

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adenylate cyclase type 4 (Homo sapiens (Human)) | BDBM50226415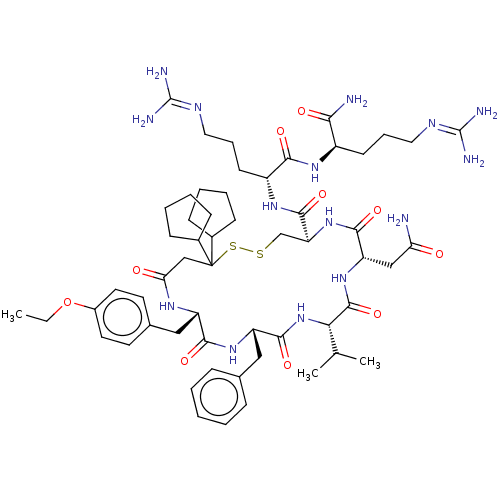 (CHEMBL3142312) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.880 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research and Development Division Curated by ChEMBL | Assay Description Binding affinity for dopamine receptor D2 was evaluated by the ability to displace [3H]spiperone | J Med Chem 30: 2291-4 (1987) BindingDB Entry DOI: 10.7270/Q2MG7RRN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50372608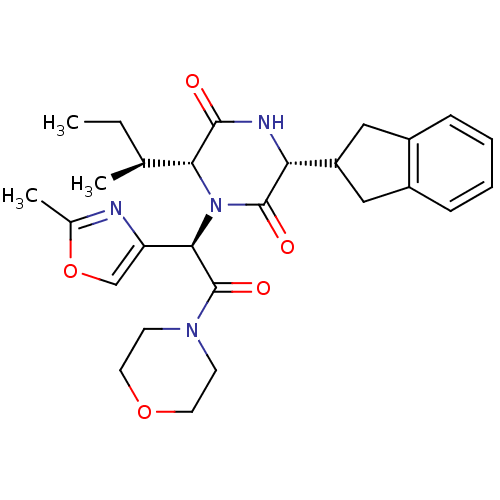 (CHEMBL429736 | GSK-221149A) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity to human oxytocin receptor | Bioorg Med Chem Lett 18: 90-4 (2008) Article DOI: 10.1016/j.bmcl.2007.11.008 BindingDB Entry DOI: 10.7270/Q2XD12H2 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50414549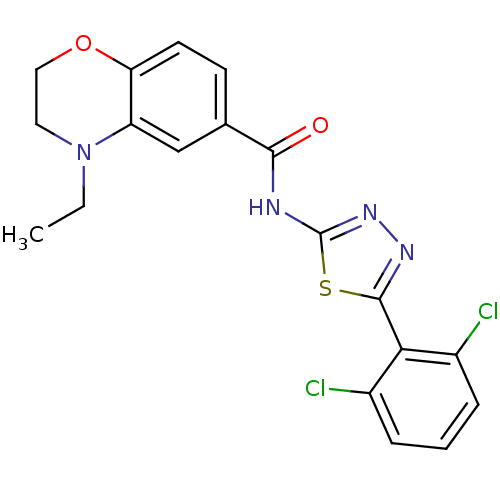 (CHEMBL563480) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in cells assessed as mobilization of intracellular calcium by FLIPR assay | Bioorg Med Chem Lett 19: 4292-5 (2009) Article DOI: 10.1016/j.bmcl.2009.05.074 BindingDB Entry DOI: 10.7270/Q2SQ90DS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50414547 (CHEMBL558644) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in cells assessed as mobilization of intracellular calcium by FLIPR assay | Bioorg Med Chem Lett 19: 4292-5 (2009) Article DOI: 10.1016/j.bmcl.2009.05.074 BindingDB Entry DOI: 10.7270/Q2SQ90DS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenylate cyclase type 4 (Homo sapiens (Human)) | BDBM50226410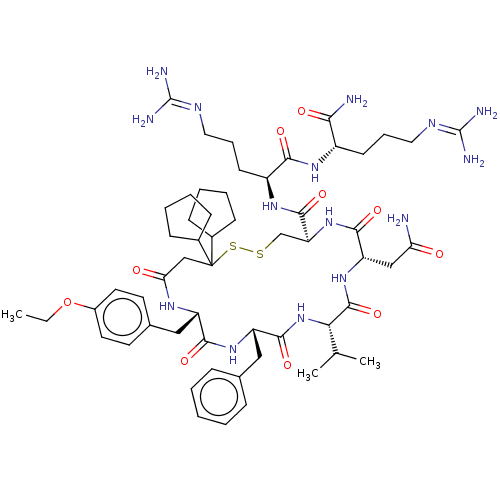 (CHEMBL3142318) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research and Development Division Curated by ChEMBL | Assay Description Binding affinity for dopamine receptor D2 was evaluated by the ability to displace [3H]spiperone | J Med Chem 30: 2291-4 (1987) BindingDB Entry DOI: 10.7270/Q2MG7RRN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenylate cyclase type 4 (Homo sapiens (Human)) | BDBM50226412 (CHEMBL3142332) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research and Development Division Curated by ChEMBL | Assay Description Binding affinity for dopamine receptor D2 was evaluated by the ability to displace [3H]spiperone | J Med Chem 30: 2291-4 (1987) BindingDB Entry DOI: 10.7270/Q2MG7RRN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenylate cyclase type 4 (Homo sapiens (Human)) | BDBM50226411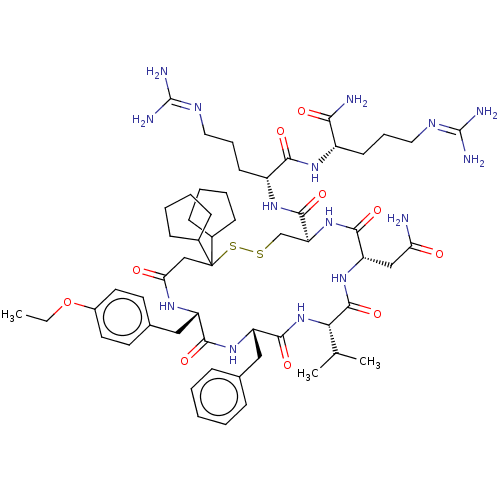 (CHEMBL3142329) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research and Development Division Curated by ChEMBL | Assay Description Binding affinity against sigma receptor | J Med Chem 30: 2291-4 (1987) BindingDB Entry DOI: 10.7270/Q2MG7RRN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenylate cyclase type 4 (Homo sapiens (Human)) | BDBM50226417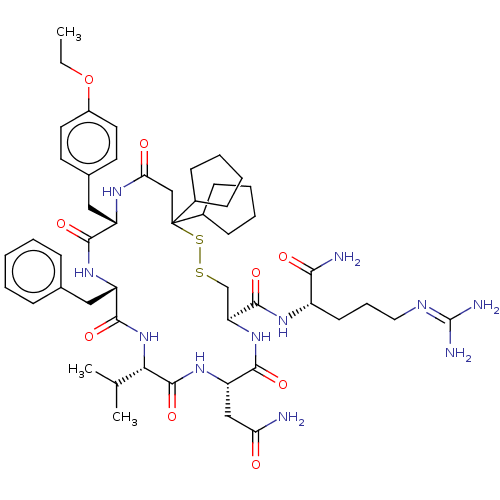 (CHEMBL3142331) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research and Development Division Curated by ChEMBL | Assay Description Binding affinity for dopamine receptor D2 was evaluated by the ability to displace [3H]spiperone | J Med Chem 30: 2291-4 (1987) BindingDB Entry DOI: 10.7270/Q2MG7RRN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenylate cyclase type 4 (Homo sapiens (Human)) | BDBM50226413 (CHEMBL2369777) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research and Development Division Curated by ChEMBL | Assay Description Binding affinity for dopamine receptor D2 was evaluated by the ability to displace [3H]spiperone | J Med Chem 30: 2291-4 (1987) BindingDB Entry DOI: 10.7270/Q2MG7RRN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (RAT) | BDBM50384444 (CHEMBL2035510) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3.16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Antagonist activity at rat EP3 receptor expressed in human U2OS cells co-expressing Gqi5 assessed as inhibition of PGE2-induced response after 24 hrs... | ACS Med Chem Lett 1: 316-320 (2010) Article DOI: 10.1021/ml100077x BindingDB Entry DOI: 10.7270/Q2BG2Q1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50414536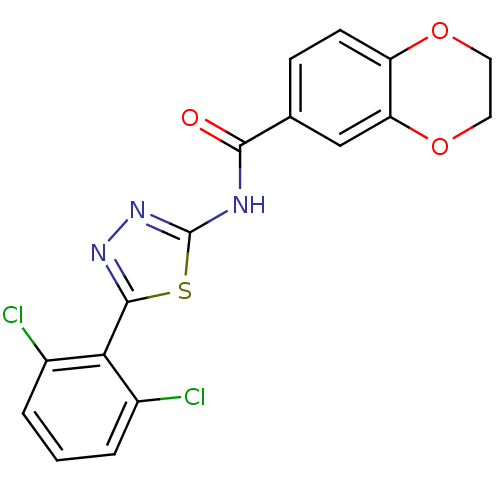 (CHEMBL551202) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3.16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in cells assessed as mobilization of intracellular calcium by FLIPR assay | Bioorg Med Chem Lett 19: 4292-5 (2009) Article DOI: 10.1016/j.bmcl.2009.05.074 BindingDB Entry DOI: 10.7270/Q2SQ90DS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50384444 (CHEMBL2035510) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 3.16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Antagonist activity at human EP3c receptor expressed in human U2OS cells assessed as inhibition of PGE2-induced calcium mobilization after 24 hrs by ... | ACS Med Chem Lett 1: 316-320 (2010) Article DOI: 10.1021/ml100077x BindingDB Entry DOI: 10.7270/Q2BG2Q1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Sus scrofa) | BDBM50020654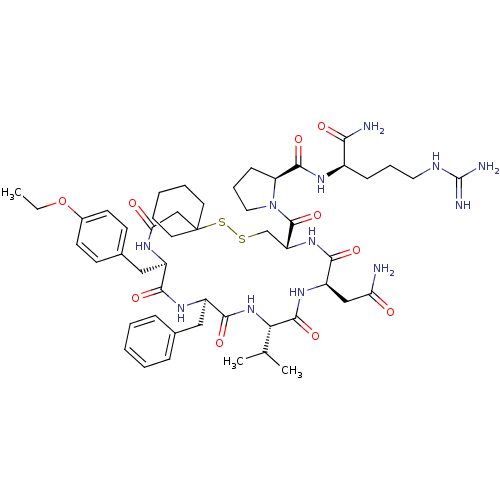 (1-[19-Benzyl-13-carbamoylmethyl-22-(4-ethoxy-benzy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was tested for inhibition against V2 vasopressin receptor in pig renal medullary membrane preparations. | J Med Chem 29: 2425-6 (1987) BindingDB Entry DOI: 10.7270/Q23T9G6Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenylate cyclase type 4 (Homo sapiens (Human)) | BDBM50226416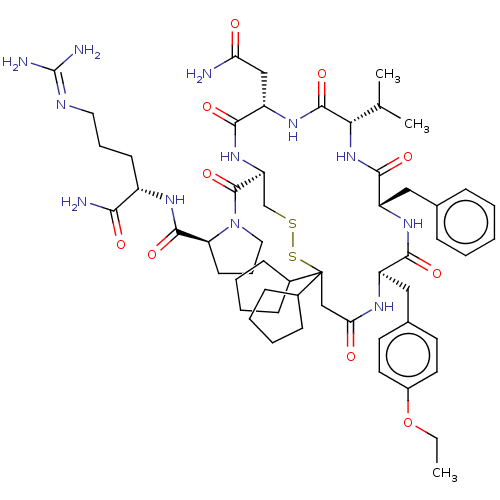 (CHEMBL2369525) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 3.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research and Development Division Curated by ChEMBL | Assay Description Binding affinity for dopamine receptor D2 was evaluated by the ability to displace [3H]spiperone | J Med Chem 30: 2291-4 (1987) BindingDB Entry DOI: 10.7270/Q2MG7RRN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50372608 (CHEMBL429736 | GSK-221149A) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 4.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity to recombinant oxytocin receptor | Bioorg Med Chem Lett 18: 90-4 (2008) Article DOI: 10.1016/j.bmcl.2007.11.008 BindingDB Entry DOI: 10.7270/Q2XD12H2 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Adenylate cyclase type 4 (Homo sapiens (Human)) | BDBM50226414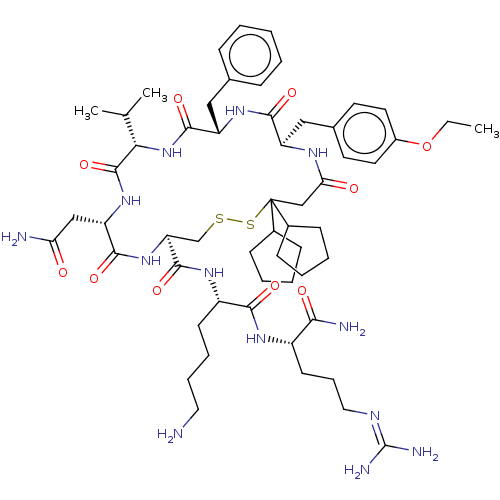 (CHEMBL2369778) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research and Development Division Curated by ChEMBL | Assay Description Binding affinity against sigma receptor | J Med Chem 30: 2291-4 (1987) BindingDB Entry DOI: 10.7270/Q2MG7RRN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50384443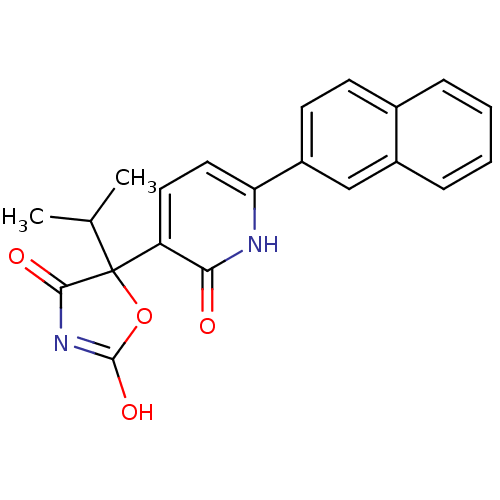 (CHEMBL1770317) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.01 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Binding affinity to human EP3 | ACS Med Chem Lett 1: 316-320 (2010) Article DOI: 10.1021/ml100077x BindingDB Entry DOI: 10.7270/Q2BG2Q1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50414543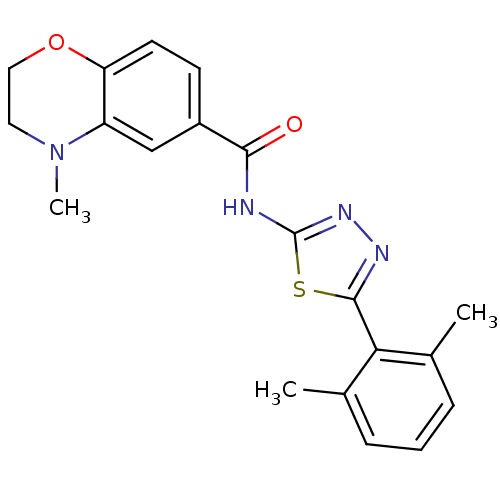 (CHEMBL550316) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.01 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in cells assessed as mobilization of intracellular calcium by FLIPR assay | Bioorg Med Chem Lett 19: 4292-5 (2009) Article DOI: 10.1016/j.bmcl.2009.05.074 BindingDB Entry DOI: 10.7270/Q2SQ90DS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Sus scrofa) | BDBM50020675 (1-[13-Benzyl-7-carbamoylmethyl-16-(4-ethoxy-benzyl...