 Found 247 hits with Last Name = 'lumeras' and Initial = 'w'
Found 247 hits with Last Name = 'lumeras' and Initial = 'w' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357914
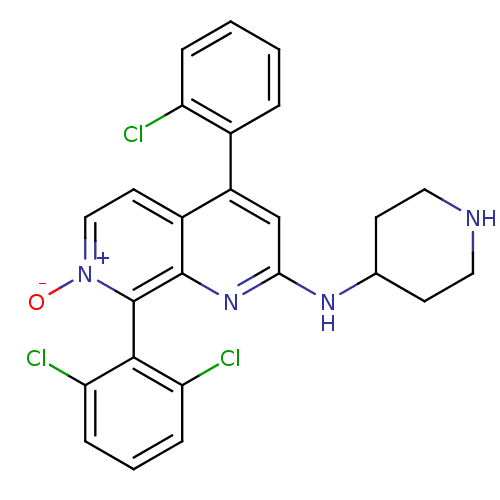
(CHEMBL1916528)Show SMILES [O-][n+]1ccc2c(cc(NC3CCNCC3)nc2c1-c1c(Cl)cccc1Cl)-c1ccccc1Cl |(20.25,-36.83,;21.59,-36.06,;21.59,-34.52,;22.92,-33.75,;24.25,-34.51,;25.59,-33.73,;26.93,-34.51,;26.93,-36.06,;28.26,-36.83,;29.59,-36.07,;30.92,-36.85,;32.25,-36.09,;32.27,-34.55,;30.93,-33.77,;29.59,-34.53,;25.58,-36.83,;24.25,-36.06,;22.92,-36.83,;22.93,-38.37,;21.59,-39.14,;20.26,-38.37,;21.59,-40.68,;22.93,-41.45,;24.27,-40.66,;24.26,-39.13,;25.59,-38.35,;25.59,-32.2,;26.93,-31.43,;26.93,-29.89,;25.59,-29.12,;24.25,-29.9,;24.26,-31.44,;22.93,-32.21,)| Show InChI InChI=1S/C25H21Cl3N4O/c26-19-5-2-1-4-16(19)18-14-22(30-15-8-11-29-12-9-15)31-24-17(18)10-13-32(33)25(24)23-20(27)6-3-7-21(23)28/h1-7,10,13-15,29H,8-9,11-12H2,(H,30,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221898
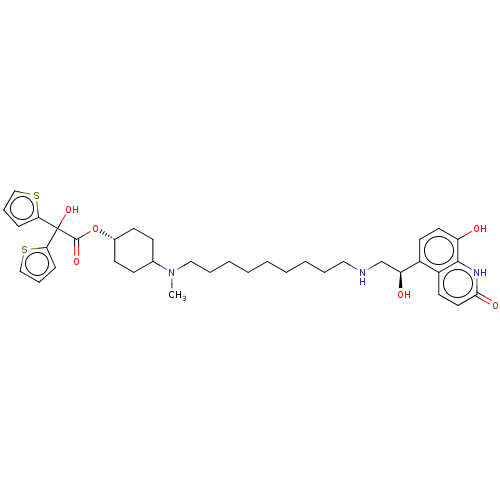
(US9315463, 1)Show SMILES CN(CCCCCCCCCNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12)C1CC[C@@H](CC1)OC(=O)C(O)(c1cccs1)c1cccs1 |r,wU:13.13,30.35,(5.28,4.23,;5.28,2.69,;3.94,1.93,;2.61,2.69,;1.27,1.93,;-.06,2.69,;-1.39,1.93,;-2.73,2.69,;-4.06,1.93,;-5.39,2.69,;-6.73,1.93,;-8.06,2.69,;-9.39,1.93,;-10.73,2.69,;-10.73,4.23,;-12.06,1.93,;-13.4,2.69,;-14.73,1.93,;-14.73,.38,;-16.06,-.38,;-13.4,-.38,;-13.4,-1.93,;-12.06,-2.69,;-12.06,-4.23,;-10.73,-1.93,;-10.73,-.38,;-12.06,.38,;6.61,1.93,;6.61,.38,;7.94,-.38,;9.28,.38,;9.28,1.93,;7.94,2.69,;10.61,-.38,;11.94,.38,;11.94,1.93,;13.28,-.38,;14.61,-1.15,;12.51,-1.72,;10.97,-1.72,;10.49,-3.18,;11.74,-4.09,;12.98,-3.18,;14.05,.95,;15.59,.95,;16.06,2.41,;14.82,3.32,;13.57,2.41,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357913
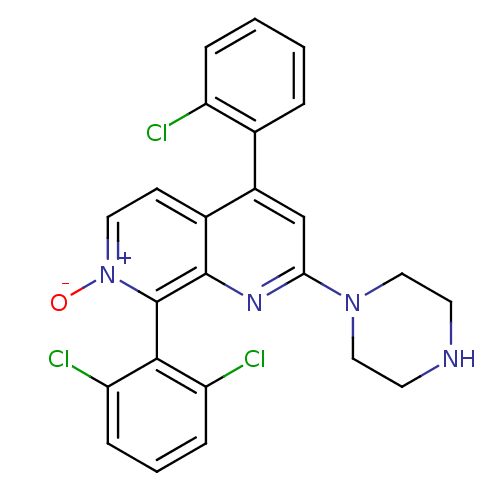
(CHEMBL1916527)Show SMILES [O-][n+]1ccc2c(cc(nc2c1-c1c(Cl)cccc1Cl)N1CCNCC1)-c1ccccc1Cl |(6.31,-36.87,;7.64,-36.1,;7.64,-34.56,;8.97,-33.79,;10.31,-34.55,;11.64,-33.77,;12.98,-34.55,;12.98,-36.1,;11.64,-36.87,;10.3,-36.1,;8.98,-36.87,;8.98,-38.4,;7.64,-39.18,;6.31,-38.41,;7.65,-40.71,;8.98,-41.48,;10.32,-40.7,;10.31,-39.17,;11.64,-38.39,;14.3,-36.87,;14.29,-38.41,;15.62,-39.18,;16.96,-38.42,;16.97,-36.88,;15.63,-36.1,;11.64,-32.24,;12.98,-31.47,;12.98,-29.93,;11.64,-29.16,;10.31,-29.94,;10.31,-31.48,;8.98,-32.25,)| Show InChI InChI=1S/C24H19Cl3N4O/c25-18-5-2-1-4-15(18)17-14-21(30-12-9-28-10-13-30)29-23-16(17)8-11-31(32)24(23)22-19(26)6-3-7-20(22)27/h1-8,11,14,28H,9-10,12-13H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357909
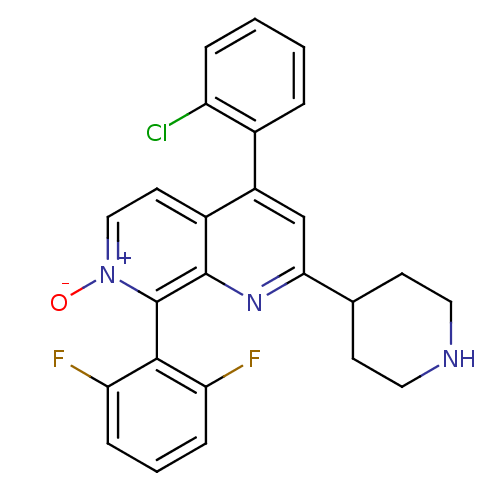
(CHEMBL1916523)Show SMILES [O-][n+]1ccc2c(cc(nc2c1-c1c(F)cccc1F)C1CCNCC1)-c1ccccc1Cl |(-8.74,-20.09,;-7.4,-19.32,;-7.4,-17.78,;-6.07,-17.01,;-4.74,-17.77,;-3.4,-16.99,;-2.06,-17.77,;-2.07,-19.32,;-3.41,-20.09,;-4.74,-19.32,;-6.07,-20.09,;-6.06,-21.63,;-7.4,-22.4,;-8.73,-21.63,;-7.4,-23.94,;-6.06,-24.71,;-4.72,-23.92,;-4.73,-22.39,;-3.4,-21.61,;-.73,-20.09,;-.74,-21.63,;.59,-22.4,;1.93,-21.64,;1.93,-20.1,;.6,-19.32,;-3.4,-15.46,;-2.07,-14.69,;-2.06,-13.15,;-3.4,-12.38,;-4.74,-13.16,;-4.73,-14.7,;-6.06,-15.47,)| Show InChI InChI=1S/C25H20ClF2N3O/c26-19-5-2-1-4-16(19)18-14-22(15-8-11-29-12-9-15)30-24-17(18)10-13-31(32)25(24)23-20(27)6-3-7-21(23)28/h1-7,10,13-15,29H,8-9,11-12H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357911
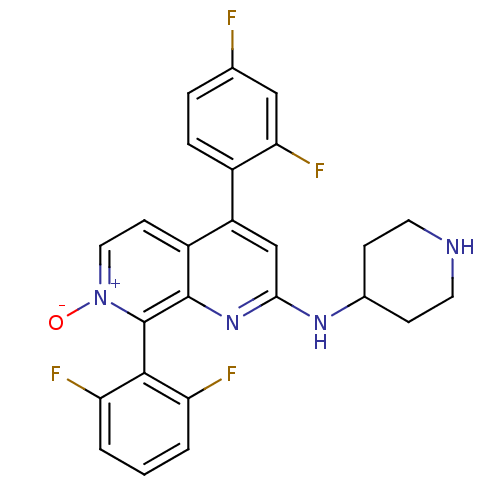
(CHEMBL1916525)Show SMILES [O-][n+]1ccc2c(cc(NC3CCNCC3)nc2c1-c1c(F)cccc1F)-c1ccc(F)cc1F |(23.08,-19.61,;24.41,-18.84,;24.41,-17.3,;25.74,-16.53,;27.08,-17.29,;28.41,-16.51,;29.75,-17.29,;29.75,-18.84,;31.08,-19.62,;32.42,-18.85,;33.75,-19.63,;35.08,-18.87,;35.09,-17.33,;33.76,-16.55,;32.41,-17.31,;28.41,-19.61,;27.08,-18.84,;25.75,-19.61,;25.75,-21.15,;27.08,-21.91,;28.41,-21.13,;27.09,-23.45,;25.76,-24.23,;24.42,-23.46,;24.42,-21.92,;23.08,-21.15,;28.41,-14.98,;29.75,-14.21,;29.75,-12.67,;28.42,-11.9,;28.41,-10.36,;27.08,-12.68,;27.08,-14.22,;25.75,-14.99,)| Show InChI InChI=1S/C25H20F4N4O/c26-14-4-5-16(21(29)12-14)18-13-22(31-15-6-9-30-10-7-15)32-24-17(18)8-11-33(34)25(24)23-19(27)2-1-3-20(23)28/h1-5,8,11-13,15,30H,6-7,9-10H2,(H,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221909
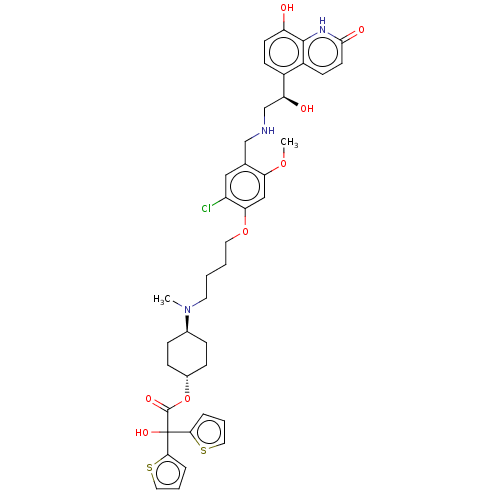
(US9315463, 25)Show SMILES COc1cc(OCCCCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:40.44,15.18,wD:12.11,(-5.39,-3.84,;-4.06,-3.07,;-4.06,-1.53,;-2.73,-.76,;-2.73,.78,;-1.39,1.55,;-.06,.78,;1.27,1.55,;2.61,.78,;3.94,1.55,;5.28,.78,;5.28,-.76,;6.61,1.55,;7.94,.78,;9.28,1.55,;9.28,3.09,;7.94,3.86,;6.61,3.09,;10.61,3.86,;11.94,3.09,;11.94,1.55,;13.28,3.86,;14.61,4.63,;14.05,2.53,;13.57,1.06,;14.82,.16,;16.06,1.06,;15.59,2.53,;12.51,5.2,;13.41,6.44,;12.51,7.69,;11.04,7.21,;11.04,5.67,;-4.06,1.55,;-4.06,3.09,;-5.39,.78,;-5.39,-.76,;-6.73,-1.53,;-8.06,-.76,;-9.39,-1.53,;-10.73,-.76,;-10.73,.78,;-12.06,-1.53,;-13.4,-.76,;-14.73,-1.53,;-14.73,-3.07,;-16.06,-3.84,;-13.4,-3.84,;-13.4,-5.38,;-12.06,-6.15,;-12.06,-7.69,;-10.73,-5.38,;-10.73,-3.84,;-12.06,-3.07,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM221910
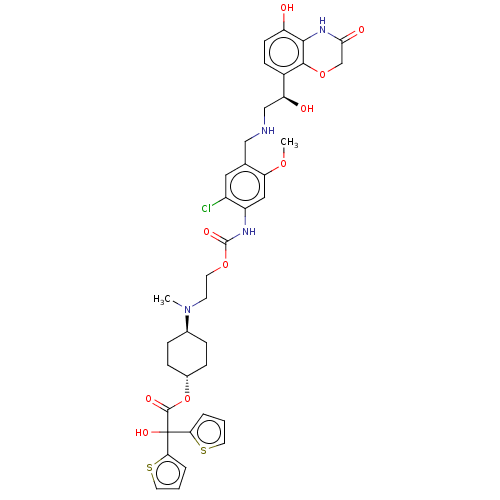
