 Found 166 hits with Last Name = 'varga' and Initial = 'e'
Found 166 hits with Last Name = 'varga' and Initial = 'e' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070376
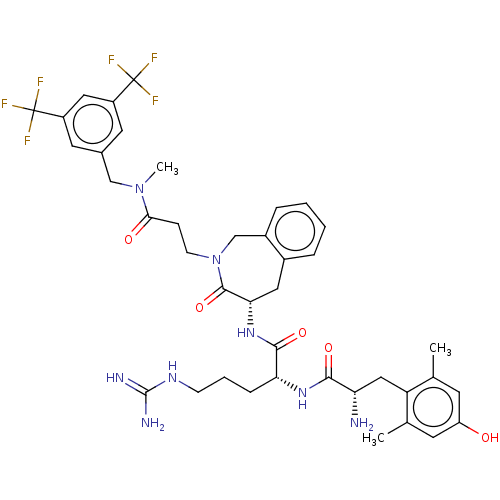
(CHEMBL3408518)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(=O)N(C)Cc2cc(cc(c2)C(F)(F)F)C(F)(F)F)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C40H48F6N8O5/c1-22-13-29(55)14-23(2)30(22)19-31(47)35(57)51-32(9-6-11-50-38(48)49)36(58)52-33-17-25-7-4-5-8-26(25)21-54(37(33)59)12-10-34(56)53(3)20-24-15-27(39(41,42)43)18-28(16-24)40(44,45)46/h4-5,7-8,13-16,18,31-33,55H,6,9-12,17,19-21,47H2,1-3H3,(H,51,57)(H,52,58)(H4,48,49,50)/t31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070377
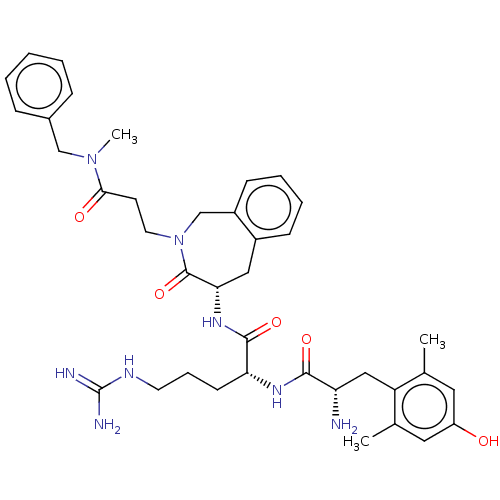
(CHEMBL3408519)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(=O)N(C)Cc2ccccc2)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C38H50N8O5/c1-24-18-29(47)19-25(2)30(24)21-31(39)35(49)43-32(14-9-16-42-38(40)41)36(50)44-33-20-27-12-7-8-13-28(27)23-46(37(33)51)17-15-34(48)45(3)22-26-10-5-4-6-11-26/h4-8,10-13,18-19,31-33,47H,9,14-17,20-23,39H2,1-3H3,(H,43,49)(H,44,50)(H4,40,41,42)/t31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070380
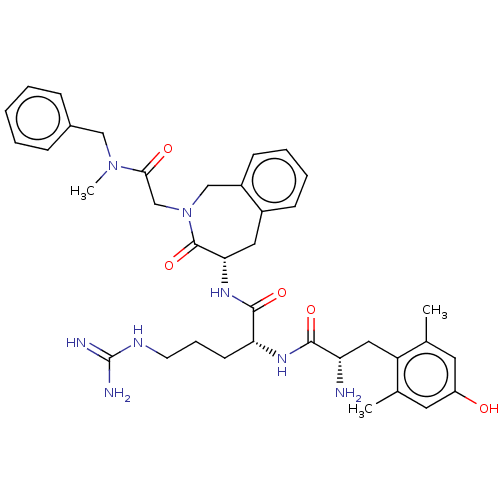
(CHEMBL3408522)Show SMILES [H][C@@]1(Cc2ccccc2CN(CC(=O)N(C)Cc2ccccc2)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C37H48N8O5/c1-23-16-28(46)17-24(2)29(23)19-30(38)34(48)42-31(14-9-15-41-37(39)40)35(49)43-32-18-26-12-7-8-13-27(26)21-45(36(32)50)22-33(47)44(3)20-25-10-5-4-6-11-25/h4-8,10-13,16-17,30-32,46H,9,14-15,18-22,38H2,1-3H3,(H,42,48)(H,43,49)(H4,39,40,41)/t30-,31+,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM81931

(CAM | CAS_56337-94-1 | N-cyclopropylmethylnorazido...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CC[C@@H](N=[N+]=[N-])[C@H]5Oc1c2[C@]45CCN3CC1CC1 Show InChI InChI=1S/C20H24N4O2/c21-23-22-14-5-4-13-15-9-12-3-6-16(25)18-17(12)20(13,19(14)26-18)7-8-24(15)10-11-1-2-11/h3,6,11,13-15,19,25H,1-2,4-5,7-10H2/t13-,14+,15+,19+,20-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
kappa-type opioid receptor isoform 1
(Frog) | BDBM81931

(CAM | CAS_56337-94-1 | N-cyclopropylmethylnorazido...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CC[C@@H](N=[N+]=[N-])[C@H]5Oc1c2[C@]45CCN3CC1CC1 Show InChI InChI=1S/C20H24N4O2/c21-23-22-14-5-4-13-15-9-12-3-6-16(25)18-17(12)20(13,19(14)26-18)7-8-24(15)10-11-1-2-11/h3,6,11,13-15,19,25H,1-2,4-5,7-10H2/t13-,14+,15+,19+,20-/m0/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070385
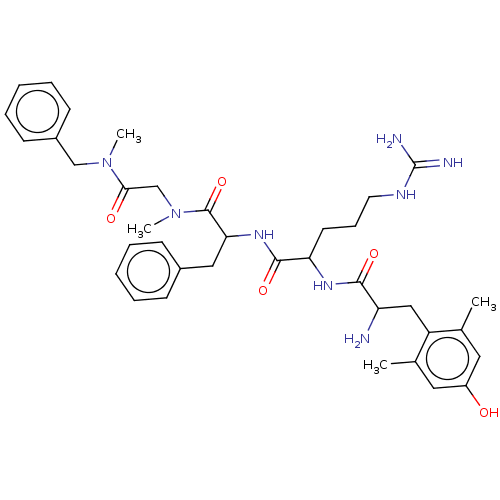
(CHEMBL3408736)Show SMILES CN(CC(=O)N(C)Cc1ccccc1)C(=O)C(Cc1ccccc1)NC(=O)C(CCCNC(N)=N)NC(=O)C(N)Cc1c(C)cc(O)cc1C Show InChI InChI=1S/C37H50N8O5/c1-24-18-28(46)19-25(2)29(24)21-30(38)34(48)42-31(16-11-17-41-37(39)40)35(49)43-32(20-26-12-7-5-8-13-26)36(50)45(4)23-33(47)44(3)22-27-14-9-6-10-15-27/h5-10,12-15,18-19,30-32,46H,11,16-17,20-23,38H2,1-4H3,(H,42,48)(H,43,49)(H4,39,40,41) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
kappa-type opioid receptor isoform 1
(Frog) | BDBM81931