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was tested for inhibition against V2 vasopressin receptor in pig renal medullary membrane preparations. | J Med Chem 29: 2425-6 (1987) BindingDB Entry DOI: 10.7270/Q23T9G6Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50483809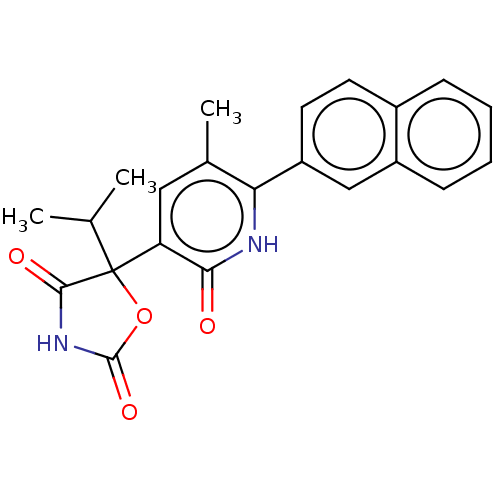 (CHEMBL1770337) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in CHO cells assessed as inhibition of PGE2-induced increase in intracellular calcium concentrati... | Bioorg Med Chem Lett 21: 2806-11 (2011) Article DOI: 10.1016/j.bmcl.2011.03.107 BindingDB Entry DOI: 10.7270/Q2NZ8BGM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50384445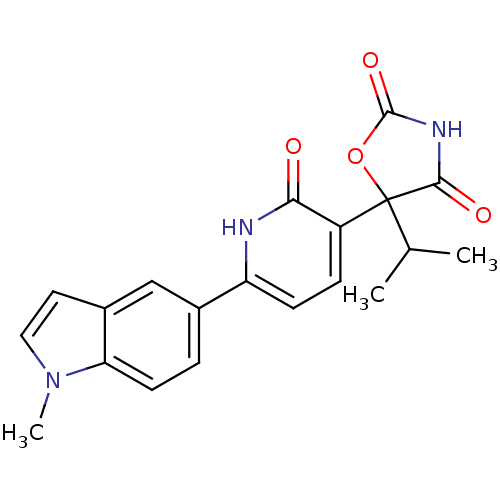 (CHEMBL2035508) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.31 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Antagonist activity at human EP3c receptor expressed in human U2OS cells assessed as inhibition of PGE2-induced calcium mobilization after 24 hrs by ... | ACS Med Chem Lett 1: 316-320 (2010) Article DOI: 10.1021/ml100077x BindingDB Entry DOI: 10.7270/Q2BG2Q1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (RAT) | BDBM50414549 (CHEMBL563480) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.31 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Inhibition of rat EP3 receptor | Bioorg Med Chem Lett 19: 4292-5 (2009) Article DOI: 10.1016/j.bmcl.2009.05.074 BindingDB Entry DOI: 10.7270/Q2SQ90DS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50417191 (CHEMBL1272136) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.31 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in human U2OS cells assessed as inhibition of PGE2-induced intracellular calcium mobilization by ... | Bioorg Med Chem Lett 20: 6744-7 (2010) Article DOI: 10.1016/j.bmcl.2010.08.137 BindingDB Entry DOI: 10.7270/Q25M65Z8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Sus scrofa) | BDBM50020653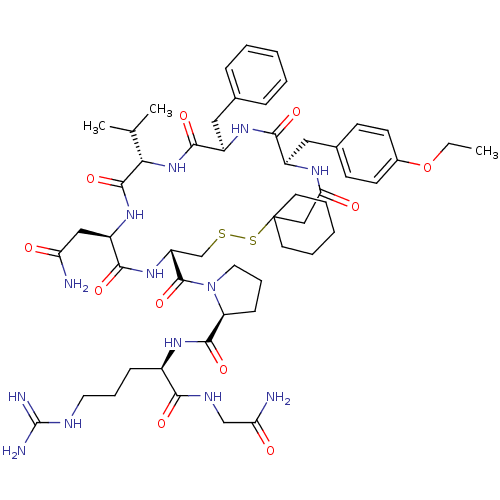 (1-[19-Benzyl-13-carbamoylmethyl-22-(4-ethoxy-benzy...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 6.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was tested for inhibition against V2 vasopressin receptor in pig renal medullary membrane preparations. | J Med Chem 29: 2425-6 (1987) BindingDB Entry DOI: 10.7270/Q23T9G6Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenylate cyclase type 4 (Homo sapiens (Human)) | BDBM50226418 (CHEMBL3142313) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 7.