

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50207816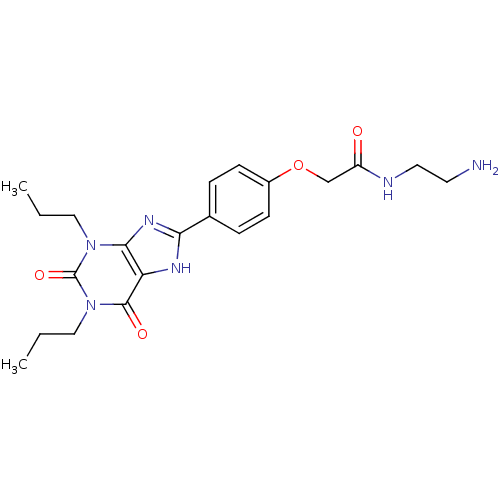 (CHEMBL273094 | N-(2-Amino-ethyl)-2-[4-(2,6-dioxo-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086173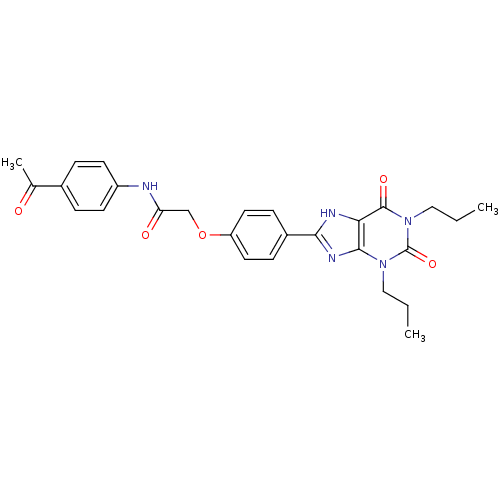 (CHEMBL17002 | N-(4-Acetyl-phenyl)-2-[4-(2,6-dioxo-...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | PubMed | 1.39 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086168 (2-(4-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.48 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086167 (2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 1.52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50025687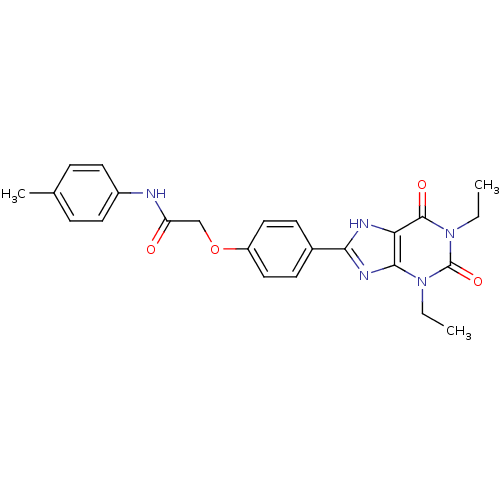 (2-[4-(1,3-Diethyl-2,6-dioxo-2,3,6,7-tetrahydro-1H-...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.78 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50021080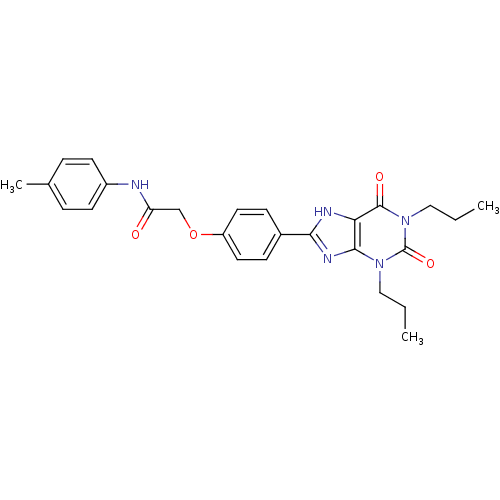 (2-(4-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.88 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086170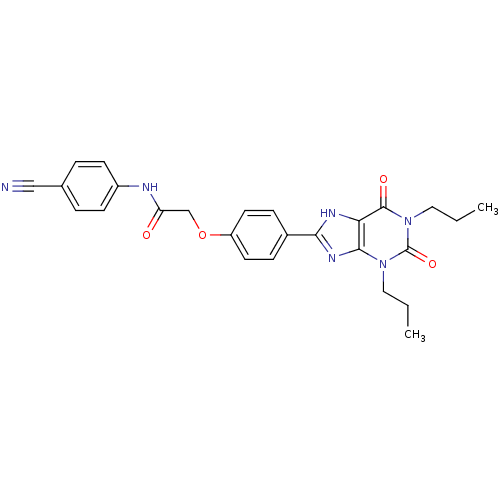 ((4-Cyano-phenyl)-carbamic acid 4-(2,6-dioxo-1,3-di...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 1.97 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086166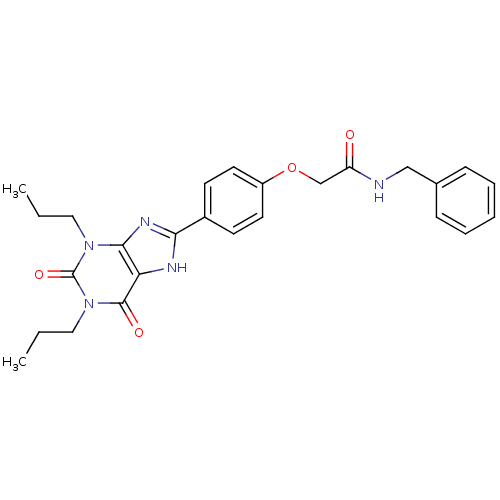 (CHEMBL275605 | N-Benzyl-2-[4-(2,6-dioxo-1,3-diprop...