 Found 23 hits of Enzyme Inhibition Constant Data
Found 23 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50326045
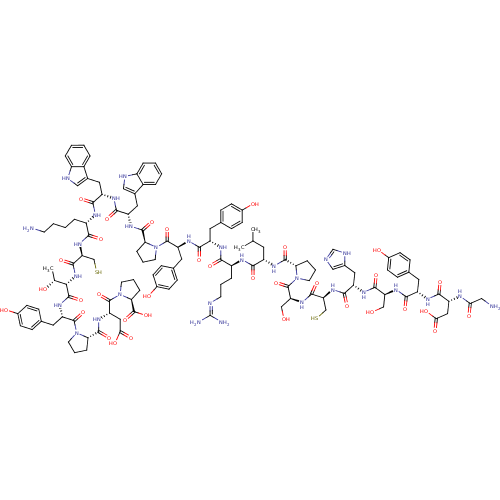
(CHEMBL1240682)Show SMILES CC(C)C[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@H](CO)NC(=O)[C@H](CS)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@@H](CC(O)=O)NC(=O)CN)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CS)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(O)=O)C(=O)N1CCC[C@H]1C(O)=O |r,wU:106.112,55.62,21.24,147.156,91.94,179.190,4.4,124.131,wD:153.162,175.187,190.203,8.7,43.54,37.41,27.35,15.18,138.147,154.165,160.169,110.115,68.70,79.81,(26.05,-7.23,;26.13,-5.69,;24.83,-4.85,;27.49,-4.99,;27.57,-3.46,;28.94,-2.76,;30.27,-3.54,;30.25,-5.08,;31.52,-2.65,;31.53,-1.11,;33,-.64,;33.89,-1.89,;32.98,-3.13,;33.67,-4.51,;32.88,-5.83,;35.21,-4.52,;35.99,-3.2,;37.53,-3.21,;35.96,-5.86,;37.5,-5.87,;38.28,-4.55,;38.26,-7.21,;37.48,-8.54,;38.23,-9.88,;39.8,-7.22,;40.58,-5.9,;39.82,-4.56,;42.12,-5.92,;42.78,-4.52,;41.91,-3.26,;40.37,-3.21,;39.94,-1.74,;41.21,-.87,;42.43,-1.81,;42.88,-7.25,;44.42,-7.27,;45.19,-5.94,;45.17,-8.61,;46.71,-8.62,;47.5,-7.31,;44.39,-9.93,;45.15,-11.27,;44.37,-12.6,;46.69,-11.28,;47.47,-9.96,;49.01,-9.98,;49.79,-8.65,;51.33,-8.66,;52.09,-10,;53.64,-10.02,;51.3,-11.33,;49.77,-11.31,;47.44,-12.63,;46.66,-13.96,;45.12,-13.94,;47.42,-15.29,;48.96,-15.31,;49.74,-13.98,;51.29,-14,;48.98,-12.64,;46.64,-16.62,;45.1,-16.6,;44.32,-17.93,;44.35,-15.27,;42.81,-15.25,;26.27,-2.62,;26.36,-1.08,;24.9,-3.31,;23.6,-2.48,;23.57,-.95,;22.22,-.2,;22.2,1.35,;20.86,2.11,;20.82,3.65,;22.14,4.44,;19.48,4.41,;22.23,-3.18,;22.15,-4.72,;20.94,-2.34,;19.56,-3.04,;18.27,-2.2,;16.9,-2.9,;15.61,-2.07,;14.23,-2.77,;14.16,-4.3,;12.81,-4.98,;15.45,-5.14,;16.82,-4.45,;19.49,-4.58,;20.78,-5.41,;18.11,-5.27,;18.02,-6.8,;19.74,-7.82,;19.73,-9.36,;21.05,-10.15,;21.03,-11.69,;19.69,-12.44,;19.69,-13.96,;18.36,-11.66,;18.39,-10.12,;16.24,-7.72,;14.96,-6.88,;16.08,-9.25,;17.22,-10.47,;16.41,-11.95,;14.76,-11.64,;14.83,-10.1,;13.48,-9.35,;13.46,-7.8,;12.16,-10.14,;10.81,-9.4,;10.78,-7.87,;9.43,-7.12,;8.19,-8.02,;6.94,-7.12,;7.43,-5.66,;6.65,-4.32,;7.43,-2.99,;8.96,-2.98,;9.73,-4.32,;8.97,-5.66,;9.49,-10.2,;9.52,-11.73,;8.14,-9.44,;6.82,-10.23,;6.85,-11.77,;5.53,-12.56,;4.26,-11.68,;3.04,-12.62,;3.55,-14.07,;2.81,-15.42,;3.61,-16.73,;5.14,-16.7,;5.88,-15.35,;5.08,-14.04,;5.48,-9.48,;5.45,-7.94,;4.15,-10.26,;2.78,-9.54,;2.77,-7.99,;1.41,-7.26,;1.38,-5.71,;.03,-4.97,;.03,-3.4,;1.47,-10.32,;1.49,-11.86,;.13,-9.56,;-1.18,-10.37,;-1.15,-11.9,;-2.49,-12.7,;-2.54,-9.62,;-2.56,-8.08,;-3.85,-10.41,;-5.19,-9.66,;-5.21,-8.12,;-3.91,-7.33,;-6.56,-7.36,;-6.52,-10.46,;-6.49,-12,;-7.86,-9.7,;-9.19,-10.48,;-10.54,-9.74,;-10.56,-8.2,;-9.24,-7.41,;-9.27,-5.88,;-10.64,-5.13,;-10.63,-3.57,;-11.94,-5.92,;-11.91,-7.45,;-9.16,-12.03,;-7.82,-12.77,;-10.34,-13.01,;-11.99,-12.72,;-12.8,-14.2,;-11.64,-15.41,;-10.36,-14.51,;-9.08,-15.37,;-7.74,-14.61,;-9.07,-16.91,;-7.76,-17.71,;-6.42,-16.97,;-5.09,-17.75,;-3.72,-16.98,;-5.1,-19.29,;-7.78,-19.26,;-6.45,-20.05,;-8.97,-20.22,;-10.61,-19.91,;-11.45,-21.38,;-10.29,-22.6,;-9,-21.72,;-7.67,-22.47,;-6.33,-21.64,;-7.62,-24,)| Show InChI InChI=1S/C128H168N30O34S2/c1-66(2)47-85(145-119(183)100-22-13-45-157(100)126(190)96(62-160)151-117(181)97(63-193)153-114(178)90(54-74-60-133-65-137-74)144-116(180)95(61-159)150-111(175)87(49-69-27-35-76(163)36-28-69)142-115(179)91(55-104(167)168)138-103(166)57-130)109(173)140-84(20-10-42-134-128(131)132)107(171)141-86(48-68-25-33-75(162)34-26-68)110(174)147-92(50-70-29-37-77(164)38-30-70)123(187)155-43-11-21-99(155)120(184)146-89(53-73-59-136-82-18-7-5-16-80(73)82)113(177)143-88(52-72-58-135-81-17-6-4-15-79(72)81)112(176)139-83(19-8-9-41-129)108(172)152-98(64-194)118(182)154-106(67(3)161)122(186)149-93(51-71-31-39-78(165)40-32-71)124(188)156-44-12-23-101(156)121(185)148-94(56-105(169)170)125(189)158-46-14-24-102(158)127(191)192/h4-7,15-18,25-40,58-60,65-67,83-102,106,135-136,159-165,193-194H,8-14,19-24,41-57,61-64,129-130H2,1-3H3,(H,133,137)(H,138,166)(H,139,176)(H,140,173)(H,141,171)(H,142,179)(H,143,177)(H,144,180)(H,145,183)(H,146,184)(H,147,174)(H,148,185)(H,149,186)(H,150,175)(H,151,181)(H,152,172)(H,153,178)(H,154,182)(H,167,168)(H,169,170)(H,191,192)(H4,131,132,134)/t67-,83+,84+,85+,86+,87+,88+,89+,90+,91-,92+,93+,94+,95+,96+,97+,98+,99+,100+,101+,102+,106+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Binding affinity to angiotensin converting enzyme 2 |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Deoxyribonuclease-2-alpha
(Homo sapiens (Human)) | BDBM50326048
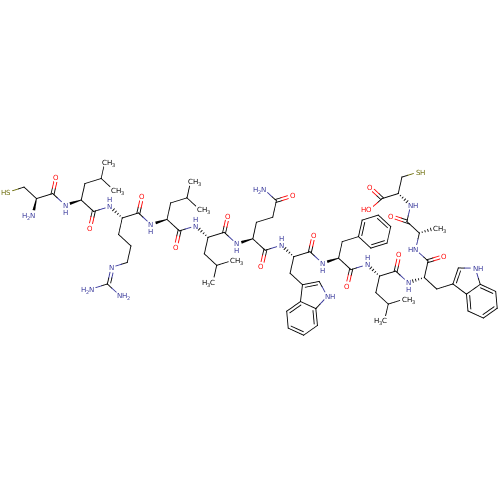
((2R,5S,8S,11S,14S,17S,20S,23S,26S,29S,32S,35R)-8,1...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H](N)CS)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](C)C(=O)N[C@@H](CS)C(O)=O |r,wU:98.103,51.50,8.8,15.14,76.78,34.33,wD:4.4,65.66,26.25,84.86,103.107,42.41,(.31,-40.62,;.28,-39.08,;1.6,-38.28,;-1.07,-38.34,;-1.09,-36.79,;-2.44,-36.06,;-3.77,-36.84,;-3.74,-38.38,;-5.12,-36.1,;-6.42,-36.89,;-5.16,-34.55,;-3.84,-33.76,;.22,-36,;.19,-34.46,;1.57,-36.74,;2.88,-35.94,;2.85,-34.4,;4.18,-33.6,;4.14,-32.07,;5.46,-31.27,;5.43,-29.73,;4.07,-28.99,;6.76,-28.94,;4.24,-36.68,;4.27,-38.22,;5.56,-35.89,;6.91,-36.63,;6.94,-38.17,;8.29,-38.91,;8.32,-40.45,;9.61,-38.12,;8.23,-35.84,;8.2,-34.29,;9.58,-36.58,;10.9,-35.78,;10.86,-34.24,;12.18,-33.45,;12.15,-31.91,;13.53,-34.19,;12.24,-36.52,;12.27,-38.06,;13.56,-35.73,;14.91,-36.47,;14.94,-38.01,;16.28,-38.75,;16.32,-40.29,;15,-41.08,;17.66,-41.04,;16.22,-35.67,;16.19,-34.14,;17.57,-36.42,;18.89,-35.62,;18.86,-34.08,;20.18,-33.29,;21.45,-34.16,;22.67,-33.23,;22.16,-31.78,;22.89,-30.42,;22.09,-29.1,;20.55,-29.15,;19.82,-30.5,;20.62,-31.81,;20.24,-36.36,;20.28,-37.9,;21.56,-35.56,;22.91,-36.3,;22.94,-37.84,;24.29,-38.58,;25.61,-37.79,;26.96,-38.53,;26.99,-40.07,;25.67,-40.87,;24.33,-40.12,;24.23,-35.51,;24.19,-33.97,;25.58,-36.24,;26.9,-35.46,;26.87,-33.91,;28.19,-33.12,;28.16,-31.58,;29.53,-33.86,;28.25,-36.2,;28.28,-37.73,;29.56,-35.39,;30.91,-36.14,;30.94,-37.68,;32.29,-38.42,;33.53,-37.52,;34.78,-38.42,;34.3,-39.89,;35.06,-41.23,;34.29,-42.56,;32.75,-42.55,;31.98,-41.21,;32.76,-39.89,;32.23,-35.34,;32.2,-33.81,;33.58,-36.08,;34.9,-35.29,;34.87,-33.75,;36.25,-36.03,;36.29,-37.57,;37.57,-35.23,;38.92,-35.97,;38.95,-37.51,;40.3,-38.25,;40.24,-35.18,;41.59,-35.92,;40.21,-33.64,)| Show