 Found 1010 hits with Last Name = 'hollis' and Initial = 'g'
Found 1010 hits with Last Name = 'hollis' and Initial = 'g' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
5-hydroxytryptamine receptor 2B
(Homo sapiens (Human)) | BDBM85530
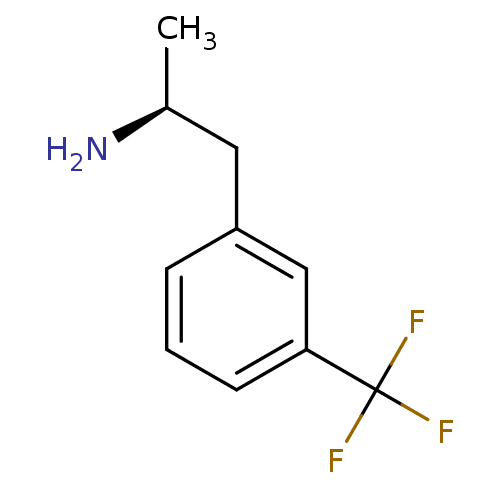
(Nor-d-fenfluramine | Nor-dexfenfluramine | Norfenf...)Show InChI InChI=1S/C10H12F3N/c1-7(14)5-8-3-2-4-9(6-8)10(11,12)13/h2-4,6-7H,5,14H2,1H3/t7-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceuticals Research Laboratories
Curated by PDSP Ki Database
| |
Mol Pharmacol 57: 75-81 (2000)
BindingDB Entry DOI: 10.7270/Q2P26WPH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2C
(Homo sapiens (Human)) | BDBM85530

(Nor-d-fenfluramine | Nor-dexfenfluramine | Norfenf...)Show InChI InChI=1S/C10H12F3N/c1-7(14)5-8-3-2-4-9(6-8)10(11,12)13/h2-4,6-7H,5,14H2,1H3/t7-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 56 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceuticals Research Laboratories
Curated by PDSP Ki Database
| |
Mol Pharmacol 57: 75-81 (2000)
BindingDB Entry DOI: 10.7270/Q2P26WPH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2B
(Homo sapiens (Human)) | BDBM85598
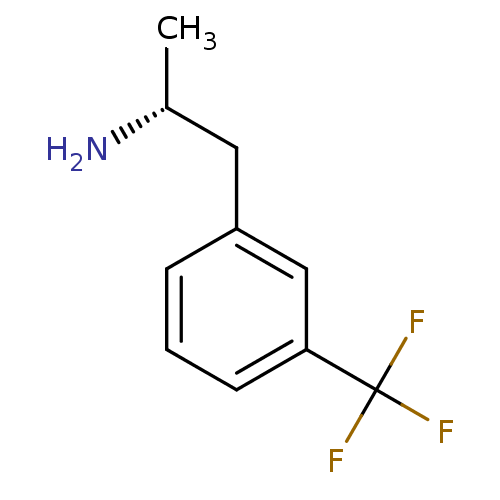
(l-norfenfluramine)Show InChI InChI=1S/C10H12F3N/c1-7(14)5-8-3-2-4-9(6-8)10(11,12)13/h2-4,6-7H,5,14H2,1H3/t7-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| 65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceuticals Research Laboratories
Curated by PDSP Ki Database
| |
Mol Pharmacol 57: 75-81 (2000)
BindingDB Entry DOI: 10.7270/Q2P26WPH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2C
(Homo sapiens (Human)) | BDBM85598

(l-norfenfluramine)Show InChI InChI=1S/C10H12F3N/c1-7(14)5-8-3-2-4-9(6-8)10(11,12)13/h2-4,6-7H,5,14H2,1H3/t7-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| 99 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceuticals Research Laboratories
Curated by PDSP Ki Database
| |
Mol Pharmacol 57: 75-81 (2000)
BindingDB Entry DOI: 10.7270/Q2P26WPH |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM13465

((2S)-N-[(1S)-1-carbamoyl-2-{4-[(5S)-1,1,3-trioxo-1...)Show SMILES CC(=O)N[C@@H](Cc1ccccc1)C(=O)N[C@@H](Cc1ccc(cc1)[C@@H]1CC(=O)NS1(=O)=O)C(N)=O |r| Show InChI InChI=1S/C23H26N4O6S/c1-14(28)25-19(12-15-5-3-2-4-6-15)23(31)26-18(22(24)30)11-16-7-9-17(10-8-16)20-13-21(29)27-34(20,32)33/h2-10,18-20H,11-13H2,1H3,(H2,24,30)(H,25,28)(H,26,31)(H,27,29)/t18-,19-,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Incyte Corporation
Curated by ChEMBL
| Assay Description
Inhibition constant against protein-tyrosine phosphatase 1B by PNPP enzyme assay |
J Med Chem 48: 6544-8 (2005)
Article DOI: 10.1021/jm0504555
BindingDB Entry DOI: 10.7270/Q2805252 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM85530

(Nor-d-fenfluramine | Nor-dexfenfluramine | Norfenf...)Show InChI InChI=1S/C10H12F3N/c1-7(14)5-8-3-2-4-9(6-8)10(11,12)13/h2-4,6-7H,5,14H2,1H3/t7-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 187 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceuticals Research Laboratories
Curated by PDSP Ki Database
| |
Mol Pharmacol 57: 75-81 (2000)
BindingDB Entry DOI: 10.7270/Q2P26WPH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM85598

(l-norfenfluramine)Show InChI InChI=1S/C10H12F3N/c1-7(14)5-8-3-2-4-9(6-8)10(11,12)13/h2-4,6-7H,5,14H2,1H3/t7-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| 267 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceuticals Research Laboratories
Curated by PDSP Ki Database
| |
Mol Pharmacol 57: 75-81 (2000)
BindingDB Entry DOI: 10.7270/Q2P26WPH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2B
(Homo sapiens (Human)) | BDBM85597
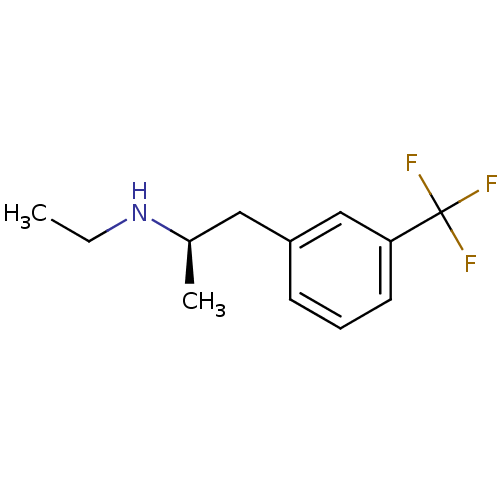
(CAS_37577-24-5 | NSC_65801 | l-Fenfluramine)Show InChI InChI=1S/C12H16F3N/c1-3-16-9(2)7-10-5-4-6-11(8-10)12(13,14)15/h4-6,8-9,16H,3,7H2,1-2H3/t9-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceuticals Research Laboratories
Curated by PDSP Ki Database
| |
Mol Pharmacol 57: 75-81 (2000)
BindingDB Entry DOI: 10.7270/Q2P26WPH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM85597