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research and Development Division Curated by ChEMBL | Assay Description Binding affinity for dopamine receptor D2 was evaluated by the ability to displace [3H]spiperone | J Med Chem 30: 2291-4 (1987) BindingDB Entry DOI: 10.7270/Q2MG7RRN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50483823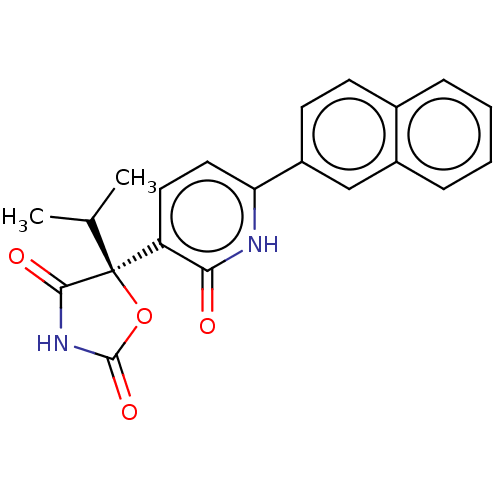 (CHEMBL1770341) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in CHO cells assessed as inhibition of PGE2-induced increase in intracellular calcium concentrati... | Bioorg Med Chem Lett 21: 2806-11 (2011) Article DOI: 10.1016/j.bmcl.2011.03.107 BindingDB Entry DOI: 10.7270/Q2NZ8BGM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50483821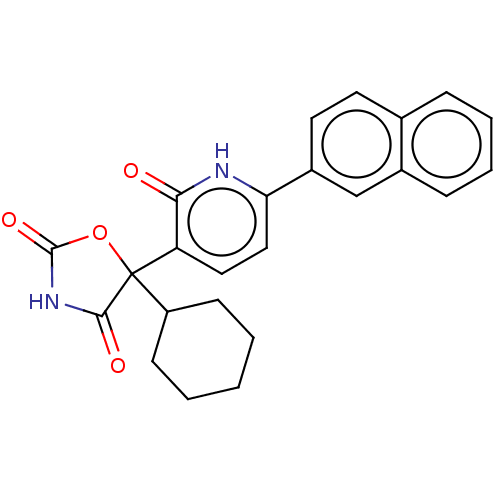 (CHEMBL1770320) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in CHO cells assessed as inhibition of PGE2-induced increase in intracellular calcium concentrati... | Bioorg Med Chem Lett 21: 2806-11 (2011) Article DOI: 10.1016/j.bmcl.2011.03.107 BindingDB Entry DOI: 10.7270/Q2NZ8BGM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50483814 (CHEMBL1770319) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in CHO cells assessed as inhibition of PGE2-induced increase in intracellular calcium concentrati... | Bioorg Med Chem Lett 21: 2806-11 (2011) Article DOI: 10.1016/j.bmcl.2011.03.107 BindingDB Entry DOI: 10.7270/Q2NZ8BGM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (RAT) | BDBM50384445 (CHEMBL2035508) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.94 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Antagonist activity at rat EP3 receptor expressed in human U2OS cells co-expressing Gqi5 assessed as inhibition of PGE2-induced response after 24 hrs... | ACS Med Chem Lett 1: 316-320 (2010) Article DOI: 10.1021/ml100077x BindingDB Entry DOI: 10.7270/Q2BG2Q1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50414548 (CHEMBL550055) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.94 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in cells assessed as mobilization of intracellular calcium by FLIPR assay | Bioorg Med Chem Lett 19: 4292-5 (2009) Article DOI: 10.1016/j.bmcl.2009.05.074 BindingDB Entry DOI: 10.7270/Q2SQ90DS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Sus scrofa) | BDBM50020674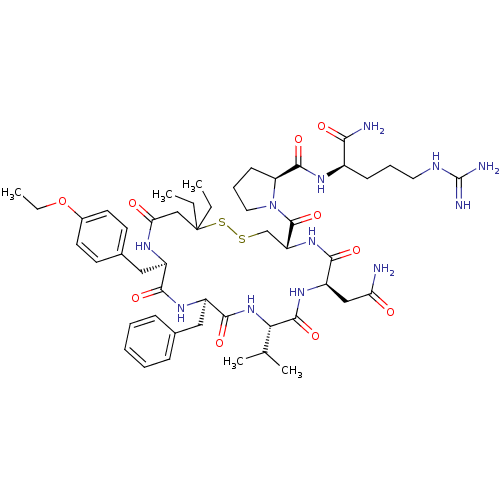 (1-[13-Benzyl-7-carbamoylmethyl-16-(4-ethoxy-benzyl...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 8.