InChI InChI=1S/C75H110N18O14S2/c1-39(2)28-54(86-64(96)49(76)37-108)68(100)84-52(24-17-27-80-75(78)79)65(97)87-56(30-41(5)6)70(102)88-55(29-40(3)4)69(101)85-53(25-26-62(77)94)66(98)91-60(34-46-36-82-51-23-16-14-21-48(46)51)73(105)90-58(32-44-18-11-10-12-19-44)72(104)89-57(31-42(7)8)71(103)92-59(33-45-35-81-50-22-15-13-20-47(45)50)67(99)83-43(9)63(95)93-61(38-109)74(106)107/h10-16,18-23,35-36,39-43,49,52-61,81-82,108-109H,17,24-34,37-38,76H2,1-9H3,(H2,77,94)(H,83,99)(H,84,100)(H,85,101)(H,86,96)(H,87,97)(H,88,102)(H,89,104)(H,90,105)(H,91,98)(H,92,103)(H,93,95)(H,106,107)(H4,78,79,80)/t43-,49-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Binding affinity to Dnase 2 |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50326049
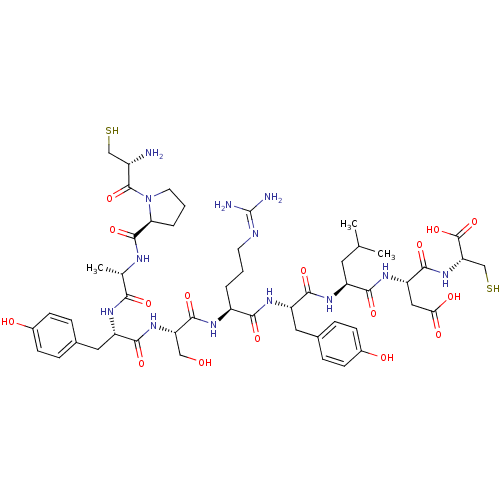
((3S,6S,9S,12S,15S,18S,21S,24R)-1-((S)-1-((R)-2-ami...)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#8])-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@@H](-[#7])-[#6]-[#16])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#8])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#16])-[#6](-[#8])=O |r| Show InChI InChI=1S/C51H75N13O16S2/c1-25(2)18-33(43(72)61-36(21-40(68)69)46(75)63-38(24-82)50(79)80)59-44(73)35(20-28-10-14-30(67)15-11-28)60-42(71)32(6-4-16-55-51(53)54)57-47(76)37(22-65)62-45(74)34(19-27-8-12-29(66)13-9-27)58-41(70)26(3)56-48(77)39-7-5-17-64(39)49(78)31(52)23-81/h8-15,25-26,31-39,65-67,81-82H,4-7,16-24,52H2,1-3H3,(H,56,77)(H,57,76)(H,58,70)(H,59,73)(H,60,71)(H,61,72)(H,62,74)(H,63,75)(H,68,69)(H,79,80)(H4,53,54,55)/t26-,31-,32-,33-,34-,35-,36-,37-,38-,39-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Binding affinity to urokinase-type plasminogen activator |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50326043
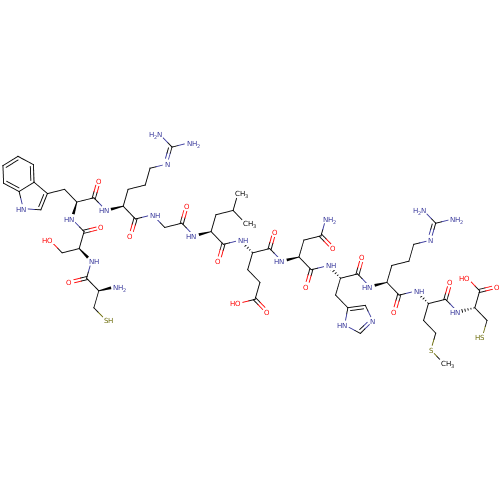
((2R,5S,8S,11S,14S,17S)-11-((1H-imidazol-5-yl)methy...)Show SMILES CSCC[C@H](NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CS)C(=O)N[C@@H](CS)C(O)=O |r,wU:37.43,19.26,69.82,4.4,89.92,wD:29.34,58.66,83.88,8.15,46.51,96.98,(29.98,-31.85,;28.63,-32.59,;28.61,-34.13,;27.26,-34.88,;27.23,-36.42,;25.89,-37.17,;24.57,-36.37,;24.59,-34.83,;23.22,-37.12,;23.19,-38.66,;24.51,-39.45,;24.49,-41,;25.81,-41.79,;25.78,-43.33,;24.43,-44.08,;27.11,-44.13,;21.9,-36.34,;20.55,-37.09,;20.53,-38.62,;19.23,-36.3,;19.25,-34.75,;20.48,-33.82,;21.96,-34.26,;22.83,-33,;21.91,-31.77,;20.45,-32.28,;17.88,-37.04,;16.56,-36.25,;16.59,-34.7,;15.21,-36.99,;15.18,-38.53,;16.51,-39.32,;17.86,-38.58,;16.48,-40.87,;13.89,-36.19,;12.54,-36.94,;12.52,-38.48,;11.23,-36.15,;11.25,-34.61,;12.6,-33.86,;12.62,-32.31,;11.3,-31.52,;13.98,-31.57,;9.88,-36.89,;8.56,-36.1,;8.58,-34.56,;7.2,-36.84,;7.18,-38.39,;8.5,-39.18,;8.48,-40.72,;9.85,-38.43,;5.88,-36.06,;4.53,-36.8,;4.5,-38.35,;3.21,-36.01,;1.87,-36.76,;.55,-35.96,;.58,-34.42,;-.8,-36.71,;-.83,-38.25,;.5,-39.05,;.47,-40.58,;1.78,-41.38,;1.77,-42.92,;.42,-43.67,;3.09,-43.72,;-2.12,-35.92,;-3.47,-36.66,;-3.49,-38.21,;-4.79,-35.87,;-4.77,-34.33,;-3.51,-33.44,;-2.25,-34.31,;-1.02,-33.38,;-1.52,-31.93,;-.78,-30.58,;-1.57,-29.26,;-3.12,-29.29,;-3.86,-30.64,;-3.07,-31.95,;-6.14,-36.62,;-7.45,-35.82,;-7.43,-34.27,;-8.81,-36.57,;-8.83,-38.1,;-7.52,-38.91,;-10.13,-35.78,;-11.48,-36.52,;-11.5,-38.05,;-12.8,-35.73,;-14.14,-36.47,;-12.77,-34.18,;-11.42,-33.43,;28.55,-37.22,;28.53,-38.76,;29.9,-36.47,;31.22,-37.26,;31.2,-38.81,;32.52,-39.59,;32.57,-36.52,;33.89,-37.31,;32.59,-34.97,)| Show InChI InChI=1S/C60H94N22O17S3/c1-29(2)18-39(73-46(85)24-71-49(89)35(10-6-15-68-59(63)64)74-54(94)40(19-30-22-70-34-9-5-4-8-32(30)34)78-57(97)43(25-83)81-48(88)33(61)26-100)53(93)76-37(12-13-47(86)87)51(91)80-42(21-45(62)84)56(96)79-41(20-31-23-67-28-72-31)55(95)75-36(11-7-16-69-60(65)66)50(90)77-38(14-17-102-3)52(92)82-44(27-101)58(98)99/h4-5,8-9,22-23,28-29,33,35-44,70,83,100-101H,6-7,10-21,24-27,61H2,1-3H3,(H2,62,84)(H,67,72)(H,71,89)(H,73,85)(H,74,94)(H,75,95)(H,76,93)(H,77,90)(H,78,97)(H,79,96)(H,80,91)(H,81,88)(H,82,92)(H,86,87)(H,98,99)(H4,63,64,68)(H4,65,66,69)/t33-,35-,36-,37-,38-,39-,40-,41-,42-,43-,44-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Binding affinity to urokinase-type plasminogen activator |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Receptor tyrosine-protein kinase erbB-2
(Homo sapiens (Human)) | BDBM50326046
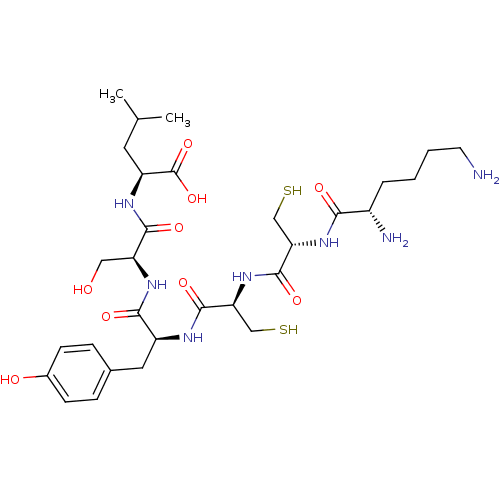
((2S,5S,8S,11R,14R,17S)-17,21-diamino-8-(4-hydroxyb...)Show SMILES CC(C)C[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CS)NC(=O)[C@H](CS)NC(=O)[C@@H](N)CCCCN)C(O)=O |r| Show InChI InChI=1S/C30H49N7O9S2/c1-16(2)11-21(30(45)46)34-27(42)22(13-38)35-26(41)20(12-17-6-8-18(39)9-7-17)33-28(43)24(15-48)37-29(44)23(14-47)36-25(40)19(32)5-3-4-10-31/h6-9,16,19-24,38-39,47-48H,3-5,10-15,31-32H2,1-2H3,(H,33,43)(H,34,42)(H,35,41)(H,36,40)(H,37,44)(H,45,46)/t19-,20-,21-,22-,23-,24-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Binding affinity to ErbB-2 |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50326044
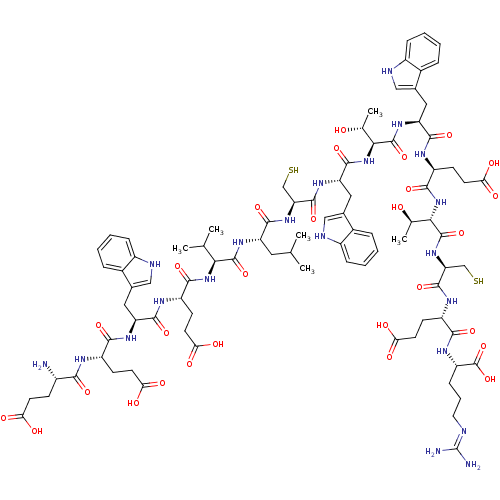