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.04 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086165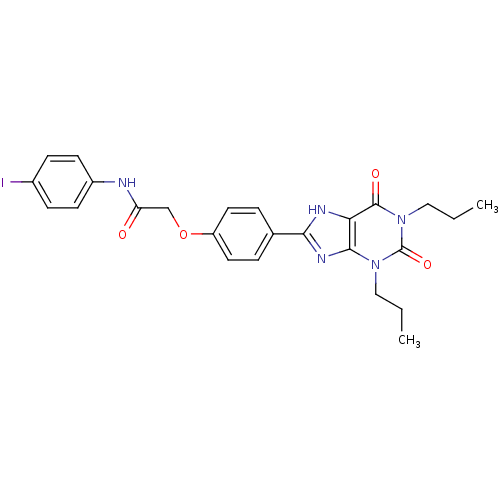 (2-(4-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086174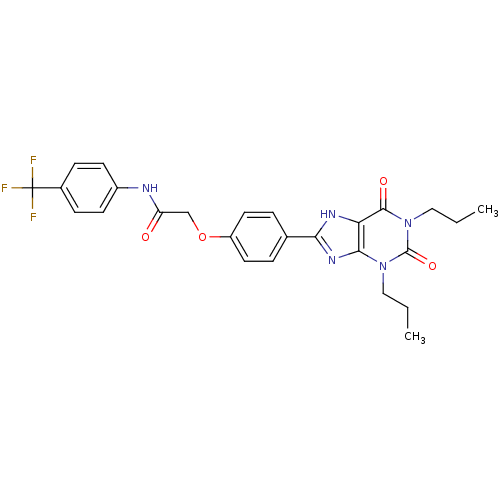 (2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086182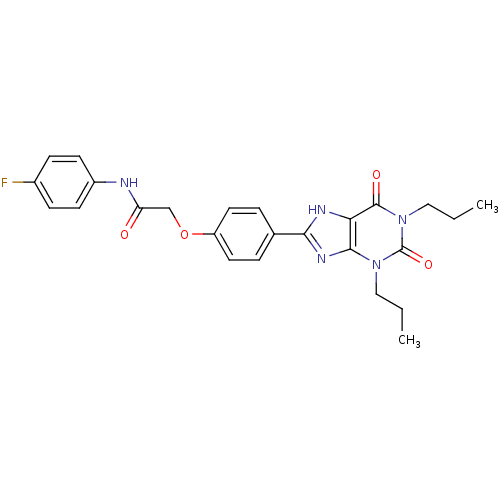 (2-(4-(2,4-dioxo-1,3-dipropyl-2,3,4,5-tetrahydro-1H...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086184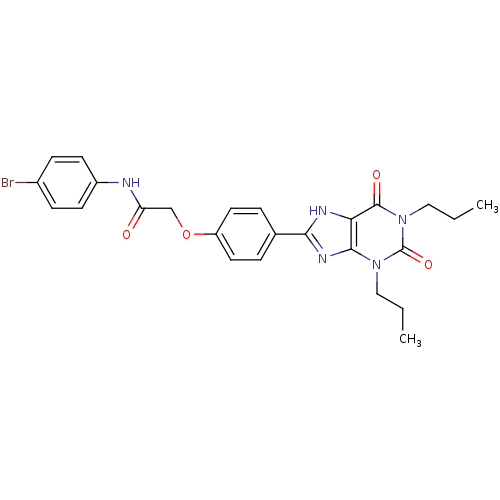 (CHEMBL17003 | N-(4-Bromo-phenyl)-2-[4-(2,6-dioxo-1...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086164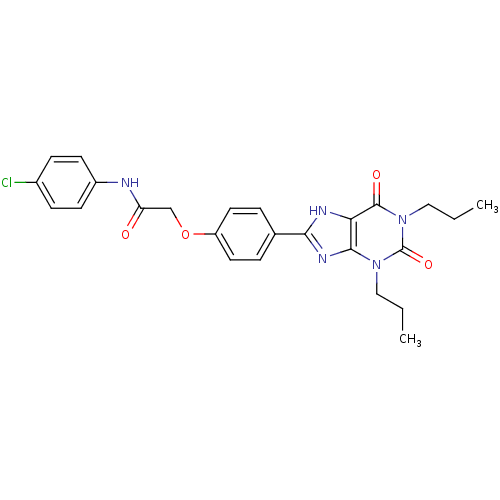 (CHEMBL275427 | N-(4-Chloro-phenyl)-2-[4-(2,6-dioxo...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.47 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086186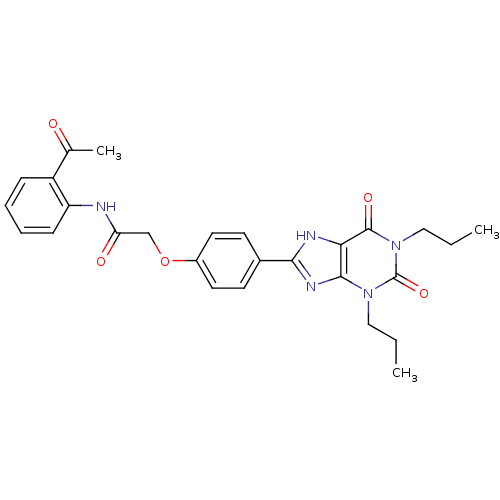 (CHEMBL17341 | N-(2-Acetyl-phenyl)-2-[4-(2,6-dioxo-...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 2.74 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086177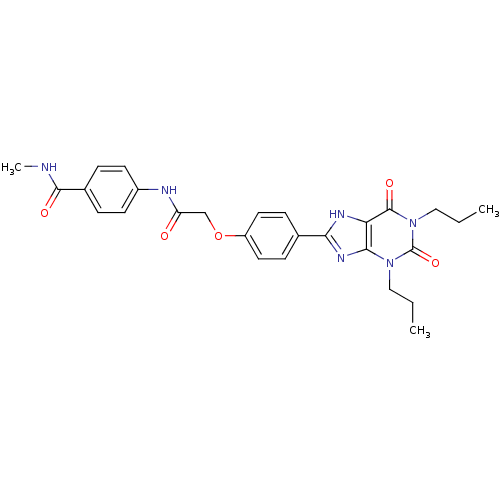 (4-{2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.34 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086181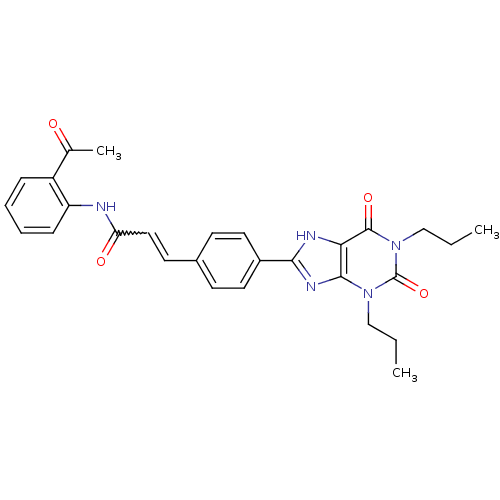 (CHEMBL17202 | N-(2-Acetyl-phenyl)-3-[4-(2,6-dioxo-...