 Found 307 hits Enz. Inhib. hit(s) with Target = 'Phosphorylase kinase'
Found 307 hits Enz. Inhib. hit(s) with Target = 'Phosphorylase kinase' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM50020310
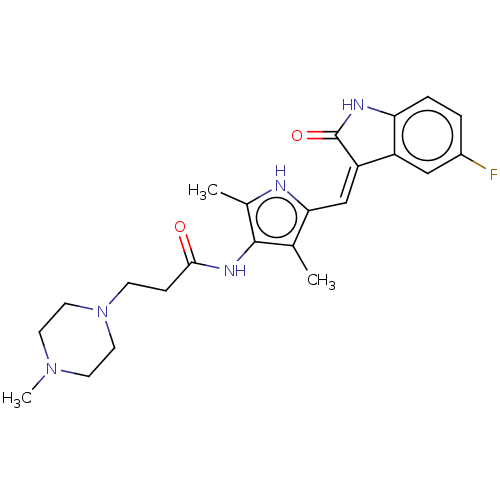
(CHEMBL3288854)Show SMILES CN1CCN(CCC(=O)Nc2c(C)[nH]c(\C=C3/C(=O)Nc4ccc(F)cc34)c2C)CC1 Show InChI InChI=1S/C23H28FN5O2/c1-14-20(13-18-17-12-16(24)4-5-19(17)26-23(18)31)25-15(2)22(14)27-21(30)6-7-29-10-8-28(3)9-11-29/h4-5,12-13,25H,6-11H2,1-3H3,(H,26,31)(H,27,30)/b18-13- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 840 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Qilu Pharmaceutical Co, Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to PHKgamma2 (unknown origin) |
Eur J Med Chem 82: 139-51 (2014)
Article DOI: 10.1016/j.ejmech.2014.05.051
BindingDB Entry DOI: 10.7270/Q2F1918D |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM50463479

(CHEMBL4249925)Show SMILES CS(=O)(=O)Nc1cccc(CC(=O)Nc2nc(cs2)-c2c[nH]c3ncccc23)c1 Show InChI InChI=1S/C19H17N5O3S2/c1-29(26,27)24-13-5-2-4-12(8-13)9-17(25)23-19-22-16(11-28-19)15-10-21-18-14(15)6-3-7-20-18/h2-8,10-11,24H,9H2,1H3,(H,20,21)(H,22,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of PHKgamma2 (unknown origin) |
Bioorg Med Chem Lett 28: 2622-2626 (2018)
Article DOI: 10.1016/j.bmcl.2018.06.040
BindingDB Entry DOI: 10.7270/Q2ZC85HX |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM50463483
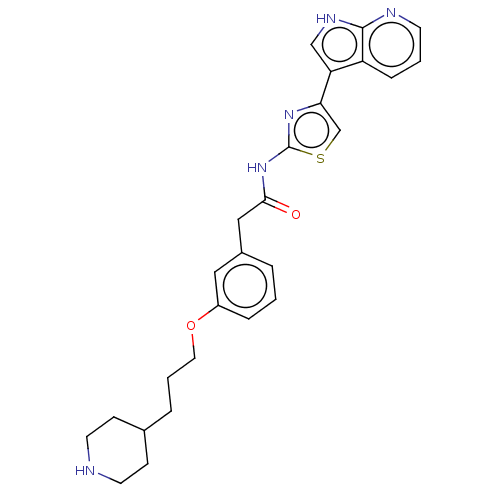
(CHEMBL4245242)Show SMILES O=C(Cc1cccc(OCCCC2CCNCC2)c1)Nc1nc(cs1)-c1c[nH]c2ncccc12 Show InChI InChI=1S/C26H29N5O2S/c32-24(31-26-30-23(17-34-26)22-16-29-25-21(22)7-2-10-28-25)15-19-4-1-6-20(14-19)33-13-3-5-18-8-11-27-12-9-18/h1-2,4,6-7,10,14,16-18,27H,3,5,8-9,11-13,15H2,(H,28,29)(H,30,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| >2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals Inc
Curated by ChEMBL
| Assay Description
Inhibition of PHKgamma2 (unknown origin) |
Bioorg Med Chem Lett 28: 2622-2626 (2018)
Article DOI: 10.1016/j.bmcl.2018.06.040
BindingDB Entry DOI: 10.7270/Q2ZC85HX |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | <0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