((2S,5S,8R,11S,14S,17S,20S,23S,26R,29S,32S,35S,38S,...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](N)CCC(O)=O)C(C)C)C(=O)N[C@@H](CS)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O |r,wU:44.46,78.83,8.8,98.103,21.33,57.58,114.119,77.80,129.134,wD:84.87,4.4,107.112,35.42,108.115,120.125,63.64,12.17,(10.8,-38.91,;10.78,-37.37,;12.1,-36.58,;9.43,-36.62,;9.4,-35.08,;8.05,-34.35,;6.73,-35.14,;6.75,-36.68,;5.38,-34.39,;4.06,-35.18,;2.73,-34.41,;2.7,-32.87,;1.4,-35.2,;1.43,-36.74,;2.78,-37.49,;2.8,-39.03,;1.45,-39.83,;4.15,-39.78,;.06,-34.45,;-1.27,-35.24,;-1.24,-36.78,;-2.61,-34.49,;-2.64,-32.95,;-1.41,-32.02,;-.16,-32.92,;1.07,-32,;.59,-30.54,;1.35,-29.19,;.57,-27.87,;-.98,-27.88,;-1.73,-29.22,;-.95,-30.55,;-3.93,-35.29,;-5.27,-34.53,;-5.3,-32.99,;-6.6,-35.32,;-6.57,-36.86,;-5.22,-37.61,;-5.2,-39.14,;-6.53,-39.94,;-3.86,-39.9,;-7.95,-34.58,;-9.27,-35.37,;-9.25,-36.91,;-10.62,-34.62,;-11.94,-35.41,;-10.64,-33.08,;-9.32,-32.29,;-9.34,-30.75,;-10.69,-30,;-8.02,-29.96,;5.36,-32.85,;6.68,-32.05,;4.02,-32.1,;10.73,-34.29,;10.7,-32.75,;12.07,-35.05,;13.39,-34.26,;13.37,-32.72,;14.69,-31.93,;14.74,-35.01,;14.76,-36.55,;16.05,-34.22,;17.4,-34.95,;17.43,-36.5,;18.67,-37.41,;19.89,-36.48,;21.16,-37.36,;20.71,-38.84,;21.51,-40.15,;20.77,-41.5,;19.23,-41.53,;18.43,-40.22,;19.18,-38.86,;18.73,-34.17,;18.71,-32.63,;20.08,-34.92,;21.4,-34.12,;21.38,-32.58,;20.03,-31.83,;22.66,-31.81,;22.74,-34.87,;22.77,-36.41,;24.06,-34.07,;25.41,-34.81,;25.43,-36.35,;26.69,-37.24,;27.94,-36.36,;29.17,-37.28,;28.67,-38.74,;29.42,-40.09,;28.63,-41.41,;27.09,-41.39,;26.34,-40.03,;27.14,-38.72,;26.73,-34.01,;26.71,-32.47,;28.06,-34.79,;29.38,-33.99,;29.36,-32.45,;30.68,-31.66,;30.66,-30.12,;29.29,-29.38,;31.98,-29.33,;30.73,-34.74,;30.76,-36.28,;32.05,-33.95,;33.41,-34.68,;33.43,-36.22,;32.11,-37.02,;34.75,-36.98,;34.73,-33.89,;34.71,-32.35,;36.06,-34.66,;37.38,-33.86,;37.36,-32.33,;38.68,-31.53,;38.73,-34.61,;38.76,-36.15,;40.05,-33.82,;41.4,-34.56,;41.43,-36.1,;42.78,-36.86,;42.8,-38.4,;41.46,-39.18,;44.14,-39.14,;42.73,-33.78,;42.7,-32.24,;44.07,-34.52,;45.39,-33.73,;45.37,-32.19,;46.7,-31.4,;46.67,-29.86,;47.99,-29.07,;47.97,-27.53,;46.61,-26.78,;49.29,-26.74,;46.74,-34.48,;48.06,-33.69,;46.77,-36.02,)| Show InChI InChI=1S/C89H123N21O28S2/c1-41(2)32-60(104-85(134)71(42(3)4)108-77(126)57(24-29-69(119)120)98-80(129)61(33-45-36-94-52-17-10-7-14-48(45)52)102-76(125)55(22-27-67(115)116)97-74(123)51(90)21-26-66(113)114)79(128)106-64(39-139)84(133)103-63(35-47-38-96-54-19-12-9-16-50(47)54)82(131)110-72(43(5)111)86(135)105-62(34-46-37-95-53-18-11-8-15-49(46)53)81(130)99-58(25-30-70(121)122)78(127)109-73(44(6)112)87(136)107-65(40-140)83(132)100-56(23-28-68(117)118)75(124)101-59(88(137)138)20-13-31-93-89(91)92/h7-12,14-19,36-38,41-44,51,55-65,71-73,94-96,111-112,139-140H,13,20-35,39-40,90H2,1-6H3,(H,97,123)(H,98,129)(H,99,130)(H,100,132)(H,101,124)(H,102,125)(H,103,133)(H,104,134)(H,105,135)(H,106,128)(H,107,136)(H,108,126)(H,109,127)(H,110,131)(H,113,114)(H,115,116)(H,117,118)(H,119,120)(H,121,122)(H,137,138)(H4,91,92,93)/t43-,44-,51+,55+,56+,57+,58+,59+,60+,61+,62+,63+,64+,65+,71+,72+,73+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Binding affinity to tissue factor/factor 7a |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM50326040
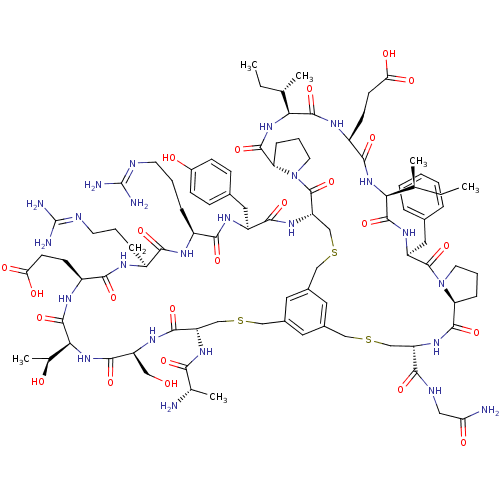
(CHEMBL1240598)Show SMILES [#6]-[#6]-[#6@H](-[#6])-[#6@@H]-1-[#7]-[#6](=O)-[#6@@H]-2-[#6]-[#6]-[#6]-[#7]-2-[#6](=O)-[#6@@H]-2-[#6]-[#16]-[#6]-c3cc(-[#6]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#8])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c4ccc(-[#8])cc4)-[#6](=O)-[#7]-2)cc(-[#6]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H]-2-[#6]-[#6]-[#6]-[#7]-2-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6]-1=O)-[#6@@H](-[#6])-[#6]-[#6])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O)c3 |r| Show InChI InChI=1S/C92H136N24O24S3/c1-7-47(3)72-87(137)107-62(38-51-16-10-9-11-17-51)89(139)115-32-14-20-67(115)84(134)110-64(76(126)101-39-69(94)120)44-141-41-53-34-54-36-55(35-53)43-143-46-66(90(140)116-33-15-21-68(116)85(135)113-73(48(4)8-2)86(136)104-60(80(130)112-72)27-29-71(123)124)111-81(131)61(37-52-22-24-56(119)25-23-52)106-78(128)58(19-13-31-100-92(97)98)102-77(127)57(18-12-30-99-91(95)96)103-79(129)59(26-28-70(121)122)105-88(138)74(50(6)118)114-82(132)63(40-117)108-83(133)65(45-142-42-54)109-75(125)49(5)93/h9-11,16-17,22-25,34-36,47-50,57-68,72-74,117-119H,7-8,12-15,18-21,26-33,37-46,93H2,1-6H3,(H2,94,120)(H,101,126)(H,102,127)(H,103,129)(H,104,136)(H,105,138)(H,106,128)(H,107,137)(H,108,133)(H,109,125)(H,110,134)(H,111,131)(H,112,130)(H,113,135)(H,114,132)(H,121,122)(H,123,124)(H4,95,96,99)(H4,97,98,100)/t47-,48-,49-,50-,57-,58-,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,72-,73?,74-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human plasma kallikrein using Z-Phe-Arg-AMC substrate after 30 mins by spectrophotometric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Cathepsin G
(Homo sapiens (Human)) | BDBM50326029
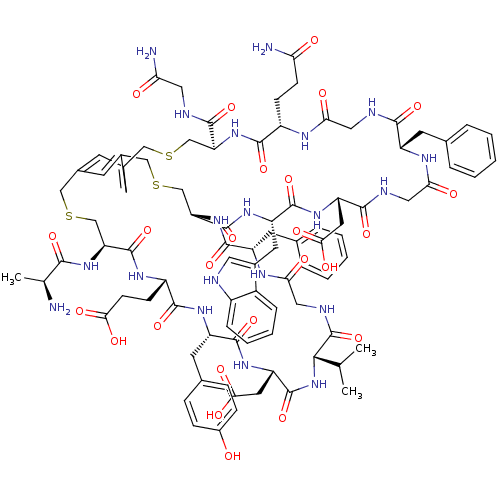