(CAS_37577-24-5 | NSC_65801 | l-Fenfluramine)Show InChI InChI=1S/C12H16F3N/c1-3-16-9(2)7-10-5-4-6-11(8-10)12(13,14)15/h4-6,8-9,16H,3,7H2,1-2H3/t9-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.43E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceuticals Research Laboratories
Curated by PDSP Ki Database
| |
Mol Pharmacol 57: 75-81 (2000)
BindingDB Entry DOI: 10.7270/Q2P26WPH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2C
(Homo sapiens (Human)) | BDBM85597

(CAS_37577-24-5 | NSC_65801 | l-Fenfluramine)Show InChI InChI=1S/C12H16F3N/c1-3-16-9(2)7-10-5-4-6-11(8-10)12(13,14)15/h4-6,8-9,16H,3,7H2,1-2H3/t9-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.62E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceuticals Research Laboratories
Curated by PDSP Ki Database
| |
Mol Pharmacol 57: 75-81 (2000)
BindingDB Entry DOI: 10.7270/Q2P26WPH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2C
(Homo sapiens (Human)) | BDBM85596

(CAS_3239-45-0 | NSC_65801 | d-Fenfluramine)Show InChI InChI=1S/C12H16F3N/c1-3-16-9(2)7-10-5-4-6-11(8-10)12(13,14)15/h4-6,8-9,16H,3,7H2,1-2H3/t9-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
PubMed
| 2.08E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceuticals Research Laboratories
Curated by PDSP Ki Database
| |
Mol Pharmacol 57: 75-81 (2000)
BindingDB Entry DOI: 10.7270/Q2P26WPH |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2B
(Homo sapiens (Human)) | BDBM85596

(CAS_3239-45-0 | NSC_65801 | d-Fenfluramine)Show InChI InChI=1S/C12H16F3N/c1-3-16-9(2)7-10-5-4-6-11(8-10)12(13,14)15/h4-6,8-9,16H,3,7H2,1-2H3/t9-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3.92E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The DuPont Pharmaceuticals Research Laboratories
Curated by PDSP Ki Database
| |
Mol Pharmacol 57: 75-81 (2000)
BindingDB Entry DOI: 10.7270/Q2P26WPH |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM301217
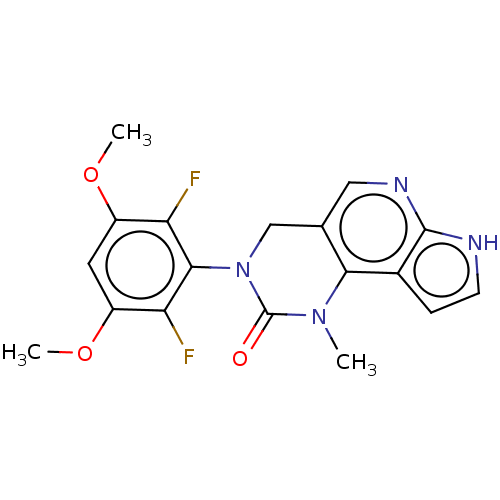
(3-(2,6-Difluoro-3,5-dimethoxyphenyl)-1-methyl-1,3,...)Show SMILES COc1cc(OC)c(F)c(N2Cc3cnc4[nH]ccc4c3N(C)C2=O)c1F Show InChI InChI=1S/C18H16F2N4O3/c1-23-15-9(7-22-17-10(15)4-5-21-17)8-24(18(23)25)16-13(19)11(26-2)6-12(27-3)14(16)20/h4-7H,8H2,1-3H3,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR1 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 2
(Homo sapiens (Human)) | BDBM301217

(3-(2,6-Difluoro-3,5-dimethoxyphenyl)-1-methyl-1,3,...)Show SMILES COc1cc(OC)c(F)c(N2Cc3cnc4[nH]ccc4c3N(C)C2=O)c1F Show InChI InChI=1S/C18H16F2N4O3/c1-23-15-9(7-22-17-10(15)4-5-21-17)8-24(18(23)25)16-13(19)11(26-2)6-12(27-3)14(16)20/h4-7H,8H2,1-3H3,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR2 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM301220
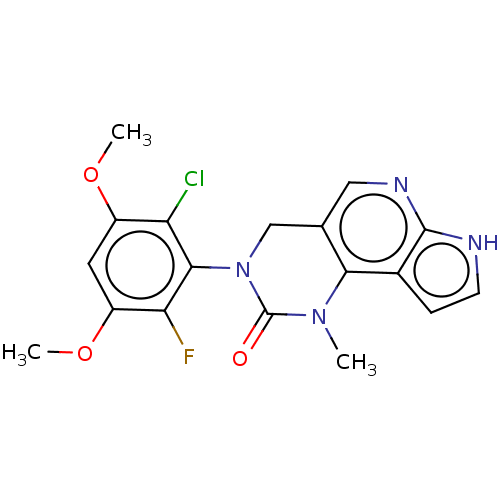
(3-(2-Chloro-6-fluoro-3,5-dimethoxyphenyl)-1-methyl...)Show SMILES COc1cc(OC)c(Cl)c(N2Cc3cnc4[nH]ccc4c3N(C)C2=O)c1F Show InChI InChI=1S/C18H16ClFN4O3/c1-23-15-9(7-22-17-10(15)4-5-21-17)8-24(18(23)25)16-13(19)11(26-2)6-12(27-3)14(16)20/h4-7H,8H2,1-3H3,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR1 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 3
(Homo sapiens (Human)) | BDBM301217

(3-(2,6-Difluoro-3,5-dimethoxyphenyl)-1-methyl-1,3,...)Show SMILES COc1cc(OC)c(F)c(N2Cc3cnc4[nH]ccc4c3N(C)C2=O)c1F Show InChI InChI=1S/C18H16F2N4O3/c1-23-15-9(7-22-17-10(15)4-5-21-17)8-24(18(23)25)16-13(19)11(26-2)6-12(27-3)14(16)20/h4-7H,8H2,1-3H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR3 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 2
(Homo sapiens (Human)) | BDBM301220

(3-(2-Chloro-6-fluoro-3,5-dimethoxyphenyl)-1-methyl...)Show SMILES COc1cc(OC)c(Cl)c(N2Cc3cnc4[nH]ccc4c3N(C)C2=O)c1F Show InChI InChI=1S/C18H16ClFN4O3/c1-23-15-9(7-22-17-10(15)4-5-21-17)8-24(18(23)25)16-13(19)11(26-2)6-12(27-3)14(16)20/h4-7H,8H2,1-3H3,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR2 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1 [1-298]
(Homo sapiens (Human)) | BDBM13467
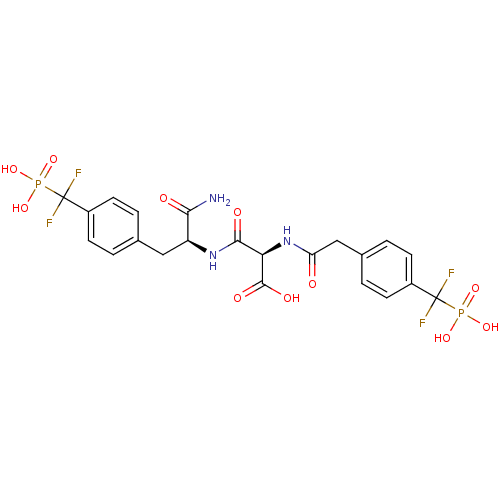
((2R)-2-{[(1S)-1-carbamoyl-2-{4-[difluoro(phosphono...)Show SMILES NC(=O)[C@H](Cc1ccc(cc1)C(F)(F)P(O)(O)=O)NC(=O)[C@@H](NC(=O)Cc1ccc(cc1)C(F)(F)P(O)(O)=O)C(O)=O |r| Show InChI InChI=1S/C22H23F4N3O11P2/c23-21(24,41(35,36)37)13-5-1-11(2-6-13)9-15(18(27)31)28-19(32)17(20(33)34)29-16(30)10-12-3-7-14(8-4-12)22(25,26)42(38,39)40/h1-8,15,17H,9-10H2,(H2,27,31)(H,28,32)(H,29,30)(H,33,34)(H2,35,36,37)(H2,38,39,40)/t15-,17+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | 7.0 | 22 |
Incyte Corporation
| Assay Description
The activity of PTP1B enzyme was assayed with 4-nitrophenyl phosphate (pNPP) as substrate. Rate of formation of the phenolate ion was monitored at 41... |
J Biol Chem 281: 38013-21 (2006)
Article DOI: 10.1074/jbc.M607913200
BindingDB Entry DOI: 10.7270/Q2JW8C4X |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 3
(Homo sapiens (Human)) | BDBM301220

