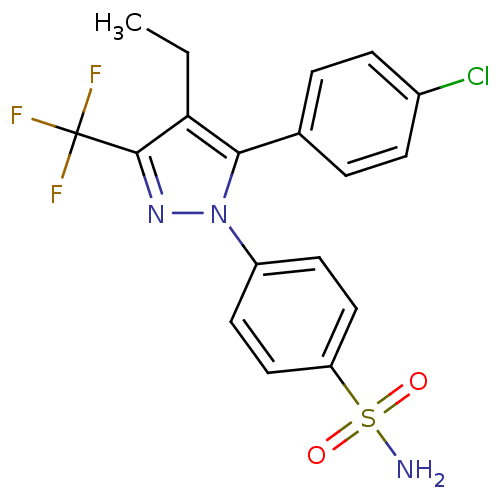
(4-[5-(4-Chloro-phenyl)-4-ethyl-3-trifluoromethyl-p...)Show SMILES CCc1c(nn(c1-c1ccc(Cl)cc1)-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C18H15ClF3N3O2S/c1-2-15-16(11-3-5-12(19)6-4-11)25(24-17(15)18(20,21)22)13-7-9-14(10-8-13)28(23,26)27/h3-10H,2H2,1H3,(H2,23,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057535
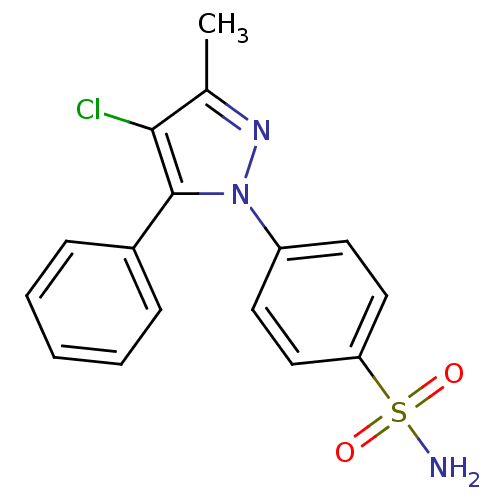
(4-(4-Chloro-3-methyl-5-phenyl-pyrazol-1-yl)-benzen...)Show SMILES Cc1nn(c(c1Cl)-c1ccccc1)-c1ccc(cc1)S(N)(=O)=O Show InChI InChI=1S/C16H14ClN3O2S/c1-11-15(17)16(12-5-3-2-4-6-12)20(19-11)13-7-9-14(10-8-13)23(18,21)22/h2-10H,1H3,(H2,18,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057561
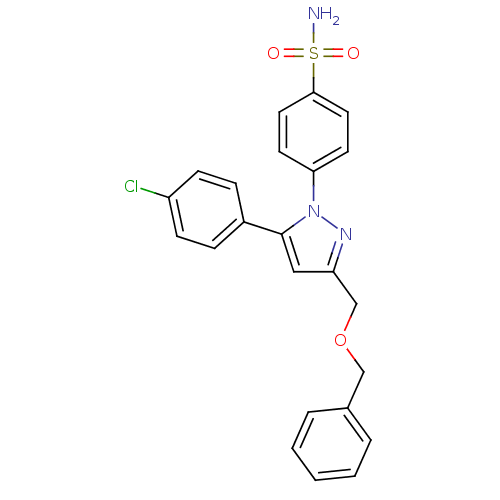
(4-[3-Benzyloxymethyl-5-(4-chloro-phenyl)-pyrazol-1...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(COCc2ccccc2)cc1-c1ccc(Cl)cc1 Show InChI InChI=1S/C23H20ClN3O3S/c24-19-8-6-18(7-9-19)23-14-20(16-30-15-17-4-2-1-3-5-17)26-27(23)21-10-12-22(13-11-21)31(25,28)29/h1-14H,15-16H2,(H2,25,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM21642

((2S)-1-[(2S)-2-methyl-3-sulfanylpropanoyl]pyrrolid...)Show InChI InChI=1S/C9H15NO3S/c1-6(5-14)8(11)10-4-2-3-7(10)9(12)13/h6-7,14H,2-5H2,1H3,(H,12,13)/t6-,7+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was tested for inhibitory activity against Angiotensin I converting enzyme |
Bioorg Med Chem Lett 3: 1953-1958 (1993)
Article DOI: 10.1016/S0960-894X(01)80994-X
BindingDB Entry DOI: 10.7270/Q2057GD5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50286052
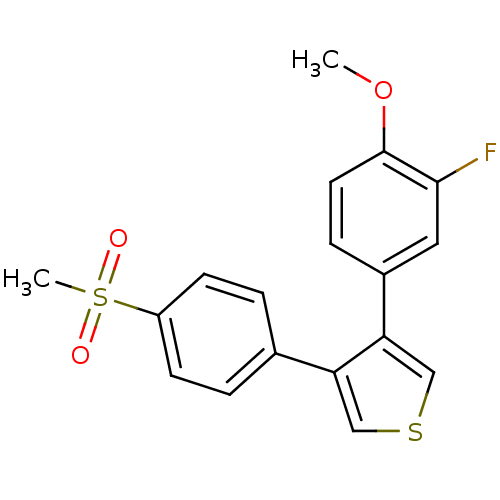