(CHEMBL1240609)Show SMILES CC(C)[C@@H]1NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CSCc2cc3CSC[C@H](NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](Cc4ccccc4)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc4c[nH]c5ccccc45)NC(=O)[C@H](CSCc(c3)c2)NC(=O)[C@H](Cc2ccccc2)NC(=O)CNC1=O)C(=O)NCC(N)=O)NC(=O)[C@H](C)N |r| Show InChI InChI=1S/C90H112N20O25S3/c1-46(2)77-90(135)98-39-73(116)101-61(30-49-14-8-5-9-15-49)84(129)109-68-45-138-42-53-27-51(26-52(28-53)41-137-44-67(107-78(123)47(3)91)88(133)102-59(23-25-74(117)118)83(128)103-62(31-50-18-20-55(111)21-19-50)85(130)106-65(34-76(121)122)87(132)110-77)40-136-43-66(81(126)95-36-70(93)113)108-82(127)58(22-24-69(92)112)99-71(114)37-96-79(124)60(29-48-12-6-4-7-13-48)100-72(115)38-97-80(125)64(33-75(119)120)105-86(131)63(104-89(68)134)32-54-35-94-57-17-11-10-16-56(54)57/h4-21,26-28,35,46-47,58-68,77,94,111H,22-25,29-34,36-45,91H2,1-3H3,(H2,92,112)(H2,93,113)(H,95,126)(H,96,124)(H,97,125)(H,98,135)(H,99,114)(H,100,115)(H,101,116)(H,102,133)(H,103,128)(H,104,134)(H,105,131)(H,106,130)(H,107,123)(H,108,127)(H,109,129)(H,110,132)(H,117,118)(H,119,120)(H,121,122)/t47-,58-,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,77-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin G using N-Suc-Ala-Ala-Phe-Pro-pNA substrate by calorimetric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Cathepsin G
(Homo sapiens (Human)) | BDBM50326028
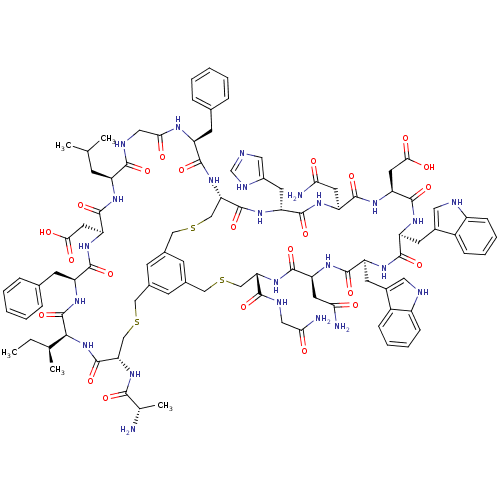
(CHEMBL1240611)Show SMILES CC[C@H](C)[C@@H]1NC(=O)[C@H](CSCc2cc3CSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc4c[nH]c5ccccc45)NC(=O)[C@H](Cc4c[nH]c5ccccc45)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](Cc4cnc[nH]4)NC(=O)[C@H](CSCc(c3)c2)NC(=O)[C@H](Cc2ccccc2)NC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc2ccccc2)NC1=O)C(=O)NCC(N)=O)NC(=O)[C@H](C)N |r| Show InChI InChI=1S/C99H124N24O23S3/c1-6-51(4)84-99(146)119-67(30-54-19-11-8-12-20-54)89(136)117-73(36-82(128)129)95(142)111-65(25-50(2)3)86(133)108-42-81(127)110-66(29-53-17-9-7-10-18-53)88(135)122-76-47-148-44-56-26-55(27-57(28-56)45-149-48-77(98(145)123-84)120-85(132)52(5)100)43-147-46-75(87(134)107-41-80(103)126)121-94(141)72(35-79(102)125)115-91(138)69(32-59-39-106-64-24-16-14-22-62(59)64)112-90(137)68(31-58-38-105-63-23-15-13-21-61(58)63)113-96(143)74(37-83(130)131)118-93(140)71(34-78(101)124)116-92(139)70(114-97(76)144)33-60-40-104-49-109-60/h7-24,26-28,38-40,49-52,65-77,84,105-106H,6,25,29-37,41-48,100H2,1-5H3,(H2,101,124)(H2,102,125)(H2,103,126)(H,104,109)(H,107,134)(H,108,133)(H,110,127)(H,111,142)(H,112,137)(H,113,143)(H,114,144)(H,115,138)(H,116,139)(H,117,136)(H,118,140)(H,119,146)(H,120,132)(H,121,141)(H,122,135)(H,123,145)(H,128,129)(H,130,131)/t51-,52-,65-,66-,67-,68-,69-,70+,71?,72-,73-,74-,75-,76-,77-,84-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin G using N-Suc-Ala-Ala-Phe-Pro-pNA substrate by calorimetric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Cathepsin G
(Homo sapiens (Human)) | BDBM50326027
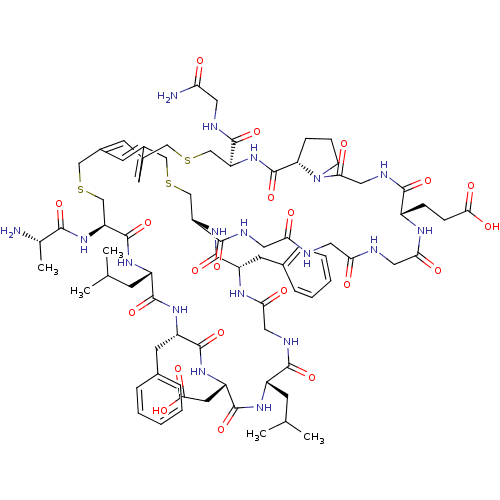
(CHEMBL1240612)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CSCc2cc3CSC[C@H](NC(=O)[C@@H]4CCCN4C(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)CNC(=O)CNC(=O)[C@H](CSCc(c3)c2)NC(=O)[C@H](Cc2ccccc2)NC(=O)CNC1=O)C(=O)NCC(N)=O)NC(=O)[C@H](C)N |r| Show InChI InChI=1S/C77H106N18O21S3/c1-41(2)21-50-69(108)84-33-63(100)87-52(26-44-13-8-6-9-14-44)73(112)93-55-38-117-35-46-23-47(25-48(24-46)37-119-40-57(92-67(106)43(5)78)76(115)89-51(22-42(3)4)72(111)90-53(27-45-15-10-7-11-16-45)74(113)91-54(28-66(104)105)75(114)88-50)36-118-39-56(70(109)82-29-59(79)96)94-77(116)58-17-12-20-95(58)64(101)34-85-68(107)49(18-19-65(102)103)86-62(99)32-81-60(97)30-80-61(98)31-83-71(55)110/h6-11,13-16,23-25,41-43,49-58H,12,17-22,26-40,78H2,1-5H3,(H2,79,96)(H,80,98)(H,81,97)(H,82,109)(H,83,110)(H,84,108)(H,85,107)(H,86,99)(H,87,100)(H,88,114)(H,89,115)(H,90,111)(H,91,113)(H,92,106)(H,93,112)(H,94,116)(H,102,103)(H,104,105)/t43-,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin G using N-Suc-Ala-Ala-Phe-Pro-pNA substrate by calorimetric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Cathepsin G
(Homo sapiens (Human)) | BDBM50326025
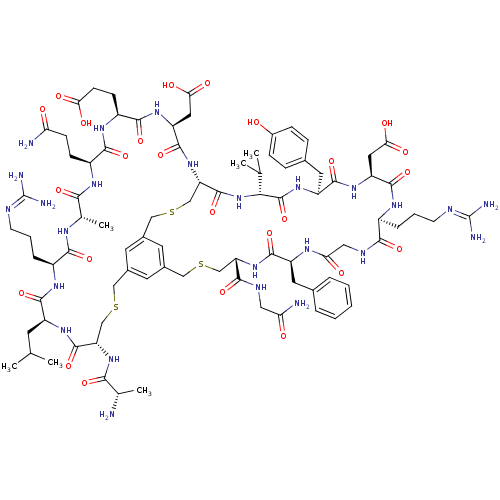
(CHEMBL1240614)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-[#16]-[#6]-c2cc3-[#6]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-c4ccccc4)-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c4ccc(-[#8])cc4)-[#7]-[#6](=O)-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#16]-[#6]-c(c3)c2)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6]-1=O)-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7] |r| Show InChI InChI=1S/C87H127N25O25S3/c1-42(2)26-56-78(130)103-53(15-11-25-96-87(93)94)75(127)99-45(6)72(124)101-54(20-22-64(89)114)76(128)104-55(21-23-67(117)118)77(129)106-60(33-69(121)122)82(134)111-63-41-140-38-50-28-48(27-49(29-50)37-139-40-62(83(135)105-56)109-71(123)44(5)88)36-138-39-61(74(126)97-34-65(90)115)110-79(131)57(30-46-12-8-7-9-13-46)100-66(116)35-98-73(125)52(14-10-24-95-86(91)92)102-81(133)59(32-68(119)120)107-80(132)58(31-47-16-18-51(113)19-17-47)108-85(137)70(43(3)4)112-84(63)136/h7-9,12-13,16-19,27-29,42-45,52-63,70,113H,10-11,14-15,20-26,30-41,88H2,1-6H3,(H2,89,114)(H2,90,115)(H,97,126)(H,98,125)(H,99,127)(H,100,116)(H,101,124)(H,102,133)(H,103,130)(H,104,128)(H,105,135)(H,106,129)(H,107,132)(H,108,137)(H,109,123)(H,110,131)(H,111,134)(H,112,136)(H,117,118)(H,119,120)(H,121,122)(H4,91,92,95)(H4,93,94,96)/t44-,45-,52-,53-,54-,55-,56-,57-,58?,59-,60-,61-,62-,63-,70+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin G using N-Suc-Ala-Ala-Phe-Pro-pNA substrate by calorimetric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Cathepsin G
(Homo sapiens (Human)) | BDBM50326024
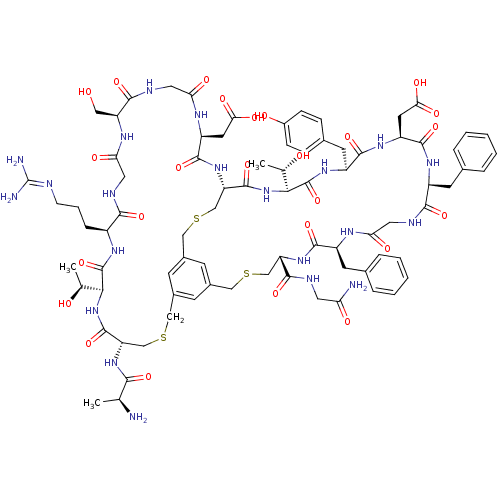
