 Found 121 hits with Last Name = 'mokube' and Initial = 'f'
Found 121 hits with Last Name = 'mokube' and Initial = 'f' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.79E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Displacement of [3H]LSD from human 5HT6 receptor expressed in CHO cells after 120 mins by scintillation counting method |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1D
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >2.40E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Activity at human 5HT1D receptor |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1F
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >2.97E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Activity at human 5HT1F receptor |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.02E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Activity at human 5HT1A receptor |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2B
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.30E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Activity at human 5T2B receptor |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1B
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.58E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Activity at human 5HT1B receptor |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 7
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.98E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Displacement of [3H]LSD from human 5HT7 receptor expressed in CHO cells after 120 mins by scintillation counting method |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1E
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >5.37E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Activity at human 5HT1E receptor |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 4
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >6.31E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Displacement of human recombinant 5HT4E receptor expressed in CHO cells after 60 mins by scintillation counting method |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 5A
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >6.90E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Displacement of human 5HT5A receptor expressed in HEK293 cells after 120 mins by scintillation counting method |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >7.47E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Activity at human 5HT2A receptor |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2C
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >7.87E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Activity at human 5T2C receptor |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207410
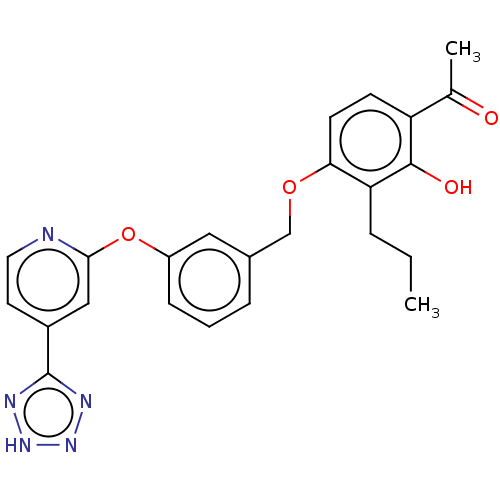
(CHEMBL3909995)Show SMILES CCCc1c(OCc2cccc(Oc3cc(ccn3)-c3nn[nH]n3)c2)ccc(C(C)=O)c1O Show InChI InChI=1S/C24H23N5O4/c1-3-5-20-21(9-8-19(15(2)30)23(20)31)32-14-16-6-4-7-18(12-16)33-22-13-17(10-11-25-22)24-26-28-29-27-24/h4,6-13,31H,3,5,14H2,1-2H3,(H,26,27,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 62 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207406
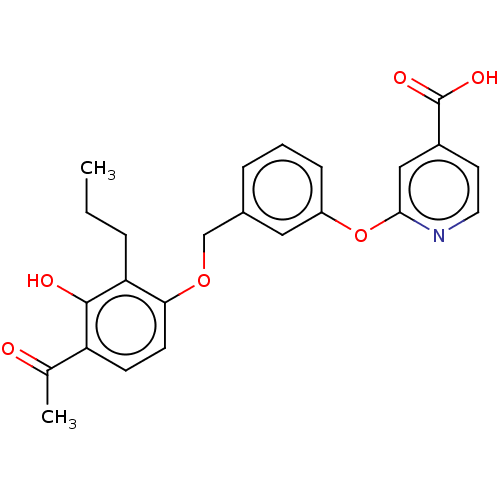
(CHEMBL3890848)Show SMILES CCCc1c(OCc2cccc(Oc3cc(ccn3)C(O)=O)c2)ccc(C(C)=O)c1O Show InChI InChI=1S/C24H23NO6/c1-3-5-20-21(9-8-19(15(2)26)23(20)27)30-14-16-6-4-7-18(12-16)31-22-13-17(24(28)29)10-11-25-22/h4,6-13,27H,3,5,14H2,1-2H3,(H,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 74 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207398
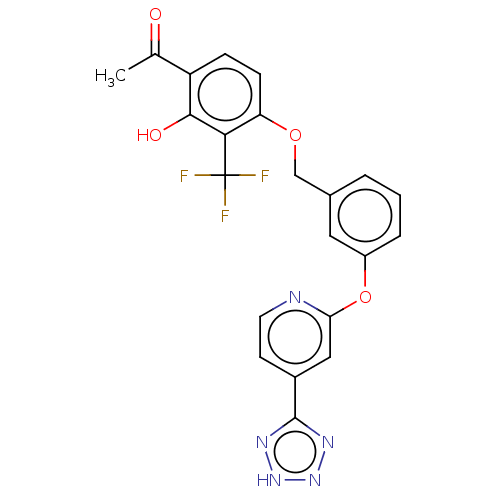
(CHEMBL3965244)Show SMILES CC(=O)c1ccc(OCc2cccc(Oc3cc(ccn3)-c3nn[nH]n3)c2)c(c1O)C(F)(F)F Show InChI InChI=1S/C22H16F3N5O4/c1-12(31)16-5-6-17(19(20(16)32)22(23,24)25)33-11-13-3-2-4-15(9-13)34-18-10-14(7-8-26-18)21-27-29-30-28-21/h2-10,32H,11H2,1H3,(H,27,28,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 112 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207404

(CHEMBL3935529)Show SMILES CCCc1c(OCc2ccc(Oc3ccc(cn3)C(O)=O)cc2)ccc(C(C)=O)c1O Show InChI InChI=1S/C24H23NO6/c1-3-4-20-21(11-10-19(15(2)26)23(20)27)30-14-16-5-8-18(9-6-16)31-22-12-7-17(13-25-22)24(28)29/h5-13,27H,3-4,14H2,1-2H3,(H,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 132 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207411
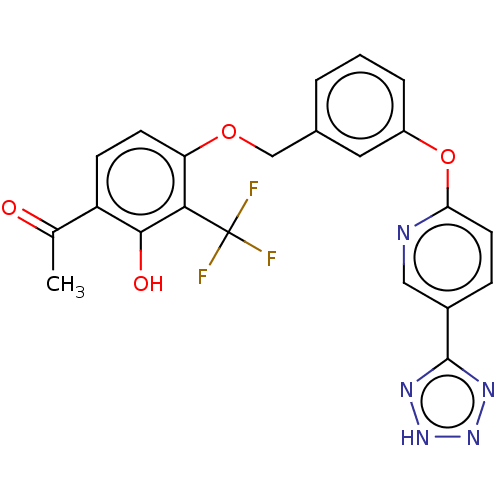
(CHEMBL3973048)Show SMILES CC(=O)c1ccc(OCc2cccc(Oc3ccc(cn3)-c3nn[nH]n3)c2)c(c1O)C(F)(F)F Show InChI InChI=1S/C22H16F3N5O4/c1-12(31)16-6-7-17(19(20(16)32)22(23,24)25)33-11-13-3-2-4-15(9-13)34-18-8-5-14(10-26-18)21-27-29-30-28-21/h2-10,32H,11H2,1H3,(H,27,28,29,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 144 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207399

