 Found 2249 hits with Last Name = 'graham' and Initial = 'k'
Found 2249 hits with Last Name = 'graham' and Initial = 'k' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50246899
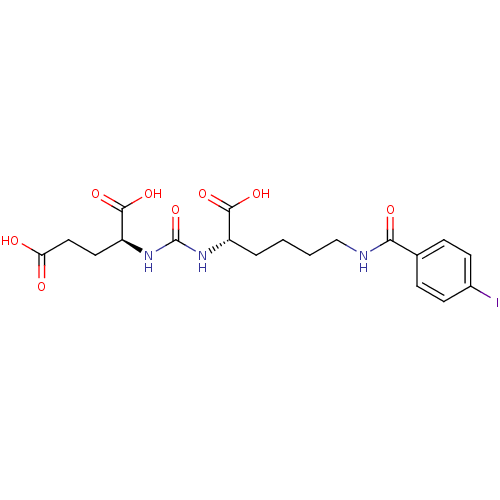
((S)-2-(3-((S)-1-carboxy-5-(4-iodobenzamido)pentyl)...)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCCCNC(=O)c1ccc(I)cc1)C(O)=O)C(O)=O |r| Show InChI InChI=1S/C19H24IN3O8/c20-12-6-4-11(5-7-12)16(26)21-10-2-1-3-13(17(27)28)22-19(31)23-14(18(29)30)8-9-15(24)25/h4-7,13-14H,1-3,8-10H2,(H,21,26)(H,24,25)(H,27,28)(H,29,30)(H2,22,23,31)/t13-,14-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare
Curated by ChEMBL
| Assay Description
Binding affinity to NAALADase |
J Med Chem 55: 9510-20 (2012)
Article DOI: 10.1021/jm300710j
BindingDB Entry DOI: 10.7270/Q28053R3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM17759
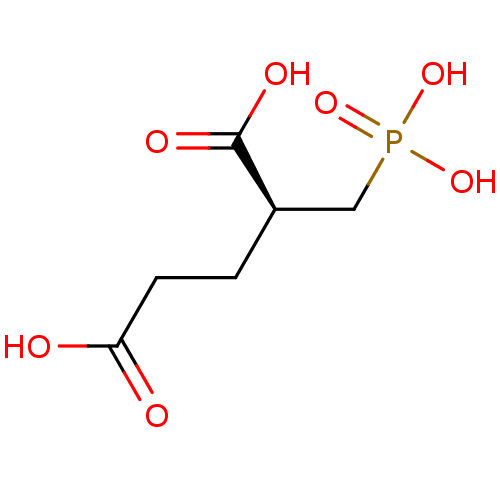
((2S)-2-(phosphonomethyl)pentanedioic acid | (S)-2-...)Show InChI InChI=1S/C6H11O7P/c7-5(8)2-1-4(6(9)10)3-14(11,12)13/h4H,1-3H2,(H,7,8)(H,9,10)(H2,11,12,13)/t4-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare
Curated by ChEMBL
| Assay Description
Binding affinity to NAALADase |
J Med Chem 55: 9510-20 (2012)
Article DOI: 10.1021/jm300710j
BindingDB Entry DOI: 10.7270/Q28053R3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase Mer
(Homo sapiens (Human)) | BDBM497267
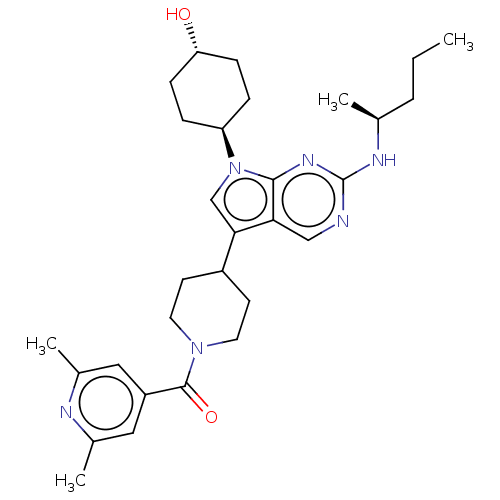
((2,6-dimethylpyridin-4- yl)(4-(7-((1R,4S)-4- hydro...)Show SMILES CCC[C@H](C)Nc1ncc2c(cn([C@H]3CC[C@H](O)CC3)c2n1)C1CCN(CC1)C(=O)c1cc(C)nc(C)c1 |r,wU:13.12,wD:3.3,16.16,(-8.93,-2.23,;-7.59,-1.46,;-6.26,-2.23,;-4.93,-1.46,;-4.93,.08,;-3.59,-2.23,;-2.26,-1.46,;-2.26,.08,;-.93,.85,;.41,.08,;1.87,.56,;2.78,-.69,;1.87,-1.94,;2.35,-3.4,;3.85,-3.72,;4.33,-5.19,;3.3,-6.33,;3.78,-7.79,;1.79,-6.01,;1.32,-4.55,;.41,-1.46,;-.93,-2.23,;2.35,2.02,;3.85,2.34,;4.33,3.81,;3.3,4.95,;1.79,4.63,;1.32,3.16,;3.78,6.41,;3.04,7.77,;5.26,6.02,;6.35,7.1,;7.84,6.71,;8.93,7.79,;8.24,5.22,;7.15,4.13,;7.55,2.64,;5.66,4.53,)| Show InChI InChI=1S/C30H42N6O2/c1-5-6-19(2)33-30-31-17-26-27(18-36(28(26)34-30)24-7-9-25(37)10-8-24)22-11-13-35(14-12-22)29(38)23-15-20(3)32-21(4)16-23/h15-19,22,24-25,37H,5-14H2,1-4H3,(H,31,33,34)/t19-,24-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
ATP competitive inhibition of MERTK (unknown origin) |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.113534
BindingDB Entry DOI: 10.7270/Q2M90DGJ |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50411736
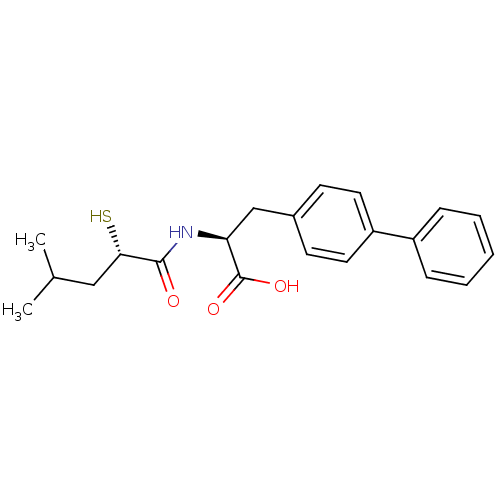
(CHEMBL271225)Show SMILES CC(C)C[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C21H25NO3S/c1-14(2)12-19(26)20(23)22-18(21(24)25)13-15-8-10-17(11-9-15)16-6-4-3-5-7-16/h3-11,14,18-19,26H,12-13H2,1-2H3,(H,22,23)(H,24,25)/t18-,19-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM17659
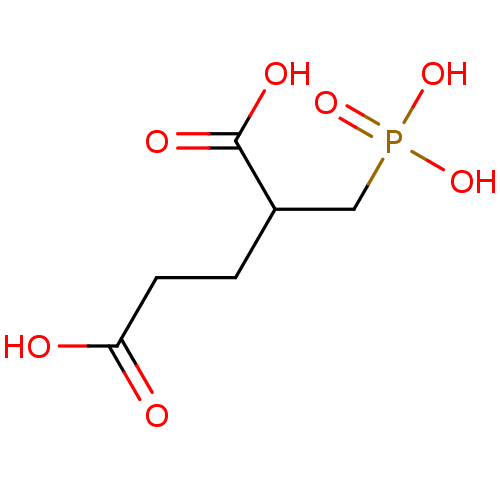
((R,S)-2-phosphonomethylpentanedioic acid | 2-(phos...)Show InChI InChI=1S/C6H11O7P/c7-5(8)2-1-4(6(9)10)3-14(11,12)13/h4H,1-3H2,(H,7,8)(H,9,10)(H2,11,12,13) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare
Curated by ChEMBL
| Assay Description
Binding affinity to NAALADase |
J Med Chem 55: 9510-20 (2012)
Article DOI: 10.1021/jm300710j
BindingDB Entry DOI: 10.7270/Q28053R3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase Mer
(Homo sapiens (Human)) | BDBM50055499

