

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50434335 (CHEMBL2386729 | US9181275, 3) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | 0.0200 | -61.1 | n/a | n/a | n/a | n/a | n/a | n/a | 25 |
AbbVie Inc. US Patent | Assay Description To determine the effectiveness of representative compounds of this invention as histamine-3 receptor ligands (H3 receptor ligands), the following tes... | US Patent US9181275 (2015) BindingDB Entry DOI: 10.7270/Q2R49PJP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine/threonine-protein kinase PAK 1 (Homo sapiens (Human)) | BDBM50148931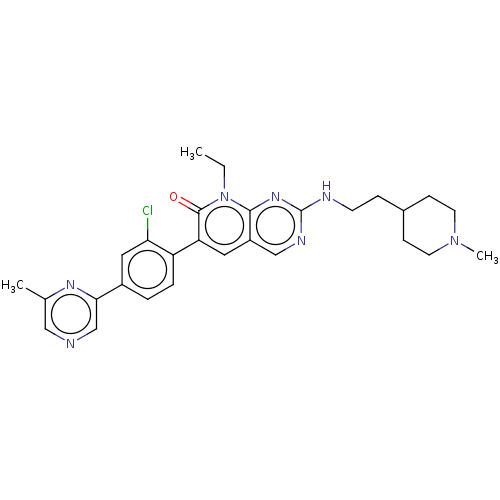 (CHEMBL3770186) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.0230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Inhibition of full length recombinant human N-terminal GST/His6-tagged PAK1 expressed in sf9 insect cells using tetra LRRWSLG as substrate preincubat... | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112517 BindingDB Entry DOI: 10.7270/Q2Q243W7 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50434337 (CHEMBL2386727 | US9181275, 1) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | 0.0320 | -59.9 | n/a | n/a | n/a | n/a | n/a | n/a | 25 |
AbbVie Inc. US Patent | Assay Description To determine the effectiveness of representative compounds of this invention as histamine-3 receptor ligands (H3 receptor ligands), the following tes... | US Patent US9181275 (2015) BindingDB Entry DOI: 10.7270/Q2R49PJP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319536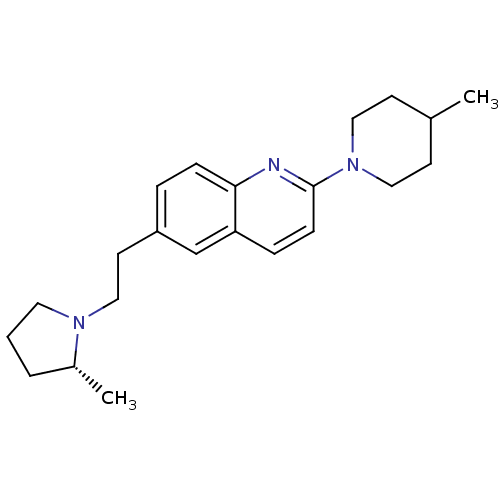 ((R)-2-(4-methylpiperidin-1-yl)-6-(2-(2-methylpyrro...