 Found 85 hits with Last Name = 'evans' and Initial = 'lm'
Found 85 hits with Last Name = 'evans' and Initial = 'lm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277145
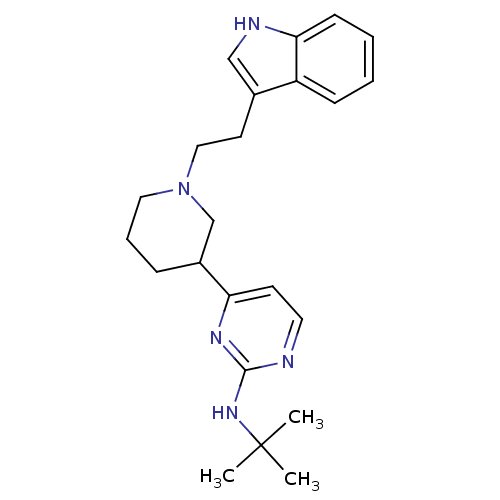
((+/-)-4-(1-(2-(1H-indol-3-yl)ethyl)piperidin-3-yl)...)Show SMILES CC(C)(C)Nc1nccc(n1)C1CCCN(CCc2c[nH]c3ccccc23)C1 Show InChI InChI=1S/C23H31N5/c1-23(2,3)27-22-24-12-10-20(26-22)18-7-6-13-28(16-18)14-11-17-15-25-21-9-5-4-8-19(17)21/h4-5,8-10,12,15,18,25H,6-7,11,13-14,16H2,1-3H3,(H,24,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277131
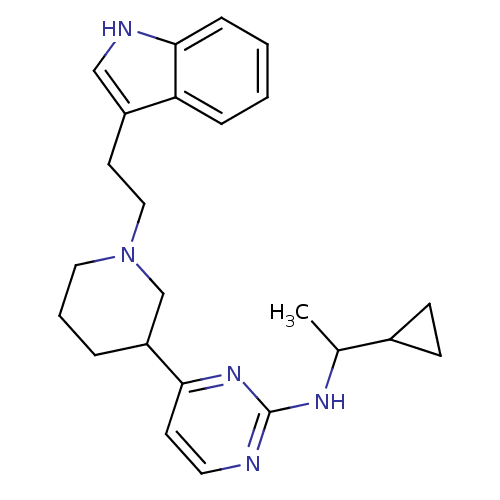
((+/-)-4-(1-(2-(1H-indol-3-yl)ethyl)piperidin-3-yl)...)Show SMILES CC(Nc1nccc(n1)C1CCCN(CCc2c[nH]c3ccccc23)C1)C1CC1 Show InChI InChI=1S/C24H31N5/c1-17(18-8-9-18)27-24-25-12-10-22(28-24)20-5-4-13-29(16-20)14-11-19-15-26-23-7-3-2-6-21(19)23/h2-3,6-7,10,12,15,17-18,20,26H,4-5,8-9,11,13-14,16H2,1H3,(H,25,27,28) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277180

(4-(1-(2-(5-chloro-1H-indol-3-yl)ethyl)piperidin-3-...)Show SMILES Clc1ccc2[nH]cc(CCN3CCCC(C3)c3ccnc(NC4CC4)n3)c2c1 Show InChI InChI=1S/C22H26ClN5/c23-17-3-6-21-19(12-17)15(13-25-21)8-11-28-10-1-2-16(14-28)20-7-9-24-22(27-20)26-18-4-5-18/h3,6-7,9,12-13,16,18,25H,1-2,4-5,8,10-11,14H2,(H,24,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10.4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277146
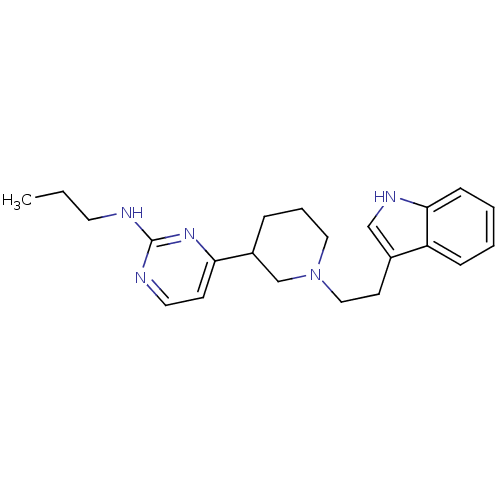
((+)-4-(1-(2-(1H-indol-3-yl)ethyl)piperidin-3-yl)-N...)Show InChI InChI=1S/C22H29N5/c1-2-11-23-22-24-12-9-20(26-22)18-6-5-13-27(16-18)14-10-17-15-25-21-8-4-3-7-19(17)21/h3-4,7-9,12,15,18,25H,2,5-6,10-11,13-14,16H2,1H3,(H,23,24,26) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 11.2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277146