(3-(3-Fluoro-4-methoxy-phenyl)-4-(4-methanesulfonyl...)Show InChI InChI=1S/C18H15FO3S2/c1-22-18-8-5-13(9-17(18)19)16-11-23-10-15(16)12-3-6-14(7-4-12)24(2,20)21/h3-11H,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against human recombinant Prostaglandin G/H synthase 2 |
Bioorg Med Chem Lett 5: 2919-2922 (1995)
Article DOI: 10.1016/0960-894X(95)00512-R
BindingDB Entry DOI: 10.7270/Q2HM58D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057590
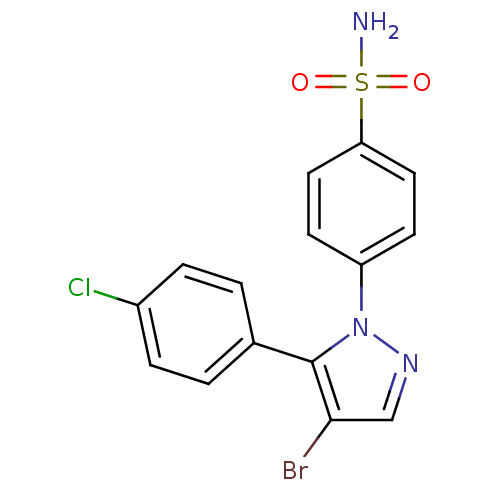
(4-[4-Bromo-5-(4-chloro-phenyl)-pyrazol-1-yl]-benze...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1ncc(Br)c1-c1ccc(Cl)cc1 Show InChI InChI=1S/C15H11BrClN3O2S/c16-14-9-19-20(15(14)10-1-3-11(17)4-2-10)12-5-7-13(8-6-12)23(18,21)22/h1-9H,(H2,18,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057580
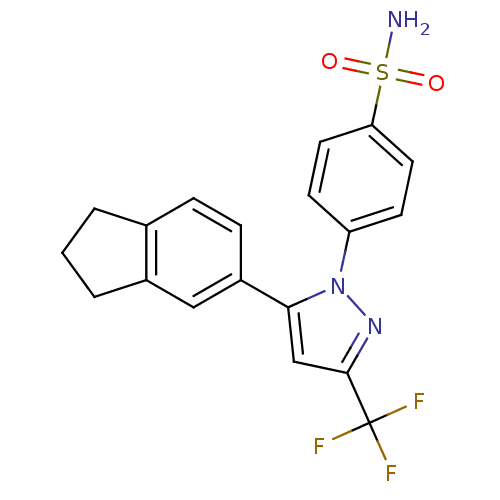
(4-(5-Indan-5-yl-3-trifluoromethyl-pyrazol-1-yl)-be...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(cc1-c1ccc2CCCc2c1)C(F)(F)F Show InChI InChI=1S/C19H16F3N3O2S/c20-19(21,22)18-11-17(14-5-4-12-2-1-3-13(12)10-14)25(24-18)15-6-8-16(9-7-15)28(23,26)27/h4-11H,1-3H2,(H2,23,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
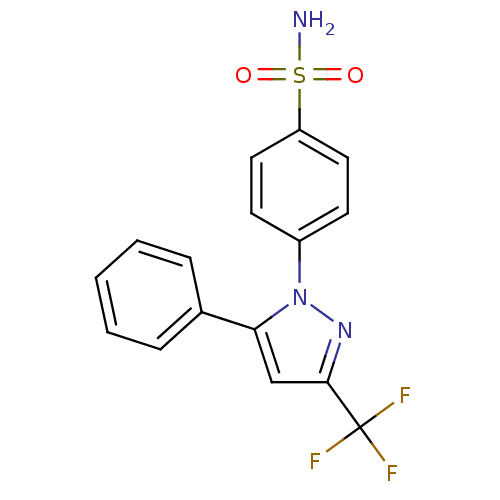
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057518
(4-(5-Phenyl-3-trifluoromethyl-pyrazol-1-yl)-benzen...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(cc1-c1ccccc1)C(F)(F)F Show InChI InChI=1S/C16H12F3N3O2S/c17-16(18,19)15-10-14(11-4-2-1-3-5-11)22(21-15)12-6-8-13(9-7-12)25(20,23)24/h1-10H,(H2,20,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM11639

