 Found 61 hits with Last Name = 'sternberg' and Initial = 'ej'
Found 61 hits with Last Name = 'sternberg' and Initial = 'ej' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403349
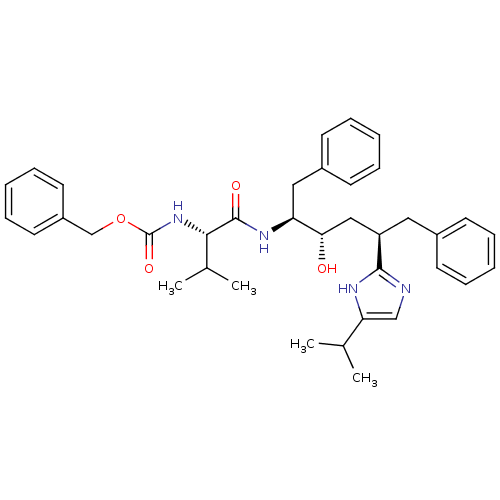
(CHEMBL407551)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccccc1)c1ncc([nH]1)C(C)C Show InChI InChI=1S/C37H46N4O4/c1-25(2)32-23-38-35(39-32)30(20-27-14-8-5-9-15-27)22-33(42)31(21-28-16-10-6-11-17-28)40-36(43)34(26(3)4)41-37(44)45-24-29-18-12-7-13-19-29/h5-19,23,25-26,30-31,33-34,42H,20-22,24H2,1-4H3,(H,38,39)(H,40,43)(H,41,44)/t30-,31+,33+,34+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403357

(CHEMBL419286)Show SMILES CC(C)[C@H](NC(C)=O)C(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccccc1)c1ncc([nH]1)C(C)C Show InChI InChI=1S/C31H42N4O3/c1-20(2)27-19-32-30(34-27)25(16-23-12-8-6-9-13-23)18-28(37)26(17-24-14-10-7-11-15-24)35-31(38)29(21(3)4)33-22(5)36/h6-15,19-21,25-26,28-29,37H,16-18H2,1-5H3,(H,32,34)(H,33,36)(H,35,38)/t25-,26+,28+,29+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403360
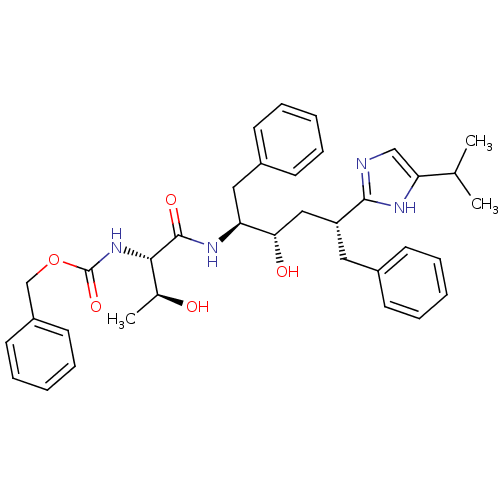
(CHEMBL1790592)Show SMILES CC(C)c1cnc([nH]1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)OCc1ccccc1)[C@H](C)O)Cc1ccccc1 Show InChI InChI=1S/C36H44N4O5/c1-24(2)31-22-37-34(38-31)29(19-26-13-7-4-8-14-26)21-32(42)30(20-27-15-9-5-10-16-27)39-35(43)33(25(3)41)40-36(44)45-23-28-17-11-6-12-18-28/h4-18,22,24-25,29-30,32-33,41-42H,19-21,23H2,1-3H3,(H,37,38)(H,39,43)(H,40,44)/t25-,29+,30-,32-,33-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403351
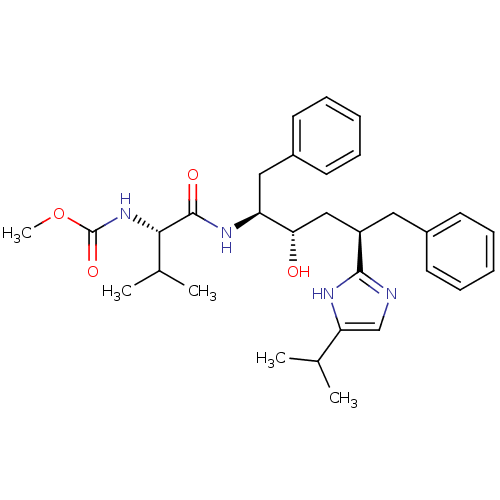
(CHEMBL79698)Show SMILES COC(=O)N[C@@H](C(C)C)C(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccccc1)c1ncc([nH]1)C(C)C Show InChI InChI=1S/C31H42N4O4/c1-20(2)26-19-32-29(33-26)24(16-22-12-8-6-9-13-22)18-27(36)25(17-23-14-10-7-11-15-23)34-30(37)28(21(3)4)35-31(38)39-5/h6-15,19-21,24-25,27-28,36H,16-18H2,1-5H3,(H,32,33)(H,34,37)(H,35,38)/t24-,25+,27+,28+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403358
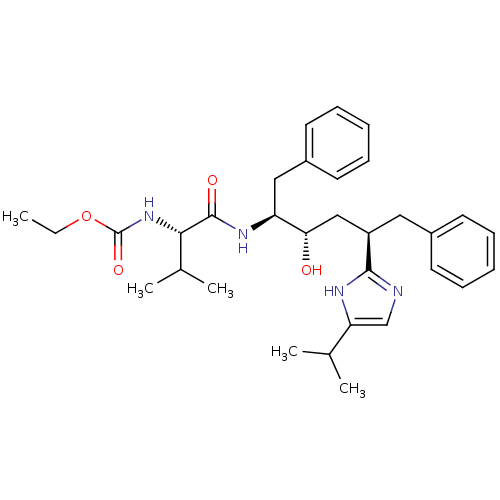
(CHEMBL78531)Show SMILES CCOC(=O)N[C@@H](C(C)C)C(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccccc1)c1ncc([nH]1)C(C)C Show InChI InChI=1S/C32H44N4O4/c1-6-40-32(39)36-29(22(4)5)31(38)35-26(18-24-15-11-8-12-16-24)28(37)19-25(17-23-13-9-7-10-14-23)30-33-20-27(34-30)21(2)3/h7-16,20-22,25-26,28-29,37H,6,17-19H2,1-5H3,(H,33,34)(H,35,38)(H,36,39)/t25-,26+,28+,29+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 2.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403352