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50434336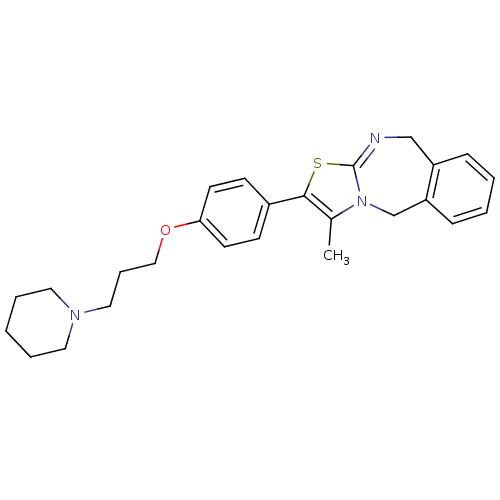 (CHEMBL2386728 | US9181275, 2) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | 0.0500 | -58.8 | n/a | n/a | n/a | n/a | n/a | n/a | 25 |
AbbVie Inc. US Patent | Assay Description To determine the effectiveness of representative compounds of this invention as histamine-3 receptor ligands (H3 receptor ligands), the following tes... | US Patent US9181275 (2015) BindingDB Entry DOI: 10.7270/Q2R49PJP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50319536 ((R)-2-(4-methylpiperidin-1-yl)-6-(2-(2-methylpyrro...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from rat cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Homo sapiens (Human)) | BDBM50599186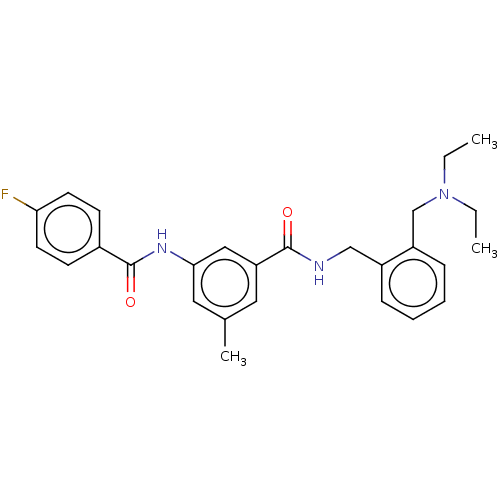 (CHEMBL5201089) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Citation and Details Article DOI: 10.1021/acs.jmedchem.2c00944 BindingDB Entry DOI: 10.7270/Q2HX1HP4 | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319552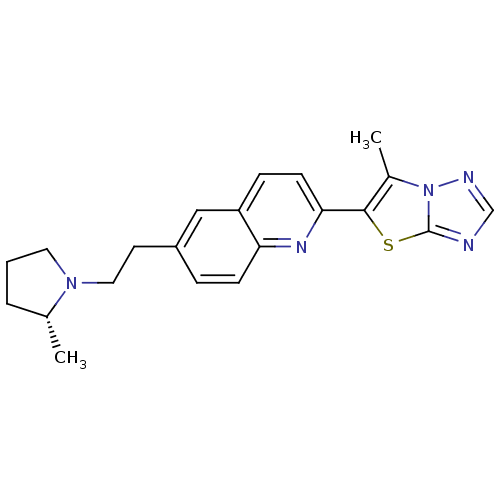 ((R)-6-methyl-5-(6-(2-(2-methylpyrrolidin-1-yl)ethy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319549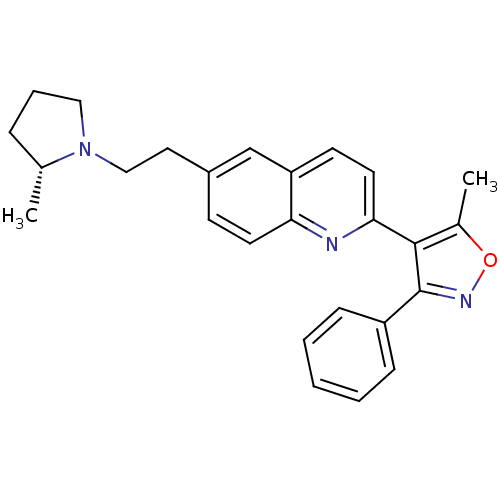 ((R)-5-methyl-4-(6-(2-(2-methylpyrrolidin-1-yl)ethy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319509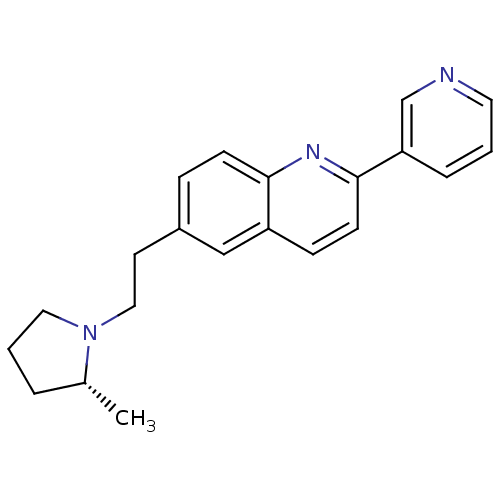 ((R)-6-(2-(2-methylpyrrolidin-1-yl)ethyl)-2-(pyridi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319517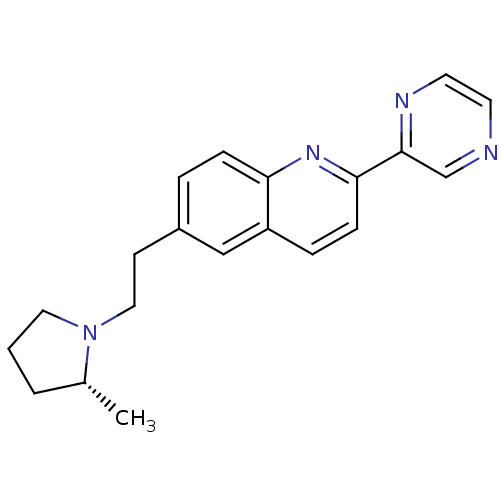 ((R)-6-(2-(2-methylpyrrolidin-1-yl)ethyl)-2-(pyrazi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319554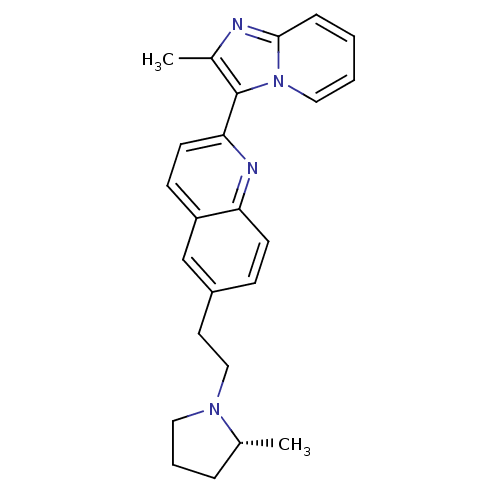 ((R)-2-(2-methylimidazo[1,2-a]pyridin-3-yl)-6-(2-(2...