 Found 282 hits with Last Name = 'koyama' and Initial = 's'
Found 282 hits with Last Name = 'koyama' and Initial = 's' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Oxytocin receptor
(Homo sapiens (Human)) | BDBM50523555
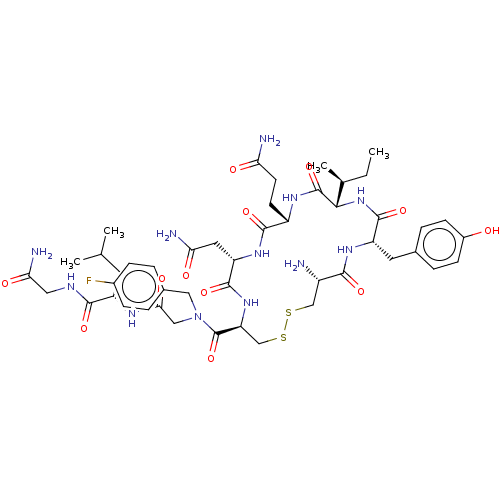
(CHEMBL4474284)Show SMILES CC[C@H](C)[C@@H]1NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)[C@@H](N)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC1=O)C(=O)N(CC(=O)N[C@@H](CC(C)C)C(=O)NCC(N)=O)Cc1ccc(F)cc1 |r| Show InChI InChI=1S/C47H67FN12O12S2/c1-5-25(4)40-46(71)55-31(14-15-36(50)62)43(68)57-34(18-37(51)63)44(69)58-35(23-74-73-22-30(49)41(66)56-33(45(70)59-40)17-26-8-12-29(61)13-9-26)47(72)60(20-27-6-10-28(48)11-7-27)21-39(65)54-32(16-24(2)3)42(67)53-19-38(52)64/h6-13,24-25,30-35,40,61H,5,14-23,49H2,1-4H3,(H2,50,62)(H2,51,63)(H2,52,64)(H,53,67)(H,54,65)(H,55,71)(H,56,66)(H,57,68)(H,58,69)(H,59,70)/t25-,30-,31-,32-,33-,34-,35-,40-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku University and Department of Pharmaceutical Sciences
Curated by ChEMBL
| Assay Description
Displacement of [3H]OT from recombinant human OTR expressed in HEK293 cell membranes measured after 1 hr |
J Med Chem 62: 3297-3310 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01691
BindingDB Entry DOI: 10.7270/Q2GF0XXQ |
More data for this
Ligand-Target Pair | |
Oxytocin receptor
(Homo sapiens (Human)) | BDBM50205990