(CHEMBL81517)Show SMILES CC(C)[C@H](NC=O)C(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccccc1)c1ncc([nH]1)C(C)C Show InChI InChI=1S/C30H40N4O3/c1-20(2)26-18-31-29(33-26)24(15-22-11-7-5-8-12-22)17-27(36)25(16-23-13-9-6-10-14-23)34-30(37)28(21(3)4)32-19-35/h5-14,18-21,24-25,27-28,36H,15-17H2,1-4H3,(H,31,33)(H,32,35)(H,34,37)/t24-,25+,27+,28+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
| 4.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403348
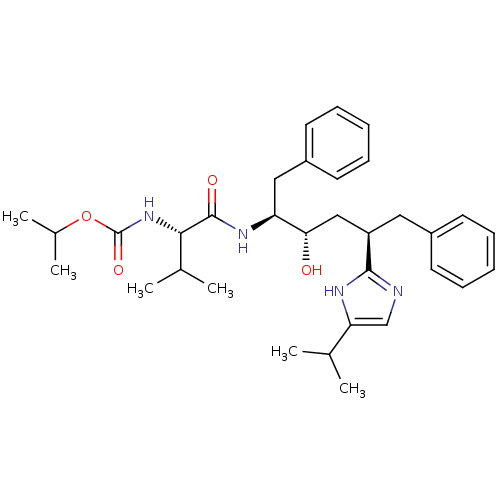
(CHEMBL421709)Show SMILES CC(C)OC(=O)N[C@@H](C(C)C)C(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccccc1)c1ncc([nH]1)C(C)C Show InChI InChI=1S/C33H46N4O4/c1-21(2)28-20-34-31(35-28)26(17-24-13-9-7-10-14-24)19-29(38)27(18-25-15-11-8-12-16-25)36-32(39)30(22(3)4)37-33(40)41-23(5)6/h7-16,20-23,26-27,29-30,38H,17-19H2,1-6H3,(H,34,35)(H,36,39)(H,37,40)/t26-,27+,29+,30+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403345
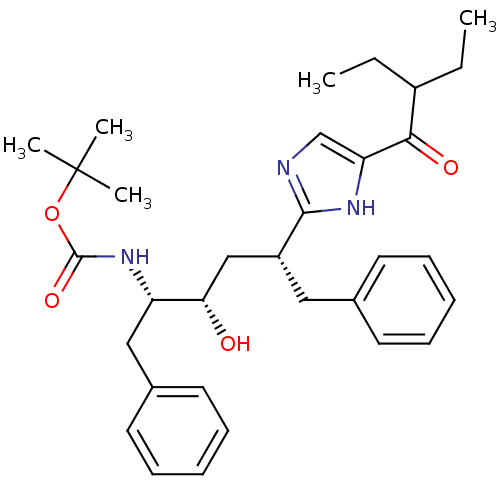
(CHEMBL83739)Show SMILES CCC(CC)C(=O)c1cnc([nH]1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)Cc1ccccc1 Show InChI InChI=1S/C32H43N3O4/c1-6-24(7-2)29(37)27-21-33-30(34-27)25(18-22-14-10-8-11-15-22)20-28(36)26(19-23-16-12-9-13-17-23)35-31(38)39-32(3,4)5/h8-17,21,24-26,28,36H,6-7,18-20H2,1-5H3,(H,33,34)(H,35,38)/t25-,26+,28+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 46 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403350
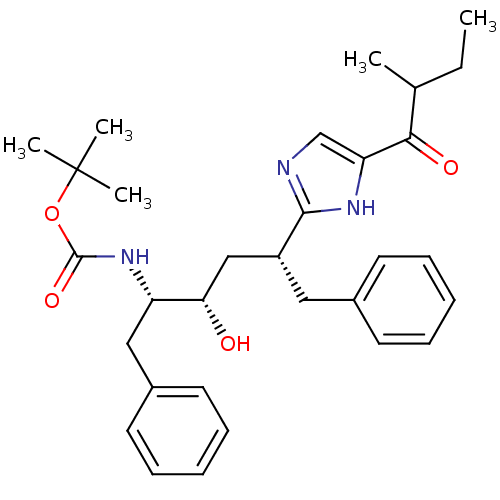
(CHEMBL81190)Show SMILES CCC(C)C(=O)c1cnc([nH]1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)Cc1ccccc1 Show InChI InChI=1S/C31H41N3O4/c1-6-21(2)28(36)26-20-32-29(33-26)24(17-22-13-9-7-10-14-22)19-27(35)25(18-23-15-11-8-12-16-23)34-30(37)38-31(3,4)5/h7-16,20-21,24-25,27,35H,6,17-19H2,1-5H3,(H,32,33)(H,34,37)/t21?,24-,25+,27+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 53 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50037126
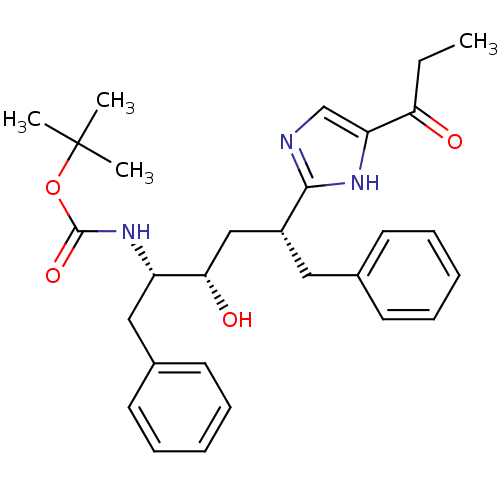
(CHEMBL80098 | [(1S,2S,4R)-1-Benzyl-2-hydroxy-5-phe...)Show SMILES CCC(=O)c1cnc([nH]1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)Cc1ccccc1 Show InChI InChI=1S/C29H37N3O4/c1-5-25(33)24-19-30-27(31-24)22(16-20-12-8-6-9-13-20)18-26(34)23(17-21-14-10-7-11-15-21)32-28(35)36-29(2,3)4/h6-15,19,22-23,26,34H,5,16-18H2,1-4H3,(H,30,31)(H,32,35)/t22-,23+,26+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 92 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403362