(US9315463, 26)Show SMILES COc1cc(NC(=O)OCCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2NC(=O)COc12 |r,wU:41.45,16.19,wD:13.12,(-2.73,-3.84,;-4.06,-3.07,;-4.06,-1.53,;-2.73,-.76,;-2.73,.78,;-1.39,1.55,;-.06,.78,;-.06,-.76,;1.27,1.55,;2.61,.78,;3.94,1.55,;5.28,.78,;5.28,-.76,;6.61,1.55,;7.94,.78,;9.28,1.55,;9.28,3.09,;7.94,3.86,;6.61,3.09,;10.61,3.86,;11.94,3.09,;11.94,1.55,;13.28,3.86,;14.61,4.63,;14.05,2.53,;13.57,1.06,;14.82,.16,;16.06,1.06,;15.59,2.53,;12.51,5.2,;13.41,6.44,;12.51,7.69,;11.04,7.21,;11.04,5.67,;-4.06,1.55,;-4.06,3.09,;-5.39,.78,;-5.39,-.76,;-6.73,-1.53,;-8.06,-.76,;-9.39,-1.53,;-10.73,-.76,;-10.73,.78,;-12.06,-1.53,;-13.4,-.76,;-14.73,-1.53,;-14.73,-3.07,;-16.06,-3.84,;-13.4,-3.84,;-13.4,-5.38,;-12.06,-6.15,;-12.06,-7.69,;-10.73,-5.38,;-10.73,-3.84,;-12.06,-3.07,)| Show InChI InChI=1S/C38H43ClN4O10S2/c1-43(23-7-9-24(10-8-23)53-36(47)38(49,31-5-3-15-54-31)32-6-4-16-55-32)13-14-51-37(48)41-27-18-30(50-2)22(17-26(27)39)19-40-20-29(45)25-11-12-28(44)34-35(25)52-21-33(46)42-34/h3-6,11-12,15-18,23-24,29,40,44-45,49H,7-10,13-14,19-21H2,1-2H3,(H,41,48)(H,42,46)/t23-,24-,29-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.380 | n/a | n/a | n/a | n/a | n/a | 25 |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human adrenergic beta1 and beta2 receptors was performed using commercial membranes prepared from Sf9 cells where they are ov... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357916
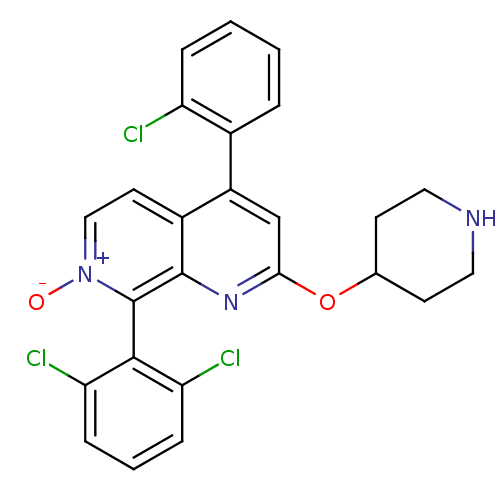
(CHEMBL1916530)Show SMILES [O-][n+]1ccc2c(cc(OC3CCNCC3)nc2c1-c1c(Cl)cccc1Cl)-c1ccccc1Cl |(8.24,-4.99,;9.58,-4.22,;9.58,-2.68,;10.91,-1.9,;12.24,-2.67,;13.58,-1.89,;14.92,-2.67,;14.91,-4.22,;16.25,-4.99,;17.58,-4.23,;18.91,-5.01,;20.25,-4.25,;20.26,-2.71,;18.93,-1.93,;17.58,-2.69,;13.57,-4.99,;12.24,-4.22,;10.91,-4.99,;10.92,-6.52,;9.58,-7.3,;8.25,-6.53,;9.58,-8.83,;10.92,-9.6,;12.26,-8.82,;12.25,-7.29,;13.58,-6.51,;13.58,-.36,;14.91,.41,;14.92,1.95,;13.58,2.72,;12.24,1.94,;12.25,.4,;10.92,-.37,)| Show InChI InChI=1S/C25H20Cl3N3O2/c26-19-5-2-1-4-16(19)18-14-22(33-15-8-11-29-12-9-15)30-24-17(18)10-13-31(32)25(24)23-20(27)6-3-7-21(23)28/h1-7,10,13-15,29H,8-9,11-12H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.460 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357903
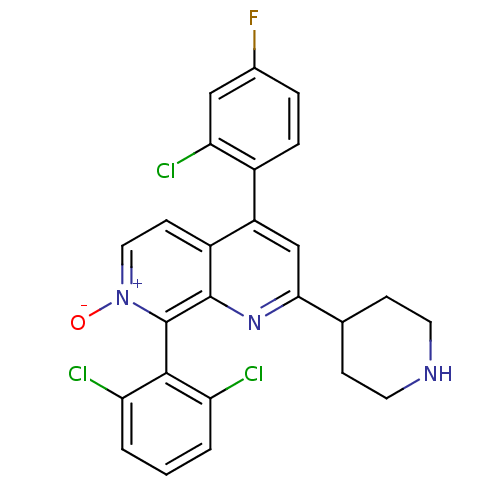
(CHEMBL1916360)Show SMILES [O-][n+]1ccc2c(cc(nc2c1-c1c(Cl)cccc1Cl)C1CCNCC1)-c1ccc(F)cc1Cl |(-9.19,-39.37,;-7.86,-38.61,;-7.86,-37.06,;-6.53,-36.29,;-5.19,-37.05,;-3.86,-36.28,;-2.52,-37.06,;-2.52,-38.6,;-3.87,-39.37,;-5.2,-38.6,;-6.53,-39.38,;-6.52,-40.91,;-7.86,-41.68,;-9.19,-40.91,;-7.85,-43.22,;-6.52,-43.99,;-5.18,-43.21,;-5.19,-41.67,;-3.86,-40.89,;-1.19,-39.37,;-1.2,-40.91,;.13,-41.69,;1.47,-40.93,;1.47,-39.38,;.14,-38.6,;-3.86,-34.75,;-2.52,-33.98,;-2.52,-32.44,;-3.86,-31.67,;-3.86,-30.13,;-5.2,-32.45,;-5.19,-33.98,;-6.52,-34.76,)| Show InChI InChI=1S/C25H19Cl3FN3O/c26-19-2-1-3-20(27)23(19)25-24-17(8-11-32(25)33)18(16-5-4-15(29)12-21(16)28)13-22(31-24)14-6-9-30-10-7-14/h1-5,8,11-14,30H,6-7,9-10H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357915
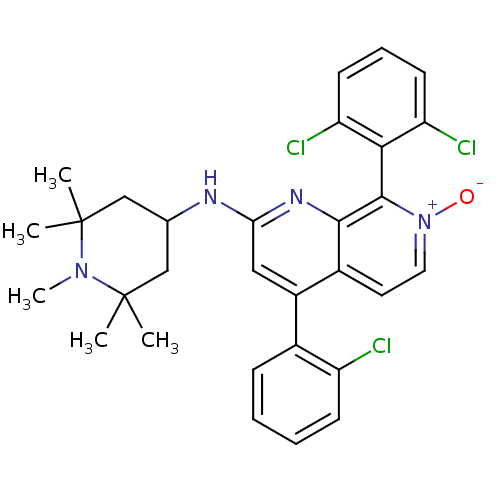
(CHEMBL1916529)Show SMILES CN1C(C)(C)CC(CC1(C)C)Nc1cc(-c2ccccc2Cl)c2cc[n+]([O-])c(-c3c(Cl)cccc3Cl)c2n1 |(4.35,-2.11,;3.01,-2.87,;2.99,-4.41,;4.53,-4.41,;3.76,-5.74,;1.66,-5.17,;.33,-4.39,;.33,-2.86,;1.67,-2.09,;.89,-.75,;2.44,-.75,;-1.01,-5.16,;-2.34,-4.38,;-2.34,-2.84,;-3.67,-2.06,;-3.67,-.52,;-2.34,.24,;-2.34,1.78,;-3.67,2.55,;-5.01,1.78,;-5,.24,;-6.33,-.53,;-5.01,-2.83,;-6.34,-2.07,;-7.67,-2.84,;-7.67,-4.38,;-9.01,-5.15,;-6.34,-5.15,;-6.33,-6.69,;-7.67,-7.46,;-9,-6.69,;-7.67,-9,;-6.33,-9.77,;-4.99,-8.98,;-5,-7.45,;-3.67,-6.67,;-5.01,-4.38,;-3.68,-5.15,)| Show InChI InChI=1S/C30H31Cl3N4O/c1-29(2)16-18(17-30(3,4)36(29)5)34-25-15-21(19-9-6-7-10-22(19)31)20-13-14-37(38)28(27(20)35-25)26-23(32)11-8-12-24(26)33/h6-15,18H,16-17H2,1-5H3,(H,34,35) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357906
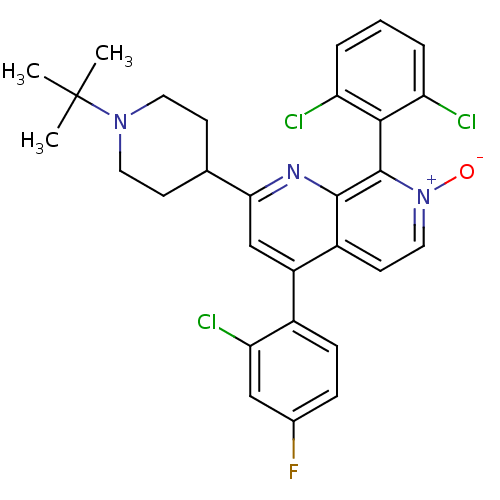
(CHEMBL1916520)Show SMILES CC(C)(C)N1CCC(CC1)c1cc(-c2ccc(F)cc2Cl)c2cc[n+]([O-])c(-c3c(Cl)cccc3Cl)c2n1 |(2.33,-7.59,;2.34,-6.04,;3.68,-5.28,;3.67,-6.81,;1.02,-5.27,;-.33,-6.03,;-1.65,-5.25,;-1.64,-3.71,;-.31,-2.94,;1.02,-3.72,;-2.98,-2.94,;-2.97,-1.4,;-4.31,-.62,;-4.31,.91,;-2.98,1.68,;-2.97,3.22,;-4.31,3.99,;-4.31,5.53,;-5.65,3.22,;-5.64,1.68,;-6.97,.91,;-5.65,-1.39,;-6.98,-.63,;-8.31,-1.4,;-8.31,-2.94,;-9.65,-3.71,;-6.98,-3.72,;-6.97,-5.25,;-8.31,-6.02,;-9.64,-5.25,;-8.31,-7.56,;-6.97,-8.33,;-5.63,-7.55,;-5.64,-6.01,;-4.31,-5.23,;-5.65,-2.94,;-4.32,-3.71,)| Show InChI InChI=1S/C29H27Cl3FN3O/c1-29(2,3)35-12-9-17(10-13-35)25-16-21(19-8-7-18(33)15-24(19)32)20-11-14-36(37)28(27(20)34-25)26-22(30)5-4-6-23(26)31/h4-8,11,14-17H,9-10,12-13H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.530 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357905
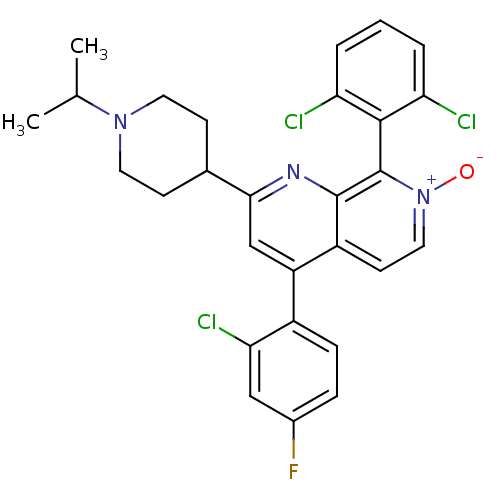
(CHEMBL1916519)Show SMILES CC(C)N1CCC(CC1)c1cc(-c2ccc(F)cc2Cl)c2cc[n+]([O-])c(-c3c(Cl)cccc3Cl)c2n1 |(32.53,-43.15,;32.54,-41.61,;33.88,-40.85,;31.21,-40.83,;29.87,-41.59,;28.54,-40.82,;28.55,-39.28,;29.88,-38.51,;31.22,-39.29,;27.23,-38.51,;27.23,-36.96,;25.89,-36.19,;25.89,-34.65,;27.23,-33.88,;27.23,-32.35,;25.89,-31.57,;25.89,-30.03,;24.55,-32.35,;24.56,-33.89,;23.23,-34.66,;24.56,-36.96,;23.22,-36.2,;21.89,-36.97,;21.89,-38.51,;20.56,-39.28,;23.22,-39.28,;23.23,-40.82,;21.89,-41.59,;20.56,-40.82,;21.9,-43.13,;23.23,-43.89,;24.57,-43.11,;24.56,-41.58,;25.89,-40.8,;24.55,-38.51,;25.88,-39.28,)| Show InChI InChI=1S/C28H25Cl3FN3O/c1-16(2)34-11-8-17(9-12-34)25-15-21(19-7-6-18(32)14-24(19)31)20-10-13-35(36)28(27(20)33-25)26-22(29)4-3-5-23(26)30/h3-7,10,13-17H,8-9,11-12H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.590 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 13
(Homo sapiens (Human)) | BDBM50420761