((+)-4-(1-(2-(1H-indol-3-yl)ethyl)piperidin-3-yl)-N...)Show InChI InChI=1S/C22H29N5/c1-2-11-23-22-24-12-9-20(26-22)18-6-5-13-27(16-18)14-10-17-15-25-21-8-4-3-7-19(17)21/h3-4,7-9,12,15,18,25H,2,5-6,10-11,13-14,16H2,1H3,(H,23,24,26) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 11.2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50001859
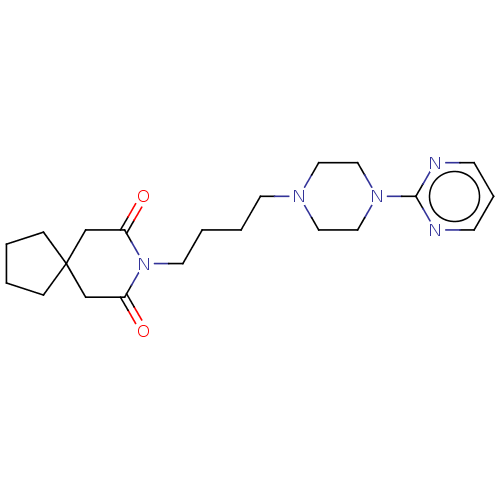
((buspirone) 8-[4-(4-Pyrimidin-2-yl-piperazin-1-yl)...)Show SMILES O=C1CC2(CCCC2)CC(=O)N1CCCCN1CCN(CC1)c1ncccn1 Show InChI InChI=1S/C21H31N5O2/c27-18-16-21(6-1-2-7-21)17-19(28)26(18)11-4-3-10-24-12-14-25(15-13-24)20-22-8-5-9-23-20/h5,8-9H,1-4,6-7,10-17H2 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277130
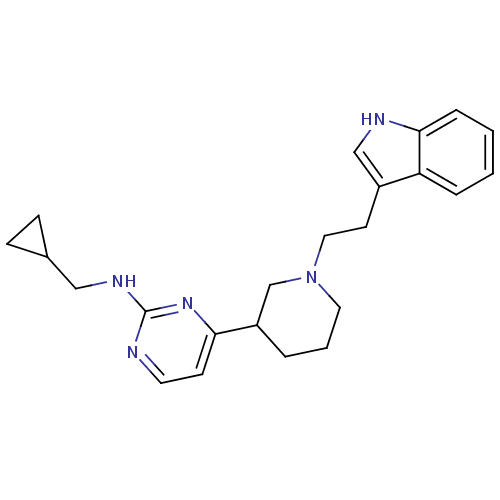
((+/-)-4-(1-(2-(1H-indol-3-yl)ethyl)piperidin-3-yl)...)Show SMILES C(Cc1c[nH]c2ccccc12)N1CCCC(C1)c1ccnc(NCC2CC2)n1 Show InChI InChI=1S/C23H29N5/c1-2-6-22-20(5-1)18(15-25-22)10-13-28-12-3-4-19(16-28)21-9-11-24-23(27-21)26-14-17-7-8-17/h1-2,5-6,9,11,15,17,19,25H,3-4,7-8,10,12-14,16H2,(H,24,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 21.3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277147

(CHEMBL473903 | N-cyclopropyl-4-(1-(2-(5-fluoro-1H-...)Show SMILES Fc1ccc2[nH]cc(CCN3CCCC(C3)c3ccnc(NC4CC4)n3)c2c1 Show InChI InChI=1S/C22H26FN5/c23-17-3-6-21-19(12-17)15(13-25-21)8-11-28-10-1-2-16(14-28)20-7-9-24-22(27-20)26-18-4-5-18/h3,6-7,9,12-13,16,18,25H,1-2,4-5,8,10-11,14H2,(H,24,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 23.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277181

(4-(1-(2-(5-chloro-2-methyl-1H-indol-3-yl)ethyl)pip...)Show SMILES Cc1[nH]c2ccc(Cl)cc2c1CCN1CCCC(C1)c1ccnc(NC2CC2)n1 Show InChI InChI=1S/C23H28ClN5/c1-15-19(20-13-17(24)4-7-22(20)26-15)9-12-29-11-2-3-16(14-29)21-8-10-25-23(28-21)27-18-5-6-18/h4,7-8,10,13,16,18,26H,2-3,5-6,9,11-12,14H2,1H3,(H,25,27,28) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 26.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277144
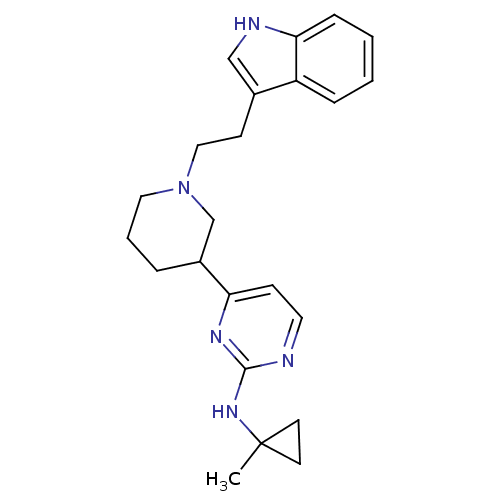
((+/-)-4-(1-(2-(1H-indol-3-yl)ethyl)piperidin-3-yl)...)Show SMILES CC1(CC1)Nc1nccc(n1)C1CCCN(CCc2c[nH]c3ccccc23)C1 Show InChI InChI=1S/C23H29N5/c1-23(10-11-23)27-22-24-12-8-20(26-22)18-5-4-13-28(16-18)14-9-17-15-25-21-7-3-2-6-19(17)21/h2-3,6-8,12,15,18,25H,4-5,9-11,13-14,16H2,1H3,(H,24,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277182
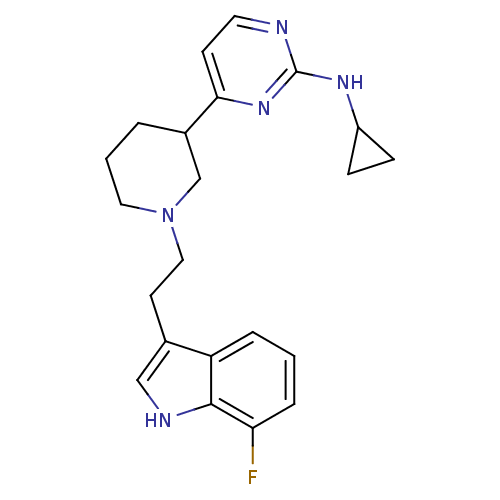
(CHEMBL475694 | N-cyclopropyl-4-(1-(2-(7-fluoro-1H-...)Show SMILES Fc1cccc2c(CCN3CCCC(C3)c3ccnc(NC4CC4)n3)c[nH]c12 Show InChI InChI=1S/C22H26FN5/c23-19-5-1-4-18-15(13-25-21(18)19)9-12-28-11-2-3-16(14-28)20-8-10-24-22(27-20)26-17-6-7-17/h1,4-5,8,10,13,16-17,25H,2-3,6-7,9,11-12,14H2,(H,24,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 35.1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277184