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319538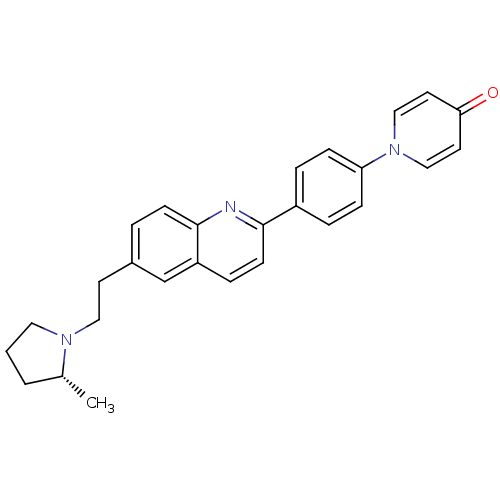 ((R)-1-(4-(6-(2-(2-methylpyrrolidin-1-yl)ethyl)quin...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319512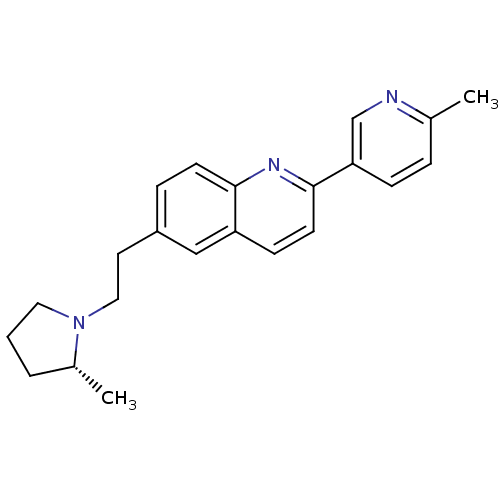 ((R)-2-(6-methylpyridin-3-yl)-6-(2-(2-methylpyrroli...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50247053 (1-(3-(3-(4-chlorophenyl)propoxy)propyl)piperidine ...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA | Assay Description Antagonist activity at recombinant human H3 receptor expressed in CHO-K1 cell membranes incubated for 60 mins by [35S]GTPgammaS binding assay | Citation and Details Article DOI: 10.1016/j.ejmech.2020.112667 BindingDB Entry DOI: 10.7270/Q2HD80BX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319547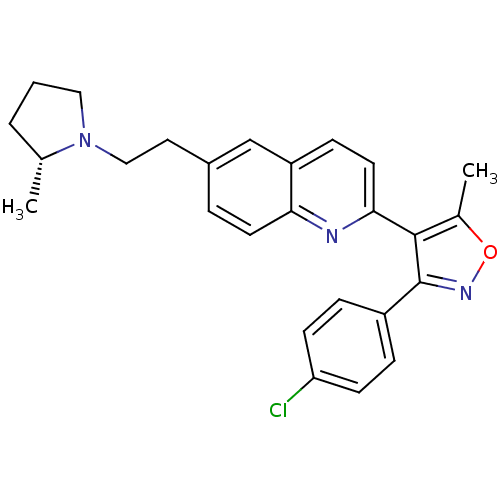 ((R)-3-(4-chlorophenyl)-5-methyl-4-(6-(2-(2-methylp...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319528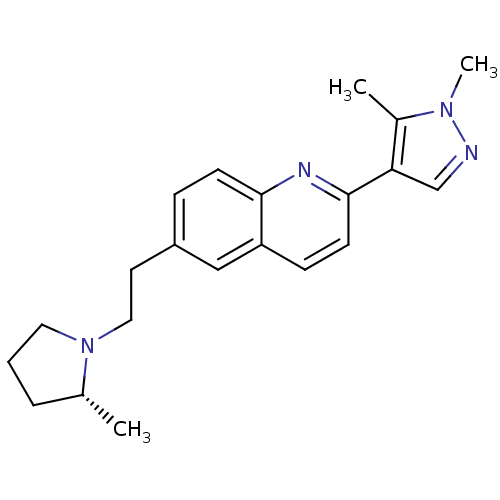 ((R)-2-(1,5-dimethyl-1H-pyrazol-4-yl)-6-(2-(2-methy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50224191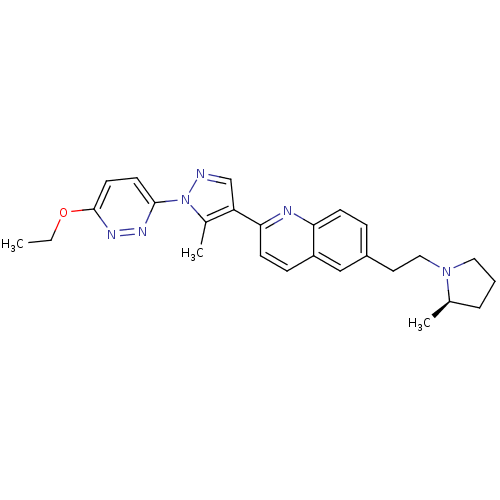 ((R)-2-(1-(6-ethoxypyridazin-3-yl)-5-methyl-1H-pyra...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha methyl histamine from human H3 receptor expressed in C6 cells | J Med Chem 50: 5439-48 (2007) Article DOI: 10.1021/jm0705051 BindingDB Entry DOI: 10.7270/Q25M65G0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50224191 ((R)-2-(1-(6-ethoxypyridazin-3-yl)-5-methyl-1H-pyra...