(3-(2-Chloro-6-fluoro-3,5-dimethoxyphenyl)-1-methyl...)Show SMILES COc1cc(OC)c(Cl)c(N2Cc3cnc4[nH]ccc4c3N(C)C2=O)c1F Show InChI InChI=1S/C18H16ClFN4O3/c1-23-15-9(7-22-17-10(15)4-5-21-17)8-24(18(23)25)16-13(19)11(26-2)6-12(27-3)14(16)20/h4-7H,8H2,1-3H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR3 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50382932
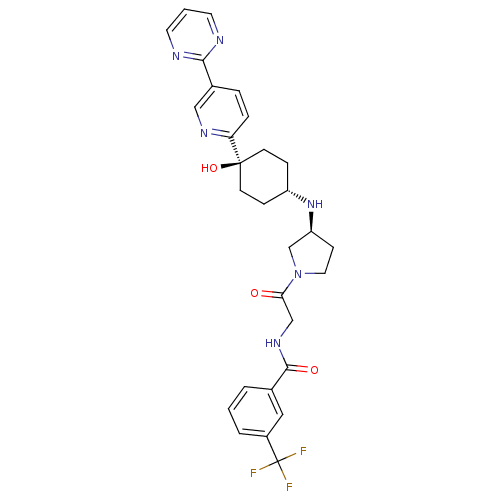
(CHEMBL2029422)Show SMILES O[C@]1(CC[C@@H](CC1)N[C@H]1CCN(C1)C(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccc(cn1)-c1ncccn1 |r,wU:4.7,wD:8.8,1.0,(25.05,5.32,;26.39,4.55,;27.72,3.79,;29.05,4.55,;29.05,6.09,;27.72,6.87,;26.39,6.09,;30.38,6.86,;31.72,6.08,;31.73,4.55,;33.2,4.09,;34.1,5.35,;33.17,6.58,;35.42,6.13,;35.41,7.67,;36.76,5.37,;38.09,6.15,;39.43,5.39,;39.44,3.85,;40.76,6.17,;40.74,7.71,;42.06,8.48,;43.41,7.73,;43.42,6.18,;42.09,5.4,;44.76,5.41,;44.77,3.87,;46.09,6.19,;46,4.52,;25.06,3.78,;23.72,4.55,;22.39,3.78,;22.39,2.24,;23.74,1.47,;25.06,2.24,;21.06,1.46,;19.73,2.22,;18.4,1.45,;18.4,-.09,;19.75,-.86,;21.07,-.08,)| Show InChI InChI=1S/C29H31F3N6O3/c30-29(31,32)21-4-1-3-19(15-21)27(40)36-17-25(39)38-14-9-23(18-38)37-22-7-10-28(41,11-8-22)24-6-5-20(16-35-24)26-33-12-2-13-34-26/h1-6,12-13,15-16,22-23,37,41H,7-11,14,17-18H2,(H,36,40)/t22-,23-,28-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of CCR2-mediated Erk phosphorylation |
ACS Med Chem Lett 2: 913-918 (2011)
Article DOI: 10.1021/ml200199c
BindingDB Entry DOI: 10.7270/Q29024TK |
More data for this
Ligand-Target Pair | |
11-beta-hydroxysteroid dehydrogenase 1
(Homo sapiens (Human)) | BDBM50595147