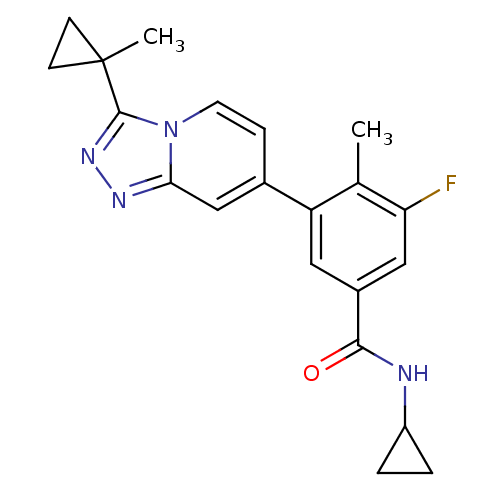
(CHEMBL2087507)Show SMILES Cc1c(F)cc(cc1-c1ccn2c(nnc2c1)C1(C)CC1)C(=O)NC1CC1 Show InChI InChI=1S/C21H21FN4O/c1-12-16(9-14(10-17(12)22)19(27)23-15-3-4-15)13-5-8-26-18(11-13)24-25-20(26)21(2)6-7-21/h5,8-11,15H,3-4,6-7H2,1-2H3,(H,23,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.640 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 22: 3431-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.099
BindingDB Entry DOI: 10.7270/Q2348MNX |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221907
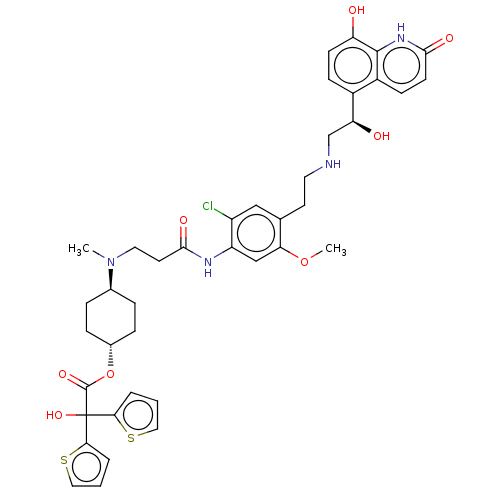
(US9315463, 20)Show SMILES COc1cc(NC(=O)CCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CCNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:41.45,15.18,wD:12.11,(-4.06,3.86,;-2.73,3.09,;-2.73,1.55,;-1.39,.78,;-1.39,-.76,;-.06,-1.53,;1.27,-.76,;1.27,.78,;2.61,-1.53,;3.94,-.76,;5.28,-1.53,;5.28,-3.07,;6.61,-.76,;7.94,-1.53,;9.28,-.76,;9.28,.78,;7.94,1.55,;6.61,.78,;10.61,1.55,;11.94,.78,;11.94,-.76,;13.28,1.55,;14.61,2.32,;14.05,.22,;13.57,-1.25,;14.82,-2.15,;16.06,-1.25,;15.59,.22,;12.51,2.89,;13.41,4.13,;12.51,5.38,;11.04,4.9,;11.04,3.36,;-2.73,-1.53,;-2.73,-3.07,;-4.06,-.76,;-4.06,.78,;-5.39,1.55,;-6.73,.78,;-8.06,1.55,;-9.39,.78,;-10.73,1.55,;-10.73,3.09,;-12.06,.78,;-13.4,1.55,;-14.73,.78,;-14.73,-.76,;-16.06,-1.53,;-13.4,-1.53,;-13.4,-3.07,;-12.06,-3.84,;-12.06,-5.38,;-10.73,-3.07,;-10.73,-1.53,;-12.06,-.76,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357904
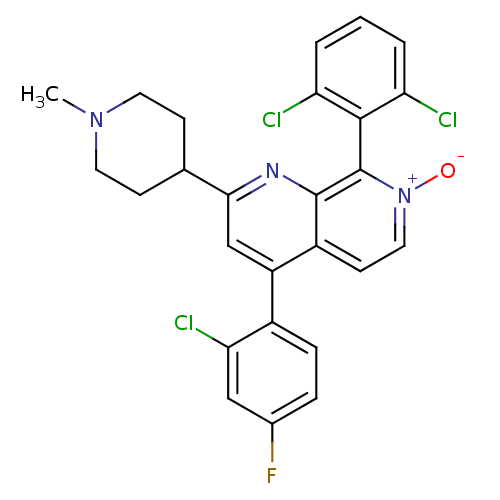
(CHEMBL1916361)Show SMILES CN1CCC(CC1)c1cc(-c2ccc(F)cc2Cl)c2cc[n+]([O-])c(-c3c(Cl)cccc3Cl)c2n1 |(17.77,-41.9,;16.45,-41.12,;15.11,-41.88,;13.78,-41.11,;13.79,-39.56,;15.12,-38.8,;16.45,-39.58,;12.46,-38.8,;12.47,-37.25,;11.13,-36.47,;11.13,-34.94,;12.46,-34.17,;12.47,-32.63,;11.13,-31.86,;11.13,-30.32,;9.79,-32.64,;9.8,-34.18,;8.47,-34.95,;9.79,-37.25,;8.46,-36.48,;7.13,-37.26,;7.13,-38.8,;5.79,-39.57,;8.46,-39.57,;8.47,-41.1,;7.13,-41.88,;5.8,-41.11,;7.13,-43.41,;8.47,-44.18,;9.81,-43.4,;9.8,-41.87,;11.13,-41.09,;9.79,-38.8,;11.12,-39.57,)| Show InChI InChI=1S/C26H21Cl3FN3O/c1-32-10-7-15(8-11-32)23-14-19(17-6-5-16(30)13-22(17)29)18-9-12-33(34)26(25(18)31-23)24-20(27)3-2-4-21(24)28/h2-6,9,12-15H,7-8,10-11H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.720 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM15242
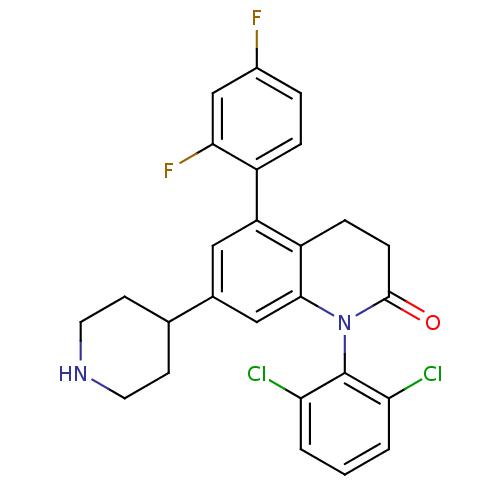
(1-(2,6-dichlorophenyl)-5-(2,4-difluorophenyl)-7-(4...)Show SMILES Fc1ccc(c(F)c1)-c1cc(cc2N(C(=O)CCc12)c1c(Cl)cccc1Cl)C1CCNCC1 Show InChI InChI=1S/C26H22Cl2F2N2O/c27-21-2-1-3-22(28)26(21)32-24-13-16(15-8-10-31-11-9-15)12-20(19(24)6-7-25(32)33)18-5-4-17(29)14-23(18)30/h1-5,12-15,31H,6-11H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 0.740 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha MAP kinase |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357902
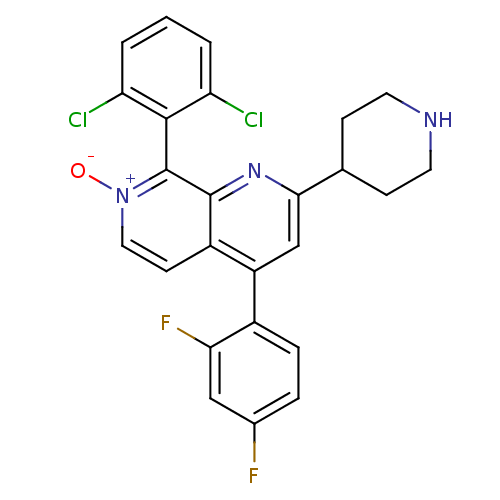
(CHEMBL1738839)Show SMILES [O-][n+]1ccc2c(cc(nc2c1-c1c(Cl)cccc1Cl)C1CCNCC1)-c1ccc(F)cc1F |(3.73,1.38,;2.66,.77,;2.66,-.77,;1.33,-1.54,;,-.77,;-1.33,-1.54,;-2.68,-.77,;-2.68,.77,;-1.33,1.54,;,.77,;1.33,1.54,;1.33,3.08,;-0,3.85,;-1.07,3.23,;-.01,5.39,;1.33,6.16,;2.66,5.39,;2.66,3.85,;3.73,3.24,;-4.01,1.54,;-5.35,.78,;-6.68,1.56,;-6.67,3.1,;-5.33,3.86,;-4,3.08,;-1.33,-3.08,;.01,-3.84,;.01,-5.38,;-1.32,-6.16,;-1.32,-7.39,;-2.65,-5.39,;-2.66,-3.85,;-3.73,-3.24,)| Show InChI InChI=1S/C25H19Cl2F2N3O/c26-19-2-1-3-20(27)23(19)25-24-17(8-11-32(25)33)18(16-5-4-15(28)12-21(16)29)13-22(31-24)14-6-9-30-10-7-14/h1-5,8,11-14,30H,6-7,9-10H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.820 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mitogen-activated protein kinase 13
(Homo sapiens (Human)) | BDBM50420769
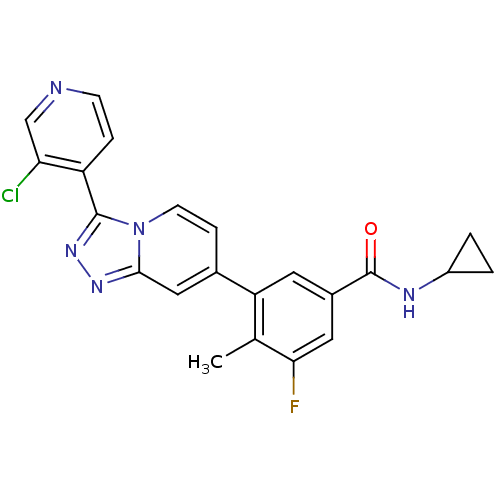
(CHEMBL2087515)Show SMILES Cc1c(F)cc(cc1-c1ccn2c(nnc2c1)-c1ccncc1Cl)C(=O)NC1CC1 Show InChI InChI=1S/C22H17ClFN5O/c1-12-17(8-14(9-19(12)24)22(30)26-15-2-3-15)13-5-7-29-20(10-13)27-28-21(29)16-4-6-25-11-18(16)23/h4-11,15H,2-3H2,1H3,(H,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.830 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 22: 3431-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.099
BindingDB Entry DOI: 10.7270/Q2348MNX |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221904
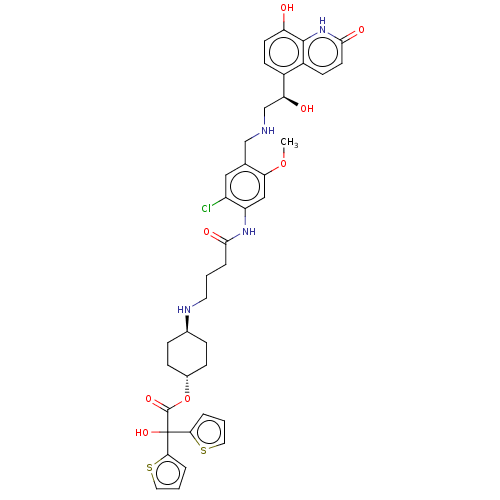
(US9315463, 15)Show SMILES COc1cc(NC(=O)CCCN[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:40.44,15.18,wD:12.11,(-5.39,-3.84,;-4.06,-3.07,;-4.06,-1.53,;-2.73,-.76,;-2.73,.78,;-1.39,1.55,;-.06,.78,;-.06,-.76,;1.27,1.55,;2.61,.78,;3.94,1.55,;5.28,.78,;6.61,1.55,;7.94,.78,;9.28,1.55,;9.28,3.09,;7.94,3.86,;6.61,3.09,;10.61,3.86,;11.94,3.09,;11.94,1.55,;13.28,3.86,;14.61,4.63,;14.05,2.53,;13.57,1.06,;14.82,.16,;16.06,1.06,;15.59,2.53,;12.51,5.2,;13.41,6.44,;12.51,7.69,;11.04,7.21,;11.04,5.67,;-4.06,1.55,;-4.06,3.09,;-5.39,.78,;-5.39,-.76,;-6.73,-1.53,;-8.06,-.76,;-9.39,-1.53,;-10.73,-.76,;-10.73,.78,;-12.06,-1.53,;-13.4,-.76,;-14.73,-1.53,;-14.73,-3.07,;-16.06,-3.84,;-13.4,-3.84,;-13.4,-5.38,;-12.06,-6.15,;-12.06,-7.69,;-10.73,-5.38,;-10.73,-3.84,;-12.06,-3.07,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM221912
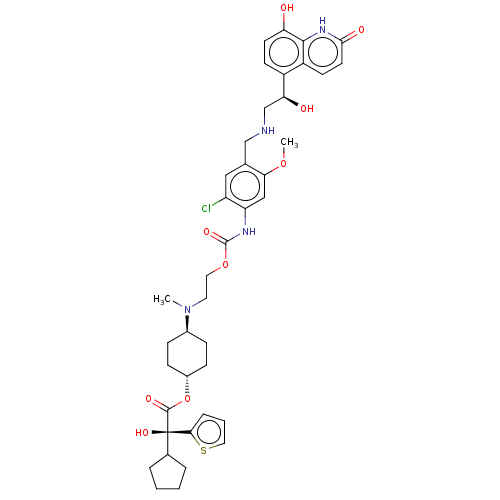