(CHEMBL312709)Show SMILES CC(C)(C)OC(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccccc1)c1ncc([nH]1)C(=O)C1CCCC1 Show InChI InChI=1S/C32H41N3O4/c1-32(2,3)39-31(38)35-26(19-23-14-8-5-9-15-23)28(36)20-25(18-22-12-6-4-7-13-22)30-33-21-27(34-30)29(37)24-16-10-11-17-24/h4-9,12-15,21,24-26,28,36H,10-11,16-20H2,1-3H3,(H,33,34)(H,35,38)/t25-,26+,28+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403353

(CHEMBL309773)Show SMILES CC(C)[C@@H](NC(C)=O)C(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccccc1)c1ncc([nH]1)C(C)C Show InChI InChI=1S/C31H42N4O3/c1-20(2)27-19-32-30(34-27)25(16-23-12-8-6-9-13-23)18-28(37)26(17-24-14-10-7-11-15-24)35-31(38)29(21(3)4)33-22(5)36/h6-15,19-21,25-26,28-29,37H,16-18H2,1-5H3,(H,32,34)(H,33,36)(H,35,38)/t25-,26+,28+,29-/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403356

(CHEMBL430437)Show SMILES CC(C)[C@H](NC(=O)OC(C)(C)C)C(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccccc1)c1ncc([nH]1)C(C)C Show InChI InChI=1S/C34H48N4O4/c1-22(2)28-21-35-31(36-28)26(18-24-14-10-8-11-15-24)20-29(39)27(19-25-16-12-9-13-17-25)37-32(40)30(23(3)4)38-33(41)42-34(5,6)7/h8-17,21-23,26-27,29-30,39H,18-20H2,1-7H3,(H,35,36)(H,37,40)(H,38,41)/t26-,27+,29+,30+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403347
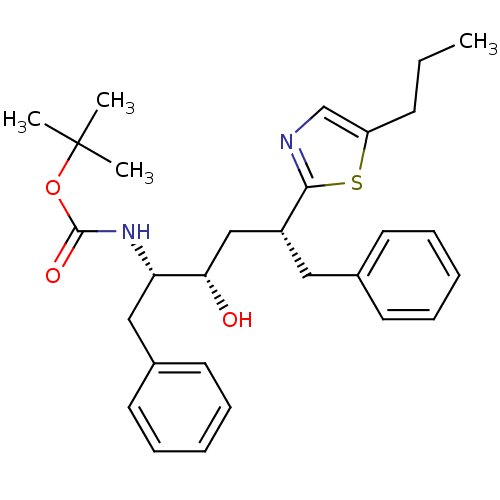
(CHEMBL2115564)Show SMILES CCCc1cnc(s1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)Cc1ccccc1 |r| Show InChI InChI=1S/C29H38N2O3S/c1-5-12-24-20-30-27(35-24)23(17-21-13-8-6-9-14-21)19-26(32)25(18-22-15-10-7-11-16-22)31-28(33)34-29(2,3)4/h6-11,13-16,20,23,25-26,32H,5,12,17-19H2,1-4H3,(H,31,33)/t23-,25+,26+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403355
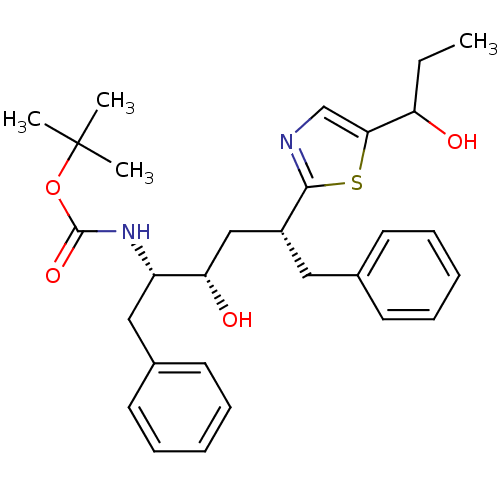
(CHEMBL315930)Show SMILES CCC(O)c1cnc(s1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)Cc1ccccc1 Show InChI InChI=1S/C29H38N2O4S/c1-5-24(32)26-19-30-27(36-26)22(16-20-12-8-6-9-13-20)18-25(33)23(17-21-14-10-7-11-15-21)31-28(34)35-29(2,3)4/h6-15,19,22-25,32-33H,5,16-18H2,1-4H3,(H,31,34)/t22-,23+,24?,25+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 720 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403342
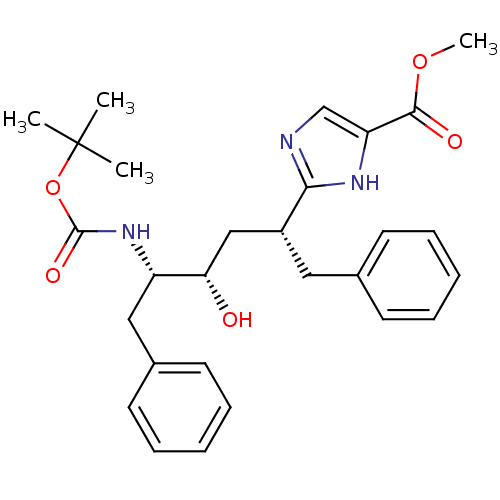
(CHEMBL79719)Show SMILES COC(=O)c1cnc([nH]1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)Cc1ccccc1 Show InChI InChI=1S/C28H35N3O5/c1-28(2,3)36-27(34)31-22(16-20-13-9-6-10-14-20)24(32)17-21(15-19-11-7-5-8-12-19)25-29-18-23(30-25)26(33)35-4/h5-14,18,21-22,24,32H,15-17H2,1-4H3,(H,29,30)(H,31,34)/t21-,22+,24+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403344