(4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyraz...)Show SMILES Cc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O2S/c1-11-2-4-12(5-3-11)15-10-16(17(18,19)20)22-23(15)13-6-8-14(9-7-13)26(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In Vitro activity of compound against human recombinant Prostaglandin G/H synthase 2 |
J Med Chem 43: 775-7 (2000)
BindingDB Entry DOI: 10.7270/Q2NV9HG1 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM11639

(4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyraz...)Show SMILES Cc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O2S/c1-11-2-4-12(5-3-11)15-10-16(17(18,19)20)22-23(15)13-6-8-14(9-7-13)26(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM11639

(4-[5-(4-methylphenyl)-3-(trifluoromethyl)-1H-pyraz...)Show SMILES Cc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O2S/c1-11-2-4-12(5-3-11)15-10-16(17(18,19)20)22-23(15)13-6-8-14(9-7-13)26(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In Vitro activity of compound against human recombinant Prostaglandin G/H synthase 2 |
J Med Chem 43: 775-7 (2000)
BindingDB Entry DOI: 10.7270/Q2NV9HG1 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057564
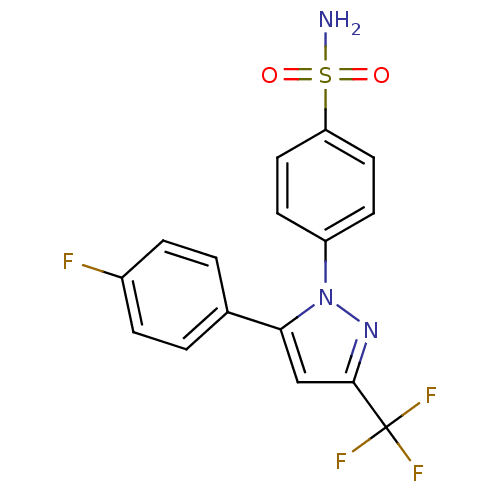
(4-[5-(4-Fluoro-phenyl)-3-trifluoromethyl-pyrazol-1...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(cc1-c1ccc(F)cc1)C(F)(F)F Show InChI InChI=1S/C16H11F4N3O2S/c17-11-3-1-10(2-4-11)14-9-15(16(18,19)20)22-23(14)12-5-7-13(8-6-12)26(21,24)25/h1-9H,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057566
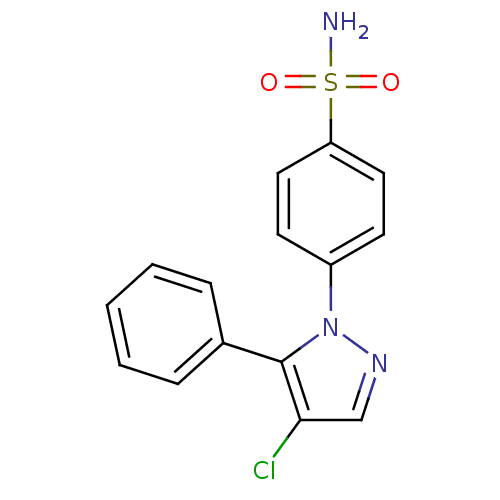