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319508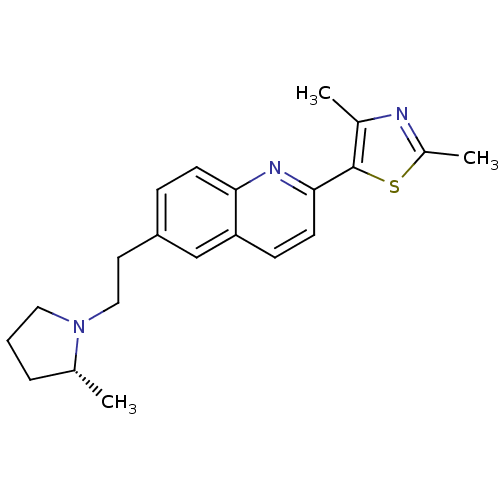 ((R)-2,4-dimethyl-5-(6-(2-(2-methylpyrrolidin-1-yl)...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319533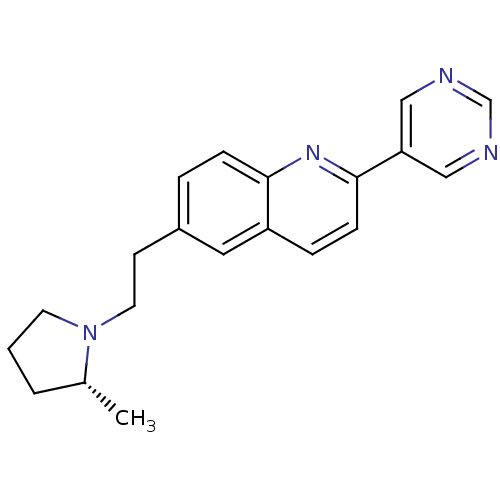 ((R)-6-(2-(2-methylpyrrolidin-1-yl)ethyl)-2-(pyrimi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319566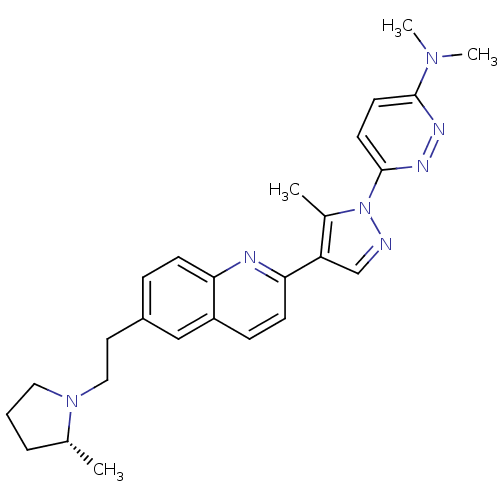 ((R)-N,N-dimethyl-6-(5-methyl-4-(6-(2-(2-methylpyrr...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319527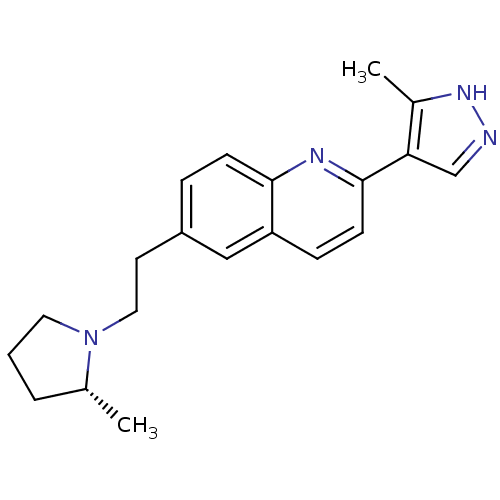 ((R)-2-(3-methyl-1H-pyrazol-4-yl)-6-(2-(2-methylpyr...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319510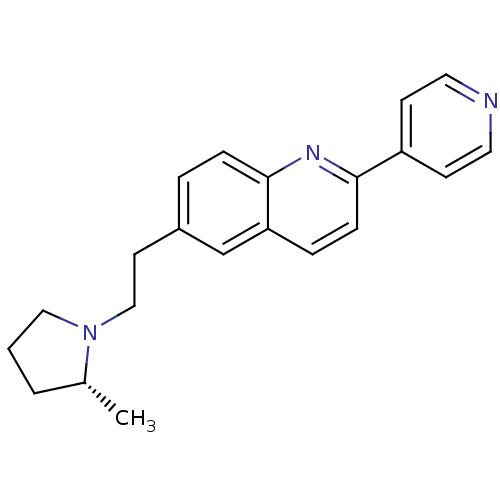 ((R)-6-(2-(2-methylpyrrolidin-1-yl)ethyl)-2-(pyridi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319534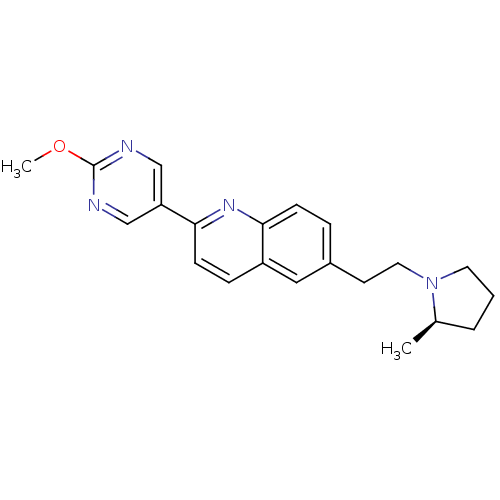 ((R)-2-(2-methoxypyrimidin-5-yl)-6-(2-(2-methylpyrr...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50224192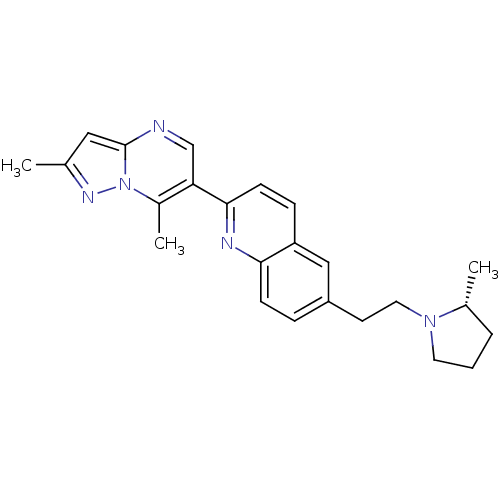 ((R)-2-(2,7-dimethylpyrazolo[1,5-a]pyrimidin-6-yl)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50224192 ((R)-2-(2,7-dimethylpyrazolo[1,5-a]pyrimidin-6-yl)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha methyl histamine from human H3 receptor expressed in C6 cells | J Med Chem 50: 5439-48 (2007) Article DOI: 10.1021/jm0705051 BindingDB Entry DOI: 10.7270/Q25M65G0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319511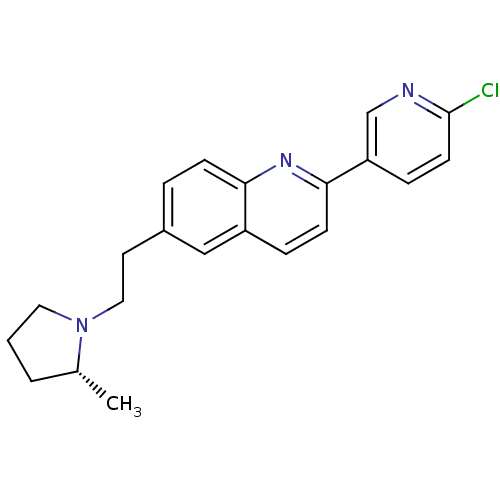 ((R)-2-(6-chloropyridin-3-yl)-6-(2-(2-methylpyrroli...