 Found 57 hits Enz. Inhib. hit(s) with all data for entry = 50034057
Found 57 hits Enz. Inhib. hit(s) with all data for entry = 50034057 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356075
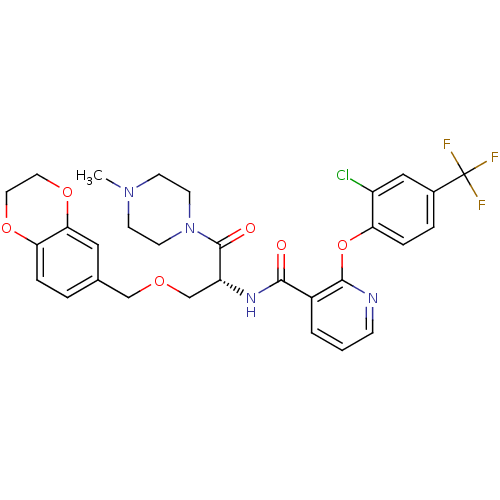
(CHEMBL1911818)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccc2OCCOc2c1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C30H30ClF3N4O6/c1-37-9-11-38(12-10-37)29(40)23(18-41-17-19-4-6-25-26(15-19)43-14-13-42-25)36-27(39)21-3-2-8-35-28(21)44-24-7-5-20(16-22(24)31)30(32,33)34/h2-8,15-16,23H,9-14,17-18H2,1H3,(H,36,39)/t23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS activity in human A549 cells assessed as amount of GM1 on the cell membrane after 72 hrs by FL-CTB-based fluorescent microscopy |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356076
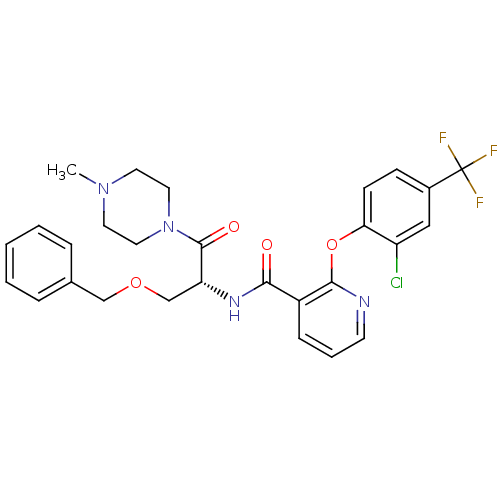
(CHEMBL1911817)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C28H28ClF3N4O4/c1-35-12-14-36(15-13-35)27(38)23(18-39-17-19-6-3-2-4-7-19)34-25(37)21-8-5-11-33-26(21)40-24-10-9-20(16-22(24)29)28(30,31)32/h2-11,16,23H,12-15,17-18H2,1H3,(H,34,37)/t23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356075

(CHEMBL1911818)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccc2OCCOc2c1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C30H30ClF3N4O6/c1-37-9-11-38(12-10-37)29(40)23(18-41-17-19-4-6-25-26(15-19)43-14-13-42-25)36-27(39)21-3-2-8-35-28(21)44-24-7-5-20(16-22(24)31)30(32,33)34/h2-8,15-16,23H,9-14,17-18H2,1H3,(H,36,39)/t23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356076