(CHEMBL5205840)Show SMILES Clc1ccc(cc1)C1(CC1)C(=O)N1CC[C@@]2(C1)OC(=O)c1ccccc21 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.128782
BindingDB Entry DOI: 10.7270/Q2FF3XBX |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM301243
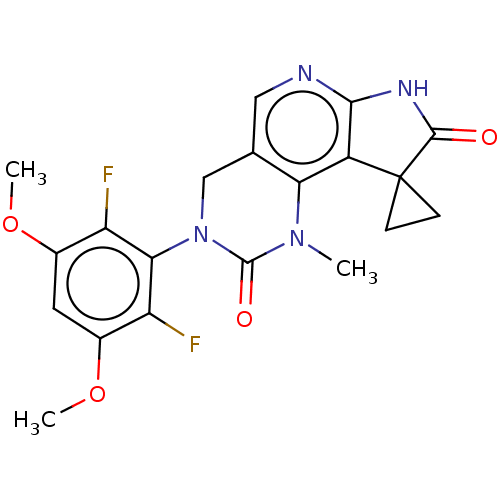
(3'-(2,6-Difluoro-3,5-dimethoxyphenyl)-1'-methyl-4'...)Show SMILES COc1cc(OC)c(F)c(N2Cc3cnc4NC(=O)C5(CC5)c4c3N(C)C2=O)c1F Show InChI InChI=1S/C20H18F2N4O4/c1-25-15-9(7-23-17-12(15)20(4-5-20)18(27)24-17)8-26(19(25)28)16-13(21)10(29-2)6-11(30-3)14(16)22/h6-7H,4-5,8H2,1-3H3,(H,23,24,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.790 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR1 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50331722
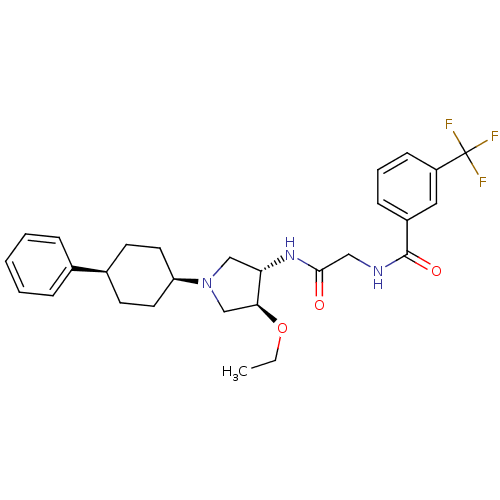
(CHEMBL1290636 | N-(2-((3S,4S)-4-ethoxy-1-(cis-4-ph...)Show SMILES CCO[C@H]1CN(C[C@@H]1NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)[C@H]1CC[C@H](CC1)c1ccccc1 |r,wU:3.2,25.26,28.33,wD:7.8,(24.27,-20.7,;23.02,-19.81,;23.17,-18.27,;21.92,-17.37,;20.46,-17.85,;19.55,-16.6,;20.45,-15.36,;21.91,-15.83,;23.25,-15.06,;24.58,-15.83,;24.58,-17.38,;25.92,-15.06,;27.26,-15.83,;28.59,-15.06,;28.59,-13.52,;29.93,-15.83,;31.25,-15.06,;32.59,-15.83,;32.59,-17.38,;31.25,-18.15,;29.93,-17.38,;31.25,-19.69,;29.93,-20.46,;32.59,-20.46,;31.24,-21.22,;18.01,-16.6,;17.24,-17.94,;15.69,-17.94,;14.93,-16.6,;15.7,-15.27,;17.23,-15.28,;13.39,-16.6,;12.62,-15.26,;11.08,-15.26,;10.31,-16.59,;11.08,-17.93,;12.62,-17.93,)| Show InChI InChI=1S/C28H34F3N3O3/c1-2-37-25-18-34(23-13-11-20(12-14-23)19-7-4-3-5-8-19)17-24(25)33-26(35)16-32-27(36)21-9-6-10-22(15-21)28(29,30)31/h3-10,15,20,23-25H,2,11-14,16-18H2,1H3,(H,32,36)(H,33,35)/t20-,23+,24-,25-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.950 | n/a | n/a | n/a | n/a | n/a | n/a |
Incyte Corporation
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from human CCR2 after 30 mins by gamma counter |
Bioorg Med Chem Lett 20: 7473-8 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.020
BindingDB Entry DOI: 10.7270/Q2TQ61S9 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363942
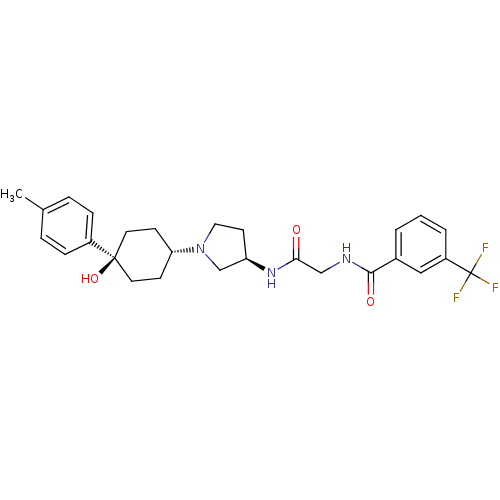
(CHEMBL1951766)Show SMILES Cc1ccc(cc1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:11.15,wD:17.21,7.8,(-10.43,2.36,;-8.89,2.39,;-8.15,3.74,;-6.61,3.78,;-5.81,2.46,;-6.55,1.1,;-8.1,1.07,;-4.27,2.5,;-5.05,3.84,;-3.46,1.18,;-1.93,1.21,;-1.18,2.56,;-1.98,3.88,;-3.53,3.85,;.36,2.59,;1.28,1.37,;2.73,1.87,;2.7,3.41,;1.23,3.86,;4.03,4.2,;5.37,3.45,;5.39,1.91,;6.7,4.23,;8.04,3.48,;9.36,4.27,;9.34,5.81,;10.71,3.51,;10.72,1.97,;12.06,1.22,;13.38,2.01,;13.36,3.55,;12.02,4.3,;14.68,4.35,;16.03,3.6,;14.66,5.89,;16.01,5.13,)| Show InChI InChI=1S/C27H32F3N3O3/c1-18-5-7-20(8-6-18)26(36)12-9-23(10-13-26)33-14-11-22(17-33)32-24(34)16-31-25(35)19-3-2-4-21(15-19)27(28,29)30/h2-8,15,22-23,36H,9-14,16-17H2,1H3,(H,31,35)(H,32,34)/t22-,23-,26-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 in human PBMC assessed as inhibition of MCP1-induced chemotaxis after 30 mins |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
High affinity nerve growth factor receptor
(Homo sapiens (Human)) | BDBM50566796
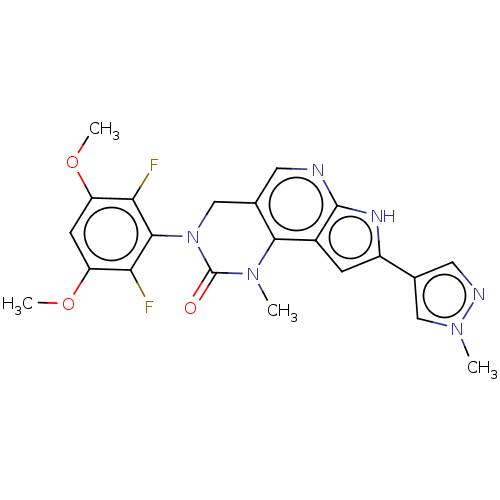
(CHEMBL4871581)Show SMILES COc1cc(OC)c(F)c(N2Cc3cnc4[nH]c(cc4c3N(C)C2=O)-c2cnn(C)c2)c1F | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of TRKA (unknown origin) incubated for 90 mins in presence of ATP by HTRF assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase JAK2
(Homo sapiens (Human)) | BDBM50566789
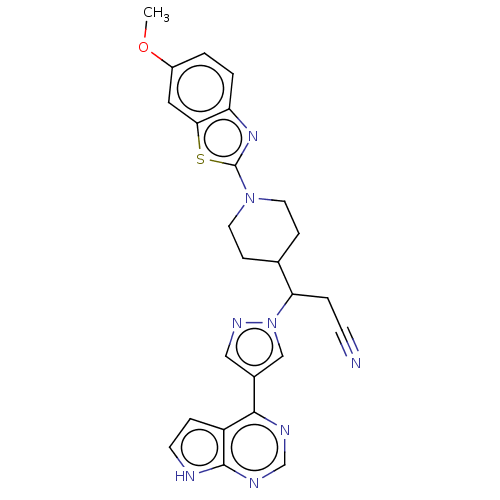
(CHEMBL4862283)Show SMILES COc1ccc2nc(sc2c1)N1CCC(CC1)C(CC#N)n1cc(cn1)-c1ncnc2[nH]ccc12 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of JAK2 (unknown origin) |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50363942

(CHEMBL1951766)Show SMILES Cc1ccc(cc1)[C@]1(O)CC[C@@H](CC1)N1CC[C@H](C1)NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F |r,wU:11.15,wD:17.21,7.8,(-10.43,2.36,;-8.89,2.39,;-8.15,3.74,;-6.61,3.78,;-5.81,2.46,;-6.55,1.1,;-8.1,1.07,;-4.27,2.5,;-5.05,3.84,;-3.46,1.18,;-1.93,1.21,;-1.18,2.56,;-1.98,3.88,;-3.53,3.85,;.36,2.59,;1.28,1.37,;2.73,1.87,;2.7,3.41,;1.23,3.86,;4.03,4.2,;5.37,3.45,;5.39,1.91,;6.7,4.23,;8.04,3.48,;9.36,4.27,;9.34,5.81,;10.71,3.51,;10.72,1.97,;12.06,1.22,;13.38,2.01,;13.36,3.55,;12.02,4.3,;14.68,4.35,;16.03,3.6,;14.66,5.89,;16.01,5.13,)| Show InChI InChI=1S/C27H32F3N3O3/c1-18-5-7-20(8-6-18)26(36)12-9-23(10-13-26)33-14-11-22(17-33)32-24(34)16-31-25(35)19-3-2-4-21(15-19)27(28,29)30/h2-8,15,22-23,36H,9-14,16-17H2,1H3,(H,31,35)(H,32,34)/t22-,23-,26-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [125I]MCP1 from CCR2 in human PBMC after 30 mins by gamma counter |
ACS Med Chem Lett 2: 450-454 (2011)
Article DOI: 10.1021/ml200030q
BindingDB Entry DOI: 10.7270/Q2WD4116 |
More data for this
Ligand-Target Pair | |
11-beta-hydroxysteroid dehydrogenase 1
(Homo sapiens (Human)) | BDBM50595148