(CHEMBL1240615)Show SMILES [#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H]-1-[#6]-[#16]-[#6]-c2cc3-[#6]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-c4ccccc4)-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@H](-[#6]-c4ccccc4)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c4ccc(-[#8])cc4)-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#16]-[#6]-c(c3)c2)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@H](-[#6]-[#8])-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6]-1=O)-[#6@H](-[#6])-[#8])-[#6@H](-[#6])-[#8])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O |r| Show InChI InChI=1S/C81H109N21O25S3/c1-40(82)68(115)98-58-38-129-35-47-21-46-22-48(23-47)36-130-39-59(100-75(122)54(27-64(111)112)92-62(109)31-90-71(118)56(33-103)93-63(110)32-88-69(116)50(15-10-20-86-81(84)85)94-79(126)66(41(2)104)101-77(58)124)78(125)102-67(42(3)105)80(127)97-53(26-45-16-18-49(106)19-17-45)74(121)96-55(28-65(113)114)76(123)95-51(24-43-11-6-4-7-12-43)70(117)89-30-61(108)91-52(25-44-13-8-5-9-14-44)73(120)99-57(37-128-34-46)72(119)87-29-60(83)107/h4-9,11-14,16-19,21-23,40-42,50-59,66-67,103-106H,10,15,20,24-39,82H2,1-3H3,(H2,83,107)(H,87,119)(H,88,116)(H,89,117)(H,90,118)(H,91,108)(H,92,109)(H,93,110)(H,94,126)(H,95,123)(H,96,121)(H,97,127)(H,98,115)(H,99,120)(H,100,122)(H,101,124)(H,102,125)(H,111,112)(H,113,114)(H4,84,85,86)/t40-,41-,42-,50-,51-,52-,53?,54-,55-,56-,57-,58-,59-,66-,67-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin G using N-Suc-Ala-Ala-Phe-Pro-pNA substrate by calorimetric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Cathepsin G
(Homo sapiens (Human)) | BDBM50326026

(CHEMBL1240613)Show SMILES [#6]-[#6]-[#6@@H](-[#6])-[#6@@H]-1-[#7]-[#6](=O)-[#6@@H]-2-[#6]-[#6]-[#6]-[#7]-2-[#6](=O)-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@@H]-2-[#6]-[#16]-[#6]-c3cc(-[#6]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6])-[#7])-[#6](=O)-[#7]-4-[#6]-[#6]-[#6]-[#6@H]-4-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6@H](-[#6])-[#6]-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6](-[#8])=O)-[#6](=O)-[#7]-[#6]-[#6](=O)-[#7]-[#6@@H](-[#6]-c4ccccc4)-[#6](=O)-[#7]-2)cc(-[#6]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O)c3 |r| Show InChI InChI=1S/C89H129N21O22S3/c1-8-48(5)72-85(129)99-57(26-27-70(114)115)75(119)96-39-69(113)97-58(35-51-19-12-10-13-20-51)78(122)105-64-45-134-42-54-32-53(33-55(34-54)43-135-46-65(106-74(118)50(7)90)88(132)110-30-17-24-66(110)83(127)98-56(77(121)107-72)23-16-28-94-89(92)93)41-133-44-63(76(120)95-38-68(91)112)104-80(124)60(37-71(116)117)100-81(125)62(40-111)103-79(123)59(36-52-21-14-11-15-22-52)101-86(130)73(49(6)9-2)108-84(128)67-25-18-29-109(67)87(131)61(31-47(3)4)102-82(64)126/h10-15,19-22,32-34,47-50,56-67,72-73,111H,8-9,16-18,23-31,35-46,90H2,1-7H3,(H2,91,112)(H,95,120)(H,96,119)(H,97,113)(H,98,127)(H,99,129)(H,100,125)(H,101,130)(H,102,126)(H,103,123)(H,104,124)(H,105,122)(H,106,118)(H,107,121)(H,108,128)(H,114,115)(H,116,117)(H4,92,93,94)/t48-,49-,50+,56+,57+,58+,59+,60-,61-,62-,63+,64+,65+,66+,67+,72+,73+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin G using N-Suc-Ala-Ala-Phe-Pro-pNA substrate by calorimetric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM50326042
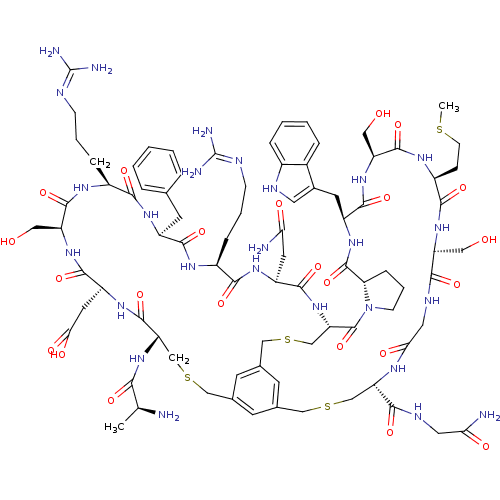
(CHEMBL1240595)Show SMILES CSCC[C@@H]1NC(=O)[C@H](CO)NC(=O)[C@H](Cc2c[nH]c3ccccc23)NC(=O)[C@@H]2CCCN2C(=O)[C@@H]2CSCc3cc(CSC[C@H](NC(=O)CNC(=O)[C@H](CO)NC1=O)C(=O)NCC(N)=O)cc(CSC[C@H](NC(=O)[C@H](C)N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CC(N)=O)C(=O)N2)c3 |r,wU:8.8,89.93,95.99,117.122,14.14,81.85,4.3,53.56,128.133,106.110,75.80,71.75,wD:28.34,45.62,35.140,(16.59,-5.46,;15.87,-4.08,;16.7,-2.77,;15.98,-1.39,;14.42,-1.34,;13.68,.02,;14.53,1.3,;16.07,1.25,;13.82,2.67,;14.64,3.96,;16.12,3.88,;12.28,2.71,;11.58,4.09,;12.41,5.38,;10.06,4.15,;10.1,5.7,;11.41,6.54,;12.87,6.02,;13.79,7.25,;12.93,8.53,;13.3,10.04,;12.17,11.11,;10.69,10.66,;10.32,9.16,;11.44,8.1,;8.52,4.19,;7.75,2.86,;6.22,2.89,;8.48,1.52,;10.09,1.46,;10.38,.29,;8.96,-.36,;7.79,.63,;6.01,.37,;6.05,1.9,;4.89,-.35,;4.89,-2.04,;4.1,-3.62,;3.33,-4.95,;1.8,-4.96,;1.04,-6.29,;-.49,-6.29,;.33,-8.23,;2.73,-7.61,;3.57,-6.04,;4.26,-7.62,;5.57,-7.5,;6.56,-6.32,;7.63,-7.4,;7.38,-5.02,;8.92,-5.11,;9.73,-3.8,;9.02,-2.44,;11.25,-3.86,;11.98,-5.24,;11.15,-6.55,;12.06,-2.55,;13.6,-2.63,;14.31,-3.98,;3.71,-9.14,;2.19,-9.41,;4.7,-10.32,;4.18,-11.77,;2.66,-12.04,;1.67,-10.86,;2.14,-13.48,;-1.26,-4.97,;-.49,-3.66,;-.46,-2.13,;-1.8,-1.39,;-1.83,.14,;-3.17,.87,;-3.29,2.41,;-1.97,3.2,;-.63,2.46,;-2.01,4.73,;-.7,5.52,;-3.42,5.44,;-4.48,.09,;-4.44,-1.45,;-5.83,.83,;-7.14,.02,;-7.1,-1.53,;-8.43,-2.33,;-9.88,-1.6,;-8.4,-3.89,;-8.47,.75,;-9.78,-.04,;-8.49,2.29,;-9.84,3.03,;-11.17,2.22,;-12.61,2.95,;-9.88,4.55,;-11.22,5.29,;-8.56,5.35,;-8.61,6.89,;-9.96,7.63,;-11.29,6.83,;-12.66,7.58,;-13.98,6.79,;-15.29,7.6,;-16.7,6.77,;-15.4,9.11,;-7.3,7.67,;-7.32,9.2,;-5.95,6.92,;-4.64,7.73,;-5.46,9.03,;-4.74,10.4,;-3.19,10.46,;-2.46,11.84,;-3.29,13.15,;-4.86,13.08,;-5.57,11.71,;-3.11,7.79,;-2.4,9.14,;-2.31,6.48,;-.77,6.56,;-.76,8.11,;.58,8.88,;.59,10.44,;1.93,11.21,;1.99,12.74,;3.29,13.47,;.6,13.48,;.59,5.85,;1.88,6.66,;.64,4.31,;2.01,3.61,;2.7,4.97,;4.26,5.05,;5.06,3.72,;4.96,6.43,;2.83,2.32,;4.37,2.4,;2.06,.95,;1.03,-3.64,)| Show InChI InChI=1S/C84H120N26O23S4/c1-42(85)68(119)108-61-40-136-37-45-23-44-24-46(25-45)38-137-41-62(109-76(127)55(28-64(86)114)102-72(123)50(15-8-19-92-83(88)89)98-74(125)53(26-43-11-4-3-5-12-43)101-71(122)51(16-9-20-93-84(90)91)99-78(129)58(34-112)107-77(128)56(29-67(117)118)103-80(61)131)82(133)110-21-10-17-63(110)81(132)104-54(27-47-30-94-49-14-7-6-13-48(47)49)75(126)106-59(35-113)79(130)100-52(18-22-134-2)73(124)105-57(33-111)69(120)96-32-66(116)97-60(39-135-36-44)70(121)95-31-65(87)115/h3-7,11-14,23-25,30,42,50-63,94,111-113H,8-10,15-22,26-29,31-41,85H2,1-2H3,(H2,86,114)(H2,87,115)(H,95,121)(H,96,120)(H,97,116)(H,98,125)(H,99,129)(H,100,130)(H,101,122)(H,102,123)(H,103,131)(H,104,132)(H,105,124)(H,106,126)(H,107,128)(H,108,119)(H,109,127)(H,117,118)(H4,88,89,92)(H4,90,91,93)/t42-,50-,51-,52-,53-,54?,55-,56-,57-,58-,59-,60-,61-,62-,63-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human plasma kallikrein using Z-Phe-Arg-AMC substrate after 30 mins by spectrophotometric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM50326037