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was tested for inhibition against V2 vasopressin receptor in pig renal medullary membrane preparations. | J Med Chem 29: 2425-6 (1987) BindingDB Entry DOI: 10.7270/Q23T9G6Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50384443 (CHEMBL1770317) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Antagonist activity at human EP3c receptor expressed in human U2OS cells assessed as inhibition of PGE2-induced calcium mobilization after 24 hrs by ... | ACS Med Chem Lett 1: 316-320 (2010) Article DOI: 10.1021/ml100077x BindingDB Entry DOI: 10.7270/Q2BG2Q1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (RAT) | BDBM50384443 (CHEMBL1770317) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Antagonist activity at rat EP3 receptor expressed in human U2OS cells co-expressing Gqi5 assessed as inhibition of PGE2-induced response after 24 hrs... | ACS Med Chem Lett 1: 316-320 (2010) Article DOI: 10.1021/ml100077x BindingDB Entry DOI: 10.7270/Q2BG2Q1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50384443 (CHEMBL1770317) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in CHO cells assessed as inhibition of PGE2-induced increase in intracellular calcium concentrati... | Bioorg Med Chem Lett 21: 2806-11 (2011) Article DOI: 10.1016/j.bmcl.2011.03.107 BindingDB Entry DOI: 10.7270/Q2NZ8BGM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50177595 ((2S)-5-amino-2-{[(2S)-1-{[(4R,7S,10S,13S,16R)-13-[...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity to human oxytocin receptor | Bioorg Med Chem Lett 18: 90-4 (2008) Article DOI: 10.1016/j.bmcl.2007.11.008 BindingDB Entry DOI: 10.7270/Q2XD12H2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50414537 (CHEMBL554297) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 12.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in cells assessed as mobilization of intracellular calcium by FLIPR assay | Bioorg Med Chem Lett 19: 4292-5 (2009) Article DOI: 10.1016/j.bmcl.2009.05.074 BindingDB Entry DOI: 10.7270/Q2SQ90DS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50414533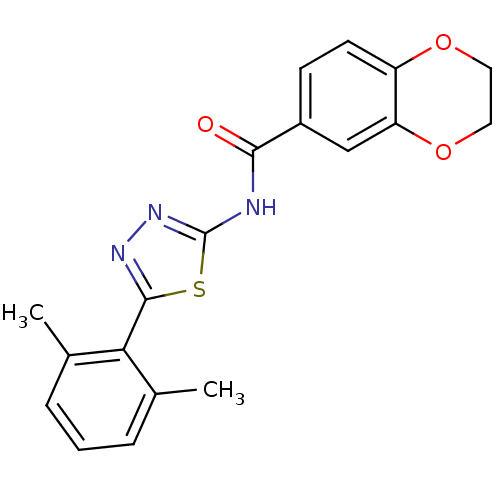 (CHEMBL549437) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 12.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in cells assessed as mobilization of intracellular calcium by FLIPR assay | Bioorg Med Chem Lett 19: 4292-5 (2009) Article DOI: 10.1016/j.bmcl.2009.05.074 BindingDB Entry DOI: 10.7270/Q2SQ90DS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (RAT) | BDBM50384446 (CHEMBL2035509) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 12.