(CHEMBL83384)Show SMILES CCc1cnc(s1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)Cc1ccccc1 Show InChI InChI=1S/C28H36N2O3S/c1-5-23-19-29-26(34-23)22(16-20-12-8-6-9-13-20)18-25(31)24(17-21-14-10-7-11-15-21)30-27(32)33-28(2,3)4/h6-15,19,22,24-25,31H,5,16-18H2,1-4H3,(H,30,32)/t22-,24+,25+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403359

(CHEMBL312136)Show SMILES CC(C)(C)OC(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccccc1)c1nccs1 Show InChI InChI=1S/C26H32N2O3S/c1-26(2,3)31-25(30)28-22(17-20-12-8-5-9-13-20)23(29)18-21(24-27-14-15-32-24)16-19-10-6-4-7-11-19/h4-15,21-23,29H,16-18H2,1-3H3,(H,28,30)/t21-,22+,23+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403343
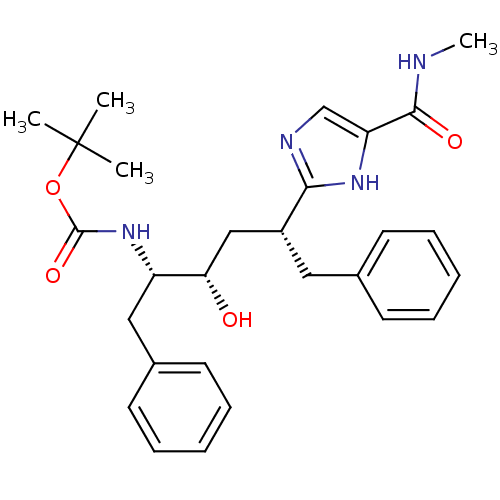
(CHEMBL309195)Show SMILES CNC(=O)c1cnc([nH]1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)Cc1ccccc1 Show InChI InChI=1S/C28H36N4O4/c1-28(2,3)36-27(35)32-22(16-20-13-9-6-10-14-20)24(33)17-21(15-19-11-7-5-8-12-19)25-30-18-23(31-25)26(34)29-4/h5-14,18,21-22,24,33H,15-17H2,1-4H3,(H,29,34)(H,30,31)(H,32,35)/t21-,22+,24+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 3.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403346
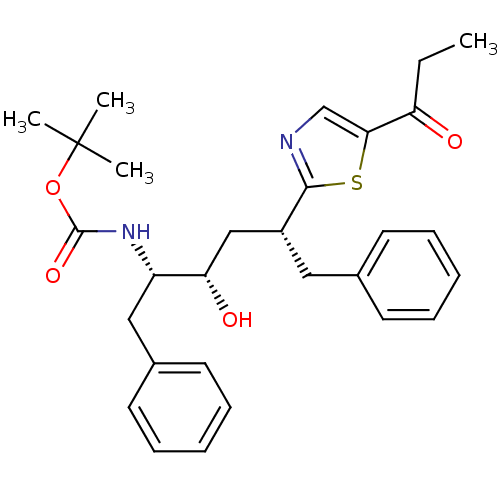
(CHEMBL81775)Show SMILES CCC(=O)c1cnc(s1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)Cc1ccccc1 Show InChI InChI=1S/C29H36N2O4S/c1-5-24(32)26-19-30-27(36-26)22(16-20-12-8-6-9-13-20)18-25(33)23(17-21-14-10-7-11-15-21)31-28(34)35-29(2,3)4/h6-15,19,22-23,25,33H,5,16-18H2,1-4H3,(H,31,34)/t22-,23+,25+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 3.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403347

(CHEMBL2115564)Show SMILES CCCc1cnc(s1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)Cc1ccccc1 |r| Show InChI InChI=1S/C29H38N2O3S/c1-5-12-24-20-30-27(35-24)23(17-21-13-8-6-9-14-21)19-26(32)25(18-22-15-10-7-11-16-22)31-28(33)34-29(2,3)4/h6-11,13-16,20,23,25-26,32H,5,12,17-19H2,1-4H3,(H,31,33)/t23-,25+,26+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 4.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403347

(CHEMBL2115564)Show SMILES CCCc1cnc(s1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)Cc1ccccc1 |r| Show InChI InChI=1S/C29H38N2O3S/c1-5-12-24-20-30-27(35-24)23(17-21-13-8-6-9-14-21)19-26(32)25(18-22-15-10-7-11-16-22)31-28(33)34-29(2,3)4/h6-11,13-16,20,23,25-26,32H,5,12,17-19H2,1-4H3,(H,31,33)/t23-,25+,26+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 4.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403354
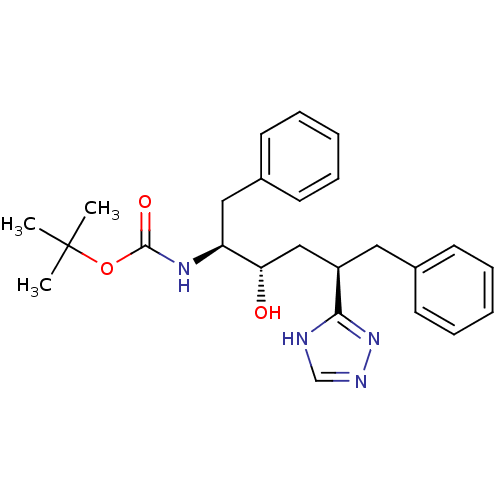
(CHEMBL79570)Show SMILES CC(C)(C)OC(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccccc1)c1nnc[nH]1 Show InChI InChI=1S/C25H32N4O3/c1-25(2,3)32-24(31)28-21(15-19-12-8-5-9-13-19)22(30)16-20(23-26-17-27-29-23)14-18-10-6-4-7-11-18/h4-13,17,20-22,30H,14-16H2,1-3H3,(H,28,31)(H,26,27,29)/t20-,21+,22+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
| 5.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403361