(CHEMBL474282 | N-cyclopropyl-4-(1-(2-((S)-2,3-dihy...)Show SMILES C(CN1CCCC(C1)c1ccnc(NC2CC2)n1)[C@H]1COc2ccccc2O1 |r| Show InChI InChI=1S/C22H28N4O2/c1-2-6-21-20(5-1)27-15-18(28-21)10-13-26-12-3-4-16(14-26)19-9-11-23-22(25-19)24-17-7-8-17/h1-2,5-6,9,11,16-18H,3-4,7-8,10,12-15H2,(H,23,24,25)/t16?,18-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 37.8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277210
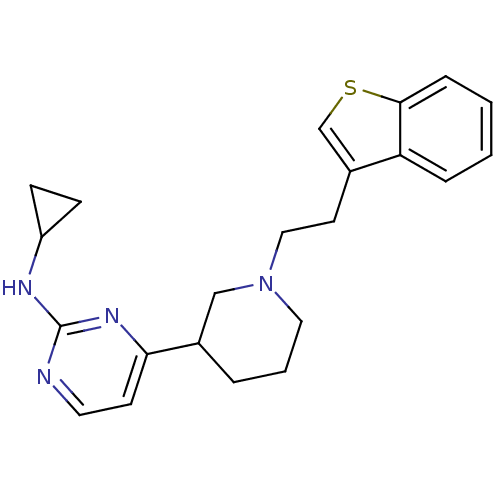
(4-(1-(2-(benzo[b]thiophen-3-yl)ethyl)piperidin-3-y...)Show SMILES C(Cc1csc2ccccc12)N1CCCC(C1)c1ccnc(NC2CC2)n1 Show InChI InChI=1S/C22H26N4S/c1-2-6-21-19(5-1)17(15-27-21)10-13-26-12-3-4-16(14-26)20-9-11-23-22(25-20)24-18-7-8-18/h1-2,5-6,9,11,15-16,18H,3-4,7-8,10,12-14H2,(H,23,24,25) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 38.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277212
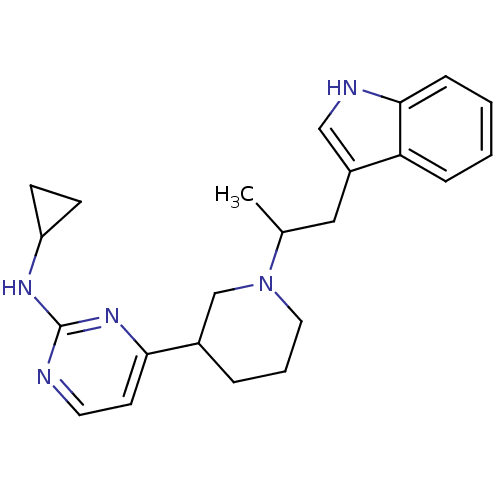
(4-(1-(1-(1H-indol-3-yl)propan-2-yl)piperidin-3-yl)...)Show SMILES CC(Cc1c[nH]c2ccccc12)N1CCCC(C1)c1ccnc(NC2CC2)n1 Show InChI InChI=1S/C23H29N5/c1-16(13-18-14-25-22-7-3-2-6-20(18)22)28-12-4-5-17(15-28)21-10-11-24-23(27-21)26-19-8-9-19/h2-3,6-7,10-11,14,16-17,19,25H,4-5,8-9,12-13,15H2,1H3,(H,24,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 40.2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277211
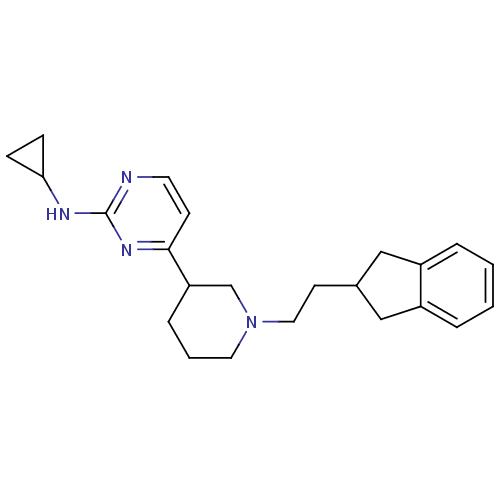
(CHEMBL475090 | N-cyclopropyl-4-(1-(2-(2,3-dihydro-...)Show SMILES C(CN1CCCC(C1)c1ccnc(NC2CC2)n1)C1Cc2ccccc2C1 Show InChI InChI=1S/C23H30N4/c1-2-5-19-15-17(14-18(19)4-1)10-13-27-12-3-6-20(16-27)22-9-11-24-23(26-22)25-21-7-8-21/h1-2,4-5,9,11,17,20-21H,3,6-8,10,12-16H2,(H,24,25,26) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 42.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277183
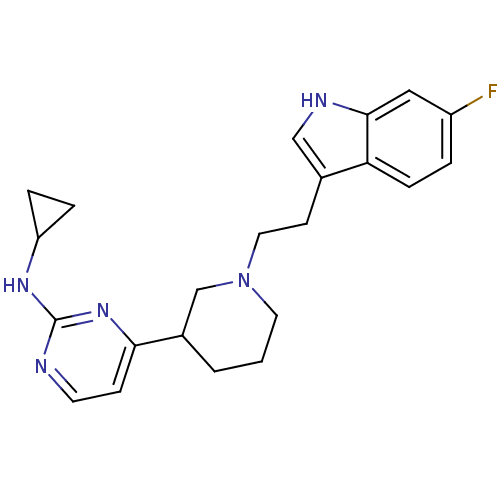
(CHEMBL514010 | N-cyclopropyl-4-(1-(2-(6-fluoro-1H-...)Show SMILES Fc1ccc2c(CCN3CCCC(C3)c3ccnc(NC4CC4)n3)c[nH]c2c1 Show InChI InChI=1S/C22H26FN5/c23-17-3-6-19-15(13-25-21(19)12-17)8-11-28-10-1-2-16(14-28)20-7-9-24-22(27-20)26-18-4-5-18/h3,6-7,9,12-13,16,18,25H,1-2,4-5,8,10-11,14H2,(H,24,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 64.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277252
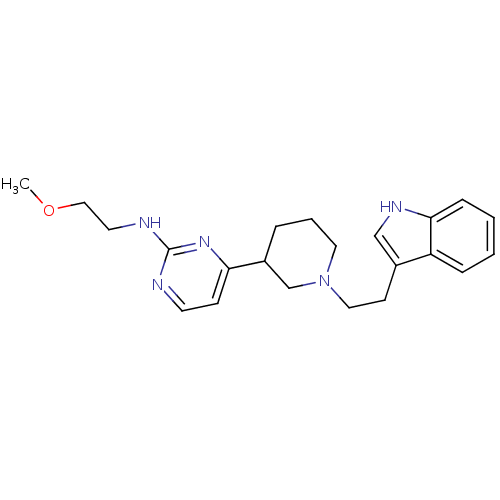
(4-(1-(2-(1H-indol-3-yl)ethyl)piperidin-3-yl)-N-(2-...)Show SMILES COCCNc1nccc(n1)C1CCCN(CCc2c[nH]c3ccccc23)C1 Show InChI InChI=1S/C22H29N5O/c1-28-14-11-24-22-23-10-8-20(26-22)18-5-4-12-27(16-18)13-9-17-15-25-21-7-3-2-6-19(17)21/h2-3,6-8,10,15,18,25H,4-5,9,11-14,16H2,1H3,(H,23,24,26) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 66.3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277129