(CAM | CAS_56337-94-1 | N-cyclopropylmethylnorazido...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CC[C@@H](N=[N+]=[N-])[C@H]5Oc1c2[C@]45CCN3CC1CC1 Show InChI InChI=1S/C20H24N4O2/c21-23-22-14-5-4-13-15-9-12-3-6-16(25)18-17(12)20(13,19(14)26-18)7-8-24(15)10-11-1-2-11/h3,6,11,13-15,19,25H,1-2,4-5,7-10H2/t13-,14+,15+,19+,20-/m0/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
kappa-type opioid receptor isoform 1
(Frog) | BDBM81931

(CAM | CAS_56337-94-1 | N-cyclopropylmethylnorazido...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CC[C@@H](N=[N+]=[N-])[C@H]5Oc1c2[C@]45CCN3CC1CC1 Show InChI InChI=1S/C20H24N4O2/c21-23-22-14-5-4-13-15-9-12-3-6-16(25)18-17(12)20(13,19(14)26-18)7-8-24(15)10-11-1-2-11/h3,6,11,13-15,19,25H,1-2,4-5,7-10H2/t13-,14+,15+,19+,20-/m0/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50010859
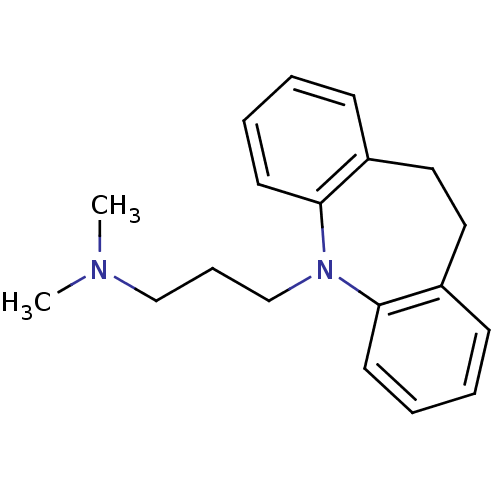
(CHEMBL11 | IMIPRAMINE HYDROCHLORIDE | IMIPRAMINE P...)Show InChI InChI=1S/C19H24N2/c1-20(2)14-7-15-21-18-10-5-3-8-16(18)12-13-17-9-4-6-11-19(17)21/h3-6,8-11H,7,12-15H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 500-7 (1998)
BindingDB Entry DOI: 10.7270/Q2HX1B6S |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070381
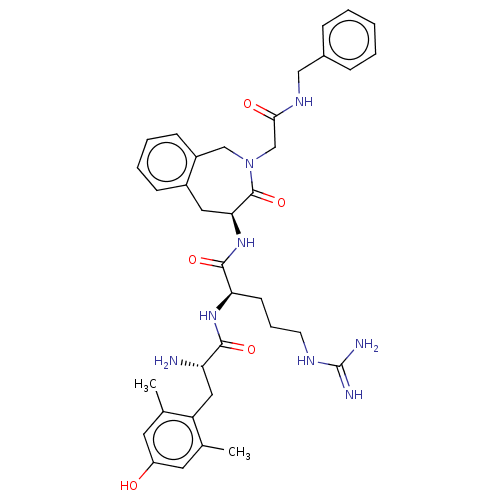
(CHEMBL3408730)Show SMILES [H][C@@]1(Cc2ccccc2CN(CC(=O)NCc2ccccc2)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C36H46N8O5/c1-22-15-27(45)16-23(2)28(22)18-29(37)33(47)42-30(13-8-14-40-36(38)39)34(48)43-31-17-25-11-6-7-12-26(25)20-44(35(31)49)21-32(46)41-19-24-9-4-3-5-10-24/h3-7,9-12,15-16,29-31,45H,8,13-14,17-21,37H2,1-2H3,(H,41,46)(H,42,47)(H,43,48)(H4,38,39,40)/t29-,30+,31-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50346330
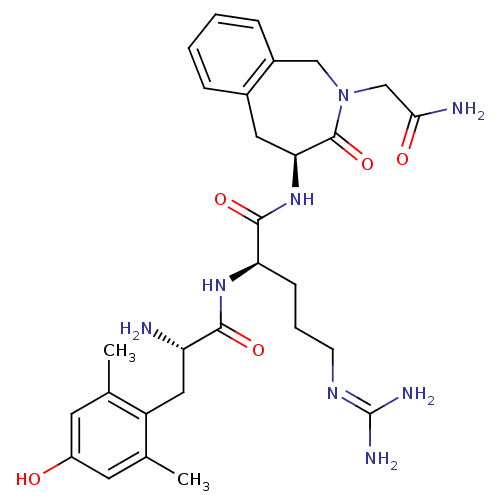
((R)-N-((S)-2-(2-amino-2-oxoethyl)-3-oxo-2,3,4,5-te...)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@H]-1-[#6]-c2ccccc2-[#6]-[#7](-[#6]-[#6](-[#7])=O)-[#6]-1=O |r| Show InChI InChI=1S/C29H40N8O5/c1-16-10-20(38)11-17(2)21(16)13-22(30)26(40)35-23(8-5-9-34-29(32)33)27(41)36-24-12-18-6-3-4-7-19(18)14-37(28(24)42)15-25(31)39/h3-4,6-7,10-11,22-24,38H,5,8-9,12-15,30H2,1-2H3,(H2,31,39)(H,35,40)(H,36,41)(H4,32,33,34)/t22-,23+,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50346330

((R)-N-((S)-2-(2-amino-2-oxoethyl)-3-oxo-2,3,4,5-te...)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@H]-1-[#6]-c2ccccc2-[#6]-[#7](-[#6]-[#6](-[#7])=O)-[#6]-1=O |r| Show InChI InChI=1S/C29H40N8O5/c1-16-10-20(38)11-17(2)21(16)13-22(30)26(40)35-23(8-5-9-34-29(32)33)27(41)36-24-12-18-6-3-4-7-19(18)14-37(28(24)42)15-25(31)39/h3-4,6-7,10-11,22-24,38H,5,8-9,12-15,30H2,1-2H3,(H2,31,39)(H,35,40)(H,36,41)(H4,32,33,34)/t22-,23+,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes after 2 hrs |
J Med Chem 54: 2467-76 (2011)
Article DOI: 10.1021/jm1016285
BindingDB Entry DOI: 10.7270/Q2416XD1 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM81931