(CHEMBL395429 | OXYTOCIN)Show SMILES CC[C@H](C)[C@@H]1NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)[C@@H](N)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)NCC(N)=O |r| Show InChI InChI=1S/C43H66N12O12S2/c1-5-22(4)35-42(66)49-26(12-13-32(45)57)38(62)51-29(17-33(46)58)39(63)53-30(20-69-68-19-25(44)36(60)50-28(40(64)54-35)16-23-8-10-24(56)11-9-23)43(67)55-14-6-7-31(55)41(65)52-27(15-21(2)3)37(61)48-18-34(47)59/h8-11,21-22,25-31,35,56H,5-7,12-20,44H2,1-4H3,(H2,45,57)(H2,46,58)(H2,47,59)(H,48,61)(H,49,66)(H,50,60)(H,51,62)(H,52,65)(H,53,63)(H,54,64)/t22-,25-,26-,27-,28-,29-,30-,31-,35-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku University and Department of Pharmaceutical Sciences
Curated by ChEMBL
| Assay Description
Displacement of [3H]OT from recombinant human OTR expressed in HEK293 cell membranes measured after 1 hr |
J Med Chem 62: 3297-3310 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01691
BindingDB Entry DOI: 10.7270/Q2GF0XXQ |
More data for this
Ligand-Target Pair | |
Oxytocin receptor
(Homo sapiens (Human)) | BDBM50523556
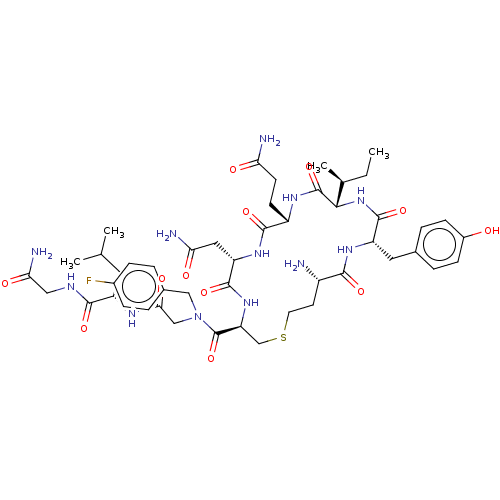
(CHEMBL4458988)Show SMILES CC[C@H](C)[C@@H]1NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)[C@@H](N)CCSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC1=O)C(=O)N(CC(=O)N[C@@H](CC(C)C)C(=O)NCC(N)=O)Cc1ccc(F)cc1 |r| Show InChI InChI=1S/C48H69FN12O12S/c1-5-26(4)41-47(72)56-32(14-15-37(51)63)44(69)58-35(20-38(52)64)45(70)59-36(24-74-17-16-31(50)42(67)57-34(46(71)60-41)19-27-8-12-30(62)13-9-27)48(73)61(22-28-6-10-29(49)11-7-28)23-40(66)55-33(18-25(2)3)43(68)54-21-39(53)65/h6-13,25-26,31-36,41,62H,5,14-24,50H2,1-4H3,(H2,51,63)(H2,52,64)(H2,53,65)(H,54,68)(H,55,66)(H,56,72)(H,57,67)(H,58,69)(H,59,70)(H,60,71)/t26-,31-,32-,33-,34-,35-,36-,41-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku University and Department of Pharmaceutical Sciences
Curated by ChEMBL
| Assay Description
Displacement of [3H]OT from recombinant human OTR expressed in HEK293 cell membranes measured after 1 hr |
J Med Chem 62: 3297-3310 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01691
BindingDB Entry DOI: 10.7270/Q2GF0XXQ |
More data for this
Ligand-Target Pair | |
Oxytocin receptor
(Homo sapiens (Human)) | BDBM50044686
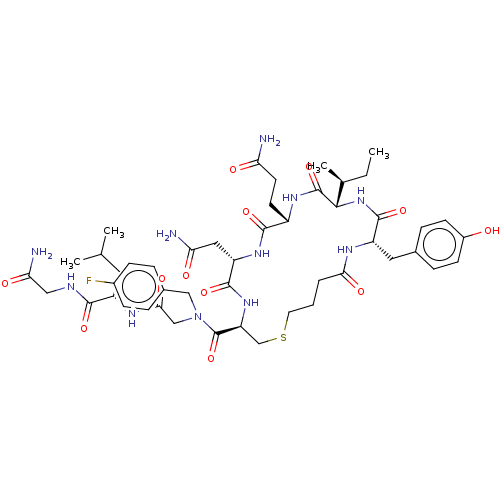
(CHEMBL3354594)Show SMILES [H][C@]1(NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCCSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC1=O)C(=O)N(CC(=O)N[C@@H](CC(C)C)C(=O)NCC(N)=O)Cc1ccc(F)cc1)[C@@H](C)CC |r| Show InChI InChI=1S/C48H68FN11O12S/c1-5-27(4)42-47(71)56-32(16-17-37(50)62)44(68)57-35(21-38(51)63)45(69)58-36(25-73-18-6-7-40(65)54-34(46(70)59-42)20-28-10-14-31(61)15-11-28)48(72)60(23-29-8-12-30(49)13-9-29)24-41(66)55-33(19-26(2)3)43(67)53-22-39(52)64/h8-15,26-27,32-36,42,61H,5-7,16-25H2,1-4H3,(H2,50,62)(H2,51,63)(H2,52,64)(H,53,67)(H,54,65)(H,55,66)(H,56,71)(H,57,68)(H,58,69)(H,59,70)/t27-,32-,33-,34-,35-,36-,42-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku University and Department of Pharmaceutical Sciences
Curated by ChEMBL
| Assay Description
Displacement of [3H]OT from recombinant human OTR expressed in HEK293 cell membranes measured after 1 hr |
J Med Chem 62: 3297-3310 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01691
BindingDB Entry DOI: 10.7270/Q2GF0XXQ |
More data for this
Ligand-Target Pair | |
Oxytocin receptor
(Homo sapiens (Human)) | BDBM50523557
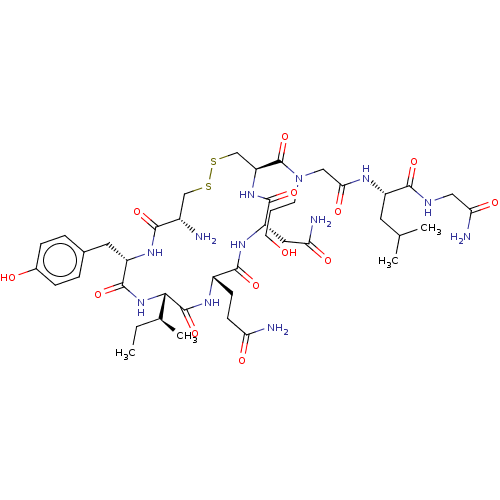
(CHEMBL4583231)Show SMILES CC[C@H](C)[C@@H]1NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)[C@@H](N)CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC1=O)C(=O)N(CCCO)CC(=O)N[C@@H](CC(C)C)C(=O)NCC(N)=O |r| Show InChI InChI=1S/C43H68N12O13S2/c1-5-23(4)36-42(67)50-27(11-12-32(45)58)39(64)52-30(17-33(46)59)40(65)53-31(21-70-69-20-26(44)37(62)51-29(41(66)54-36)16-24-7-9-25(57)10-8-24)43(68)55(13-6-14-56)19-35(61)49-28(15-22(2)3)38(63)48-18-34(47)60/h7-10,22-23,26-31,36,56-57H,5-6,11-21,44H2,1-4H3,(H2,45,58)(H2,46,59)(H2,47,60)(H,48,63)(H,49,61)(H,50,67)(H,51,62)(H,52,64)(H,53,65)(H,54,66)/t23-,26-,27-,28-,29-,30-,31-,36-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku University and Department of Pharmaceutical Sciences
Curated by ChEMBL
| Assay Description
Displacement of [3H]OT from recombinant human OTR expressed in HEK293 cell membranes measured after 1 hr |
J Med Chem 62: 3297-3310 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01691
BindingDB Entry DOI: 10.7270/Q2GF0XXQ |
More data for this
Ligand-Target Pair | |
Oxytocin receptor
(Homo sapiens (Human)) | BDBM50044742
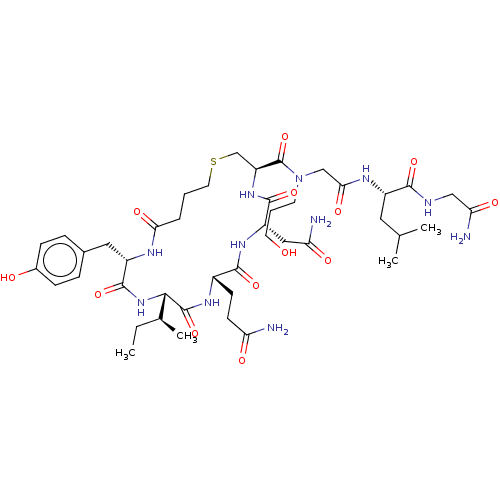