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50021085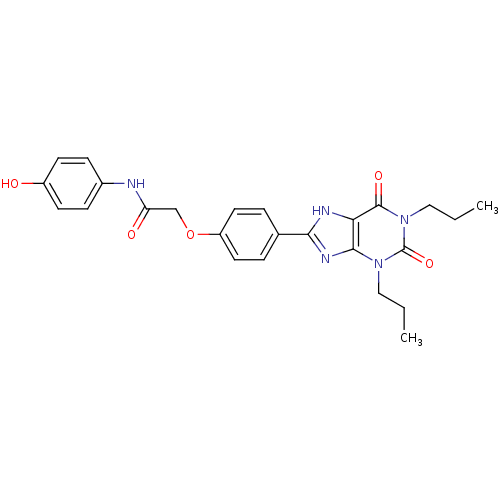 (2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.71 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086172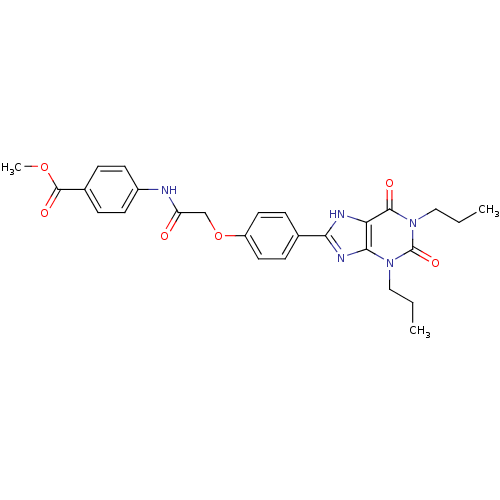 (4-{2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.93 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50086163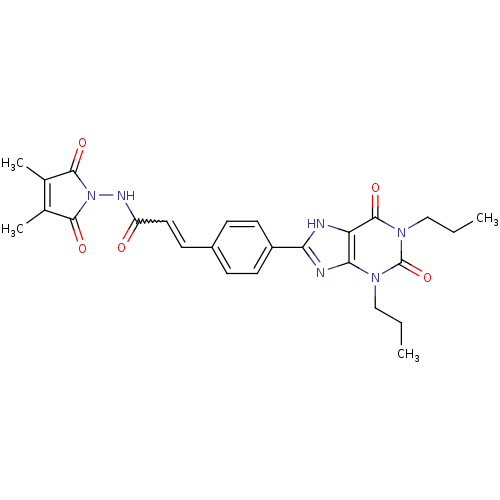 (CHEMBL17294 | N-(3,4-Dimethyl-2,5-dioxo-2,5-dihydr...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.94 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50086168 (2-(4-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 4.22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086187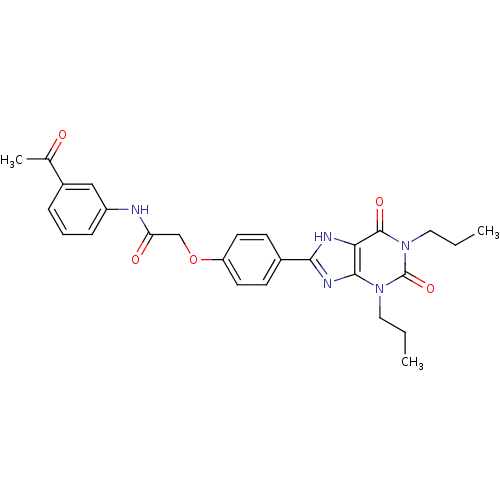 (CHEMBL16534 | N-(3-Acetyl-phenyl)-2-[4-(2,6-dioxo-...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 4.92 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50086166 (CHEMBL275605 | N-Benzyl-2-[4-(2,6-dioxo-1,3-diprop...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 5.02 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086178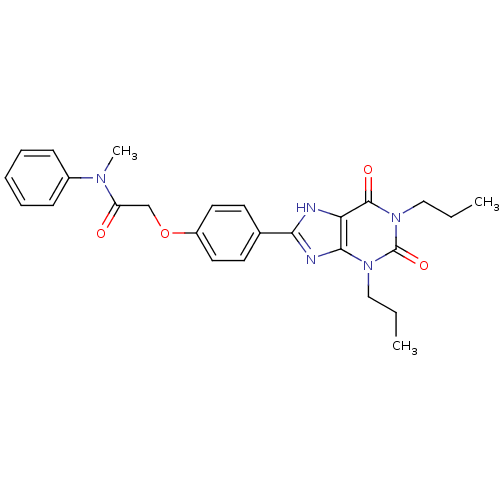 (2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 5.42 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50021085 (2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 5.88 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50086164 (CHEMBL275427 | N-(4-Chloro-phenyl)-2-[4-(2,6-dioxo...