((+/-)-4-(1-(2-(1H-indol-3-yl)ethyl)piperidin-3-yl)...)Show SMILES C(Cc1c[nH]c2ccccc12)N1CCCC(C1)c1ccnc(NC2CC2)n1 Show InChI InChI=1S/C22H27N5/c1-2-6-21-19(5-1)16(14-24-21)10-13-27-12-3-4-17(15-27)20-9-11-23-22(26-20)25-18-7-8-18/h1-2,5-6,9,11,14,17-18,24H,3-4,7-8,10,12-13,15H2,(H,23,25,26) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 75.3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277251
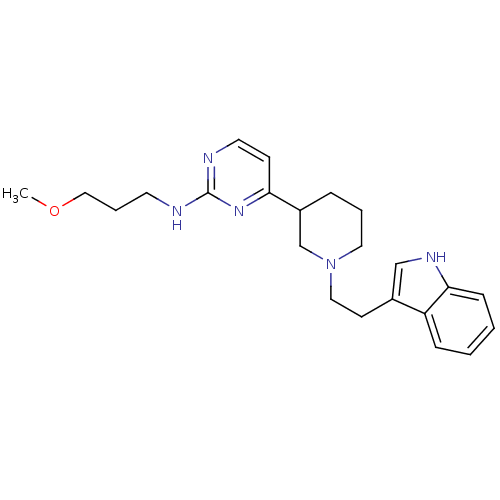
(4-(1-(2-(1H-indol-3-yl)ethyl)piperidin-3-yl)-N-(3-...)Show SMILES COCCCNc1nccc(n1)C1CCCN(CCc2c[nH]c3ccccc23)C1 Show InChI InChI=1S/C23H31N5O/c1-29-15-5-11-24-23-25-12-9-21(27-23)19-6-4-13-28(17-19)14-10-18-16-26-22-8-3-2-7-20(18)22/h2-3,7-9,12,16,19,26H,4-6,10-11,13-15,17H2,1H3,(H,24,25,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 78.3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277250
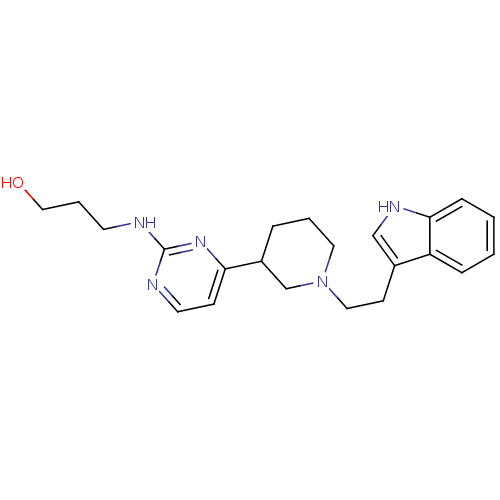
(3-(4-(1-(2-(1H-indol-3-yl)ethyl)piperidin-3-yl)pyr...)Show SMILES OCCCNc1nccc(n1)C1CCCN(CCc2c[nH]c3ccccc23)C1 Show InChI InChI=1S/C22H29N5O/c28-14-4-10-23-22-24-11-8-20(26-22)18-5-3-12-27(16-18)13-9-17-15-25-21-7-2-1-6-19(17)21/h1-2,6-8,11,15,18,25,28H,3-5,9-10,12-14,16H2,(H,23,24,26) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 175 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277214
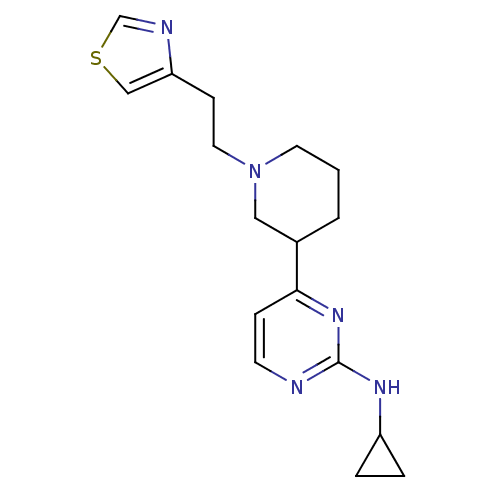
(CHEMBL475092 | N-cyclopropyl-4-(1-(2-(thiazol-4-yl...)Show InChI InChI=1S/C17H23N5S/c1-2-13(10-22(8-1)9-6-15-11-23-12-19-15)16-5-7-18-17(21-16)20-14-3-4-14/h5,7,11-14H,1-4,6,8-10H2,(H,18,20,21) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277213

