 Found 264 hits with Last Name = 'guillon' and Initial = 'g'
Found 264 hits with Last Name = 'guillon' and Initial = 'g' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Vasopressin V1b receptor
(RAT) | BDBM50205313
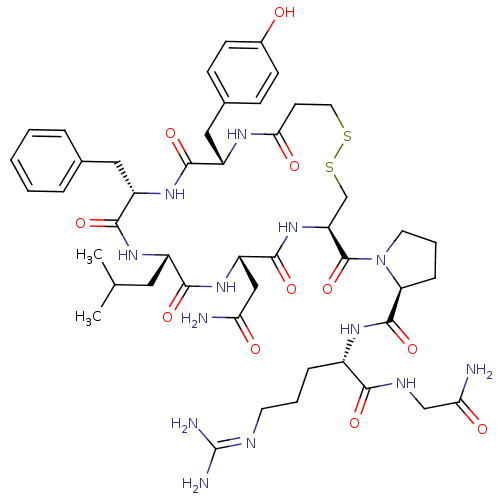
(CHEMBL265859 | d[Leu4]AVP)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C47H67N13O11S2/c1-26(2)20-31-41(66)58-34(23-37(48)62)44(69)59-35(46(71)60-18-7-11-36(60)45(70)55-30(10-6-17-52-47(50)51)40(65)53-24-38(49)63)25-73-72-19-16-39(64)54-32(22-28-12-14-29(61)15-13-28)42(67)57-33(43(68)56-31)21-27-8-4-3-5-9-27/h3-5,8-9,12-15,26,30-36,61H,6-7,10-11,16-25H2,1-2H3,(H2,48,62)(H2,49,63)(H,53,65)(H,54,64)(H,55,70)(H,56,68)(H,57,67)(H,58,66)(H,59,69)(H4,50,51,52)/t30-,31-,32-,33-,34-,35-,36-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50038601
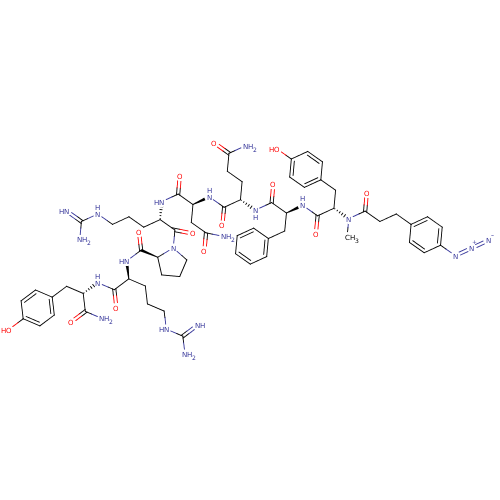
(4-N3-C6H4CH2CH2CO-D-Tyr(Me)-Phe-Gln-Asn-Arg-Pro-Ar...)Show SMILES CN([C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O)C(=O)CCc1ccc(cc1)N=[N+]=[N-] Show InChI InChI=1S/C63H84N20O13/c1-82(53(88)28-19-36-13-20-40(21-14-36)80-81-71)50(34-39-17-24-42(85)25-18-39)60(95)79-47(33-37-8-3-2-4-9-37)57(92)74-44(26-27-51(64)86)56(91)78-48(35-52(65)87)58(93)76-45(11-6-30-73-63(69)70)61(96)83-31-7-12-49(83)59(94)75-43(10-5-29-72-62(67)68)55(90)77-46(54(66)89)32-38-15-22-41(84)23-16-38/h2-4,8-9,13-18,20-25,43-50,84-85H,5-7,10-12,19,26-35H2,1H3,(H2,64,86)(H2,65,87)(H2,66,89)(H,74,92)(H,75,94)(H,76,93)(H,77,90)(H,78,91)(H,79,95)(H4,67,68,72)(H4,69,70,73)/t43-,44-,45-,46-,47-,48-,49-,50-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UPR 9023 CNRS
Curated by ChEMBL
| Assay Description
Tested for inhibition constant determined from vasopressin induced inositol phosphates accumulation performed on WRK1 cell line of V1a receptor subty... |
J Med Chem 37: 1841-9 (1994)
BindingDB Entry DOI: 10.7270/Q2R49PST |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50038602
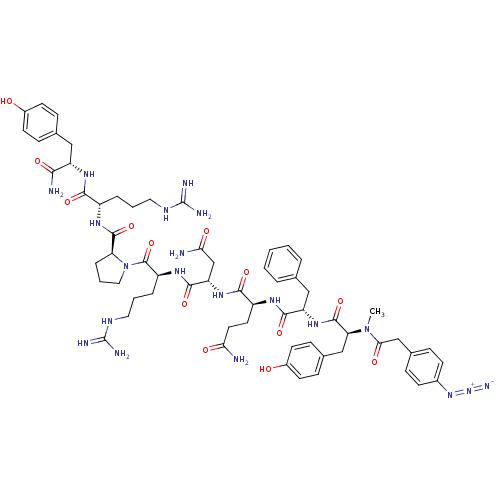
(4-N3-C6H4CH2CO-D-Tyr(Me)-Phe-Gln-Asn-Arg-Pro-Arg-T...)Show SMILES CN([C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O)C(=O)Cc1ccc(cc1)N=[N+]=[N-] Show InChI InChI=1S/C62H82N20O13/c1-81(52(87)33-38-13-19-39(20-14-38)79-80-70)49(32-37-17-23-41(84)24-18-37)59(94)78-46(31-35-8-3-2-4-9-35)56(91)73-43(25-26-50(63)85)55(90)77-47(34-51(64)86)57(92)75-44(11-6-28-72-62(68)69)60(95)82-29-7-12-48(82)58(93)74-42(10-5-27-71-61(66)67)54(89)76-45(53(65)88)30-36-15-21-40(83)22-16-36/h2-4,8-9,13-24,42-49,83-84H,5-7,10-12,25-34H2,1H3,(H2,63,85)(H2,64,86)(H2,65,88)(H,73,91)(H,74,93)(H,75,92)(H,76,89)(H,77,90)(H,78,94)(H4,66,67,71)(H4,68,69,72)/t42-,43-,44-,45-,46-,47-,48-,49-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UPR 9023 CNRS
Curated by ChEMBL
| Assay Description
Inhibition constant for V1a receptor of rat liver membrane |
J Med Chem 37: 1841-9 (1994)
BindingDB Entry DOI: 10.7270/Q2R49PST |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205305
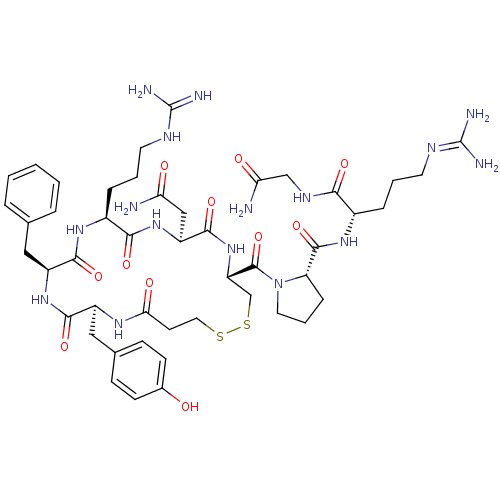
(CHEMBL375324 | d[Arg4]AVP)Show SMILES [#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@@H]-1-[#6]-[#16]-[#16]-[#6]-[#6]-[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccc(-[#8])cc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccccc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#7]-[#6](-[#7])=[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-1 Show InChI InChI=1S/C47H68N16O11S2/c48-36(65)23-33-43(72)62-34(45(74)63-19-6-11-35(63)44(73)59-29(9-4-17-54-46(50)51)39(68)56-24-37(49)66)25-76-75-20-16-38(67)57-31(22-27-12-14-28(64)15-13-27)41(70)60-32(21-26-7-2-1-3-8-26)42(71)58-30(40(69)61-33)10-5-18-55-47(52)53/h1-3,7-8,12-15,29-35,64H,4-6,9-11,16-25H2,(H2,48,65)(H2,49,66)(H,56,68)(H,57,67)(H,58,71)(H,59,73)(H,60,70)(H,61,69)(H,62,72)(H4,50,51,54)(H4,52,53,55)/t29-,30-,31-,32-,33-,34-,35-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50038603
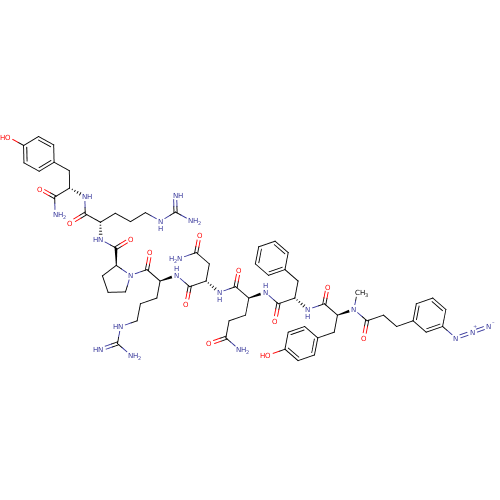
(3-N3-C6H4CH2CH2CO-D-Tyr(Me)-Phe-Gln-Asn-Arg-Pro-Ar...)Show SMILES CN([C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O)C(=O)CCc1cccc(c1)N=[N+]=[N-] Show InChI InChI=1S/C63H84N20O13/c1-82(53(88)27-20-37-11-5-12-40(31-37)80-81-71)50(34-39-18-23-42(85)24-19-39)60(95)79-47(33-36-9-3-2-4-10-36)57(92)74-44(25-26-51(64)86)56(91)78-48(35-52(65)87)58(93)76-45(14-7-29-73-63(69)70)61(96)83-30-8-15-49(83)59(94)75-43(13-6-28-72-62(67)68)55(90)77-46(54(66)89)32-38-16-21-41(84)22-17-38/h2-5,9-12,16-19,21-24,31,43-50,84-85H,6-8,13-15,20,25-30,32-35H2,1H3,(H2,64,86)(H2,65,87)(H2,66,89)(H,74,92)(H,75,94)(H,76,93)(H,77,90)(H,78,91)(H,79,95)(H4,67,68,72)(H4,69,70,73)/t43-,44-,45-,46-,47-,48-,49-,50-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UPR 9023 CNRS
Curated by ChEMBL
| Assay Description
Tested for inhibition constant determined from vasopressin induced inositol phosphates accumulation performed on WRK1 cell line of V1a receptor subty... |
J Med Chem 37: 1841-9 (1994)
BindingDB Entry DOI: 10.7270/Q2R49PST |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205309
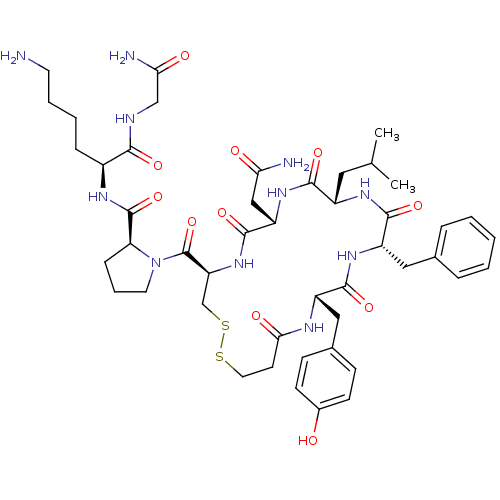
(CHEMBL412972 | d[Leu4,Lys8]VP)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)NCC(N)=O Show InChI InChI=1S/C47H67N11O11S2/c1-27(2)21-32-42(64)56-35(24-38(49)60)45(67)57-36(47(69)58-19-8-12-37(58)46(68)53-31(11-6-7-18-48)41(63)51-25-39(50)61)26-71-70-20-17-40(62)52-33(23-29-13-15-30(59)16-14-29)43(65)55-34(44(66)54-32)22-28-9-4-3-5-10-28/h3-5,9-10,13-16,27,31-37,59H,6-8,11-12,17-26,48H2,1-2H3,(H2,49,60)(H2,50,61)(H,51,63)(H,52,62)(H,53,68)(H,54,66)(H,55,65)(H,56,64)(H,57,67)/t31-,32-,33-,34-,35-,36-,37-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50038603