(CHEMBL5202600)Show SMILES Clc1ccc(cc1)C1(CC1)C(=O)N1CC[C@@]2(C1)OC(=O)c1cnccc21 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.128782
BindingDB Entry DOI: 10.7270/Q2FF3XBX |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 2
(Homo sapiens (Human)) | BDBM301192
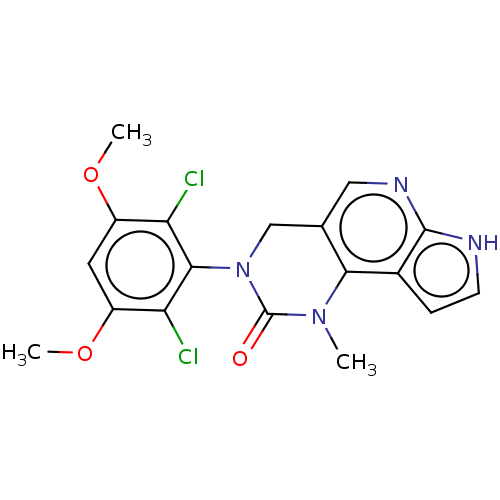
(3-(2,6-Dichloro-3,5-dimethoxyphenyl)-1-methyl-1,3,...)Show SMILES COc1cc(OC)c(Cl)c(N2Cc3cnc4[nH]ccc4c3N(C)C2=O)c1Cl Show InChI InChI=1S/C18H16Cl2N4O3/c1-23-15-9(7-22-17-10(15)4-5-21-17)8-24(18(23)25)16-13(19)11(26-2)6-12(27-3)14(16)20/h4-7H,8H2,1-3H3,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR2 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
11-beta-hydroxysteroid dehydrogenase 1
(Homo sapiens (Human)) | BDBM50595161

(CHEMBL5172072)Show SMILES CNC(=O)c1ccc(cn1)-c1ccc(cc1)C1(CC1)C(=O)N1CC[C@@]2(C1)OC(=O)c1ccccc21 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.128782
BindingDB Entry DOI: 10.7270/Q2FF3XBX |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM301192

(3-(2,6-Dichloro-3,5-dimethoxyphenyl)-1-methyl-1,3,...)Show SMILES COc1cc(OC)c(Cl)c(N2Cc3cnc4[nH]ccc4c3N(C)C2=O)c1Cl Show InChI InChI=1S/C18H16Cl2N4O3/c1-23-15-9(7-22-17-10(15)4-5-21-17)8-24(18(23)25)16-13(19)11(26-2)6-12(27-3)14(16)20/h4-7H,8H2,1-3H3,(H,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR1 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM301216
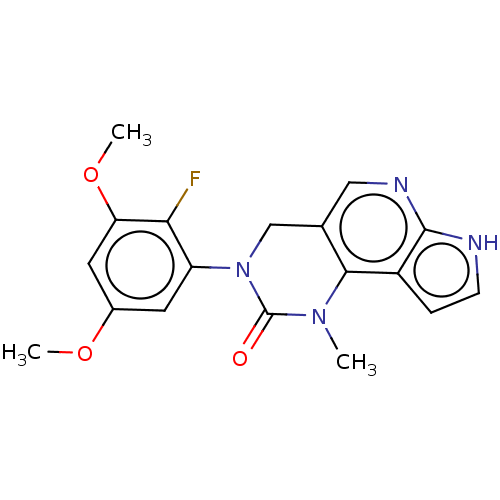
(3-(2-Fluoro-3,5-dimethoxyphenyl)-1-methyl-1,3,4,7-...)Show SMILES COc1cc(OC)c(F)c(c1)N1Cc2cnc3[nH]ccc3c2N(C)C1=O Show InChI InChI=1S/C18H17FN4O3/c1-22-16-10(8-21-17-12(16)4-5-20-17)9-23(18(22)24)13-6-11(25-2)7-14(26-3)15(13)19/h4-8H,9H2,1-3H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR1 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50339029

(5-({4-[(3S)-4-(5-Bromo-2,3-dihydro-1H-inden-1-yl)-...)Show SMILES C[C@H]1CN(CCN1C1CCc2cc(Br)ccc12)C1(C)CCN(CC1)C(=O)c1c(C)ncnc1C |r| Show InChI InChI=1S/C27H36BrN5O/c1-18-16-32(13-14-33(18)24-8-5-21-15-22(28)6-7-23(21)24)27(4)9-11-31(12-10-27)26(34)25-19(2)29-17-30-20(25)3/h6-7,15,17-18,24H,5,8-14,16H2,1-4H3/t18-,24?/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR5 in IL-10 stimulated human PBMC cells assessed as MIP-1beta induced chemotaxis |
ACS Med Chem Lett 1: 483-487 (2010)
Article DOI: 10.1021/ml1001536
BindingDB Entry DOI: 10.7270/Q2SQ90PC |
More data for this
Ligand-Target Pair | |
11-beta-hydroxysteroid dehydrogenase 1
(Homo sapiens (Human)) | BDBM50595158
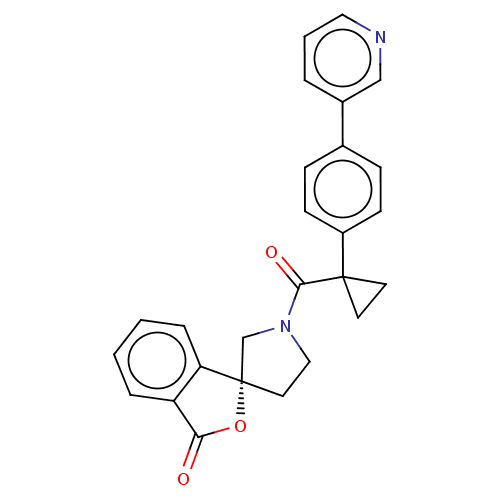
(CHEMBL5203601)Show SMILES O=C(N1CC[C@@]2(C1)OC(=O)c1ccccc21)C1(CC1)c1ccc(cc1)-c1cccnc1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.128782
BindingDB Entry DOI: 10.7270/Q2FF3XBX |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50339030
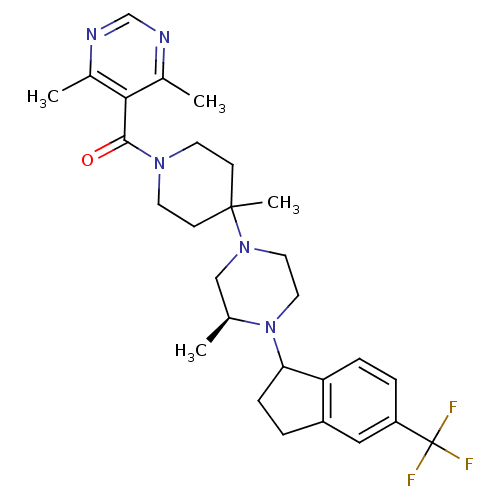
(4,6-Dimethyl-5-[(4-methyl-4-{(3S)-3-methyl-4-[5-(t...)Show SMILES C[C@H]1CN(CCN1C1CCc2cc(ccc12)C(F)(F)F)C1(C)CCN(CC1)C(=O)c1c(C)ncnc1C |r| Show InChI InChI=1S/C28H36F3N5O/c1-18-16-35(13-14-36(18)24-8-5-21-15-22(28(29,30)31)6-7-23(21)24)27(4)9-11-34(12-10-27)26(37)25-19(2)32-17-33-20(25)3/h6-7,15,17-18,24H,5,8-14,16H2,1-4H3/t18-,24?/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR5 in IL-10 stimulated human PBMC cells assessed as MIP-1beta induced chemotaxis |
ACS Med Chem Lett 1: 483-487 (2010)
Article DOI: 10.1021/ml1001536
BindingDB Entry DOI: 10.7270/Q2SQ90PC |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 3
(Homo sapiens (Human)) | BDBM301192