(2-(3-(2-(cyclopropylamino)pyrimidin-4-yl)piperidin...)Show SMILES O=C(CN1CCCC(C1)c1ccnc(NC2CC2)n1)c1c[nH]c2ccccc12 Show InChI InChI=1S/C22H25N5O/c28-21(18-12-24-20-6-2-1-5-17(18)20)14-27-11-3-4-15(13-27)19-9-10-23-22(26-19)25-16-7-8-16/h1-2,5-6,9-10,12,15-16,24H,3-4,7-8,11,13-14H2,(H,23,25,26) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.42E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50277249

(4-(1-(2-(1H-pyrazol-4-yl)ethyl)piperidin-3-yl)-N-c...)Show InChI InChI=1S/C17H24N6/c1-2-14(12-23(8-1)9-6-13-10-19-20-11-13)16-5-7-18-17(22-16)21-15-3-4-15/h5,7,10-11,14-15H,1-4,6,8-9,12H2,(H,19,20)(H,18,21,22) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 4.45E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Displacement of [3H]8-OH-DPAT from human 5HT1A receptor |
Bioorg Med Chem Lett 19: 1159-63 (2009)
Article DOI: 10.1016/j.bmcl.2008.12.087
BindingDB Entry DOI: 10.7270/Q29886VW |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50063698

(4-Guanidino-benzoic acid 6-carbamimidoyl-naphthale...)Show SMILES [#7]\[#6](-[#7])=[#7]/c1ccc(cc1)-[#6](=O)-[#8]-c1ccc2cc(ccc2c1)-[#6](-[#7])=[#7] Show InChI InChI=1S/C19H17N5O2/c20-17(21)14-2-1-13-10-16(8-5-12(13)9-14)26-18(25)11-3-6-15(7-4-11)24-19(22)23/h1-10H,(H3,20,21)(H4,22,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro for inhibition of purified bovine trypsin. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063745
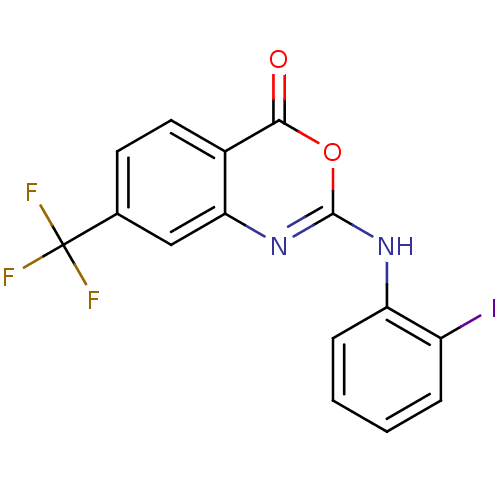
(2-(2-Iodo-phenylamino)-7-trifluoromethyl-benzo[d][...)Show InChI InChI=1S/C15H8F3IN2O2/c16-15(17,18)8-5-6-9-12(7-8)21-14(23-13(9)22)20-11-4-2-1-3-10(11)19/h1-7H,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Compound was evaluated in vitro for inhibitory activity against purified human C1r protease protease |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075982

(4-Methoxy-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxazin...)Show SMILES COc1ccc(cc1)S(=O)(=O)N(C)c1nc2ccccc2c(=O)o1 Show InChI InChI=1S/C16H14N2O5S/c1-18(24(20,21)12-9-7-11(22-2)8-10-12)16-17-14-6-4-3-5-13(14)15(19)23-16/h3-10H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50075980
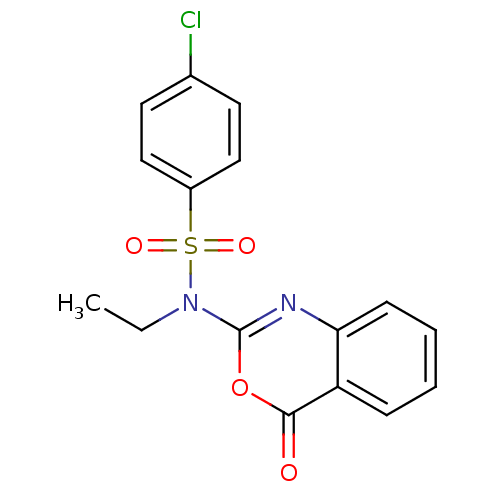
(4-Chloro-N-ethyl-N-(4-oxo-4H-benzo[d][1,3]oxazin-2...)Show SMILES CCN(c1nc2ccccc2c(=O)o1)S(=O)(=O)c1ccc(Cl)cc1 Show InChI InChI=1S/C16H13ClN2O4S/c1-2-19(24(21,22)12-9-7-11(17)8-10-12)16-18-14-6-4-3-5-13(14)15(20)23-16/h3-10H,2H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 930 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro for inhibition of purified bovine trypsin. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50075979