(3-N3-C6H4CH2CH2CO-D-Tyr(Me)-Phe-Gln-Asn-Arg-Pro-Ar...)Show SMILES CN([C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O)C(=O)CCc1cccc(c1)N=[N+]=[N-] Show InChI InChI=1S/C63H84N20O13/c1-82(53(88)27-20-37-11-5-12-40(31-37)80-81-71)50(34-39-18-23-42(85)24-19-39)60(95)79-47(33-36-9-3-2-4-10-36)57(92)74-44(25-26-51(64)86)56(91)78-48(35-52(65)87)58(93)76-45(14-7-29-73-63(69)70)61(96)83-30-8-15-49(83)59(94)75-43(13-6-28-72-62(67)68)55(90)77-46(54(66)89)32-38-16-21-41(84)22-17-38/h2-5,9-12,16-19,21-24,31,43-50,84-85H,6-8,13-15,20,25-30,32-35H2,1H3,(H2,64,86)(H2,65,87)(H2,66,89)(H,74,92)(H,75,94)(H,76,93)(H,77,90)(H,78,91)(H,79,95)(H4,67,68,72)(H4,69,70,73)/t43-,44-,45-,46-,47-,48-,49-,50-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UPR 9023 CNRS
Curated by ChEMBL
| Assay Description
Inhibition constant for V1a receptor of rat liver membrane |
J Med Chem 37: 1841-9 (1994)
BindingDB Entry DOI: 10.7270/Q2R49PST |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50038599
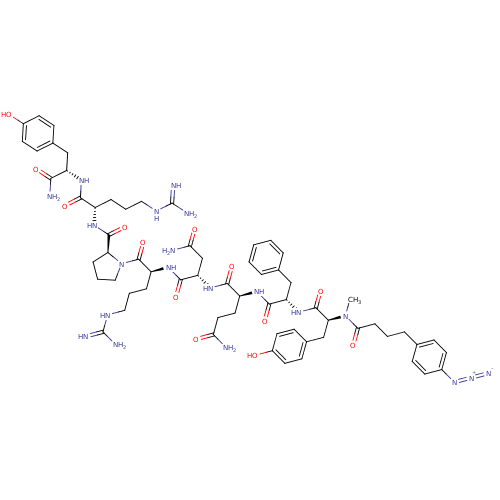
(4-N3-C6H4CH2CH2CH2CO-D-Tyr(Me)-Phe-Gln-Asn-Arg-Pro...)Show SMILES CN([C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O)C(=O)CCCc1ccc(cc1)N=[N+]=[N-] Show InChI InChI=1S/C64H86N20O13/c1-83(54(89)15-5-11-37-16-22-41(23-17-37)81-82-72)51(35-40-20-26-43(86)27-21-40)61(96)80-48(34-38-9-3-2-4-10-38)58(93)75-45(28-29-52(65)87)57(92)79-49(36-53(66)88)59(94)77-46(13-7-31-74-64(70)71)62(97)84-32-8-14-50(84)60(95)76-44(12-6-30-73-63(68)69)56(91)78-47(55(67)90)33-39-18-24-42(85)25-19-39/h2-4,9-10,16-27,44-51,85-86H,5-8,11-15,28-36H2,1H3,(H2,65,87)(H2,66,88)(H2,67,90)(H,75,93)(H,76,95)(H,77,94)(H,78,91)(H,79,92)(H,80,96)(H4,68,69,73)(H4,70,71,74)/t44-,45-,46-,47-,48-,49-,50-,51-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UPR 9023 CNRS
Curated by ChEMBL
| Assay Description
Inhibition constant for V1a receptor of rat liver membrane |
J Med Chem 37: 1841-9 (1994)
BindingDB Entry DOI: 10.7270/Q2R49PST |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205296

(CHEMBL385739 | d[Arg4,Dab8]VP)Show SMILES NCC[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1CSSCCC(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(N)=O)C(=O)N1)C(=O)NCC(N)=O Show InChI InChI=1S/C45H64N14O11S2/c46-16-14-29(38(64)52-23-36(48)62)55-43(69)34-9-5-18-59(34)44(70)33-24-72-71-19-15-37(63)53-30(21-26-10-12-27(60)13-11-26)40(66)56-31(20-25-6-2-1-3-7-25)41(67)54-28(8-4-17-51-45(49)50)39(65)57-32(22-35(47)61)42(68)58-33/h1-3,6-7,10-13,28-34,60H,4-5,8-9,14-24,46H2,(H2,47,61)(H2,48,62)(H,52,64)(H,53,63)(H,54,67)(H,55,69)(H,56,66)(H,57,65)(H,58,68)(H4,49,50,51)/t28-,29-,30-,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205291
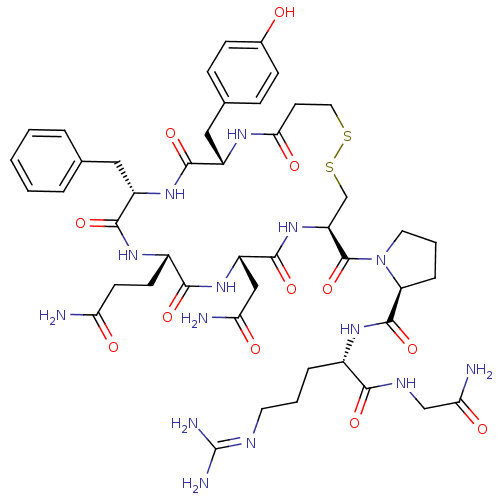
((S)-1-((4R,7S,10S,13S,16S)-7-(2-amino-2-oxoethyl)-...)Show SMILES [#7]-[#6](=O)-[#6]-[#6]-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H64N14O12S2/c47-35(62)15-14-29-40(67)58-32(22-36(48)63)43(70)59-33(45(72)60-18-5-9-34(60)44(71)56-28(8-4-17-52-46(50)51)39(66)53-23-37(49)64)24-74-73-19-16-38(65)54-30(21-26-10-12-27(61)13-11-26)41(68)57-31(42(69)55-29)20-25-6-2-1-3-7-25/h1-3,6-7,10-13,28-34,61H,4-5,8-9,14-24H2,(H2,47,62)(H2,48,63)(H2,49,64)(H,53,66)(H,54,65)(H,55,69)(H,56,71)(H,57,68)(H,58,67)(H,59,70)(H4,50,51,52)/t28-,29-,30-,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V2 receptor
(Rattus norvegicus (Rat)) | BDBM50205305

(CHEMBL375324 | d[Arg4]AVP)Show SMILES [#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@@H]-1-[#6]-[#16]-[#16]-[#6]-[#6]-[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccc(-[#8])cc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-c2ccccc2)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#7]-[#6](-[#7])=[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#7])=O)-[#6](=O)-[#7]-1 Show InChI InChI=1S/C47H68N16O11S2/c48-36(65)23-33-43(72)62-34(45(74)63-19-6-11-35(63)44(73)59-29(9-4-17-54-46(50)51)39(68)56-24-37(49)66)25-76-75-20-16-38(67)57-31(22-27-12-14-28(64)15-13-27)41(70)60-32(21-26-7-2-1-3-8-26)42(71)58-30(40(69)61-33)10-5-18-55-47(52)53/h1-3,7-8,12-15,29-35,64H,4-6,9-11,16-25H2,(H2,48,65)(H2,49,66)(H,56,68)(H,57,67)(H,58,71)(H,59,73)(H,60,70)(H,61,69)(H,62,72)(H4,50,51,54)(H4,52,53,55)/t29-,30-,31-,32-,33-,34-,35-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from vasopressin V2 receptor in rat kidney membranes |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50038602

(4-N3-C6H4CH2CO-D-Tyr(Me)-Phe-Gln-Asn-Arg-Pro-Arg-T...)Show SMILES CN([C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O)C(=O)Cc1ccc(cc1)N=[N+]=[N-] Show InChI InChI=1S/C62H82N20O13/c1-81(52(87)33-38-13-19-39(20-14-38)79-80-70)49(32-37-17-23-41(84)24-18-37)59(94)78-46(31-35-8-3-2-4-9-35)56(91)73-43(25-26-50(63)85)55(90)77-47(34-51(64)86)57(92)75-44(11-6-28-72-62(68)69)60(95)82-29-7-12-48(82)58(93)74-42(10-5-27-71-61(66)67)54(89)76-45(53(65)88)30-36-15-21-40(83)22-16-36/h2-4,8-9,13-24,42-49,83-84H,5-7,10-12,25-34H2,1H3,(H2,63,85)(H2,64,86)(H2,65,88)(H,73,91)(H,74,93)(H,75,92)(H,76,89)(H,77,90)(H,78,94)(H4,66,67,71)(H4,68,69,72)/t42-,43-,44-,45-,46-,47-,48-,49-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UPR 9023 CNRS
Curated by ChEMBL
| Assay Description
Tested for inhibition constant determined from vasopressin induced inositol phosphates accumulation performed on WRK1 cell line of V1a receptor subty... |
J Med Chem 37: 1841-9 (1994)
BindingDB Entry DOI: 10.7270/Q2R49PST |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50145126

