 Found 205 hits with Last Name = 'burgess-henry' and Initial = 'j'
Found 205 hits with Last Name = 'burgess-henry' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50103651

(2-(3'-Amino-5-chloro-2-hydroxy-biphenyl-3-yl)-1H-b...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(Cl)cc(-c2cccc(N)c2)c1O Show InChI InChI=1S/C20H16ClN5O/c21-12-8-14(10-2-1-3-13(22)6-10)18(27)15(9-12)20-25-16-5-4-11(19(23)24)7-17(16)26-20/h1-9,27H,22H2,(H3,23,24)(H,25,26) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for factor VIIa/TF |
Bioorg Med Chem Lett 11: 2253-6 (2001)
BindingDB Entry DOI: 10.7270/Q2XG9QD9 |
More data for this
Ligand-Target Pair | |
Urokinase-type plasminogen activator
(Homo sapiens (Human)) | BDBM50103651

(2-(3'-Amino-5-chloro-2-hydroxy-biphenyl-3-yl)-1H-b...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(Cl)cc(-c2cccc(N)c2)c1O Show InChI InChI=1S/C20H16ClN5O/c21-12-8-14(10-2-1-3-13(22)6-10)18(27)15(9-12)20-25-16-5-4-11(19(23)24)7-17(16)26-20/h1-9,27H,22H2,(H3,23,24)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity of the compound towards urokinase-type plasminogen activator (microPa) |
Bioorg Med Chem Lett 11: 2253-6 (2001)
BindingDB Entry DOI: 10.7270/Q2XG9QD9 |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM50180517
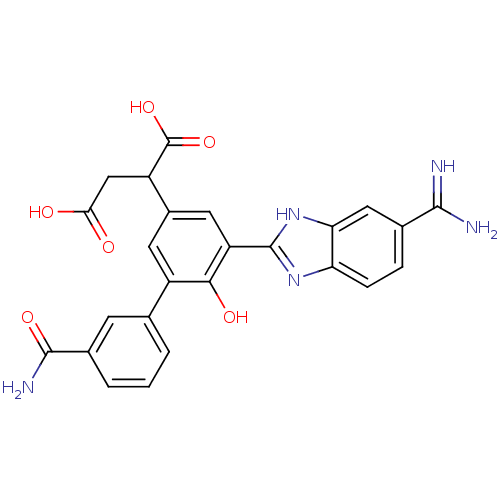
(2-[5-(5-carbamimidoyl-1H-benzoimidazol-2-yl)-3'-ca...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(-c2cccc(c2)C(N)=O)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C25H21N5O6/c26-22(27)12-4-5-18-19(9-12)30-24(29-18)17-8-14(16(25(35)36)10-20(31)32)7-15(21(17)33)11-2-1-3-13(6-11)23(28)34/h1-9,16,33H,10H2,(H3,26,27)(H2,28,34)(H,29,30)(H,31,32)(H,35,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50103651

(2-(3'-Amino-5-chloro-2-hydroxy-biphenyl-3-yl)-1H-b...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(Cl)cc(-c2cccc(N)c2)c1O Show InChI InChI=1S/C20H16ClN5O/c21-12-8-14(10-2-1-3-13(22)6-10)18(27)15(9-12)20-25-16-5-4-11(19(23)24)7-17(16)26-20/h1-9,27H,22H2,(H3,23,24)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity of the compound towards Coagulation factor X |
Bioorg Med Chem Lett 11: 2253-6 (2001)
BindingDB Entry DOI: 10.7270/Q2XG9QD9 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101871