(CHEMBL3354579)Show SMILES [H][C@]1(NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCCSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC1=O)C(=O)N(CCCO)CC(=O)N[C@@H](CC(C)C)C(=O)NCC(N)=O)[C@@H](C)CC |r| Show InChI InChI=1S/C44H69N11O13S/c1-5-25(4)38-43(67)51-28(13-14-33(45)58)40(64)52-31(20-34(46)59)41(65)53-32(23-69-17-6-8-36(61)49-30(42(66)54-38)19-26-9-11-27(57)12-10-26)44(68)55(15-7-16-56)22-37(62)50-29(18-24(2)3)39(63)48-21-35(47)60/h9-12,24-25,28-32,38,56-57H,5-8,13-23H2,1-4H3,(H2,45,58)(H2,46,59)(H2,47,60)(H,48,63)(H,49,61)(H,50,62)(H,51,67)(H,52,64)(H,53,65)(H,54,66)/t25-,28-,29-,30-,31-,32-,38-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku University and Department of Pharmaceutical Sciences
Curated by ChEMBL
| Assay Description
Displacement of [3H]OT from recombinant human OTR expressed in HEK293 cell membranes measured after 1 hr |
J Med Chem 62: 3297-3310 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01691
BindingDB Entry DOI: 10.7270/Q2GF0XXQ |
More data for this
Ligand-Target Pair | |
Oxytocin receptor
(Homo sapiens (Human)) | BDBM50044677
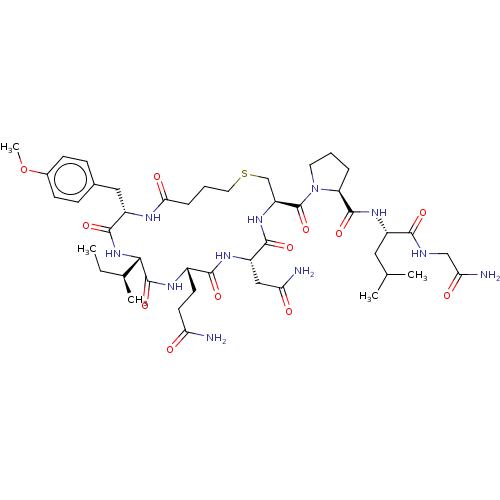
(CHEBI:59204 | Carbetocin)Show SMILES [H][C@]1(NC(=O)[C@H](Cc2ccc(OC)cc2)NC(=O)CCCSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)NCC(N)=O)[C@@H](C)CC Show InChI InChI=1S/C45H69N11O12S/c1-6-25(4)38-44(66)51-28(15-16-34(46)57)40(62)52-31(21-35(47)58)41(63)54-32(23-69-18-8-10-37(60)50-30(42(64)55-38)20-26-11-13-27(68-5)14-12-26)45(67)56-17-7-9-33(56)43(65)53-29(19-24(2)3)39(61)49-22-36(48)59/h11-14,24-25,28-33,38H,6-10,15-23H2,1-5H3,(H2,46,57)(H2,47,58)(H2,48,59)(H,49,61)(H,50,60)(H,51,66)(H,52,62)(H,53,65)(H,54,63)(H,55,64)/t25-,28-,29-,30-,31-,32-,33-,38-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku University and Department of Pharmaceutical Sciences
Curated by ChEMBL
| Assay Description
Displacement of [3H]OT from recombinant human OTR expressed in HEK293 cell membranes measured after 1 hr |
J Med Chem 62: 3297-3310 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01691
BindingDB Entry DOI: 10.7270/Q2GF0XXQ |
More data for this
Ligand-Target Pair | |
Oxytocin receptor
(Homo sapiens (Human)) | BDBM50044777
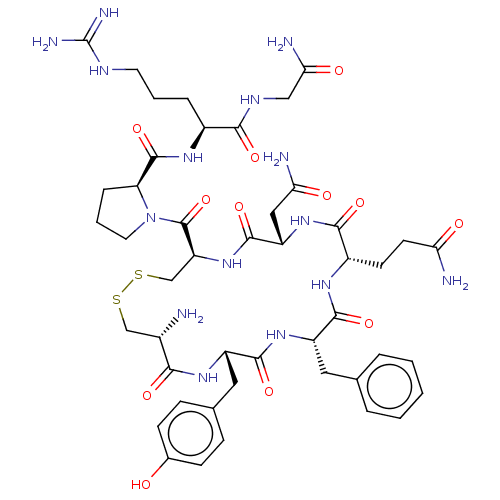
(Arginine Vasopressin | Beta-Hypophamine | CHEBI:34...)Show SMILES N[C@H]1CSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](Cc2ccc(O)cc2)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(N)=O Show InChI InChI=1S/C46H65N15O12S2/c47-27-22-74-75-23-33(45(73)61-17-5-9-34(61)44(72)56-28(8-4-16-53-46(51)52)39(67)54-21-37(50)65)60-43(71)32(20-36(49)64)59-40(68)29(14-15-35(48)63)55-41(69)31(18-24-6-2-1-3-7-24)58-42(70)30(57-38(27)66)19-25-10-12-26(62)13-11-25/h1-3,6-7,10-13,27-34,62H,4-5,8-9,14-23,47H2,(H2,48,63)(H2,49,64)(H2,50,65)(H,54,67)(H,55,69)(H,56,72)(H,57,66)(H,58,70)(H,59,68)(H,60,71)(H4,51,52,53)/t27-,28-,29-,30-,31-,32-,33-,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku University and Department of Pharmaceutical Sciences
Curated by ChEMBL
| Assay Description
Displacement of [3H]OT from recombinant human OTR expressed in HEK293 cell membranes measured after 1 hr |
J Med Chem 62: 3297-3310 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01691
BindingDB Entry DOI: 10.7270/Q2GF0XXQ |
More data for this
Ligand-Target Pair | |
Oxytocin receptor
(Homo sapiens (Human)) | BDBM50523554
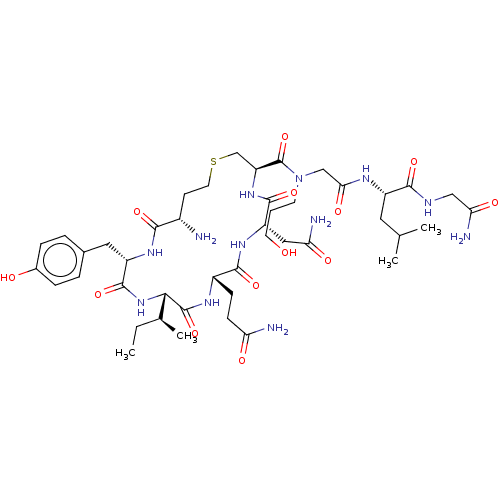
(CHEMBL4467042)Show SMILES CC[C@H](C)[C@@H]1NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)[C@@H](N)CCSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC1=O)C(=O)N(CCCO)CC(=O)N[C@@H](CC(C)C)C(=O)NCC(N)=O |r| Show InChI InChI=1S/C44H70N12O13S/c1-5-24(4)37-43(68)51-28(11-12-33(46)59)40(65)53-31(19-34(47)60)41(66)54-32(22-70-16-13-27(45)38(63)52-30(42(67)55-37)18-25-7-9-26(58)10-8-25)44(69)56(14-6-15-57)21-36(62)50-29(17-23(2)3)39(64)49-20-35(48)61/h7-10,23-24,27-32,37,57-58H,5-6,11-22,45H2,1-4H3,(H2,46,59)(H2,47,60)(H2,48,61)(H,49,64)(H,50,62)(H,51,68)(H,52,63)(H,53,65)(H,54,66)(H,55,67)/t24-,27-,28-,29-,30-,31-,32-,37-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Tohoku University and Department of Pharmaceutical Sciences
Curated by ChEMBL
| Assay Description
Displacement of [3H]OT from recombinant human OTR expressed in HEK293 cell membranes measured after 1 hr |
J Med Chem 62: 3297-3310 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01691
BindingDB Entry DOI: 10.7270/Q2GF0XXQ |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50168287
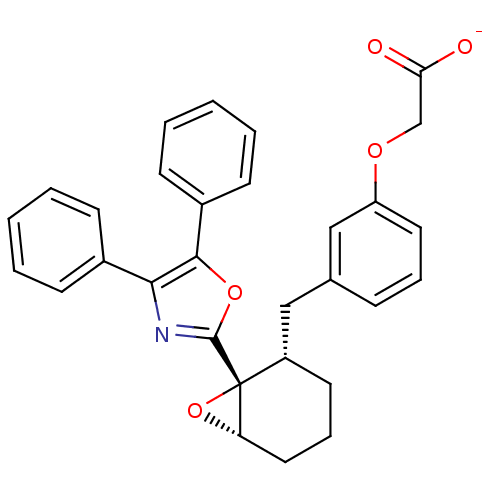
(CHEMBL363800 | Sodium; {3-[(1R,2S,6S)-1-(4,5-diphe...)Show SMILES [O-]C(=O)COc1cccc(C[C@@H]2CCC[C@@H]3O[C@]23c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 Show InChI InChI=1S/C30H27NO5/c32-26(33)19-34-24-15-7-9-20(18-24)17-23-14-8-16-25-30(23,36-25)29-31-27(21-10-3-1-4-11-21)28(35-29)22-12-5-2-6-13-22/h1-7,9-13,15,18,23,25H,8,14,16-17,19H2,(H,32,33)/p-1/t23-,25-,30+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-Iloprost binding to human Prostanoid IP receptor |
Bioorg Med Chem Lett 15: 3284-7 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.076
BindingDB Entry DOI: 10.7270/Q2C53KCV |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM23954