(CHEMBL3326002)Show SMILES CCCCNc1ncc2c(nn([C@H]3CC[C@H](O)CC3)c2n1)-c1ccc(cc1)S(=O)(=O)N1CCOCC1 |r,wU:12.11,wD:15.15,(-2.75,-7.06,;-1.43,-6.29,;-.1,-7.06,;1.25,-6.3,;2.58,-7.07,;3.91,-6.3,;3.91,-4.75,;5.24,-3.99,;6.57,-4.75,;8.05,-4.27,;8.96,-5.52,;8.05,-6.78,;8.53,-8.25,;10.03,-8.56,;10.5,-10.03,;9.47,-11.17,;9.94,-12.64,;7.96,-10.85,;7.49,-9.39,;6.57,-6.3,;5.24,-7.07,;8.53,-2.8,;10.04,-2.49,;10.51,-1.03,;9.49,.13,;7.98,-.2,;7.5,-1.67,;9.97,1.58,;8.48,1.99,;9.57,3.09,;11.48,1.89,;12.49,.74,;13.99,1.03,;14.49,2.48,;13.47,3.64,;11.96,3.35,)| Show InChI InChI=1S/C25H34N6O4S/c1-2-3-12-26-25-27-17-22-23(29-31(24(22)28-25)19-6-8-20(32)9-7-19)18-4-10-21(11-5-18)36(33,34)30-13-15-35-16-14-30/h4-5,10-11,17,19-20,32H,2-3,6-9,12-16H2,1H3,(H,26,27,28)/t19-,20- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by ChEMBL
| Assay Description
Inhibition of Mer (unknown origin) by Off-chip Mobility Shift Assay |
J Med Chem 57: 7031-41 (2014)
Article DOI: 10.1021/jm500749d
BindingDB Entry DOI: 10.7270/Q2K075XQ |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM17659