(3-(2,6-Dichloro-3,5-dimethoxyphenyl)-1-methyl-1,3,...)Show SMILES COc1cc(OC)c(Cl)c(N2Cc3cnc4[nH]ccc4c3N(C)C2=O)c1Cl Show InChI InChI=1S/C18H16Cl2N4O3/c1-23-15-9(7-22-17-10(15)4-5-21-17)8-24(18(23)25)16-13(19)11(26-2)6-12(27-3)14(16)20/h4-7H,8H2,1-3H3,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR3 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50145685
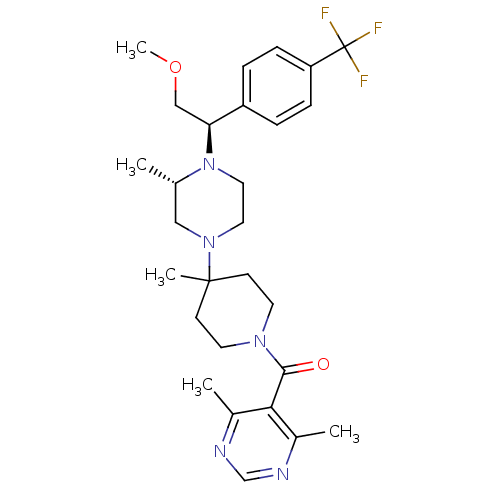
((4,6-Dimethyl-pyrimidin-5-yl)-(4-{(S)-4-[(R)-2-met...)Show SMILES COC[C@H](N1CCN(C[C@@H]1C)C1(C)CCN(CC1)C(=O)c1c(C)ncnc1C)c1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C28H38F3N5O2/c1-19-16-35(14-15-36(19)24(17-38-5)22-6-8-23(9-7-22)28(29,30)31)27(4)10-12-34(13-11-27)26(37)25-20(2)32-18-33-21(25)3/h6-9,18-19,24H,10-17H2,1-5H3/t19-,24-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR5 in IL-10 stimulated human PBMC cells assessed as MIP-1beta induced chemotaxis |
ACS Med Chem Lett 1: 483-487 (2010)
Article DOI: 10.1021/ml1001536
BindingDB Entry DOI: 10.7270/Q2SQ90PC |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50566796

(CHEMBL4871581)Show SMILES COc1cc(OC)c(F)c(N2Cc3cnc4[nH]c(cc4c3N(C)C2=O)-c2cnn(C)c2)c1F | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of VEGFR2 (unknown origin) incubated for 90 mins in presence of ATP by HTRF assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 3
(Homo sapiens (Human)) | BDBM301216

(3-(2-Fluoro-3,5-dimethoxyphenyl)-1-methyl-1,3,4,7-...)Show SMILES COc1cc(OC)c(F)c(c1)N1Cc2cnc3[nH]ccc3c2N(C)C1=O Show InChI InChI=1S/C18H17FN4O3/c1-22-16-10(8-21-17-12(16)4-5-20-17)9-23(18(22)24)13-6-11(25-2)7-14(26-3)15(13)19/h4-8H,9H2,1-3H3,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR3 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50331727
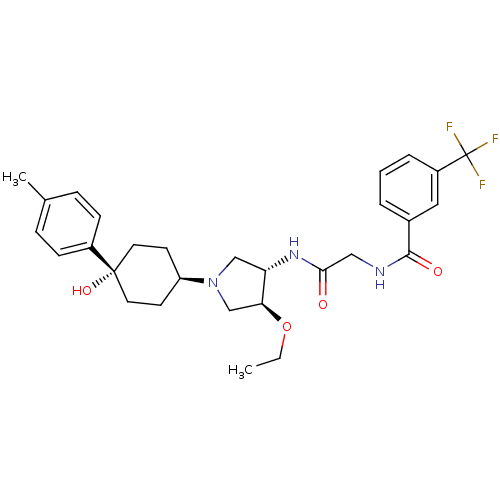
(CHEMBL1289203 | N-(2-((3S,4S)-4-ethoxy-1-(cis-4-hy...)Show SMILES CCO[C@H]1CN(C[C@@H]1NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)[C@H]1CC[C@](O)(CC1)c1ccc(C)cc1 |r,wU:3.2,25.26,wD:7.8,28.30,(4.62,-48.68,;3.37,-47.78,;3.52,-46.25,;2.27,-45.35,;.81,-45.83,;-.1,-44.58,;.81,-43.34,;2.26,-43.81,;3.6,-43.04,;4.93,-43.81,;4.93,-45.35,;6.26,-43.04,;7.6,-43.81,;8.93,-43.04,;8.93,-41.5,;10.26,-43.81,;11.59,-43.04,;12.93,-43.81,;12.93,-45.36,;11.6,-46.13,;10.26,-45.36,;11.6,-47.67,;10.26,-48.44,;12.93,-48.44,;11.58,-49.2,;-1.64,-44.58,;-2.41,-45.92,;-3.95,-45.92,;-4.72,-44.58,;-5.13,-43.09,;-3.94,-43.25,;-2.41,-43.25,;-6.25,-44.58,;-7.02,-43.24,;-8.56,-43.24,;-9.33,-44.57,;-10.87,-44.57,;-8.56,-45.91,;-7.02,-45.91,)| Show InChI InChI=1S/C29H36F3N3O4/c1-3-39-25-18-35(23-11-13-28(38,14-12-23)21-9-7-19(2)8-10-21)17-24(25)34-26(36)16-33-27(37)20-5-4-6-22(15-20)29(30,31)32/h4-10,15,23-25,38H,3,11-14,16-18H2,1-2H3,(H,33,37)(H,34,36)/t23-,24-,25-,28-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Incyte Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CCR2 assessed as inhibition of MCP1 induced chemotaxis after 30 mins |
Bioorg Med Chem Lett 20: 7473-8 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.020
BindingDB Entry DOI: 10.7270/Q2TQ61S9 |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 2
(Homo sapiens (Human)) | BDBM301191
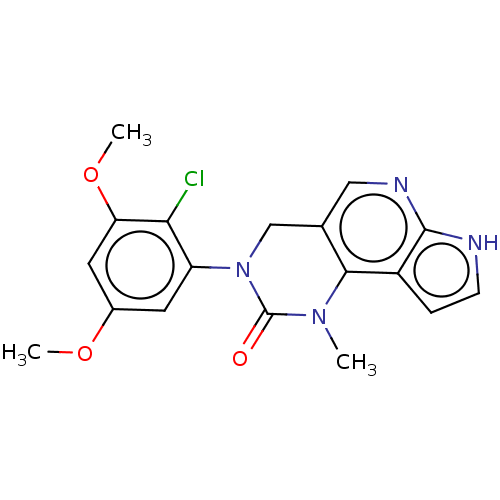
(3-(2-Chloro-3,5-dimethoxyphenyl)-1-methyl-1,3,4,7-...)Show SMILES COc1cc(OC)c(Cl)c(c1)N1Cc2cnc3[nH]ccc3c2N(C)C1=O Show InChI InChI=1S/C18H17ClN4O3/c1-22-16-10(8-21-17-12(16)4-5-20-17)9-23(18(22)24)13-6-11(25-2)7-14(26-3)15(13)19/h4-8H,9H2,1-3H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR2 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 2
(Homo sapiens (Human)) | BDBM301216