(5-[(2E,3aS,4R,5R,6aS)-5-hydroxy-4-[(1E,3S)-3-hydro...)Show SMILES [H][C@]12C[C@@H](O)[C@H](\C=C\[C@@H](O)C(C)CC#CC)[C@@]1([H])C\C(C2)=C\CCCC(O)=O Show InChI InChI=1S/C22H32O4/c1-3-4-7-15(2)20(23)11-10-18-19-13-16(8-5-6-9-22(25)26)12-17(19)14-21(18)24/h8,10-11,15,17-21,23-24H,5-7,9,12-14H2,1-2H3,(H,25,26)/b11-10+,16-8+/t15?,17-,18+,19-,20+,21+/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Ability to inhibit binding of [3H]iloprost to cloned human prostaglandin I2 receptor |
Bioorg Med Chem Lett 15: 3279-83 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.042
BindingDB Entry DOI: 10.7270/Q2J103X8 |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50167887

((R)-2-(6-((diphenylcarbamoyloxy)methyl)-6-hydroxy-...)Show SMILES OC(=O)COc1cccc2C[C@@](O)(COC(=O)N(c3ccccc3)c3ccccc3)CCc12 Show InChI InChI=1S/C26H25NO6/c28-24(29)17-32-23-13-7-8-19-16-26(31,15-14-22(19)23)18-33-25(30)27(20-9-3-1-4-10-20)21-11-5-2-6-12-21/h1-13,31H,14-18H2,(H,28,29)/t26-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]iloprost from human Prostanoid IP receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50167890
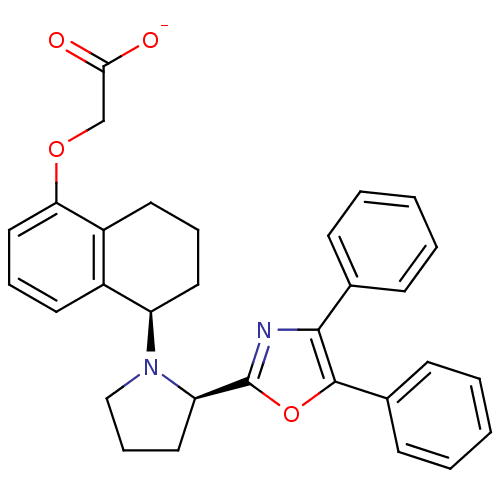
(CHEMBL190497 | FR-193262 | Sodium; {(R)-5-[(R)-2-(...)Show SMILES [O-]C(=O)COc1cccc2[C@@H](CCCc12)N1CCC[C@@H]1c1nc(c(o1)-c1ccccc1)-c1ccccc1 Show InChI InChI=1S/C31H30N2O4/c34-28(35)20-36-27-18-8-14-23-24(27)15-7-16-25(23)33-19-9-17-26(33)31-32-29(21-10-3-1-4-11-21)30(37-31)22-12-5-2-6-13-22/h1-6,8,10-14,18,25-26H,7,9,15-17,19-20H2,(H,34,35)/p-1/t25-,26-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]iloprost from human Prostanoid IP receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50167890

(CHEMBL190497 | FR-193262 | Sodium; {(R)-5-[(R)-2-(...)Show SMILES [O-]C(=O)COc1cccc2[C@@H](CCCc12)N1CCC[C@@H]1c1nc(c(o1)-c1ccccc1)-c1ccccc1 Show InChI InChI=1S/C31H30N2O4/c34-28(35)20-36-27-18-8-14-23-24(27)15-7-16-25(23)33-19-9-17-26(33)31-32-29(21-10-3-1-4-11-21)30(37-31)22-12-5-2-6-13-22/h1-6,8,10-14,18,25-26H,7,9,15-17,19-20H2,(H,34,35)/p-1/t25-,26-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Ability to inhibit binding of [3H]iloprost to cloned human prostaglandin I2 receptor |
Bioorg Med Chem Lett 15: 3279-83 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.042
BindingDB Entry DOI: 10.7270/Q2J103X8 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240130
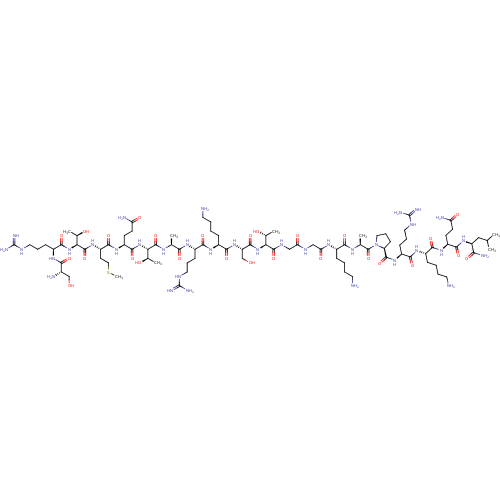
(CHEMBL4060961)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](N)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C90H165N35O27S/c1-44(2)39-60(70(97)135)120-78(143)57(26-28-63(95)131)116-74(139)52(20-10-13-32-92)114-76(141)55(23-16-35-105-89(100)101)118-83(148)62-25-18-37-125(62)87(152)46(4)110-73(138)51(19-9-12-31-91)111-66(134)41-107-65(133)40-108-84(149)67(47(5)128)122-82(147)61(43-127)121-77(142)53(21-11-14-33-93)115-75(140)54(22-15-34-104-88(98)99)112-71(136)45(3)109-85(150)68(48(6)129)123-81(146)58(27-29-64(96)132)117-79(144)59(30-38-153-8)119-86(151)69(49(7)130)124-80(145)56(24-17-36-106-90(102)103)113-72(137)50(94)42-126/h44-62,67-69,126-130H,9-43,91-94H2,1-8H3,(H2,95,131)(H2,96,132)(H2,97,135)(H,107,133)(H,108,149)(H,109,150)(H,110,138)(H,111,134)(H,112,136)(H,113,137)(H,114,141)(H,115,140)(H,116,139)(H,117,144)(H,118,148)(H,119,151)(H,120,143)(H,121,142)(H,122,147)(H,123,146)(H,124,145)(H4,98,99,104)(H4,100,101,105)(H4,102,103,106)/t45-,46-,47+,48+,49+,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,67-,68-,69-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240124
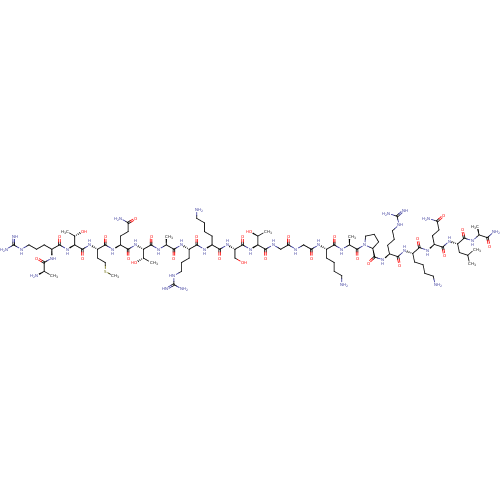
(CHEMBL4090728)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](C)N)[C@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(N)=O |r| Show InChI InChI=1S/C93H170N36O27S/c1-45(2)41-62(84(150)112-47(4)72(100)138)124-80(146)59(28-30-65(98)134)120-76(142)54(22-12-15-34-95)118-78(144)57(25-18-37-108-92(103)104)122-86(152)64-27-20-39-129(64)90(156)49(6)114-75(141)53(21-11-14-33-94)115-68(137)43-110-67(136)42-111-87(153)69(50(7)131)126-85(151)63(44-130)125-79(145)55(23-13-16-35-96)119-77(143)56(24-17-36-107-91(101)102)117-74(140)48(5)113-88(154)70(51(8)132)127-83(149)60(29-31-66(99)135)121-81(147)61(32-40-157-10)123-89(155)71(52(9)133)128-82(148)58(116-73(139)46(3)97)26-19-38-109-93(105)106/h45-64,69-71,130-133H,11-44,94-97H2,1-10H3,(H2,98,134)(H2,99,135)(H2,100,138)(H,110,136)(H,111,153)(H,112,150)(H,113,154)(H,114,141)(H,115,137)(H,116,139)(H,117,140)(H,118,144)(H,119,143)(H,120,142)(H,121,147)(H,122,152)(H,123,155)(H,124,146)(H,125,145)(H,126,151)(H,127,149)(H,128,148)(H4,101,102,107)(H4,103,104,108)(H4,105,106,109)/t46-,47+,48+,49+,50+,51+,52+,53+,54+,55+,56+,57+,58+,59+,60+,61+,62+,63+,64+,69+,70+,71+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 assessed as reduction in H2O2 production using pLys4Met H3 peptide as substrate by peroxidase coupled UV-visible spectrophot... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50168291
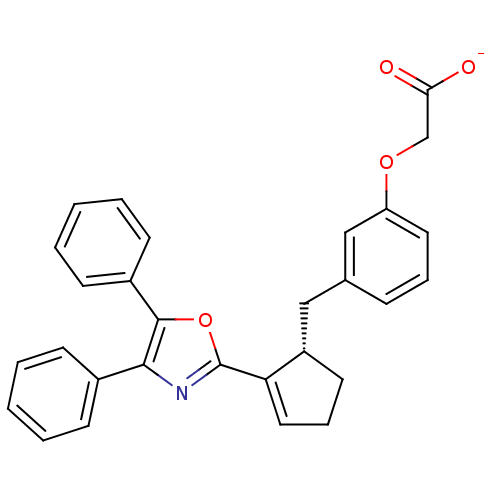
(CHEMBL363350 | Sodium; {3-[(S)-2-(4,5-diphenyl-oxa...)Show SMILES [O-]C(=O)COc1cccc(C[C@@H]2CCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:14| Show InChI InChI=1S/C29H25NO4/c31-26(32)19-33-24-15-7-9-20(18-24)17-23-14-8-16-25(23)29-30-27(21-10-3-1-4-11-21)28(34-29)22-12-5-2-6-13-22/h1-7,9-13,15-16,18,23H,8,14,17,19H2,(H,31,32)/p-1/t23-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 41 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-Iloprost binding to human Prostanoid IP receptor |
Bioorg Med Chem Lett 15: 3284-7 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.076
BindingDB Entry DOI: 10.7270/Q2C53KCV |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50370452
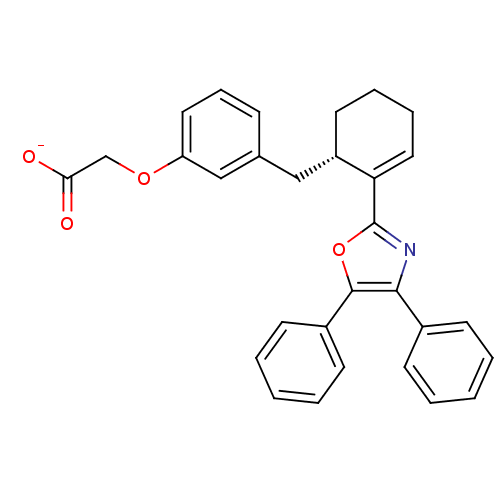
(CHEMBL132589 | FR-181157)Show SMILES [O-]C(=O)COc1cccc(C[C@@H]2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1/t24-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 54 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Ability to inhibit binding of [3H]iloprost to cloned human prostaglandin I2 receptor |
Bioorg Med Chem Lett 15: 3279-83 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.042
BindingDB Entry DOI: 10.7270/Q2J103X8 |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50136234
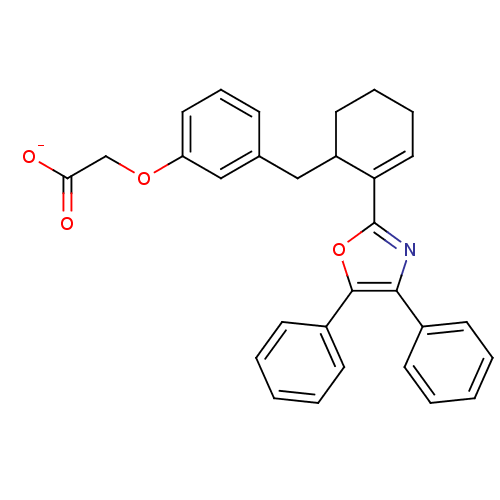
(CHEMBL132589 | FR-181157 | Sodium; {3-[(R)-2-(4,5-...)Show SMILES [O-]C(=O)COc1cccc(CC2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 54 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-Iloprost binding to human Prostanoid IP receptor |
Bioorg Med Chem Lett 15: 3284-7 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.076
BindingDB Entry DOI: 10.7270/Q2C53KCV |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50370452