(CAM | CAS_56337-94-1 | N-cyclopropylmethylnorazido...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CC[C@@H](N=[N+]=[N-])[C@H]5Oc1c2[C@]45CCN3CC1CC1 Show InChI InChI=1S/C20H24N4O2/c21-23-22-14-5-4-13-15-9-12-3-6-16(25)18-17(12)20(13,19(14)26-18)7-8-24(15)10-11-1-2-11/h3,6,11,13-15,19,25H,1-2,4-5,7-10H2/t13-,14+,15+,19+,20-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070377

(CHEMBL3408519)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(=O)N(C)Cc2ccccc2)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C38H50N8O5/c1-24-18-29(47)19-25(2)30(24)21-31(39)35(49)43-32(14-9-16-42-38(40)41)36(50)44-33-20-27-12-7-8-13-28(27)23-46(37(33)51)17-15-34(48)45(3)22-26-10-5-4-6-11-26/h4-8,10-13,18-19,31-33,47H,9,14-17,20-23,39H2,1-3H3,(H,43,49)(H,44,50)(H4,40,41,42)/t31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from rat brain DOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070379
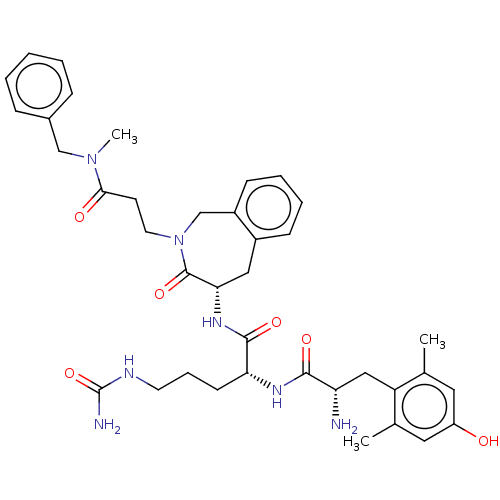
(CHEMBL3408521)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(=O)N(C)Cc2ccccc2)C1=O)NC(=O)[C@@H](CCCNC(N)=O)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C38H49N7O6/c1-24-18-29(46)19-25(2)30(24)21-31(39)35(48)42-32(14-9-16-41-38(40)51)36(49)43-33-20-27-12-7-8-13-28(27)23-45(37(33)50)17-15-34(47)44(3)22-26-10-5-4-6-11-26/h4-8,10-13,18-19,31-33,46H,9,14-17,20-23,39H2,1-3H3,(H,42,48)(H,43,49)(H3,40,41,51)/t31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM81931

(CAM | CAS_56337-94-1 | N-cyclopropylmethylnorazido...)Show SMILES Oc1ccc2C[C@@H]3[C@@H]4CC[C@@H](N=[N+]=[N-])[C@H]5Oc1c2[C@]45CCN3CC1CC1 Show InChI InChI=1S/C20H24N4O2/c21-23-22-14-5-4-13-15-9-12-3-6-16(25)18-17(12)20(13,19(14)26-18)7-8-24(15)10-11-1-2-11/h3,6,11,13-15,19,25H,1-2,4-5,7-10H2/t13-,14+,15+,19+,20-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070378

(CHEMBL3408520)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(=O)N(C)Cc2cc(cc(c2)C(F)(F)F)C(F)(F)F)C1=O)NC(=O)[C@@H](CCCNC(N)=O)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C40H47F6N7O6/c1-22-13-29(54)14-23(2)30(22)19-31(47)35(56)50-32(9-6-11-49-38(48)59)36(57)51-33-17-25-7-4-5-8-26(25)21-53(37(33)58)12-10-34(55)52(3)20-24-15-27(39(41,42)43)18-28(16-24)40(44,45)46/h4-5,7-8,13-16,18,31-33,54H,6,9-12,17,19-21,47H2,1-3H3,(H,50,56)(H,51,57)(H3,48,49,59)/t31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
kappa-type opioid receptor isoform 1
(Frog) | BDBM81935
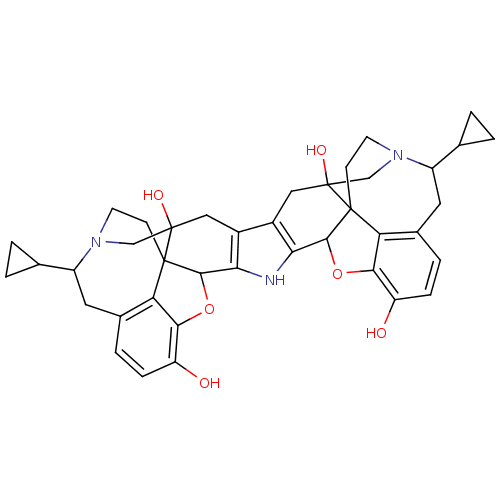
(CAS_105618-26-6 | NSC_3034416 | Nor-BNI)Show SMILES Oc1ccc2CC(C3CC3)N3CCC45C(Oc1c24)c1[nH]c2C4Oc6c7c(CC(C8CC8)N8CCC47C(O)(C8)Cc2c1CC5(O)C3)ccc6O |TLB:7:6:12.11:44.42,43:42:6.5.4.17:12.11,28:27:35.37:32.33,THB:23:24:35.37:32.33,45:25:35.37:32.33,41:42:6.5.4.17:12.11,16:17:12.11:44.42,3:4:12.11:44.42,36:35:32.33:25.24.26.27| Show InChI InChI=1S/C40H43N3O6/c44-27-7-5-21-13-25(19-1-2-19)42-11-9-39-29(21)33(27)48-35(39)31-23(15-37(39,46)17-42)24-16-38(47)18-43-12-10-40(38)30-22(14-26(43)20-3-4-20)6-8-28(45)34(30)49-36(40)32(24)41-31/h5-8,19-20,25-26,35-36,41,44-47H,1-4,9-18H2 | PDB
Reactome pathway
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50346329
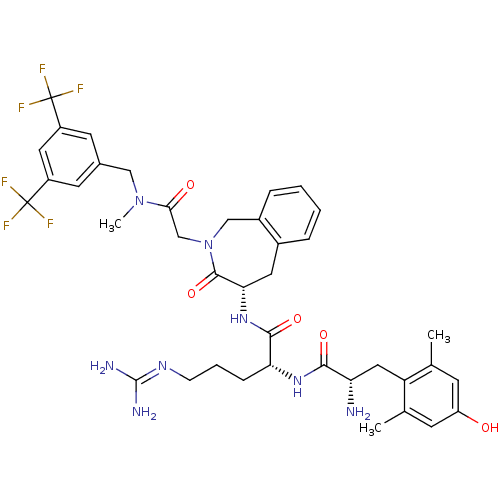
((R)-2-((S)-2-amino-3-(4-hydroxy-2,6-dimethylphenyl...)Show SMILES [#6]-[#7](-[#6]-c1cc(cc(c1)C(F)(F)F)C(F)(F)F)-[#6](=O)-[#6]-[#7]-1-[#6]-c2ccccc2-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-c2c(-[#6])cc(-[#8])cc2-[#6])-[#6]-1=O |r| Show InChI InChI=1S/C39H46F6N8O5/c1-21-11-28(54)12-22(2)29(21)17-30(46)34(56)50-31(9-6-10-49-37(47)48)35(57)51-32-15-24-7-4-5-8-25(24)19-53(36(32)58)20-33(55)52(3)18-23-13-26(38(40,41)42)16-27(14-23)39(43,44)45/h4-5,7-8,11-14,16,30-32,54H,6,9-10,15,17-20,46H2,1-3H3,(H,50,56)(H,51,57)(H4,47,48,49)/t30-,31+,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.416 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from mu opioid receptor in rat brain membranes after 2 hrs |
J Med Chem 54: 2467-76 (2011)
Article DOI: 10.1021/jm1016285
BindingDB Entry DOI: 10.7270/Q2416XD1 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50346329