(US9315463, 29)Show SMILES COc1cc(NC(=O)OCCN(C)[C@H]2CC[C@@H](CC2)OC(=O)[C@@](O)(C2CCCC2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:41.45,16.19,22.30,wD:13.12,22.23,(-5.85,-3.78,;-4.52,-3.01,;-4.52,-1.47,;-3.19,-.7,;-3.19,.84,;-1.85,1.61,;-.52,.84,;-.52,-.7,;.82,1.61,;2.15,.84,;3.48,1.61,;4.82,.84,;4.82,-.7,;6.15,1.61,;7.48,.84,;8.82,1.61,;8.82,3.15,;7.48,3.92,;6.15,3.15,;10.15,3.92,;11.49,3.15,;11.49,1.61,;12.82,3.92,;14.15,4.69,;12.05,5.26,;12.52,6.72,;11.28,7.63,;10.03,6.72,;10.51,5.26,;14.15,3.15,;14.15,1.61,;15.62,1.14,;16.52,2.38,;15.62,3.63,;-4.52,1.61,;-4.52,3.15,;-5.85,.84,;-5.85,-.7,;-7.19,-1.47,;-8.52,-.7,;-9.85,-1.47,;-11.19,-.7,;-11.19,.84,;-12.52,-1.47,;-13.85,-.7,;-15.19,-1.47,;-15.19,-3.01,;-16.52,-3.78,;-13.85,-3.78,;-13.85,-5.32,;-12.52,-6.09,;-12.52,-7.63,;-11.19,-5.32,;-11.19,-3.78,;-12.52,-3.01,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | 25 |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human adrenergic beta1 and beta2 receptors was performed using commercial membranes prepared from Sf9 cells where they are ov... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221900
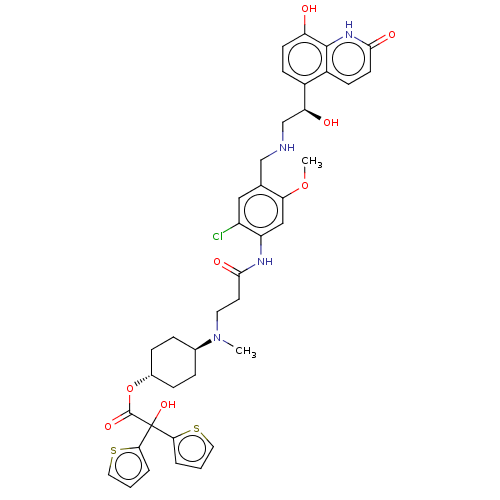
(US9315463, 9)Show SMILES COc1cc(NC(=O)CCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:40.44,15.18,wD:12.11,(-7.85,3.08,;-6.52,3.85,;-5.19,3.08,;-3.85,3.85,;-2.52,3.08,;-1.18,3.85,;.15,3.08,;.15,1.54,;1.48,3.85,;2.82,3.08,;4.15,3.85,;4.15,5.39,;5.48,3.08,;5.48,1.54,;6.82,.77,;8.15,1.54,;8.15,3.08,;6.82,3.85,;9.48,.77,;10.82,1.54,;10.82,3.08,;12.15,.77,;13.49,,;12.15,-.77,;10.91,-1.68,;11.38,-3.14,;12.92,-3.14,;13.4,-1.68,;13.49,1.54,;14.95,1.06,;15.86,2.31,;14.95,3.56,;13.49,3.08,;-2.52,1.54,;-1.18,.77,;-3.85,.77,;-5.19,1.54,;-6.52,.77,;-7.85,1.54,;-9.19,.77,;-10.52,1.54,;-10.52,3.08,;-11.85,.77,;-13.19,1.54,;-14.52,.77,;-14.52,-.77,;-15.86,-1.54,;-13.19,-1.54,;-13.19,-3.08,;-11.85,-3.85,;-11.85,-5.39,;-10.52,-3.08,;-10.52,-1.54,;-11.85,-.77,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221899
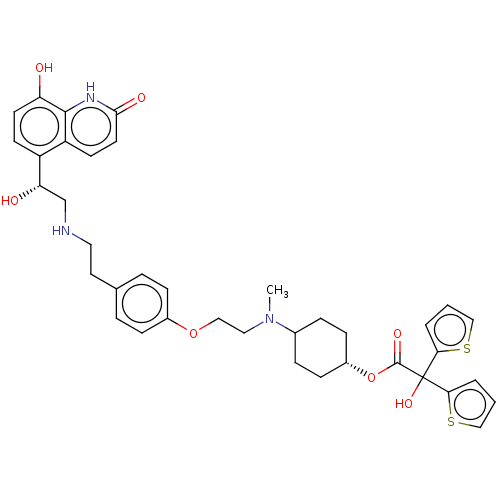
(US9315463, 2)Show SMILES CN(CCOc1ccc(CCNC[C@H](O)c2ccc(O)c3[nH]c(=O)ccc23)cc1)C1CC[C@@H](CC1)OC(=O)C(O)(c1cccs1)c1cccs1 |r,wU:13.13,32.38,(4.61,3.07,;4.61,1.53,;3.27,.76,;1.94,1.53,;.61,.76,;-.73,1.53,;-2.06,.76,;-3.39,1.53,;-3.39,3.07,;-4.73,3.84,;-6.06,3.07,;-7.39,3.84,;-8.73,3.07,;-10.06,3.84,;-10.06,5.38,;-11.4,3.07,;-12.73,3.84,;-14.06,3.07,;-14.06,1.53,;-15.4,.76,;-12.73,.76,;-12.73,-.78,;-11.4,-1.55,;-11.4,-3.09,;-10.06,-.78,;-10.06,.76,;-11.4,1.53,;-2.06,3.84,;-.73,3.07,;5.94,.76,;5.94,-.78,;7.28,-1.55,;8.61,-.78,;8.61,.76,;7.28,1.53,;9.94,-1.55,;11.28,-.78,;11.28,.76,;12.61,-1.55,;13.94,-2.32,;13.38,-.22,;14.92,-.22,;15.4,1.25,;14.15,2.15,;12.9,1.25,;11.84,-2.89,;10.38,-3.36,;10.38,-4.9,;11.84,-5.38,;12.75,-4.13,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM221911
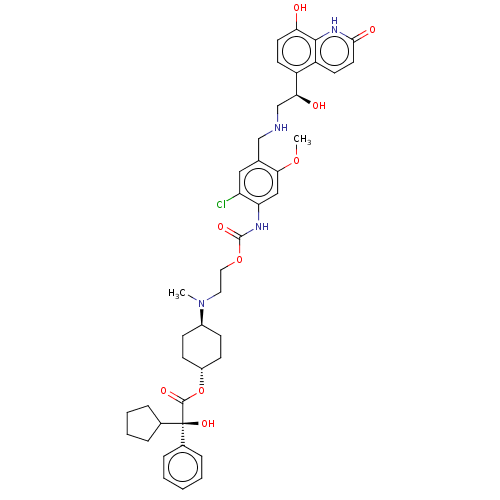
(US9315463, 28)Show SMILES COc1cc(NC(=O)OCCN(C)[C@H]2CC[C@@H](CC2)OC(=O)[C@@](O)(C2CCCC2)c2ccccc2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:42.46,16.19,22.22,wD:13.12,22.23,(-6.31,-3.88,;-4.98,-3.11,;-4.98,-1.57,;-3.64,-.8,;-3.64,.74,;-2.31,1.51,;-.98,.74,;-.98,-.8,;.36,1.51,;1.69,.74,;3.02,1.51,;4.36,.74,;4.36,-.8,;5.69,1.51,;7.03,.74,;8.36,1.51,;8.36,3.05,;7.03,3.82,;5.69,3.05,;9.69,3.82,;11.03,3.05,;11.03,1.51,;12.36,3.82,;13.13,2.49,;12.36,5.36,;13.61,6.27,;13.13,7.73,;11.59,7.73,;11.11,6.27,;13.9,3.82,;14.67,2.49,;16.21,2.49,;16.98,3.82,;16.21,5.15,;14.67,5.15,;-4.98,1.51,;-4.98,3.05,;-6.31,.74,;-6.31,-.8,;-7.64,-1.57,;-8.98,-.8,;-10.31,-1.57,;-11.65,-.8,;-11.65,.74,;-12.98,-1.57,;-14.31,-.8,;-15.65,-1.57,;-15.65,-3.11,;-16.98,-3.88,;-14.31,-3.88,;-14.31,-5.42,;-12.98,-6.19,;-12.98,-7.73,;-11.65,-5.42,;-11.65,-3.88,;-12.98,-3.11,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | 25 |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human adrenergic beta1 and beta2 receptors was performed using commercial membranes prepared from Sf9 cells where they are ov... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221908
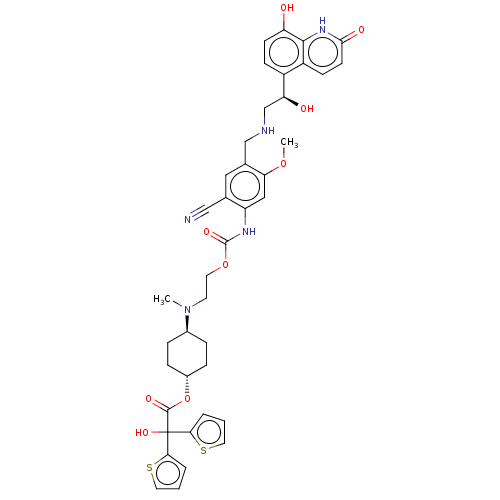
(US9315463, 22)Show SMILES COc1cc(NC(=O)OCCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12)C#N |r,wU:40.44,16.19,wD:13.12,(-5.39,-3.84,;-4.06,-3.07,;-4.06,-1.53,;-2.73,-.76,;-2.73,.78,;-1.39,1.55,;-.06,.78,;-.06,-.76,;1.27,1.55,;2.61,.78,;3.94,1.55,;5.28,.78,;5.28,-.76,;6.61,1.55,;7.94,.78,;9.28,1.55,;9.28,3.09,;7.94,3.86,;6.61,3.09,;10.61,3.86,;11.94,3.09,;11.94,1.55,;13.28,3.86,;14.61,4.63,;14.05,2.53,;13.57,1.06,;14.82,.16,;16.06,1.06,;15.59,2.53,;12.51,5.2,;13.41,6.44,;12.51,7.69,;11.04,7.21,;11.04,5.67,;-4.06,1.55,;-5.39,.78,;-5.39,-.76,;-6.73,-1.53,;-8.06,-.76,;-9.39,-1.53,;-10.73,-.76,;-10.73,.78,;-12.06,-1.53,;-13.4,-.76,;-14.73,-1.53,;-14.73,-3.07,;-16.06,-3.84,;-13.4,-3.84,;-13.4,-5.38,;-12.06,-6.15,;-12.06,-7.69,;-10.73,-5.38,;-10.73,-3.84,;-12.06,-3.07,;-4.06,3.09,;-4.06,4.63,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 13
(Homo sapiens (Human)) | BDBM50420767
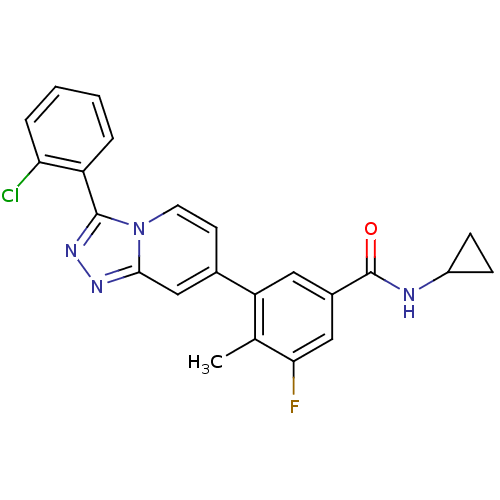
(CHEMBL2087513)Show SMILES Cc1c(F)cc(cc1-c1ccn2c(nnc2c1)-c1ccccc1Cl)C(=O)NC1CC1 Show InChI InChI=1S/C23H18ClFN4O/c1-13-18(10-15(11-20(13)25)23(30)26-16-6-7-16)14-8-9-29-21(12-14)27-28-22(29)17-4-2-3-5-19(17)24/h2-5,8-12,16H,6-7H2,1H3,(H,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 22: 3431-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.099
BindingDB Entry DOI: 10.7270/Q2348MNX |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM221902
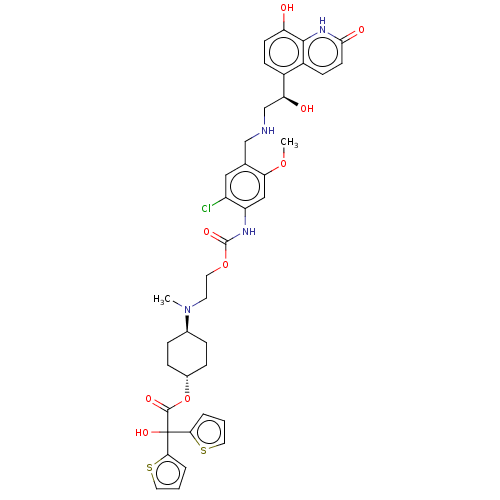