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Antagonist activity at rat EP3 receptor expressed in human U2OS cells co-expressing Gqi5 assessed as inhibition of PGE2-induced response after 24 hrs... | ACS Med Chem Lett 1: 316-320 (2010) Article DOI: 10.1021/ml100077x BindingDB Entry DOI: 10.7270/Q2BG2Q1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50414544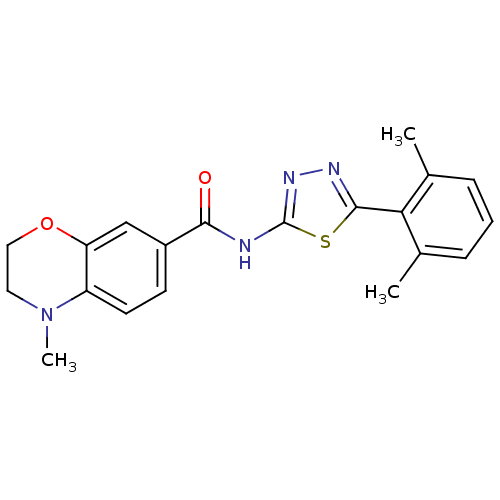 (CHEMBL562329) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 15.8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in cells assessed as mobilization of intracellular calcium by FLIPR assay | Bioorg Med Chem Lett 19: 4292-5 (2009) Article DOI: 10.1016/j.bmcl.2009.05.074 BindingDB Entry DOI: 10.7270/Q2SQ90DS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50483827 (CHEMBL1770322) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in CHO cells assessed as inhibition of PGE2-induced increase in intracellular calcium concentrati... | Bioorg Med Chem Lett 21: 2806-11 (2011) Article DOI: 10.1016/j.bmcl.2011.03.107 BindingDB Entry DOI: 10.7270/Q2NZ8BGM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50384446 (CHEMBL2035509) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 19.9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Antagonist activity at human EP3c receptor expressed in human U2OS cells assessed as inhibition of PGE2-induced calcium mobilization after 24 hrs by ... | ACS Med Chem Lett 1: 316-320 (2010) Article DOI: 10.1021/ml100077x BindingDB Entry DOI: 10.7270/Q2BG2Q1F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50414534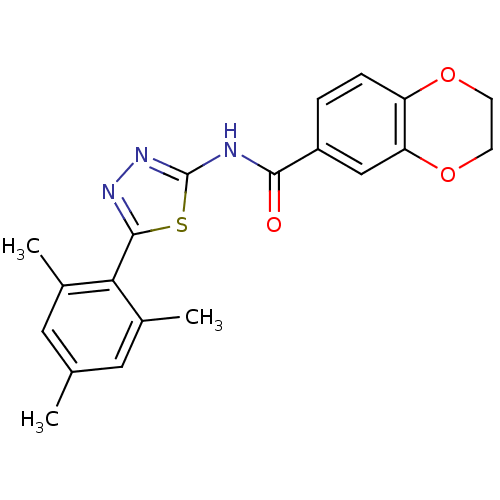 (CHEMBL564190) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 19.9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in cells assessed as mobilization of intracellular calcium by FLIPR assay | Bioorg Med Chem Lett 19: 4292-5 (2009) Article DOI: 10.1016/j.bmcl.2009.05.074 BindingDB Entry DOI: 10.7270/Q2SQ90DS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50483811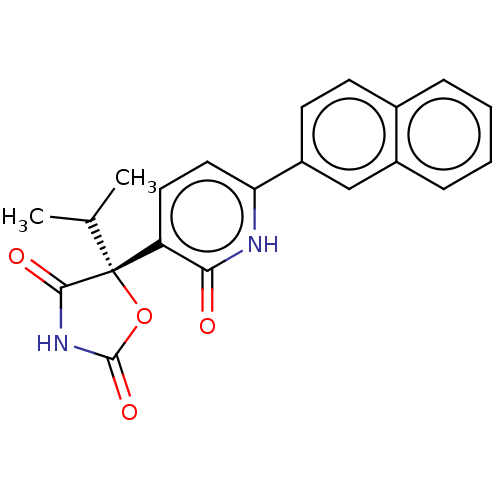 (CHEMBL1770340) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid UniChem | Article PubMed | 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in CHO cells assessed as inhibition of PGE2-induced increase in intracellular calcium concentrati... | Bioorg Med Chem Lett 21: 2806-11 (2011) Article DOI: 10.1016/j.bmcl.2011.03.107 BindingDB Entry DOI: 10.7270/Q2NZ8BGM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50483812 (CHEMBL1770316) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in CHO cells assessed as inhibition of PGE2-induced increase in intracellular calcium concentrati... | Bioorg Med Chem Lett 21: 2806-11 (2011) Article DOI: 10.1016/j.bmcl.2011.03.107 BindingDB Entry DOI: 10.7270/Q2NZ8BGM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50329821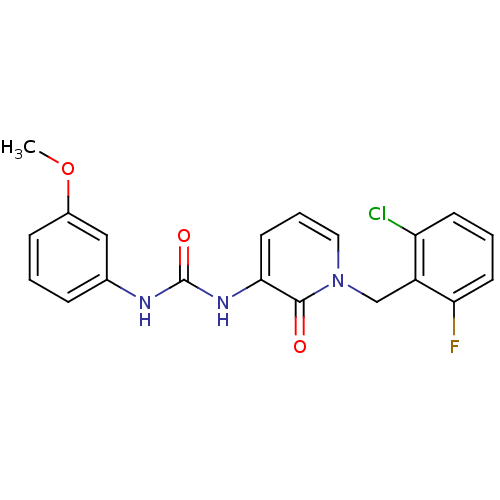 (1-(1-(2-chloro-6-fluorobenzyl)-2-oxo-1,2-dihydropy...) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 25.1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in human U2OS cells assessed as inhibition of PGE2-induced intracellular calcium mobilization by ... | Bioorg Med Chem Lett 20: 6744-7 (2010) Article DOI: 10.1016/j.bmcl.2010.08.137 BindingDB Entry DOI: 10.7270/Q25M65Z8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50414542 (CHEMBL562994) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 25.1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Pharmaceuticals Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in cells assessed as mobilization of intracellular calcium by FLIPR assay | Bioorg Med Chem Lett 19: 4292-5 (2009) Article DOI: 10.1016/j.bmcl.2009.05.074 BindingDB Entry DOI: 10.7270/Q2SQ90DS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50483815 (CHEMBL1770321) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in CHO cells assessed as inhibition of PGE2-induced increase in intracellular calcium concentrati... | Bioorg Med Chem Lett 21: 2806-11 (2011) Article DOI: 10.1016/j.bmcl.2011.03.107 BindingDB Entry DOI: 10.7270/Q2NZ8BGM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50177595 ((2S)-5-amino-2-{[(2S)-1-{[(4R,7S,10S,13S,16R)-13-[...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 32 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Research and Development Curated by ChEMBL | Assay Description Binding affinity to recombinant oxytocin receptor | Bioorg Med Chem Lett 18: 90-4 (2008) Article DOI: 10.1016/j.bmcl.2007.11.008 BindingDB Entry DOI: 10.7270/Q2XD12H2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50483818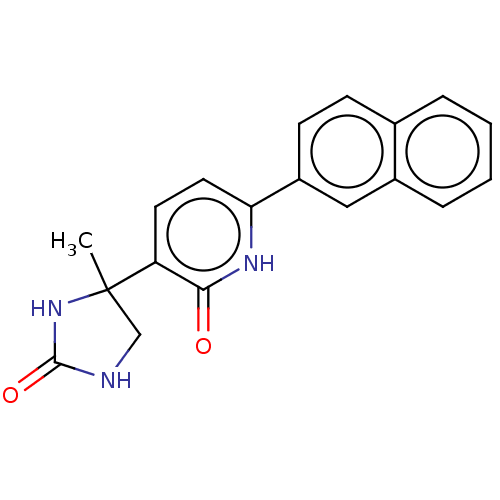 (CHEMBL1770328) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in CHO cells assessed as inhibition of PGE2-induced increase in intracellular calcium concentrati... | Bioorg Med Chem Lett 21: 2806-11 (2011) Article DOI: 10.1016/j.bmcl.2011.03.107 BindingDB Entry DOI: 10.7270/Q2NZ8BGM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prostaglandin E2 receptor EP3 subtype (Homo sapiens (Human)) | BDBM50483808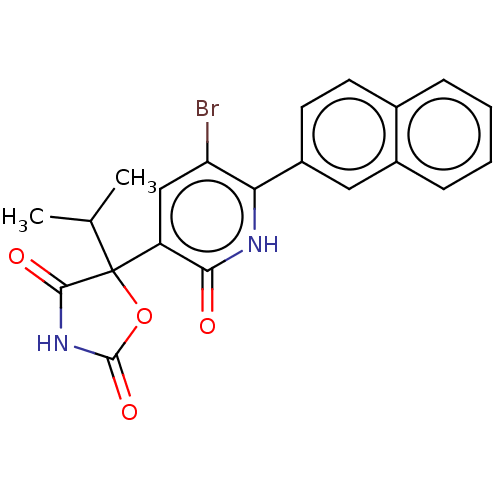 (CHEMBL1770336) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Antagonist activity at human EP3 receptor expressed in CHO cells assessed as inhibition of PGE2-induced increase in intracellular calcium concentrati... | Bioorg Med Chem Lett 21: 2806-11 (2011) Article DOI: 10.1016/j.bmcl.2011.03.107 BindingDB Entry DOI: 10.7270/Q2NZ8BGM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 213 total ) | Next | Last >> |