(3-(2-Fluoro-3,5-dimethoxyphenyl)-1-methyl-1,3,4,7-...)Show SMILES COc1cc(OC)c(F)c(c1)N1Cc2cnc3[nH]ccc3c2N(C)C1=O Show InChI InChI=1S/C18H17FN4O3/c1-22-16-10(8-21-17-12(16)4-5-20-17)9-23(18(22)24)13-6-11(25-2)7-14(26-3)15(13)19/h4-8H,9H2,1-3H3,(H,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR2 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM301261
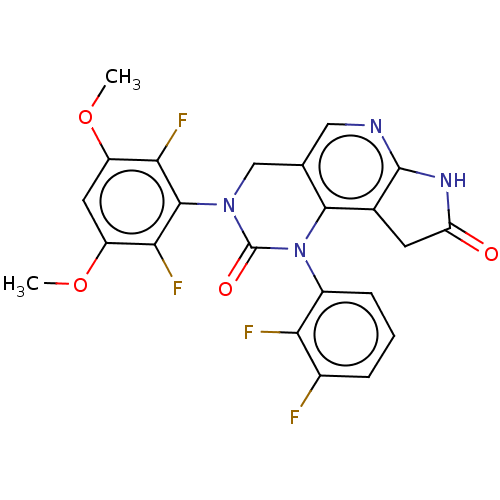
(3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-(2,3-difluo...)Show SMILES COc1cc(OC)c(F)c(N2Cc3cnc4NC(=O)Cc4c3N(C2=O)c2cccc(F)c2F)c1F Show InChI InChI=1S/C23H16F4N4O4/c1-34-14-7-15(35-2)19(27)21(18(14)26)30-9-10-8-28-22-11(6-16(32)29-22)20(10)31(23(30)33)13-5-3-4-12(24)17(13)25/h3-5,7-8H,6,9H2,1-2H3,(H,28,29,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human FGFR1 using biotinylated-EQEDEPEGDYFEWLE peptide as substrate incubated for 1 hr in presence of ATP by FRET assay |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c00713
BindingDB Entry DOI: 10.7270/Q2S1867R |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50339033
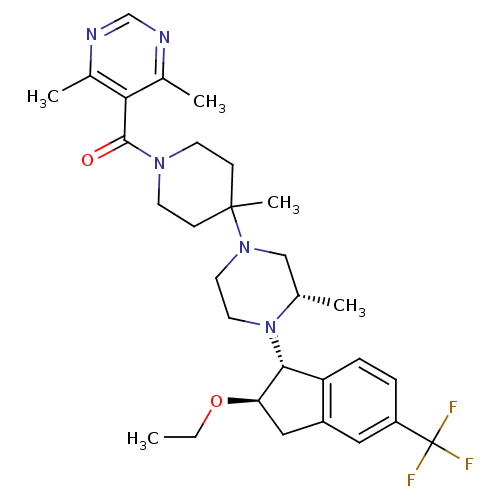
(5-[(4-(3S)-4-[(1R,2R)-2-ethoxy-5-(trifluoromethyl)...)Show SMILES CCO[C@@H]1Cc2cc(ccc2[C@H]1N1CCN(C[C@@H]1C)C1(C)CCN(CC1)C(=O)c1c(C)ncnc1C)C(F)(F)F |r| Show InChI InChI=1S/C30H40F3N5O2/c1-6-40-25-16-22-15-23(30(31,32)33)7-8-24(22)27(25)38-14-13-37(17-19(38)2)29(5)9-11-36(12-10-29)28(39)26-20(3)34-18-35-21(26)4/h7-8,15,18-19,25,27H,6,9-14,16-17H2,1-5H3/t19-,25+,27+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR5 assessed as inhibition of receptor internalization |
ACS Med Chem Lett 1: 483-487 (2010)
Article DOI: 10.1021/ml1001536
BindingDB Entry DOI: 10.7270/Q2SQ90PC |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM11868

(CHEMBL256157 | N-Hydroxy-4-{[(4-phenoxyphenyl)sulf...)Show SMILES ONC(=O)C1(CS(=O)(=O)c2ccc(Oc3ccccc3)cc2)CCN(CC#C)CC1 Show InChI InChI=1S/C22H24N2O5S/c1-2-14-24-15-12-22(13-16-24,21(25)23-26)17-30(27,28)20-10-8-19(9-11-20)29-18-6-4-3-5-7-18/h1,3-11,26H,12-17H2,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Incyte Corporation
Curated by ChEMBL
| Assay Description
Inhibition of MMP9 |
Bioorg Med Chem Lett 18: 560-4 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.086
BindingDB Entry DOI: 10.7270/Q2CN73MJ |
More data for this
Ligand-Target Pair | |
72 kDa type IV collagenase
(Homo sapiens (Human)) | BDBM11868

(CHEMBL256157 | N-Hydroxy-4-{[(4-phenoxyphenyl)sulf...)Show SMILES ONC(=O)C1(CS(=O)(=O)c2ccc(Oc3ccccc3)cc2)CCN(CC#C)CC1 Show InChI InChI=1S/C22H24N2O5S/c1-2-14-24-15-12-22(13-16-24,21(25)23-26)17-30(27,28)20-10-8-19(9-11-20)29-18-6-4-3-5-7-18/h1,3-11,26H,12-17H2,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Incyte Corporation
Curated by ChEMBL
| Assay Description
Inhibition of MMP2 |
Bioorg Med Chem Lett 18: 560-4 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.086
BindingDB Entry DOI: 10.7270/Q2CN73MJ |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM11868