(CHEMBL132589 | FR-181157)Show SMILES [O-]C(=O)COc1cccc(C[C@@H]2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1/t24-/m0/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 54 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]iloprost from human Prostanoid IP receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240122

(CHEMBL4103690)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](N)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCNC(=O)[C@@H](N)CO)C(N)=O |r| Show InChI InChI=1S/C99H182N38O30S/c1-49(2)43-67(90(161)123-57(77(107)148)21-12-16-37-114-79(150)55(103)46-138)132-86(157)64(29-31-70(105)144)128-82(153)59(23-10-14-35-101)126-84(155)62(26-18-39-116-98(110)111)130-92(163)69-28-20-41-137(69)96(167)51(4)121-81(152)58(22-9-13-34-100)122-73(147)45-118-72(146)44-119-93(164)74(52(5)141)134-91(162)68(48-140)133-85(156)60(24-11-15-36-102)127-83(154)61(25-17-38-115-97(108)109)124-78(149)50(3)120-94(165)75(53(6)142)135-89(160)65(30-32-71(106)145)129-87(158)66(33-42-168-8)131-95(166)76(54(7)143)136-88(159)63(27-19-40-117-99(112)113)125-80(151)56(104)47-139/h49-69,74-76,138-143H,9-48,100-104H2,1-8H3,(H2,105,144)(H2,106,145)(H2,107,148)(H,114,150)(H,118,146)(H,119,164)(H,120,165)(H,121,152)(H,122,147)(H,123,161)(H,124,149)(H,125,151)(H,126,155)(H,127,154)(H,128,153)(H,129,158)(H,130,163)(H,131,166)(H,132,157)(H,133,156)(H,134,162)(H,135,160)(H,136,159)(H4,108,109,115)(H4,110,111,116)(H4,112,113,117)/t50-,51-,52+,53+,54+,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,69-,74-,75-,76-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240126

(CHEMBL4105288)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)N)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C90H165N35O26S/c1-44(2)40-60(70(97)134)120-78(142)57(27-29-63(95)130)116-74(138)52(21-11-14-33-92)114-76(140)55(24-17-36-105-89(100)101)118-83(147)62-26-19-38-125(62)87(151)47(5)110-73(137)51(20-10-13-32-91)111-66(133)42-107-65(132)41-108-84(148)67(48(6)127)122-82(146)61(43-126)121-77(141)53(22-12-15-34-93)115-75(139)54(23-16-35-104-88(98)99)113-72(136)46(4)109-85(149)68(49(7)128)123-81(145)58(28-30-64(96)131)117-79(143)59(31-39-152-9)119-86(150)69(50(8)129)124-80(144)56(112-71(135)45(3)94)25-18-37-106-90(102)103/h44-62,67-69,126-129H,10-43,91-94H2,1-9H3,(H2,95,130)(H2,96,131)(H2,97,134)(H,107,132)(H,108,148)(H,109,149)(H,110,137)(H,111,133)(H,112,135)(H,113,136)(H,114,140)(H,115,139)(H,116,138)(H,117,143)(H,118,147)(H,119,150)(H,120,142)(H,121,141)(H,122,146)(H,123,145)(H,124,144)(H4,98,99,104)(H4,100,101,105)(H4,102,103,106)/t45-,46-,47-,48+,49+,50+,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,67-,68-,69-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 98 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50167892