(4-Chloro-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxazin-...)Show SMILES CN(c1nc2ccccc2c(=O)o1)S(=O)(=O)c1ccc(Cl)cc1 Show InChI InChI=1S/C15H11ClN2O4S/c1-18(23(20,21)11-8-6-10(16)7-9-11)15-17-13-5-3-2-4-12(13)14(19)22-15/h2-9H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro for inhibition of purified bovine trypsin. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50075986
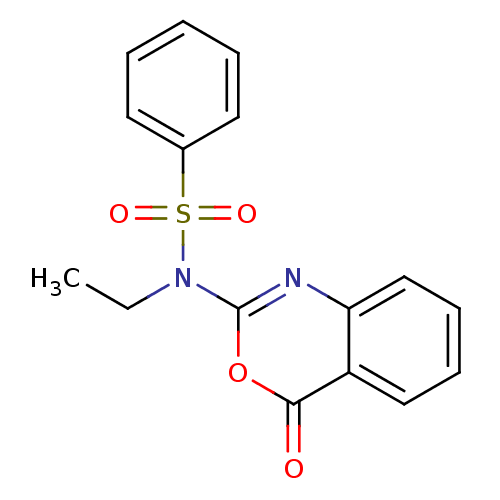
(CHEMBL168038 | N-Ethyl-N-(4-oxo-4H-benzo[d][1,3]ox...)Show InChI InChI=1S/C16H14N2O4S/c1-2-18(23(20,21)12-8-4-3-5-9-12)16-17-14-11-7-6-10-13(14)15(19)22-16/h3-11H,2H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro for inhibition of purified bovine trypsin. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075992

(4-Guanidino-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxaz...)Show SMILES [#6]-[#7](-c1nc2ccccc2c(=O)o1)S(=O)(=O)c1ccc(cc1)\[#7]=[#6](/[#7])-[#7] Show InChI InChI=1S/C16H15N5O4S/c1-21(16-20-13-5-3-2-4-12(13)14(22)25-16)26(23,24)11-8-6-10(7-9-11)19-15(17)18/h2-9H,1H3,(H4,17,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50075981
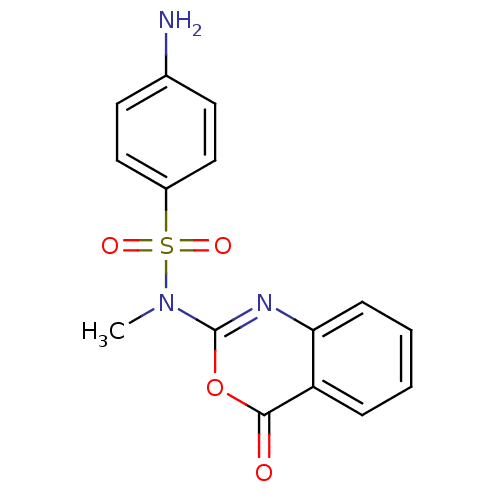
(4-Amino-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxazin-2...)Show InChI InChI=1S/C15H13N3O4S/c1-18(23(20,21)11-8-6-10(16)7-9-11)15-17-13-5-3-2-4-12(13)14(19)22-15/h2-9H,16H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro for inhibition of purified bovine trypsin. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075978
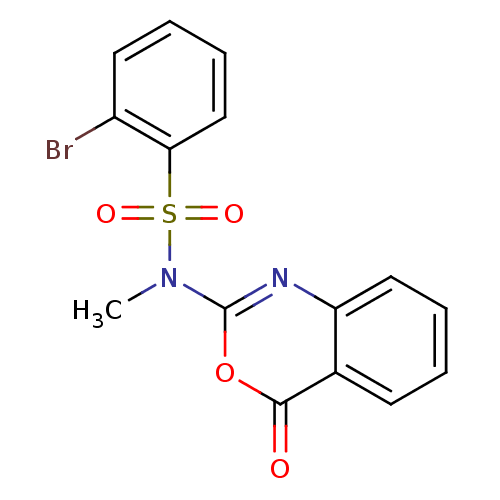
(2-Bromo-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxazin-2...)Show InChI InChI=1S/C15H11BrN2O4S/c1-18(23(20,21)13-9-5-3-7-11(13)16)15-17-12-8-4-2-6-10(12)14(19)22-15/h2-9H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075992

(4-Guanidino-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxaz...)Show SMILES [#6]-[#7](-c1nc2ccccc2c(=O)o1)S(=O)(=O)c1ccc(cc1)\[#7]=[#6](/[#7])-[#7] Show InChI InChI=1S/C16H15N5O4S/c1-21(16-20-13-5-3-2-4-12(13)14(22)25-16)26(23,24)11-8-6-10(7-9-11)19-15(17)18/h2-9H,1H3,(H4,17,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075986