(CHEMBL3913281)Show SMILES CCCc1c(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)ccc(C(C)=O)c1O Show InChI InChI=1S/C24H23N5O4/c1-3-4-20-21(10-9-19(15(2)30)23(20)31)32-14-16-5-7-18(8-6-16)33-22-13-17(11-12-25-22)24-26-28-29-27-24/h5-13,31H,3-4,14H2,1-2H3,(H,26,27,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207405
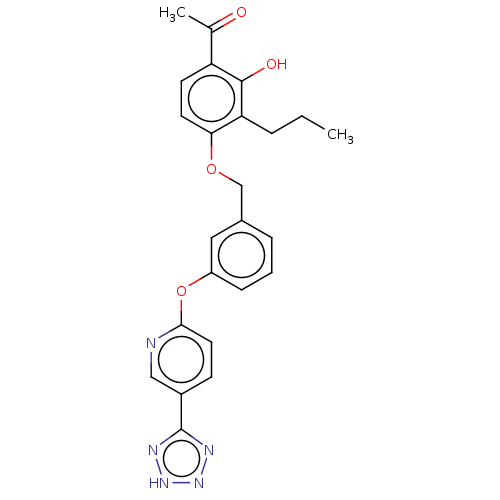
(CHEMBL3918914)Show SMILES CCCc1c(OCc2cccc(Oc3ccc(cn3)-c3nn[nH]n3)c2)ccc(C(C)=O)c1O Show InChI InChI=1S/C24H23N5O4/c1-3-5-20-21(10-9-19(15(2)30)23(20)31)32-14-16-6-4-7-18(12-16)33-22-11-8-17(13-25-22)24-26-28-29-27-24/h4,6-13,31H,3,5,14H2,1-2H3,(H,26,27,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 203 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207413

(CHEMBL3945882)Show SMILES CCCc1c(OCc2ccc(Oc3ncccc3-c3nn[nH]n3)cc2)ccc(C(C)=O)c1O Show InChI InChI=1S/C24H23N5O4/c1-3-5-19-21(12-11-18(15(2)30)22(19)31)32-14-16-7-9-17(10-8-16)33-24-20(6-4-13-25-24)23-26-28-29-27-23/h4,6-13,31H,3,5,14H2,1-2H3,(H,26,27,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 330 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207397
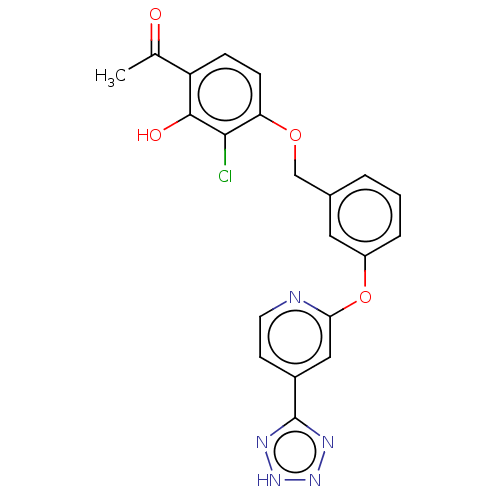
(CHEMBL3927834)Show SMILES CC(=O)c1ccc(OCc2cccc(Oc3cc(ccn3)-c3nn[nH]n3)c2)c(Cl)c1O Show InChI InChI=1S/C21H16ClN5O4/c1-12(28)16-5-6-17(19(22)20(16)29)30-11-13-3-2-4-15(9-13)31-18-10-14(7-8-23-18)21-24-26-27-25-21/h2-10,29H,11H2,1H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 342 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207403
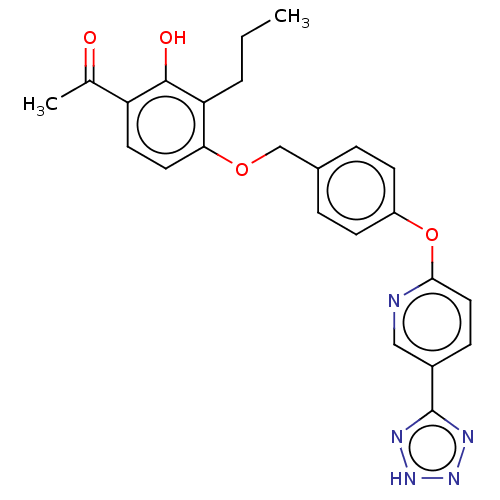
(CHEMBL3911282)Show SMILES CCCc1c(OCc2ccc(Oc3ccc(cn3)-c3nn[nH]n3)cc2)ccc(C(C)=O)c1O Show InChI InChI=1S/C24H23N5O4/c1-3-4-20-21(11-10-19(15(2)30)23(20)31)32-14-16-5-8-18(9-6-16)33-22-12-7-17(13-25-22)24-26-28-29-27-24/h5-13,31H,3-4,14H2,1-2H3,(H,26,27,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 346 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207402
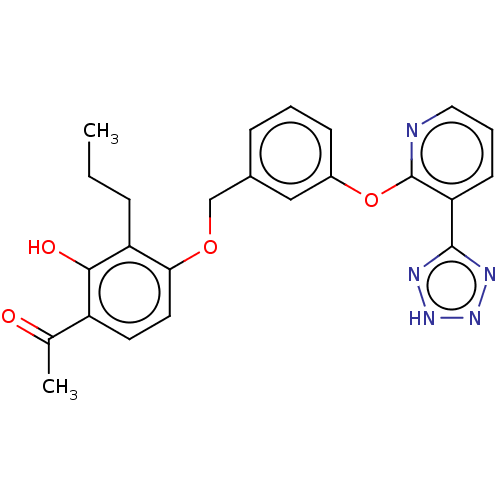
(CHEMBL3980125)Show SMILES CCCc1c(OCc2cccc(Oc3ncccc3-c3nn[nH]n3)c2)ccc(C(C)=O)c1O Show InChI InChI=1S/C24H23N5O4/c1-3-6-19-21(11-10-18(15(2)30)22(19)31)32-14-16-7-4-8-17(13-16)33-24-20(9-5-12-25-24)23-26-28-29-27-23/h4-5,7-13,31H,3,6,14H2,1-2H3,(H,26,27,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 390 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207407
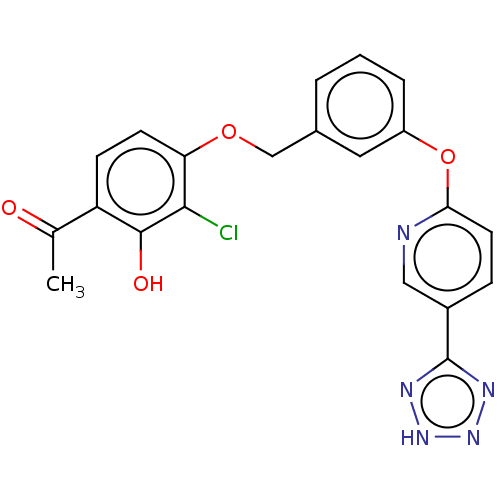