(CHEMBL195456 | {(S)-6-[(Diphenylcarbamoyloxy)-meth...)Show SMILES O[C@@H]1c2cccc(OCC(O)=O)c2CC[C@]1(O)COC(=O)N(c1ccccc1)c1ccccc1 Show InChI InChI=1S/C26H25NO7/c28-23(29)16-33-22-13-7-12-21-20(22)14-15-26(32,24(21)30)17-34-25(31)27(18-8-3-1-4-9-18)19-10-5-2-6-11-19/h1-13,24,30,32H,14-17H2,(H,28,29)/t24-,26+/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]iloprost from human Prostanoid IP receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostacyclin receptor
(Homo sapiens (Human)) | BDBM50167891
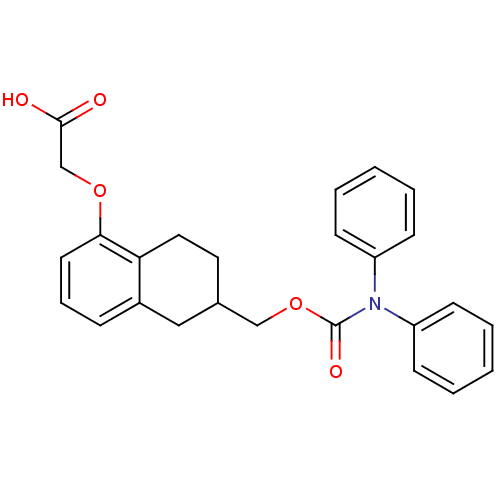
(CHEMBL197319 | {6-[(Diphenylcarbamoyloxy)-methyl]-...)Show SMILES OC(=O)COc1cccc2CC(COC(=O)N(c3ccccc3)c3ccccc3)CCc12 Show InChI InChI=1S/C26H25NO5/c28-25(29)18-31-24-13-7-8-20-16-19(14-15-23(20)24)17-32-26(30)27(21-9-3-1-4-10-21)22-11-5-2-6-12-22/h1-13,19H,14-18H2,(H,28,29) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]iloprost from human Prostanoid IP receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240123
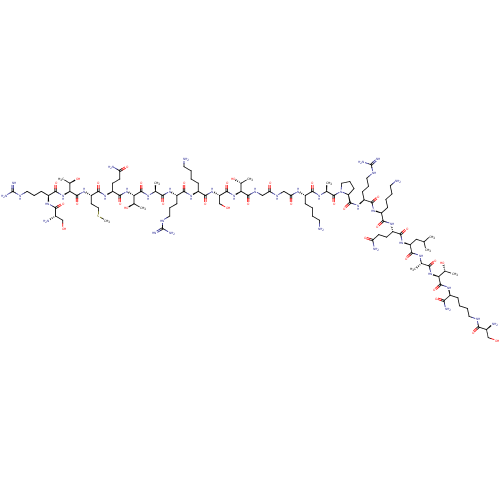
(CHEMBL4085763)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](N)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCCNC(=O)[C@@H](N)CO)C(N)=O |r| Show InChI InChI=1S/C106H194N40O33S/c1-51(2)45-71(96(172)127-53(4)84(160)142-80(57(8)152)101(177)131-61(82(114)158)23-14-18-39-121-85(161)59(110)48-147)140-92(168)68(31-33-74(112)154)136-88(164)63(25-12-16-37-108)134-90(166)66(28-20-41-123-105(117)118)138-98(174)73-30-22-43-146(73)103(179)54(5)129-87(163)62(24-11-15-36-107)130-77(157)47-125-76(156)46-126-99(175)78(55(6)150)143-97(173)72(50-149)141-91(167)64(26-13-17-38-109)135-89(165)65(27-19-40-122-104(115)116)132-83(159)52(3)128-100(176)79(56(7)151)144-95(171)69(32-34-75(113)155)137-93(169)70(35-44-180-10)139-102(178)81(58(9)153)145-94(170)67(29-21-42-124-106(119)120)133-86(162)60(111)49-148/h51-73,78-81,147-153H,11-50,107-111H2,1-10H3,(H2,112,154)(H2,113,155)(H2,114,158)(H,121,161)(H,125,156)(H,126,175)(H,127,172)(H,128,176)(H,129,163)(H,130,157)(H,131,177)(H,132,159)(H,133,162)(H,134,166)(H,135,165)(H,136,164)(H,137,169)(H,138,174)(H,139,178)(H,140,168)(H,141,167)(H,142,160)(H,143,173)(H,144,171)(H,145,170)(H4,115,116,122)(H4,117,118,123)(H4,119,120,124)/t52-,53-,54-,55+,56+,57+,58+,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,69-,70-,71-,72-,73-,78-,79-,80-,81-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240121
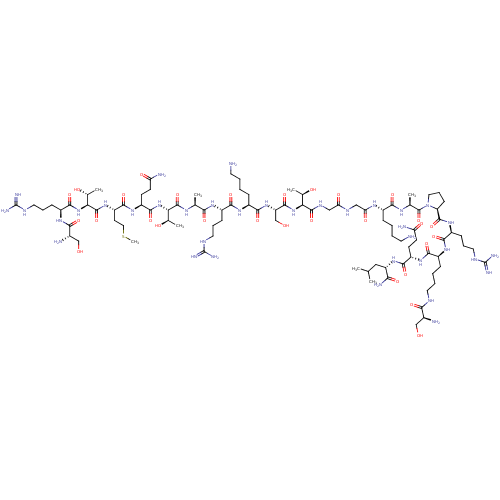
(CHEMBL4089148)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](N)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCNC(=O)[C@@H](N)CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C93H170N36O29S/c1-45(2)39-62(72(100)140)124-81(149)59(26-28-65(98)136)120-77(145)55(21-11-14-33-107-74(142)51(96)42-130)119-79(147)57(23-16-35-109-92(103)104)122-86(154)64-25-18-37-129(64)90(158)47(4)114-76(144)53(19-9-12-31-94)115-68(139)41-111-67(138)40-112-87(155)69(48(5)133)126-85(153)63(44-132)125-80(148)54(20-10-13-32-95)118-78(146)56(22-15-34-108-91(101)102)116-73(141)46(3)113-88(156)70(49(6)134)127-84(152)60(27-29-66(99)137)121-82(150)61(30-38-159-8)123-89(157)71(50(7)135)128-83(151)58(24-17-36-110-93(105)106)117-75(143)52(97)43-131/h45-64,69-71,130-135H,9-44,94-97H2,1-8H3,(H2,98,136)(H2,99,137)(H2,100,140)(H,107,142)(H,111,138)(H,112,155)(H,113,156)(H,114,144)(H,115,139)(H,116,141)(H,117,143)(H,118,146)(H,119,147)(H,120,145)(H,121,150)(H,122,154)(H,123,157)(H,124,149)(H,125,148)(H,126,153)(H,127,152)(H,128,151)(H4,101,102,108)(H4,103,104,109)(H4,105,106,110)/t46-,47-,48+,49+,50+,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,69-,70-,71-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM50168287