(US9315463, 12)Show SMILES COc1cc(NC(=O)OCCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:41.45,13.12,wD:16.19,(-5.85,-3.32,;-4.52,-2.55,;-4.52,-1.01,;-3.19,-.24,;-3.19,1.3,;-1.85,2.07,;-.52,1.3,;-.52,-.24,;.82,2.07,;2.15,1.3,;3.48,2.07,;4.82,1.3,;4.82,-.24,;6.15,2.07,;6.15,3.61,;7.48,4.38,;8.82,3.61,;8.82,2.07,;7.48,1.3,;10.15,4.38,;11.49,3.61,;11.49,2.07,;12.82,4.38,;12.05,5.72,;13.59,3.05,;15.13,3.05,;15.6,1.58,;14.36,.68,;13.11,1.58,;14.15,5.15,;14.15,6.69,;15.62,7.17,;16.52,5.92,;15.62,4.68,;-4.52,2.07,;-4.52,3.61,;-5.85,1.3,;-5.85,-.24,;-7.19,-1.01,;-8.52,-.24,;-9.85,-1.01,;-11.19,-.24,;-11.19,1.3,;-12.52,-1.01,;-13.85,-.24,;-15.19,-1.01,;-15.19,-2.55,;-16.52,-3.32,;-13.85,-3.32,;-13.85,-4.86,;-12.52,-5.63,;-12.52,-7.17,;-11.19,-4.86,;-11.19,-3.32,;-12.52,-2.55,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | 25 |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human adrenergic beta1 and beta2 receptors was performed using commercial membranes prepared from Sf9 cells where they are ov... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221902

(US9315463, 12)Show SMILES COc1cc(NC(=O)OCCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:41.45,13.12,wD:16.19,(-5.85,-3.32,;-4.52,-2.55,;-4.52,-1.01,;-3.19,-.24,;-3.19,1.3,;-1.85,2.07,;-.52,1.3,;-.52,-.24,;.82,2.07,;2.15,1.3,;3.48,2.07,;4.82,1.3,;4.82,-.24,;6.15,2.07,;6.15,3.61,;7.48,4.38,;8.82,3.61,;8.82,2.07,;7.48,1.3,;10.15,4.38,;11.49,3.61,;11.49,2.07,;12.82,4.38,;12.05,5.72,;13.59,3.05,;15.13,3.05,;15.6,1.58,;14.36,.68,;13.11,1.58,;14.15,5.15,;14.15,6.69,;15.62,7.17,;16.52,5.92,;15.62,4.68,;-4.52,2.07,;-4.52,3.61,;-5.85,1.3,;-5.85,-.24,;-7.19,-1.01,;-8.52,-.24,;-9.85,-1.01,;-11.19,-.24,;-11.19,1.3,;-12.52,-1.01,;-13.85,-.24,;-15.19,-1.01,;-15.19,-2.55,;-16.52,-3.32,;-13.85,-3.32,;-13.85,-4.86,;-12.52,-5.63,;-12.52,-7.17,;-11.19,-4.86,;-11.19,-3.32,;-12.52,-2.55,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221906
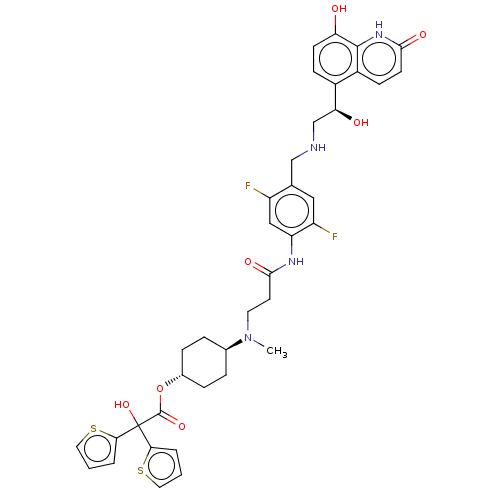
(US9315463, 18)Show SMILES CN(CCC(=O)Nc1cc(F)c(CNC[C@H](O)c2ccc(O)c3[nH]c(=O)ccc23)cc1F)[C@H]1CC[C@@H](CC1)OC(=O)C(O)(c1cccs1)c1cccs1 |r,wU:15.15,35.41,wD:32.34,(4.61,5.39,;4.61,3.85,;3.27,3.08,;1.94,3.85,;.61,3.08,;.61,1.54,;-.73,3.85,;-2.06,3.08,;-2.06,1.54,;-3.39,.77,;-3.39,-.77,;-4.73,1.54,;-6.06,.77,;-7.39,1.54,;-8.73,.77,;-10.06,1.54,;-10.06,3.08,;-11.4,.77,;-12.73,1.54,;-14.06,.77,;-14.06,-.77,;-15.4,-1.54,;-12.73,-1.54,;-12.73,-3.08,;-11.4,-3.85,;-11.4,-5.39,;-10.06,-3.08,;-10.06,-1.54,;-11.4,-.77,;-4.73,3.08,;-3.39,3.85,;-3.39,5.39,;5.94,3.08,;5.94,1.54,;7.28,.77,;8.61,1.54,;8.61,3.08,;7.28,3.85,;9.94,.77,;11.28,1.54,;11.28,3.08,;12.61,.77,;13.94,,;13.38,2.1,;12.9,3.57,;14.15,4.47,;15.4,3.57,;14.92,2.1,;11.84,-.56,;10.38,-1.04,;10.38,-2.58,;11.84,-3.06,;12.75,-1.81,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 13
(Homo sapiens (Human)) | BDBM50420746
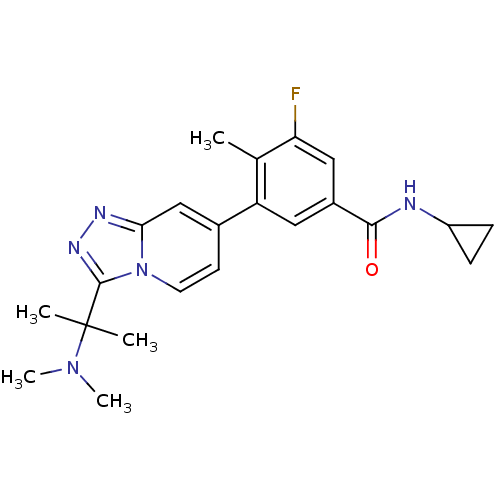
(CHEMBL2087524)Show SMILES CN(C)C(C)(C)c1nnc2cc(ccn12)-c1cc(cc(F)c1C)C(=O)NC1CC1 Show InChI InChI=1S/C22H26FN5O/c1-13-17(10-15(11-18(13)23)20(29)24-16-6-7-16)14-8-9-28-19(12-14)25-26-21(28)22(2,3)27(4)5/h8-12,16H,6-7H2,1-5H3,(H,24,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 22: 3431-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.099
BindingDB Entry DOI: 10.7270/Q2348MNX |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221910

(US9315463, 26)Show SMILES COc1cc(NC(=O)OCCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2NC(=O)COc12 |r,wU:41.45,16.19,wD:13.12,(-2.73,-3.84,;-4.06,-3.07,;-4.06,-1.53,;-2.73,-.76,;-2.73,.78,;-1.39,1.55,;-.06,.78,;-.06,-.76,;1.27,1.55,;2.61,.78,;3.94,1.55,;5.28,.78,;5.28,-.76,;6.61,1.55,;7.94,.78,;9.28,1.55,;9.28,3.09,;7.94,3.86,;6.61,3.09,;10.61,3.86,;11.94,3.09,;11.94,1.55,;13.28,3.86,;14.61,4.63,;14.05,2.53,;13.57,1.06,;14.82,.16,;16.06,1.06,;15.59,2.53,;12.51,5.2,;13.41,6.44,;12.51,7.69,;11.04,7.21,;11.04,5.67,;-4.06,1.55,;-4.06,3.09,;-5.39,.78,;-5.39,-.76,;-6.73,-1.53,;-8.06,-.76,;-9.39,-1.53,;-10.73,-.76,;-10.73,.78,;-12.06,-1.53,;-13.4,-.76,;-14.73,-1.53,;-14.73,-3.07,;-16.06,-3.84,;-13.4,-3.84,;-13.4,-5.38,;-12.06,-6.15,;-12.06,-7.69,;-10.73,-5.38,;-10.73,-3.84,;-12.06,-3.07,)| Show InChI InChI=1S/C38H43ClN4O10S2/c1-43(23-7-9-24(10-8-23)53-36(47)38(49,31-5-3-15-54-31)32-6-4-16-55-32)13-14-51-37(48)41-27-18-30(50-2)22(17-26(27)39)19-40-20-29(45)25-11-12-28(44)34-35(25)52-21-33(46)42-34/h3-6,11-12,15-18,23-24,29,40,44-45,49H,7-10,13-14,19-21H2,1-2H3,(H,41,48)(H,42,46)/t23-,24-,29-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 13
(Homo sapiens (Human)) | BDBM50420763
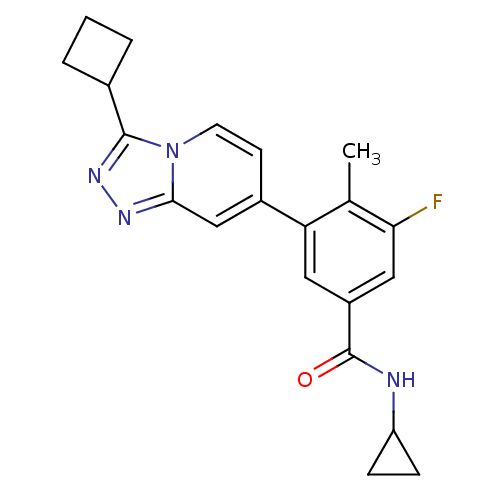
(CHEMBL2087509)Show SMILES Cc1c(F)cc(cc1-c1ccn2c(nnc2c1)C1CCC1)C(=O)NC1CC1 Show InChI InChI=1S/C21H21FN4O/c1-12-17(9-15(10-18(12)22)21(27)23-16-5-6-16)14-7-8-26-19(11-14)24-25-20(26)13-3-2-4-13/h7-11,13,16H,2-6H2,1H3,(H,23,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 22: 3431-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.099
BindingDB Entry DOI: 10.7270/Q2348MNX |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221905
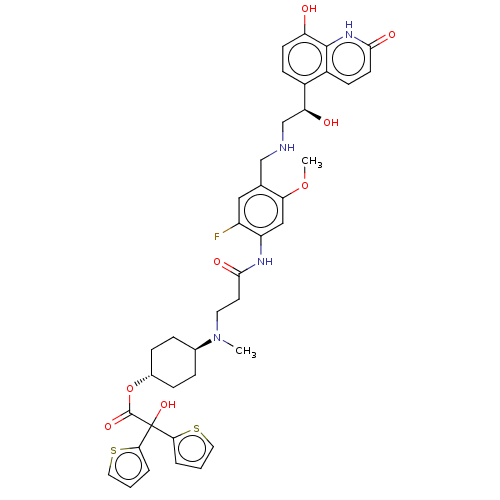
(US9315463, 16)Show SMILES COc1cc(NC(=O)CCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(F)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:40.44,15.18,wD:12.11,(-2.06,-1.54,;-3.39,-.77,;-3.39,.77,;-2.06,1.54,;-2.06,3.08,;-.73,3.85,;.61,3.08,;.61,1.54,;1.94,3.85,;3.27,3.08,;4.61,3.85,;4.61,5.39,;5.94,3.08,;5.94,1.54,;7.28,.77,;8.61,1.54,;8.61,3.08,;7.28,3.85,;9.94,.77,;11.28,1.54,;11.28,3.08,;12.61,.77,;13.94,,;13.38,2.1,;12.9,3.57,;14.15,4.47,;15.4,3.57,;14.92,2.1,;11.84,-.56,;10.38,-1.04,;10.38,-2.58,;11.84,-3.06,;12.75,-1.81,;-3.39,3.85,;-3.39,5.39,;-4.73,3.08,;-4.73,1.54,;-6.06,.77,;-7.39,1.54,;-8.73,.77,;-10.06,1.54,;-10.06,3.08,;-11.4,.77,;-12.73,1.54,;-14.06,.77,;-14.06,-.77,;-15.4,-1.54,;-12.73,-1.54,;-12.73,-3.08,;-11.4,-3.85,;-11.4,-5.39,;-10.06,-3.08,;-10.06,-1.54,;-11.4,-.77,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 13