((2S)-2-{[(2R)-1-{[(4S,7R,10S,13R,16S)-13-benzyl-7-...)Show SMILES CC(C)C[C@@H]1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@@H](NC(=O)[C@@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O Show InChI InChI=1S/C47H67N13O11S2/c1-26(2)20-31-41(66)58-34(23-37(48)62)44(69)59-35(46(71)60-18-7-11-36(60)45(70)55-30(10-6-17-52-47(50)51)40(65)53-24-38(49)63)25-73-72-19-16-39(64)54-32(22-28-12-14-29(61)15-13-28)42(67)57-33(43(68)56-31)21-27-8-4-3-5-9-27/h3-5,8-9,12-15,26,30-36,61H,6-7,10-11,16-25H2,1-2H3,(H2,48,62)(H2,49,63)(H,53,65)(H,54,64)(H,55,70)(H,56,68)(H,57,67)(H,58,66)(H,59,69)(H4,50,51,52)/t30-,31-,32-,33+,34+,35+,36+/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Ohio
Curated by ChEMBL
| Assay Description
Binding affinity against human vasopressin V1b receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT recepto... |
J Med Chem 47: 2375-88 (2004)
Article DOI: 10.1021/jm030611c
BindingDB Entry DOI: 10.7270/Q2GF0V8C |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50038604
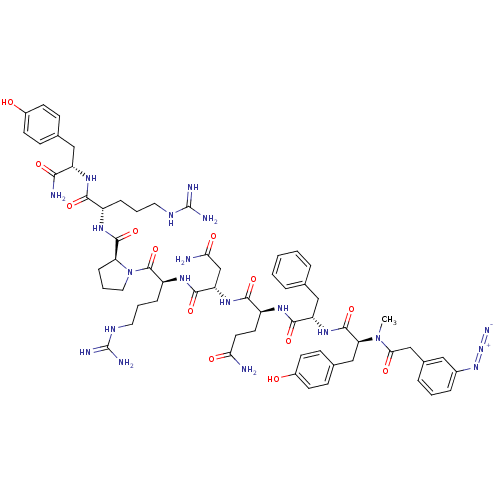
(3-N3-C6H4CH2CO-D-Tyr(Me)-Phe-Gln-Asn-Arg-Pro-Arg-T...)Show SMILES CN([C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O)C(=O)Cc1cccc(c1)N=[N+]=[N-] Show InChI InChI=1S/C62H82N20O13/c1-81(52(87)33-38-11-5-12-39(29-38)79-80-70)49(32-37-18-22-41(84)23-19-37)59(94)78-46(31-35-9-3-2-4-10-35)56(91)73-43(24-25-50(63)85)55(90)77-47(34-51(64)86)57(92)75-44(14-7-27-72-62(68)69)60(95)82-28-8-15-48(82)58(93)74-42(13-6-26-71-61(66)67)54(89)76-45(53(65)88)30-36-16-20-40(83)21-17-36/h2-5,9-12,16-23,29,42-49,83-84H,6-8,13-15,24-28,30-34H2,1H3,(H2,63,85)(H2,64,86)(H2,65,88)(H,73,91)(H,74,93)(H,75,92)(H,76,89)(H,77,90)(H,78,94)(H4,66,67,71)(H4,68,69,72)/t42-,43-,44-,45-,46-,47-,48-,49-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UPR 9023 CNRS
Curated by ChEMBL
| Assay Description
Tested for inhibition constant determined from vasopressin induced inositol phosphates accumulation performed on WRK1 cell line of V1a receptor subty... |
J Med Chem 37: 1841-9 (1994)
BindingDB Entry DOI: 10.7270/Q2R49PST |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205297
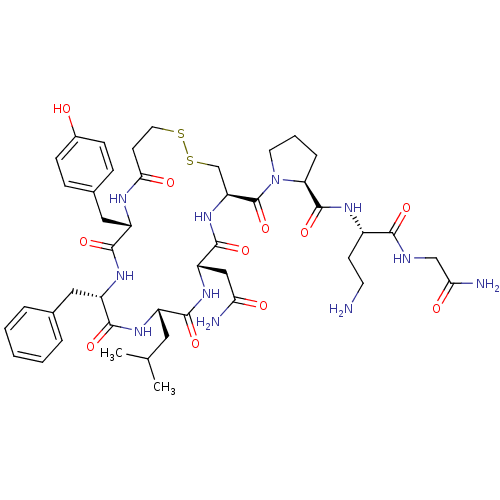
(CHEMBL412973 | d[Leu4,Dab8]VP)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCN)C(=O)NCC(N)=O Show InChI InChI=1S/C45H63N11O11S2/c1-25(2)19-30-40(62)54-33(22-36(47)58)43(65)55-34(45(67)56-17-6-9-35(56)44(66)51-29(14-16-46)39(61)49-23-37(48)59)24-69-68-18-15-38(60)50-31(21-27-10-12-28(57)13-11-27)41(63)53-32(42(64)52-30)20-26-7-4-3-5-8-26/h3-5,7-8,10-13,25,29-35,57H,6,9,14-24,46H2,1-2H3,(H2,47,58)(H2,48,59)(H,49,61)(H,50,60)(H,51,66)(H,52,64)(H,53,63)(H,54,62)(H,55,65)/t29-,30-,31-,32-,33-,34-,35-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205300
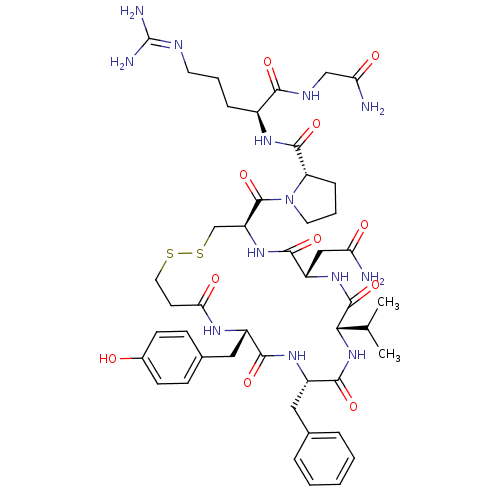
(CHEMBL221436 | d[Val4]AVP)Show SMILES [#6]-[#6](-[#6])-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H65N13O11S2/c1-25(2)38-44(69)56-32(22-35(47)61)41(66)57-33(45(70)59-18-7-11-34(59)43(68)54-29(10-6-17-51-46(49)50)39(64)52-23-36(48)62)24-72-71-19-16-37(63)53-30(21-27-12-14-28(60)15-13-27)40(65)55-31(42(67)58-38)20-26-8-4-3-5-9-26/h3-5,8-9,12-15,25,29-34,38,60H,6-7,10-11,16-24H2,1-2H3,(H2,47,61)(H2,48,62)(H,52,64)(H,53,63)(H,54,68)(H,55,65)(H,56,69)(H,57,66)(H,58,67)(H4,49,50,51)/t29-,30-,31-,32-,33-,34-,38-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V2 receptor
(Rattus norvegicus (Rat)) | BDBM50205304
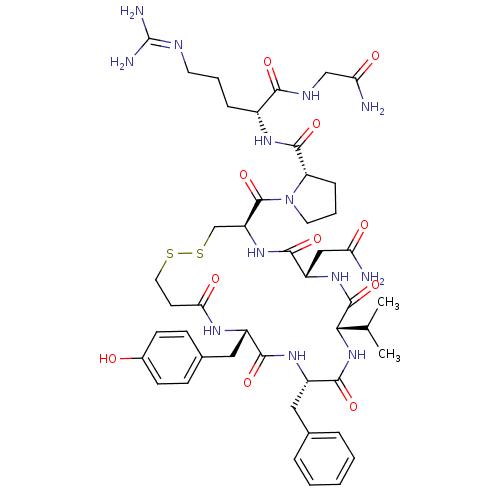
(CHEMBL435323 | dVDAVP)Show SMILES [#6]-[#6](-[#6])-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H65N13O11S2/c1-25(2)38-44(69)56-32(22-35(47)61)41(66)57-33(45(70)59-18-7-11-34(59)43(68)54-29(10-6-17-51-46(49)50)39(64)52-23-36(48)62)24-72-71-19-16-37(63)53-30(21-27-12-14-28(60)15-13-27)40(65)55-31(42(67)58-38)20-26-8-4-3-5-9-26/h3-5,8-9,12-15,25,29-34,38,60H,6-7,10-11,16-24H2,1-2H3,(H2,47,61)(H2,48,62)(H,52,64)(H,53,63)(H,54,68)(H,55,65)(H,56,69)(H,57,66)(H,58,67)(H4,49,50,51)/t29-,30+,31+,32+,33+,34+,38+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from vasopressin V2 receptor in rat kidney membranes |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205301
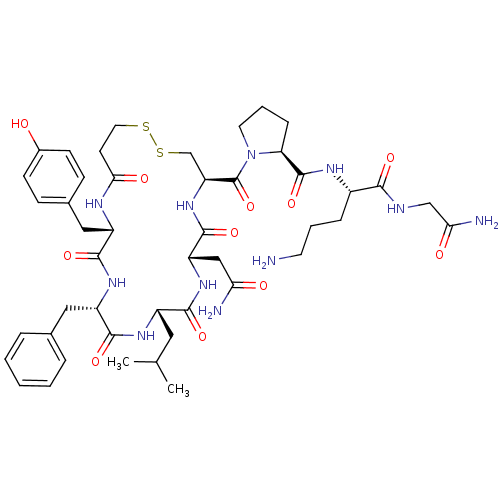
(CHEMBL375188 | d[Leu4,Orn8]VP)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCN)C(=O)NCC(N)=O Show InChI InChI=1S/C46H65N11O11S2/c1-26(2)20-31-41(63)55-34(23-37(48)59)44(66)56-35(46(68)57-18-7-11-36(57)45(67)52-30(10-6-17-47)40(62)50-24-38(49)60)25-70-69-19-16-39(61)51-32(22-28-12-14-29(58)15-13-28)42(64)54-33(43(65)53-31)21-27-8-4-3-5-9-27/h3-5,8-9,12-15,26,30-36,58H,6-7,10-11,16-25,47H2,1-2H3,(H2,48,59)(H2,49,60)(H,50,62)(H,51,61)(H,52,67)(H,53,65)(H,54,64)(H,55,63)(H,56,66)/t30-,31-,32-,33-,34-,35-,36-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50145118
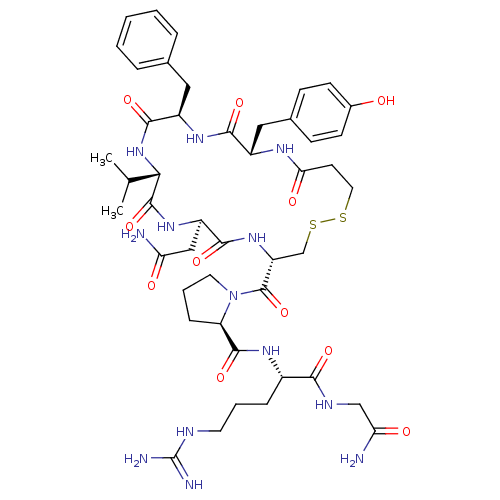
(CHEMBL412353 | d[Val4]AVP)Show SMILES CC(C)[C@@H]1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@@H](NC(=O)[C@@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O Show InChI InChI=1S/C46H65N13O11S2/c1-25(2)38-44(69)56-32(22-35(47)61)41(66)57-33(45(70)59-18-7-11-34(59)43(68)54-29(10-6-17-51-46(49)50)39(64)52-23-36(48)62)24-72-71-19-16-37(63)53-30(21-27-12-14-28(60)15-13-27)40(65)55-31(42(67)58-38)20-26-8-4-3-5-9-26/h3-5,8-9,12-15,25,29-34,38,60H,6-7,10-11,16-24H2,1-2H3,(H2,47,61)(H2,48,62)(H,52,64)(H,53,63)(H,54,68)(H,55,65)(H,56,69)(H,57,66)(H,58,67)(H4,49,50,51)/t29-,30-,31+,32+,33+,34+,38-/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Ohio
Curated by ChEMBL
| Assay Description
Binding affinity against human vasopressin V1b receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT recepto... |
J Med Chem 47: 2375-88 (2004)
Article DOI: 10.1021/jm030611c
BindingDB Entry DOI: 10.7270/Q2GF0V8C |
More data for this
Ligand-Target Pair | |
Vasopressin V2 receptor
(Rattus norvegicus (Rat)) | BDBM50205308
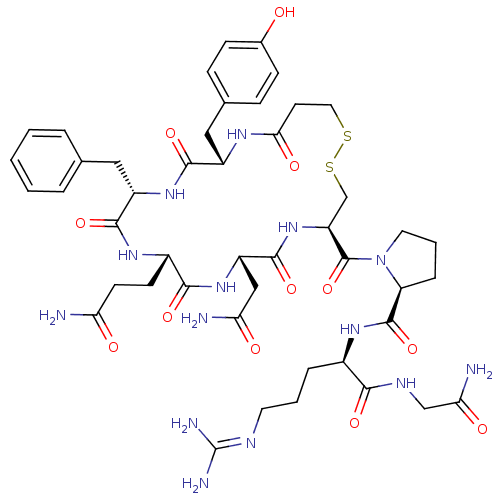
((S)-1-((4R,7S,10S,13S,16S)-7-(2-amino-2-oxoethyl)-...)Show SMILES [#7]-[#6](=O)-[#6]-[#6]-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H64N14O12S2/c47-35(62)15-14-29-40(67)58-32(22-36(48)63)43(70)59-33(45(72)60-18-5-9-34(60)44(71)56-28(8-4-17-52-46(50)51)39(66)53-23-37(49)64)24-74-73-19-16-38(65)54-30(21-26-10-12-27(61)13-11-26)41(68)57-31(42(69)55-29)20-25-6-2-1-3-7-25/h1-3,6-7,10-13,28-34,61H,4-5,8-9,14-24H2,(H2,47,62)(H2,48,63)(H2,49,64)(H,53,66)(H,54,65)(H,55,69)(H,56,71)(H,57,68)(H,58,67)(H,59,70)(H4,50,51,52)/t28-,29+,30+,31+,32+,33+,34+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from vasopressin V2 receptor in rat kidney membranes |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V2 receptor
(Rattus norvegicus (Rat)) | BDBM50205300