(3-[3-(3-Benzyl-5-carbamimidoyl-1H-indol-2-yl)-5-br...)Show SMILES NC(=N)c1ccc2[nH]c(c(Cc3ccccc3)c2c1)-c1cc(CCC(O)=O)cc(Br)c1O Show InChI InChI=1S/C25H22BrN3O3/c26-20-12-15(6-9-22(30)31)11-19(24(20)32)23-18(10-14-4-2-1-3-5-14)17-13-16(25(27)28)7-8-21(17)29-23/h1-5,7-8,11-13,29,32H,6,9-10H2,(H3,27,28)(H,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Coagulation factor X in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101882
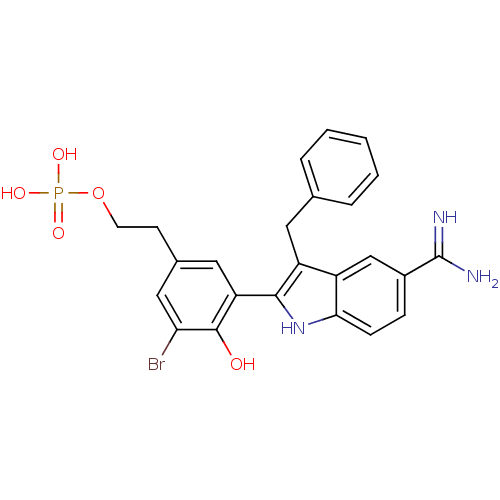
(CHEMBL53829 | Phosphoric acid mono-{2-[3-(3-benzyl...)Show SMILES NC(=N)c1ccc2[nH]c(c(Cc3ccccc3)c2c1)-c1cc(CCOP(O)(O)=O)cc(Br)c1O Show InChI InChI=1S/C24H23BrN3O5P/c25-20-12-15(8-9-33-34(30,31)32)11-19(23(20)29)22-18(10-14-4-2-1-3-5-14)17-13-16(24(26)27)6-7-21(17)28-22/h1-7,11-13,28-29H,8-10H2,(H3,26,27)(H2,30,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Coagulation factor X in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101881
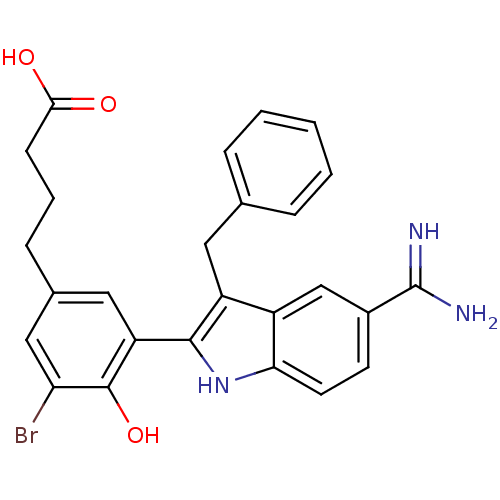
(4-[3-(3-Benzyl-5-carbamimidoyl-1H-indol-2-yl)-5-br...)Show SMILES NC(=N)c1ccc2[nH]c(c(Cc3ccccc3)c2c1)-c1cc(CCCC(O)=O)cc(Br)c1O Show InChI InChI=1S/C26H24BrN3O3/c27-21-13-16(7-4-8-23(31)32)12-20(25(21)33)24-19(11-15-5-2-1-3-6-15)18-14-17(26(28)29)9-10-22(18)30-24/h1-3,5-6,9-10,12-14,30,33H,4,7-8,11H2,(H3,28,29)(H,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Coagulation factor X in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101885
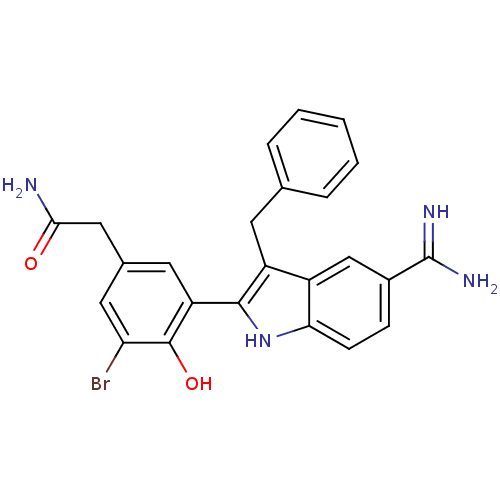
(2-[3-(3-Benzyl-5-carbamimidoyl-1H-indol-2-yl)-5-br...)Show SMILES NC(=O)Cc1cc(Br)c(O)c(c1)-c1[nH]c2ccc(cc2c1Cc1ccccc1)C(N)=N Show InChI InChI=1S/C24H21BrN4O2/c25-19-10-14(11-21(26)30)9-18(23(19)31)22-17(8-13-4-2-1-3-5-13)16-12-15(24(27)28)6-7-20(16)29-22/h1-7,9-10,12,29,31H,8,11H2,(H2,26,30)(H3,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Coagulation factor X in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101883
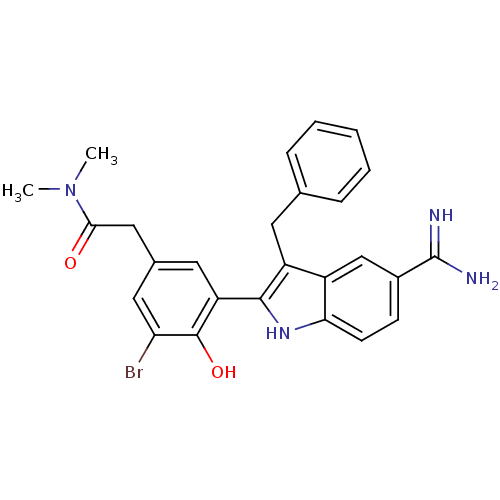
(2-[3-(3-Benzyl-5-carbamimidoyl-1H-indol-2-yl)-5-br...)Show SMILES CN(C)C(=O)Cc1cc(Br)c(O)c(c1)-c1[nH]c2ccc(cc2c1Cc1ccccc1)C(N)=N Show InChI InChI=1S/C26H25BrN4O2/c1-31(2)23(32)13-16-11-20(25(33)21(27)12-16)24-19(10-15-6-4-3-5-7-15)18-14-17(26(28)29)8-9-22(18)30-24/h3-9,11-12,14,30,33H,10,13H2,1-2H3,(H3,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Coagulation factor X in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50103655
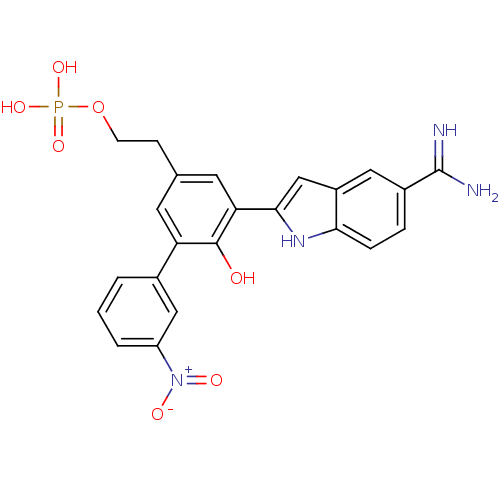
(CHEMBL72231 | Phosphoric acid mono-{2-[5-(5-carbam...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(CCO[P+](O)(O)[O-])cc(c1O)-c1cccc(c1)[N+]([O-])=O Show InChI InChI=1S/C23H21N4O7P/c24-23(25)15-4-5-20-16(10-15)12-21(26-20)19-9-13(6-7-34-35(31,32)33)8-18(22(19)28)14-2-1-3-17(11-14)27(29)30/h1-5,8-12,26,28H,6-7H2,(H3,24,25)(H2,31,32,33) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for factor VIIa/TF |
Bioorg Med Chem Lett 11: 2253-6 (2001)
BindingDB Entry DOI: 10.7270/Q2XG9QD9 |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM14917
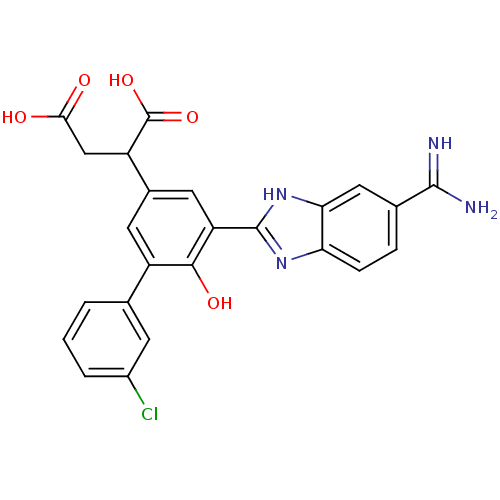
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-5-(...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(-c2cccc(Cl)c2)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H19ClN4O5/c25-14-3-1-2-11(6-14)15-7-13(16(24(33)34)10-20(30)31)8-17(21(15)32)23-28-18-5-4-12(22(26)27)9-19(18)29-23/h1-9,16,32H,10H2,(H3,26,27)(H,28,29)(H,30,31)(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101879
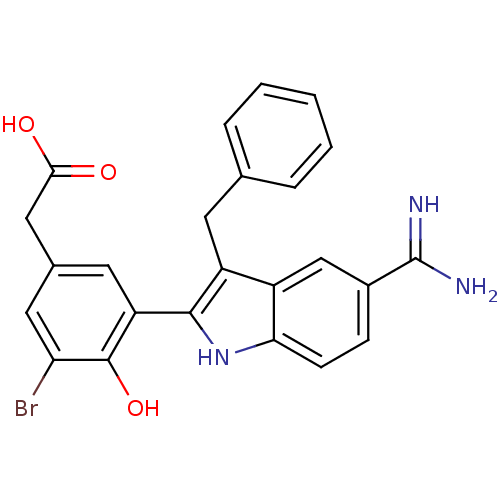
(CHEMBL50924 | [3-(3-Benzyl-5-carbamimidoyl-1H-indo...)Show SMILES NC(=N)c1ccc2[nH]c(c(Cc3ccccc3)c2c1)-c1cc(CC(O)=O)cc(Br)c1O Show InChI InChI=1S/C24H20BrN3O3/c25-19-10-14(11-21(29)30)9-18(23(19)31)22-17(8-13-4-2-1-3-5-13)16-12-15(24(26)27)6-7-20(16)28-22/h1-7,9-10,12,28,31H,8,11H2,(H3,26,27)(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Coagulation factor X in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101880
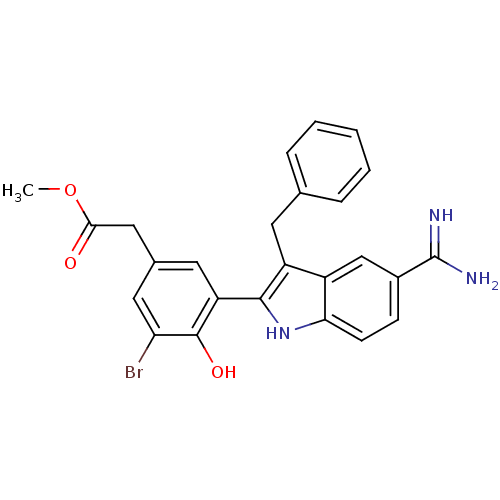
(CHEMBL416127 | [3-(3-Benzyl-5-carbamimidoyl-1H-ind...)Show SMILES COC(=O)Cc1cc(Br)c(O)c(c1)-c1[nH]c2ccc(cc2c1Cc1ccccc1)C(N)=N Show InChI InChI=1S/C25H22BrN3O3/c1-32-22(30)12-15-10-19(24(31)20(26)11-15)23-18(9-14-5-3-2-4-6-14)17-13-16(25(27)28)7-8-21(17)29-23/h2-8,10-11,13,29,31H,9,12H2,1H3,(H3,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Coagulation factor X in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM13778