(Homo sapiens (Human)) | BDBM50420756
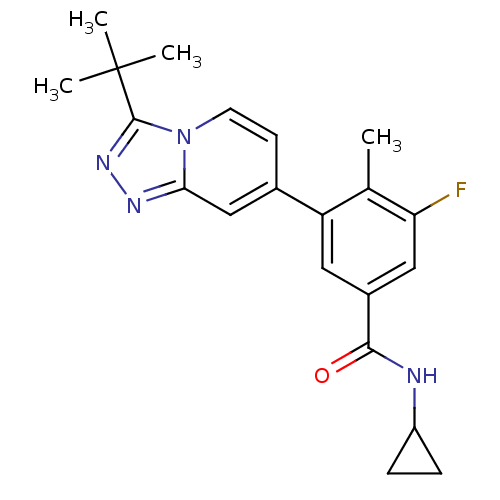
(CHEMBL2087501)Show SMILES Cc1c(F)cc(cc1-c1ccn2c(nnc2c1)C(C)(C)C)C(=O)NC1CC1 Show InChI InChI=1S/C21H23FN4O/c1-12-16(9-14(10-17(12)22)19(27)23-15-5-6-15)13-7-8-26-18(11-13)24-25-20(26)21(2,3)4/h7-11,15H,5-6H2,1-4H3,(H,23,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 22: 3431-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.099
BindingDB Entry DOI: 10.7270/Q2348MNX |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221903
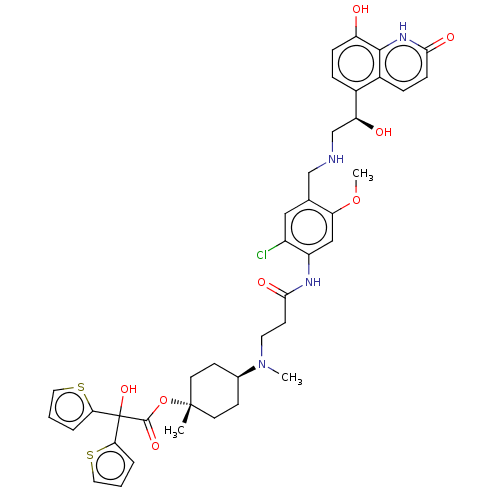
(US9315463, 13)Show SMILES COc1cc(NC(=O)CCN(C)[C@H]2CC[C@@](C)(CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:41.45,15.19,wD:12.11,15.15,(-4.73,-1.54,;-3.39,-.77,;-3.39,.77,;-2.06,1.54,;-2.06,3.08,;-.73,3.85,;.61,3.08,;.61,1.54,;1.94,3.85,;3.27,3.08,;4.61,3.85,;4.61,5.39,;5.94,3.08,;5.94,1.54,;7.28,.77,;8.61,1.54,;9.94,2.31,;8.61,3.08,;7.28,3.85,;9.94,.77,;11.28,1.54,;11.28,3.08,;12.61,.77,;13.94,,;13.38,2.1,;12.9,3.57,;14.15,4.47,;15.4,3.57,;14.92,2.1,;11.84,-.56,;10.38,-1.04,;10.38,-2.58,;11.84,-3.06,;12.75,-1.81,;-3.39,3.85,;-3.39,5.39,;-4.73,3.08,;-4.73,1.54,;-6.06,.77,;-7.39,1.54,;-8.73,.77,;-10.06,1.54,;-10.06,3.08,;-11.4,.77,;-12.73,1.54,;-14.06,.77,;-14.06,-.77,;-15.4,-1.54,;-12.73,-1.54,;-12.73,-3.08,;-11.4,-3.85,;-11.4,-5.39,;-10.06,-3.08,;-10.06,-1.54,;-11.4,-.77,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM221904

(US9315463, 15)Show SMILES COc1cc(NC(=O)CCCN[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:40.44,15.18,wD:12.11,(-5.39,-3.84,;-4.06,-3.07,;-4.06,-1.53,;-2.73,-.76,;-2.73,.78,;-1.39,1.55,;-.06,.78,;-.06,-.76,;1.27,1.55,;2.61,.78,;3.94,1.55,;5.28,.78,;6.61,1.55,;7.94,.78,;9.28,1.55,;9.28,3.09,;7.94,3.86,;6.61,3.09,;10.61,3.86,;11.94,3.09,;11.94,1.55,;13.28,3.86,;14.61,4.63,;14.05,2.53,;13.57,1.06,;14.82,.16,;16.06,1.06,;15.59,2.53,;12.51,5.2,;13.41,6.44,;12.51,7.69,;11.04,7.21,;11.04,5.67,;-4.06,1.55,;-4.06,3.09,;-5.39,.78,;-5.39,-.76,;-6.73,-1.53,;-8.06,-.76,;-9.39,-1.53,;-10.73,-.76,;-10.73,.78,;-12.06,-1.53,;-13.4,-.76,;-14.73,-1.53,;-14.73,-3.07,;-16.06,-3.84,;-13.4,-3.84,;-13.4,-5.38,;-12.06,-6.15,;-12.06,-7.69,;-10.73,-5.38,;-10.73,-3.84,;-12.06,-3.07,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | 25 |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human adrenergic beta1 and beta2 receptors was performed using commercial membranes prepared from Sf9 cells where they are ov... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221901
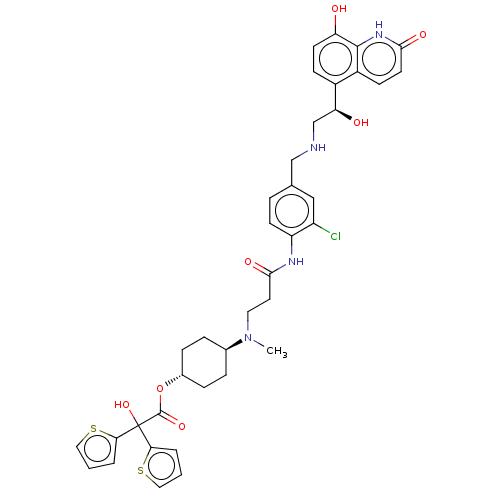
(US9315463, 10)Show SMILES CN(CCC(=O)Nc1ccc(CNC[C@H](O)c2ccc(O)c3[nH]c(=O)ccc23)cc1Cl)[C@H]1CC[C@@H](CC1)OC(=O)C(O)(c1cccs1)c1cccs1 |r,wU:14.14,34.40,wD:31.33,(4.15,5.39,;4.15,3.85,;2.82,3.08,;1.48,3.85,;.15,3.08,;.15,1.54,;-1.18,3.85,;-2.52,3.08,;-2.52,1.54,;-3.85,.77,;-5.19,1.54,;-6.52,.77,;-7.85,1.54,;-9.19,.77,;-10.52,1.54,;-10.52,3.08,;-11.85,.77,;-13.19,1.54,;-14.52,.77,;-14.52,-.77,;-15.86,-1.54,;-13.19,-1.54,;-13.19,-3.08,;-11.85,-3.85,;-11.85,-5.39,;-10.52,-3.08,;-10.52,-1.54,;-11.85,-.77,;-5.19,3.08,;-3.85,3.85,;-3.85,5.39,;5.48,3.08,;5.48,1.54,;6.82,.77,;8.15,1.54,;8.15,3.08,;6.82,3.85,;9.48,.77,;10.82,1.54,;10.82,3.08,;12.15,.77,;13.49,,;12.15,-.77,;13.4,-1.68,;12.92,-3.14,;11.38,-3.14,;10.91,-1.68,;13.49,1.54,;14.95,1.06,;15.86,2.31,;14.95,3.56,;13.49,3.08,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM221903

(US9315463, 13)Show SMILES COc1cc(NC(=O)CCN(C)[C@H]2CC[C@@](C)(CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:41.45,15.19,wD:12.11,15.15,(-4.73,-1.54,;-3.39,-.77,;-3.39,.77,;-2.06,1.54,;-2.06,3.08,;-.73,3.85,;.61,3.08,;.61,1.54,;1.94,3.85,;3.27,3.08,;4.61,3.85,;4.61,5.39,;5.94,3.08,;5.94,1.54,;7.28,.77,;8.61,1.54,;9.94,2.31,;8.61,3.08,;7.28,3.85,;9.94,.77,;11.28,1.54,;11.28,3.08,;12.61,.77,;13.94,,;13.38,2.1,;12.9,3.57,;14.15,4.47,;15.4,3.57,;14.92,2.1,;11.84,-.56,;10.38,-1.04,;10.38,-2.58,;11.84,-3.06,;12.75,-1.81,;-3.39,3.85,;-3.39,5.39,;-4.73,3.08,;-4.73,1.54,;-6.06,.77,;-7.39,1.54,;-8.73,.77,;-10.06,1.54,;-10.06,3.08,;-11.4,.77,;-12.73,1.54,;-14.06,.77,;-14.06,-.77,;-15.4,-1.54,;-12.73,-1.54,;-12.73,-3.08,;-11.4,-3.85,;-11.4,-5.39,;-10.06,-3.08,;-10.06,-1.54,;-11.4,-.77,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | 25 |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human adrenergic beta1 and beta2 receptors was performed using commercial membranes prepared from Sf9 cells where they are ov... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 13
(Homo sapiens (Human)) | BDBM50420758

(CHEMBL2087503)Show SMILES CC(C)c1nnc2cc(ccn12)-c1cc(cc(F)c1C)C(=O)NC1CC1 Show InChI InChI=1S/C20H21FN4O/c1-11(2)19-24-23-18-10-13(6-7-25(18)19)16-8-14(9-17(21)12(16)3)20(26)22-15-4-5-15/h6-11,15H,4-5H2,1-3H3,(H,22,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 22: 3431-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.099
BindingDB Entry DOI: 10.7270/Q2348MNX |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 13
(Homo sapiens (Human)) | BDBM50420747
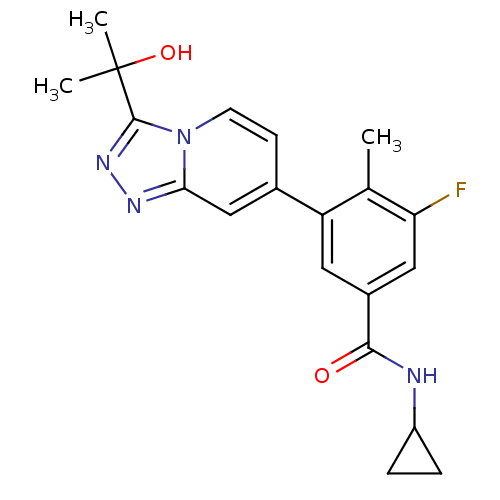
(CHEMBL2087526)Show SMILES Cc1c(F)cc(cc1-c1ccn2c(nnc2c1)C(C)(C)O)C(=O)NC1CC1 Show InChI InChI=1S/C20H21FN4O2/c1-11-15(8-13(9-16(11)21)18(26)22-14-4-5-14)12-6-7-25-17(10-12)23-24-19(25)20(2,3)27/h6-10,14,27H,4-5H2,1-3H3,(H,22,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha |
Bioorg Med Chem Lett 22: 3431-6 (2012)