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50319552 ((R)-6-methyl-5-(6-(2-(2-methylpyrrolidin-1-yl)ethy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from rat cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Serine/threonine-protein kinase PLK4 (Homo sapiens (Human)) | BDBM50073587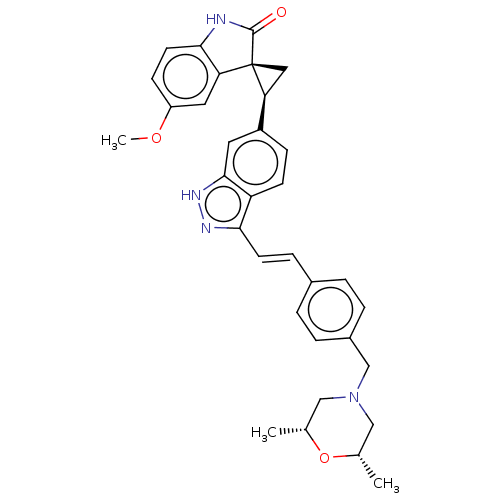 (CHEMBL3408947 | US10358436, Example A185 | US20230...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Zhengzhou University Curated by ChEMBL | Assay Description Competitive inhibition of PLK-4 (unknown origin) | Eur J Med Chem 95: 35-40 (2015) Article DOI: 10.1016/j.ejmech.2015.03.020 BindingDB Entry DOI: 10.7270/Q2NG4SBV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50224187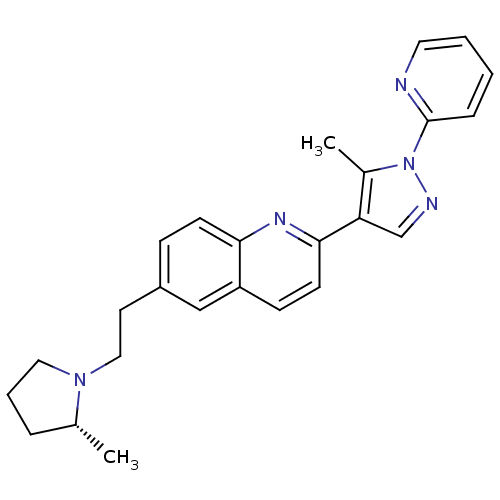 ((R)-2-(5-methyl-1-(pyridin-2-yl)-1H-pyrazol-4-yl)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50224187 ((R)-2-(5-methyl-1-(pyridin-2-yl)-1H-pyrazol-4-yl)-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha methyl histamine from human H3 receptor expressed in C6 cells | J Med Chem 50: 5439-48 (2007) Article DOI: 10.1021/jm0705051 BindingDB Entry DOI: 10.7270/Q25M65G0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319514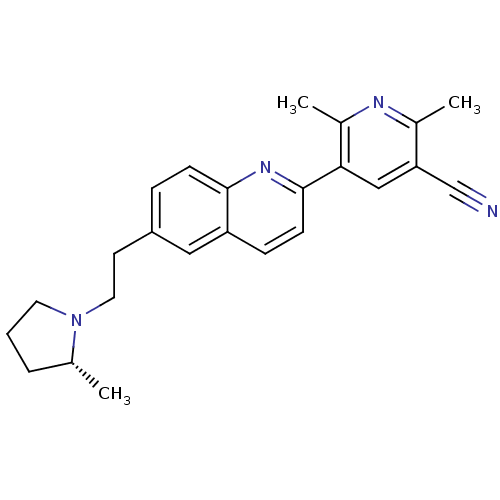 ((R)-2,6-dimethyl-5-(6-(2-(2-methylpyrrolidin-1-yl)...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D(2) dopamine receptor (Homo sapiens (Human)) | BDBM21397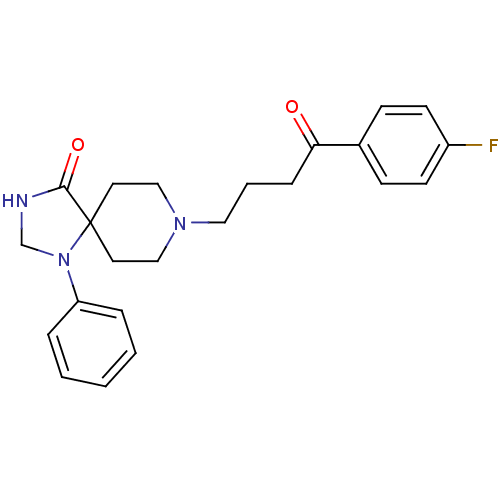 (8-[4-(4-fluorophenyl)-4-keto-butyl]-1-phenyl-1,3,8...