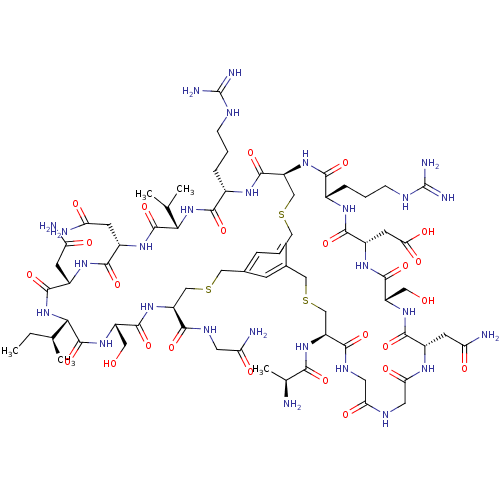
(CHEMBL1240604)Show SMILES CC[C@H](C)[C@@H]1NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]2CSCc3cc(CSC[C@H](NC(=O)[C@H](CO)NC1=O)C(=O)NCC(N)=O)cc(CSC[C@H](NC(=O)[C@H](C)N)C(=O)NCC(=O)NCC(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N2)c3)C(C)C |r| Show InChI InChI=1S/C72H115N27O24S3/c1-6-32(4)56-70(123)94-44(24-101)67(120)96-46(58(111)85-20-51(77)105)29-125-26-35-13-34-14-36(15-35)27-126-30-47(68(121)89-38(10-8-12-83-72(80)81)61(114)98-55(31(2)3)69(122)92-40(17-49(75)103)63(116)90-41(18-50(76)104)65(118)99-56)97-60(113)37(9-7-11-82-71(78)79)88-64(117)42(19-54(108)109)91-66(119)43(23-100)93-62(115)39(16-48(74)102)87-53(107)22-84-52(106)21-86-59(112)45(28-124-25-34)95-57(110)33(5)73/h13-15,31-33,37-47,55-56,100-101H,6-12,16-30,73H2,1-5H3,(H2,74,102)(H2,75,103)(H2,76,104)(H2,77,105)(H,84,106)(H,85,111)(H,86,112)(H,87,107)(H,88,117)(H,89,121)(H,90,116)(H,91,119)(H,92,122)(H,93,115)(H,94,123)(H,95,110)(H,96,120)(H,97,113)(H,98,114)(H,99,118)(H,108,109)(H4,78,79,82)(H4,80,81,83)/t32-,33-,37-,38-,39-,40-,41-,42-,43-,44-,45-,46-,47-,55-,56-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human plasma kallikrein using Z-Phe-Arg-AMC substrate after 30 mins by spectrophotometric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Kallikrein-2
(Homo sapiens (Human)) | BDBM50326050
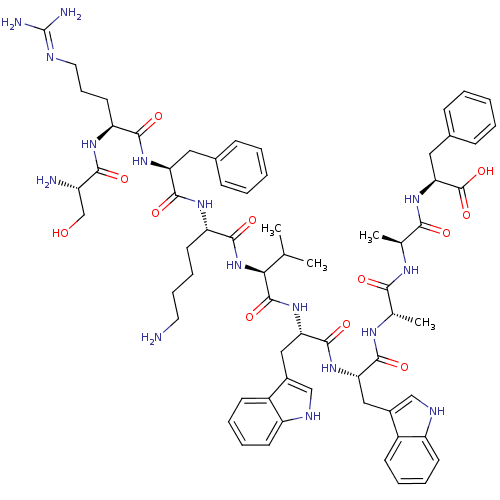
(CHEMBL1240617)Show SMILES CC(C)[C@H](NC(=O)[C@H](CCCCN)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@@H](N)CO)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](C)C(=O)N[C@@H](C)C(=O)N[C@@H](Cc1ccccc1)C(O)=O |r,wU:78.83,3.3,38.39,59.61,16.24,wD:73.78,45.45,27.35,7.12,83.87,(16.62,-15.91,;15.28,-16.68,;13.95,-15.91,;15.28,-18.23,;13.95,-18.99,;12.61,-18.23,;12.61,-16.7,;11.28,-19,;11.28,-20.54,;12.61,-21.31,;12.61,-22.86,;13.93,-23.62,;13.94,-25.16,;9.95,-18.22,;8.61,-18.99,;8.61,-20.53,;7.28,-18.22,;7.28,-16.68,;8.61,-15.91,;9.95,-16.69,;11.28,-15.92,;11.29,-14.38,;9.95,-13.61,;8.62,-14.37,;5.94,-18.98,;4.6,-18.23,;4.6,-16.69,;3.27,-19,;3.27,-20.54,;4.6,-21.32,;4.6,-22.85,;5.94,-23.6,;5.94,-25.14,;4.6,-25.92,;7.27,-25.9,;1.95,-18.22,;.61,-18.99,;.61,-20.53,;-.72,-18.22,;-2.04,-18.97,;-.72,-16.68,;.62,-15.89,;16.62,-18.99,;16.62,-20.53,;17.95,-18.22,;19.28,-19,;19.28,-20.54,;20.56,-21.41,;22,-20.88,;22.94,-22.1,;22.08,-23.38,;22.45,-24.87,;21.33,-25.95,;19.85,-25.51,;19.48,-24.02,;20.6,-22.96,;20.61,-18.23,;20.61,-16.7,;21.95,-18.99,;23.29,-18.22,;23.29,-16.69,;24.51,-15.74,;25.76,-16.65,;27.01,-15.74,;26.53,-14.28,;27.29,-12.94,;26.53,-11.61,;24.99,-11.61,;24.21,-12.94,;24.98,-14.28,;24.62,-18.99,;24.62,-20.54,;25.96,-18.23,;27.29,-19,;27.29,-20.54,;28.62,-18.24,;28.62,-16.69,;29.96,-18.99,;31.29,-18.22,;31.29,-16.68,;32.63,-18.99,;32.63,-20.53,;33.96,-18.22,;35.3,-18.99,;35.3,-20.53,;36.63,-21.31,;37.97,-20.54,;39.3,-21.31,;39.3,-22.85,;37.96,-23.62,;36.63,-22.85,;36.63,-18.22,;37.97,-18.99,;36.63,-16.69,)| Show InChI InChI=1S/C66H88N16O12/c1-37(2)55(82-60(88)49(26-15-16-28-67)77-62(90)51(30-40-18-7-5-8-19-40)78-59(87)50(27-17-29-71-66(69)70)76-58(86)46(68)36-83)64(92)80-53(33-43-35-73-48-25-14-12-23-45(43)48)63(91)79-52(32-42-34-72-47-24-13-11-22-44(42)47)61(89)75-38(3)56(84)74-39(4)57(85)81-54(65(93)94)31-41-20-9-6-10-21-41/h5-14,18-25,34-35,37-39,46,49-55,72-73,83H,15-17,26-33,36,67-68H2,1-4H3,(H,74,84)(H,75,89)(H,76,86)(H,77,90)(H,78,87)(H,79,91)(H,80,92)(H,81,85)(H,82,88)(H,93,94)(H4,69,70,71)/t38-,39-,46-,49-,50-,51-,52-,53-,54-,55-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Binding affinity to human kallikrein 2 |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM50326021
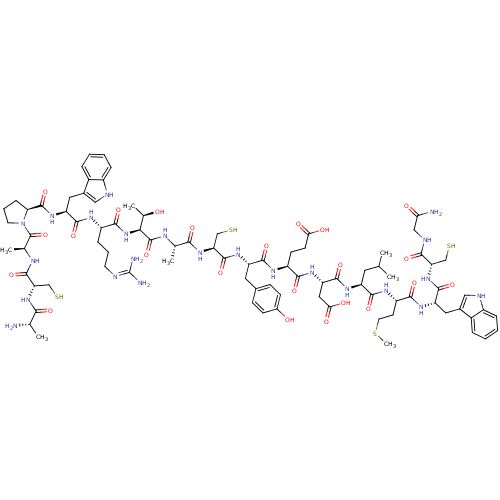
(CHEMBL1240699)Show SMILES CSCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CS)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@@H]1CCCN1C(=O)[C@H](C)NC(=O)[C@H](CS)NC(=O)[C@H](C)N)[C@@H](C)O)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CS)C(=O)NCC(N)=O |r,wU:97.103,71.84,24.29,56.57,8.12,106.111,112.115,103.108,45.48,wD:92.96,85.87,51.52,33.42,60.68,16.20,4.4,126.131,(43.17,-16.52,;41.84,-15.75,;41.84,-14.21,;40.5,-13.44,;40.5,-11.91,;39.17,-11.13,;37.84,-11.9,;37.84,-13.44,;36.51,-11.13,;36.51,-9.59,;37.84,-8.82,;37.84,-7.28,;39.17,-9.59,;35.17,-11.9,;33.84,-11.14,;33.84,-9.59,;32.5,-11.91,;32.5,-13.45,;33.84,-14.22,;35.18,-13.44,;33.84,-15.75,;31.17,-11.13,;29.83,-11.9,;29.83,-13.44,;28.5,-11.13,;28.5,-9.59,;29.83,-8.81,;29.83,-7.28,;28.51,-6.5,;31.16,-6.5,;27.17,-11.91,;25.83,-11.15,;25.83,-9.61,;24.49,-11.92,;24.49,-13.45,;25.83,-14.22,;27.16,-13.45,;28.49,-14.22,;28.5,-15.76,;29.85,-16.53,;27.16,-16.53,;25.83,-15.77,;23.16,-11.14,;21.82,-11.9,;21.82,-13.44,;20.49,-11.13,;20.49,-9.59,;21.82,-8.82,;19.15,-11.9,;17.82,-11.13,;17.82,-9.59,;16.48,-11.9,;16.48,-13.43,;15.15,-11.13,;13.8,-11.89,;13.8,-13.42,;12.47,-11.12,;11.14,-11.9,;9.81,-11.13,;9.81,-9.58,;8.47,-11.9,;8.47,-13.44,;9.8,-14.21,;9.8,-15.74,;11.14,-16.51,;11.12,-18.05,;9.83,-18.83,;12.5,-18.84,;7.14,-11.13,;5.8,-11.88,;5.8,-13.42,;4.46,-11.11,;4.48,-9.56,;5.74,-8.69,;7.03,-9.53,;8.23,-8.57,;7.68,-7.13,;8.38,-5.75,;7.55,-4.46,;6.02,-4.54,;5.31,-5.91,;6.14,-7.2,;3.14,-11.9,;1.81,-11.13,;1.81,-9.59,;.56,-12.04,;.57,-13.58,;-.9,-14.06,;-1.81,-12.81,;-.9,-11.57,;-1.58,-10.19,;-.8,-8.86,;-3.12,-10.17,;-3.91,-11.5,;-3.88,-8.83,;-5.42,-8.8,;-6.2,-10.13,;-6.18,-7.45,;-5.39,-6.14,;-6.14,-4.79,;-7.71,-7.44,;-8.49,-8.77,;-7.74,-10.11,;-10.05,-8.75,;-10.78,-7.41,;-10.8,-10.08,;12.47,-9.57,;11.13,-8.8,;13.82,-8.81,;41.84,-11.13,;41.84,-9.59,;43.18,-11.9,;44.51,-11.13,;44.51,-9.58,;45.72,-8.64,;46.94,-9.58,;48.21,-8.7,;47.77,-7.23,;48.57,-5.91,;47.84,-4.56,;46.3,-4.52,;45.5,-5.83,;46.24,-7.19,;45.84,-11.9,;45.84,-13.43,;47.18,-11.13,;48.51,-11.9,;48.51,-13.44,;49.85,-14.21,;49.85,-11.13,;49.85,-9.59,;51.18,-11.9,;52.52,-11.13,;53.85,-11.91,;55.18,-11.13,;53.85,-13.44,)| Show