((R,S)-2-phosphonomethylpentanedioic acid | 2-(phos...)Show InChI InChI=1S/C6H11O7P/c7-5(8)2-1-4(6(9)10)3-14(11,12)13/h4H,1-3H2,(H,7,8)(H,9,10)(H2,11,12,13) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare
Curated by ChEMBL
| Assay Description
Inhibition of NAALADase activity in human LNCaP cell membranes assessed as inhibition of [3H]NAG conversion to [3H]glutamate after 30 mins by liquid ... |
J Med Chem 55: 9510-20 (2012)
Article DOI: 10.1021/jm300710j
BindingDB Entry DOI: 10.7270/Q28053R3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase Mer
(Homo sapiens (Human)) | BDBM50469353
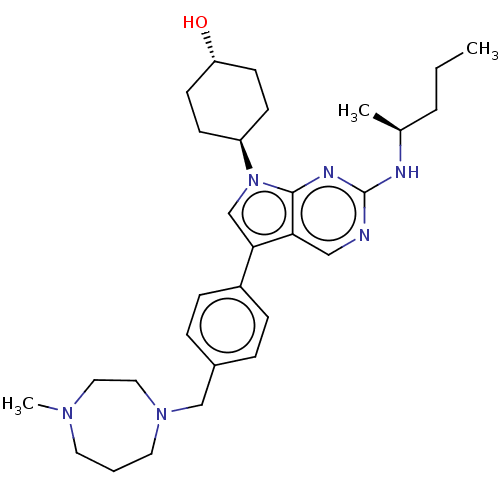
(CHEMBL4283353)Show SMILES CCC[C@H](C)Nc1ncc2c(cn([C@H]3CC[C@H](O)CC3)c2n1)-c1ccc(CN2CCCN(C)CC2)cc1 |r,wU:13.12,wD:16.16,3.3,(17.07,-10.92,;18.4,-10.15,;19.74,-10.92,;21.07,-10.15,;21.07,-8.61,;22.4,-10.92,;23.74,-10.15,;23.74,-8.61,;25.07,-7.84,;26.4,-8.6,;27.88,-8.12,;28.79,-9.38,;27.88,-10.63,;28.63,-11.97,;30.16,-12,;30.91,-13.35,;30.11,-14.67,;30.85,-16.02,;28.57,-14.64,;27.83,-13.29,;26.4,-10.15,;25.07,-10.92,;28.59,-6.76,;30.13,-6.7,;30.84,-5.34,;30.02,-4.03,;30.73,-2.67,;32.27,-2.6,;33,-3.97,;34.51,-4.24,;35.67,-3.22,;35.61,-1.69,;36.96,-.95,;34.36,-.79,;32.87,-1.2,;28.47,-4.1,;27.77,-5.47,)| Show InChI InChI=1S/C30H44N6O/c1-4-6-22(2)32-30-31-19-27-28(21-36(29(27)33-30)25-11-13-26(37)14-12-25)24-9-7-23(8-10-24)20-35-16-5-15-34(3)17-18-35/h7-10,19,21-22,25-26,37H,4-6,11-18,20H2,1-3H3,(H,31,32,33)/t22-,25-,26-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UNC Eshelman School of Pharmacy
Curated by ChEMBL
| Assay Description
Inhibition of MERTK (unknown origin) using 5'-FAM-EFPIYDFLPAKKK-CONH2 as substrate after 180 mins by MCE assay |
J Med Chem 61: 10242-10254 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01229
BindingDB Entry DOI: 10.7270/Q2K076ZK |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Mer
(Homo sapiens (Human)) | BDBM50384584
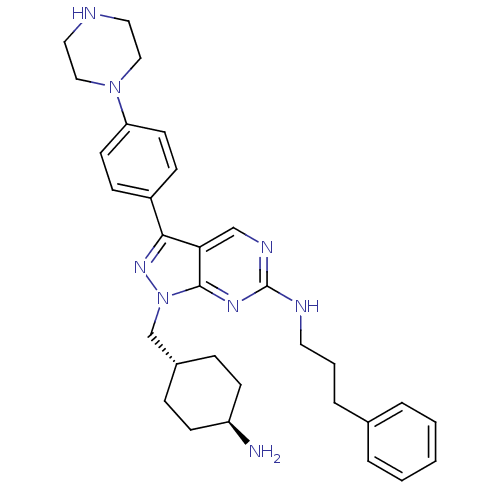
(CHEMBL2036807 | US9744172, Compound UNC607A)Show SMILES N[C@H]1CC[C@H](Cn2nc(-c3ccc(cc3)N3CCNCC3)c3cnc(NCCCc4ccccc4)nc23)CC1 |r,wU:4.4,wD:1.0,(41.81,-28.1,;42.85,-26.96,;44.35,-27.28,;45.39,-26.14,;44.91,-24.68,;45.94,-23.54,;45.47,-22.07,;46.38,-20.82,;45.47,-19.56,;45.94,-18.1,;47.45,-17.78,;47.93,-16.32,;46.9,-15.17,;45.38,-15.5,;44.91,-16.96,;47.36,-13.71,;48.87,-13.39,;49.35,-11.94,;48.32,-10.79,;46.81,-11.11,;46.33,-12.57,;43.99,-20.04,;42.66,-19.28,;41.33,-20.05,;41.33,-21.6,;39.99,-22.36,;38.66,-21.59,;38.66,-20.05,;37.33,-19.28,;37.33,-17.74,;38.67,-16.98,;38.67,-15.44,;37.34,-14.67,;36,-15.45,;36,-16.98,;42.66,-22.37,;43.99,-21.6,;43.41,-24.35,;42.38,-25.49,)| Show InChI InChI=1S/C31H40N8/c32-26-12-8-24(9-13-26)22-39-30-28(21-35-31(36-30)34-16-4-7-23-5-2-1-3-6-23)29(37-39)25-10-14-27(15-11-25)38-19-17-33-18-20-38/h1-3,5-6,10-11,14-15,21,24,26,33H,4,7-9,12-13,16-20,22,32H2,(H,34,35,36)/t24-,26- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Mer using EFPIYDFLPAKKK-CONH2 as substrate by Michaelis-Menton equation |
ACS Med Chem Lett 3: 129-134 (2012)
Article DOI: 10.1021/ml200239k
BindingDB Entry DOI: 10.7270/Q2F76DMC |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Mer
(Homo sapiens (Human)) | BDBM50384583
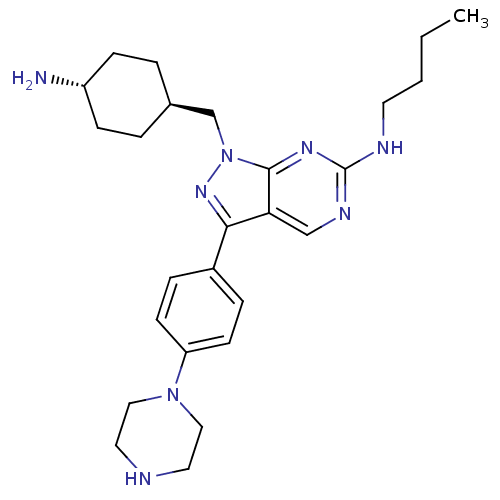
(CHEMBL2036806)Show SMILES CCCCNc1ncc2c(nn(C[C@H]3CC[C@H](N)CC3)c2n1)-c1ccc(cc1)N1CCNCC1 |r,wU:13.12,wD:16.16,(19.9,-18.03,;19.9,-19.57,;21.23,-20.34,;21.23,-21.88,;22.56,-22.65,;23.9,-21.88,;23.9,-20.34,;25.23,-19.57,;26.56,-20.33,;28.04,-19.85,;28.95,-21.11,;28.04,-22.36,;28.51,-23.83,;27.48,-24.97,;27.96,-26.43,;26.92,-27.57,;25.42,-27.25,;24.38,-28.39,;24.95,-25.78,;25.98,-24.64,;26.56,-21.88,;25.23,-22.65,;28.51,-18.39,;30.02,-18.07,;30.5,-16.61,;29.47,-15.46,;27.95,-15.79,;27.48,-17.25,;29.93,-14,;31.44,-13.68,;31.92,-12.22,;30.89,-11.08,;29.38,-11.39,;28.9,-12.86,)| Show InChI InChI=1S/C26H38N8/c1-2-3-12-29-26-30-17-23-24(20-6-10-22(11-7-20)33-15-13-28-14-16-33)32-34(25(23)31-26)18-19-4-8-21(27)9-5-19/h6-7,10-11,17,19,21,28H,2-5,8-9,12-16,18,27H2,1H3,(H,29,30,31)/t19-,21- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Mer using EFPIYDFLPAKKK-CONH2 as substrate by Michaelis-Menton equation |
ACS Med Chem Lett 3: 129-134 (2012)
Article DOI: 10.1021/ml200239k
BindingDB Entry DOI: 10.7270/Q2F76DMC |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50411731
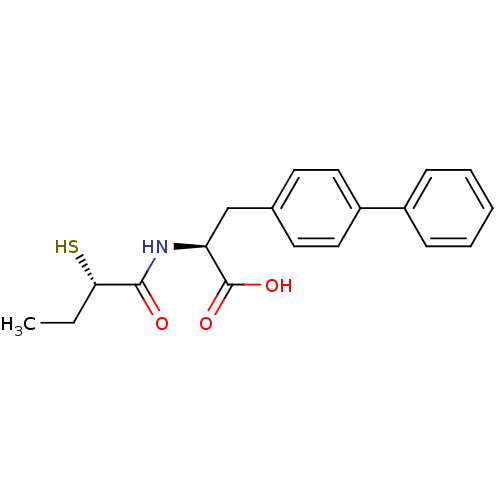
(CHEMBL257726)Show SMILES CC[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C19H21NO3S/c1-2-17(24)18(21)20-16(19(22)23)12-13-8-10-15(11-9-13)14-6-4-3-5-7-14/h3-11,16-17,24H,2,12H2,1H3,(H,20,21)(H,22,23)/t16-,17-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50286715
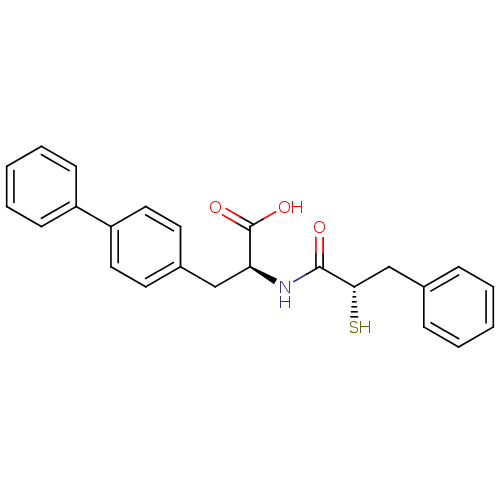
((S)-3-biphenyl-4-yl-2-((S)-2-mercapto-3-phenyl-pro...)Show SMILES OC(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC(=O)[C@@H](S)Cc1ccccc1 Show InChI InChI=1S/C24H23NO3S/c26-23(22(29)16-17-7-3-1-4-8-17)25-21(24(27)28)15-18-11-13-20(14-12-18)19-9-5-2-6-10-19/h1-14,21-22,29H,15-16H2,(H,25,26)(H,27,28)/t21-,22-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50411730
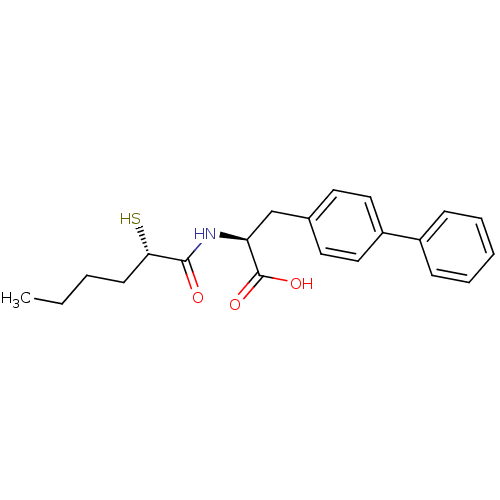
(CHEMBL257270)Show SMILES CCCC[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C21H25NO3S/c1-2-3-9-19(26)20(23)22-18(21(24)25)14-15-10-12-17(13-11-15)16-7-5-4-6-8-16/h4-8,10-13,18-19,26H,2-3,9,14H2,1H3,(H,22,23)(H,24,25)/t18-,19-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50286724
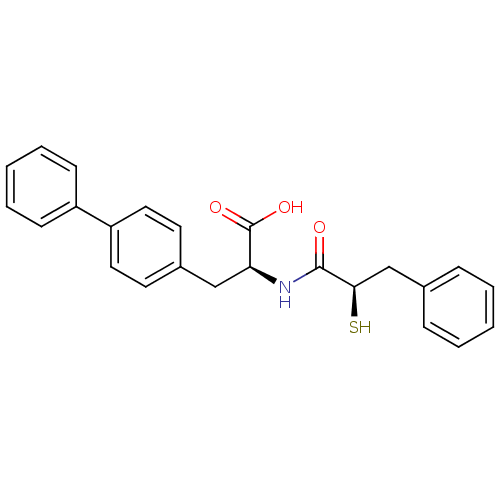
((S)-3-Biphenyl-4-yl-2-((R)-2-mercapto-3-phenyl-pro...)Show SMILES OC(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC(=O)[C@H](S)Cc1ccccc1 Show InChI InChI=1S/C24H23NO3S/c26-23(22(29)16-17-7-3-1-4-8-17)25-21(24(27)28)15-18-11-13-20(14-12-18)19-9-5-2-6-10-19/h1-14,21-22,29H,15-16H2,(H,25,26)(H,27,28)/t21-,22+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50411736

(CHEMBL271225)Show SMILES CC(C)C[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C21H25NO3S/c1-14(2)12-19(26)20(23)22-18(21(24)25)13-15-8-10-17(11-9-15)16-6-4-3-5-7-16/h3-11,14,18-19,26H,12-13H2,1-2H3,(H,22,23)(H,24,25)/t18-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE2 by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50411731