(2-[3-(5-carbamimidoyl-1H-indol-2-yl)-4-hydroxy-5-(...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(CC(O)=O)cc(c1O)-c1cccc(c1)[N+]([O-])=O Show InChI InChI=1S/C23H18N4O5/c24-23(25)14-4-5-19-15(9-14)11-20(26-19)18-7-12(8-21(28)29)6-17(22(18)30)13-2-1-3-16(10-13)27(31)32/h1-7,9-11,26,30H,8H2,(H3,24,25)(H,28,29) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for factor VIIa/TF |
Bioorg Med Chem Lett 11: 2253-6 (2001)
BindingDB Entry DOI: 10.7270/Q2XG9QD9 |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM14921
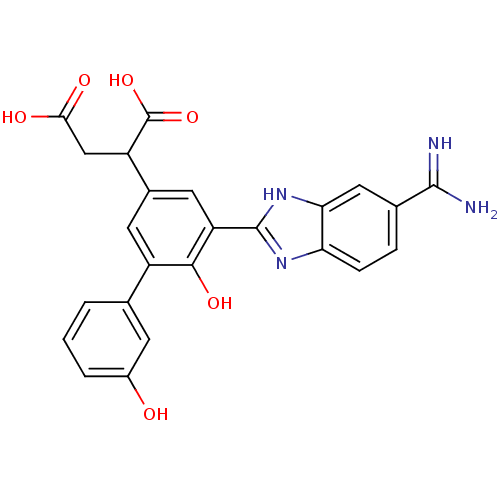
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(-c2cccc(O)c2)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H20N4O6/c25-22(26)12-4-5-18-19(9-12)28-23(27-18)17-8-13(16(24(33)34)10-20(30)31)7-15(21(17)32)11-2-1-3-14(29)6-11/h1-9,16,29,32H,10H2,(H3,25,26)(H,27,28)(H,30,31)(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Plasminogen
(Homo sapiens (Human)) | BDBM50103651

(2-(3'-Amino-5-chloro-2-hydroxy-biphenyl-3-yl)-1H-b...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(Cl)cc(-c2cccc(N)c2)c1O Show InChI InChI=1S/C20H16ClN5O/c21-12-8-14(10-2-1-3-13(22)6-10)18(27)15(9-12)20-25-16-5-4-11(19(23)24)7-17(16)26-20/h1-9,27H,22H2,(H3,23,24)(H,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity of the compound towards Plasmin |
Bioorg Med Chem Lett 11: 2253-6 (2001)
BindingDB Entry DOI: 10.7270/Q2XG9QD9 |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14898
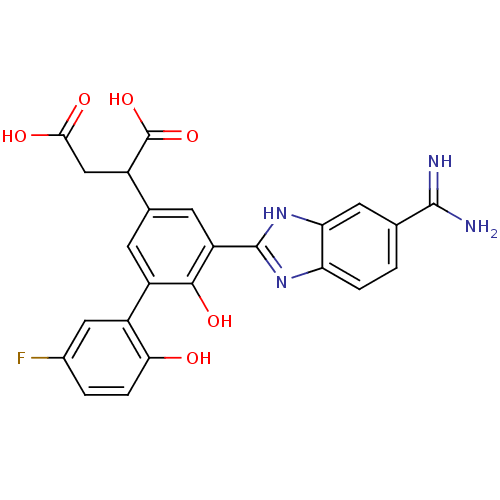
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-5-(...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(c1O)-c1cc(F)ccc1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H19FN4O6/c25-12-2-4-19(30)14(8-12)15-5-11(13(24(34)35)9-20(31)32)6-16(21(15)33)23-28-17-3-1-10(22(26)27)7-18(17)29-23/h1-8,13,30,33H,9H2,(H3,26,27)(H,28,29)(H,31,32)(H,34,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | -47.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM14914

(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES Cc1cccc(c1)-c1cc(cc(-c2nc3ccc(cc3[nH]2)C(N)=N)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C25H22N4O5/c1-12-3-2-4-13(7-12)16-8-15(17(25(33)34)11-21(30)31)9-18(22(16)32)24-28-19-6-5-14(23(26)27)10-20(19)29-24/h2-10,17,32H,11H2,1H3,(H3,26,27)(H,28,29)(H,30,31)(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101870
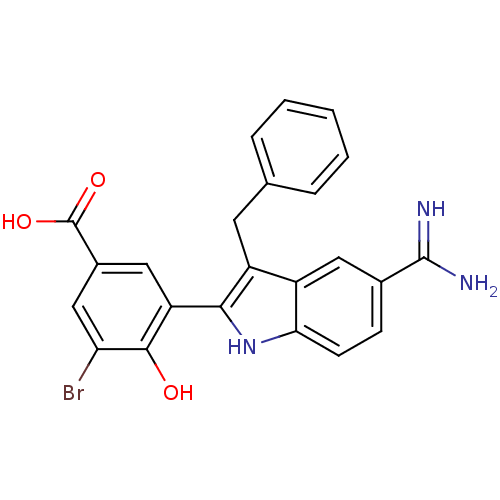
(3-(3-Benzyl-5-carbamimidoyl-1H-indol-2-yl)-5-bromo...)Show SMILES NC(=N)c1ccc2[nH]c(c(Cc3ccccc3)c2c1)-c1cc(cc(Br)c1O)C(O)=O Show InChI InChI=1S/C23H18BrN3O3/c24-18-11-14(23(29)30)10-17(21(18)28)20-16(8-12-4-2-1-3-5-12)15-9-13(22(25)26)6-7-19(15)27-20/h1-7,9-11,27-28H,8H2,(H3,25,26)(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Coagulation factor X in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50103651

(2-(3'-Amino-5-chloro-2-hydroxy-biphenyl-3-yl)-1H-b...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(Cl)cc(-c2cccc(N)c2)c1O Show InChI InChI=1S/C20H16ClN5O/c21-12-8-14(10-2-1-3-13(22)6-10)18(27)15(9-12)20-25-16-5-4-11(19(23)24)7-17(16)26-20/h1-9,27H,22H2,(H3,23,24)(H,25,26) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity of the compound towards Coagulation factor II |
Bioorg Med Chem Lett 11: 2253-6 (2001)
BindingDB Entry DOI: 10.7270/Q2XG9QD9 |
More data for this
Ligand-Target Pair | |
Serine protease 1
(Homo sapiens (Human)) | BDBM50103651