(CHEMBL221436 | d[Val4]AVP)Show SMILES [#6]-[#6](-[#6])-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H65N13O11S2/c1-25(2)38-44(69)56-32(22-35(47)61)41(66)57-33(45(70)59-18-7-11-34(59)43(68)54-29(10-6-17-51-46(49)50)39(64)52-23-36(48)62)24-72-71-19-16-37(63)53-30(21-27-12-14-28(60)15-13-27)40(65)55-31(42(67)58-38)20-26-8-4-3-5-9-26/h3-5,8-9,12-15,25,29-34,38,60H,6-7,10-11,16-24H2,1-2H3,(H2,47,61)(H2,48,62)(H,52,64)(H,53,63)(H,54,68)(H,55,65)(H,56,69)(H,57,66)(H,58,67)(H4,49,50,51)/t29-,30-,31-,32-,33-,34-,38-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from vasopressin V2 receptor in rat kidney membranes |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50038604

(3-N3-C6H4CH2CO-D-Tyr(Me)-Phe-Gln-Asn-Arg-Pro-Arg-T...)Show SMILES CN([C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O)C(=O)Cc1cccc(c1)N=[N+]=[N-] Show InChI InChI=1S/C62H82N20O13/c1-81(52(87)33-38-11-5-12-39(29-38)79-80-70)49(32-37-18-22-41(84)23-19-37)59(94)78-46(31-35-9-3-2-4-10-35)56(91)73-43(24-25-50(63)85)55(90)77-47(34-51(64)86)57(92)75-44(14-7-27-72-62(68)69)60(95)82-28-8-15-48(82)58(93)74-42(13-6-26-71-61(66)67)54(89)76-45(53(65)88)30-36-16-20-40(83)21-17-36/h2-5,9-12,16-23,29,42-49,83-84H,6-8,13-15,24-28,30-34H2,1H3,(H2,63,85)(H2,64,86)(H2,65,88)(H,73,91)(H,74,93)(H,75,92)(H,76,89)(H,77,90)(H,78,94)(H4,66,67,71)(H4,68,69,72)/t42-,43-,44-,45-,46-,47-,48-,49-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UPR 9023 CNRS
Curated by ChEMBL
| Assay Description
Inhibition constant for V1a receptor of rat liver membrane |
J Med Chem 37: 1841-9 (1994)
BindingDB Entry DOI: 10.7270/Q2R49PST |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50038604

(3-N3-C6H4CH2CO-D-Tyr(Me)-Phe-Gln-Asn-Arg-Pro-Arg-T...)Show SMILES CN([C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O)C(=O)Cc1cccc(c1)N=[N+]=[N-] Show InChI InChI=1S/C62H82N20O13/c1-81(52(87)33-38-11-5-12-39(29-38)79-80-70)49(32-37-18-22-41(84)23-19-37)59(94)78-46(31-35-9-3-2-4-10-35)56(91)73-43(24-25-50(63)85)55(90)77-47(34-51(64)86)57(92)75-44(14-7-27-72-62(68)69)60(95)82-28-8-15-48(82)58(93)74-42(13-6-26-71-61(66)67)54(89)76-45(53(65)88)30-36-16-20-40(83)21-17-36/h2-5,9-12,16-23,29,42-49,83-84H,6-8,13-15,24-28,30-34H2,1H3,(H2,63,85)(H2,64,86)(H2,65,88)(H,73,91)(H,74,93)(H,75,92)(H,76,89)(H,77,90)(H,78,94)(H4,66,67,71)(H4,68,69,72)/t42-,43-,44-,45-,46-,47-,48-,49-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UPR 9023 CNRS
Curated by ChEMBL
| Assay Description
Inhibition constant for V1a receptor of rat liver membrane |
J Med Chem 37: 1841-9 (1994)
BindingDB Entry DOI: 10.7270/Q2R49PST |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50145112
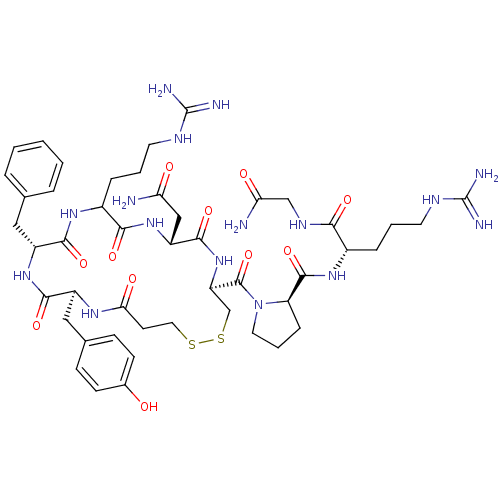
((2S)-2-{[(2R)-1-{[(4S,7R,13R,16S)-13-benzyl-7-(car...)Show SMILES NC(=O)CNC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H]1CCCN1C(=O)[C@H]1CSSCCC(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N[C@H](Cc2ccccc2)C(=O)NC(CCCNC(N)=N)C(=O)N[C@H](CC(N)=O)C(=O)N1 Show InChI InChI=1S/C47H68N16O11S2/c48-36(65)23-33-43(72)62-34(45(74)63-19-6-11-35(63)44(73)59-29(9-4-17-54-46(50)51)39(68)56-24-37(49)66)25-76-75-20-16-38(67)57-31(22-27-12-14-28(64)15-13-27)41(70)60-32(21-26-7-2-1-3-8-26)42(71)58-30(40(69)61-33)10-5-18-55-47(52)53/h1-3,7-8,12-15,29-35,64H,4-6,9-11,16-25H2,(H2,48,65)(H2,49,66)(H,56,68)(H,57,67)(H,58,71)(H,59,73)(H,60,70)(H,61,69)(H,62,72)(H4,50,51,54)(H4,52,53,55)/t29-,30?,31-,32+,33+,34+,35+/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Ohio
Curated by ChEMBL
| Assay Description
Binding affinity against human vasopressin V1b receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT recepto... |
J Med Chem 47: 2375-88 (2004)
Article DOI: 10.1021/jm030611c
BindingDB Entry DOI: 10.7270/Q2GF0V8C |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205303

(CHEMBL373968 | d[Leu4,Dap8]VP)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CN)C(=O)NCC(N)=O Show InChI InChI=1S/C44H61N11O11S2/c1-24(2)17-28-39(61)52-31(20-35(46)57)42(64)54-33(44(66)55-15-6-9-34(55)43(65)53-32(21-45)38(60)48-22-36(47)58)23-68-67-16-14-37(59)49-29(19-26-10-12-27(56)13-11-26)40(62)51-30(41(63)50-28)18-25-7-4-3-5-8-25/h3-5,7-8,10-13,24,28-34,56H,6,9,14-23,45H2,1-2H3,(H2,46,57)(H2,47,58)(H,48,60)(H,49,59)(H,50,63)(H,51,62)(H,52,61)(H,53,65)(H,54,64)/t28-,29-,30-,31-,32-,33-,34-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50370387