(CHEMBL3936830)Show SMILES CC(=O)c1ccc(OCc2cccc(Oc3ccc(cn3)-c3nn[nH]n3)c2)c(Cl)c1O Show InChI InChI=1S/C21H16ClN5O4/c1-12(28)16-6-7-17(19(22)20(16)29)30-11-13-3-2-4-15(9-13)31-18-8-5-14(10-23-18)21-24-26-27-25-21/h2-10,29H,11H2,1H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 475 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207412

(CHEMBL3920609)Show SMILES CCCc1c(OCc2ccc(Oc3cccc(c3)-c3nn[nH]n3)nc2)ccc(C(C)=O)c1O Show InChI InChI=1S/C24H23N5O4/c1-3-5-20-21(10-9-19(15(2)30)23(20)31)32-14-16-8-11-22(25-13-16)33-18-7-4-6-17(12-18)24-26-28-29-27-24/h4,6-13,31H,3,5,14H2,1-2H3,(H,26,27,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 484 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207409
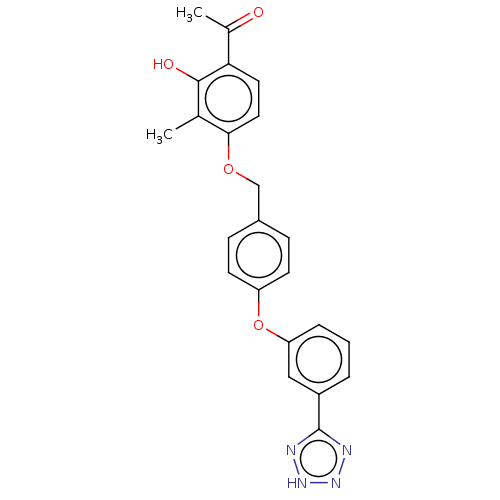
(CHEMBL3892611)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cccc(c3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C23H20N4O4/c1-14-21(11-10-20(15(2)28)22(14)29)30-13-16-6-8-18(9-7-16)31-19-5-3-4-17(12-19)23-24-26-27-25-23/h3-12,29H,13H2,1-2H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.43E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(Homo sapiens (Human)) | BDBM50207408

(CHEMBL3943547)Show SMILES CC(=O)c1ccc(OCc2cccc(Oc3ccc(cn3)-c3nn[nH]n3)c2)c(F)c1O Show InChI InChI=1S/C21H16FN5O4/c1-12(28)16-6-7-17(19(22)20(16)29)30-11-13-3-2-4-15(9-13)31-18-8-5-14(10-23-18)21-24-26-27-25-21/h2-10,29H,11H2,1H3,(H,24,25,26,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at human CysLT1 receptor expressed in Syrian hamster AV12-664 cells assessed as decrease in LTD4-induced intracellular calcium le... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 1
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >8.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human mGlu1 receptor expressed in hamster AV12 cells expressing EAAT1 assessed as inhibition of glutamate-induced ... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 5
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.25E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human mGlu5 receptor expressed in hamster AV12 cells expressing EAAT1 assessed as inhibition of glutamate-induced ... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 3
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.25E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human mGlu3 receptor expressed in hamster AV12 cells expressing EAAT1 assessed as inhibition of glutamate-induced ... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 2