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 6.35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Homo sapiens (Human)) | BDBM50207816 (CHEMBL273094 | N-(2-Amino-ethyl)-2-[4-(2,6-dioxo-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 6.82 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against recombinant human adenosine A1 receptor expressed in HEK-293 cells in presence of [125I]-IABA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50086184 (CHEMBL17003 | N-(4-Bromo-phenyl)-2-[4-(2,6-dioxo-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 7.46 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50086181 (CHEMBL17202 | N-(2-Acetyl-phenyl)-3-[4-(2,6-dioxo-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 7.67 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50207816 (CHEMBL273094 | N-(2-Amino-ethyl)-2-[4-(2,6-dioxo-1...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 7.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086171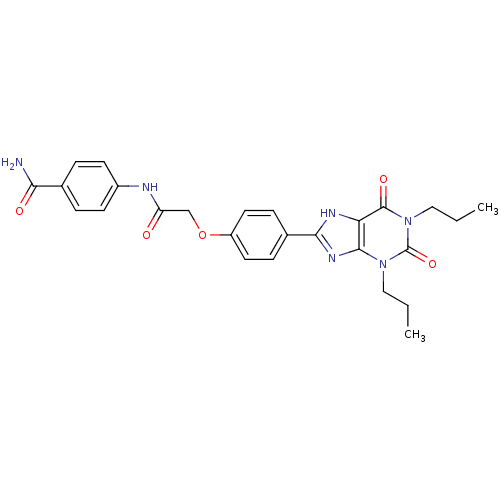 (4-{2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 7.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086176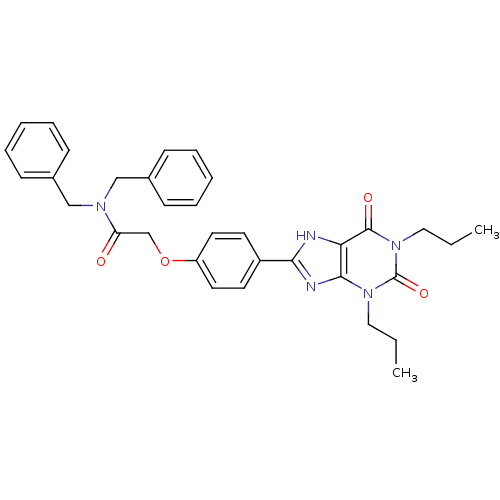 (CHEMBL275149 | N,N-Dibenzyl-2-[4-(2,6-dioxo-1,3-di...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 9.88 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50086171 (4-{2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50086179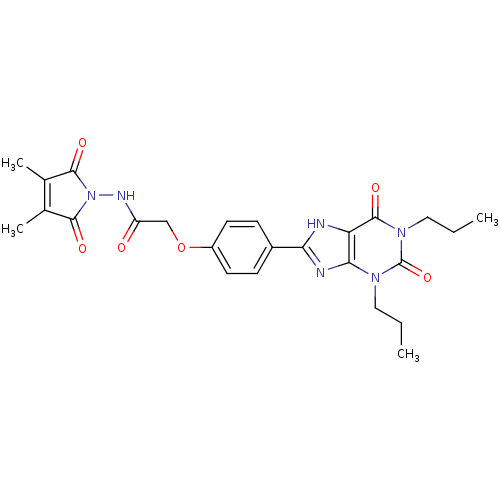 (CHEMBL17173 | N-(3,4-Dimethyl-2,5-dioxo-2,5-dihydr...