(CHEMBL1790575)Show SMILES [#6]-[#6]-[#6@H](-[#6])-[#6@@H]-1-[#7]-[#6](=O)-[#6@@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C47H67N13O11S2/c1-3-26(2)39-45(70)57-33(23-36(48)62)42(67)58-34(46(71)60-19-8-12-35(60)44(69)55-30(11-7-18-52-47(50)51)40(65)53-24-37(49)63)25-73-72-20-17-38(64)54-31(22-28-13-15-29(61)16-14-28)41(66)56-32(43(68)59-39)21-27-9-5-4-6-10-27/h4-6,9-10,13-16,26,30-35,39,61H,3,7-8,11-12,17-25H2,1-2H3,(H2,48,62)(H2,49,63)(H,53,65)(H,54,64)(H,55,69)(H,56,66)(H,57,70)(H,58,67)(H,59,68)(H4,50,51,52)/t26-,30-,31-,32+,33+,34+,35+,39-/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Ohio
Curated by ChEMBL
| Assay Description
Binding affinity against human vasopressin V1b receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT recepto... |
J Med Chem 47: 2375-88 (2004)
Article DOI: 10.1021/jm030611c
BindingDB Entry DOI: 10.7270/Q2GF0V8C |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50145124

((2S)-2-{[(2R)-1-{[(4S,7R,13R,16S)-13-benzyl-10-but...)Show SMILES CCCCC1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@@H](NC(=O)[C@@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O Show InChI InChI=1S/C47H67N13O11S2/c1-2-3-11-31-41(66)58-34(24-37(48)62)44(69)59-35(46(71)60-20-8-13-36(60)45(70)56-30(12-7-19-52-47(50)51)40(65)53-25-38(49)63)26-73-72-21-18-39(64)54-32(23-28-14-16-29(61)17-15-28)42(67)57-33(43(68)55-31)22-27-9-5-4-6-10-27/h4-6,9-10,14-17,30-36,61H,2-3,7-8,11-13,18-26H2,1H3,(H2,48,62)(H2,49,63)(H,53,65)(H,54,64)(H,55,68)(H,56,70)(H,57,67)(H,58,66)(H,59,69)(H4,50,51,52)/t30-,31?,32-,33+,34+,35+,36+/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Ohio
Curated by ChEMBL
| Assay Description
Binding affinity against human vasopressin V1b receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT recepto... |
J Med Chem 47: 2375-88 (2004)
Article DOI: 10.1021/jm030611c
BindingDB Entry DOI: 10.7270/Q2GF0V8C |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205311
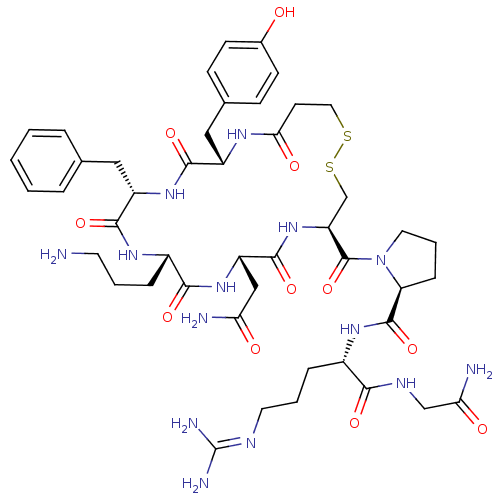
(CHEMBL375096 | d[Orn4]AVP)Show SMILES [#7]-[#6]-[#6]-[#6]-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H66N14O11S2/c47-17-4-9-30-40(66)58-33(23-36(48)62)43(69)59-34(45(71)60-19-6-11-35(60)44(70)56-29(10-5-18-52-46(50)51)39(65)53-24-37(49)63)25-73-72-20-16-38(64)54-31(22-27-12-14-28(61)15-13-27)41(67)57-32(42(68)55-30)21-26-7-2-1-3-8-26/h1-3,7-8,12-15,29-35,61H,4-6,9-11,16-25,47H2,(H2,48,62)(H2,49,63)(H,53,65)(H,54,64)(H,55,68)(H,56,70)(H,57,67)(H,58,66)(H,59,69)(H4,50,51,52)/t29-,30-,31-,32-,33-,34-,35-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205306
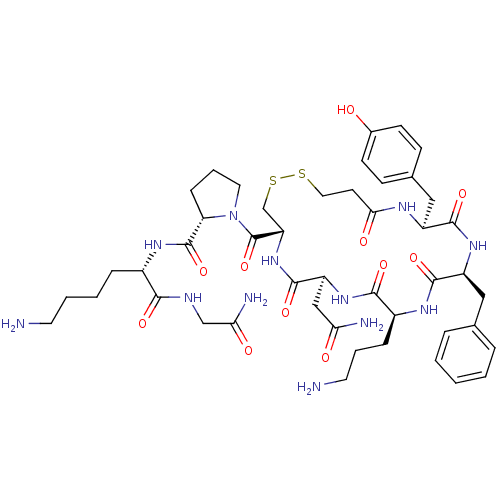
(CHEMBL375323 | d[Orn4,Lys8]VP)Show SMILES NCCCC[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1CSSCCC(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@@H](CCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N1)C(=O)NCC(N)=O Show InChI InChI=1S/C46H66N12O11S2/c47-18-5-4-10-30(40(63)51-25-38(50)61)54-45(68)36-12-7-20-58(36)46(69)35-26-71-70-21-17-39(62)52-32(23-28-13-15-29(59)16-14-28)42(65)55-33(22-27-8-2-1-3-9-27)43(66)53-31(11-6-19-48)41(64)56-34(24-37(49)60)44(67)57-35/h1-3,8-9,13-16,30-36,59H,4-7,10-12,17-26,47-48H2,(H2,49,60)(H2,50,61)(H,51,63)(H,52,62)(H,53,66)(H,54,68)(H,55,65)(H,56,64)(H,57,67)/t30-,31-,32-,33-,34-,35-,36-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50038599

(4-N3-C6H4CH2CH2CH2CO-D-Tyr(Me)-Phe-Gln-Asn-Arg-Pro...)Show SMILES CN([C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O)C(=O)CCCc1ccc(cc1)N=[N+]=[N-] Show InChI InChI=1S/C64H86N20O13/c1-83(54(89)15-5-11-37-16-22-41(23-17-37)81-82-72)51(35-40-20-26-43(86)27-21-40)61(96)80-48(34-38-9-3-2-4-10-38)58(93)75-45(28-29-52(65)87)57(92)79-49(36-53(66)88)59(94)77-46(13-7-31-74-64(70)71)62(97)84-32-8-14-50(84)60(95)76-44(12-6-30-73-63(68)69)56(91)78-47(55(67)90)33-39-18-24-42(85)25-19-39/h2-4,9-10,16-27,44-51,85-86H,5-8,11-15,28-36H2,1H3,(H2,65,87)(H2,66,88)(H2,67,90)(H,75,93)(H,76,95)(H,77,94)(H,78,91)(H,79,92)(H,80,96)(H4,68,69,73)(H4,70,71,74)/t44-,45-,46-,47-,48-,49-,50-,51-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UPR 9023 CNRS
Curated by ChEMBL
| Assay Description
Tested for inhibition constant determined from vasopressin induced inositol phosphates accumulation performed on WRK1 cell line of V1a receptor subty... |
J Med Chem 37: 1841-9 (1994)
BindingDB Entry DOI: 10.7270/Q2R49PST |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50038601

(4-N3-C6H4CH2CH2CO-D-Tyr(Me)-Phe-Gln-Asn-Arg-Pro-Ar...)Show SMILES CN([C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O)C(=O)CCc1ccc(cc1)N=[N+]=[N-] Show InChI InChI=1S/C63H84N20O13/c1-82(53(88)28-19-36-13-20-40(21-14-36)80-81-71)50(34-39-17-24-42(85)25-18-39)60(95)79-47(33-37-8-3-2-4-9-37)57(92)74-44(26-27-51(64)86)56(91)78-48(35-52(65)87)58(93)76-45(11-6-30-73-63(69)70)61(96)83-31-7-12-49(83)59(94)75-43(10-5-29-72-62(67)68)55(90)77-46(54(66)89)32-38-15-22-41(84)23-16-38/h2-4,8-9,13-18,20-25,43-50,84-85H,5-7,10-12,19,26-35H2,1H3,(H2,64,86)(H2,65,87)(H2,66,89)(H,74,92)(H,75,94)(H,76,93)(H,77,90)(H,78,91)(H,79,95)(H4,67,68,72)(H4,69,70,73)/t43-,44-,45-,46-,47-,48-,49-,50-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UPR 9023 CNRS
Curated by ChEMBL
| Assay Description
Inhibition constant for V1a receptor of rat liver membrane |
J Med Chem 37: 1841-9 (1994)
BindingDB Entry DOI: 10.7270/Q2R49PST |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50145107
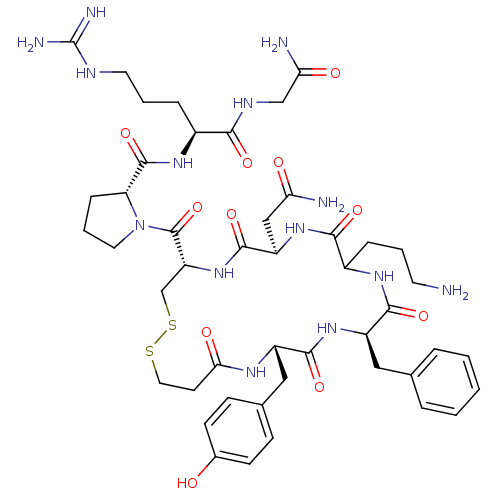
((2S)-2-{[(2R)-1-{[(4S,7R,13R,16S)-10-(3-aminopropy...)Show SMILES NCCCC1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@@H](NC(=O)[C@@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O Show InChI InChI=1S/C46H66N14O11S2/c47-17-4-9-30-40(66)58-33(23-36(48)62)43(69)59-34(45(71)60-19-6-11-35(60)44(70)56-29(10-5-18-52-46(50)51)39(65)53-24-37(49)63)25-73-72-20-16-38(64)54-31(22-27-12-14-28(61)15-13-27)41(67)57-32(42(68)55-30)21-26-7-2-1-3-8-26/h1-3,7-8,12-15,29-35,61H,4-6,9-11,16-25,47H2,(H2,48,62)(H2,49,63)(H,53,65)(H,54,64)(H,55,68)(H,56,70)(H,57,67)(H,58,66)(H,59,69)(H4,50,51,52)/t29-,30?,31-,32+,33+,34+,35+/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Ohio
Curated by ChEMBL
| Assay Description
Binding affinity against human vasopressin V1b receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT recepto... |
J Med Chem 47: 2375-88 (2004)
Article DOI: 10.1021/jm030611c
BindingDB Entry DOI: 10.7270/Q2GF0V8C |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50342028
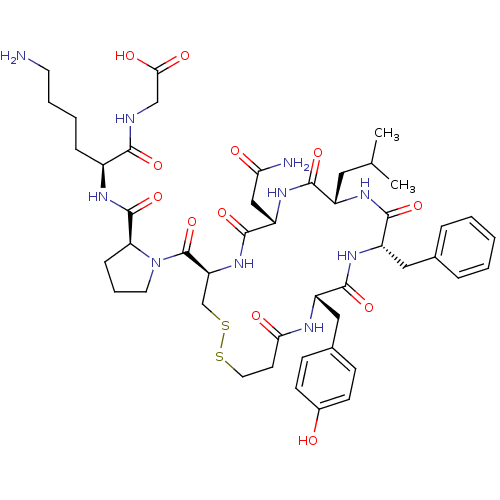
(2-((S)-6-amino-2-((S)-1-((4R,7S,10S,13S,16S)-7-(2-...)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCN)C(=O)NCC(O)=O |r| Show InChI InChI=1S/C47H66N10O12S2/c1-27(2)21-32-42(64)55-35(24-38(49)59)45(67)56-36(47(69)57-19-8-12-37(57)46(68)52-31(11-6-7-18-48)41(63)50-25-40(61)62)26-71-70-20-17-39(60)51-33(23-29-13-15-30(58)16-14-29)43(65)54-34(44(66)53-32)22-28-9-4-3-5-10-28/h3-5,9-10,13-16,27,31-37,58H,6-8,11-12,17-26,48H2,1-2H3,(H2,49,59)(H,50,63)(H,51,60)(H,52,68)(H,53,66)(H,54,65)(H,55,64)(H,56,67)(H,61,62)/t31-,32-,33-,34-,35-,36-,37-/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from human vasopressin V1b receptor expressed CHO cells after 60 mins |
J Med Chem 54: 2864-77 (2011)
Article DOI: 10.1021/jm1016208
BindingDB Entry DOI: 10.7270/Q23B60GJ |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50145117