(Homo sapiens (Human)) | BDBM50207400

(CHEMBL3899832)Show SMILES CC(=O)c1ccc(OCc2ccc(Oc3cc(ccn3)-c3nn[nH]n3)cc2)c(C)c1O Show InChI InChI=1S/C22H19N5O4/c1-13-19(8-7-18(14(2)28)21(13)29)30-12-15-3-5-17(6-4-15)31-20-11-16(9-10-23-20)22-24-26-27-25-22/h3-11,29H,12H2,1-2H3,(H,24,25,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.25E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Eli Lilly and Company
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human mGlu2 receptor expressed in hamster AV12 cells expressing EAAT1 assessed as inhibition of glutamate-induced ... |
Bioorg Med Chem Lett 27: 323-328 (2017)
Article DOI: 10.1016/j.bmcl.2016.11.049
BindingDB Entry DOI: 10.7270/Q2DN471R |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM10759
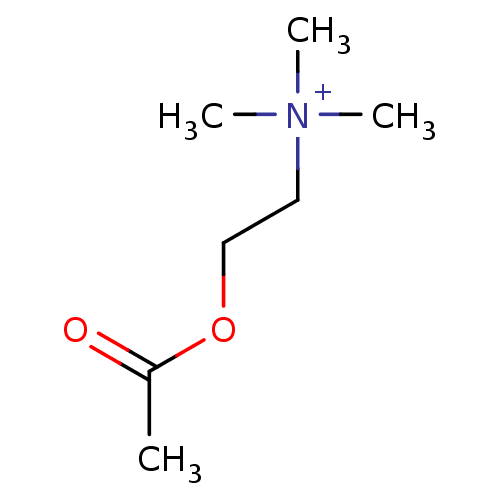
(2-acetoxyethyl(trimethyl)ammonium;bromide | 2-acet...)Show InChI InChI=1S/C7H16NO2/c1-7(9)10-6-5-8(2,3)4/h5-6H2,1-4H3/q+1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 7 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M2 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50038210
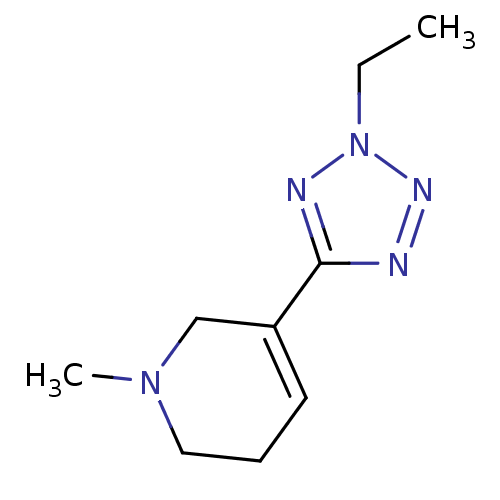
(5-(2-Ethyl-2H-tetrazol-5-yl)-1-methyl-1,2,3,6-tetr...)Show InChI InChI=1S/C9H15N5/c1-3-14-11-9(10-12-14)8-5-4-6-13(2)7-8/h5H,3-4,6-7H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 296 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M2 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50061705

((R)-1-Aza-bicyclo[2.2.2]oct-3-yl-[(Z)-methoxyimino...)Show SMILES CO\N=C(/C#N)[C@H]1CN2CCC1CC2 |wU:6.5,(10.77,-7.56,;9.23,-7.56,;8.14,-8.66,;8.14,-10.2,;9.41,-10.96,;10.69,-11.69,;6.8,-10.98,;6.8,-12.53,;5.46,-13.28,;4.14,-12.53,;4.14,-10.98,;5.46,-10.2,;4.95,-11.51,;5.89,-12.1,)| Show InChI InChI=1S/C10H15N3O/c1-14-12-10(6-11)9-7-13-4-2-8(9)3-5-13/h8-9H,2-5,7H2,1H3/b12-10+/t9-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 62 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M2 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50119648
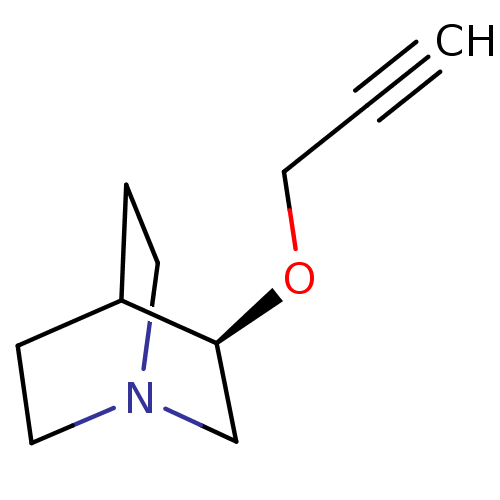
(Talsaclidine)Show SMILES C#CCO[C@H]1CN2CCC1CC2 |wU:4.3,(10.21,-8.05,;8.84,-8.8,;7.52,-9.57,;6.13,-8.8,;4.75,-9.57,;4.75,-11.19,;3.37,-11.99,;2.03,-11.19,;2.03,-9.66,;3.37,-8.88,;3.81,-10.1,;3,-10.66,)| Show InChI InChI=1S/C10H15NO/c1-2-7-12-10-8-11-5-3-9(10)4-6-11/h1,9-10H,3-8H2/t10-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 849 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M2 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50003359