National Cancer Institute
Curated by ChEMBL
| Assay Description
Inhibition of PHKgamma2 |
J Med Chem 52: 3191-204 (2009)
Article DOI: 10.1021/jm800861c
BindingDB Entry DOI: 10.7270/Q23J3DWT |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.336 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PHKgamma2 using MBP as substrate by [gamma-33P]-ATP assay |
Citation and Details
Article DOI: 10.1016/j.bmc.2018.02.022
BindingDB Entry DOI: 10.7270/Q2DJ5KB8 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Institute of Science and Technology (KIST)
Curated by ChEMBL
| Assay Description
Inhibition of PHKg2 (unknown origin) |
Eur J Med Chem 141: 657-675 (2017)
Article DOI: 10.1016/j.ejmech.2017.10.003
BindingDB Entry DOI: 10.7270/Q2CV4M86 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.620 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Florida
Curated by ChEMBL
| Assay Description
Inhibition of human PHKgamma2 using MBP as substrate by [gamma-33P]-ATP assay |
Eur J Med Chem 161: 456-467 (2019)
Article DOI: 10.1016/j.ejmech.2018.10.052
BindingDB Entry DOI: 10.7270/Q2W380MT |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human PHKgamma1 using MBP as substrate by [gamma-33P]-ATP assay |
Citation and Details
Article DOI: 10.1016/j.bmc.2018.02.022
BindingDB Entry DOI: 10.7270/Q2DJ5KB8 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Florida
Curated by ChEMBL
| Assay Description
Inhibition of human PHKgamma1 using MBP as substrate by [gamma-33P]-ATP assay |
Eur J Med Chem 161: 456-467 (2019)
Article DOI: 10.1016/j.ejmech.2018.10.052
BindingDB Entry DOI: 10.7270/Q2W380MT |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM50392791
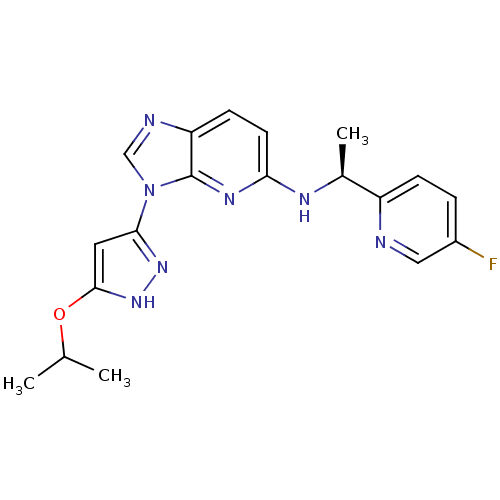
(CHEMBL2151321 | US8486966, 1)Show SMILES CC(C)Oc1cc(n[nH]1)-n1cnc2ccc(N[C@@H](C)c3ccc(F)cn3)nc12 |r| Show InChI InChI=1S/C19H20FN7O/c1-11(2)28-18-8-17(25-26-18)27-10-22-15-6-7-16(24-19(15)27)23-12(3)14-5-4-13(20)9-21-14/h4-12H,1-3H3,(H,23,24)(H,25,26)/t12-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.10 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of PhKg2 |
ACS Med Chem Lett 3: 705-709 (2012)