((2S)-2-{[(2R)-1-{[(4S,7R,13R,16S)-13-benzyl-7-(car...)Show SMILES NC(=O)CNC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H]1CCCN1C(=O)[C@H]1CSSCCC(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N[C@H](Cc2ccccc2)C(=O)NC(CCCCNC(N)=N)C(=O)N[C@H](CC(N)=O)C(=O)N1 Show InChI InChI=1S/C48H70N16O11S2/c49-37(66)24-34-44(73)63-35(46(75)64-20-7-12-36(64)45(74)60-30(11-6-19-56-48(53)54)40(69)57-25-38(50)67)26-77-76-21-17-39(68)58-32(23-28-13-15-29(65)16-14-28)42(71)61-33(22-27-8-2-1-3-9-27)43(72)59-31(41(70)62-34)10-4-5-18-55-47(51)52/h1-3,8-9,13-16,30-36,65H,4-7,10-12,17-26H2,(H2,49,66)(H2,50,67)(H,57,69)(H,58,68)(H,59,72)(H,60,74)(H,61,71)(H,62,70)(H,63,73)(H4,51,52,55)(H4,53,54,56)/t30-,31?,32-,33+,34+,35+,36+/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Ohio
Curated by ChEMBL
| Assay Description
Binding affinity against human vasopressin V1b receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT recepto... |
J Med Chem 47: 2375-88 (2004)
Article DOI: 10.1021/jm030611c
BindingDB Entry DOI: 10.7270/Q2GF0V8C |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205292

(CHEMBL385068 | d[Arg4,Orn8]VP)Show SMILES NCCC[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1CSSCCC(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(N)=O)C(=O)N1)C(=O)NCC(N)=O Show InChI InChI=1S/C46H66N14O11S2/c47-17-4-9-29(39(65)53-24-37(49)63)56-44(70)35-11-6-19-60(35)45(71)34-25-73-72-20-16-38(64)54-31(22-27-12-14-28(61)15-13-27)41(67)57-32(21-26-7-2-1-3-8-26)42(68)55-30(10-5-18-52-46(50)51)40(66)58-33(23-36(48)62)43(69)59-34/h1-3,7-8,12-15,29-35,61H,4-6,9-11,16-25,47H2,(H2,48,62)(H2,49,63)(H,53,65)(H,54,64)(H,55,68)(H,56,70)(H,57,67)(H,58,66)(H,59,69)(H4,50,51,52)/t29-,30-,31-,32-,33-,34-,35-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205298
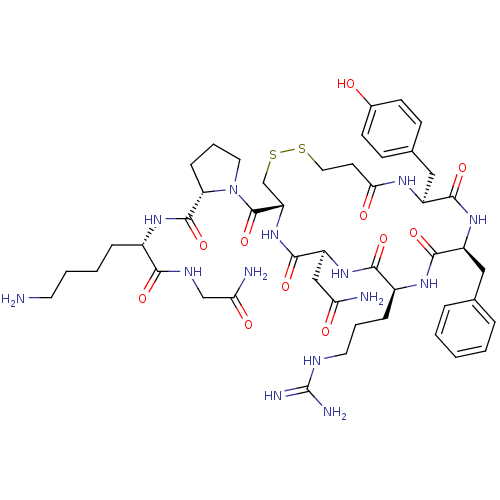
(CHEMBL375325 | d[Arg4,Lys8]VP)Show SMILES NCCCC[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1CSSCCC(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(N)=O)C(=O)N1)C(=O)NCC(N)=O Show InChI InChI=1S/C47H68N14O11S2/c48-18-5-4-10-30(40(66)54-25-38(50)64)57-45(71)36-12-7-20-61(36)46(72)35-26-74-73-21-17-39(65)55-32(23-28-13-15-29(62)16-14-28)42(68)58-33(22-27-8-2-1-3-9-27)43(69)56-31(11-6-19-53-47(51)52)41(67)59-34(24-37(49)63)44(70)60-35/h1-3,8-9,13-16,30-36,62H,4-7,10-12,17-26,48H2,(H2,49,63)(H2,50,64)(H,54,66)(H,55,65)(H,56,69)(H,57,71)(H,58,68)(H,59,67)(H,60,70)(H4,51,52,53)/t30-,31-,32-,33-,34-,35-,36-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50342032

(2-((S)-2-((S)-1-((4R,7S,10S,13S,16S)-7-(2-amino-2-...)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCNC(=O)CCCCCCN)C(=O)NCC(O)=O |r| Show InChI InChI=1S/C54H79N11O13S2/c1-33(2)27-38-49(73)63-41(30-44(56)67)52(76)64-42(54(78)65-25-12-16-43(65)53(77)60-37(48(72)58-31-47(70)71)15-9-11-24-57-45(68)17-8-3-4-10-23-55)32-80-79-26-22-46(69)59-39(29-35-18-20-36(66)21-19-35)50(74)62-40(51(75)61-38)28-34-13-6-5-7-14-34/h5-7,13-14,18-21,33,37-43,66H,3-4,8-12,15-17,22-32,55H2,1-2H3,(H2,56,67)(H,57,68)(H,58,72)(H,59,69)(H,60,77)(H,61,75)(H,62,74)(H,63,73)(H,64,76)(H,70,71)/t37-,38-,39-,40-,41-,42-,43-/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.610 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from human vasopressin V1b receptor expressed CHO cells after 60 mins |
J Med Chem 54: 2864-77 (2011)
Article DOI: 10.1021/jm1016208
BindingDB Entry DOI: 10.7270/Q23B60GJ |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50342029
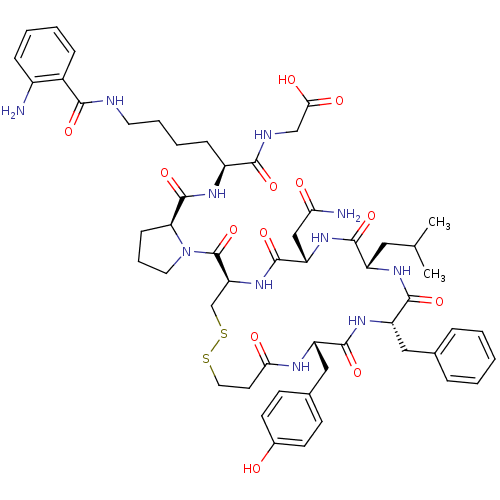
(2-((S)-2-((S)-1-((4R,7S,10S,13S,16S)-7-(2-amino-2-...)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCNC(=O)c1ccccc1N)C(=O)NCC(O)=O |r| Show InChI InChI=1S/C54H71N11O13S2/c1-31(2)25-38-49(73)63-41(28-44(56)67)52(76)64-42(30-80-79-24-21-45(68)59-39(27-33-17-19-34(66)20-18-33)50(74)62-40(51(75)61-38)26-32-11-4-3-5-12-32)54(78)65-23-10-16-43(65)53(77)60-37(48(72)58-29-46(69)70)15-8-9-22-57-47(71)35-13-6-7-14-36(35)55/h3-7,11-14,17-20,31,37-43,66H,8-10,15-16,21-30,55H2,1-2H3,(H2,56,67)(H,57,71)(H,58,72)(H,59,68)(H,60,77)(H,61,75)(H,62,74)(H,63,73)(H,64,76)(H,69,70)/t37-,38-,39-,40-,41-,42-,43-/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from human vasopressin V1b receptor expressed CHO cells after 60 mins |
J Med Chem 54: 2864-77 (2011)
Article DOI: 10.1021/jm1016208
BindingDB Entry DOI: 10.7270/Q23B60GJ |
More data for this
Ligand-Target Pair | |
Oxytocin receptor
(RAT) | BDBM50038604