(5-(4-Hexyloxy-[1,2,5]thiadiazol-3-yl)-1-methyl-1,2...)Show InChI InChI=1S/C14H23N3OS/c1-3-4-5-6-10-18-14-13(15-19-16-14)12-8-7-9-17(2)11-12/h8H,3-7,9-11H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 121 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M2 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM10759

(2-acetoxyethyl(trimethyl)ammonium;bromide | 2-acet...)Show InChI InChI=1S/C7H16NO2/c1-7(9)10-6-5-8(2,3)4/h5-6H2,1-4H3/q+1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 205 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M3 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50061705

((R)-1-Aza-bicyclo[2.2.2]oct-3-yl-[(Z)-methoxyimino...)Show SMILES CO\N=C(/C#N)[C@H]1CN2CCC1CC2 |wU:6.5,(10.77,-7.56,;9.23,-7.56,;8.14,-8.66,;8.14,-10.2,;9.41,-10.96,;10.69,-11.69,;6.8,-10.98,;6.8,-12.53,;5.46,-13.28,;4.14,-12.53,;4.14,-10.98,;5.46,-10.2,;4.95,-11.51,;5.89,-12.1,)| Show InChI InChI=1S/C10H15N3O/c1-14-12-10(6-11)9-7-13-4-2-8(9)3-5-13/h8-9H,2-5,7H2,1H3/b12-10+/t9-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 117 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M3 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50119648

(Talsaclidine)Show SMILES C#CCO[C@H]1CN2CCC1CC2 |wU:4.3,(10.21,-8.05,;8.84,-8.8,;7.52,-9.57,;6.13,-8.8,;4.75,-9.57,;4.75,-11.19,;3.37,-11.99,;2.03,-11.19,;2.03,-9.66,;3.37,-8.88,;3.81,-10.1,;3,-10.66,)| Show InChI InChI=1S/C10H15NO/c1-2-7-12-10-8-11-5-3-9(10)4-6-11/h1,9-10H,3-8H2/t10-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 2.70E+3 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M3 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50119623
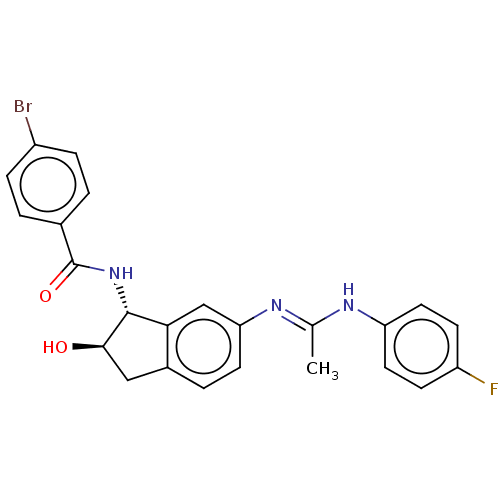
(CHEMBL3618436)Show SMILES C\C(Nc1ccc(F)cc1)=N/c1ccc2C[C@@H](O)[C@H](NC(=O)c3ccc(Br)cc3)c2c1 |r| Show InChI InChI=1S/C24H21BrFN3O2/c1-14(27-19-10-7-18(26)8-11-19)28-20-9-4-16-12-22(30)23(21(16)13-20)29-24(31)15-2-5-17(25)6-3-15/h2-11,13,22-23,30H,12H2,1H3,(H,27,28)(H,29,31)/t22-,23-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.11E+3 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M4 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50119626
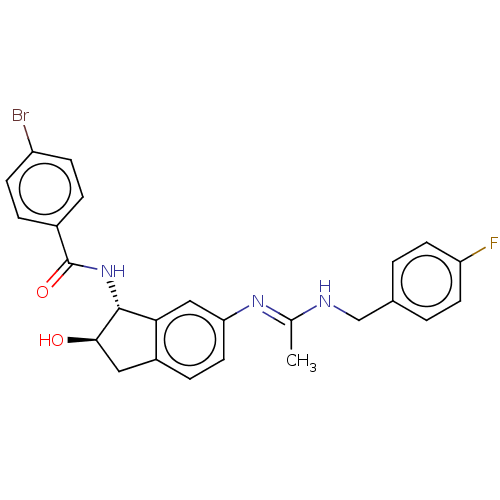