(4-(4-Chloro-5-phenyl-pyrazol-1-yl)-benzenesulfonam...)Show InChI InChI=1S/C15H12ClN3O2S/c16-14-10-18-19(15(14)11-4-2-1-3-5-11)12-6-8-13(9-7-12)22(17,20)21/h1-10H,(H2,17,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 49 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057583
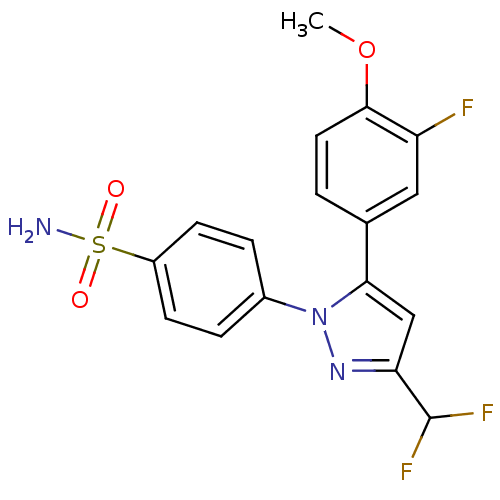
(4-[3-Difluoromethyl-5-(3-fluoro-4-methoxy-phenyl)-...)Show SMILES COc1ccc(cc1F)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)F Show InChI InChI=1S/C17H14F3N3O3S/c1-26-16-7-2-10(8-13(16)18)15-9-14(17(19)20)22-23(15)11-3-5-12(6-4-11)27(21,24)25/h2-9,17H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50286051
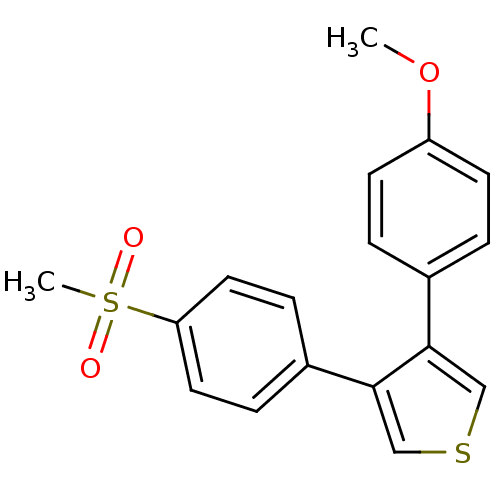
(3-(4-Methanesulfonyl-phenyl)-4-(4-methoxy-phenyl)-...)Show InChI InChI=1S/C18H16O3S2/c1-21-15-7-3-13(4-8-15)17-11-22-12-18(17)14-5-9-16(10-6-14)23(2,19)20/h3-12H,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against human recombinant Prostaglandin G/H synthase 2 |
Bioorg Med Chem Lett 5: 2919-2922 (1995)
Article DOI: 10.1016/0960-894X(95)00512-R
BindingDB Entry DOI: 10.7270/Q2HM58D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057533
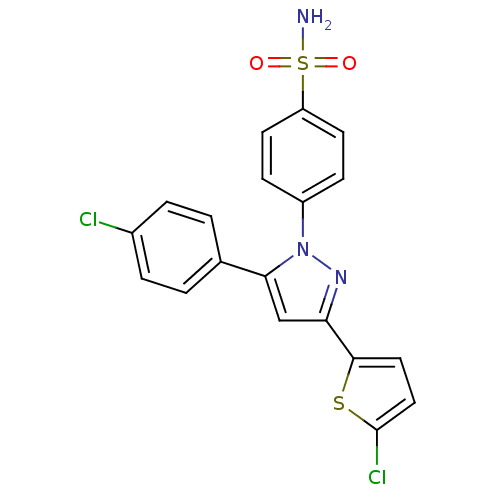
(4-[5-(4-Chloro-phenyl)-3-(5-chloro-thiophen-2-yl)-...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(cc1-c1ccc(Cl)cc1)-c1ccc(Cl)s1 Show InChI InChI=1S/C19H13Cl2N3O2S2/c20-13-3-1-12(2-4-13)17-11-16(18-9-10-19(21)27-18)23-24(17)14-5-7-15(8-6-14)28(22,25)26/h1-11H,(H2,22,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 52 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data