(2-(3'-Amino-5-chloro-2-hydroxy-biphenyl-3-yl)-1H-b...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(Cl)cc(-c2cccc(N)c2)c1O Show InChI InChI=1S/C20H16ClN5O/c21-12-8-14(10-2-1-3-13(22)6-10)18(27)15(9-12)20-25-16-5-4-11(19(23)24)7-17(16)26-20/h1-9,27H,22H2,(H3,23,24)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity of the compound towards Trypsin |
Bioorg Med Chem Lett 11: 2253-6 (2001)
BindingDB Entry DOI: 10.7270/Q2XG9QD9 |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM50180516
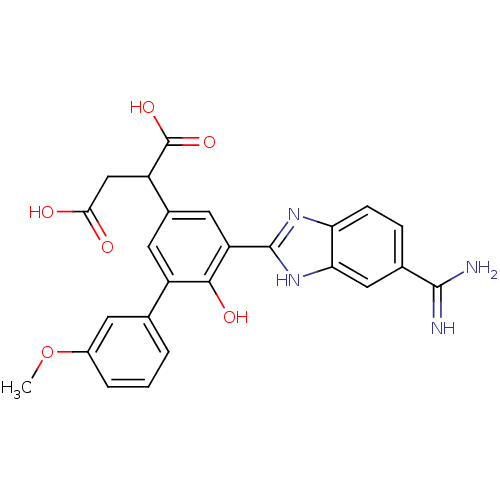
(2-[5-(5-carbamimidoyl-1H-benzoimidazol-2-yl)-6-hyd...)Show SMILES COc1cccc(c1)-c1cc(cc(-c2nc3ccc(cc3[nH]2)C(N)=N)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C25H22N4O6/c1-35-15-4-2-3-12(7-15)16-8-14(17(25(33)34)11-21(30)31)9-18(22(16)32)24-28-19-6-5-13(23(26)27)10-20(19)29-24/h2-10,17,32H,11H2,1H3,(H3,26,27)(H,28,29)(H,30,31)(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50103652
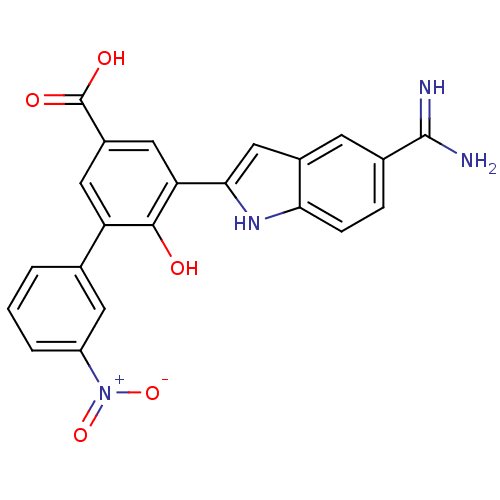
(5-(5-Carbamimidoyl-1H-indol-2-yl)-6-hydroxy-3'-nit...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(cc(c1O)-c1cccc(c1)[N+]([O-])=O)C(O)=O Show InChI InChI=1S/C22H16N4O5/c23-21(24)12-4-5-18-13(6-12)10-19(25-18)17-9-14(22(28)29)8-16(20(17)27)11-2-1-3-15(7-11)26(30)31/h1-10,25,27H,(H3,23,24)(H,28,29) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for factor VIIa/TF |
Bioorg Med Chem Lett 11: 2253-6 (2001)
BindingDB Entry DOI: 10.7270/Q2XG9QD9 |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14899