(CHEMBL3618440)Show SMILES C\C(NCc1ccc(F)cc1)=N/c1ccc2C[C@@H](O)[C@H](NC(=O)c3ccc(Br)cc3)c2c1 |r| Show InChI InChI=1S/C25H23BrFN3O2/c1-15(28-14-16-2-9-20(27)10-3-16)29-21-11-6-18-12-23(31)24(22(18)13-21)30-25(32)17-4-7-19(26)8-5-17/h2-11,13,23-24,31H,12,14H2,1H3,(H,28,29)(H,30,32)/t23-,24-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 115 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M4 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50119630
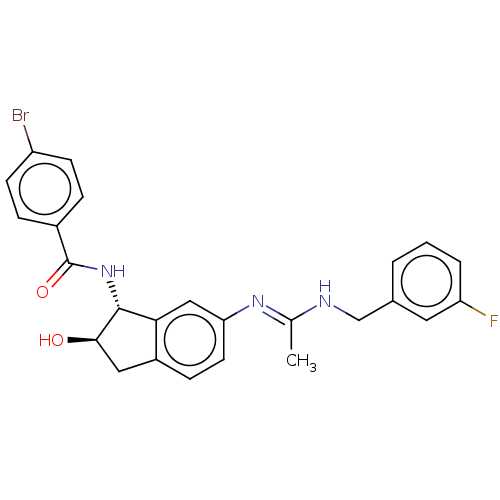
(CHEMBL3618444)Show SMILES C\C(NCc1cccc(F)c1)=N/c1ccc2C[C@@H](O)[C@H](NC(=O)c3ccc(Br)cc3)c2c1 |r| Show InChI InChI=1S/C25H23BrFN3O2/c1-15(28-14-16-3-2-4-20(27)11-16)29-21-10-7-18-12-23(31)24(22(18)13-21)30-25(32)17-5-8-19(26)9-6-17/h2-11,13,23-24,31H,12,14H2,1H3,(H,28,29)(H,30,32)/t23-,24-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 355 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M4 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50119632
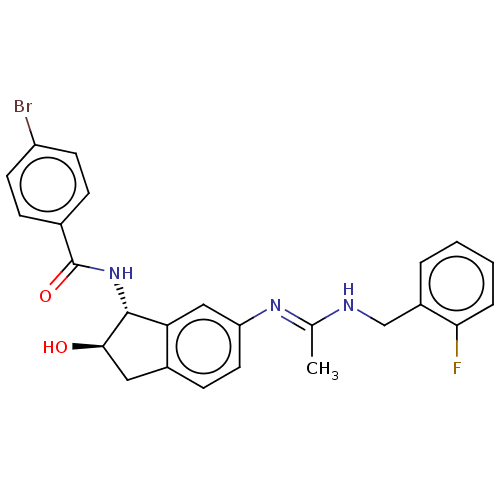
(CHEMBL3618445)Show SMILES C\C(NCc1ccccc1F)=N/c1ccc2C[C@@H](O)[C@H](NC(=O)c3ccc(Br)cc3)c2c1 |r| Show InChI InChI=1S/C25H23BrFN3O2/c1-15(28-14-18-4-2-3-5-22(18)27)29-20-11-8-17-12-23(31)24(21(17)13-20)30-25(32)16-6-9-19(26)10-7-16/h2-11,13,23-24,31H,12,14H2,1H3,(H,28,29)(H,30,32)/t23-,24-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 227 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M4 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50119636
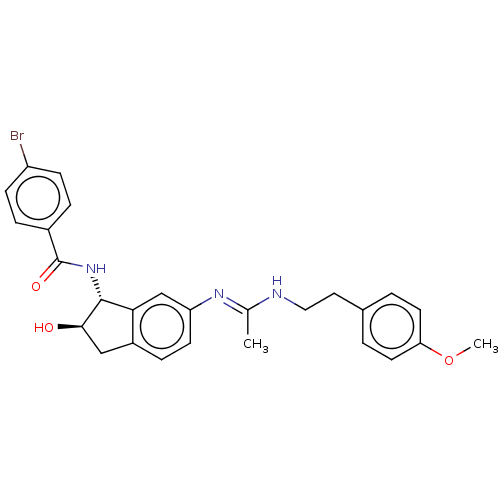
(CHEMBL3616497)Show SMILES COc1ccc(CCN\C(C)=N\c2ccc3C[C@@H](O)[C@H](NC(=O)c4ccc(Br)cc4)c3c2)cc1 |r| Show InChI InChI=1S/C27H28BrN3O3/c1-17(29-14-13-18-3-11-23(34-2)12-4-18)30-22-10-7-20-15-25(32)26(24(20)16-22)31-27(33)19-5-8-21(28)9-6-19/h3-12,16,25-26,32H,13-15H2,1-2H3,(H,29,30)(H,31,33)/t25-,26-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 1.36E+3 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M4 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50119637
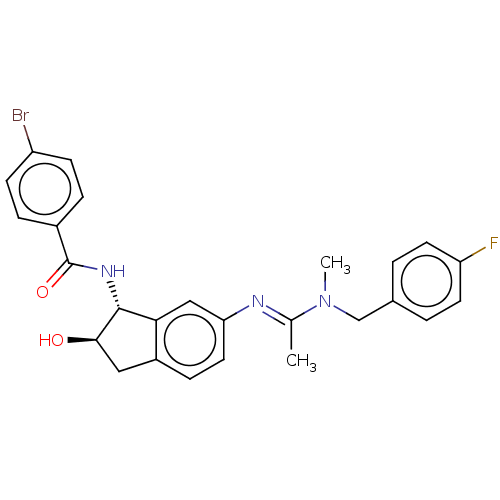
(CHEMBL3618447)Show SMILES CN(Cc1ccc(F)cc1)C(\C)=N\c1ccc2C[C@@H](O)[C@H](NC(=O)c3ccc(Br)cc3)c2c1 |r| Show InChI InChI=1S/C26H25BrFN3O2/c1-16(31(2)15-17-3-10-21(28)11-4-17)29-22-12-7-19-13-24(32)25(23(19)14-22)30-26(33)18-5-8-20(27)9-6-18/h3-12,14,24-25,32H,13,15H2,1-2H3,(H,30,33)/b29-16+/t24-,25-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 3.72E+3 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M4 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50119639