InChI InChI=1S/C86H123N23O23S4/c1-41(2)30-57(76(123)99-56(26-29-136-7)74(121)102-59(32-47-35-92-52-16-10-8-14-50(47)52)79(126)107-62(38-133)72(119)94-37-66(88)112)100-80(127)61(34-68(115)116)103-73(120)55(24-25-67(113)114)98-77(124)58(31-46-20-22-49(111)23-21-46)101-82(129)64(40-135)106-71(118)43(4)95-84(131)69(45(6)110)108-75(122)54(18-12-27-91-86(89)90)97-78(125)60(33-48-36-93-53-17-11-9-15-51(48)53)104-83(130)65-19-13-28-109(65)85(132)44(5)96-81(128)63(39-134)105-70(117)42(3)87/h8-11,14-17,20-23,35-36,41-45,54-65,69,92-93,110-111,133-135H,12-13,18-19,24-34,37-40,87H2,1-7H3,(H2,88,112)(H,94,119)(H,95,131)(H,96,128)(H,97,125)(H,98,124)(H,99,123)(H,100,127)(H,101,129)(H,102,121)(H,103,120)(H,104,130)(H,105,117)(H,106,118)(H,107,126)(H,108,122)(H,113,114)(H,115,116)(H4,89,90,91)/t42-,43-,44-,45+,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,65-,69-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.71E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human plasma kallikrein using Z-Phe-Arg-AMC substrate after 30 mins by spectrophotometric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM50326022
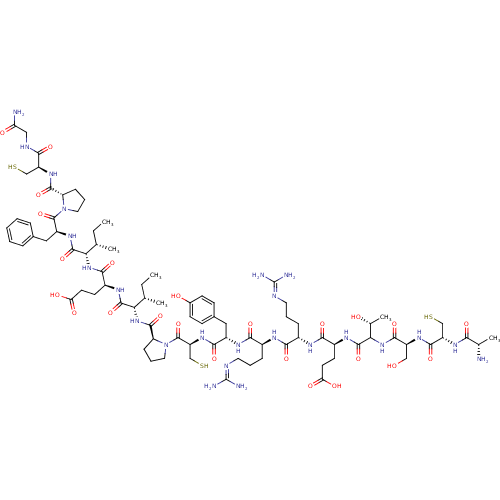
(CHEMBL1240698)Show SMILES [#6]-[#6]-[#6@H](-[#6])-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@@H](-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-[#16])-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccc(-[#8])cc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6](-[#7]-[#6](=O)-[#6@H](-[#6]-[#8])-[#7]-[#6](=O)-[#6@H](-[#6]-[#16])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7])-[#6@@H](-[#6])-[#8])-[#6@@H](-[#6])-[#6]-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccccc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#16])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O |r| Show InChI InChI=1S/C83H130N24O24S3/c1-7-41(3)63(78(128)98-53(35-45-16-10-9-11-17-45)80(130)106-32-14-20-58(106)75(125)101-55(38-132)67(117)92-36-60(85)111)103-71(121)51(27-29-62(114)115)95-77(127)64(42(4)8-2)104-76(126)59-21-15-33-107(59)81(131)57(40-134)102-72(122)52(34-46-22-24-47(110)25-23-46)97-69(119)49(19-13-31-91-83(88)89)93-68(118)48(18-12-30-90-82(86)87)94-70(120)50(26-28-61(112)113)96-79(129)65(44(6)109)105-73(123)54(37-108)99-74(124)56(39-133)100-66(116)43(5)84/h9-11,16-17,22-25,41-44,48-59,63-65,108-110,132-134H,7-8,12-15,18-21,26-40,84H2,1-6H3,(H2,85,111)(H,92,117)(H,93,118)(H,94,120)(H,95,127)(H,96,129)(H,97,119)(H,98,128)(H,99,124)(H,100,116)(H,101,125)(H,102,122)(H,103,121)(H,104,126)(H,105,123)(H,112,113)(H,114,115)(H4,86,87,90)(H4,88,89,91)/t41-,42-,43-,44+,48-,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,63-,64-,65?/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7.18E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human plasma kallikrein using Z-Phe-Arg-AMC substrate after 30 mins by spectrophotometric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM50326023
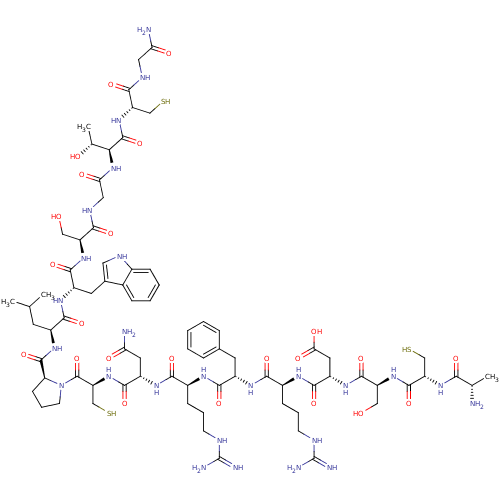
((2R,5S,8S,11S,14S,17S)-1-((S)-2-((5R,8S,14S,17S,20...)Show SMILES CC(C)C[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@H](CS)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CS)NC(=O)[C@H](C)N)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CS)C(=O)NCC(N)=O |r| Show InChI InChI=1S/C77H118N26O23S3/c1-36(2)23-45(66(117)93-47(25-40-28-87-42-16-9-8-15-41(40)42)68(119)97-50(31-104)62(113)89-30-58(109)102-60(38(4)106)74(125)100-52(33-127)63(114)88-29-57(80)108)96-73(124)55-19-12-22-103(55)75(126)54(35-129)101-69(120)48(26-56(79)107)94-65(116)44(18-11-21-86-77(83)84)90-67(118)46(24-39-13-6-5-7-14-39)92-64(115)43(17-10-20-85-76(81)82)91-70(121)49(27-59(110)111)95-71(122)51(32-105)98-72(123)53(34-128)99-61(112)37(3)78/h5-9,13-16,28,36-38,43-55,60,87,104-106,127-129H,10-12,17-27,29-35,78H2,1-4H3,(H2,79,107)(H2,80,108)(H,88,114)(H,89,113)(H,90,118)(H,91,121)(H,92,115)(H,93,117)(H,94,116)(H,95,122)(H,96,124)(H,97,119)(H,98,123)(H,99,112)(H,100,125)(H,101,120)(H,102,109)(H,110,111)(H4,81,82,85)(H4,83,84,86)/t37-,38+,43-,44-,45-,46-,47-,48-,49-,50-,51-,52-,53-,54-,55-,60-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human plasma kallikrein using Z-Phe-Arg-AMC substrate after 30 mins by spectrophotometric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM50326019
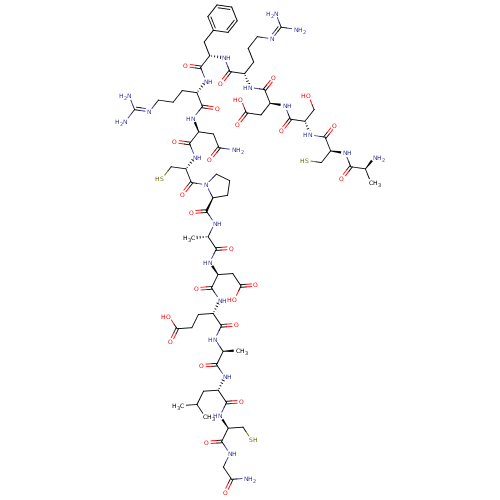
((2R,5S,8S,11S,14S,17S)-1-((S)-2-((5R,8S,11S,14S,17...)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-[#16])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#8])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#8])-[#7]-[#6](=O)-[#6@H](-[#6]-[#16])-[#7]-[#6](=O)-[#6@H](-[#6])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#16])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O |r| Show InChI InChI=1S/C72H115N25O25S3/c1-32(2)22-40(62(114)95-46(29-123)58(110)82-27-51(75)100)88-56(108)34(4)83-59(111)39(17-18-52(101)102)87-65(117)43(25-53(103)104)89-57(109)35(5)84-69(121)49-16-11-21-97(49)70(122)48(31-125)96-64(116)42(24-50(74)99)91-61(113)38(15-10-20-81-72(78)79)85-63(115)41(23-36-12-7-6-8-13-36)90-60(112)37(14-9-19-80-71(76)77)86-66(118)44(26-54(105)106)92-67(119)45(28-98)93-68(120)47(30-124)94-55(107)33(3)73/h6-8,12-13,32-35,37-49,98,123-125H,9-11,14-31,73H2,1-5H3,(H2,74,99)(H2,75,100)(H,82,110)(H,83,111)(H,84,121)(H,85,115)(H,86,118)(H,87,117)(H,88,108)(H,89,109)(H,90,112)(H,91,113)(H,92,119)(H,93,120)(H,94,107)(H,95,114)(H,96,116)(H,101,102)(H,103,104)(H,105,106)(H4,76,77,80)(H4,78,79,81)/t33-,34-,35-,37-,38-,39-,40-,41-,42-,43-,44-,45-,46-,47-,48-,49-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human plasma kallikrein using Z-Phe-Arg-AMC substrate after 30 mins by spectrophotometric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM50326020