(3-N3-C6H4CH2CO-D-Tyr(Me)-Phe-Gln-Asn-Arg-Pro-Arg-T...)Show SMILES CN([C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1ccc(O)cc1)C(N)=O)C(=O)Cc1cccc(c1)N=[N+]=[N-] Show InChI InChI=1S/C62H82N20O13/c1-81(52(87)33-38-11-5-12-39(29-38)79-80-70)49(32-37-18-22-41(84)23-19-37)59(94)78-46(31-35-9-3-2-4-10-35)56(91)73-43(24-25-50(63)85)55(90)77-47(34-51(64)86)57(92)75-44(14-7-27-72-62(68)69)60(95)82-28-8-15-48(82)58(93)74-42(13-6-26-71-61(66)67)54(89)76-45(53(65)88)30-36-16-20-40(83)21-17-36/h2-5,9-12,16-23,29,42-49,83-84H,6-8,13-15,24-28,30-34H2,1H3,(H2,63,85)(H2,64,86)(H2,65,88)(H,73,91)(H,74,93)(H,75,92)(H,76,89)(H,77,90)(H,78,94)(H4,66,67,71)(H4,68,69,72)/t42-,43-,44-,45-,46-,47-,48-,49-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UPR 9023 CNRS
Curated by ChEMBL
| Assay Description
Tested for inhibition constant at OT receptor of rat mamary glands |
J Med Chem 37: 1841-9 (1994)
BindingDB Entry DOI: 10.7270/Q2R49PST |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50145108
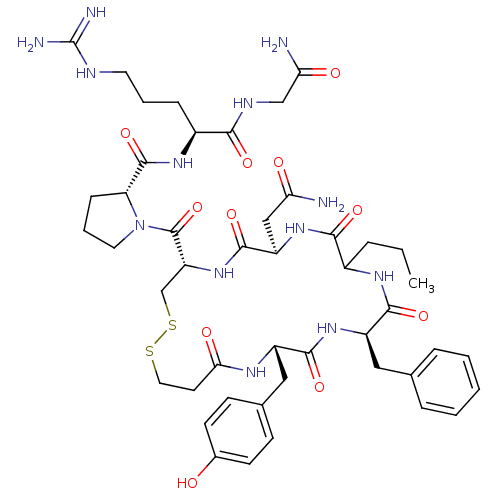
((2S)-2-{[(2R)-1-{[(4S,7R,13R,16S)-13-benzyl-7-(car...)Show SMILES CCCC1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@@H](NC(=O)[C@@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O Show InChI InChI=1S/C46H65N13O11S2/c1-2-8-29-40(65)57-33(23-36(47)61)43(68)58-34(45(70)59-19-7-12-35(59)44(69)55-30(11-6-18-51-46(49)50)39(64)52-24-37(48)62)25-72-71-20-17-38(63)53-31(22-27-13-15-28(60)16-14-27)41(66)56-32(42(67)54-29)21-26-9-4-3-5-10-26/h3-5,9-10,13-16,29-35,60H,2,6-8,11-12,17-25H2,1H3,(H2,47,61)(H2,48,62)(H,52,64)(H,53,63)(H,54,67)(H,55,69)(H,56,66)(H,57,65)(H,58,68)(H4,49,50,51)/t29?,30-,31-,32+,33+,34+,35+/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Ohio
Curated by ChEMBL
| Assay Description
Binding affinity against human vasopressin V1b receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT recepto... |
J Med Chem 47: 2375-88 (2004)
Article DOI: 10.1021/jm030611c
BindingDB Entry DOI: 10.7270/Q2GF0V8C |
More data for this
Ligand-Target Pair | |
Vasopressin V2 receptor
(Rattus norvegicus (Rat)) | BDBM50205291

((S)-1-((4R,7S,10S,13S,16S)-7-(2-amino-2-oxoethyl)-...)Show SMILES [#7]-[#6](=O)-[#6]-[#6]-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H64N14O12S2/c47-35(62)15-14-29-40(67)58-32(22-36(48)63)43(70)59-33(45(72)60-18-5-9-34(60)44(71)56-28(8-4-17-52-46(50)51)39(66)53-23-37(49)64)24-74-73-19-16-38(65)54-30(21-26-10-12-27(61)13-11-26)41(68)57-31(42(69)55-29)20-25-6-2-1-3-7-25/h1-3,6-7,10-13,28-34,61H,4-5,8-9,14-24H2,(H2,47,62)(H2,48,63)(H2,49,64)(H,53,66)(H,54,65)(H,55,69)(H,56,71)(H,57,68)(H,58,67)(H,59,70)(H4,50,51,52)/t28-,29-,30-,31-,32-,33-,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from vasopressin V2 receptor in rat kidney membranes |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205307

(CHEMBL412742 | d[Cha4,Dab8]VP)Show SMILES NCC[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1CSSCCC(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@@H](CC2CCCCC2)C(=O)N[C@@H](CC(N)=O)C(=O)N1)C(=O)NCC(N)=O Show InChI InChI=1S/C48H67N11O11S2/c49-19-17-32(42(64)52-26-40(51)62)54-47(69)38-12-7-20-59(38)48(70)37-27-72-71-21-18-41(63)53-33(24-30-13-15-31(60)16-14-30)43(65)55-34(22-28-8-3-1-4-9-28)44(66)56-35(23-29-10-5-2-6-11-29)45(67)57-36(25-39(50)61)46(68)58-37/h1,3-4,8-9,13-16,29,32-38,60H,2,5-7,10-12,17-27,49H2,(H2,50,61)(H2,51,62)(H,52,64)(H,53,63)(H,54,69)(H,55,65)(H,56,66)(H,57,67)(H,58,68)/t32-,33-,34-,35-,36-,37-,38-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(RAT) | BDBM50205310

(CHEMBL219272 | d[Orn4,Orn8]VP)Show SMILES NCCC[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H]1CSSCCC(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@@H](CCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N1)C(=O)NCC(N)=O Show InChI InChI=1S/C45H64N12O11S2/c46-17-4-9-29(39(62)50-24-37(49)60)53-44(67)35-11-6-19-57(35)45(68)34-25-70-69-20-16-38(61)51-31(22-27-12-14-28(58)15-13-27)41(64)54-32(21-26-7-2-1-3-8-26)42(65)52-30(10-5-18-47)40(63)55-33(23-36(48)59)43(66)56-34/h1-3,7-8,12-15,29-35,58H,4-6,9-11,16-25,46-47H2,(H2,48,59)(H2,49,60)(H,50,62)(H,51,61)(H,52,65)(H,53,67)(H,54,64)(H,55,63)(H,56,66)/t29-,30-,31-,32-,33-,34-,35-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.910 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Oxytocin receptor
(RAT) | BDBM50205291

((S)-1-((4R,7S,10S,13S,16S)-7-(2-amino-2-oxoethyl)-...)Show SMILES [#7]-[#6](=O)-[#6]-[#6]-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H64N14O12S2/c47-35(62)15-14-29-40(67)58-32(22-36(48)63)43(70)59-33(45(72)60-18-5-9-34(60)44(71)56-28(8-4-17-52-46(50)51)39(66)53-23-37(49)64)24-74-73-19-16-38(65)54-30(21-26-10-12-27(61)13-11-26)41(68)57-31(42(69)55-29)20-25-6-2-1-3-7-25/h1-3,6-7,10-13,28-34,61H,4-5,8-9,14-24H2,(H2,47,62)(H2,48,63)(H2,49,64)(H,53,66)(H,54,65)(H,55,69)(H,56,71)(H,57,68)(H,58,67)(H,59,70)(H4,50,51,52)/t28-,29-,30-,31-,32-,33-,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.970 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat OT receptor expressed in CHO cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50145111
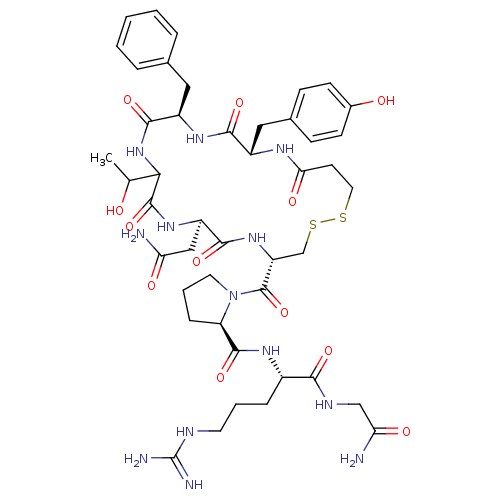
((2S)-2-{[(2R)-1-{[(4S,7R,13R,16S)-13-benzyl-7-(car...)Show SMILES CC(O)C1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@@H](NC(=O)[C@@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O Show InChI InChI=1S/C45H63N13O12S2/c1-24(59)37-43(69)55-31(21-34(46)61)40(66)56-32(44(70)58-17-6-10-33(58)42(68)53-28(9-5-16-50-45(48)49)38(64)51-22-35(47)62)23-72-71-18-15-36(63)52-29(20-26-11-13-27(60)14-12-26)39(65)54-30(41(67)57-37)19-25-7-3-2-4-8-25/h2-4,7-8,11-14,24,28-33,37,59-60H,5-6,9-10,15-23H2,1H3,(H2,46,61)(H2,47,62)(H,51,64)(H,52,63)(H,53,68)(H,54,65)(H,55,69)(H,56,66)(H,57,67)(H4,48,49,50)/t24?,28-,29-,30+,31+,32+,33+,37?/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Ohio
Curated by ChEMBL
| Assay Description
Binding affinity against human vasopressin V1b receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT recepto... |
J Med Chem 47: 2375-88 (2004)
Article DOI: 10.1021/jm030611c
BindingDB Entry DOI: 10.7270/Q2GF0V8C |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50145109
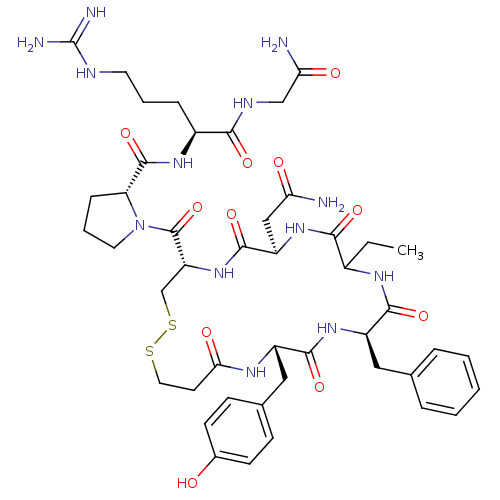
((2S)-2-{[(2R)-1-{[(4S,7R,13R,16S)-13-benzyl-7-(car...)Show SMILES CCC1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@@H](NC(=O)[C@@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O Show InChI InChI=1S/C45H63N13O11S2/c1-2-28-39(64)56-32(22-35(46)60)42(67)57-33(44(69)58-18-7-11-34(58)43(68)54-29(10-6-17-50-45(48)49)38(63)51-23-36(47)61)24-71-70-19-16-37(62)52-30(21-26-12-14-27(59)15-13-26)40(65)55-31(41(66)53-28)20-25-8-4-3-5-9-25/h3-5,8-9,12-15,28-34,59H,2,6-7,10-11,16-24H2,1H3,(H2,46,60)(H2,47,61)(H,51,63)(H,52,62)(H,53,66)(H,54,68)(H,55,65)(H,56,64)(H,57,67)(H4,48,49,50)/t28?,29-,30-,31+,32+,33+,34+/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Ohio
Curated by ChEMBL
| Assay Description
Binding affinity against human vasopressin V1b receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT recepto... |
J Med Chem 47: 2375-88 (2004)
Article DOI: 10.1021/jm030611c
BindingDB Entry DOI: 10.7270/Q2GF0V8C |
More data for this
Ligand-Target Pair | |
Oxytocin receptor
(RAT) | BDBM35667
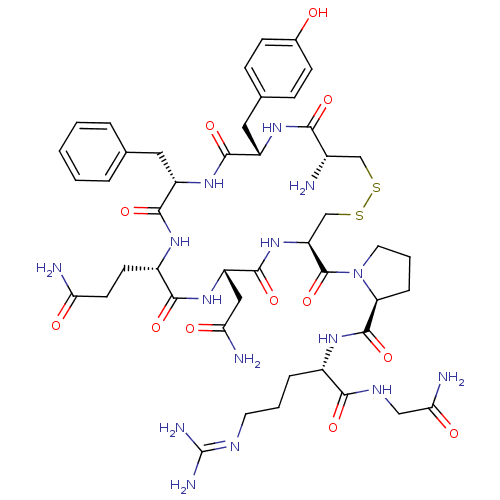
(AVP | CHEMBL373742 | US10131692, 44 (AVP) | [3H]Ar...)Show SMILES [#7]-[#6@H]-1-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C46H65N15O12S2/c47-27-22-74-75-23-33(45(73)61-17-5-9-34(61)44(72)56-28(8-4-16-53-46(51)52)39(67)54-21-37(50)65)60-43(71)32(20-36(49)64)59-40(68)29(14-15-35(48)63)55-41(69)31(18-24-6-2-1-3-7-24)58-42(70)30(57-38(27)66)19-25-10-12-26(62)13-11-25/h1-3,6-7,10-13,27-34,62H,4-5,8-9,14-23,47H2,(H2,48,63)(H2,49,64)(H2,50,65)(H,54,67)(H,55,69)(H,56,72)(H,57,66)(H,58,70)(H,59,68)(H,60,71)(H4,51,52,53)/t27-,28-,29-,30-,31-,32-,33-,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat OT receptor expressed in CHO cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
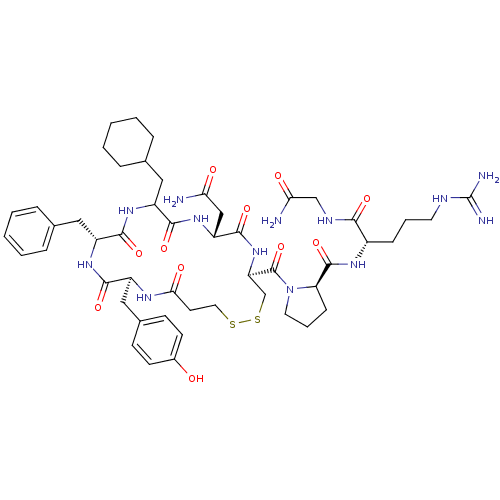
(Homo sapiens (Human)) | BDBM50145123
(CHEMBL410573 | d[Cha4]AVP)Show SMILES NC(=O)CNC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H]1CCCN1C(=O)[C@H]1CSSCCC(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N[C@H](Cc2ccccc2)C(=O)NC(CC2CCCCC2)C(=O)N[C@H](CC(N)=O)C(=O)N1 Show InChI InChI=1S/C50H71N13O11S2/c51-40(65)26-37-47(72)62-38(49(74)63-21-8-14-39(63)48(73)58-33(13-7-20-55-50(53)54)43(68)56-27-41(52)66)28-76-75-22-19-42(67)57-34(25-31-15-17-32(64)18-16-31)44(69)59-35(23-29-9-3-1-4-10-29)45(70)60-36(46(71)61-37)24-30-11-5-2-6-12-30/h1,3-4,9-10,15-18,30,33-39,64H,2,5-8,11-14,19-28H2,(H2,51,65)(H2,52,66)(H,56,68)(H,57,67)(H,58,73)(H,59,69)(H,60,70)(H,61,71)(H,62,72)(H4,53,54,55)/t33-,34-,35+,36?,37+,38+,39+/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Ohio
Curated by ChEMBL
| Assay Description
Binding affinity against human vasopressin V1b receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT recepto... |
J Med Chem 47: 2375-88 (2004)
Article DOI: 10.1021/jm030611c
BindingDB Entry DOI: 10.7270/Q2GF0V8C |
More data for this
Ligand-Target Pair | |
Vasopressin V2 receptor
(Homo sapiens (Human)) | BDBM50145118