((R)-2-((S)-2-amino-3-(4-hydroxy-2,6-dimethylphenyl...)Show SMILES [#6]-[#7](-[#6]-c1cc(cc(c1)C(F)(F)F)C(F)(F)F)-[#6](=O)-[#6]-[#7]-1-[#6]-c2ccccc2-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-c2c(-[#6])cc(-[#8])cc2-[#6])-[#6]-1=O |r| Show InChI InChI=1S/C39H46F6N8O5/c1-21-11-28(54)12-22(2)29(21)17-30(46)34(56)50-31(9-6-10-49-37(47)48)35(57)51-32-15-24-7-4-5-8-25(24)19-53(36(32)58)20-33(55)52(3)18-23-13-26(38(40,41)42)16-27(14-23)39(43,44)45/h4-5,7-8,11-14,16,30-32,54H,6,9-10,15,17-20,46H2,1-3H3,(H,50,56)(H,51,57)(H4,47,48,49)/t30-,31+,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070379

(CHEMBL3408521)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(=O)N(C)Cc2ccccc2)C1=O)NC(=O)[C@@H](CCCNC(N)=O)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C38H49N7O6/c1-24-18-29(46)19-25(2)30(24)21-31(39)35(48)42-32(14-9-16-41-38(40)51)36(49)43-33-20-27-12-7-8-13-28(27)23-45(37(33)50)17-15-34(47)44(3)22-26-10-5-4-6-11-26/h4-8,10-13,18-19,31-33,46H,9,14-17,20-23,39H2,1-3H3,(H,42,48)(H,43,49)(H3,40,41,51)/t31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from rat brain DOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50346329

((R)-2-((S)-2-amino-3-(4-hydroxy-2,6-dimethylphenyl...)Show SMILES [#6]-[#7](-[#6]-c1cc(cc(c1)C(F)(F)F)C(F)(F)F)-[#6](=O)-[#6]-[#7]-1-[#6]-c2ccccc2-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-c2c(-[#6])cc(-[#8])cc2-[#6])-[#6]-1=O |r| Show InChI InChI=1S/C39H46F6N8O5/c1-21-11-28(54)12-22(2)29(21)17-30(46)34(56)50-31(9-6-10-49-37(47)48)35(57)51-32-15-24-7-4-5-8-25(24)19-53(36(32)58)20-33(55)52(3)18-23-13-26(38(40,41)42)16-27(14-23)39(43,44)45/h4-5,7-8,11-14,16,30-32,54H,6,9-10,15,17-20,46H2,1-3H3,(H,50,56)(H,51,57)(H4,47,48,49)/t30-,31+,32-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]SP from human recombinant NK1 receptor expressed in CHO cells after 20 mins by liquid scintillation counting |
J Med Chem 54: 2467-76 (2011)
Article DOI: 10.1021/jm1016285
BindingDB Entry DOI: 10.7270/Q2416XD1 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM21015

((2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydroxyphenyl...)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N(C)[C@@H](Cc1ccccc1)C(=O)NCCO Show InChI InChI=1S/C26H35N5O6/c1-17(30-25(36)21(27)14-19-8-10-20(33)11-9-19)24(35)29-16-23(34)31(2)22(26(37)28-12-13-32)15-18-6-4-3-5-7-18/h3-11,17,21-22,32-33H,12-16,27H2,1-2H3,(H,28,37)(H,29,35)(H,30,36)/t17-,21+,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Substance-P receptor
(Homo sapiens (Human)) | BDBM50346329

((R)-2-((S)-2-amino-3-(4-hydroxy-2,6-dimethylphenyl...)Show SMILES [#6]-[#7](-[#6]-c1cc(cc(c1)C(F)(F)F)C(F)(F)F)-[#6](=O)-[#6]-[#7]-1-[#6]-c2ccccc2-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-c2c(-[#6])cc(-[#8])cc2-[#6])-[#6]-1=O |r| Show InChI InChI=1S/C39H46F6N8O5/c1-21-11-28(54)12-22(2)29(21)17-30(46)34(56)50-31(9-6-10-49-37(47)48)35(57)51-32-15-24-7-4-5-8-25(24)19-53(36(32)58)20-33(55)52(3)18-23-13-26(38(40,41)42)16-27(14-23)39(43,44)45/h4-5,7-8,11-14,16,30-32,54H,6,9-10,15,17-20,46H2,1-3H3,(H,50,56)(H,51,57)(H4,47,48,49)/t30-,31+,32-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]SP from human NK1 receptor expressed in CHO cells after 20 mins by liquid scintillation counting analysis |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000788
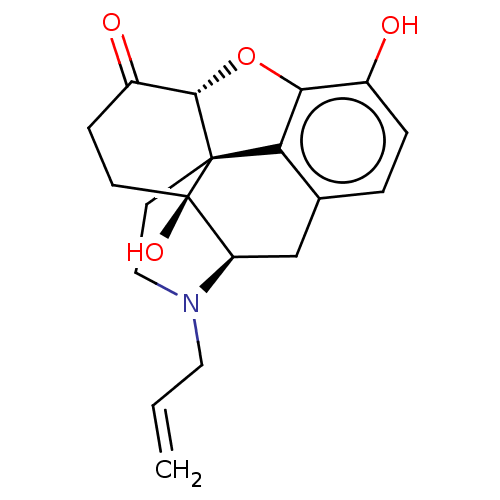
((morphine)4-methyl-(1S,5R,13R,14S,17R)-12-oxa-4-az...)Show SMILES Oc1ccc2C[C@H]3N(CC=C)CC[C@@]45[C@@H](Oc1c24)C(=O)CC[C@@]35O |r| Show InChI InChI=1S/C19H21NO4/c1-2-8-20-9-7-18-15-11-3-4-12(21)16(15)24-17(18)13(22)5-6-19(18,23)14(20)10-11/h2-4,14,17,21,23H,1,5-10H2/t14-,17+,18+,19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070378