((2R,3S,6S,9S,12S,15S,18R,21S,27S)-6-((1H-indol-3-y...)Show SMILES CC(C)[C@H](NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CS)NC(=O)[C@H](CCCN=C(N)N)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CS)NC(=O)[C@@H](C)N)[C@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H]([C@@H](C)O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CS)C(=O)NCC(N)=O |r,wU:39.44,52.52,3.3,60.62,83.82,66.67,24.31,wD:107.110,18.20,7.14,75.74,69.70,97.98,98.101,111.113,(31.86,-9.36,;30.53,-10.14,;29.2,-9.37,;30.53,-11.67,;29.2,-12.44,;27.87,-11.66,;27.87,-10.12,;26.54,-12.43,;26.54,-13.97,;27.87,-14.74,;27.88,-16.28,;29.2,-17.07,;29.24,-18.61,;27.87,-19.38,;30.54,-19.37,;25.2,-11.67,;23.87,-10.89,;23.87,-9.35,;22.53,-11.66,;22.53,-13.2,;21.2,-13.97,;21.19,-10.9,;19.86,-11.68,;19.86,-13.22,;18.52,-10.91,;18.52,-9.37,;17.19,-8.6,;17.19,-7.06,;15.85,-6.29,;15.85,-4.75,;17.18,-3.98,;14.51,-3.97,;17.18,-11.67,;15.85,-10.9,;15.85,-9.35,;14.51,-11.67,;13.17,-10.91,;11.84,-11.67,;11.84,-13.21,;10.5,-10.9,;10.51,-9.36,;9.17,-8.6,;9.17,-7.05,;10.48,-6.3,;7.83,-6.29,;9.18,-11.68,;7.86,-10.89,;7.87,-9.35,;6.53,-11.67,;5.2,-10.89,;3.87,-11.67,;3.87,-13.21,;2.53,-10.91,;1.2,-11.69,;-.12,-10.9,;-.13,-9.36,;-1.46,-11.67,;-2.8,-10.9,;-4.13,-11.67,;-4.14,-13.21,;-5.47,-10.9,;-5.46,-9.36,;-4.14,-8.59,;-6.8,-11.68,;-8.13,-10.91,;-8.13,-9.37,;-9.47,-11.68,;-9.46,-13.22,;-10.8,-10.92,;2.53,-9.36,;1.19,-8.61,;3.85,-8.6,;31.86,-12.44,;31.87,-13.98,;33.19,-11.66,;34.53,-12.44,;34.53,-13.97,;35.86,-14.74,;37.19,-13.97,;35.86,-16.28,;35.86,-11.66,;35.85,-10.12,;37.2,-12.43,;38.53,-11.67,;38.53,-10.13,;39.75,-9.19,;41.02,-10.06,;42.23,-9.11,;41.7,-7.66,;42.41,-6.3,;41.59,-5,;40.06,-5.06,;39.34,-6.42,;40.17,-7.72,;39.86,-12.43,;39.86,-13.97,;41.19,-11.65,;42.52,-12.42,;42.53,-13.97,;41.19,-14.74,;43.85,-14.73,;43.86,-11.65,;43.85,-10.11,;45.11,-12.56,;45.11,-14.11,;46.58,-14.56,;47.47,-13.32,;46.56,-12.07,;47.25,-10.7,;46.41,-9.41,;48.79,-10.6,;49.64,-11.89,;48.88,-13.22,;49.65,-14.55,;51.17,-11.88,;51.93,-10.54,;51.95,-13.21,;53.49,-13.21,;54.27,-14.54,;55.8,-14.53,;53.5,-15.88,)| Show InChI InChI=1S/C70H112N26O22S3/c1-31(2)53(67(117)89-42(22-47(72)99)62(112)88-41(21-35-23-80-37-12-7-6-11-36(35)37)63(113)95-55(34(5)98)68(118)96-20-10-15-46(96)65(115)92-44(29-120)58(108)81-24-48(73)100)94-61(111)39(14-9-19-79-70(76)77)87-64(114)45(30-121)91-60(110)38(13-8-18-78-69(74)75)85-49(101)25-82-57(107)40(16-17-52(104)105)86-50(102)26-84-66(116)54(33(4)97)93-51(103)27-83-59(109)43(28-119)90-56(106)32(3)71/h6-7,11-12,23,31-34,38-46,53-55,80,97-98,119-121H,8-10,13-22,24-30,71H2,1-5H3,(H2,72,99)(H2,73,100)(H,81,108)(H,82,107)(H,83,109)(H,84,116)(H,85,101)(H,86,102)(H,87,114)(H,88,112)(H,89,117)(H,90,106)(H,91,110)(H,92,115)(H,93,103)(H,94,111)(H,95,113)(H,104,105)(H4,74,75,78)(H4,76,77,79)/t32-,33+,34-,38+,39+,40+,41+,42+,43+,44+,45+,46+,53+,54+,55+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Inhibition of human plasma kallikrein using Z-Phe-Arg-AMC substrate after 30 mins by spectrophotometric assay |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Pancreatic triacylglycerol lipase
(Homo sapiens (Human)) | BDBM50326047
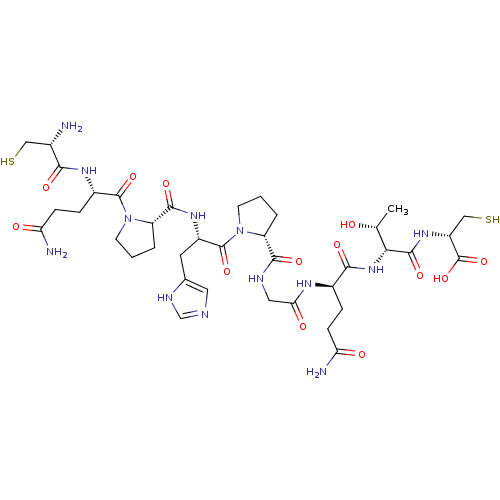
((6S,9R,12R)-1-((R)-1-((R)-2-((S)-1-((S)-5-amino-2-...)Show SMILES C[C@@H](O)[C@@H](NC(=O)[C@@H](CCC(N)=O)NC(=O)CNC(=O)[C@H]1CCCN1C(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](N)CS)C(=O)N[C@H](CS)C(O)=O |r| Show InChI InChI=1S/C38H59N13O13S2/c1-18(52)30(35(60)48-24(16-66)38(63)64)49-32(57)21(6-8-27(40)53)45-29(55)14-43-33(58)25-4-2-10-50(25)37(62)23(12-19-13-42-17-44-19)47-34(59)26-5-3-11-51(26)36(61)22(7-9-28(41)54)46-31(56)20(39)15-65/h13,17-18,20-26,30,52,65-66H,2-12,14-16,39H2,1H3,(H2,40,53)(H2,41,54)(H,42,44)(H,43,58)(H,45,55)(H,46,56)(H,47,59)(H,48,60)(H,49,57)(H,63,64)/t18-,20+,21-,22+,23+,24-,25-,26+,30-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Binding affinity to pancreatic lipase |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |
Prostate-specific antigen
(Homo sapiens (Human)) | BDBM50326051
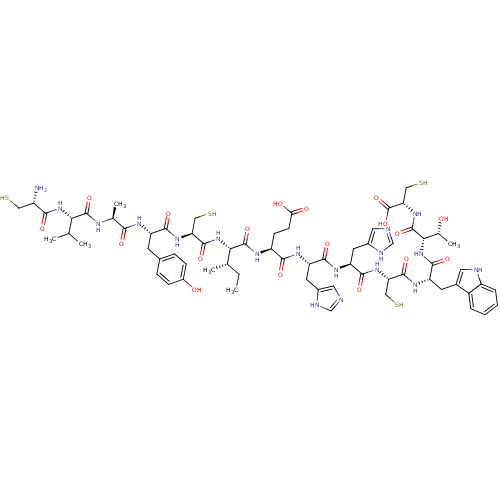
((2R,5S,8S,11R,14S,17S,20S)-14,17-bis((1H-imidazol-...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CS)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@@H](N)CS)C(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@@H](CS)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CS)C(O)=O |r| Show InChI InChI=1S/C67H94N18O18S4/c1-7-32(4)53(84-63(98)49(27-106)81-58(93)44(18-35-12-14-39(87)15-13-35)76-55(90)33(5)74-64(99)52(31(2)3)83-56(91)41(68)25-104)65(100)75-43(16-17-51(88)89)57(92)78-46(20-37-23-69-29-72-37)59(94)79-47(21-38-24-70-30-73-38)60(95)80-48(26-105)62(97)77-45(19-36-22-71-42-11-9-8-10-40(36)42)61(96)85-54(34(6)86)66(101)82-50(28-107)67(102)103/h8-15,22-24,29-34,41,43-50,52-54,71,86-87,104-107H,7,16-21,25-28,68H2,1-6H3,(H,69,72)(H,70,73)(H,74,99)(H,75,100)(H,76,90)(H,77,97)(H,78,92)(H,79,94)(H,80,95)(H,81,93)(H,82,101)(H,83,91)(H,84,98)(H,85,96)(H,88,89)(H,102,103)/t32-,33-,34+,41-,43-,44-,45-,46-,47-,48-,49-,50-,52-,53-,54-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a |
Laboratory of Molecular Biology, Medical Research Council
Curated by ChEMBL
| Assay Description
Binding affinity to prostrate specific antigen |
Nat Chem Biol 5: 502-7 (2009)
Article DOI: 10.1038/nchembio.184
BindingDB Entry DOI: 10.7270/Q2Z60P9P |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data






