(CHEMBL1911817)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C28H28ClF3N4O4/c1-35-12-14-36(15-13-35)27(38)23(18-39-17-19-6-3-2-4-7-19)34-25(37)21-8-5-11-33-26(21)40-24-10-9-20(16-22(24)29)28(30,31)32/h2-11,16,23H,12-15,17-18H2,1H3,(H,34,37)/t23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS activity in human A549 cells assessed as amount of GM1 on the cell membrane after 72 hrs by FL-CTB-based fluorescent microscopy |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356091
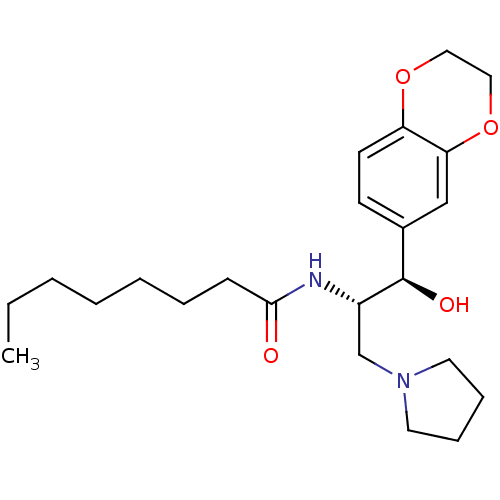
(CHEMBL1911679)Show SMILES CCCCCCCC(=O)N[C@@H](CN1CCCC1)[C@H](O)c1ccc2OCCOc2c1 |r| Show InChI InChI=1S/C23H36N2O4/c1-2-3-4-5-6-9-22(26)24-19(17-25-12-7-8-13-25)23(27)18-10-11-20-21(16-18)29-15-14-28-20/h10-11,16,19,23,27H,2-9,12-15,17H2,1H3,(H,24,26)/t19-,23+/m0/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS activity in human A549 cells assessed as amount of GM1 on the cell membrane after 72 hrs by FL-CTB-based fluorescent microscopy |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356090
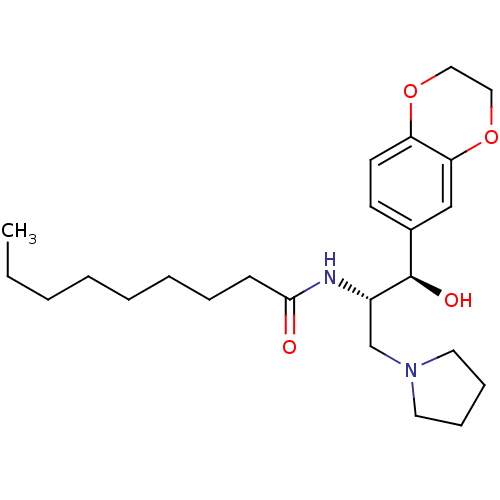
(CHEMBL1911678)Show SMILES CCCCCCCCC(=O)N[C@@H](CN1CCCC1)[C@H](O)c1ccc2OCCOc2c1 |r| Show InChI InChI=1S/C24H38N2O4/c1-2-3-4-5-6-7-10-23(27)25-20(18-26-13-8-9-14-26)24(28)19-11-12-21-22(17-19)30-16-15-29-21/h11-12,17,20,24,28H,2-10,13-16,18H2,1H3,(H,25,27)/t20-,24+/m0/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS activity in human A549 cells assessed as amount of GM1 on the cell membrane after 72 hrs by FL-CTB-based fluorescent microscopy |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356078
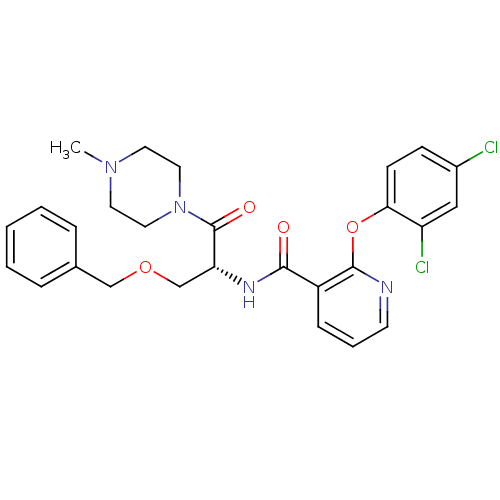
(CHEMBL1911815)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(Cl)cc1Cl |r| Show InChI InChI=1S/C27H28Cl2N4O4/c1-32-12-14-33(15-13-32)27(35)23(18-36-17-19-6-3-2-4-7-19)31-25(34)21-8-5-11-30-26(21)37-24-10-9-20(28)16-22(24)29/h2-11,16,23H,12-15,17-18H2,1H3,(H,31,34)/t23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356077
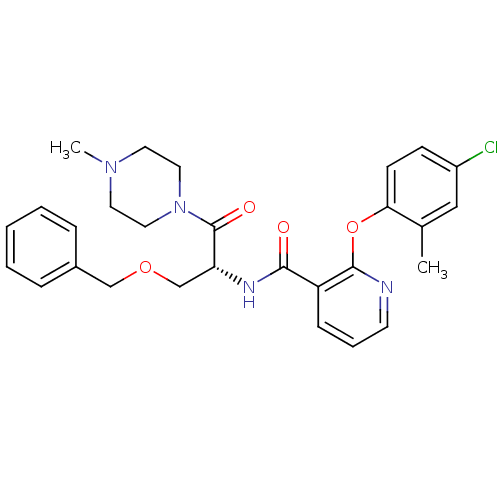
(CHEMBL1911816)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(Cl)cc1C |r| Show InChI InChI=1S/C28H31ClN4O4/c1-20-17-22(29)10-11-25(20)37-27-23(9-6-12-30-27)26(34)31-24(19-36-18-21-7-4-3-5-8-21)28(35)33-15-13-32(2)14-16-33/h3-12,17,24H,13-16,18-19H2,1-2H3,(H,31,34)/t24-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356079
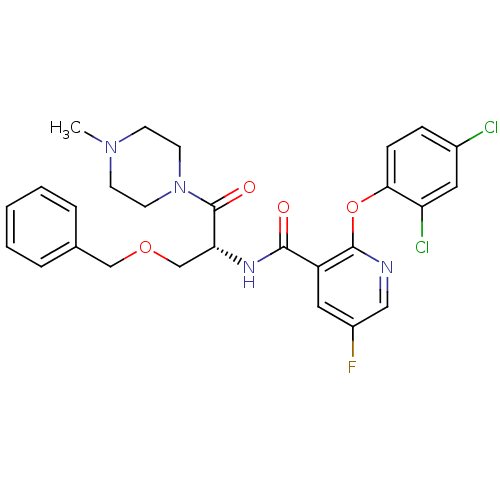
(CHEMBL1911814)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cc(F)cnc1Oc1ccc(Cl)cc1Cl |r| Show InChI InChI=1S/C27H27Cl2FN4O4/c1-33-9-11-34(12-10-33)27(36)23(17-37-16-18-5-3-2-4-6-18)32-25(35)21-14-20(30)15-31-26(21)38-24-8-7-19(28)13-22(24)29/h2-8,13-15,23H,9-12,16-17H2,1H3,(H,32,35)/t23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356094

(CHEMBL1911829)Show SMILES FC(F)(F)c1ccc(Oc2ncccc2C(=O)N[C@H](COCc2ccccc2)C(=O)N[C@@H]2CN3CCC2CC3)c(Cl)c1 |r,wD:18.18,31.32,(8.76,-41.16,;8.77,-39.62,;10.11,-38.85,;10.1,-40.39,;7.44,-38.85,;6.1,-39.61,;4.77,-38.83,;4.79,-37.3,;3.46,-36.52,;3.47,-34.98,;4.8,-34.21,;4.8,-32.67,;3.46,-31.9,;2.13,-32.67,;2.14,-34.2,;.8,-34.97,;.8,-36.51,;-.53,-34.2,;-1.86,-34.96,;-1.87,-36.5,;-3.2,-37.27,;-3.2,-38.81,;-1.87,-39.59,;-1.87,-41.13,;-.54,-41.91,;.8,-41.14,;.79,-39.59,;-.54,-38.83,;-3.2,-34.19,;-3.19,-32.65,;-4.53,-34.96,;-5.86,-34.19,;-7.2,-34.96,;-8.53,-34.18,;-8.53,-32.64,;-7.19,-31.88,;-5.86,-32.65,;-7.4,-32.68,;-7,-34.18,;6.11,-36.53,;6.11,-34.99,;7.44,-37.3,)| Show InChI InChI=1S/C30H30ClF3N4O4/c31-23-15-21(30(32,33)34)8-9-26(23)42-29-22(7-4-12-35-29)27(39)37-25(18-41-17-19-5-2-1-3-6-19)28(40)36-24-16-38-13-10-20(24)11-14-38/h1-9,12,15,20,24-25H,10-11,13-14,16-18H2,(H,36,40)(H,37,39)/t24-,25-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS activity in human A549 cells assessed as amount of GM1 on the cell membrane after 72 hrs by FL-CTB-based fluorescent microscopy |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356094