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Displacement of [3H]Spiprone from human dopamine D2 receptor expressed in HEK293 cells | Bioorg Med Chem 20: 4862-71 (2012) Article DOI: 10.1016/j.bmc.2012.05.057 BindingDB Entry DOI: 10.7270/Q28P61K9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50224189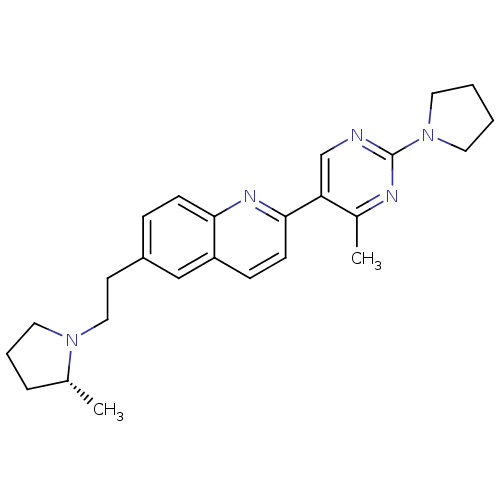 ((R)-2-(4-methyl-2-(pyrrolidin-1-yl)pyrimidin-5-yl)...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha methyl histamine from human H3 receptor expressed in C6 cells | J Med Chem 50: 5439-48 (2007) Article DOI: 10.1021/jm0705051 BindingDB Entry DOI: 10.7270/Q25M65G0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319545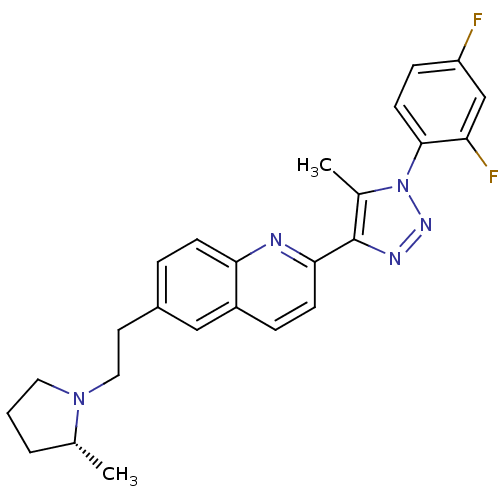 (2-(1-(2,4-difluorophenyl)-5-methyl-1H-1,2,3-triazo...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50224189 ((R)-2-(4-methyl-2-(pyrrolidin-1-yl)pyrimidin-5-yl)...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50319538 ((R)-1-(4-(6-(2-(2-methylpyrrolidin-1-yl)ethyl)quin...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from rat cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50256492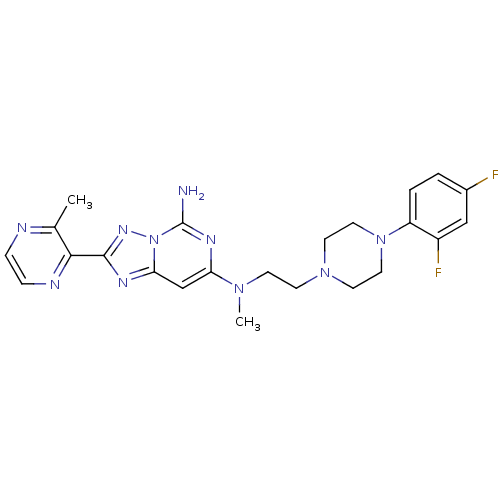 (CHEMBL480570 | N7-(2-(4-(2,4-difluorophenyl)pipera...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute Curated by ChEMBL | Assay Description Displacement of [3H]SCH58261 from human adenosine A2A receptor expressed in HEK293 cells | Bioorg Med Chem Lett 19: 967-71 (2009) Article DOI: 10.1016/j.bmcl.2008.11.075 BindingDB Entry DOI: 10.7270/Q2CC10KT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50256419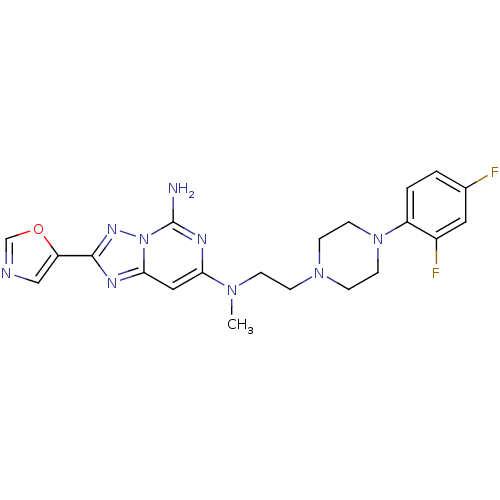 (CHEMBL480542 | N7-(2-(4-(2,4-difluorophenyl)pipera...