(CHEMBL257726)Show SMILES CC[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C19H21NO3S/c1-2-17(24)18(21)20-16(19(22)23)12-13-8-10-15(11-9-13)14-6-4-3-5-7-14/h3-11,16-17,24H,2,12H2,1H3,(H,20,21)(H,22,23)/t16-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE2 by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Mer
(Homo sapiens (Human)) | BDBM50384582
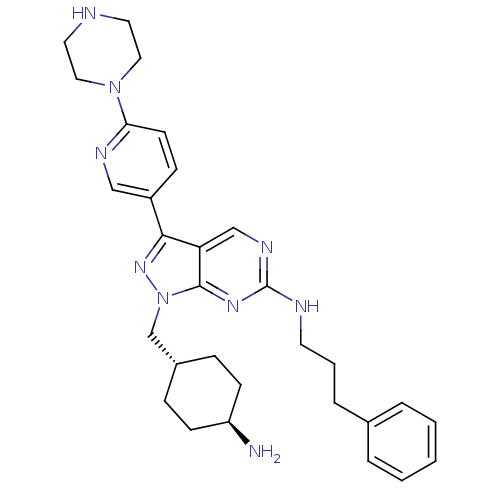
(CHEMBL2036805)Show SMILES N[C@H]1CC[C@H](Cn2nc(-c3ccc(nc3)N3CCNCC3)c3cnc(NCCCc4ccccc4)nc23)CC1 |r,wU:4.4,wD:1.0,(11.21,-28.56,;12.24,-27.41,;13.75,-27.74,;14.79,-26.59,;14.31,-25.14,;15.34,-24,;14.86,-22.53,;15.77,-21.28,;14.86,-20.02,;15.34,-18.56,;14.31,-17.42,;14.78,-15.96,;16.29,-15.63,;17.32,-16.78,;16.84,-18.24,;16.76,-14.17,;18.27,-13.85,;18.74,-12.39,;17.71,-11.25,;16.21,-11.56,;15.72,-13.03,;13.39,-20.5,;12.05,-19.74,;10.72,-20.51,;10.72,-22.05,;9.39,-22.82,;8.05,-22.05,;8.05,-20.51,;6.72,-19.74,;6.72,-18.2,;8.06,-17.44,;8.06,-15.9,;6.73,-15.13,;5.39,-15.9,;5.4,-17.44,;12.05,-22.82,;13.39,-22.05,;12.8,-24.81,;11.77,-25.95,)| Show InChI InChI=1S/C30H39N9/c31-25-11-8-23(9-12-25)21-39-29-26(20-35-30(36-29)33-14-4-7-22-5-2-1-3-6-22)28(37-39)24-10-13-27(34-19-24)38-17-15-32-16-18-38/h1-3,5-6,10,13,19-20,23,25,32H,4,7-9,11-12,14-18,21,31H2,(H,33,35,36)/t23-,25- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Mer using EFPIYDFLPAKKK-CONH2 as substrate by Michaelis-Menton equation |
ACS Med Chem Lett 3: 129-134 (2012)
Article DOI: 10.1021/ml200239k
BindingDB Entry DOI: 10.7270/Q2F76DMC |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50398811
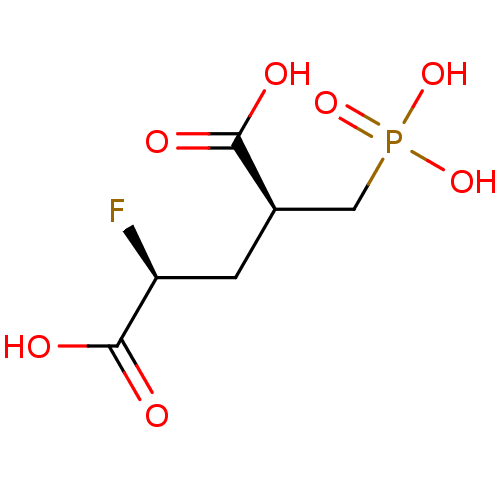
(CHEMBL2177507)Show SMILES OC(=O)[C@@H](F)C[C@H](CP(O)(O)=O)C(O)=O |r| Show InChI InChI=1S/C6H10FO7P/c7-4(6(10)11)1-3(5(8)9)2-15(12,13)14/h3-4H,1-2H2,(H,8,9)(H,10,11)(H2,12,13,14)/t3-,4+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare
Curated by ChEMBL
| Assay Description
Inhibition of NAALADase activity in human LNCaP cell membranes assessed as inhibition of [3H]NAG conversion to [3H]glutamate after 30 mins by liquid ... |
J Med Chem 55: 9510-20 (2012)
Article DOI: 10.1021/jm300710j
BindingDB Entry DOI: 10.7270/Q28053R3 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50411733
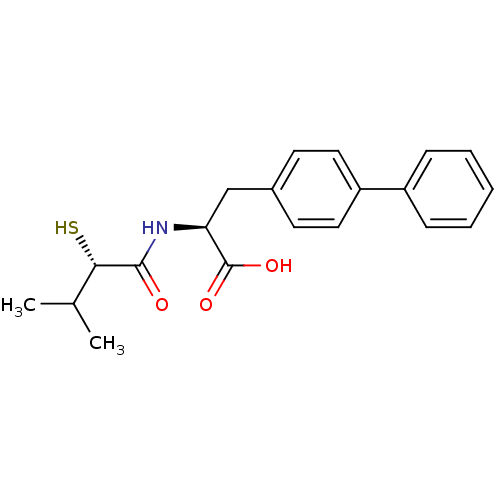
(CHEMBL269997)Show SMILES CC(C)[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C20H23NO3S/c1-13(2)18(25)19(22)21-17(20(23)24)12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,13,17-18,25H,12H2,1-2H3,(H,21,22)(H,23,24)/t17-,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE2 by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50411605
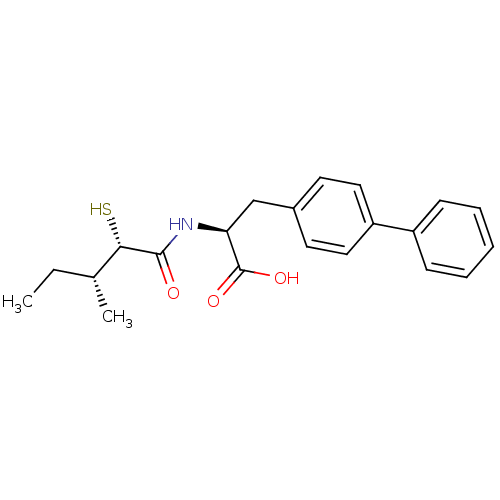
(CHEMBL252391)Show SMILES CC[C@@H](C)[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C21H25NO3S/c1-3-14(2)19(26)20(23)22-18(21(24)25)13-15-9-11-17(12-10-15)16-7-5-4-6-8-16/h4-12,14,18-19,26H,3,13H2,1-2H3,(H,22,23)(H,24,25)/t14-,18+,19+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE2 by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50411729
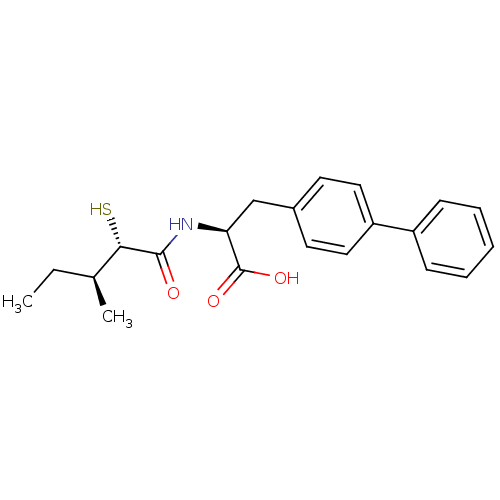
(CHEMBL269996)Show SMILES CC[C@H](C)[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C21H25NO3S/c1-3-14(2)19(26)20(23)22-18(21(24)25)13-15-9-11-17(12-10-15)16-7-5-4-6-8-16/h4-12,14,18-19,26H,3,13H2,1-2H3,(H,22,23)(H,24,25)/t14-,18-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE2 by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50411737
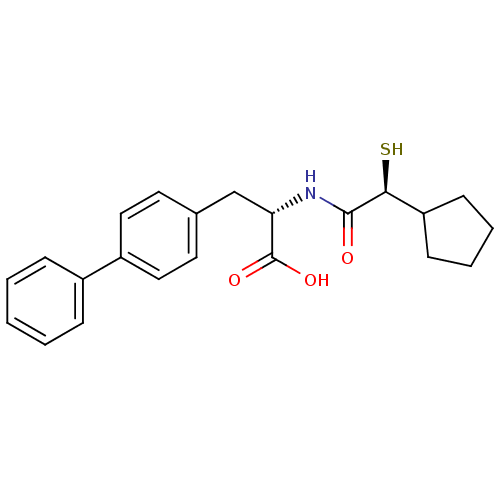
(CHEMBL404117)Show SMILES OC(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC(=O)[C@@H](S)C1CCCC1 Show InChI InChI=1S/C22H25NO3S/c24-21(20(27)18-8-4-5-9-18)23-19(22(25)26)14-15-10-12-17(13-11-15)16-6-2-1-3-7-16/h1-3,6-7,10-13,18-20,27H,4-5,8-9,14H2,(H,23,24)(H,25,26)/t19-,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE2 by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50411730