Article DOI: 10.1021/ml300074j
BindingDB Entry DOI: 10.7270/Q2GH9K2G |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM50177715
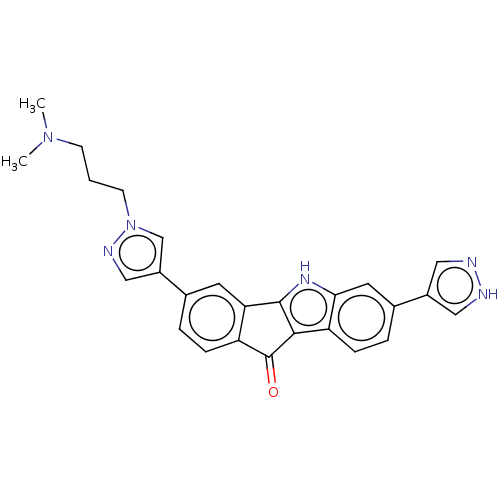
(CHEMBL3815054)Show SMILES CN(C)CCCn1cc(cn1)-c1ccc2C(=O)c3c([nH]c4cc(ccc34)-c3cn[nH]c3)-c2c1 Show InChI InChI=1S/C26H24N6O/c1-31(2)8-3-9-32-15-19(14-29-32)16-4-6-20-22(10-16)25-24(26(20)33)21-7-5-17(11-23(21)30-25)18-12-27-28-13-18/h4-7,10-15,30H,3,8-9H2,1-2H3,(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 78 | n/a | n/a | n/a | n/a | n/a | n/a |
Harvard Medical School
Curated by ChEMBL
| Assay Description
Inhibition of PHKGI (unknown origin) by kinomescan analysis |
ACS Med Chem Lett 7: 476-81 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00498
BindingDB Entry DOI: 10.7270/Q2HD7XJD |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM317462
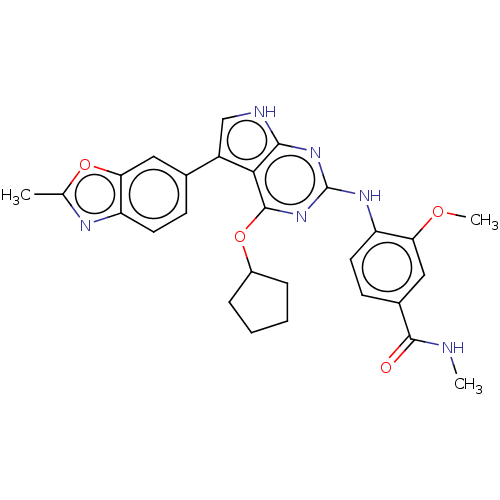
(4-(4-(cyclopentyloxy)-5-(2- methylbenzo[d]oxazol-6...)Show SMILES CNC(=O)c1ccc(Nc2nc(OC3CCCC3)c3c(c[nH]c3n2)-c2ccc3nc(C)oc3c2)c(OC)c1 Show InChI InChI=1S/C28H28N6O4/c1-15-31-21-10-8-16(12-23(21)37-15)19-14-30-25-24(19)27(38-18-6-4-5-7-18)34-28(33-25)32-20-11-9-17(26(35)29-2)13-22(20)36-3/h8-14,18H,4-7H2,1-3H3,(H,29,35)(H2,30,32,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 136 | n/a | n/a | n/a | n/a | n/a | n/a |
Celgene Corporation
Curated by ChEMBL
| Assay Description
Inhibition of full length human GST-tagged PHKG1 expressed in baculovirus by Z'-LYTE assay |
J Med Chem 60: 8989-9002 (2017)
Article DOI: 10.1021/acs.jmedchem.7b01223
BindingDB Entry DOI: 10.7270/Q2GF0WX9 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
(Oryctolagus cuniculus (rabbit)) | BDBM2691
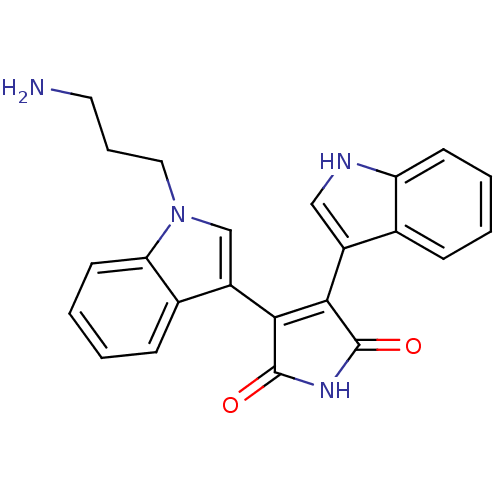
(3-[1-(3-aminopropyl)-1H-indol-3-yl]-4-(1H-indol-3-...)Show SMILES NCCCn1cc(C2=C(C(=O)NC2=O)c2c[nH]c3ccccc23)c2ccccc12 |t:7| Show InChI InChI=1S/C23H20N4O2/c24-10-5-11-27-13-17(15-7-2-4-9-19(15)27)21-20(22(28)26-23(21)29)16-12-25-18-8-3-1-6-14(16)18/h1-4,6-9,12-13,25H,5,10-11,24H2,(H,26,28,29) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratoires Glaxo
| Assay Description