(CHEMBL363800 | Sodium; {3-[(1R,2S,6S)-1-(4,5-diphe...)Show SMILES [O-]C(=O)COc1cccc(C[C@@H]2CCC[C@@H]3O[C@]23c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 Show InChI InChI=1S/C30H27NO5/c32-26(33)19-34-24-15-7-9-20(18-24)17-23-14-8-16-25-30(23,36-25)29-31-27(21-10-3-1-4-11-21)28(35-29)22-12-5-2-6-13-22/h1-7,9-13,15,18,23,25H,8,14,16-17,19H2,(H,32,33)/p-1/t23-,25-,30+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 930 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-SQ-29,548 binding to human Prostanoid TP receptor |
Bioorg Med Chem Lett 15: 3284-7 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.076
BindingDB Entry DOI: 10.7270/Q2C53KCV |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM50136234

(CHEMBL132589 | FR-181157 | Sodium; {3-[(R)-2-(4,5-...)Show SMILES [O-]C(=O)COc1cccc(CC2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-SQ-29,548 binding to human Prostanoid TP receptor |
Bioorg Med Chem Lett 15: 3284-7 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.076
BindingDB Entry DOI: 10.7270/Q2C53KCV |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50136234

(CHEMBL132589 | FR-181157 | Sodium; {3-[(R)-2-(4,5-...)Show SMILES [O-]C(=O)COc1cccc(CC2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-PGE-2 binding to Prostanoid EP2 receptor |
Bioorg Med Chem Lett 15: 3284-7 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.076
BindingDB Entry DOI: 10.7270/Q2C53KCV |
More data for this
Ligand-Target Pair | |
Prostaglandin D2 receptor
(Homo sapiens (Human)) | BDBM50136234

(CHEMBL132589 | FR-181157 | Sodium; {3-[(R)-2-(4,5-...)Show SMILES [O-]C(=O)COc1cccc(CC2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-PGD-2 binding to human Prostanoid DP receptor |
Bioorg Med Chem Lett 15: 3284-7 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.076
BindingDB Entry DOI: 10.7270/Q2C53KCV |
More data for this
Ligand-Target Pair | |
Prostaglandin F2-alpha receptor
(Homo sapiens (Human)) | BDBM50136234

(CHEMBL132589 | FR-181157 | Sodium; {3-[(R)-2-(4,5-...)Show SMILES [O-]C(=O)COc1cccc(CC2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-PGF-2 binding to human Prostanoid FP receptor |
Bioorg Med Chem Lett 15: 3284-7 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.076
BindingDB Entry DOI: 10.7270/Q2C53KCV |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM50136234

(CHEMBL132589 | FR-181157 | Sodium; {3-[(R)-2-(4,5-...)Show SMILES [O-]C(=O)COc1cccc(CC2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-PGE-2 binding to Prostanoid EP1 receptor |
Bioorg Med Chem Lett 15: 3284-7 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.076
BindingDB Entry DOI: 10.7270/Q2C53KCV |
More data for this
Ligand-Target Pair | |
Prostaglandin D2 receptor
(Homo sapiens (Human)) | BDBM50167890

(CHEMBL190497 | FR-193262 | Sodium; {(R)-5-[(R)-2-(...)Show SMILES [O-]C(=O)COc1cccc2[C@@H](CCCc12)N1CCC[C@@H]1c1nc(c(o1)-c1ccccc1)-c1ccccc1 Show InChI InChI=1S/C31H30N2O4/c34-28(35)20-36-27-18-8-14-23-24(27)15-7-16-25(23)33-19-9-17-26(33)31-32-29(21-10-3-1-4-11-21)30(37-31)22-12-5-2-6-13-22/h1-6,8,10-14,18,25-26H,7,9,15-17,19-20H2,(H,34,35)/p-1/t25-,26-/m1/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]PGD-2 from human Prostanoid DP receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM50370452

(CHEMBL132589 | FR-181157)Show SMILES [O-]C(=O)COc1cccc(C[C@@H]2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1/t24-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]SQ-29,548 from human Prostanoid TP receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM50167891

(CHEMBL197319 | {6-[(Diphenylcarbamoyloxy)-methyl]-...)Show SMILES OC(=O)COc1cccc2CC(COC(=O)N(c3ccccc3)c3ccccc3)CCc12 Show InChI InChI=1S/C26H25NO5/c28-25(29)18-31-24-13-7-8-20-16-19(14-15-23(20)24)17-32-26(30)27(21-9-3-1-4-10-21)22-11-5-2-6-12-22/h1-13,19H,14-18H2,(H,28,29) | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]PGE-2 from human Prostanoid EP1 receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostaglandin D2 receptor
(Homo sapiens (Human)) | BDBM50370452

(CHEMBL132589 | FR-181157)Show SMILES [O-]C(=O)COc1cccc(C[C@@H]2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1/t24-/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]PGD-2 from human Prostanoid DP receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50167891

(CHEMBL197319 | {6-[(Diphenylcarbamoyloxy)-methyl]-...)Show SMILES OC(=O)COc1cccc2CC(COC(=O)N(c3ccccc3)c3ccccc3)CCc12 Show InChI InChI=1S/C26H25NO5/c28-25(29)18-31-24-13-7-8-20-16-19(14-15-23(20)24)17-32-26(30)27(21-9-3-1-4-10-21)22-11-5-2-6-12-22/h1-13,19H,14-18H2,(H,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-PGE-2 from human Prostanoid EP2 receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostaglandin F2-alpha receptor
(Homo sapiens (Human)) | BDBM50167890

(CHEMBL190497 | FR-193262 | Sodium; {(R)-5-[(R)-2-(...)Show SMILES [O-]C(=O)COc1cccc2[C@@H](CCCc12)N1CCC[C@@H]1c1nc(c(o1)-c1ccccc1)-c1ccccc1 Show InChI InChI=1S/C31H30N2O4/c34-28(35)20-36-27-18-8-14-23-24(27)15-7-16-25(23)33-19-9-17-26(33)31-32-29(21-10-3-1-4-11-21)30(37-31)22-12-5-2-6-13-22/h1-6,8,10-14,18,25-26H,7,9,15-17,19-20H2,(H,34,35)/p-1/t25-,26-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]PGF-2 from human Prostanoid FP receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM50167890

(CHEMBL190497 | FR-193262 | Sodium; {(R)-5-[(R)-2-(...)Show SMILES [O-]C(=O)COc1cccc2[C@@H](CCCc12)N1CCC[C@@H]1c1nc(c(o1)-c1ccccc1)-c1ccccc1 Show InChI InChI=1S/C31H30N2O4/c34-28(35)20-36-27-18-8-14-23-24(27)15-7-16-25(23)33-19-9-17-26(33)31-32-29(21-10-3-1-4-11-21)30(37-31)22-12-5-2-6-13-22/h1-6,8,10-14,18,25-26H,7,9,15-17,19-20H2,(H,34,35)/p-1/t25-,26-/m1/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]PGE-2 from human Prostanoid EP1 receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM50167892

(CHEMBL195456 | {(S)-6-[(Diphenylcarbamoyloxy)-meth...)Show SMILES O[C@@H]1c2cccc(OCC(O)=O)c2CC[C@]1(O)COC(=O)N(c1ccccc1)c1ccccc1 Show InChI InChI=1S/C26H25NO7/c28-23(29)16-33-22-13-7-12-21-20(22)14-15-26(32,24(21)30)17-34-25(31)27(18-8-3-1-4-9-18)19-10-5-2-6-11-19/h1-13,24,30,32H,14-17H2,(H,28,29)/t24-,26+/m1/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]PGE-2 from human Prostanoid EP1 receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50167892