(CHEMBL257270)Show SMILES CCCC[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C21H25NO3S/c1-2-3-9-19(26)20(23)22-18(21(24)25)14-15-10-12-17(13-11-15)16-7-5-4-6-8-16/h4-8,10-13,18-19,26H,2-3,9,14H2,1H3,(H,22,23)(H,24,25)/t18-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE2 by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Mer
(Homo sapiens (Human)) | BDBM50384585
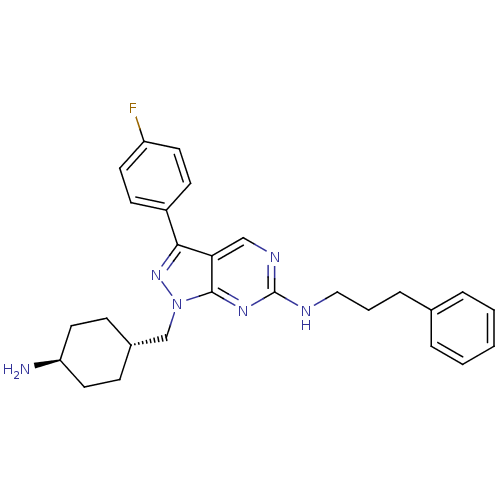
(CHEMBL2036809)Show SMILES N[C@H]1CC[C@H](Cn2nc(-c3ccc(F)cc3)c3cnc(NCCCc4ccccc4)nc23)CC1 |r,wU:4.4,wD:1.0,(-.82,-49.25,;.22,-48.11,;1.72,-48.43,;2.76,-47.29,;2.28,-45.83,;3.31,-44.69,;2.84,-43.22,;3.75,-41.97,;2.84,-40.71,;3.31,-39.25,;4.82,-38.93,;5.3,-37.47,;4.27,-36.32,;4.74,-34.86,;2.75,-36.65,;2.28,-38.11,;1.36,-41.19,;.03,-40.43,;-1.3,-41.2,;-1.3,-42.74,;-2.64,-43.51,;-3.97,-42.74,;-3.97,-41.2,;-5.3,-40.43,;-5.3,-38.89,;-3.96,-38.13,;-3.96,-36.59,;-5.29,-35.82,;-6.63,-36.59,;-6.63,-38.13,;.03,-43.51,;1.36,-42.74,;.78,-45.5,;-.25,-46.64,)| Show InChI InChI=1S/C27H31FN6/c28-22-12-10-21(11-13-22)25-24-17-31-27(30-16-4-7-19-5-2-1-3-6-19)32-26(24)34(33-25)18-20-8-14-23(29)15-9-20/h1-3,5-6,10-13,17,20,23H,4,7-9,14-16,18,29H2,(H,30,31,32)/t20-,23- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Mer using EFPIYDFLPAKKK-CONH2 as substrate by Michaelis-Menton equation |
ACS Med Chem Lett 3: 129-134 (2012)
Article DOI: 10.1021/ml200239k
BindingDB Entry DOI: 10.7270/Q2F76DMC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50411728
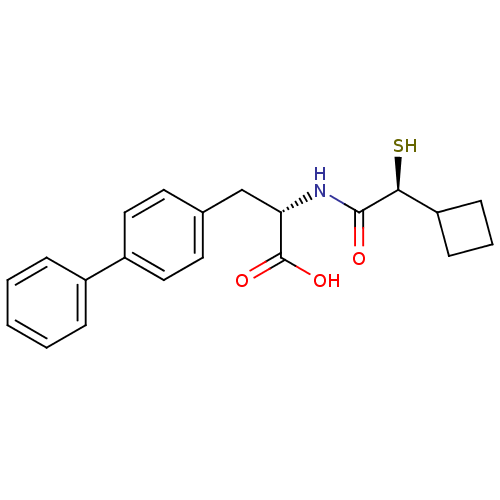
(CHEMBL257229)Show SMILES OC(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC(=O)[C@@H](S)C1CCC1 Show InChI InChI=1S/C21H23NO3S/c23-20(19(26)17-7-4-8-17)22-18(21(24)25)13-14-9-11-16(12-10-14)15-5-2-1-3-6-15/h1-3,5-6,9-12,17-19,26H,4,7-8,13H2,(H,22,23)(H,24,25)/t18-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE2 by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50411729