(CHEMBL432179)Show SMILES CC(C)(C)OC(=O)N[C@@H](Cc1ccccc1)[C@@H](O)C[C@@H](Cc1ccccc1)c1ncc([nH]1)C(=O)c1ccccc1 Show InChI InChI=1S/C33H37N3O4/c1-33(2,3)40-32(39)36-27(20-24-15-9-5-10-16-24)29(37)21-26(19-23-13-7-4-8-14-23)31-34-22-28(35-31)30(38)25-17-11-6-12-18-25/h4-18,22,26-27,29,37H,19-21H2,1-3H3,(H,34,35)(H,36,39)/t26-,27+,29+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 7.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50403363

(CHEMBL78534)Show SMILES CCCCc1cnc(s1)[C@@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)Cc1ccccc1 Show InChI InChI=1S/C30H40N2O3S/c1-5-6-17-25-21-31-28(36-25)24(18-22-13-9-7-10-14-22)20-27(33)26(19-23-15-11-8-12-16-23)32-29(34)35-30(2,3)4/h7-16,21,24,26-27,33H,5-6,17-20H2,1-4H3,(H,32,34)/t24-,26+,27+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 2.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against HIV-1 protease |
Bioorg Med Chem Lett 4: 2441-2446 (1994)
Article DOI: 10.1016/S0960-894X(01)80406-6
BindingDB Entry DOI: 10.7270/Q2X63P48 |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human cytomegalovirus (strain AD169) (HHV-5) (Huma...) | BDBM50075178
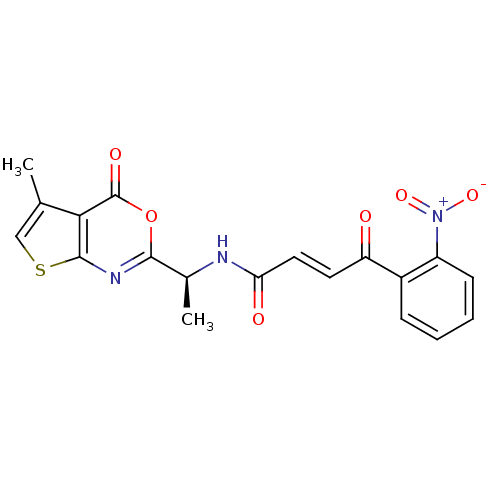
((E)-4-(2-Nitro-phenyl)-4-oxo-but-2-enoic acid [(S)...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1ccccc1[N+]([O-])=O)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C19H15N3O6S/c1-10-9-29-18-16(10)19(25)28-17(21-18)11(2)20-15(24)8-7-14(23)12-5-3-4-6-13(12)22(26)27/h3-9,11H,1-2H3,(H,20,24)/b8-7+/t11-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against cytomegalovirus protease (CMV) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human cytomegalovirus (strain AD169) (HHV-5) (Huma...) | BDBM50075173
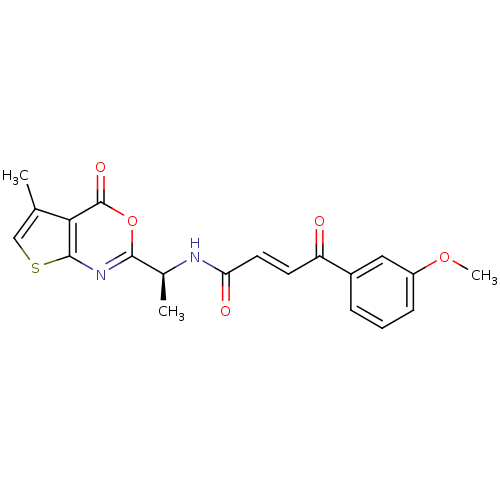
((E)-4-(3-Methoxy-phenyl)-4-oxo-but-2-enoic acid [(...)Show SMILES COc1cccc(c1)C(=O)\C=C\C(=O)N[C@@H](C)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C20H18N2O5S/c1-11-10-28-19-17(11)20(25)27-18(22-19)12(2)21-16(24)8-7-15(23)13-5-4-6-14(9-13)26-3/h4-10,12H,1-3H3,(H,21,24)/b8-7+/t12-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against cytomegalovirus protease (CMV) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human cytomegalovirus (strain AD169) (HHV-5) (Huma...) | BDBM50075171
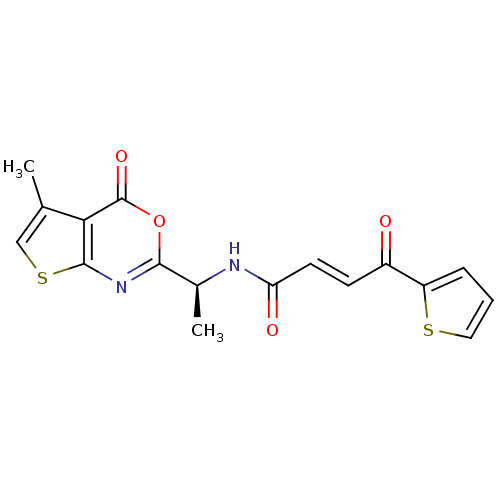
((E)-4-Oxo-4-thiophen-2-yl-but-2-enoic acid [(S)-1-...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1cccs1)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C17H14N2O4S2/c1-9-8-25-16-14(9)17(22)23-15(19-16)10(2)18-13(21)6-5-11(20)12-4-3-7-24-12/h3-8,10H,1-2H3,(H,18,21)/b6-5+/t10-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against cytomegalovirus protease (CMV) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human cytomegalovirus (strain AD169) (HHV-5) (Huma...) | BDBM50075176