(CHEMBL195456 | {(S)-6-[(Diphenylcarbamoyloxy)-meth...)Show SMILES O[C@@H]1c2cccc(OCC(O)=O)c2CC[C@]1(O)COC(=O)N(c1ccccc1)c1ccccc1 Show InChI InChI=1S/C26H25NO7/c28-23(29)16-33-22-13-7-12-21-20(22)14-15-26(32,24(21)30)17-34-25(31)27(18-8-3-1-4-9-18)19-10-5-2-6-11-19/h1-13,24,30,32H,14-17H2,(H,28,29)/t24-,26+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-PGE-2 from human Prostanoid EP2 receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP1 subtype
(Homo sapiens (Human)) | BDBM50370452

(CHEMBL132589 | FR-181157)Show SMILES [O-]C(=O)COc1cccc(C[C@@H]2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1/t24-/m0/s1 | KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]PGE-2 from human Prostanoid EP1 receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50167890

(CHEMBL190497 | FR-193262 | Sodium; {(R)-5-[(R)-2-(...)Show SMILES [O-]C(=O)COc1cccc2[C@@H](CCCc12)N1CCC[C@@H]1c1nc(c(o1)-c1ccccc1)-c1ccccc1 Show InChI InChI=1S/C31H30N2O4/c34-28(35)20-36-27-18-8-14-23-24(27)15-7-16-25(23)33-19-9-17-26(33)31-32-29(21-10-3-1-4-11-21)30(37-31)22-12-5-2-6-13-22/h1-6,8,10-14,18,25-26H,7,9,15-17,19-20H2,(H,34,35)/p-1/t25-,26-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-PGE-2 from human Prostanoid EP2 receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50370452

(CHEMBL132589 | FR-181157)Show SMILES [O-]C(=O)COc1cccc(C[C@@H]2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1/t24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]-PGE-2 from human Prostanoid EP2 receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostaglandin F2-alpha receptor
(Homo sapiens (Human)) | BDBM50370452

(CHEMBL132589 | FR-181157)Show SMILES [O-]C(=O)COc1cccc(C[C@@H]2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1/t24-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]PGF-2 from human Prostanoid FP receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM50167890

(CHEMBL190497 | FR-193262 | Sodium; {(R)-5-[(R)-2-(...)Show SMILES [O-]C(=O)COc1cccc2[C@@H](CCCc12)N1CCC[C@@H]1c1nc(c(o1)-c1ccccc1)-c1ccccc1 Show InChI InChI=1S/C31H30N2O4/c34-28(35)20-36-27-18-8-14-23-24(27)15-7-16-25(23)33-19-9-17-26(33)31-32-29(21-10-3-1-4-11-21)30(37-31)22-12-5-2-6-13-22/h1-6,8,10-14,18,25-26H,7,9,15-17,19-20H2,(H,34,35)/p-1/t25-,26-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]SQ-29,548 from human Prostanoid TP receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP4 subtype
(Homo sapiens (Human)) | BDBM50136234

(CHEMBL132589 | FR-181157 | Sodium; {3-[(R)-2-(4,5-...)Show SMILES [O-]C(=O)COc1cccc(CC2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.02E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-PGE-2 binding to Prostanoid EP4 receptor |
Bioorg Med Chem Lett 15: 3284-7 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.076
BindingDB Entry DOI: 10.7270/Q2C53KCV |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP4 subtype
(Homo sapiens (Human)) | BDBM50370452

(CHEMBL132589 | FR-181157)Show SMILES [O-]C(=O)COc1cccc(C[C@@H]2CCCC=C2c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 |c:15| Show InChI InChI=1S/C30H27NO4/c32-27(33)20-34-25-16-9-10-21(19-25)18-24-15-7-8-17-26(24)30-31-28(22-11-3-1-4-12-22)29(35-30)23-13-5-2-6-14-23/h1-6,9-14,16-17,19,24H,7-8,15,18,20H2,(H,32,33)/p-1/t24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.02E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]PGE-2 from human Prostanoid EP4 receptor |
Bioorg Med Chem Lett 15: 3091-5 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.047
BindingDB Entry DOI: 10.7270/Q2SF2WXV |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240129
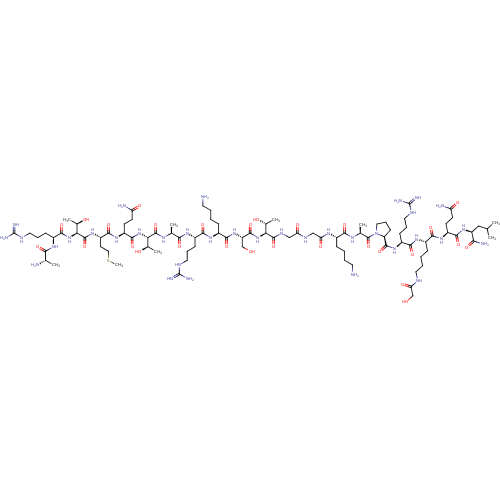
(CHEMBL4080345)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)N)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCNC(=O)CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C92H167N35O28S/c1-45(2)40-61(72(98)138)122-80(146)58(27-29-64(96)133)118-76(142)54(22-12-15-34-105-68(137)44-129)117-78(144)56(24-17-36-107-91(101)102)120-85(151)63-26-19-38-127(63)89(155)48(5)112-75(141)52(20-10-13-32-93)113-67(136)42-109-66(135)41-110-86(152)69(49(6)130)124-84(150)62(43-128)123-79(145)53(21-11-14-33-94)116-77(143)55(23-16-35-106-90(99)100)115-74(140)47(4)111-87(153)70(50(7)131)125-83(149)59(28-30-65(97)134)119-81(147)60(31-39-156-9)121-88(154)71(51(8)132)126-82(148)57(114-73(139)46(3)95)25-18-37-108-92(103)104/h45-63,69-71,128-132H,10-44,93-95H2,1-9H3,(H2,96,133)(H2,97,134)(H2,98,138)(H,105,137)(H,109,135)(H,110,152)(H,111,153)(H,112,141)(H,113,136)(H,114,139)(H,115,140)(H,116,143)(H,117,144)(H,118,142)(H,119,147)(H,120,151)(H,121,154)(H,122,146)(H,123,145)(H,124,150)(H,125,149)(H,126,148)(H4,99,100,106)(H4,101,102,107)(H4,103,104,108)/t46-,47-,48-,49+,50+,51+,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,69-,70-,71-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Prostaglandin E2 receptor EP2 subtype
(Homo sapiens (Human)) | BDBM50168287

(CHEMBL363800 | Sodium; {3-[(1R,2S,6S)-1-(4,5-diphe...)Show SMILES [O-]C(=O)COc1cccc(C[C@@H]2CCC[C@@H]3O[C@]23c2nc(c(o2)-c2ccccc2)-c2ccccc2)c1 Show InChI InChI=1S/C30H27NO5/c32-26(33)19-34-24-15-7-9-20(18-24)17-23-14-8-16-25-30(23,36-25)29-31-27(21-10-3-1-4-11-21)28(35-29)22-12-5-2-6-13-22/h1-7,9-13,15,18,23,25H,8,14,16-17,19H2,(H,32,33)/p-1/t23-,25-,30+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Fujisawa Pharmaceutical Co., Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of [3H]-PGE-2 binding to Prostanoid EP2 receptor |
Bioorg Med Chem Lett 15: 3284-7 (2005)
Article DOI: 10.1016/j.bmcl.2005.04.076
BindingDB Entry DOI: 10.7270/Q2C53KCV |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data