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 11.1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50086167 (2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 13.1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50037440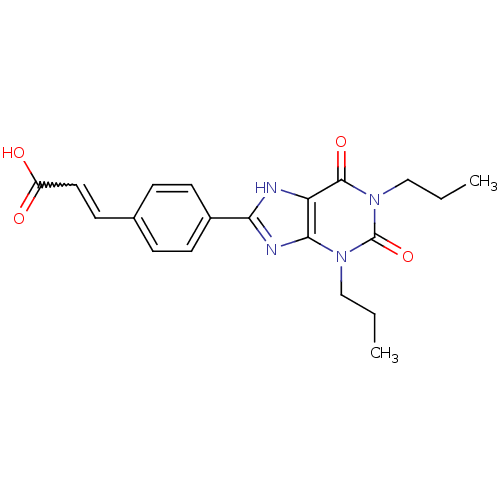 ((E)-3-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydr...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50025690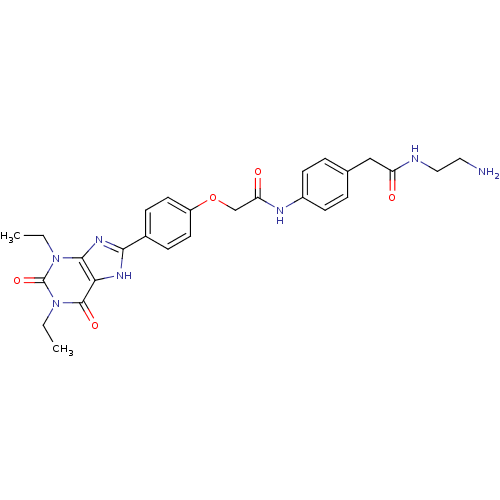 (CHEMBL17141 | N-{4-[(2-Amino-ethylcarbamoyl)-methy...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 15.2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50086165 (2-(4-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 15.7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086169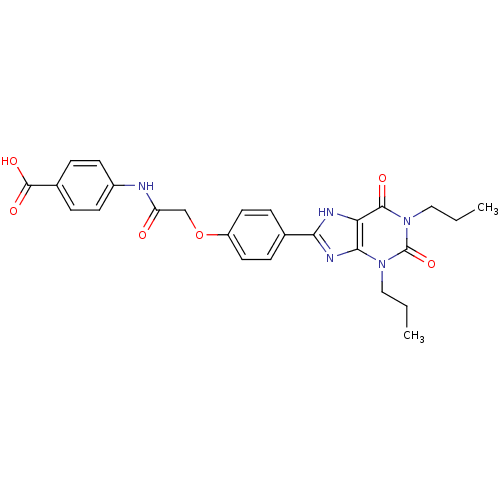 (4-{2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 16.1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50021077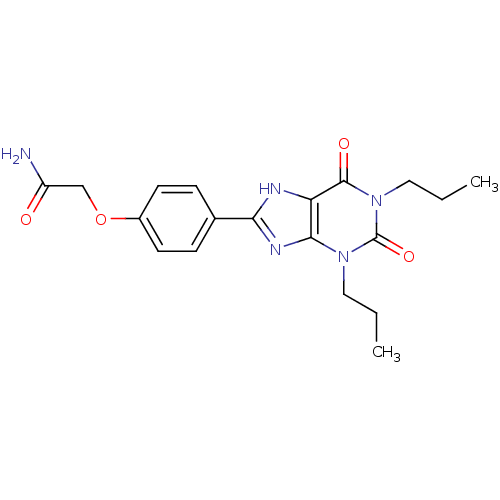 (2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 16.3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50086182 (2-(4-(2,4-dioxo-1,3-dipropyl-2,3,4,5-tetrahydro-1H...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 16.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against recombinant human adenosine A2A receptor expressed in HEK-293 cells in presence of [125I]-iodo-ZM241385 radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2b (Homo sapiens (Human)) | BDBM50086163 (CHEMBL17294 | N-(3,4-Dimethyl-2,5-dioxo-2,5-dihydr...