(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-5-(...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(c1O)-c1cc(Cl)ccc1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H19ClN4O6/c25-12-2-4-19(30)14(8-12)15-5-11(13(24(34)35)9-20(31)32)6-16(21(15)33)23-28-17-3-1-10(22(26)27)7-18(17)29-23/h1-8,13,30,33H,9H2,(H3,26,27)(H,28,29)(H,31,32)(H,34,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5.40 | -46.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14900
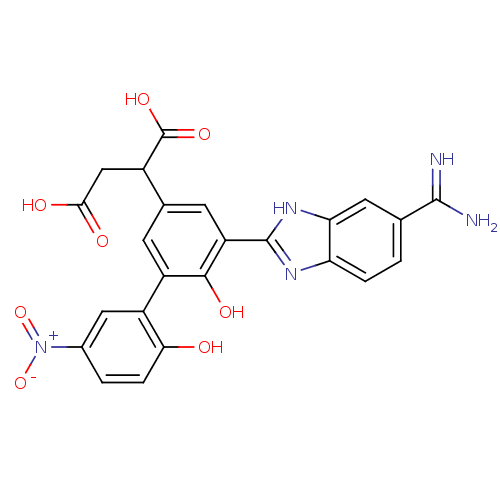
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(c1O)-c1cc(ccc1O)[N+]([O-])=O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H19N5O8/c25-22(26)10-1-3-17-18(7-10)28-23(27-17)16-6-11(13(24(34)35)9-20(31)32)5-15(21(16)33)14-8-12(29(36)37)2-4-19(14)30/h1-8,13,30,33H,9H2,(H3,25,26)(H,27,28)(H,31,32)(H,34,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | -46.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM14912
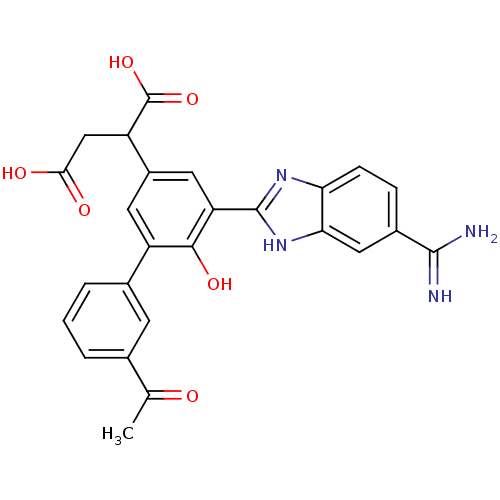
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-5-(...)Show SMILES CC(=O)c1cccc(c1)-c1cc(cc(-c2nc3ccc(cc3[nH]2)C(N)=N)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C26H22N4O6/c1-12(31)13-3-2-4-14(7-13)17-8-16(18(26(35)36)11-22(32)33)9-19(23(17)34)25-29-20-6-5-15(24(27)28)10-21(20)30-25/h2-10,18,34H,11H2,1H3,(H3,27,28)(H,29,30)(H,32,33)(H,35,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM14939
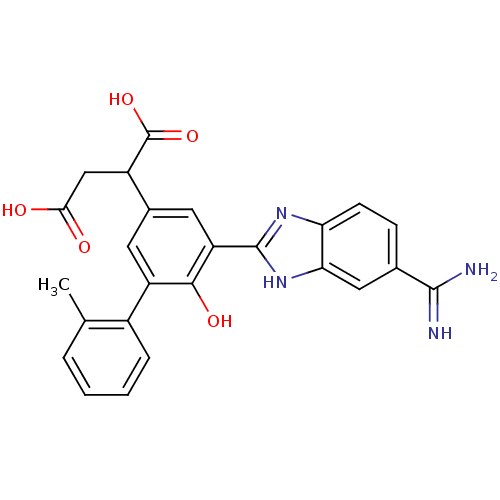
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES Cc1ccccc1-c1cc(cc(-c2nc3ccc(cc3[nH]2)C(N)=N)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C25H22N4O5/c1-12-4-2-3-5-15(12)17-8-14(16(25(33)34)11-21(30)31)9-18(22(17)32)24-28-19-7-6-13(23(26)27)10-20(19)29-24/h2-10,16,32H,11H2,1H3,(H3,26,27)(H,28,29)(H,30,31)(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM14927
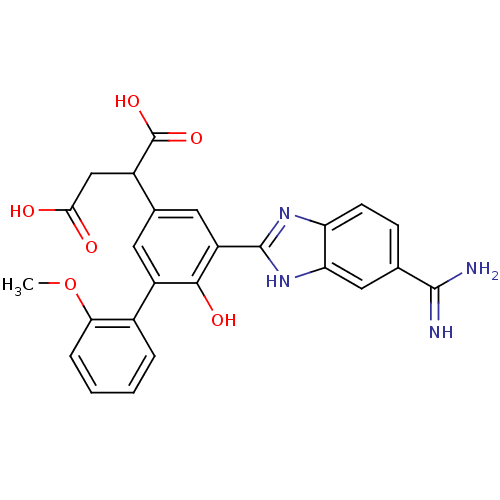
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES COc1ccccc1-c1cc(cc(-c2nc3ccc(cc3[nH]2)C(N)=N)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C25H22N4O6/c1-35-20-5-3-2-4-14(20)16-8-13(15(25(33)34)11-21(30)31)9-17(22(16)32)24-28-18-7-6-12(23(26)27)10-19(18)29-24/h2-10,15,32H,11H2,1H3,(H3,26,27)(H,28,29)(H,30,31)(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM14897
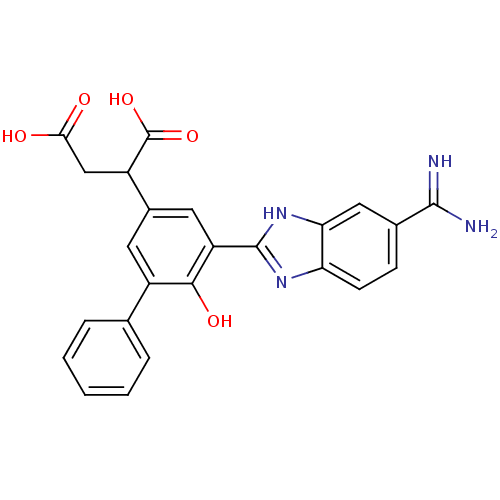
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(-c2ccccc2)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H20N4O5/c25-22(26)13-6-7-18-19(10-13)28-23(27-18)17-9-14(16(24(32)33)11-20(29)30)8-15(21(17)31)12-4-2-1-3-5-12/h1-10,16,31H,11H2,(H3,25,26)(H,27,28)(H,29,30)(H,32,33) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14904
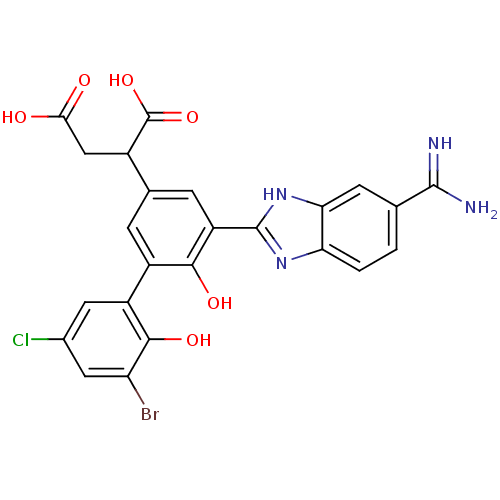
(2-[3-(3-bromo-5-chloro-2-hydroxyphenyl)-5-(5-carba...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(c1O)-c1cc(Cl)cc(Br)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H18BrClN4O6/c25-16-7-11(26)6-14(21(16)34)13-3-10(12(24(35)36)8-19(31)32)4-15(20(13)33)23-29-17-2-1-9(22(27)28)5-18(17)30-23/h1-7,12,33-34H,8H2,(H3,27,28)(H,29,30)(H,31,32)(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | -45.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101869

(2-[3-(3-Benzyl-5-carbamimidoyl-1H-indol-2-yl)-5-br...)Show SMILES CC(C)(C(O)=O)c1cc(Br)c(O)c(c1)-c1[nH]c2ccc(cc2c1Cc1ccccc1)C(N)=N Show InChI InChI=1S/C26H24BrN3O3/c1-26(2,25(32)33)16-12-19(23(31)20(27)13-16)22-18(10-14-6-4-3-5-7-14)17-11-15(24(28)29)8-9-21(17)30-22/h3-9,11-13,30-31H,10H2,1-2H3,(H3,28,29)(H,32,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Coagulation factor X in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14901
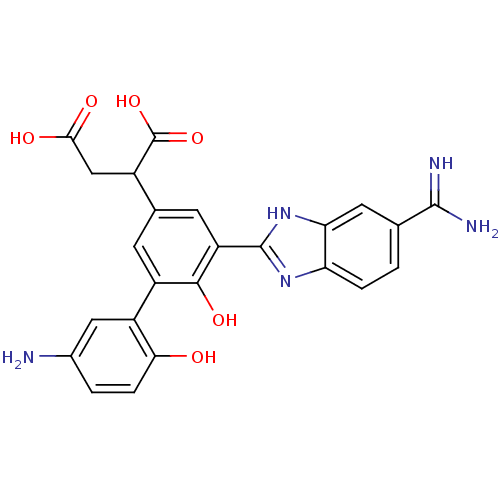
(2-[3-(5-amino-2-hydroxyphenyl)-5-(5-carbamimidoyl-...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(c1O)-c1cc(N)ccc1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H21N5O6/c25-12-2-4-19(30)14(8-12)15-5-11(13(24(34)35)9-20(31)32)6-16(21(15)33)23-28-17-3-1-10(22(26)27)7-18(17)29-23/h1-8,13,30,33H,9,25H2,(H3,26,27)(H,28,29)(H,31,32)(H,34,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 10 | -45.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101867