(CHEMBL168038 | N-Ethyl-N-(4-oxo-4H-benzo[d][1,3]ox...)Show InChI InChI=1S/C16H14N2O4S/c1-2-18(23(20,21)12-8-4-3-5-9-12)16-17-14-11-7-6-10-13(14)15(19)22-16/h3-11H,2H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075993
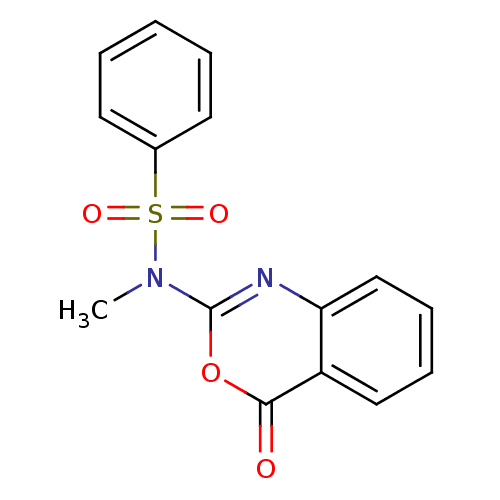
(CHEMBL166632 | N-Methyl-N-(4-oxo-4H-benzo[d][1,3]o...)Show InChI InChI=1S/C15H12N2O4S/c1-17(22(19,20)11-7-3-2-4-8-11)15-16-13-10-6-5-9-12(13)14(18)21-15/h2-10H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50063745

(2-(2-Iodo-phenylamino)-7-trifluoromethyl-benzo[d][...)Show InChI InChI=1S/C15H8F3IN2O2/c16-15(17,18)8-5-6-9-12(7-8)21-14(23-13(9)22)20-11-4-2-1-3-10(11)19/h1-7H,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Evaluated in vitro for inhibitory activity against purified human C1r protease incubated in buffer for 60 minutes |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50075991
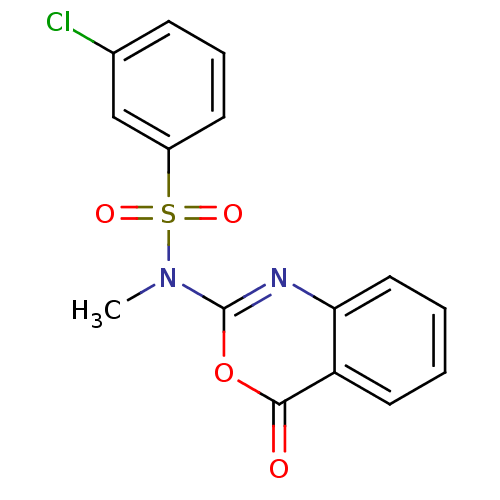
(3-Chloro-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxazin-...)Show SMILES CN(c1nc2ccccc2c(=O)o1)S(=O)(=O)c1cccc(Cl)c1 Show InChI InChI=1S/C15H11ClN2O4S/c1-18(23(20,21)11-6-4-5-10(16)9-11)15-17-13-8-3-2-7-12(13)14(19)22-15/h2-9H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro for inhibition of purified bovine trypsin. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50075989
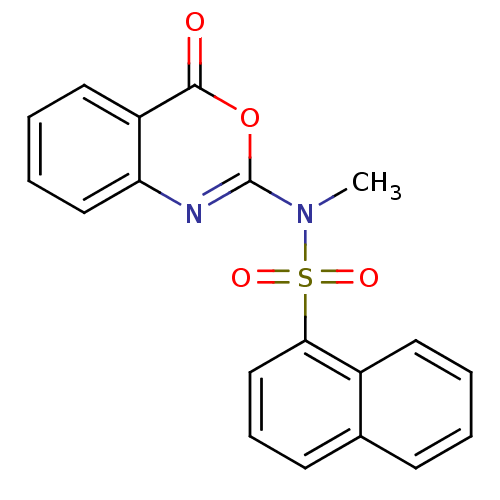
(CHEMBL167163 | Naphthalene-1-sulfonic acid methyl-...)Show SMILES CN(c1nc2ccccc2c(=O)o1)S(=O)(=O)c1cccc2ccccc12 Show InChI InChI=1S/C19H14N2O4S/c1-21(19-20-16-11-5-4-10-15(16)18(22)25-19)26(23,24)17-12-6-8-13-7-2-3-9-14(13)17/h2-12H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro for inhibition of purified bovine trypsin. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50075987
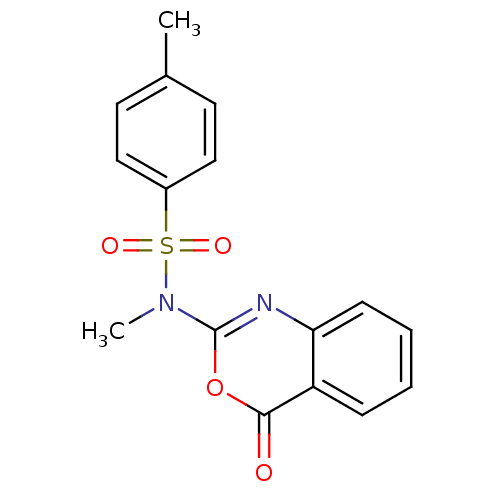
(4,N-Dimethyl-N-(4-oxo-4H-benzo[d][1,3]oxazin-2-yl)...)Show InChI InChI=1S/C16H14N2O4S/c1-11-7-9-12(10-8-11)23(20,21)18(2)16-17-14-6-4-3-5-13(14)15(19)22-16/h3-10H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro for inhibition of purified bovine trypsin. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50075990
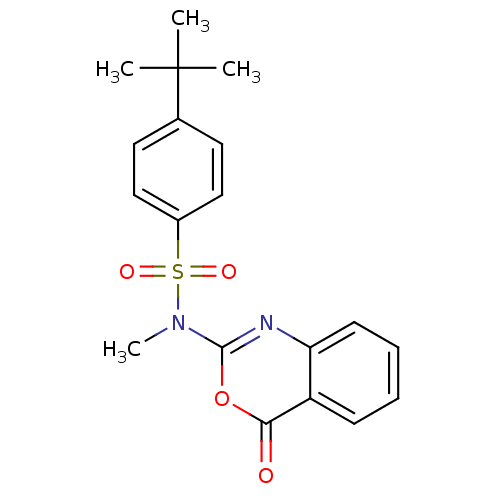
(4-tert-Butyl-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxa...)Show SMILES CN(c1nc2ccccc2c(=O)o1)S(=O)(=O)c1ccc(cc1)C(C)(C)C Show InChI InChI=1S/C19H20N2O4S/c1-19(2,3)13-9-11-14(12-10-13)26(23,24)21(4)18-20-16-8-6-5-7-15(16)17(22)25-18/h5-12H,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5.83E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro for inhibition of purified bovine trypsin. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075993