(CHEMBL1911829)Show SMILES FC(F)(F)c1ccc(Oc2ncccc2C(=O)N[C@H](COCc2ccccc2)C(=O)N[C@@H]2CN3CCC2CC3)c(Cl)c1 |r,wD:18.18,31.32,(8.76,-41.16,;8.77,-39.62,;10.11,-38.85,;10.1,-40.39,;7.44,-38.85,;6.1,-39.61,;4.77,-38.83,;4.79,-37.3,;3.46,-36.52,;3.47,-34.98,;4.8,-34.21,;4.8,-32.67,;3.46,-31.9,;2.13,-32.67,;2.14,-34.2,;.8,-34.97,;.8,-36.51,;-.53,-34.2,;-1.86,-34.96,;-1.87,-36.5,;-3.2,-37.27,;-3.2,-38.81,;-1.87,-39.59,;-1.87,-41.13,;-.54,-41.91,;.8,-41.14,;.79,-39.59,;-.54,-38.83,;-3.2,-34.19,;-3.19,-32.65,;-4.53,-34.96,;-5.86,-34.19,;-7.2,-34.96,;-8.53,-34.18,;-8.53,-32.64,;-7.19,-31.88,;-5.86,-32.65,;-7.4,-32.68,;-7,-34.18,;6.11,-36.53,;6.11,-34.99,;7.44,-37.3,)| Show InChI InChI=1S/C30H30ClF3N4O4/c31-23-15-21(30(32,33)34)8-9-26(23)42-29-22(7-4-12-35-29)27(39)37-25(18-41-17-19-5-2-1-3-6-19)28(40)36-24-16-38-13-10-20(24)11-14-38/h1-9,12,15,20,24-25H,10-11,13-14,16-18H2,(H,36,40)(H,37,39)/t24-,25-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356066
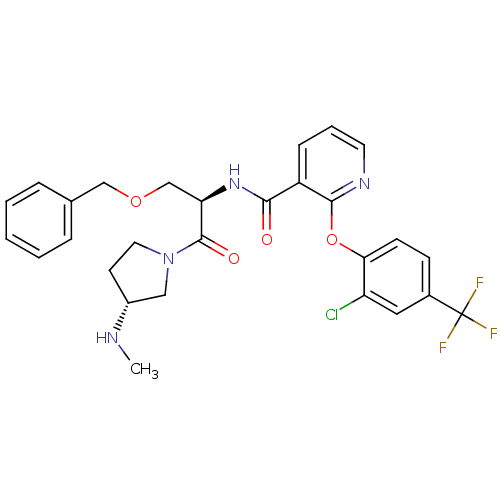
(CHEMBL1911827)Show SMILES CN[C@@H]1CCN(C1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C28H28ClF3N4O4/c1-33-20-11-13-36(15-20)27(38)23(17-39-16-18-6-3-2-4-7-18)35-25(37)21-8-5-12-34-26(21)40-24-10-9-19(14-22(24)29)28(30,31)32/h2-10,12,14,20,23,33H,11,13,15-17H2,1H3,(H,35,37)/t20-,23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS activity in human A549 cells assessed as amount of GM1 on the cell membrane after 72 hrs by FL-CTB-based fluorescent microscopy |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356074
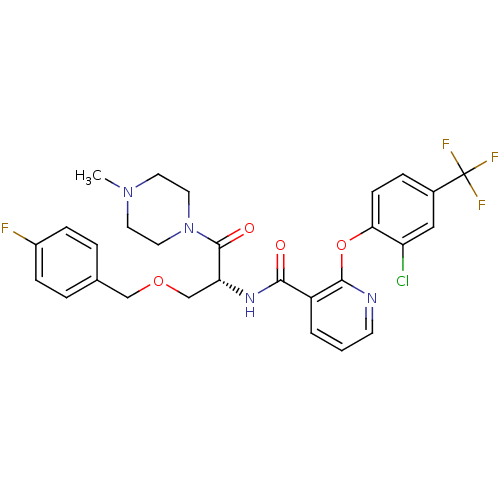
(CHEMBL1911819)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccc(F)cc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C28H27ClF4N4O4/c1-36-11-13-37(14-12-36)27(39)23(17-40-16-18-4-7-20(30)8-5-18)35-25(38)21-3-2-10-34-26(21)41-24-9-6-19(15-22(24)29)28(31,32)33/h2-10,15,23H,11-14,16-17H2,1H3,(H,35,38)/t23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356095
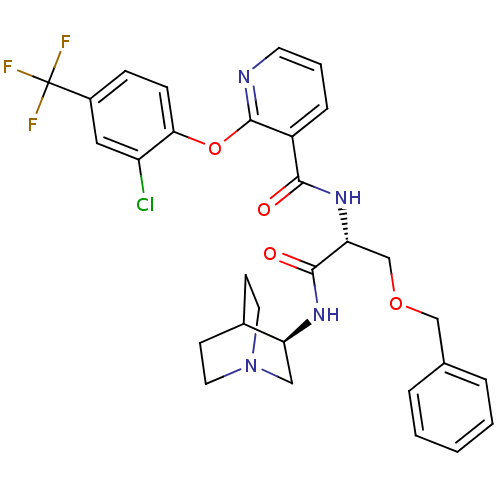
(CHEMBL1911830)Show SMILES FC(F)(F)c1ccc(Oc2ncccc2C(=O)N[C@H](COCc2ccccc2)C(=O)N[C@H]2CN3CCC2CC3)c(Cl)c1 |r,wU:31.32,wD:18.18,(32.59,-40.93,;32.59,-39.39,;33.93,-38.62,;33.92,-40.16,;31.26,-38.61,;29.92,-39.38,;28.59,-38.6,;28.61,-37.06,;27.28,-36.29,;27.29,-34.75,;28.62,-33.98,;28.63,-32.44,;27.29,-31.67,;25.96,-32.44,;25.96,-33.97,;24.63,-34.74,;24.62,-36.28,;23.3,-33.96,;21.96,-34.73,;21.96,-36.27,;20.62,-37.04,;20.62,-38.58,;21.96,-39.36,;21.95,-40.9,;23.28,-41.67,;24.62,-40.9,;24.62,-39.36,;23.28,-38.59,;20.63,-33.96,;20.63,-32.42,;19.29,-34.73,;17.96,-33.95,;16.62,-34.73,;15.29,-33.95,;15.3,-32.41,;16.64,-31.64,;17.96,-32.42,;16.42,-32.45,;16.82,-33.94,;29.94,-36.29,;29.93,-34.75,;31.26,-37.06,)| Show InChI InChI=1S/C30H30ClF3N4O4/c31-23-15-21(30(32,33)34)8-9-26(23)42-29-22(7-4-12-35-29)27(39)37-25(18-41-17-19-5-2-1-3-6-19)28(40)36-24-16-38-13-10-20(24)11-14-38/h1-9,12,15,20,24-25H,10-11,13-14,16-18H2,(H,36,40)(H,37,39)/t24-,25+/m0/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356090