(CHEMBL269996)Show SMILES CC[C@H](C)[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C21H25NO3S/c1-3-14(2)19(26)20(23)22-18(21(24)25)13-15-9-11-17(12-10-15)16-7-5-4-6-8-16/h4-12,14,18-19,26H,3,13H2,1-2H3,(H,22,23)(H,24,25)/t14-,18-,19-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50411725
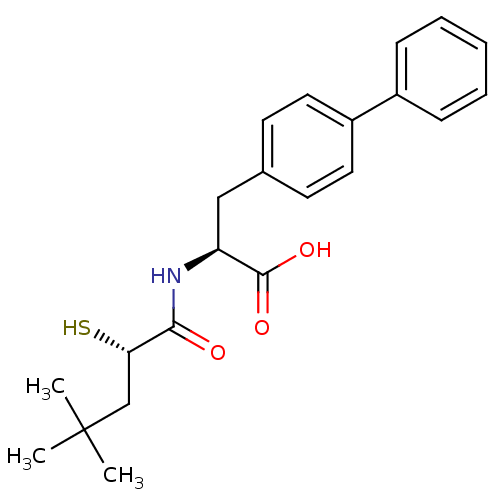
(CHEMBL271223)Show SMILES CC(C)(C)C[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C22H27NO3S/c1-22(2,3)14-19(27)20(24)23-18(21(25)26)13-15-9-11-17(12-10-15)16-7-5-4-6-8-16/h4-12,18-19,27H,13-14H2,1-3H3,(H,23,24)(H,25,26)/t18-,19-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50398808
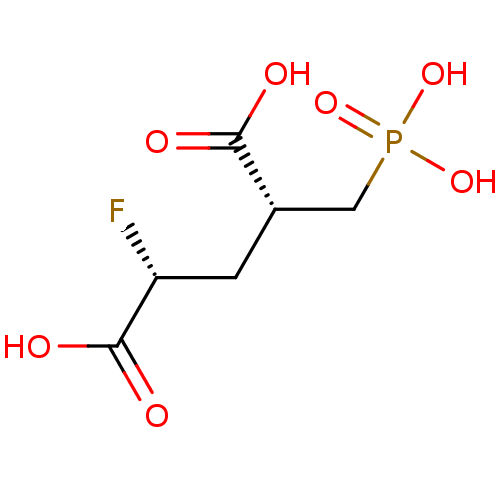
(CHEMBL2177506)Show SMILES OC(=O)[C@H](F)C[C@@H](CP(O)(O)=O)C(O)=O |r| Show InChI InChI=1S/C6H10FO7P/c7-4(6(10)11)1-3(5(8)9)2-15(12,13)14/h3-4H,1-2H2,(H,8,9)(H,10,11)(H2,12,13,14)/t3-,4+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare
Curated by ChEMBL
| Assay Description
Inhibition of NAALADase activity in human LNCaP cell membranes assessed as inhibition of [3H]NAG conversion to [3H]glutamate after 30 mins by liquid ... |
J Med Chem 55: 9510-20 (2012)
Article DOI: 10.1021/jm300710j
BindingDB Entry DOI: 10.7270/Q28053R3 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Neprilysin
(Homo sapiens (Human)) | BDBM50411735
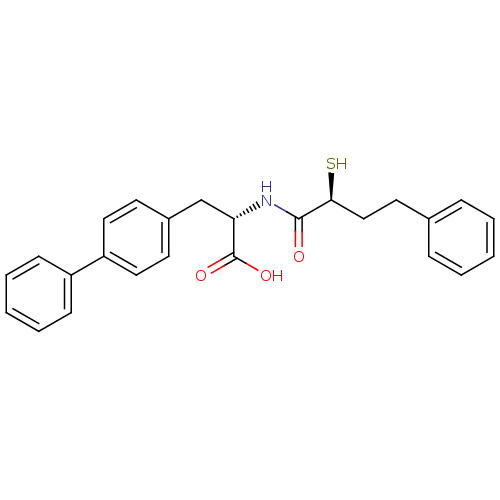
(CHEMBL402987)Show SMILES OC(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC(=O)[C@@H](S)CCc1ccccc1 Show InChI InChI=1S/C25H25NO3S/c27-24(23(30)16-13-18-7-3-1-4-8-18)26-22(25(28)29)17-19-11-14-21(15-12-19)20-9-5-2-6-10-20/h1-12,14-15,22-23,30H,13,16-17H2,(H,26,27)(H,28,29)/t22-,23-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Mer
(Homo sapiens (Human)) | BDBM50384581
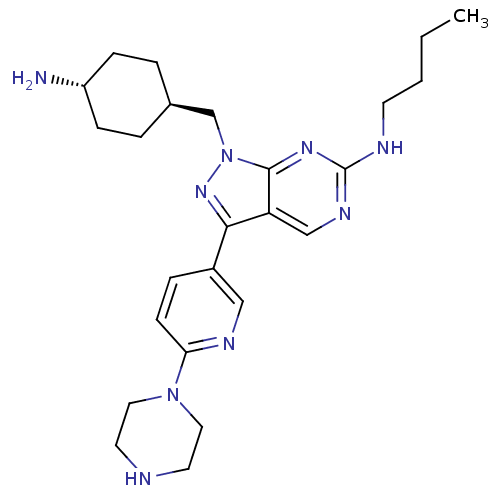
(CHEMBL2036804)Show SMILES CCCCNc1ncc2c(nn(C[C@H]3CC[C@H](N)CC3)c2n1)-c1ccc(nc1)N1CCNCC1 |r,wU:13.12,wD:16.16,(-8.83,-18.78,;-8.83,-20.32,;-7.5,-21.09,;-7.5,-22.63,;-6.17,-23.41,;-4.83,-22.64,;-4.83,-21.09,;-3.5,-20.32,;-2.17,-21.08,;-.69,-20.61,;.22,-21.86,;-.69,-23.12,;-.22,-24.58,;-1.25,-25.72,;-.77,-27.18,;-1.81,-28.32,;-3.31,-28,;-4.35,-29.14,;-3.78,-26.53,;-2.76,-25.39,;-2.17,-22.64,;-3.5,-23.41,;-.22,-19.14,;-1.25,-18.01,;-.78,-16.54,;.73,-16.22,;1.77,-17.36,;1.29,-18.82,;1.2,-14.75,;2.71,-14.44,;3.19,-12.98,;2.16,-11.83,;.65,-12.15,;.17,-13.62,)| Show InChI InChI=1S/C25H37N9/c1-2-3-10-28-25-30-16-21-23(19-6-9-22(29-15-19)33-13-11-27-12-14-33)32-34(24(21)31-25)17-18-4-7-20(26)8-5-18/h6,9,15-16,18,20,27H,2-5,7-8,10-14,17,26H2,1H3,(H,28,30,31)/t18-,20- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Mer using EFPIYDFLPAKKK-CONH2 as substrate by Michaelis-Menton equation |
ACS Med Chem Lett 3: 129-134 (2012)
Article DOI: 10.1021/ml200239k
BindingDB Entry DOI: 10.7270/Q2F76DMC |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50398810
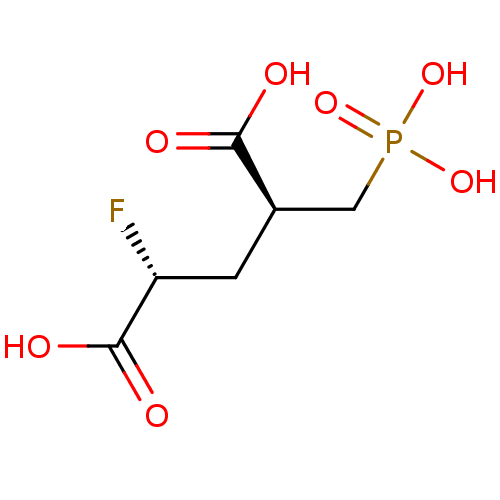
(CHEMBL2177509)Show InChI InChI=1S/C6H10FO7P/c7-4(6(10)11)1-3(5(8)9)2-15(12,13)14/h3-4H,1-2H2,(H,8,9)(H,10,11)(H2,12,13,14)/t3-,4-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare
Curated by ChEMBL
| Assay Description
Inhibition of NAALADase activity in human LNCaP cell membranes assessed as inhibition of [3H]NAG conversion to [3H]glutamate after 30 mins by liquid ... |
J Med Chem 55: 9510-20 (2012)
Article DOI: 10.1021/jm300710j
BindingDB Entry DOI: 10.7270/Q28053R3 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50411736

(CHEMBL271225)Show SMILES CC(C)C[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C21H25NO3S/c1-14(2)12-19(26)20(23)22-18(21(24)25)13-15-8-10-17(11-9-15)16-6-4-3-5-7-16/h3-11,14,18-19,26H,12-13H2,1-2H3,(H,22,23)(H,24,25)/t18-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase receptor TYRO3
(Homo sapiens (Human)) | BDBM50469353

(CHEMBL4283353)Show SMILES CCC[C@H](C)Nc1ncc2c(cn([C@H]3CC[C@H](O)CC3)c2n1)-c1ccc(CN2CCCN(C)CC2)cc1 |r,wU:13.12,wD:16.16,3.3,(17.07,-10.92,;18.4,-10.15,;19.74,-10.92,;21.07,-10.15,;21.07,-8.61,;22.4,-10.92,;23.74,-10.15,;23.74,-8.61,;25.07,-7.84,;26.4,-8.6,;27.88,-8.12,;28.79,-9.38,;27.88,-10.63,;28.63,-11.97,;30.16,-12,;30.91,-13.35,;30.11,-14.67,;30.85,-16.02,;28.57,-14.64,;27.83,-13.29,;26.4,-10.15,;25.07,-10.92,;28.59,-6.76,;30.13,-6.7,;30.84,-5.34,;30.02,-4.03,;30.73,-2.67,;32.27,-2.6,;33,-3.97,;34.51,-4.24,;35.67,-3.22,;35.61,-1.69,;36.96,-.95,;34.36,-.79,;32.87,-1.2,;28.47,-4.1,;27.77,-5.47,)| Show InChI InChI=1S/C30H44N6O/c1-4-6-22(2)32-30-31-19-27-28(21-36(29(27)33-30)25-11-13-26(37)14-12-25)24-9-7-23(8-10-24)20-35-16-5-15-34(3)17-18-35/h7-10,19,21-22,25-26,37H,4-6,11-18,20H2,1-3H3,(H,31,32,33)/t22-,25-,26-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 3.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UNC Eshelman School of Pharmacy
Curated by ChEMBL
| Assay Description
Inhibition of TYRO3 (unknown origin) using 5'-FAM-EFPIYDFLPAKKK-CONH2 as substrate after 180 mins by MCE assay |
J Med Chem 61: 10242-10254 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01229
BindingDB Entry DOI: 10.7270/Q2K076ZK |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Mer
(Homo sapiens (Human)) | BDBM50384576