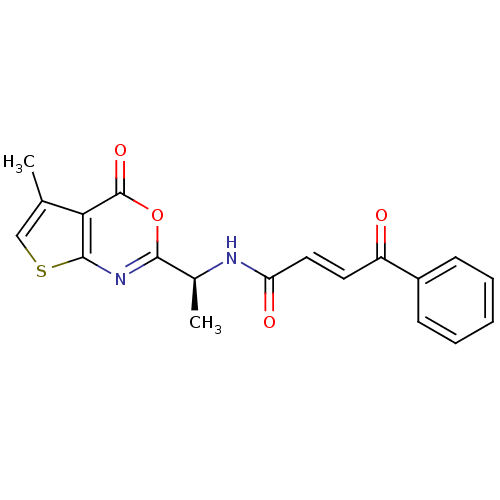
((E)-4-Oxo-4-phenyl-but-2-enoic acid [(S)-1-(5-meth...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1ccccc1)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C19H16N2O4S/c1-11-10-26-18-16(11)19(24)25-17(21-18)12(2)20-15(23)9-8-14(22)13-6-4-3-5-7-13/h3-10,12H,1-2H3,(H,20,23)/b9-8+/t12-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against cytomegalovirus protease (CMV) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human cytomegalovirus (strain AD169) (HHV-5) (Huma...) | BDBM50075175
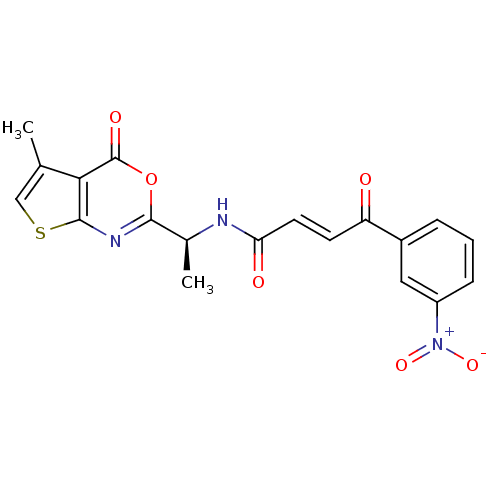
((E)-4-(3-Nitro-phenyl)-4-oxo-but-2-enoic acid [(S)...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1cccc(c1)[N+]([O-])=O)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C19H15N3O6S/c1-10-9-29-18-16(10)19(25)28-17(21-18)11(2)20-15(24)7-6-14(23)12-4-3-5-13(8-12)22(26)27/h3-9,11H,1-2H3,(H,20,24)/b7-6+/t11-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against cytomegalovirus protease (CMV) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human cytomegalovirus (strain AD169) (HHV-5) (Huma...) | BDBM50075177
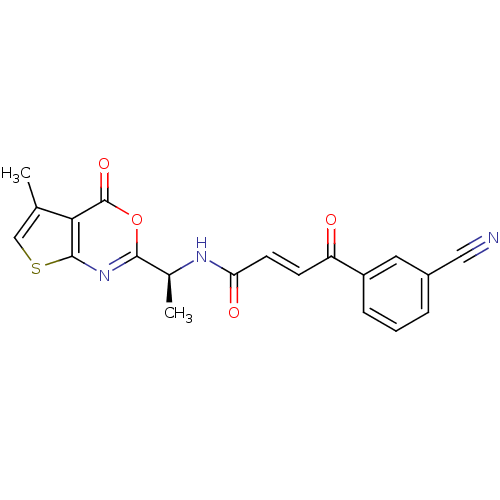
((E)-4-(3-Cyano-phenyl)-4-oxo-but-2-enoic acid [(S)...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1cccc(c1)C#N)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C20H15N3O4S/c1-11-10-28-19-17(11)20(26)27-18(23-19)12(2)22-16(25)7-6-15(24)14-5-3-4-13(8-14)9-21/h3-8,10,12H,1-2H3,(H,22,25)/b7-6+/t12-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against cytomegalovirus protease (CMV) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human cytomegalovirus (strain AD169) (HHV-5) (Huma...) | BDBM50075180
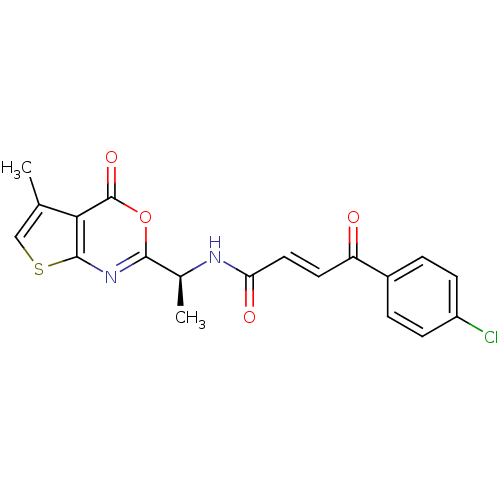
((E)-4-(4-Chloro-phenyl)-4-oxo-but-2-enoic acid [(S...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1ccc(Cl)cc1)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C19H15ClN2O4S/c1-10-9-27-18-16(10)19(25)26-17(22-18)11(2)21-15(24)8-7-14(23)12-3-5-13(20)6-4-12/h3-9,11H,1-2H3,(H,21,24)/b8-7+/t11-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against cytomegalovirus protease (CMV) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human cytomegalovirus (strain AD169) (HHV-5) (Huma...) | BDBM50075170
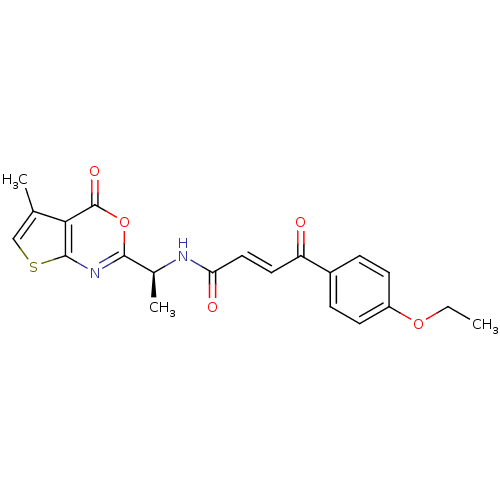
((E)-4-(4-Ethoxy-phenyl)-4-oxo-but-2-enoic acid [(S...)Show SMILES CCOc1ccc(cc1)C(=O)\C=C\C(=O)N[C@@H](C)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C21H20N2O5S/c1-4-27-15-7-5-14(6-8-15)16(24)9-10-17(25)22-13(3)19-23-20-18(21(26)28-19)12(2)11-29-20/h5-11,13H,4H2,1-3H3,(H,22,25)/b10-9+/t13-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against cytomegalovirus protease (CMV) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50075173