(CHEMBL256157 | N-Hydroxy-4-{[(4-phenoxyphenyl)sulf...)Show SMILES ONC(=O)C1(CS(=O)(=O)c2ccc(Oc3ccccc3)cc2)CCN(CC#C)CC1 Show InChI InChI=1S/C22H24N2O5S/c1-2-14-24-15-12-22(13-16-24,21(25)23-26)17-30(27,28)20-10-8-19(9-11-20)29-18-6-4-3-5-7-18/h1,3-11,26H,12-17H2,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Incyte Corporation
Curated by ChEMBL
| Assay Description
Inhibition of MMP3 |
Bioorg Med Chem Lett 18: 560-4 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.086
BindingDB Entry DOI: 10.7270/Q2CN73MJ |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1 [1-298]
(Homo sapiens (Human)) | BDBM13469
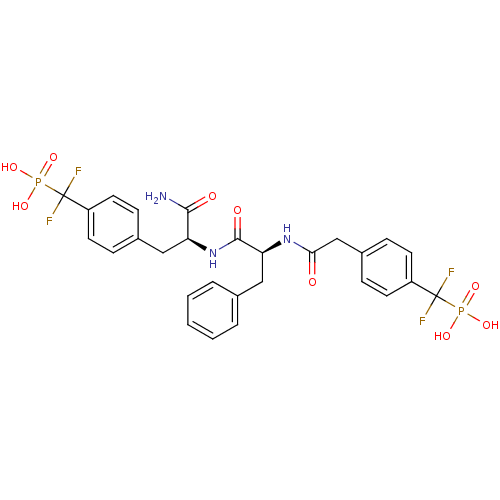
(({4-[(2S)-2-carbamoyl-2-[(2S)-2-(1-{4-[difluoro(ph...)Show SMILES NC(=O)[C@H](Cc1ccc(cc1)C(F)(F)P(O)(O)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)Cc1ccc(cc1)C(F)(F)P(O)(O)=O |r| Show InChI InChI=1S/C28H29F4N3O9P2/c29-27(30,45(39,40)41)20-10-6-18(7-11-20)14-22(25(33)37)35-26(38)23(15-17-4-2-1-3-5-17)34-24(36)16-19-8-12-21(13-9-19)28(31,32)46(42,43)44/h1-13,22-23H,14-16H2,(H2,33,37)(H,34,36)(H,35,38)(H2,39,40,41)(H2,42,43,44)/t22-,23-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | 7.0 | 22 |
Incyte Corporation
| Assay Description
The activity of PTP1B enzyme was assayed with 4-nitrophenyl phosphate (pNPP) as substrate. Rate of formation of the phenolate ion was monitored at 41... |
J Biol Chem 281: 38013-21 (2006)
Article DOI: 10.1074/jbc.M607913200
BindingDB Entry DOI: 10.7270/Q2JW8C4X |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50331723
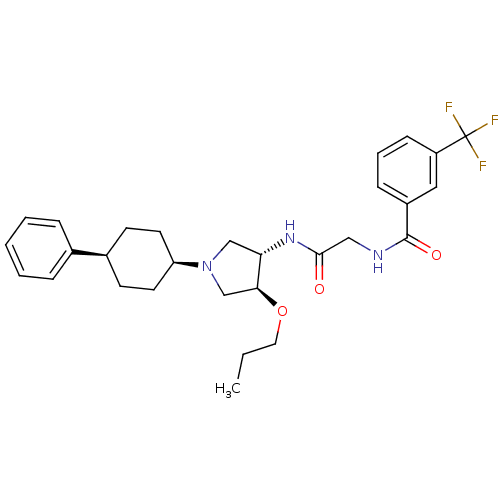
(CHEMBL1290637 | N-(2-oxo-2-((3S,4S)-1-(cis-4-pheny...)Show SMILES CCCO[C@H]1CN(C[C@@H]1NC(=O)CNC(=O)c1cccc(c1)C(F)(F)F)[C@H]1CC[C@H](CC1)c1ccccc1 |r,wU:4.3,26.27,29.34,wD:8.9,(2.69,-29.05,;2.84,-27.52,;1.59,-26.62,;1.75,-25.09,;.5,-24.19,;-.97,-24.67,;-1.87,-23.42,;-.97,-22.17,;.49,-22.65,;1.82,-21.88,;3.15,-22.65,;3.16,-24.19,;4.49,-21.88,;5.82,-22.65,;7.16,-21.88,;7.16,-20.34,;8.49,-22.65,;9.81,-21.88,;11.15,-22.65,;11.16,-24.2,;9.82,-24.96,;8.49,-24.2,;9.82,-26.5,;8.49,-27.28,;11.15,-27.28,;9.8,-28.04,;-3.41,-23.42,;-4.18,-24.76,;-5.72,-24.76,;-6.49,-23.42,;-5.72,-22.09,;-4.19,-22.09,;-8.02,-23.42,;-8.79,-22.08,;-10.33,-22.07,;-11.1,-23.41,;-10.33,-24.75,;-8.8,-24.75,)| Show InChI InChI=1S/C29H36F3N3O3/c1-2-15-38-26-19-35(24-13-11-21(12-14-24)20-7-4-3-5-8-20)18-25(26)34-27(36)17-33-28(37)22-9-6-10-23(16-22)29(30,31)32/h3-10,16,21,24-26H,2,11-15,17-19H2,1H3,(H,33,37)(H,34,36)/t21-,24+,25-,26-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Incyte Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CCR2 assessed as inhibition of MCP1 induced chemotaxis after 30 mins |
Bioorg Med Chem Lett 20: 7473-8 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.020
BindingDB Entry DOI: 10.7270/Q2TQ61S9 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 2
(Homo sapiens (Human)) | BDBM50382942
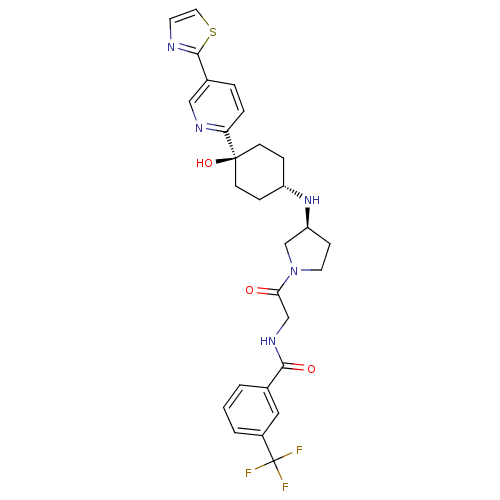
(CHEMBL2029423)Show SMILES O[C@]1(CC[C@@H](CC1)N[C@H]1CCN(C1)C(=O)CNC(=O)c1cccc(c1)C(F)(F)F)c1ccc(cn1)-c1nccs1 |r,wU:4.7,wD:8.8,1.0,(-4.43,-4.47,;-3.09,-5.24,;-1.76,-6,;-.43,-5.24,;-.43,-3.7,;-1.76,-2.92,;-3.09,-3.7,;.91,-2.94,;2.24,-3.71,;2.26,-5.24,;3.73,-5.7,;4.62,-4.44,;3.7,-3.21,;5.95,-3.67,;5.94,-2.13,;7.29,-4.43,;8.61,-3.65,;9.95,-4.41,;9.96,-5.95,;11.28,-3.63,;11.26,-2.1,;12.58,-1.32,;13.93,-2.08,;13.94,-3.62,;12.62,-4.4,;15.28,-4.38,;15.29,-5.92,;16.61,-3.6,;16.53,-5.27,;-4.42,-6.01,;-5.75,-5.24,;-7.09,-6.01,;-7.08,-7.56,;-5.74,-8.32,;-4.41,-7.55,;-8.42,-8.33,;-9.83,-7.71,;-10.85,-8.86,;-10.08,-10.19,;-8.58,-9.87,)| Show InChI InChI=1S/C28H30F3N5O3S/c29-28(30,31)20-3-1-2-18(14-20)25(38)34-16-24(37)36-12-8-22(17-36)35-21-6-9-27(39,10-7-21)23-5-4-19(15-33-23)26-32-11-13-40-26/h1-5,11,13-15,21-22,35,39H,6-10,12,16-17H2,(H,34,38)/t21-,22-,27-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Antagonist activity at CCR2 receptor in human PBMC assessed as inhibition of MCP1-mediated leukocyte chemotaxis after 30 mins by microscopy |
ACS Med Chem Lett 2: 913-918 (2011)
Article DOI: 10.1021/ml200199c
BindingDB Entry DOI: 10.7270/Q29024TK |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data