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 16.7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human A2B adenosine receptor expressed in HEK-293 cells uisng [3H]-ZM-241,385 or [125I]-IABOPX | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50086170 ((4-Cyano-phenyl)-carbamic acid 4-(2,6-dioxo-1,3-di...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 16.8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50021080 (2-(4-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 17.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Homo sapiens (Human)) | BDBM50086182 (2-(4-(2,4-dioxo-1,3-dipropyl-2,3,4,5-tetrahydro-1H...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 17.9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against recombinant human adenosine A1 receptor expressed in HEK-293 cells in presence of [125I]-IABA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50207816 (CHEMBL273094 | N-(2-Amino-ethyl)-2-[4-(2,6-dioxo-1...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB PubMed | 18.4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against recombinant human adenosine A2A receptor expressed in HEK-293 cells in presence of [125I]-iodo-ZM241385 radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50021077 (2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50086166 (CHEMBL275605 | N-Benzyl-2-[4-(2,6-dioxo-1,3-diprop...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 23.8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against recombinant human adenosine A2A receptor expressed in HEK-293 cells in presence of [125I]-iodo-ZM241385 radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50086177 (4-{2-[4-(2,6-Dioxo-1,3-dipropyl-2,3,6,7-tetrahydro...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 24.8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against adenosine A1 receptor in rat brain membrane in presence of [3H]-R-PIA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A3 (Homo sapiens (Human)) | BDBM50207816 (CHEMBL273094 | N-(2-Amino-ethyl)-2-[4-(2,6-dioxo-1...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PubMed | 25.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against human adenosine A3 receptor expressed in HEK cells in presence of [125]IAB-MECA or [125I]-IABA radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50086168 (2-(4-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 25.8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Antagonist activity against recombinant human adenosine A2A receptor expressed in HEK-293 cells in presence of [125I]-iodo-ZM241385 radioligand. | J Med Chem 43: 1165-72 (2000) BindingDB Entry DOI: 10.7270/Q2M044NW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 182 total ) | Next | Last >> |