Article DOI: 10.1016/j.bmcl.2012.03.099
BindingDB Entry DOI: 10.7270/Q2348MNX |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM221912

(US9315463, 29)Show SMILES COc1cc(NC(=O)OCCN(C)[C@H]2CC[C@@H](CC2)OC(=O)[C@@](O)(C2CCCC2)c2cccs2)c(Cl)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:41.45,16.19,22.30,wD:13.12,22.23,(-5.85,-3.78,;-4.52,-3.01,;-4.52,-1.47,;-3.19,-.7,;-3.19,.84,;-1.85,1.61,;-.52,.84,;-.52,-.7,;.82,1.61,;2.15,.84,;3.48,1.61,;4.82,.84,;4.82,-.7,;6.15,1.61,;7.48,.84,;8.82,1.61,;8.82,3.15,;7.48,3.92,;6.15,3.15,;10.15,3.92,;11.49,3.15,;11.49,1.61,;12.82,3.92,;14.15,4.69,;12.05,5.26,;12.52,6.72,;11.28,7.63,;10.03,6.72,;10.51,5.26,;14.15,3.15,;14.15,1.61,;15.62,1.14,;16.52,2.38,;15.62,3.63,;-4.52,1.61,;-4.52,3.15,;-5.85,.84,;-5.85,-.7,;-7.19,-1.47,;-8.52,-.7,;-9.85,-1.47,;-11.19,-.7,;-11.19,.84,;-12.52,-1.47,;-13.85,-.7,;-15.19,-1.47,;-15.19,-3.01,;-16.52,-3.78,;-13.85,-3.78,;-13.85,-5.32,;-12.52,-6.09,;-12.52,-7.63,;-11.19,-5.32,;-11.19,-3.78,;-12.52,-3.01,)| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 3.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human muscarinic M1, M2, M3, M4 and M5 receptors was performed using commercial membranes (Perkin Elmer) prepared from CHO-K1... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM221908

(US9315463, 22)Show SMILES COc1cc(NC(=O)OCCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12)C#N |r,wU:40.44,16.19,wD:13.12,(-5.39,-3.84,;-4.06,-3.07,;-4.06,-1.53,;-2.73,-.76,;-2.73,.78,;-1.39,1.55,;-.06,.78,;-.06,-.76,;1.27,1.55,;2.61,.78,;3.94,1.55,;5.28,.78,;5.28,-.76,;6.61,1.55,;7.94,.78,;9.28,1.55,;9.28,3.09,;7.94,3.86,;6.61,3.09,;10.61,3.86,;11.94,3.09,;11.94,1.55,;13.28,3.86,;14.61,4.63,;14.05,2.53,;13.57,1.06,;14.82,.16,;16.06,1.06,;15.59,2.53,;12.51,5.2,;13.41,6.44,;12.51,7.69,;11.04,7.21,;11.04,5.67,;-4.06,1.55,;-5.39,.78,;-5.39,-.76,;-6.73,-1.53,;-8.06,-.76,;-9.39,-1.53,;-10.73,-.76,;-10.73,.78,;-12.06,-1.53,;-13.4,-.76,;-14.73,-1.53,;-14.73,-3.07,;-16.06,-3.84,;-13.4,-3.84,;-13.4,-5.38,;-12.06,-6.15,;-12.06,-7.69,;-10.73,-5.38,;-10.73,-3.84,;-12.06,-3.07,;-4.06,3.09,;-4.06,4.63,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | 25 |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human adrenergic beta1 and beta2 receptors was performed using commercial membranes prepared from Sf9 cells where they are ov... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM221905

(US9315463, 16)Show SMILES COc1cc(NC(=O)CCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(F)cc1CNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:40.44,15.18,wD:12.11,(-2.06,-1.54,;-3.39,-.77,;-3.39,.77,;-2.06,1.54,;-2.06,3.08,;-.73,3.85,;.61,3.08,;.61,1.54,;1.94,3.85,;3.27,3.08,;4.61,3.85,;4.61,5.39,;5.94,3.08,;5.94,1.54,;7.28,.77,;8.61,1.54,;8.61,3.08,;7.28,3.85,;9.94,.77,;11.28,1.54,;11.28,3.08,;12.61,.77,;13.94,,;13.38,2.1,;12.9,3.57,;14.15,4.47,;15.4,3.57,;14.92,2.1,;11.84,-.56,;10.38,-1.04,;10.38,-2.58,;11.84,-3.06,;12.75,-1.81,;-3.39,3.85,;-3.39,5.39,;-4.73,3.08,;-4.73,1.54,;-6.06,.77,;-7.39,1.54,;-8.73,.77,;-10.06,1.54,;-10.06,3.08,;-11.4,.77,;-12.73,1.54,;-14.06,.77,;-14.06,-.77,;-15.4,-1.54,;-12.73,-1.54,;-12.73,-3.08,;-11.4,-3.85,;-11.4,-5.39,;-10.06,-3.08,;-10.06,-1.54,;-11.4,-.77,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | 25 |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human adrenergic beta1 and beta2 receptors was performed using commercial membranes prepared from Sf9 cells where they are ov... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM31629
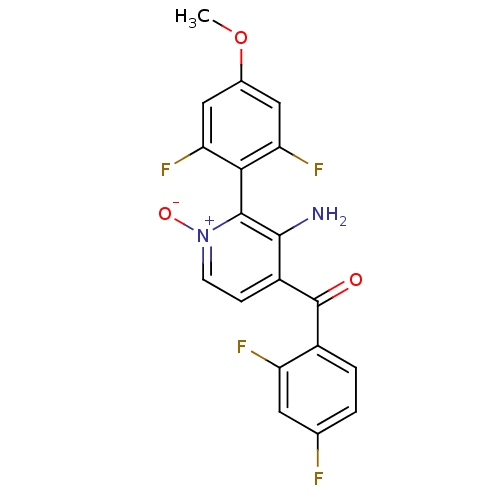
(Aminopyridine N-oxide, 55)Show SMILES COc1cc(F)c(c(F)c1)-c1c(N)c(cc[n+]1[O-])C(=O)c1ccc(F)cc1F |(5.09,2.69,;3.75,1.92,;2.42,2.69,;1.08,1.92,;-.25,2.69,;-1.58,1.92,;-.25,4.23,;1.08,5.01,;1.08,6.55,;2.42,4.23,;-1.58,5,;-1.58,6.55,;-.25,7.32,;-2.92,7.32,;-4.25,6.55,;-4.25,5,;-2.92,4.23,;-2.92,2.69,;-2.92,8.86,;-1.58,9.63,;-4.25,9.63,;-5.59,8.87,;-6.92,9.66,;-6.9,11.2,;-8.22,11.99,;-5.55,11.95,;-4.23,11.16,;-2.89,11.92,)| Show InChI InChI=1S/C19H12F4N2O3/c1-28-10-7-14(22)16(15(23)8-10)18-17(24)12(4-5-25(18)27)19(26)11-3-2-9(20)6-13(11)21/h2-8H,24H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | 7.5 | 25 |
Almirall Research Center
| Assay Description
Enzymatic activity assay was performed in 96-well microtiter plates. Various concentrations of the test compound or vehicle controls were preincubate... |
J Med Chem 52: 5531-45 (2009)
Article DOI: 10.1021/jm9008604
BindingDB Entry DOI: 10.7270/Q25719B5 |
More data for this
Ligand-Target Pair | |
Isocitrate dehydrogenase [NADP] cytoplasmic [R132C]
(Homo sapiens (Human)) | BDBM448754
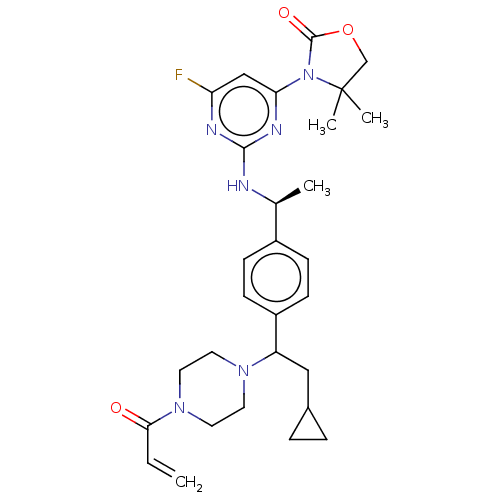
(3-[2-[[(1S)-1-[4-[2-Cyclopropyl-1-(4-prop-2-enoylp...)Show SMILES C[C@H](Nc1nc(F)cc(n1)N1C(=O)OCC1(C)C)c1ccc(cc1)C(CC1CC1)N1CCN(CC1)C(=O)C=C |r| Show InChI InChI=1S/C29H37FN6O3/c1-5-26(37)35-14-12-34(13-15-35)23(16-20-6-7-20)22-10-8-21(9-11-22)19(2)31-27-32-24(30)17-25(33-27)36-28(38)39-18-29(36,3)4/h5,8-11,17,19-20,23H,1,6-7,12-16,18H2,2-4H3,(H,31,32,33)/t19-,23?/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <5.08 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
US Patent
| Assay Description
IDH1 mutant (R132H and R132C) and IDH2 mutant (R140Q and R172K) proteins containing N-terminal His-tag are expressed in E. coli and purified using ni... |
US Patent US10696665 (2020)
BindingDB Entry DOI: 10.7270/Q2ZK5KP2 |
More data for this
Ligand-Target Pair | |
Isocitrate dehydrogenase [NADP] cytoplasmic [R132H]