(CHEMBL3408520)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(=O)N(C)Cc2cc(cc(c2)C(F)(F)F)C(F)(F)F)C1=O)NC(=O)[C@@H](CCCNC(N)=O)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C40H47F6N7O6/c1-22-13-29(54)14-23(2)30(22)19-31(47)35(56)50-32(9-6-11-49-38(48)59)36(57)51-33-17-25-7-4-5-8-26(25)21-53(37(33)58)12-10-34(55)52(3)20-24-15-27(39(41,42)43)18-28(16-24)40(44,45)46/h4-5,7-8,13-16,18,31-33,54H,6,9-12,17,19-21,47H2,1-3H3,(H,50,56)(H,51,57)(H3,48,49,59)/t31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from rat brain DOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50346330

((R)-N-((S)-2-(2-amino-2-oxoethyl)-3-oxo-2,3,4,5-te...)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@H]-1-[#6]-c2ccccc2-[#6]-[#7](-[#6]-[#6](-[#7])=O)-[#6]-1=O |r| Show InChI InChI=1S/C29H40N8O5/c1-16-10-20(38)11-17(2)21(16)13-22(30)26(40)35-23(8-5-9-34-29(32)33)27(41)36-24-12-18-6-3-4-7-19(18)14-37(28(24)42)15-25(31)39/h3-4,6-7,10-11,22-24,38H,5,8-9,12-15,30H2,1-2H3,(H2,31,39)(H,35,40)(H,36,41)(H4,32,33,34)/t22-,23+,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from rat brain DOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000092

((-)-(etorphine) | (-)-morphine | (1S,5R,13R,14S)-1...)Show SMILES CN1CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4O)ccc5O |r,c:16,TLB:13:12:8.9.10:3.2.1| Show InChI InChI=1S/C17H19NO3/c1-18-7-6-17-10-3-5-13(20)16(17)21-15-12(19)4-2-9(14(15)17)8-11(10)18/h2-5,10-11,13,16,19-20H,6-8H2,1H3/t10-,11+,13-,16-,17-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50346330

((R)-N-((S)-2-(2-amino-2-oxoethyl)-3-oxo-2,3,4,5-te...)Show SMILES [#6]-c1cc(-[#8])cc(-[#6])c1-[#6]-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6@H]-1-[#6]-c2ccccc2-[#6]-[#7](-[#6]-[#6](-[#7])=O)-[#6]-1=O |r| Show InChI InChI=1S/C29H40N8O5/c1-16-10-20(38)11-17(2)21(16)13-22(30)26(40)35-23(8-5-9-34-29(32)33)27(41)36-24-12-18-6-3-4-7-19(18)14-37(28(24)42)15-25(31)39/h3-4,6-7,10-11,22-24,38H,5,8-9,12-15,30H2,1-2H3,(H2,31,39)(H,35,40)(H,36,41)(H4,32,33,34)/t22-,23+,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from delta opioid receptor in rat brain membranes after 2 hrs |
J Med Chem 54: 2467-76 (2011)
Article DOI: 10.1021/jm1016285
BindingDB Entry DOI: 10.7270/Q2416XD1 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070381

(CHEMBL3408730)Show SMILES [H][C@@]1(Cc2ccccc2CN(CC(=O)NCc2ccccc2)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C36H46N8O5/c1-22-15-27(45)16-23(2)28(22)18-29(37)33(47)42-30(13-8-14-40-36(38)39)34(48)43-31-17-25-11-6-7-12-26(25)20-44(35(31)49)21-32(46)41-19-24-9-4-3-5-10-24/h3-7,9-12,15-16,29-31,45H,8,13-14,17-21,37H2,1-2H3,(H,41,46)(H,42,47)(H,43,48)(H4,38,39,40)/t29-,30+,31-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from rat brain DOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070384
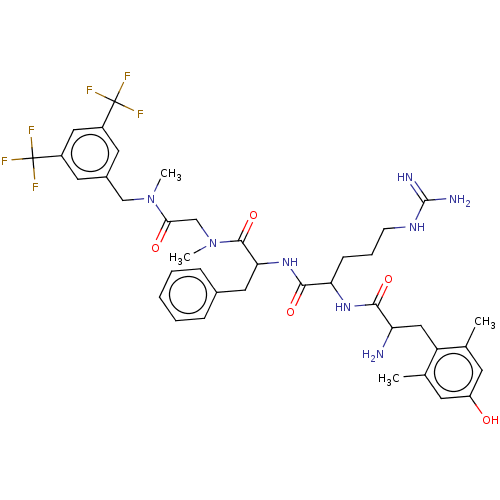
(CHEMBL3408735)Show SMILES CN(Cc1cc(cc(c1)C(F)(F)F)C(F)(F)F)C(=O)CN(C)C(=O)C(Cc1ccccc1)NC(=O)C(CCCNC(N)=N)NC(=O)C(N)Cc1c(C)cc(O)cc1C Show InChI InChI=1S/C39H48F6N8O5/c1-22-13-28(54)14-23(2)29(22)19-30(46)34(56)50-31(11-8-12-49-37(47)48)35(57)51-32(17-24-9-6-5-7-10-24)36(58)53(4)21-33(55)52(3)20-25-15-26(38(40,41)42)18-27(16-25)39(43,44)45/h5-7,9-10,13-16,18,30-32,54H,8,11-12,17,19-21,46H2,1-4H3,(H,50,56)(H,51,57)(H4,47,48,49) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM86231
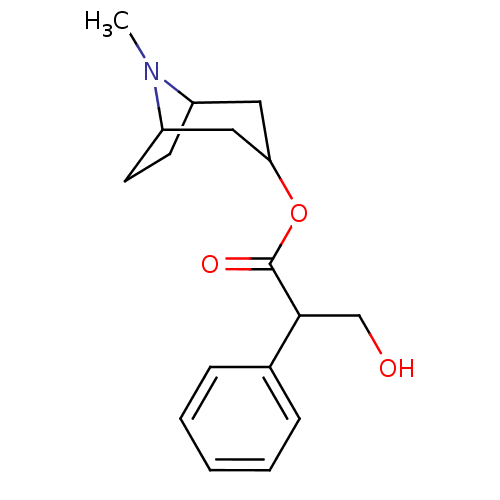
(ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...)Show SMILES CN1C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |THB:9:7:1:3.4| Show InChI InChI=1S/C17H23NO3/c1-18-13-7-8-14(18)10-15(9-13)21-17(20)16(11-19)12-5-3-2-4-6-12/h2-6,13-16,19H,7-11H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.630 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 500-7 (1998)
BindingDB Entry DOI: 10.7270/Q2HX1B6S |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(RAT) | BDBM86231