(CHEMBL412353 | d[Val4]AVP)Show SMILES CC(C)[C@@H]1NC(=O)[C@@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@@H](NC(=O)[C@@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O Show InChI InChI=1S/C46H65N13O11S2/c1-25(2)38-44(69)56-32(22-35(47)61)41(66)57-33(45(70)59-18-7-11-34(59)43(68)54-29(10-6-17-51-46(49)50)39(64)52-23-36(48)62)24-72-71-19-16-37(63)53-30(21-27-12-14-28(60)15-13-27)40(65)55-31(42(67)58-38)20-26-8-4-3-5-9-26/h3-5,8-9,12-15,25,29-34,38,60H,6-7,10-11,16-24H2,1-2H3,(H2,47,61)(H2,48,62)(H,52,64)(H,53,63)(H,54,68)(H,55,65)(H,56,69)(H,57,66)(H,58,67)(H4,49,50,51)/t29-,30-,31+,32+,33+,34+,38-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medical College of Ohio
Curated by ChEMBL
| Assay Description
Binding affinity against human vasopressin V2 receptor was determined by using plasma membranes from CHO cells stably transfected with VP/OT receptor... |
J Med Chem 47: 2375-88 (2004)
Article DOI: 10.1021/jm030611c
BindingDB Entry DOI: 10.7270/Q2GF0V8C |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50342030
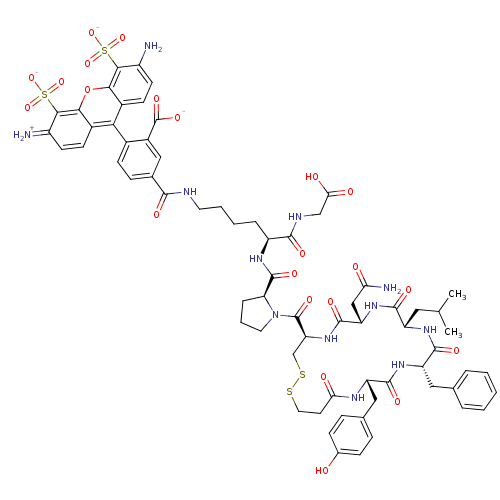
(CHEMBL1765665 | triethylamine hemi(2-((S)-2-((S)-1...)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCNC(=O)c1ccc(c(c1)C([O-])=O)-c1c2ccc(N)c(c2oc2c(c(=[NH2+])ccc12)S([O-])(=O)=O)S([O-])(=O)=O)C(=O)NCC(O)=O |r,wU:19.20,4.3,54.58,wD:8.8,40.42,58.61,36.50,(11.79,6.5,;11.79,4.96,;13.13,4.18,;10.45,4.18,;10.45,2.64,;9.11,1.87,;7.78,2.64,;7.78,4.18,;6.44,1.87,;6.44,.33,;7.78,-.45,;9.11,.33,;10.45,-.45,;10.45,-1.99,;9.11,-2.77,;7.78,-1.99,;5.1,2.64,;3.76,1.87,;3.76,.33,;2.42,2.64,;2.42,4.18,;3.76,4.96,;5.1,4.18,;6.44,4.96,;6.44,6.5,;7.78,7.28,;5.1,7.28,;3.76,6.5,;1.09,1.87,;-.25,1.1,;-1.58,1.87,;-.25,-.44,;-1.58,-2.38,;9.79,-6.18,;19.65,-1.87,;18.5,-.42,;18.48,1.12,;17.14,1.87,;15.8,2.64,;15.8,4.18,;14.47,1.87,;14.39,.33,;15.68,-.52,;17.06,.19,;15.6,-2.05,;13.13,2.64,;11.79,1.87,;11.79,.33,;19.8,1.9,;19.79,3.44,;21.15,1.15,;21.61,2.74,;23.28,2.8,;23.84,1.25,;22.49,.51,;22.54,-1.01,;21.2,-1.73,;23.84,-1.8,;23.8,-3.33,;22.46,-4.06,;22.41,-5.58,;21.07,-6.31,;21.03,-7.83,;19.69,-8.56,;18.36,-7.77,;18.38,-6.23,;17.02,-8.53,;16.99,-10.07,;15.65,-10.82,;14.33,-10.02,;14.35,-8.49,;15.68,-7.74,;13.59,-7.15,;14.38,-5.83,;12.05,-7.14,;12.98,-10.78,;12.96,-12.31,;14.29,-13.1,;14.26,-14.64,;12.92,-15.39,;12.9,-16.93,;11.59,-14.6,;11.61,-13.07,;10.28,-12.28,;10.31,-10.73,;8.99,-9.95,;9.01,-8.42,;7.68,-7.63,;10.35,-7.66,;11.67,-8.45,;11.65,-9.98,;7.65,-10.71,;6.32,-9.92,;6.86,-12.03,;8.4,-12.05,;10.25,-15.36,;8.93,-14.58,;9.47,-16.69,;11.01,-16.7,;25.09,-4.13,;26.44,-3.4,;25.05,-5.65,;26.35,-6.45,;26.31,-7.97,;27.61,-8.77,;24.97,-8.7,)| Show InChI InChI=1S/C68H78N12O22S4/c1-34(2)27-46-62(88)78-49(31-52(71)82)65(91)79-50(33-104-103-26-23-53(83)74-47(29-36-13-16-38(81)17-14-36)63(89)77-48(64(90)76-46)28-35-9-4-3-5-10-35)67(93)80-25-8-12-51(80)66(92)75-45(61(87)73-32-54(84)85)11-6-7-24-72-60(86)37-15-18-39(42(30-37)68(94)95)55-40-19-21-43(69)58(105(96,97)98)56(40)102-57-41(55)20-22-44(70)59(57)106(99,100)101/h3-5,9-10,13-22,30,34,45-51,69,81H,6-8,11-12,23-29,31-33,70H2,1-2H3,(H2,71,82)(H,72,86)(H,73,87)(H,74,83)(H,75,92)(H,76,90)(H,77,89)(H,78,88)(H,79,91)(H,84,85)(H,94,95)(H,96,97,98)(H,99,100,101)/p-2/t45-,46-,47-,48-,49-,50-,51-/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from human vasopressin V1b receptor expressed CHO cells after 60 mins |
J Med Chem 54: 2864-77 (2011)
Article DOI: 10.1021/jm1016208
BindingDB Entry DOI: 10.7270/Q23B60GJ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

















