((E)-4-(3-Methoxy-phenyl)-4-oxo-but-2-enoic acid [(...)Show SMILES COc1cccc(c1)C(=O)\C=C\C(=O)N[C@@H](C)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C20H18N2O5S/c1-11-10-28-19-17(11)20(25)27-18(22-19)12(2)21-16(24)8-7-15(23)13-5-4-6-14(9-13)26-3/h4-10,12H,1-3H3,(H,21,24)/b8-7+/t12-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against varicella zoster virus (VZV) protease |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human cytomegalovirus (strain AD169) (HHV-5) (Huma...) | BDBM50075179
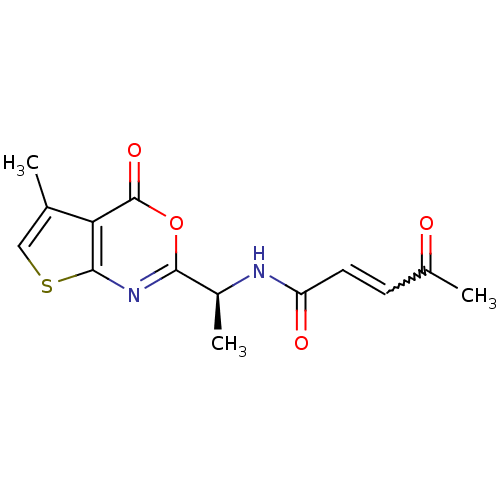
((E)-4-Oxo-pent-2-enoic acid [(S)-1-(5-methyl-4-oxo...)Show SMILES C[C@H](NC(=O)C=CC(C)=O)c1nc2scc(C)c2c(=O)o1 |w:6.6| Show InChI InChI=1S/C14H14N2O4S/c1-7-6-21-13-11(7)14(19)20-12(16-13)9(3)15-10(18)5-4-8(2)17/h4-6,9H,1-3H3,(H,15,18)/t9-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 370 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against cytomegalovirus protease (CMV) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50075177

((E)-4-(3-Cyano-phenyl)-4-oxo-but-2-enoic acid [(S)...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1cccc(c1)C#N)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C20H15N3O4S/c1-11-10-28-19-17(11)20(26)27-18(23-19)12(2)22-16(25)7-6-15(24)14-5-3-4-13(8-14)9-21/h3-8,10,12H,1-2H3,(H,22,25)/b7-6+/t12-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 410 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against varicella zoster virus (VZV) protease |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50075175

((E)-4-(3-Nitro-phenyl)-4-oxo-but-2-enoic acid [(S)...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1cccc(c1)[N+]([O-])=O)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C19H15N3O6S/c1-10-9-29-18-16(10)19(25)28-17(21-18)11(2)20-15(24)7-6-14(23)12-4-3-5-13(8-12)22(26)27/h3-9,11H,1-2H3,(H,20,24)/b7-6+/t11-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 480 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against varicella zoster virus (VZV) protease |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50075171

((E)-4-Oxo-4-thiophen-2-yl-but-2-enoic acid [(S)-1-...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1cccs1)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C17H14N2O4S2/c1-9-8-25-16-14(9)17(22)23-15(19-16)10(2)18-13(21)6-5-11(20)12-4-3-7-24-12/h3-8,10H,1-2H3,(H,18,21)/b6-5+/t10-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 650 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against varicella zoster virus (VZV) protease |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human herpesvirus 2 (strain HG52) (HHV-2) (Human h...) | BDBM50075173

((E)-4-(3-Methoxy-phenyl)-4-oxo-but-2-enoic acid [(...)Show SMILES COc1cccc(c1)C(=O)\C=C\C(=O)N[C@@H](C)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C20H18N2O5S/c1-11-10-28-19-17(11)20(25)27-18(22-19)12(2)21-16(24)8-7-15(23)13-5-4-6-14(9-13)26-3/h4-10,12H,1-3H3,(H,21,24)/b8-7+/t12-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 680 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against herpes simplex virus protease (HSV-2) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human herpesvirus 2 (strain HG52) (HHV-2) (Human h...) | BDBM50075179

((E)-4-Oxo-pent-2-enoic acid [(S)-1-(5-methyl-4-oxo...)Show SMILES C[C@H](NC(=O)C=CC(C)=O)c1nc2scc(C)c2c(=O)o1 |w:6.6| Show InChI InChI=1S/C14H14N2O4S/c1-7-6-21-13-11(7)14(19)20-12(16-13)9(3)15-10(18)5-4-8(2)17/h4-6,9H,1-3H3,(H,15,18)/t9-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 790 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against herpes simplex virus protease (HSV-2) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50075172
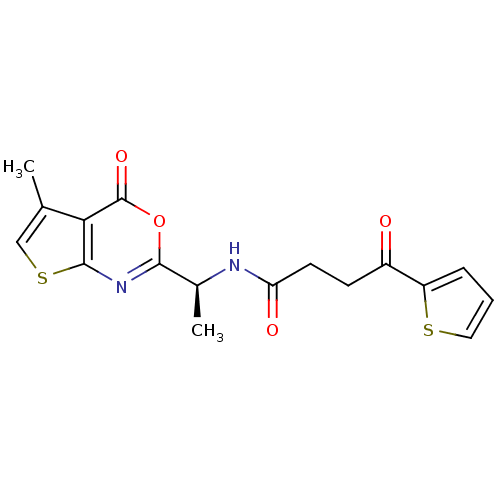
(CHEMBL138045 | N-[(S)-1-(5-Methyl-4-oxo-4H-thieno[...)Show SMILES C[C@H](NC(=O)CCC(=O)c1cccs1)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C17H16N2O4S2/c1-9-8-25-16-14(9)17(22)23-15(19-16)10(2)18-13(21)6-5-11(20)12-4-3-7-24-12/h3-4,7-8,10H,5-6H2,1-2H3,(H,18,21)/t10-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 820 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against varicella zoster virus (VZV) protease |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50075178