(CHEMBL2036808)Show SMILES CCCCNc1ncc2c(nn(C[C@H]3CC[C@H](N)CC3)c2n1)-c1ccc(F)cc1 |r,wU:13.12,wD:16.16,(54.06,-16.98,;54.06,-18.52,;55.39,-19.29,;55.39,-20.83,;56.72,-21.6,;58.06,-20.83,;58.06,-19.29,;59.39,-18.52,;60.72,-19.28,;62.2,-18.8,;63.11,-20.06,;62.2,-21.31,;62.67,-22.78,;61.64,-23.92,;62.12,-25.38,;61.08,-26.52,;59.58,-26.2,;58.54,-27.34,;59.11,-24.73,;60.14,-23.59,;60.72,-20.83,;59.39,-21.6,;62.67,-17.34,;64.18,-17.02,;64.66,-15.56,;63.63,-14.41,;64.1,-12.95,;62.11,-14.74,;61.64,-16.2,)| Show InChI InChI=1S/C22H29FN6/c1-2-3-12-25-22-26-13-19-20(16-6-8-17(23)9-7-16)28-29(21(19)27-22)14-15-4-10-18(24)11-5-15/h6-9,13,15,18H,2-5,10-12,14,24H2,1H3,(H,25,26,27)/t15-,18- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 4.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Mer using EFPIYDFLPAKKK-CONH2 as substrate by Michaelis-Menton equation |
ACS Med Chem Lett 3: 129-134 (2012)
Article DOI: 10.1021/ml200239k
BindingDB Entry DOI: 10.7270/Q2F76DMC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50304731
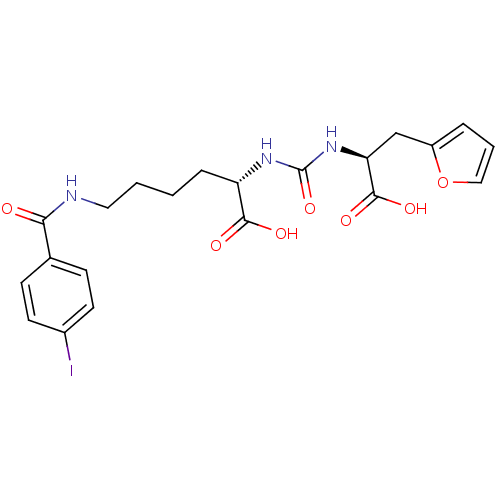
((S)-2-(3-((S)-1-Carboxy-2-(furan-2-yl)ethyl)ureido...)Show SMILES OC(=O)[C@H](CCCCNC(=O)c1ccc(I)cc1)NC(=O)N[C@@H](Cc1ccco1)C(O)=O |r| Show InChI InChI=1S/C21H24IN3O7/c22-14-8-6-13(7-9-14)18(26)23-10-2-1-5-16(19(27)28)24-21(31)25-17(20(29)30)12-15-4-3-11-32-15/h3-4,6-9,11,16-17H,1-2,5,10,12H2,(H,23,26)(H,27,28)(H,29,30)(H2,24,25,31)/t16-,17-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 5.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare
Curated by ChEMBL
| Assay Description
Binding affinity to NAALADase |
J Med Chem 55: 9510-20 (2012)
Article DOI: 10.1021/jm300710j
BindingDB Entry DOI: 10.7270/Q28053R3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50304722
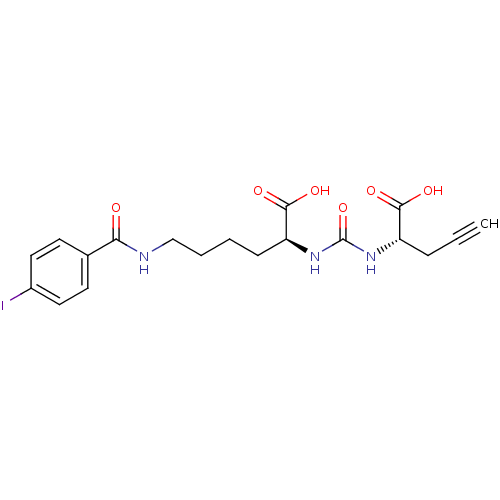
((S)-2-(3-((S)-1-Carboxybut-3-ynyl)ureido)-6-(4-iod...)Show SMILES OC(=O)[C@H](CCCCNC(=O)c1ccc(I)cc1)NC(=O)N[C@@H](CC#C)C(O)=O |r| Show InChI InChI=1S/C19H22IN3O6/c1-2-5-14(17(25)26)22-19(29)23-15(18(27)28)6-3-4-11-21-16(24)12-7-9-13(20)10-8-12/h1,7-10,14-15H,3-6,11H2,(H,21,24)(H,25,26)(H,27,28)(H2,22,23,29)/t14-,15-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 5.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare
Curated by ChEMBL
| Assay Description
Binding affinity to NAALADase |
J Med Chem 55: 9510-20 (2012)
Article DOI: 10.1021/jm300710j
BindingDB Entry DOI: 10.7270/Q28053R3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neprilysin
(Homo sapiens (Human)) | BDBM50411728

(CHEMBL257229)Show SMILES OC(=O)[C@H](Cc1ccc(cc1)-c1ccccc1)NC(=O)[C@@H](S)C1CCC1 Show InChI InChI=1S/C21H23NO3S/c23-20(19(26)17-7-4-8-17)22-18(21(24)25)13-14-9-11-16(12-10-14)15-5-2-1-3-6-15/h1-3,5-6,9-12,17-19,26H,4,7-8,13H2,(H,22,23)(H,24,25)/t18-,19-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50411734

(CHEMBL257727)Show SMILES C[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C18H19NO3S/c1-12(23)17(20)19-16(18(21)22)11-13-7-9-15(10-8-13)14-5-3-2-4-6-14/h2-10,12,16,23H,11H2,1H3,(H,19,20)(H,21,22)/t12-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE2 by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50411725

(CHEMBL271223)Show SMILES CC(C)(C)C[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C22H27NO3S/c1-22(2,3)14-19(27)20(24)23-18(21(25)26)13-15-9-11-17(12-10-15)16-7-5-4-6-8-16/h4-12,18-19,27H,13-14H2,1-3H3,(H,23,24)(H,25,26)/t18-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE2 by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50102258
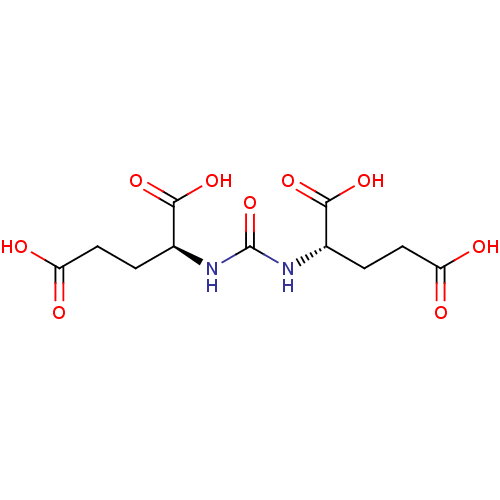
((S)-2-[3-((S)-3-Carboxy-1-carboxy-propyl)-ureido]-...)Show SMILES OC(=O)CC[C@H](NC(=O)N[C@@H](CCC(O)=O)C(O)=O)C(O)=O Show InChI InChI=1S/C11H16N2O9/c14-7(15)3-1-5(9(18)19)12-11(22)13-6(10(20)21)2-4-8(16)17/h5-6H,1-4H2,(H,14,15)(H,16,17)(H,18,19)(H,20,21)(H2,12,13,22)/t5-,6-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare
Curated by ChEMBL
| Assay Description
Binding affinity to NAALADase |
J Med Chem 55: 9510-20 (2012)
Article DOI: 10.1021/jm300710j
BindingDB Entry DOI: 10.7270/Q28053R3 |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50411731

(CHEMBL257726)Show SMILES CC[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C19H21NO3S/c1-2-17(24)18(21)20-16(19(22)23)12-13-8-10-15(11-9-13)14-6-4-3-5-7-14/h3-11,16-17,24H,2,12H2,1H3,(H,20,21)(H,22,23)/t16-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50411730

(CHEMBL257270)Show SMILES CCCC[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C21H25NO3S/c1-2-3-9-19(26)20(23)22-18(21(24)25)14-15-10-12-17(13-11-15)16-7-5-4-6-8-16/h4-8,10-13,18-19,26H,2-3,9,14H2,1H3,(H,22,23)(H,24,25)/t18-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50411606
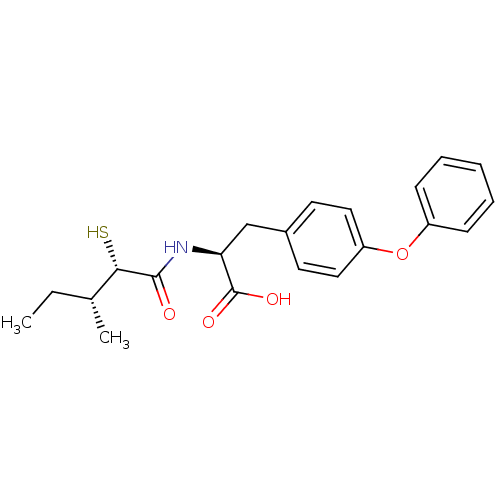
(CHEMBL254282)Show SMILES CC[C@@H](C)[C@H](S)C(=O)N[C@@H](Cc1ccc(Oc2ccccc2)cc1)C(O)=O Show InChI InChI=1S/C21H25NO4S/c1-3-14(2)19(27)20(23)22-18(21(24)25)13-15-9-11-17(12-10-15)26-16-7-5-4-6-8-16/h4-12,14,18-19,27H,3,13H2,1-2H3,(H,22,23)(H,24,25)/t14-,18+,19+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 1681-7 (2008)
Article DOI: 10.1016/j.bmcl.2008.01.046
BindingDB Entry DOI: 10.7270/Q29K49Z2 |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50304751
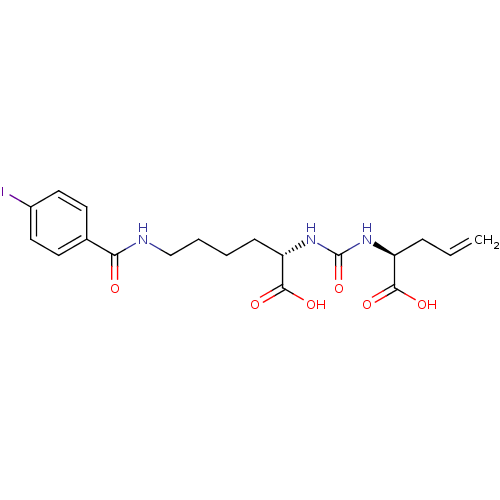
((S)-2-(3-((S)-1-Carboxybut-3-enyl)ureido)-6-(4-iod...)Show SMILES OC(=O)[C@H](CCCCNC(=O)c1ccc(I)cc1)NC(=O)N[C@@H](CC=C)C(O)=O |r| Show InChI InChI=1S/C19H24IN3O6/c1-2-5-14(17(25)26)22-19(29)23-15(18(27)28)6-3-4-11-21-16(24)12-7-9-13(20)10-8-12/h2,7-10,14-15H,1,3-6,11H2,(H,21,24)(H,25,26)(H,27,28)(H2,22,23,29)/t14-,15-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare
Curated by ChEMBL
| Assay Description
Binding affinity to NAALADase |
J Med Chem 55: 9510-20 (2012)
Article DOI: 10.1021/jm300710j
BindingDB Entry DOI: 10.7270/Q28053R3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neprilysin
(Homo sapiens (Human)) | BDBM50411608