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute Curated by ChEMBL | Assay Description Displacement of [3H]SCH58261 from human adenosine A2A receptor expressed in HEK293 cells | Bioorg Med Chem Lett 19: 967-71 (2009) Article DOI: 10.1016/j.bmcl.2008.11.075 BindingDB Entry DOI: 10.7270/Q2CC10KT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A2a (Homo sapiens (Human)) | BDBM50256362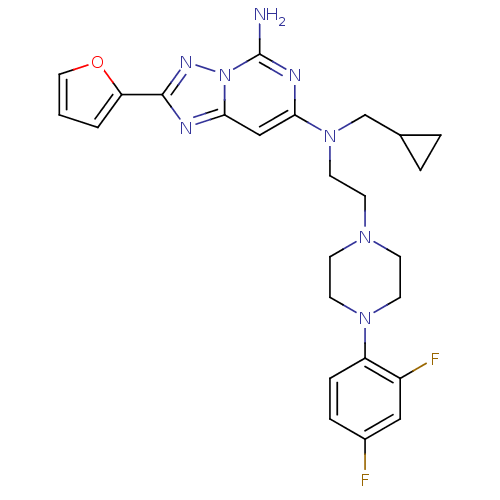 (CHEMBL470942 | N7-(cyclopropylmethyl)-N7-(2-(4-(2,...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute Curated by ChEMBL | Assay Description Displacement of [3H]SCH58261 from human adenosine A2A receptor expressed in HEK293 cells | Bioorg Med Chem Lett 19: 967-71 (2009) Article DOI: 10.1016/j.bmcl.2008.11.075 BindingDB Entry DOI: 10.7270/Q2CC10KT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50319549 ((R)-5-methyl-4-(6-(2-(2-methylpyrrolidin-1-yl)ethy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from rat cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319557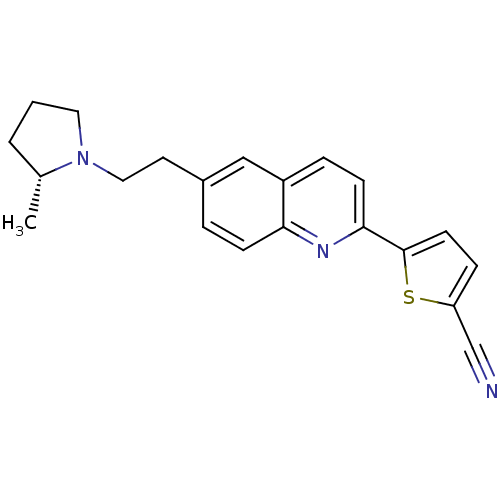 ((R)-5-(6-(2-(2-methylpyrrolidin-1-yl)ethyl)quinoli...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319560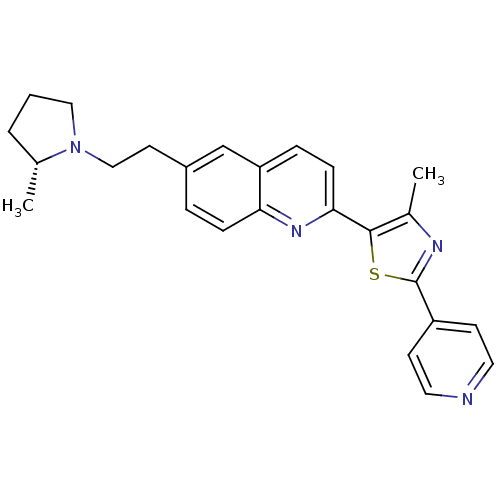 ((R)-4-methyl-5-(6-(2-(2-methylpyrrolidin-1-yl)ethy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50434334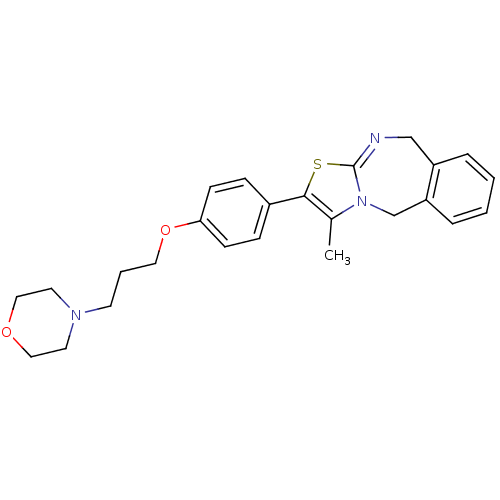 (CHEMBL2386730 | US9181275, 4) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | 0.324 | -54.2 | n/a | n/a | n/a | n/a | n/a | n/a | 25 |