(CHEMBL1911678)Show SMILES CCCCCCCCC(=O)N[C@@H](CN1CCCC1)[C@H](O)c1ccc2OCCOc2c1 |r| Show InChI InChI=1S/C24H38N2O4/c1-2-3-4-5-6-7-10-23(27)25-20(18-26-13-8-9-14-26)24(28)19-11-12-21-22(17-19)30-16-15-29-21/h11-12,17,20,24,28H,2-10,13-16,18H2,1H3,(H,25,27)/t20-,24+/m0/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356067
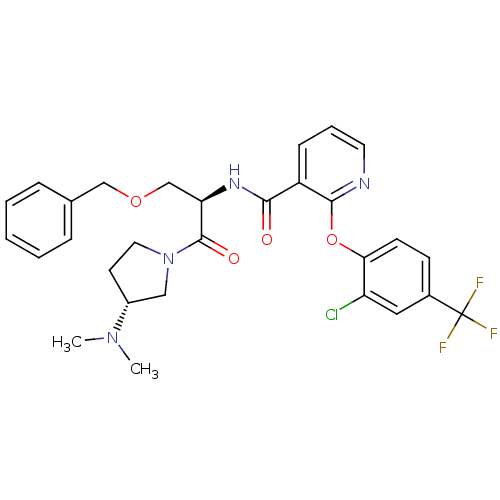
(CHEMBL1911826)Show SMILES CN(C)[C@@H]1CCN(C1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C29H30ClF3N4O4/c1-36(2)21-12-14-37(16-21)28(39)24(18-40-17-19-7-4-3-5-8-19)35-26(38)22-9-6-13-34-27(22)41-25-11-10-20(15-23(25)30)29(31,32)33/h3-11,13,15,21,24H,12,14,16-18H2,1-2H3,(H,35,38)/t21-,24-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356095

(CHEMBL1911830)Show SMILES FC(F)(F)c1ccc(Oc2ncccc2C(=O)N[C@H](COCc2ccccc2)C(=O)N[C@H]2CN3CCC2CC3)c(Cl)c1 |r,wU:31.32,wD:18.18,(32.59,-40.93,;32.59,-39.39,;33.93,-38.62,;33.92,-40.16,;31.26,-38.61,;29.92,-39.38,;28.59,-38.6,;28.61,-37.06,;27.28,-36.29,;27.29,-34.75,;28.62,-33.98,;28.63,-32.44,;27.29,-31.67,;25.96,-32.44,;25.96,-33.97,;24.63,-34.74,;24.62,-36.28,;23.3,-33.96,;21.96,-34.73,;21.96,-36.27,;20.62,-37.04,;20.62,-38.58,;21.96,-39.36,;21.95,-40.9,;23.28,-41.67,;24.62,-40.9,;24.62,-39.36,;23.28,-38.59,;20.63,-33.96,;20.63,-32.42,;19.29,-34.73,;17.96,-33.95,;16.62,-34.73,;15.29,-33.95,;15.3,-32.41,;16.64,-31.64,;17.96,-32.42,;16.42,-32.45,;16.82,-33.94,;29.94,-36.29,;29.93,-34.75,;31.26,-37.06,)| Show InChI InChI=1S/C30H30ClF3N4O4/c31-23-15-21(30(32,33)34)8-9-26(23)42-29-22(7-4-12-35-29)27(39)37-25(18-41-17-19-5-2-1-3-6-19)28(40)36-24-16-38-13-10-20(24)11-14-38/h1-9,12,15,20,24-25H,10-11,13-14,16-18H2,(H,36,40)(H,37,39)/t24-,25+/m0/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS activity in human A549 cells assessed as amount of GM1 on the cell membrane after 72 hrs by FL-CTB-based fluorescent microscopy |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356074

(CHEMBL1911819)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccc(F)cc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C28H27ClF4N4O4/c1-36-11-13-37(14-12-36)27(39)23(17-40-16-18-4-7-20(30)8-5-18)35-25(38)21-3-2-10-34-26(21)41-24-9-6-19(15-22(24)29)28(31,32)33/h2-10,15,23H,11-14,16-17H2,1H3,(H,35,38)/t23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS activity in human A549 cells assessed as amount of GM1 on the cell membrane after 72 hrs by FL-CTB-based fluorescent microscopy |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356091

(CHEMBL1911679)Show SMILES CCCCCCCC(=O)N[C@@H](CN1CCCC1)[C@H](O)c1ccc2OCCOc2c1 |r| Show InChI InChI=1S/C23H36N2O4/c1-2-3-4-5-6-9-22(26)24-19(17-25-12-7-8-13-25)23(27)18-10-11-20-21(16-18)29-15-14-28-20/h10-11,16,19,23,27H,2-9,12-15,17H2,1H3,(H,24,26)/t19-,23+/m0/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356066