(Homo sapiens (Human)) | BDBM448752

((4S)-3-[2-[[(1S)-1-[4-[2-Cyclopropyl-1-(4-prop-2-e...)Show SMILES CC(C)[C@H]1COC(=O)N1c1ccnc(N[C@@H](C)c2ccc(cc2)C(CC2CC2)N2CCN(CC2)C(=O)C=C)n1 |r| Show InChI InChI=1S/C30H40N6O3/c1-5-28(37)35-16-14-34(15-17-35)25(18-22-6-7-22)24-10-8-23(9-11-24)21(4)32-29-31-13-12-27(33-29)36-26(20(2)3)19-39-30(36)38/h5,8-13,20-22,25-26H,1,6-7,14-19H2,2-4H3,(H,31,32,33)/t21-,25?,26+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <5.08 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
US Patent
| Assay Description
IDH1 mutant (R132H and R132C) and IDH2 mutant (R140Q and R172K) proteins containing N-terminal His-tag are expressed in E. coli and purified using ni... |
US Patent US10696665 (2020)
BindingDB Entry DOI: 10.7270/Q2ZK5KP2 |
More data for this
Ligand-Target Pair | |
Isocitrate dehydrogenase [NADP] cytoplasmic [R132H]
(Homo sapiens (Human)) | BDBM448754

(3-[2-[[(1S)-1-[4-[2-Cyclopropyl-1-(4-prop-2-enoylp...)Show SMILES C[C@H](Nc1nc(F)cc(n1)N1C(=O)OCC1(C)C)c1ccc(cc1)C(CC1CC1)N1CCN(CC1)C(=O)C=C |r| Show InChI InChI=1S/C29H37FN6O3/c1-5-26(37)35-14-12-34(13-15-35)23(16-20-6-7-20)22-10-8-21(9-11-22)19(2)31-27-32-24(30)17-25(33-27)36-28(38)39-18-29(36,3)4/h5,8-11,17,19-20,23H,1,6-7,12-16,18H2,2-4H3,(H,31,32,33)/t19-,23?/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <5.08 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
US Patent
| Assay Description
IDH1 mutant (R132H and R132C) and IDH2 mutant (R140Q and R172K) proteins containing N-terminal His-tag are expressed in E. coli and purified using ni... |
US Patent US10696665 (2020)
BindingDB Entry DOI: 10.7270/Q2ZK5KP2 |
More data for this
Ligand-Target Pair | |
Isocitrate dehydrogenase [NADP] cytoplasmic [R132H]
(Homo sapiens (Human)) | BDBM448752

((4S)-3-[2-[[(1S)-1-[4-[2-Cyclopropyl-1-(4-prop-2-e...)Show SMILES CC(C)[C@H]1COC(=O)N1c1ccnc(N[C@@H](C)c2ccc(cc2)C(CC2CC2)N2CCN(CC2)C(=O)C=C)n1 |r| Show InChI InChI=1S/C30H40N6O3/c1-5-28(37)35-16-14-34(15-17-35)25(18-22-6-7-22)24-10-8-23(9-11-24)21(4)32-29-31-13-12-27(33-29)36-26(20(2)3)19-39-30(36)38/h5,8-13,20-22,25-26H,1,6-7,14-19H2,2-4H3,(H,31,32,33)/t21-,25?,26+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <5.08 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
US Patent
| Assay Description
IDH1 mutant (R132H and R132C) and IDH2 mutant (R140Q and R172K) proteins containing N-terminal His-tag are expressed in E. coli and purified using ni... |
US Patent US10696665 (2020)
BindingDB Entry DOI: 10.7270/Q2ZK5KP2 |
More data for this
Ligand-Target Pair | |
Isocitrate dehydrogenase [NADP] cytoplasmic [R132C]
(Homo sapiens (Human)) | BDBM448752

((4S)-3-[2-[[(1S)-1-[4-[2-Cyclopropyl-1-(4-prop-2-e...)Show SMILES CC(C)[C@H]1COC(=O)N1c1ccnc(N[C@@H](C)c2ccc(cc2)C(CC2CC2)N2CCN(CC2)C(=O)C=C)n1 |r| Show InChI InChI=1S/C30H40N6O3/c1-5-28(37)35-16-14-34(15-17-35)25(18-22-6-7-22)24-10-8-23(9-11-24)21(4)32-29-31-13-12-27(33-29)36-26(20(2)3)19-39-30(36)38/h5,8-13,20-22,25-26H,1,6-7,14-19H2,2-4H3,(H,31,32,33)/t21-,25?,26+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <5.08 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
US Patent
| Assay Description
IDH1 mutant (R132H and R132C) and IDH2 mutant (R140Q and R172K) proteins containing N-terminal His-tag are expressed in E. coli and purified using ni... |
US Patent US10696665 (2020)
BindingDB Entry DOI: 10.7270/Q2ZK5KP2 |
More data for this
Ligand-Target Pair | |
Isocitrate dehydrogenase [NADP] cytoplasmic [R132C]
(Homo sapiens (Human)) | BDBM448752

((4S)-3-[2-[[(1S)-1-[4-[2-Cyclopropyl-1-(4-prop-2-e...)Show SMILES CC(C)[C@H]1COC(=O)N1c1ccnc(N[C@@H](C)c2ccc(cc2)C(CC2CC2)N2CCN(CC2)C(=O)C=C)n1 |r| Show InChI InChI=1S/C30H40N6O3/c1-5-28(37)35-16-14-34(15-17-35)25(18-22-6-7-22)24-10-8-23(9-11-24)21(4)32-29-31-13-12-27(33-29)36-26(20(2)3)19-39-30(36)38/h5,8-13,20-22,25-26H,1,6-7,14-19H2,2-4H3,(H,31,32,33)/t21-,25?,26+/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <5.08 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
US Patent
| Assay Description
IDH1 mutant (R132H and R132C) and IDH2 mutant (R140Q and R172K) proteins containing N-terminal His-tag are expressed in E. coli and purified using ni... |
US Patent US10696665 (2020)
BindingDB Entry DOI: 10.7270/Q2ZK5KP2 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM221907

(US9315463, 20)Show SMILES COc1cc(NC(=O)CCN(C)[C@H]2CC[C@@H](CC2)OC(=O)C(O)(c2cccs2)c2cccs2)c(Cl)cc1CCNC[C@H](O)c1ccc(O)c2[nH]c(=O)ccc12 |r,wU:41.45,15.18,wD:12.11,(-4.06,3.86,;-2.73,3.09,;-2.73,1.55,;-1.39,.78,;-1.39,-.76,;-.06,-1.53,;1.27,-.76,;1.27,.78,;2.61,-1.53,;3.94,-.76,;5.28,-1.53,;5.28,-3.07,;6.61,-.76,;7.94,-1.53,;9.28,-.76,;9.28,.78,;7.94,1.55,;6.61,.78,;10.61,1.55,;11.94,.78,;11.94,-.76,;13.28,1.55,;14.61,2.32,;14.05,.22,;13.57,-1.25,;14.82,-2.15,;16.06,-1.25,;15.59,.22,;12.51,2.89,;13.41,4.13,;12.51,5.38,;11.04,4.9,;11.04,3.36,;-2.73,-1.53,;-2.73,-3.07,;-4.06,-.76,;-4.06,.78,;-5.39,1.55,;-6.73,.78,;-8.06,1.55,;-9.39,.78,;-10.73,1.55,;-10.73,3.09,;-12.06,.78,;-13.4,1.55,;-14.73,.78,;-14.73,-.76,;-16.06,-1.53,;-13.4,-1.53,;-13.4,-3.07,;-12.06,-3.84,;-12.06,-5.38,;-10.73,-3.07,;-10.73,-1.53,;-12.06,-.76,)| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | 25 |
Almirall, S.A.
US Patent
| Assay Description
The study of binding to human adrenergic beta1 and beta2 receptors was performed using commercial membranes prepared from Sf9 cells where they are ov... |
US Patent US9315463 (2016)
BindingDB Entry DOI: 10.7270/Q24X56MM |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data