(CHEMBL400527)Show SMILES CC[C@@H](C)[C@H](S)C(=O)N[C@@H](Cc1cccc2ccccc12)C(O)=O Show InChI InChI=1S/C19H23NO3S/c1-3-12(2)17(24)18(21)20-16(19(22)23)11-14-9-6-8-13-7-4-5-10-15(13)14/h4-10,12,16-17,24H,3,11H2,1-2H3,(H,20,21)(H,22,23)/t12-,16+,17+/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 1681-7 (2008)
Article DOI: 10.1016/j.bmcl.2008.01.046
BindingDB Entry DOI: 10.7270/Q29K49Z2 |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50411722
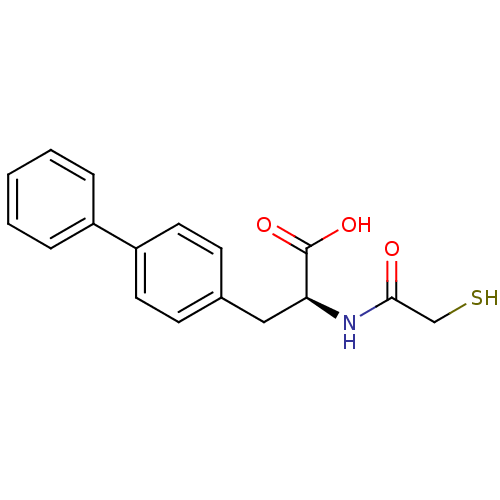
(CHEMBL258333)Show InChI InChI=1S/C17H17NO3S/c19-16(11-22)18-15(17(20)21)10-12-6-8-14(9-7-12)13-4-2-1-3-5-13/h1-9,15,22H,10-11H2,(H,18,19)(H,20,21)/t15-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Neprilysin
(Homo sapiens (Human)) | BDBM50411733

(CHEMBL269997)Show SMILES CC(C)[C@H](S)C(=O)N[C@@H](Cc1ccc(cc1)-c1ccccc1)C(O)=O Show InChI InChI=1S/C20H23NO3S/c1-13(2)18(25)19(22)21-17(20(23)24)12-14-8-10-16(11-9-14)15-6-4-3-5-7-15/h3-11,13,17-18,25H,12H2,1-2H3,(H,21,22)(H,23,24)/t17-,18-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant NEP by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50411722

(CHEMBL258333)Show InChI InChI=1S/C17H17NO3S/c19-16(11-22)18-15(17(20)21)10-12-6-8-14(9-7-12)13-4-2-1-3-5-13/h1-9,15,22H,10-11H2,(H,18,19)(H,20,21)/t15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACE by fluorescence assay |
Bioorg Med Chem Lett 18: 732-7 (2008)
Article DOI: 10.1016/j.bmcl.2007.11.048
BindingDB Entry DOI: 10.7270/Q2GT5PCB |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50469353

(CHEMBL4283353)Show SMILES CCC[C@H](C)Nc1ncc2c(cn([C@H]3CC[C@H](O)CC3)c2n1)-c1ccc(CN2CCCN(C)CC2)cc1 |r,wU:13.12,wD:16.16,3.3,(17.07,-10.92,;18.4,-10.15,;19.74,-10.92,;21.07,-10.15,;21.07,-8.61,;22.4,-10.92,;23.74,-10.15,;23.74,-8.61,;25.07,-7.84,;26.4,-8.6,;27.88,-8.12,;28.79,-9.38,;27.88,-10.63,;28.63,-11.97,;30.16,-12,;30.91,-13.35,;30.11,-14.67,;30.85,-16.02,;28.57,-14.64,;27.83,-13.29,;26.4,-10.15,;25.07,-10.92,;28.59,-6.76,;30.13,-6.7,;30.84,-5.34,;30.02,-4.03,;30.73,-2.67,;32.27,-2.6,;33,-3.97,;34.51,-4.24,;35.67,-3.22,;35.61,-1.69,;36.96,-.95,;34.36,-.79,;32.87,-1.2,;28.47,-4.1,;27.77,-5.47,)| Show InChI InChI=1S/C30H44N6O/c1-4-6-22(2)32-30-31-19-27-28(21-36(29(27)33-30)25-11-13-26(37)14-12-25)24-9-7-23(8-10-24)20-35-16-5-15-34(3)17-18-35/h7-10,19,21-22,25-26,37H,4-6,11-18,20H2,1-3H3,(H,31,32,33)/t22-,25-,26-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
UNC Eshelman School of Pharmacy
Curated by ChEMBL
| Assay Description
Inhibition of FLT3 (unknown origin) using 5'-FAM-KKKKEEIYFFF-CONH2 as substrate after 180 mins by MCE assay |
J Med Chem 61: 10242-10254 (2018)
Article DOI: 10.1021/acs.jmedchem.8b01229
BindingDB Entry DOI: 10.7270/Q2K076ZK |
More data for this
Ligand-Target Pair | |
Glutamate carboxypeptidase 2
(Homo sapiens (Human)) | BDBM50304735
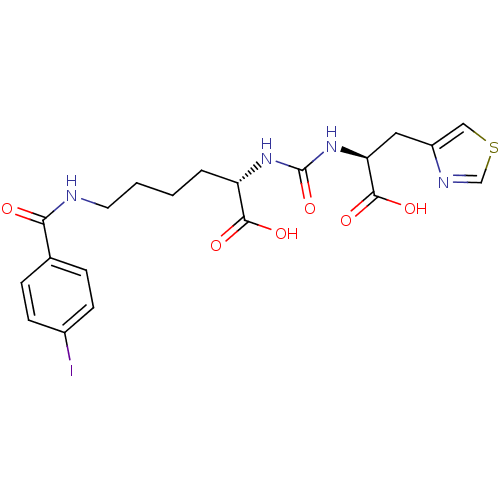
((S)-2-(3-((S)-1-Carboxy-2-(thiazol-4-yl)ethyl)urei...)Show SMILES OC(=O)[C@H](CCCCNC(=O)c1ccc(I)cc1)NC(=O)N[C@@H](Cc1cscn1)C(O)=O |r| Show InChI InChI=1S/C20H23IN4O6S/c21-13-6-4-12(5-7-13)17(26)22-8-2-1-3-15(18(27)28)24-20(31)25-16(19(29)30)9-14-10-32-11-23-14/h4-7,10-11,15-16H,1-3,8-9H2,(H,22,26)(H,27,28)(H,29,30)(H2,24,25,31)/t15-,16-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 16.1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bayer HealthCare
Curated by ChEMBL
| Assay Description
Binding affinity to NAALADase |
J Med Chem 55: 9510-20 (2012)
Article DOI: 10.1021/jm300710j
BindingDB Entry DOI: 10.7270/Q28053R3 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data