(CHEMBL1911827)Show SMILES CN[C@@H]1CCN(C1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C28H28ClF3N4O4/c1-33-20-11-13-36(15-20)27(38)23(17-39-16-18-6-3-2-4-7-18)35-25(37)21-8-5-12-34-26(21)40-24-10-9-19(14-22(24)29)28(30,31)32/h2-10,12,14,20,23,33H,11,13,15-17H2,1H3,(H,35,37)/t20-,23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356073
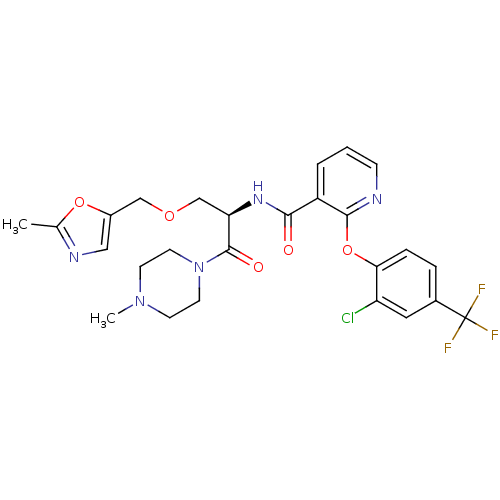
(CHEMBL1911820)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1cnc(C)o1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C26H27ClF3N5O5/c1-16-32-13-18(39-16)14-38-15-21(25(37)35-10-8-34(2)9-11-35)33-23(36)19-4-3-7-31-24(19)40-22-6-5-17(12-20(22)27)26(28,29)30/h3-7,12-13,21H,8-11,14-15H2,1-2H3,(H,33,36)/t21-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356092

(CHEMBL1911680)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(Cl)cc1 |r| Show InChI InChI=1S/C27H29ClN4O4/c1-31-14-16-32(17-15-31)27(34)24(19-35-18-20-6-3-2-4-7-20)30-25(33)23-8-5-13-29-26(23)36-22-11-9-21(28)10-12-22/h2-13,24H,14-19H2,1H3,(H,30,33)/t24-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356068
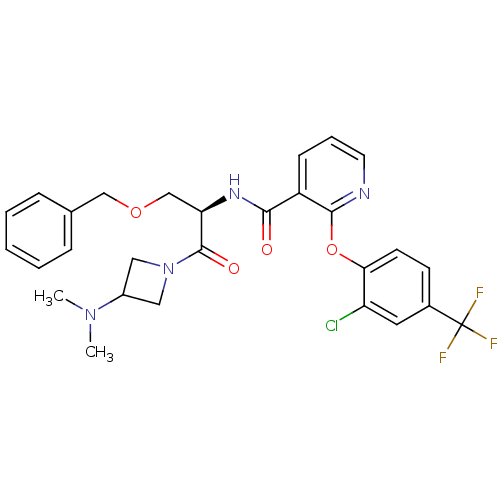
(CHEMBL1911825)Show SMILES CN(C)C1CN(C1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C28H28ClF3N4O4/c1-35(2)20-14-36(15-20)27(38)23(17-39-16-18-7-4-3-5-8-18)34-25(37)21-9-6-12-33-26(21)40-24-11-10-19(13-22(24)29)28(30,31)32/h3-13,20,23H,14-17H2,1-2H3,(H,34,37)/t23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356071

(CHEMBL1911822)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccnc(Cl)c1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C27H26Cl2F3N5O4/c1-36-9-11-37(12-10-36)26(39)21(16-40-15-17-6-8-33-23(29)13-17)35-24(38)19-3-2-7-34-25(19)41-22-5-4-18(14-20(22)28)27(30,31)32/h2-8,13-14,21H,9-12,15-16H2,1H3,(H,35,38)/t21-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356069
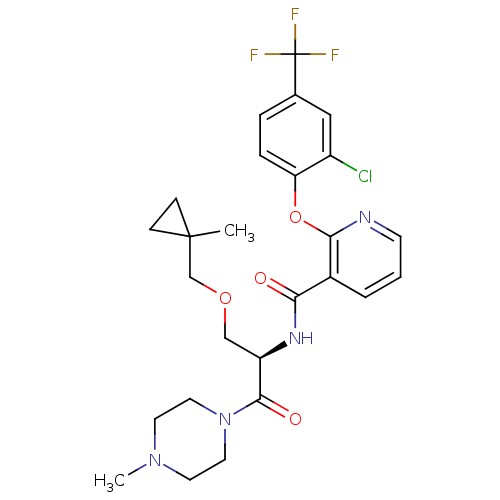
(CHEMBL1911824)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCC1(C)CC1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C26H30ClF3N4O4/c1-25(7-8-25)16-37-15-20(24(36)34-12-10-33(2)11-13-34)32-22(35)18-4-3-9-31-23(18)38-21-6-5-17(14-19(21)27)26(28,29)30/h3-6,9,14,20H,7-8,10-13,15-16H2,1-2H3,(H,32,35)/t20-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356073

(CHEMBL1911820)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1cnc(C)o1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C26H27ClF3N5O5/c1-16-32-13-18(39-16)14-38-15-21(25(37)35-10-8-34(2)9-11-35)33-23(36)19-4-3-7-31-24(19)40-22-6-5-17(12-20(22)27)26(28,29)30/h3-7,12-13,21H,8-11,14-15H2,1-2H3,(H,33,36)/t21-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS activity in human A549 cells assessed as amount of GM1 on the cell membrane after 72 hrs by FL-CTB-based fluorescent microscopy |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356089

(CHEMBL1911682)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(C)cc1 |r| Show InChI InChI=1S/C28H32N4O4/c1-21-10-12-23(13-11-21)36-27-24(9-6-14-29-27)26(33)30-25(20-35-19-22-7-4-3-5-8-22)28(34)32-17-15-31(2)16-18-32/h3-14,25H,15-20H2,1-2H3,(H,30,33)/t25-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 53 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356065