(3-Benzyl-2-[3-bromo-2-hydroxy-5-(2H-tetrazol-5-yl)...)Show SMILES NC(=N)c1ccc2[nH]c(c(Cc3ccccc3)c2c1)-c1cc(cc(Br)c1O)-c1nnn[nH]1 Show InChI InChI=1S/C23H18BrN7O/c24-18-11-14(23-28-30-31-29-23)10-17(21(18)32)20-16(8-12-4-2-1-3-5-12)15-9-13(22(25)26)6-7-19(15)27-20/h1-7,9-11,27,32H,8H2,(H3,25,26)(H,28,29,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Coagulation factor X in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM14920
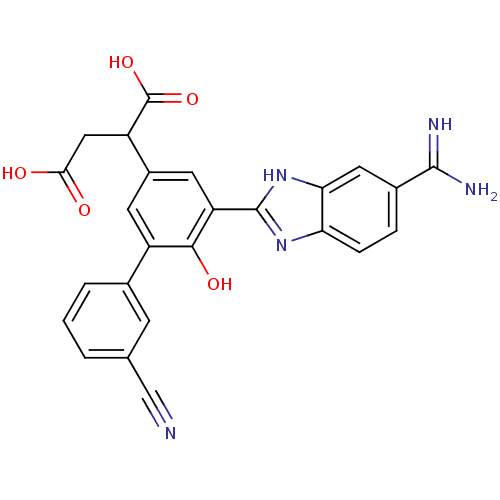
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-5-(...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(-c2cccc(c2)C#N)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C25H19N5O5/c26-11-12-2-1-3-13(6-12)16-7-15(17(25(34)35)10-21(31)32)8-18(22(16)33)24-29-19-5-4-14(23(27)28)9-20(19)30-24/h1-9,17,33H,10H2,(H3,27,28)(H,29,30)(H,31,32)(H,34,35) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14902
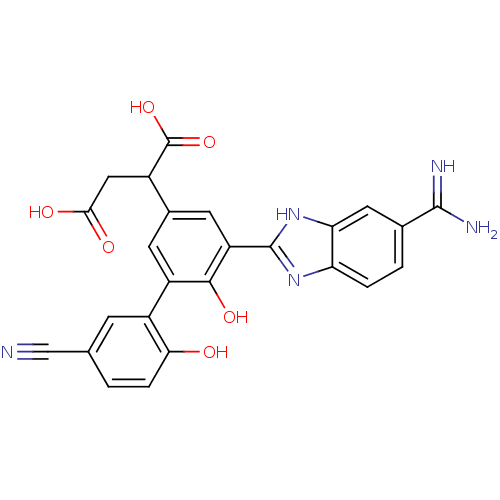
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-5-(...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(c1O)-c1cc(ccc1O)C#N)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C25H19N5O6/c26-10-11-1-4-20(31)15(5-11)16-6-13(14(25(35)36)9-21(32)33)7-17(22(16)34)24-29-18-3-2-12(23(27)28)8-19(18)30-24/h1-8,14,31,34H,9H2,(H3,27,28)(H,29,30)(H,32,33)(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 12 | -44.8 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50103662
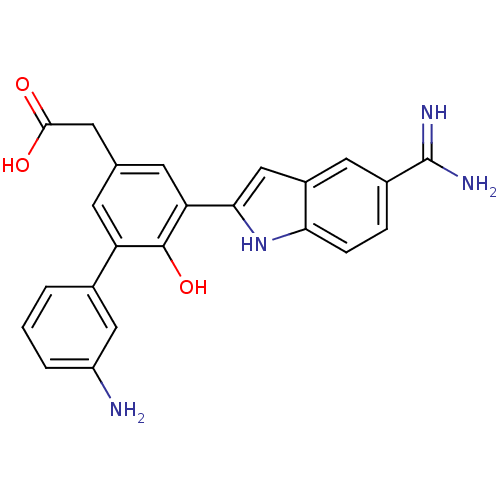
(CHEMBL73162 | [3'-Amino-5-(5-carbamimidoyl-1H-indo...)Show SMILES NC(=N)c1ccc2[nH]c(cc2c1)-c1cc(CC(O)=O)cc(c1O)-c1cccc(N)c1 Show InChI InChI=1S/C23H20N4O3/c24-16-3-1-2-13(10-16)17-6-12(8-21(28)29)7-18(22(17)30)20-11-15-9-14(23(25)26)4-5-19(15)27-20/h1-7,9-11,27,30H,8,24H2,(H3,25,26)(H,28,29) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for factor VIIa/TF |
Bioorg Med Chem Lett 11: 2253-6 (2001)
BindingDB Entry DOI: 10.7270/Q2XG9QD9 |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM14929
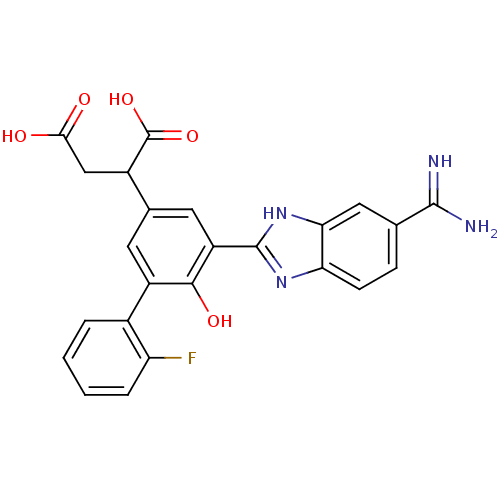
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-5-(...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(c1O)-c1ccccc1F)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H19FN4O5/c25-17-4-2-1-3-13(17)15-7-12(14(24(33)34)10-20(30)31)8-16(21(15)32)23-28-18-6-5-11(22(26)27)9-19(18)29-23/h1-9,14,32H,10H2,(H3,26,27)(H,28,29)(H,30,31)(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM14903
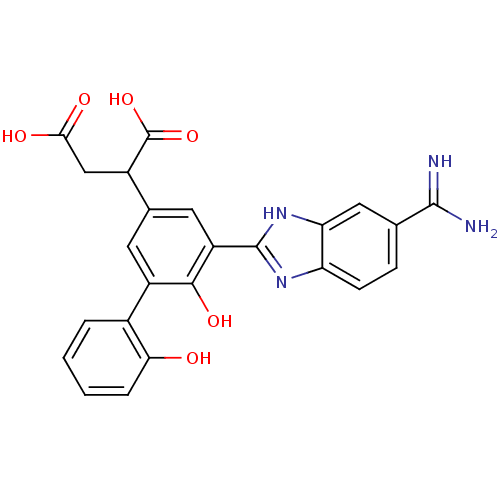
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(c1O)-c1ccccc1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H20N4O6/c25-22(26)11-5-6-17-18(9-11)28-23(27-17)16-8-12(14(24(33)34)10-20(30)31)7-15(21(16)32)13-3-1-2-4-19(13)29/h1-9,14,29,32H,10H2,(H3,25,26)(H,27,28)(H,30,31)(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14903