(ATR | ATROPINE | Atropine,(-) | CAS_51-55-8 | CHEM...)Show SMILES CN1C2CCC1CC(C2)OC(=O)C(CO)c1ccccc1 |THB:9:7:1:3.4| Show InChI InChI=1S/C17H23NO3/c1-18-13-7-8-14(18)10-15(9-13)21-17(20)16(11-19)12-5-3-2-4-6-12/h2-6,13-16,19H,7-11H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 0.870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 500-7 (1998)
BindingDB Entry DOI: 10.7270/Q2HX1B6S |
More data for this
Ligand-Target Pair | |
kappa-type opioid receptor isoform 1
(Frog) | BDBM50000788

((morphine)4-methyl-(1S,5R,13R,14S,17R)-12-oxa-4-az...)Show SMILES Oc1ccc2C[C@H]3N(CC=C)CC[C@@]45[C@@H](Oc1c24)C(=O)CC[C@@]35O |r| Show InChI InChI=1S/C19H21NO4/c1-2-8-20-9-7-18-15-11-3-4-12(21)16(15)24-17(18)13(22)5-6-19(18,23)14(20)10-11/h2-4,14,17,21,23H,1,5-10H2/t14-,17+,18+,19-/m1/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000092

((-)-(etorphine) | (-)-morphine | (1S,5R,13R,14S)-1...)Show SMILES CN1CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4O)ccc5O |r,c:16,TLB:13:12:8.9.10:3.2.1| Show InChI InChI=1S/C17H19NO3/c1-18-7-6-17-10-3-5-13(20)16(17)21-15-12(19)4-2-9(14(15)17)8-11(10)18/h2-5,10-11,13,16,19-20H,6-8H2,1H3/t10-,11+,13-,16-,17-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
kappa-type opioid receptor isoform 1
(Frog) | BDBM50000091
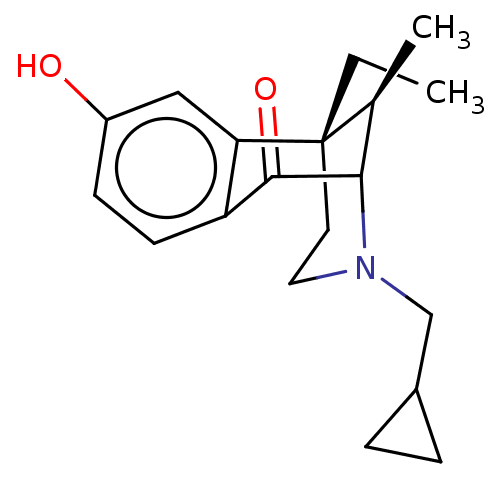
((6S,11R)-3-Cyclopropylmethyl-6-ethyl-8-hydroxy-11-...)Show SMILES CC[C@]12CCN(CC3CC3)C([C@@H]1C)C(=O)c1ccc(O)cc21 |TLB:6:5:13.21.15:11,14:13:11:5.3.4,THB:20:21:11:5.3.4| Show InChI InChI=1S/C19H25NO2/c1-3-19-8-9-20(11-13-4-5-13)17(12(19)2)18(22)15-7-6-14(21)10-16(15)19/h6-7,10,12-13,17,21H,3-5,8-9,11H2,1-2H3/t12-,17?,19-/m0/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50018550
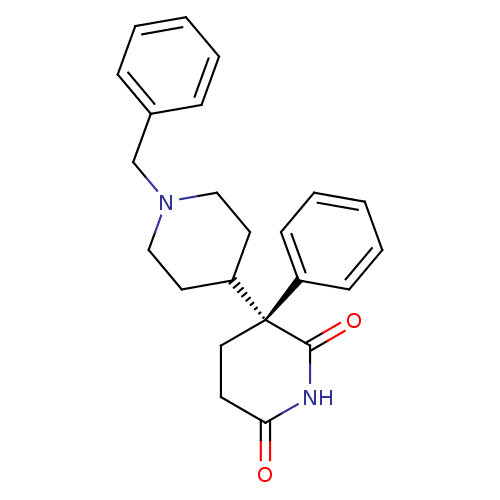
(1'-Benzyl-3-phenyl-[3,4']bipiperidinyl-2,6-dione |...)Show SMILES O=C1CC[C@](C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27)/t23-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 500-7 (1998)
BindingDB Entry DOI: 10.7270/Q2HX1B6S |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070386
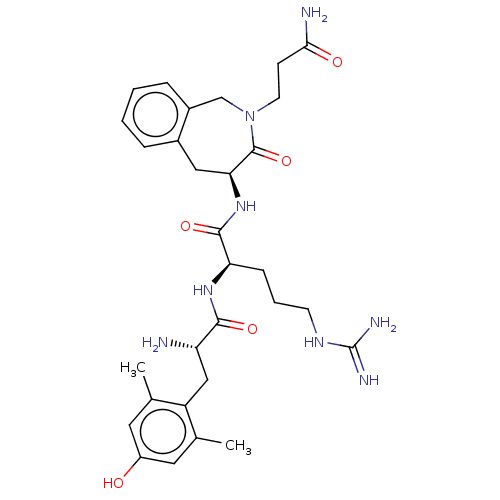
(CHEMBL3408737)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(N)=O)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C30H42N8O5/c1-17-12-21(39)13-18(2)22(17)15-23(31)27(41)36-24(8-5-10-35-30(33)34)28(42)37-25-14-19-6-3-4-7-20(19)16-38(29(25)43)11-9-26(32)40/h3-4,6-7,12-13,23-25,39H,5,8-11,14-16,31H2,1-2H3,(H2,32,40)(H,36,41)(H,37,42)(H4,33,34,35)/t23-,24+,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DAMGO from rat brain MOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(RAT) | BDBM50018550