(CHEMBL1911828)Show SMILES CN[C@H]1CCN(C1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C28H28ClF3N4O4/c1-33-20-11-13-36(15-20)27(38)23(17-39-16-18-6-3-2-4-7-18)35-25(37)21-8-5-12-34-26(21)40-24-10-9-19(14-22(24)29)28(30,31)32/h2-10,12,14,20,23,33H,11,13,15-17H2,1H3,(H,35,37)/t20-,23+/m0/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 65 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50299749
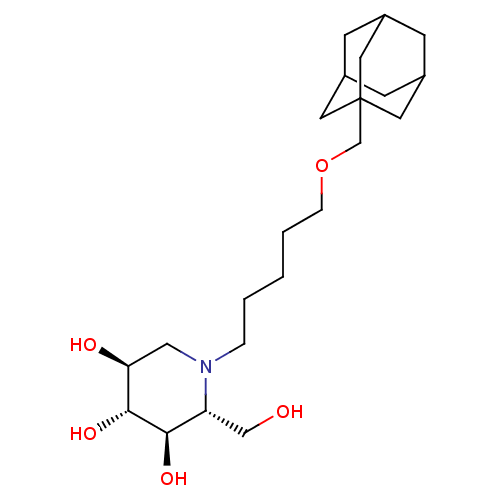
((2R,3R,4R,5S)-1-[5-(Adamantan-1-ylmethoxy)-pentyl]...)Show SMILES OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)CN1CCCCCOCC12CC3CC(CC(C3)C1)C2 |r,TLB:21:22:19.20.25:26,THB:21:20:26:27.22.23,23:22:19:25.24.26,23:24:19:27.21.22| Show InChI InChI=1S/C22H39NO5/c24-13-18-20(26)21(27)19(25)12-23(18)4-2-1-3-5-28-14-22-9-15-6-16(10-22)8-17(7-15)11-22/h15-21,24-27H,1-14H2/t15?,16?,17?,18-,19+,20-,21-,22?/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356082
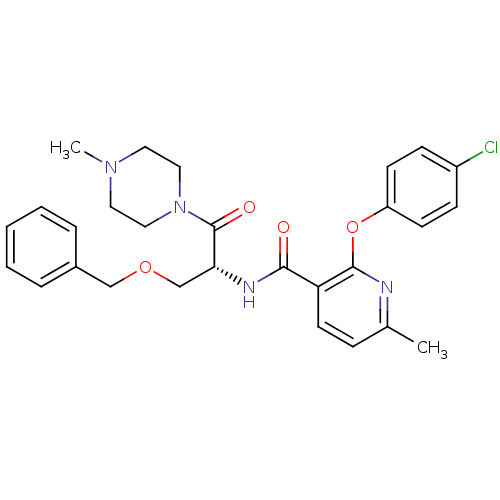
(CHEMBL1911811)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1ccc(C)nc1Oc1ccc(Cl)cc1 |r| Show InChI InChI=1S/C28H31ClN4O4/c1-20-8-13-24(27(30-20)37-23-11-9-22(29)10-12-23)26(34)31-25(19-36-18-21-6-4-3-5-7-21)28(35)33-16-14-32(2)15-17-33/h3-13,25H,14-19H2,1-2H3,(H,31,34)/t25-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356081
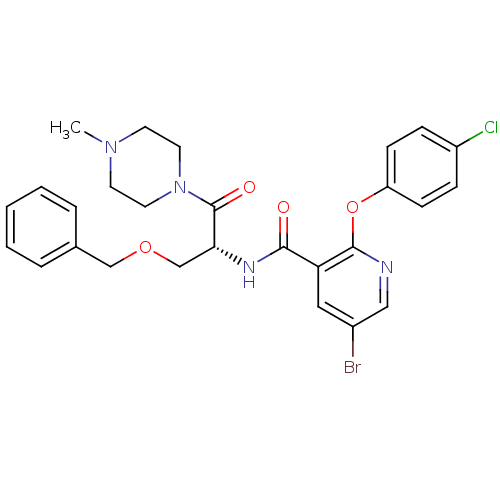
(CHEMBL1911812)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cc(Br)cnc1Oc1ccc(Cl)cc1 |r| Show InChI InChI=1S/C27H28BrClN4O4/c1-32-11-13-33(14-12-32)27(35)24(18-36-17-19-5-3-2-4-6-19)31-25(34)23-15-20(28)16-30-26(23)37-22-9-7-21(29)8-10-22/h2-10,15-16,24H,11-14,17-18H2,1H3,(H,31,34)/t24-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356070

(CHEMBL1911823)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCC(C)(C)C)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C26H32ClF3N4O4/c1-25(2,3)16-37-15-20(24(36)34-12-10-33(4)11-13-34)32-22(35)18-6-5-9-31-23(18)38-21-8-7-17(14-19(21)27)26(28,29)30/h5-9,14,20H,10-13,15-16H2,1-4H3,(H,32,35)/t20-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356080
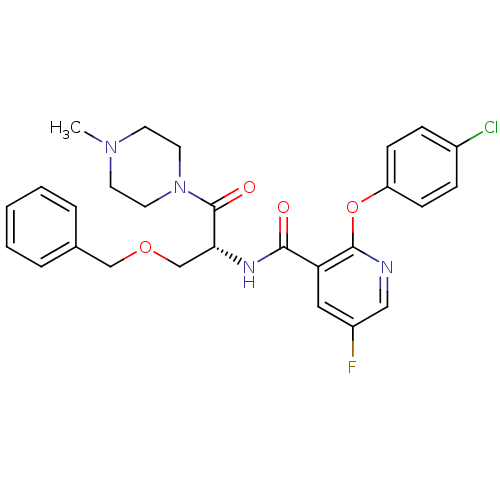
(CHEMBL1911813)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cc(F)cnc1Oc1ccc(Cl)cc1 |r| Show InChI InChI=1S/C27H28ClFN4O4/c1-32-11-13-33(14-12-32)27(35)24(18-36-17-19-5-3-2-4-6-19)31-25(34)23-15-21(29)16-30-26(23)37-22-9-7-20(28)8-10-22/h2-10,15-16,24H,11-14,17-18H2,1H3,(H,31,34)/t24-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356086
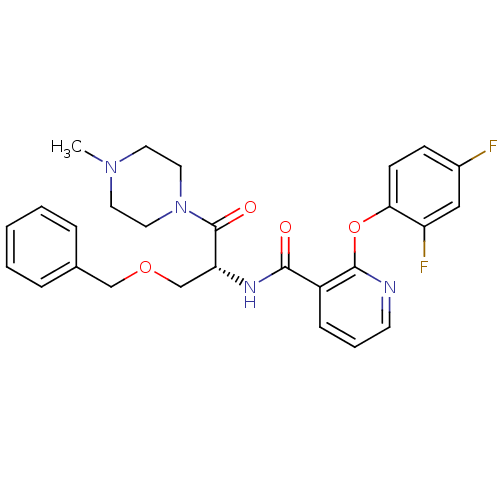
(CHEMBL1911685)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(F)cc1F |r| Show InChI InChI=1S/C27H28F2N4O4/c1-32-12-14-33(15-13-32)27(35)23(18-36-17-19-6-3-2-4-7-19)31-25(34)21-8-5-11-30-26(21)37-24-10-9-20(28)16-22(24)29/h2-11,16,23H,12-15,17-18H2,1H3,(H,31,34)/t23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356072