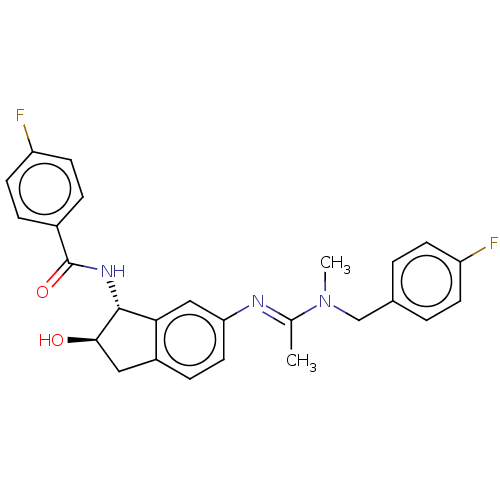
(CHEMBL3618449)Show SMILES CN(Cc1ccc(F)cc1)C(\C)=N\c1ccc2C[C@@H](O)[C@H](NC(=O)c3ccc(F)cc3)c2c1 |r| Show InChI InChI=1S/C26H25F2N3O2/c1-16(31(2)15-17-3-8-20(27)9-4-17)29-22-12-7-19-13-24(32)25(23(19)14-22)30-26(33)18-5-10-21(28)11-6-18/h3-12,14,24-25,32H,13,15H2,1-2H3,(H,30,33)/b29-16+/t24-,25-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 392 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M4 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50119641
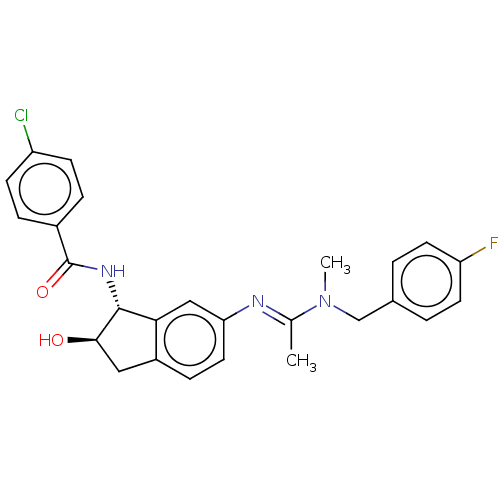
(CHEMBL3618450)Show SMILES CN(Cc1ccc(F)cc1)C(\C)=N\c1ccc2C[C@@H](O)[C@H](NC(=O)c3ccc(Cl)cc3)c2c1 |r| Show InChI InChI=1S/C26H25ClFN3O2/c1-16(31(2)15-17-3-10-21(28)11-4-17)29-22-12-7-19-13-24(32)25(23(19)14-22)30-26(33)18-5-8-20(27)9-6-18/h3-12,14,24-25,32H,13,15H2,1-2H3,(H,30,33)/b29-16+/t24-,25-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 405 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M4 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50119644
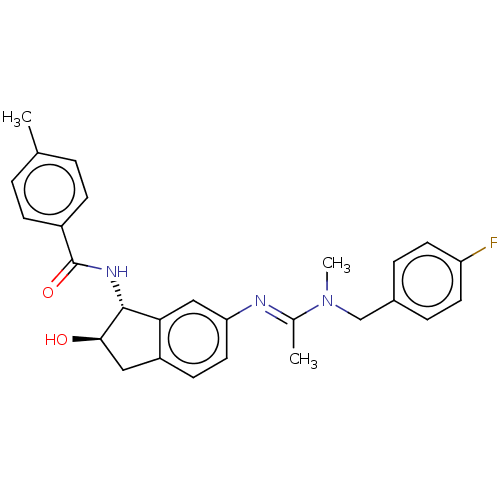
(CHEMBL3616499)Show SMILES CN(Cc1ccc(F)cc1)C(\C)=N\c1ccc2C[C@@H](O)[C@H](NC(=O)c3ccc(C)cc3)c2c1 |r| Show InChI InChI=1S/C27H28FN3O2/c1-17-4-8-20(9-5-17)27(33)30-26-24-15-23(13-10-21(24)14-25(26)32)29-18(2)31(3)16-19-6-11-22(28)12-7-19/h4-13,15,25-26,32H,14,16H2,1-3H3,(H,30,33)/b29-18+/t25-,26-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 235 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M4 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50119645

(CHEMBL3616500)Show SMILES CN(Cc1ccc(F)cc1)C(\C)=N\c1ccc2C[C@@H](O)[C@H](NC(=O)c3ccc(cc3)C(F)(F)F)c2c1 |r| Show InChI InChI=1S/C27H25F4N3O2/c1-16(34(2)15-17-3-10-21(28)11-4-17)32-22-12-7-19-13-24(35)25(23(19)14-22)33-26(36)18-5-8-20(9-6-18)27(29,30)31/h3-12,14,24-25,35H,13,15H2,1-2H3,(H,33,36)/b32-16+/t24-,25-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 393 | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Agonist activity at human muscarinic M4 receptor expressed in CHO cells after 30 mins by GTPgamma35S binding assay |
Bioorg Med Chem Lett 25: 4158-63 (2015)
Article DOI: 10.1016/j.bmcl.2015.08.011
BindingDB Entry DOI: 10.7270/Q2GM8938 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data