((E)-4-(2-Nitro-phenyl)-4-oxo-but-2-enoic acid [(S)...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1ccccc1[N+]([O-])=O)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C19H15N3O6S/c1-10-9-29-18-16(10)19(25)28-17(21-18)11(2)20-15(24)8-7-14(23)12-5-3-4-6-13(12)22(26)27/h3-9,11H,1-2H3,(H,20,24)/b8-7+/t11-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 840 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against varicella zoster virus (VZV) protease |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50075176

((E)-4-Oxo-4-phenyl-but-2-enoic acid [(S)-1-(5-meth...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1ccccc1)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C19H16N2O4S/c1-11-10-26-18-16(11)19(24)25-17(21-18)12(2)20-15(23)9-8-14(22)13-6-4-3-5-7-13/h3-10,12H,1-2H3,(H,20,23)/b9-8+/t12-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against varicella zoster virus (VZV) protease |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50075181
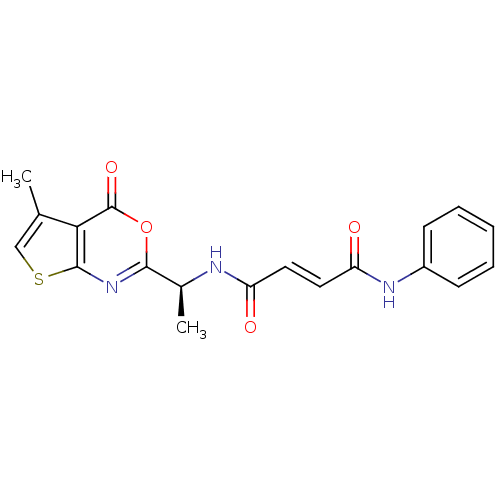
((E)-But-2-enedioic acid [(S)-1-(5-methyl-4-oxo-4H-...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)Nc1ccccc1)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C19H17N3O4S/c1-11-10-27-18-16(11)19(25)26-17(22-18)12(2)20-14(23)8-9-15(24)21-13-6-4-3-5-7-13/h3-10,12H,1-2H3,(H,20,23)(H,21,24)/b9-8+/t12-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against varicella zoster virus (VZV) protease |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50075179

((E)-4-Oxo-pent-2-enoic acid [(S)-1-(5-methyl-4-oxo...)Show SMILES C[C@H](NC(=O)C=CC(C)=O)c1nc2scc(C)c2c(=O)o1 |w:6.6| Show InChI InChI=1S/C14H14N2O4S/c1-7-6-21-13-11(7)14(19)20-12(16-13)9(3)15-10(18)5-4-8(2)17/h4-6,9H,1-3H3,(H,15,18)/t9-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.36E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against varicella zoster virus (VZV) protease |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Human rhinovirus B) | BDBM50075170

((E)-4-(4-Ethoxy-phenyl)-4-oxo-but-2-enoic acid [(S...)Show SMILES CCOc1ccc(cc1)C(=O)\C=C\C(=O)N[C@@H](C)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C21H20N2O5S/c1-4-27-15-7-5-14(6-8-15)16(24)9-10-17(25)22-13(3)19-23-20-18(21(26)28-19)12(2)11-29-20/h5-11,13H,4H2,1-3H3,(H,22,25)/b10-9+/t13-/m0/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against varicella zoster virus (VZV) protease |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human herpesvirus 2 (strain HG52) (HHV-2) (Human h...) | BDBM50075177

((E)-4-(3-Cyano-phenyl)-4-oxo-but-2-enoic acid [(S)...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1cccc(c1)C#N)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C20H15N3O4S/c1-11-10-28-19-17(11)20(26)27-18(23-19)12(2)22-16(25)7-6-15(24)14-5-3-4-13(8-14)9-21/h3-8,10,12H,1-2H3,(H,22,25)/b7-6+/t12-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against herpes simplex virus protease (HSV-2) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human herpesvirus 2 (strain HG52) (HHV-2) (Human h...) | BDBM50075178

((E)-4-(2-Nitro-phenyl)-4-oxo-but-2-enoic acid [(S)...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)c1ccccc1[N+]([O-])=O)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C19H15N3O6S/c1-10-9-29-18-16(10)19(25)28-17(21-18)11(2)20-15(24)8-7-14(23)12-5-3-4-6-13(12)22(26)27/h3-9,11H,1-2H3,(H,20,24)/b8-7+/t11-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against herpes simplex virus protease (HSV-2) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human cytomegalovirus (strain AD169) (HHV-5) (Huma...) | BDBM50075181

((E)-But-2-enedioic acid [(S)-1-(5-methyl-4-oxo-4H-...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)Nc1ccccc1)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C19H17N3O4S/c1-11-10-27-18-16(11)19(25)26-17(22-18)12(2)20-14(23)8-9-15(24)21-13-6-4-3-5-7-13/h3-10,12H,1-2H3,(H,20,23)(H,21,24)/b9-8+/t12-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against cytomegalovirus protease (CMV) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |
Capsid scaffolding protein
(Human herpesvirus 2 (strain HG52) (HHV-2) (Human h...) | BDBM50075181

((E)-But-2-enedioic acid [(S)-1-(5-methyl-4-oxo-4H-...)Show SMILES C[C@H](NC(=O)\C=C\C(=O)Nc1ccccc1)c1nc2scc(C)c2c(=O)o1 Show InChI InChI=1S/C19H17N3O4S/c1-11-10-27-18-16(11)19(25)26-17(22-18)12(2)20-14(23)8-9-15(24)21-13-6-4-3-5-7-13/h3-10,12H,1-2H3,(H,20,23)(H,21,24)/b9-8+/t12-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibitory activity against herpes simplex virus protease (HSV-2) |
Bioorg Med Chem Lett 9: 449-52 (1999)
BindingDB Entry DOI: 10.7270/Q2TM799P |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data