(CHEMBL1911821)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccncc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C27H27ClF3N5O4/c1-35-11-13-36(14-12-35)26(38)22(17-39-16-18-6-9-32-10-7-18)34-24(37)20-3-2-8-33-25(20)40-23-5-4-19(15-21(23)28)27(29,30)31/h2-10,15,22H,11-14,16-17H2,1H3,(H,34,37)/t22-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 275 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356087
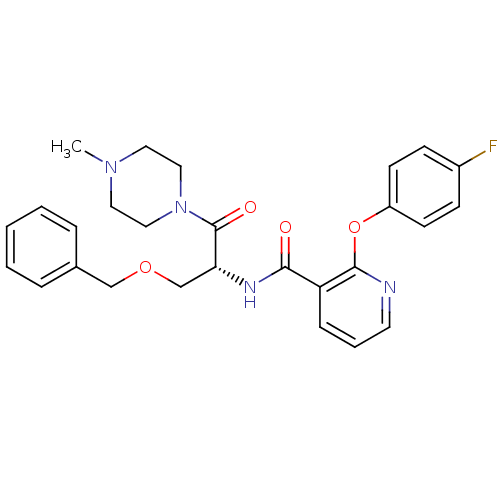
(CHEMBL1911684)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(F)cc1 |r| Show InChI InChI=1S/C27H29FN4O4/c1-31-14-16-32(17-15-31)27(34)24(19-35-18-20-6-3-2-4-7-20)30-25(33)23-8-5-13-29-26(23)36-22-11-9-21(28)10-12-22/h2-13,24H,14-19H2,1H3,(H,30,33)/t24-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356085
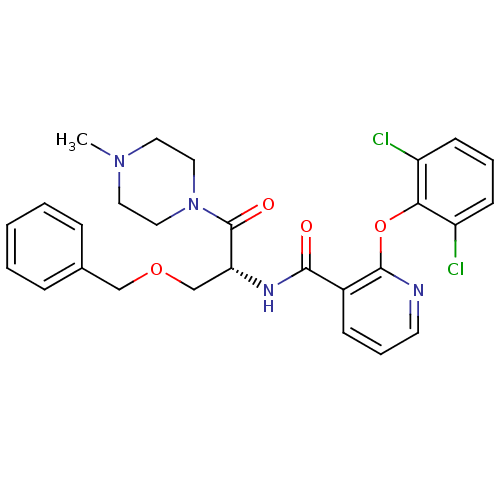
(CHEMBL1911686)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1c(Cl)cccc1Cl |r| Show InChI InChI=1S/C27H28Cl2N4O4/c1-32-13-15-33(16-14-32)27(35)23(18-36-17-19-7-3-2-4-8-19)31-25(34)20-9-6-12-30-26(20)37-24-21(28)10-5-11-22(24)29/h2-12,23H,13-18H2,1H3,(H,31,34)/t23-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356084
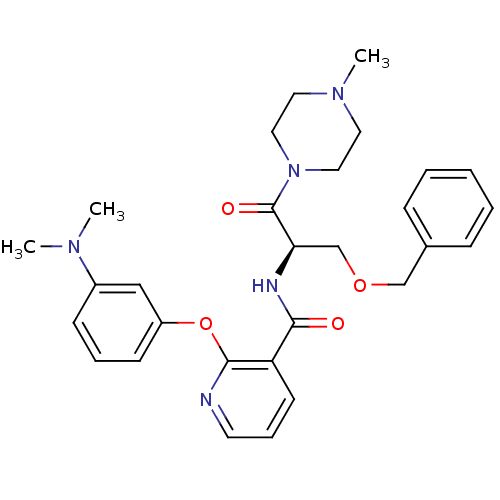
(CHEMBL1911809)Show SMILES CN(C)c1cccc(Oc2ncccc2C(=O)N[C@H](COCc2ccccc2)C(=O)N2CCN(C)CC2)c1 |r| Show InChI InChI=1S/C29H35N5O4/c1-32(2)23-11-7-12-24(19-23)38-28-25(13-8-14-30-28)27(35)31-26(21-37-20-22-9-5-4-6-10-22)29(36)34-17-15-33(3)16-18-34/h4-14,19,26H,15-18,20-21H2,1-3H3,(H,31,35)/t26-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.08E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356083

(CHEMBL1911810)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(C)c(C)c1 |r| Show InChI InChI=1S/C29H34N4O4/c1-21-11-12-24(18-22(21)2)37-28-25(10-7-13-30-28)27(34)31-26(20-36-19-23-8-5-4-6-9-23)29(35)33-16-14-32(3)15-17-33/h4-13,18,26H,14-17,19-20H2,1-3H3,(H,31,34)/t26-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.08E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356088