(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(c1O)-c1ccccc1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H20N4O6/c25-22(26)11-5-6-17-18(9-11)28-23(27-17)16-8-12(14(24(33)34)10-20(30)31)7-15(21(16)32)13-3-1-2-4-19(13)29/h1-9,14,29,32H,10H2,(H3,25,26)(H,27,28)(H,30,31)(H,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 13 | -44.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14905
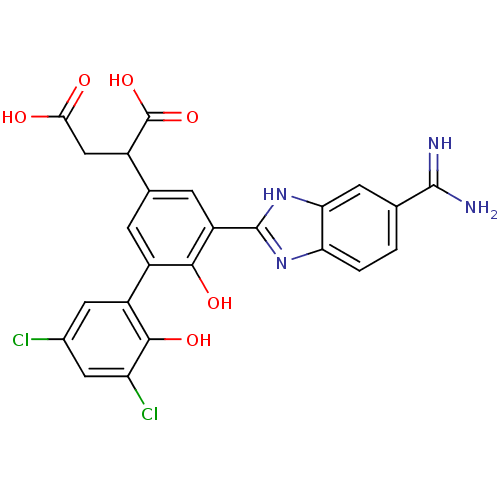
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-5-(...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(c1O)-c1cc(Cl)cc(Cl)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H18Cl2N4O6/c25-11-6-14(21(34)16(26)7-11)13-3-10(12(24(35)36)8-19(31)32)4-15(20(13)33)23-29-17-2-1-9(22(27)28)5-18(17)30-23/h1-7,12,33-34H,8H2,(H3,27,28)(H,29,30)(H,31,32)(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 14 | -44.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM14937

(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES Cc1ccc(cc1)-c1cc(cc(-c2nc3ccc(cc3[nH]2)C(N)=N)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C25H22N4O5/c1-12-2-4-13(5-3-12)16-8-15(17(25(33)34)11-21(30)31)9-18(22(16)32)24-28-19-7-6-14(23(26)27)10-20(19)29-24/h2-10,17,32H,11H2,1H3,(H3,26,27)(H,28,29)(H,30,31)(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101878
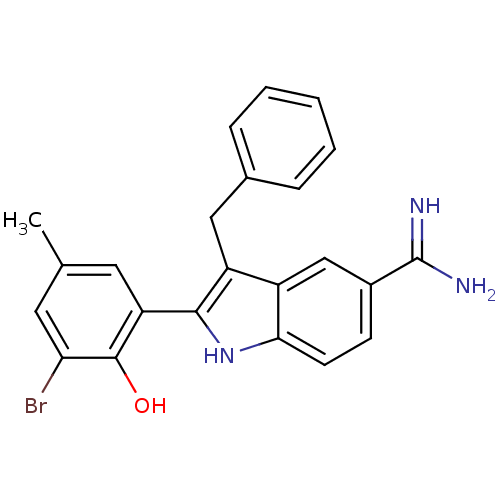
(3-Benzyl-2-(3-bromo-2-hydroxy-5-methyl-phenyl)-1H-...)Show SMILES Cc1cc(Br)c(O)c(c1)-c1[nH]c2ccc(cc2c1Cc1ccccc1)C(N)=N Show InChI InChI=1S/C23H20BrN3O/c1-13-9-18(22(28)19(24)10-13)21-17(11-14-5-3-2-4-6-14)16-12-15(23(25)26)7-8-20(16)27-21/h2-10,12,27-28H,11H2,1H3,(H3,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Coagulation factor X in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50101868
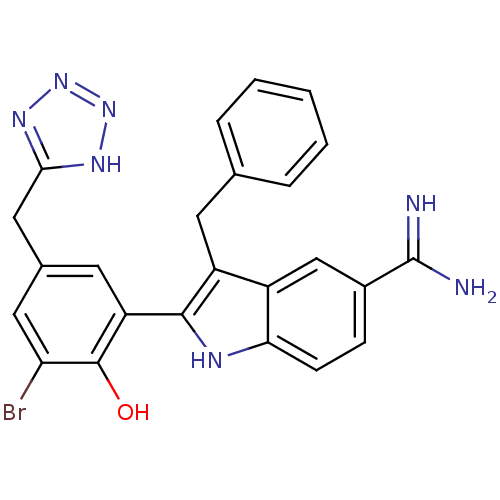
(3-Benzyl-2-[3-bromo-2-hydroxy-5-(2H-tetrazol-5-ylm...)Show SMILES NC(=N)c1ccc2[nH]c(c(Cc3ccccc3)c2c1)-c1cc(Cc2nnn[nH]2)cc(Br)c1O Show InChI InChI=1S/C24H20BrN7O/c25-19-10-14(11-21-29-31-32-30-21)9-18(23(19)33)22-17(8-13-4-2-1-3-5-13)16-12-15(24(26)27)6-7-20(16)28-22/h1-7,9-10,12,28,33H,8,11H2,(H3,26,27)(H,29,30,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against Coagulation factor X in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14907
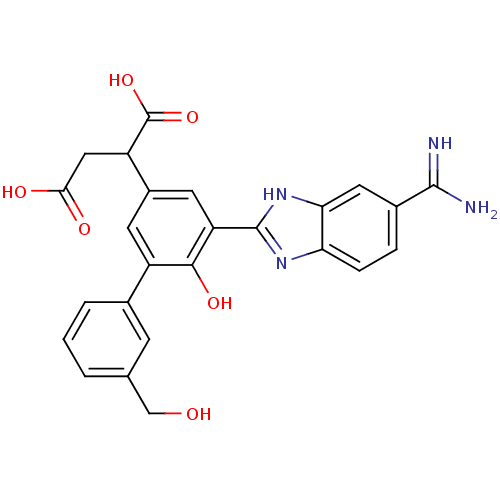
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(-c2cccc(CO)c2)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C25H22N4O6/c26-23(27)14-4-5-19-20(9-14)29-24(28-19)18-8-15(17(25(34)35)10-21(31)32)7-16(22(18)33)13-3-1-2-12(6-13)11-30/h1-9,17,30,33H,10-11H2,(H3,26,27)(H,28,29)(H,31,32)(H,34,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 21 | -43.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14909
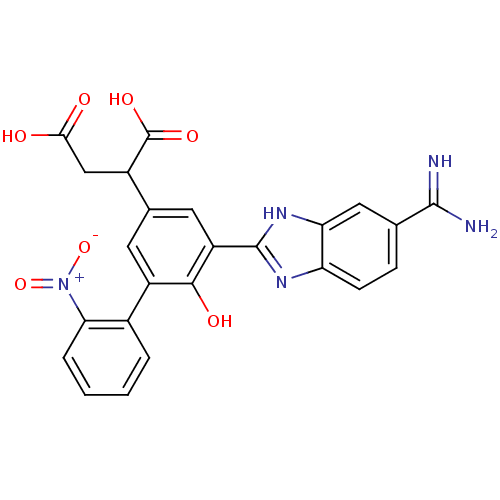
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(c1O)-c1ccccc1[N+]([O-])=O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H19N5O7/c25-22(26)11-5-6-17-18(9-11)28-23(27-17)16-8-12(14(24(33)34)10-20(30)31)7-15(21(16)32)13-3-1-2-4-19(13)29(35)36/h1-9,14,32H,10H2,(H3,25,26)(H,27,28)(H,30,31)(H,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 22 | -43.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14908
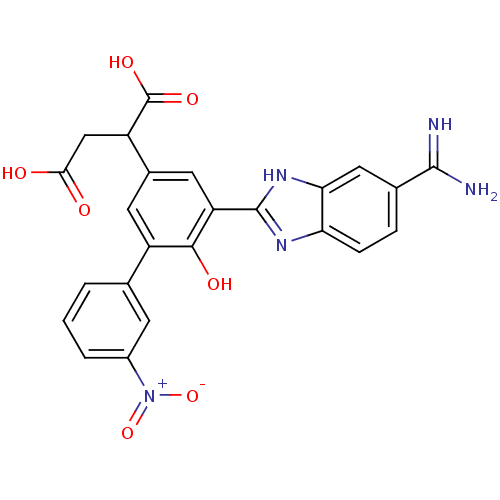
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(-c2cccc(c2)[N+]([O-])=O)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H19N5O7/c25-22(26)12-4-5-18-19(9-12)28-23(27-18)17-8-13(16(24(33)34)10-20(30)31)7-15(21(17)32)11-2-1-3-14(6-11)29(35)36/h1-9,16,32H,10H2,(H3,25,26)(H,27,28)(H,30,31)(H,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 22 | -43.3 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14906
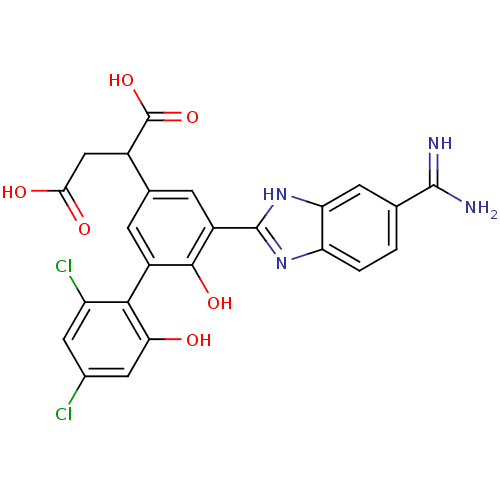
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-5-(...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(c1O)-c1c(O)cc(Cl)cc1Cl)C(CC(O)=O)C(O)=O |(-12.53,-2.27,;-11.93,-.85,;-12.87,.37,;-10.41,-.66,;-9.47,-1.88,;-7.95,-1.68,;-7.35,-.26,;-5.9,.25,;-5.93,1.78,;-7.41,2.23,;-8.29,.96,;-9.81,.77,;-4.6,2.55,;-4.6,4.1,;-3.27,4.87,;-1.93,4.1,;-1.93,2.55,;-3.27,1.78,;-3.27,.24,;-.6,1.78,;.73,2.56,;.73,4.1,;2.07,1.78,;2.07,.24,;3.4,-.53,;.73,-.53,;-.6,.24,;-1.93,-.53,;-3.27,6.41,;-1.93,7.18,;-.6,6.41,;-.6,4.87,;.73,7.18,;-4.6,7.18,;-5.93,6.41,;-4.6,8.72,)| Show InChI InChI=1S/C24H18Cl2N4O6/c25-11-6-15(26)20(18(31)7-11)13-3-10(12(24(35)36)8-19(32)33)4-14(21(13)34)23-29-16-2-1-9(22(27)28)5-17(16)30-23/h1-7,12,31,34H,8H2,(H3,27,28)(H,29,30)(H,32,33)(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 25 | -43.0 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50101885