(1'-Benzyl-3-phenyl-[3,4']bipiperidinyl-2,6-dione |...)Show SMILES O=C1CC[C@](C2CCN(Cc3ccccc3)CC2)(C(=O)N1)c1ccccc1 Show InChI InChI=1S/C23H26N2O2/c26-21-11-14-23(22(27)24-21,19-9-5-2-6-10-19)20-12-15-25(16-13-20)17-18-7-3-1-4-8-18/h1-10,20H,11-17H2,(H,24,26,27)/t23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Arizona
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 284: 500-7 (1998)
BindingDB Entry DOI: 10.7270/Q2HX1B6S |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070384

(CHEMBL3408735)Show SMILES CN(Cc1cc(cc(c1)C(F)(F)F)C(F)(F)F)C(=O)CN(C)C(=O)C(Cc1ccccc1)NC(=O)C(CCCNC(N)=N)NC(=O)C(N)Cc1c(C)cc(O)cc1C Show InChI InChI=1S/C39H48F6N8O5/c1-22-13-28(54)14-23(2)29(22)19-30(46)34(56)50-31(11-8-12-49-37(47)48)35(57)51-32(17-24-9-6-5-7-10-24)36(58)53(4)21-33(55)52(3)20-25-15-26(38(40,41)42)18-27(16-25)39(43,44)45/h5-7,9-10,13-16,18,30-32,54H,8,11-12,17,19-21,46H2,1-4H3,(H,50,56)(H,51,57)(H4,47,48,49) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from rat brain DOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
kappa-type opioid receptor isoform 1
(Frog) | BDBM50000091

((6S,11R)-3-Cyclopropylmethyl-6-ethyl-8-hydroxy-11-...)Show SMILES CC[C@]12CCN(CC3CC3)C([C@@H]1C)C(=O)c1ccc(O)cc21 |TLB:6:5:13.21.15:11,14:13:11:5.3.4,THB:20:21:11:5.3.4| Show InChI InChI=1S/C19H25NO2/c1-3-19-8-9-20(11-13-4-5-13)17(12(19)2)18(22)15-7-6-14(21)10-16(15)19/h6-7,10,12-13,17,21H,3-5,8-9,11H2,1-2H3/t12-,17?,19-/m0/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070376

(CHEMBL3408518)Show SMILES [H][C@@]1(Cc2ccccc2CN(CCC(=O)N(C)Cc2cc(cc(c2)C(F)(F)F)C(F)(F)F)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C40H48F6N8O5/c1-22-13-29(55)14-23(2)30(22)19-31(47)35(57)51-32(9-6-11-50-38(48)49)36(58)52-33-17-25-7-4-5-8-26(25)21-54(37(33)59)12-10-34(56)53(3)20-24-15-27(39(41,42)43)18-28(16-24)40(44,45)46/h4-5,7-8,13-16,18,31-33,55H,6,9-12,17,19-21,47H2,1-3H3,(H,51,57)(H,52,58)(H4,48,49,50)/t31-,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from rat brain DOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM81934
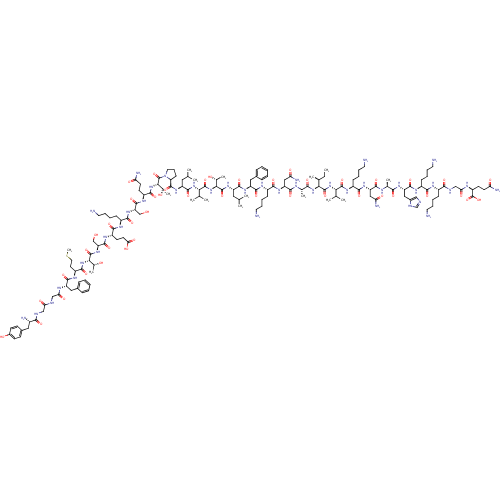
(Beta-Endorphin)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](C)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H]1CCCN1C(=O)[C@@H](NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CCSC)NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)[C@@H](C)O)[C@@H](C)O)C(C)C)[C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCC(N)=O)C(O)=O Show InChI InChI=1S/C154H248N42O44S/c1-17-82(10)123(150(235)191-121(80(6)7)148(233)181-98(43-28-33-60-159)133(218)185-108(69-115(163)205)138(223)170-83(11)127(212)182-107(68-91-71-165-77-169-91)143(228)177-95(40-25-30-57-156)131(216)174-94(39-24-29-56-155)130(215)168-74-119(209)172-102(154(239)240)50-53-114(162)204)192-128(213)84(12)171-139(224)109(70-116(164)206)184-132(217)96(41-26-31-58-157)176-142(227)106(67-89-37-22-19-23-38-89)183-140(225)103(63-78(2)3)187-151(236)125(86(14)200)194-149(234)122(81(8)9)190-144(229)104(64-79(4)5)186-147(232)112-44-34-61-196(112)153(238)126(87(15)201)195-136(221)99(49-52-113(161)203)178-145(230)110(75-197)188-134(219)97(42-27-32-59-158)175-135(220)100(51-54-120(210)211)179-146(231)111(76-198)189-152(237)124(85(13)199)193-137(222)101(55-62-241-16)180-141(226)105(66-88-35-20-18-21-36-88)173-118(208)73-166-117(207)72-167-129(214)93(160)65-90-45-47-92(202)48-46-90/h18-23,35-38,45-48,71,77-87,93-112,121-126,197-202H,17,24-34,39-44,49-70,72-76,155-160H2,1-16H3,(H2,161,203)(H2,162,204)(H2,163,205)(H2,164,206)(H,165,169)(H,166,207)(H,167,214)(H,168,215)(H,170,223)(H,171,224)(H,172,209)(H,173,208)(H,174,216)(H,175,220)(H,176,227)(H,177,228)(H,178,230)(H,179,231)(H,180,226)(H,181,233)(H,182,212)(H,183,225)(H,184,217)(H,185,218)(H,186,232)(H,187,236)(H,188,219)(H,189,237)(H,190,229)(H,191,235)(H,192,213)(H,193,222)(H,194,234)(H,195,221)(H,210,211)(H,239,240)/t82-,83-,84-,85+,86+,87+,93-,94-,95-,96-,97-,98-,99-,100-,101-,102-,103-,104-,105-,106-,107-,108-,109-,110-,111-,112-,121-,122-,123-,124-,125-,126-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000091

((6S,11R)-3-Cyclopropylmethyl-6-ethyl-8-hydroxy-11-...)Show SMILES CC[C@]12CCN(CC3CC3)C([C@@H]1C)C(=O)c1ccc(O)cc21 |TLB:6:5:13.21.15:11,14:13:11:5.3.4,THB:20:21:11:5.3.4| Show InChI InChI=1S/C19H25NO2/c1-3-19-8-9-20(11-13-4-5-13)17(12(19)2)18(22)15-7-6-14(21)10-16(15)19/h6-7,10,12-13,17,21H,3-5,8-9,11H2,1-2H3/t12-,17?,19-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Delta-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50070380