(CHEMBL1911683)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccccc1 |r| Show InChI InChI=1S/C27H30N4O4/c1-30-15-17-31(18-16-30)27(33)24(20-34-19-21-9-4-2-5-10-21)29-25(32)23-13-8-14-28-26(23)35-22-11-6-3-7-12-22/h2-14,24H,15-20H2,1H3,(H,29,32)/t24-/m1/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.15E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50356072

(CHEMBL1911821)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccncc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C27H27ClF3N5O4/c1-35-11-13-36(14-12-35)26(38)22(17-39-16-18-6-9-32-10-7-18)34-24(37)20-3-2-8-33-25(20)40-23-5-4-19(15-21(23)28)27(29,30)31/h2-10,15,22H,11-14,16-17H2,1H3,(H,34,37)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as inhibition of oxidatative metabolism of testosterone after 45 mins by MS analysis in the p... |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Ceramide glucosyltransferase
(Homo sapiens (Human)) | BDBM50356093

(CHEMBL1911681)Show SMILES CN1CCN(CC1)C(=O)[C@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(Cl)cc1 |r| Show InChI InChI=1S/C27H29ClN4O4/c1-31-14-16-32(17-15-31)27(34)24(19-35-18-20-6-3-2-4-7-20)30-25(33)23-8-5-13-29-26(23)36-22-11-9-21(28)10-12-22/h2-13,24H,14-19H2,1H3,(H,30,33)/t24-/m0/s1 | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of GCS assessed as amount of UDP-glucose consumed during enzyme-catalyzed reaction |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50356075

(CHEMBL1911818)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccc2OCCOc2c1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C30H30ClF3N4O6/c1-37-9-11-38(12-10-37)29(40)23(18-41-17-19-4-6-25-26(15-19)43-14-13-42-25)36-27(39)21-3-2-8-35-28(21)44-24-7-5-20(16-22(24)31)30(32,33)34/h2-8,15-16,23H,9-14,17-18H2,1H3,(H,36,39)/t23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as inhibition of oxidatative metabolism of testosterone after 45 mins by MS analysis in the p... |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50356068

(CHEMBL1911825)Show SMILES CN(C)C1CN(C1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C28H28ClF3N4O4/c1-35(2)20-14-36(15-20)27(38)23(17-39-16-18-7-4-3-5-8-18)34-25(37)21-9-6-12-33-26(21)40-24-11-10-19(13-22(24)29)28(30,31)32/h3-13,20,23H,14-17H2,1-2H3,(H,34,37)/t23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as inhibition of oxidatative metabolism of testosterone after 45 mins by MS analysis in the p... |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50356074

(CHEMBL1911819)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccc(F)cc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C28H27ClF4N4O4/c1-36-11-13-37(14-12-36)27(39)23(17-40-16-18-4-7-20(30)8-5-18)35-25(38)21-3-2-10-34-26(21)41-24-9-6-19(15-22(24)29)28(31,32)33/h2-10,15,23H,11-14,16-17H2,1H3,(H,35,38)/t23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as inhibition of oxidatative metabolism of testosterone after 45 mins by MS analysis in the p... |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50356067

(CHEMBL1911826)Show SMILES CN(C)[C@@H]1CCN(C1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C29H30ClF3N4O4/c1-36(2)21-12-14-37(16-21)28(39)24(18-40-17-19-7-4-3-5-8-19)35-26(38)22-9-6-13-34-27(22)41-25-11-10-20(15-23(25)30)29(31,32)33/h3-11,13,15,21,24H,12,14,16-18H2,1-2H3,(H,35,38)/t21-,24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as inhibition of oxidatative metabolism of testosterone after 45 mins by MS analysis in the p... |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50356065

(CHEMBL1911828)Show SMILES CN[C@H]1CCN(C1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C28H28ClF3N4O4/c1-33-20-11-13-36(15-20)27(38)23(17-39-16-18-6-3-2-4-7-18)35-25(37)21-8-5-12-34-26(21)40-24-10-9-19(14-22(24)29)28(30,31)32/h2-10,12,14,20,23,33H,11,13,15-17H2,1H3,(H,35,37)/t20-,23+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as inhibition of oxidatative metabolism of testosterone after 45 mins by MS analysis in the p... |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50356071

(CHEMBL1911822)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCc1ccnc(Cl)c1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C27H26Cl2F3N5O4/c1-36-9-11-37(12-10-36)26(39)21(16-40-15-17-6-8-33-23(29)13-17)35-24(38)19-3-2-7-34-25(19)41-22-5-4-18(14-20(22)28)27(30,31)32/h2-8,13-14,21H,9-12,15-16H2,1H3,(H,35,38)/t21-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as inhibition of oxidatative metabolism of testosterone after 45 mins by MS analysis in the p... |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50356066

(CHEMBL1911827)Show SMILES CN[C@@H]1CCN(C1)C(=O)[C@@H](COCc1ccccc1)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C28H28ClF3N4O4/c1-33-20-11-13-36(15-20)27(38)23(17-39-16-18-6-3-2-4-7-18)35-25(37)21-8-5-12-34-26(21)40-24-10-9-19(14-22(24)29)28(30,31)32/h2-10,12,14,20,23,33H,11,13,15-17H2,1H3,(H,35,37)/t20-,23-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as inhibition of oxidatative metabolism of testosterone after 45 mins by MS analysis in the p... |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50356070

(CHEMBL1911823)Show SMILES CN1CCN(CC1)C(=O)[C@@H](COCC(C)(C)C)NC(=O)c1cccnc1Oc1ccc(cc1Cl)C(F)(F)F |r| Show InChI InChI=1S/C26H32ClF3N4O4/c1-25(2,3)16-37-15-20(24(36)34-12-10-33(4)11-13-34)32-22(35)18-6-5-9-31-23(18)38-21-8-7-17(14-19(21)27)26(28,29)30/h5-9,14,20H,10-13,15-16H2,1-4H3,(H,32,35)/t20-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes assessed as inhibition of oxidatative metabolism of testosterone after 45 mins by MS analysis in the p... |
Bioorg Med Chem Lett 21: 6773-7 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.037
BindingDB Entry DOI: 10.7270/Q2833SG2 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data