(2-[3-(3-Benzyl-5-carbamimidoyl-1H-indol-2-yl)-5-br...)Show SMILES NC(=O)Cc1cc(Br)c(O)c(c1)-c1[nH]c2ccc(cc2c1Cc1ccccc1)C(N)=N Show InChI InChI=1S/C24H21BrN4O2/c25-19-10-14(11-21(26)30)9-18(23(19)31)22-17(8-13-4-2-1-3-5-13)16-12-15(24(27)28)6-7-20(16)29-22/h1-7,9-10,12,29,31H,8,11H2,(H2,26,30)(H3,27,28) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against thrombin(fIIa) in human plasma |
Bioorg Med Chem Lett 11: 1797-800 (2001)
BindingDB Entry DOI: 10.7270/Q20K27TW |
More data for this
Ligand-Target Pair | |
Coagulation factor VII
(Homo sapiens (Human)) | BDBM14910
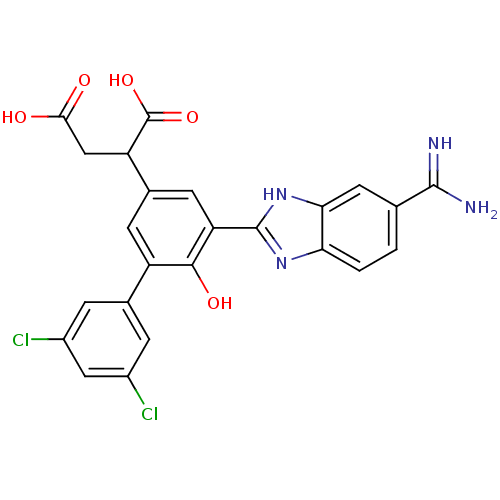
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-5-(...)Show SMILES NC(=N)c1ccc2nc([nH]c2c1)-c1cc(cc(-c2cc(Cl)cc(Cl)c2)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C24H18Cl2N4O5/c25-13-3-11(4-14(26)8-13)15-5-12(16(24(34)35)9-20(31)32)6-17(21(15)33)23-29-18-2-1-10(22(27)28)7-19(18)30-23/h1-8,16,33H,9H2,(H3,27,28)(H,29,30)(H,31,32)(H,34,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 27 | -42.8 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera
| Assay Description
The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... |
Bioorg Med Chem Lett 16: 1596-600 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.040
BindingDB Entry DOI: 10.7270/Q2VX0DSZ |
More data for this
Ligand-Target Pair | |
Plasma kallikrein
(Homo sapiens (Human)) | BDBM14944
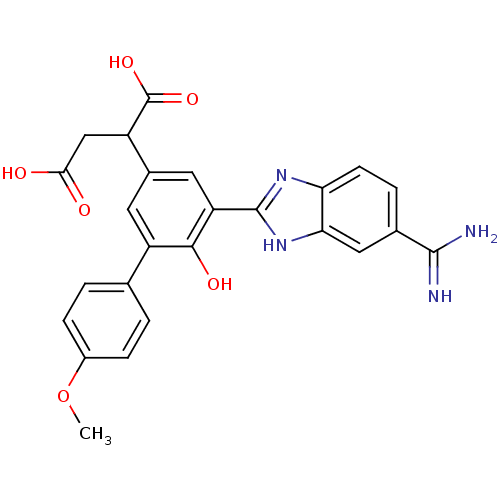
(2-[3-(5-carbamimidoyl-1H-1,3-benzodiazol-2-yl)-4-h...)Show SMILES COc1ccc(cc1)-c1cc(cc(-c2nc3ccc(cc3[nH]2)C(N)=N)c1O)C(CC(O)=O)C(O)=O Show InChI InChI=1S/C25H22N4O6/c1-35-15-5-2-12(3-6-15)16-8-14(17(25(33)34)11-21(30)31)9-18(22(16)32)24-28-19-7-4-13(23(26)27)10-20(19)29-24/h2-10,17,32H,11H2,1H3,(H3,26,27)(H,28,29)(H,30,31)(H,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Celera Genomics
Curated by ChEMBL
| Assay Description
Binding affinity to plasma kallikrein |
Bioorg Med Chem Lett 16: 2034-6 (2006)
Article DOI: 10.1016/j.bmcl.2005.12.060
BindingDB Entry DOI: 10.7270/Q2P55N3N |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data