AbbVie Inc. US Patent | Assay Description To determine the effectiveness of representative compounds of this invention as histamine-3 receptor ligands (H3 receptor ligands), the following tes... | US Patent US9181275 (2015) BindingDB Entry DOI: 10.7270/Q2R49PJP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50224188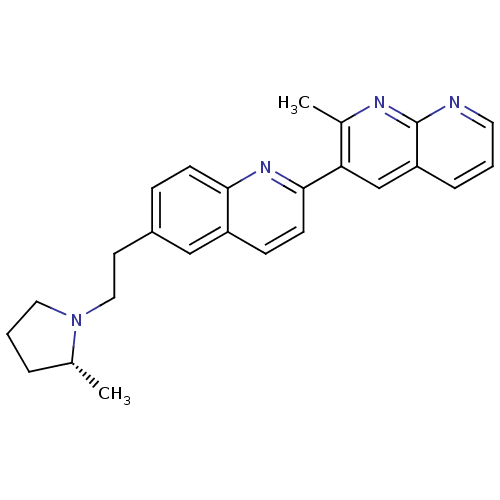 ((R)-2-methyl-3-(6-(2-(2-methylpyrrolidin-1-yl)ethy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha methyl histamine from human H3 receptor expressed in C6 cells | J Med Chem 50: 5439-48 (2007) Article DOI: 10.1021/jm0705051 BindingDB Entry DOI: 10.7270/Q25M65G0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50224188 ((R)-2-methyl-3-(6-(2-(2-methylpyrrolidin-1-yl)ethy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319543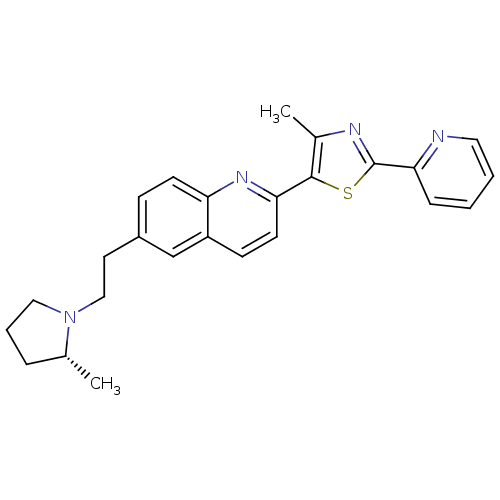 ((R)-4-methyl-5-(6-(2-(2-methylpyrrolidin-1-yl)ethy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Rattus norvegicus (rat)) | BDBM50319545 (2-(1-(2,4-difluorophenyl)-5-methyl-1H-1,2,3-triazo...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 0.350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from rat cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50319544 ((R)-4-methyl-5-(6-(2-(2-methylpyrrolidin-1-yl)ethy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Displacement of [3H]-N-alpha-methylhistamine from human cloned histamine H3 receptor | Bioorg Med Chem Lett 20: 3295-300 (2010) Article DOI: 10.1016/j.bmcl.2010.04.045 BindingDB Entry DOI: 10.7270/Q2XW4JZN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 18928 total ) | Next | Last >> |