(CHEMBL3408522)Show SMILES [H][C@@]1(Cc2ccccc2CN(CC(=O)N(C)Cc2ccccc2)C1=O)NC(=O)[C@@H](CCCNC(N)=N)NC(=O)[C@@H](N)Cc1c(C)cc(O)cc1C |r| Show InChI InChI=1S/C37H48N8O5/c1-23-16-28(46)17-24(2)29(23)19-30(38)34(48)42-31(14-9-15-41-37(39)40)35(49)43-32-18-26-12-7-8-13-27(26)21-45(36(32)50)22-33(47)44(3)20-25-10-5-4-6-11-25/h4-8,10-13,16-17,30-32,46H,9,14-15,18-22,38H2,1-3H3,(H,42,48)(H,43,49)(H4,39,40,41)/t30-,31+,32-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Brussel
Curated by ChEMBL
| Assay Description
Displacement of [3H]DSLET from rat brain DOR after 2 hrs |
Eur J Med Chem 92: 64-77 (2015)
Article DOI: 10.1016/j.ejmech.2014.12.033
BindingDB Entry DOI: 10.7270/Q2M90BCC |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM21015

((2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydroxyphenyl...)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N(C)[C@@H](Cc1ccccc1)C(=O)NCCO Show InChI InChI=1S/C26H35N5O6/c1-17(30-25(36)21(27)14-19-8-10-20(33)11-9-19)24(35)29-16-23(34)31(2)22(26(37)28-12-13-32)15-18-6-4-3-5-7-18/h3-11,17,21-22,32-33H,12-16,27H2,1-2H3,(H,28,37)(H,29,35)(H,30,36)/t17-,21+,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
kappa-type opioid receptor isoform 1
(Frog) | BDBM50000092

((-)-(etorphine) | (-)-morphine | (1S,5R,13R,14S)-1...)Show SMILES CN1CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4O)ccc5O |r,c:16,TLB:13:12:8.9.10:3.2.1| Show InChI InChI=1S/C17H19NO3/c1-18-7-6-17-10-3-5-13(20)16(17)21-15-12(19)4-2-9(14(15)17)8-11(10)18/h2-5,10-11,13,16,19-20H,6-8H2,1H3/t10-,11+,13-,16-,17-/m0/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
kappa-type opioid receptor isoform 1
(Frog) | BDBM81935

(CAS_105618-26-6 | NSC_3034416 | Nor-BNI)Show SMILES Oc1ccc2CC(C3CC3)N3CCC45C(Oc1c24)c1[nH]c2C4Oc6c7c(CC(C8CC8)N8CCC47C(O)(C8)Cc2c1CC5(O)C3)ccc6O |TLB:7:6:12.11:44.42,43:42:6.5.4.17:12.11,28:27:35.37:32.33,THB:23:24:35.37:32.33,45:25:35.37:32.33,41:42:6.5.4.17:12.11,16:17:12.11:44.42,3:4:12.11:44.42,36:35:32.33:25.24.26.27| Show InChI InChI=1S/C40H43N3O6/c44-27-7-5-21-13-25(19-1-2-19)42-11-9-39-29(21)33(27)48-35(39)31-23(15-37(39,46)17-42)24-16-38(47)18-43-12-10-40(38)30-22(14-26(43)20-3-4-20)6-8-28(45)34(30)49-36(40)32(24)41-31/h5-8,19-20,25-26,35-36,41,44-47H,1-4,9-18H2 | PDB
Reactome pathway
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
kappa-type opioid receptor isoform 1
(Frog) | BDBM224024
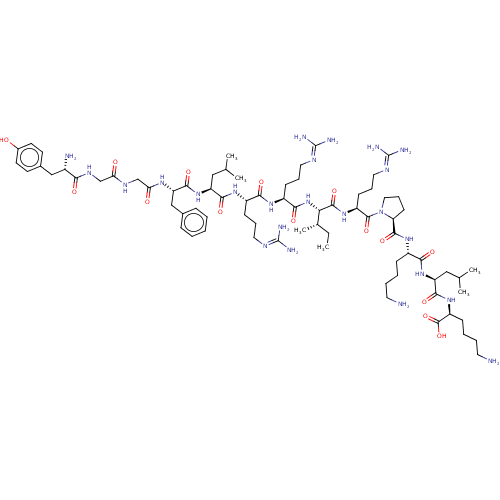
(BDBM50241435 | Dynorphin A (1-13) | YGGFLRRXRPKLK)Show SMILES [#6]-[#6]-[#6@H](-[#6])-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-c1ccc(-[#8])cc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](-[#8])=O Show InChI InChI=1S/C75H126N24O15/c1-7-45(6)61(70(111)94-53(25-17-35-87-75(83)84)71(112)99-36-18-26-58(99)69(110)93-50(21-11-13-31-76)64(105)96-56(38-44(4)5)67(108)95-54(72(113)114)22-12-14-32-77)98-65(106)52(24-16-34-86-74(81)82)91-63(104)51(23-15-33-85-73(79)80)92-66(107)55(37-43(2)3)97-68(109)57(40-46-19-9-8-10-20-46)90-60(102)42-88-59(101)41-89-62(103)49(78)39-47-27-29-48(100)30-28-47/h8-10,19-20,27-30,43-45,49-58,61,100H,7,11-18,21-26,31-42,76-78H2,1-6H3,(H,88,101)(H,89,103)(H,90,102)(H,91,104)(H,92,107)(H,93,110)(H,94,111)(H,95,108)(H,96,105)(H,97,109)(H,98,106)(H,113,114)(H4,79,80,85)(H4,81,82,86)(H4,83,84,87)/t45-,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-,61-/m0/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000091

((6S,11R)-3-Cyclopropylmethyl-6-ethyl-8-hydroxy-11-...)Show SMILES CC[C@]12CCN(CC3CC3)C([C@@H]1C)C(=O)c1ccc(O)cc21 |TLB:6:5:13.21.15:11,14:13:11:5.3.4,THB:20:21:11:5.3.4| Show InChI InChI=1S/C19H25NO2/c1-3-19-8-9-20(11-13-4-5-13)17(12(19)2)18(22)15-7-6-14(21)10-16(15)19/h6-7,10,12-13,17,21H,3-5,8-9,11H2,1-2H3/t12-,17?,19-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Biological Research Center of Hungarian Academy of Sciences
Curated by PDSP Ki Database
| |
Neurochem Res 15: 899-904 (1990)
Article DOI: 10.1007/bf00965909
BindingDB Entry DOI: 10.7270/Q25M6460 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data