(CHEMBL166632 | N-Methyl-N-(4-oxo-4H-benzo[d][1,3]o...)Show InChI InChI=1S/C15H12N2O4S/c1-17(22(19,20)11-7-3-2-4-8-11)15-16-13-10-6-5-9-12(13)14(18)21-15/h2-10H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Evaluated in vitro for inhibitory activity against purified human C1r protease incubated in buffer for 60 minutes |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075982

(4-Methoxy-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxazin...)Show SMILES COc1ccc(cc1)S(=O)(=O)N(C)c1nc2ccccc2c(=O)o1 Show InChI InChI=1S/C16H14N2O5S/c1-18(24(20,21)12-9-7-11(22-2)8-10-12)16-17-14-6-4-3-5-13(14)15(19)23-16/h3-10H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Evaluated in vitro for inhibitory activity against purified human C1r protease incubated in buffer for 60 minutes |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075989

(CHEMBL167163 | Naphthalene-1-sulfonic acid methyl-...)Show SMILES CN(c1nc2ccccc2c(=O)o1)S(=O)(=O)c1cccc2ccccc12 Show InChI InChI=1S/C19H14N2O4S/c1-21(19-20-16-11-5-4-10-15(16)18(22)25-19)26(23,24)17-12-6-8-13-7-2-3-9-14(13)17/h2-12H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 6.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075981

(4-Amino-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxazin-2...)Show InChI InChI=1S/C15H13N3O4S/c1-18(23(20,21)11-8-6-10(16)7-9-11)15-17-13-5-3-2-4-12(13)14(19)22-15/h2-9H,16H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075981

(4-Amino-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxazin-2...)Show InChI InChI=1S/C15H13N3O4S/c1-18(23(20,21)11-8-6-10(16)7-9-11)15-17-13-5-3-2-4-12(13)14(19)22-15/h2-9H,16H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 7.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075984
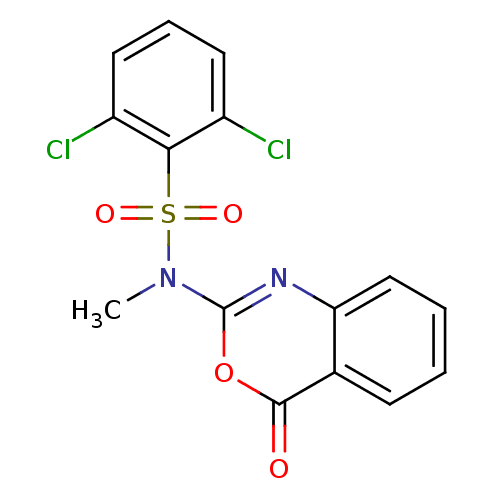
(2,6-Dichloro-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxa...)Show SMILES CN(c1nc2ccccc2c(=O)o1)S(=O)(=O)c1c(Cl)cccc1Cl Show InChI InChI=1S/C15H10Cl2N2O4S/c1-19(24(21,22)13-10(16)6-4-7-11(13)17)15-18-12-8-3-2-5-9(12)14(20)23-15/h2-8H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 7.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075987

(4,N-Dimethyl-N-(4-oxo-4H-benzo[d][1,3]oxazin-2-yl)...)Show InChI InChI=1S/C16H14N2O4S/c1-11-7-9-12(10-8-11)23(20,21)18(2)16-17-14-6-4-3-5-13(14)15(19)22-16/h3-10H,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 7.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro inhibition of purified human C1r protease. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50075985

(3,4-Dichloro-N-methyl-N-(4-oxo-4H-benzo[d][1,3]oxa...)Show SMILES CN(c1nc2ccccc2c(=O)o1)S(=O)(=O)c1ccc(Cl)c(Cl)c1 Show InChI InChI=1S/C15H10Cl2N2O4S/c1-19(24(21,22)9-6-7-11(16)12(17)8-9)15-18-13-5-3-2-4-10(13)14(20)23-15/h2-8H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
In vitro for inhibition of purified bovine trypsin. |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075986

(CHEMBL168038 | N-Ethyl-N-(4-oxo-4H-benzo[d][1,3]ox...)Show InChI InChI=1S/C16H14N2O4S/c1-2-18(23(20,21)12-8-4-3-5-9-12)16-17-14-11-7-6-10-13(14)15(19)22-16/h3-11H,2H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Evaluated in vitro for inhibitory activity against purified human C1r protease incubated in buffer for 60 minutes |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |
Complement C1r subcomponent
(Homo sapiens (Human)) | BDBM50075989

(CHEMBL167163 | Naphthalene-1-sulfonic acid methyl-...)Show SMILES CN(c1nc2ccccc2c(=O)o1)S(=O)(=O)c1cccc2ccccc12 Show InChI InChI=1S/C19H14N2O4S/c1-21(19-20-16-11-5-4-10-15(16)18(22)25-19)26(23,24)17-12-6-8-13-7-2-3-9-14(13)17/h2-12H,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Evaluated in vitro for inhibitory activity against purified human C1r protease incubated in buffer for 60 minutes |
Bioorg Med Chem Lett 9: 815-20 (1999)
BindingDB Entry DOI: 10.7270/Q29G5M0K |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data