The activity of PK is measured by its ability to transfer from [gamma-32P]ATP to phosphorylase b. |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
(Oryctolagus cuniculus (rabbit)) | BDBM2684

(3-[3-(dimethylamino)propyl]-3,13,23-triazahexacycl...)Show SMILES CN(C)CCCn1c2ccccc2c2c3C(=O)NC(=O)c3c3c4ccccc4[nH]c3c12 Show InChI InChI=1S/C25H22N4O2/c1-28(2)12-7-13-29-17-11-6-4-9-15(17)19-21-20(24(30)27-25(21)31)18-14-8-3-5-10-16(14)26-22(18)23(19)29/h3-6,8-11,26H,7,12-13H2,1-2H3,(H,27,30,31) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratoires Glaxo
| Assay Description
The activity of PK is measured by its ability to transfer from [gamma-32P]ATP to phosphorylase b. |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM50204290
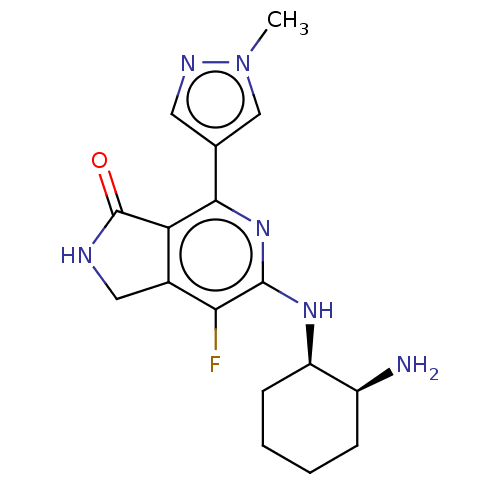
(CHEMBL3979920 | US11077111, Compound IIIa | US2023...)Show SMILES Cn1cc(cn1)-c1nc(N[C@@H]2CCCC[C@@H]2N)c(F)c2CNC(=O)c12 |r| Show InChI InChI=1S/C17H21FN6O/c1-24-8-9(6-21-24)15-13-10(7-20-17(13)25)14(18)16(23-15)22-12-5-3-2-4-11(12)19/h6,8,11-12H,2-5,7,19H2,1H3,(H,20,25)(H,22,23)/t11-,12+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 226 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda California, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant full length N-terminal GST-tagged PHKG2 expressed in baculovirus expression system by Z'-LYTE assay |
Bioorg Med Chem Lett 26: 5947-5950 (2016)
Article DOI: 10.1016/j.bmcl.2016.10.087
BindingDB Entry DOI: 10.7270/Q29888Z2 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
(Oryctolagus cuniculus (rabbit)) | BDBM2685
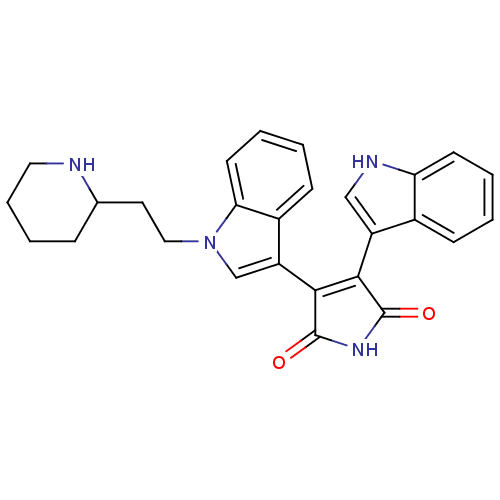
(3-(1H-indol-3-yl)-4-{1-[2-(piperidin-2-yl)ethyl]-1...)Show SMILES O=C1NC(=O)C(=C1c1c[nH]c2ccccc12)c1cn(CCC2CCCCN2)c2ccccc12 |c:5| Show InChI InChI=1S/C27H26N4O2/c32-26-24(20-15-29-22-10-3-1-8-18(20)22)25(27(33)30-26)21-16-31(23-11-4-2-9-19(21)23)14-12-17-7-5-6-13-28-17/h1-4,8-11,15-17,28-29H,5-7,12-14H2,(H,30,32,33) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratoires Glaxo
| Assay Description
The activity of PK is measured by its ability to transfer from [gamma-32P]ATP to phosphorylase b. |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
(Oryctolagus cuniculus (rabbit)) | BDBM2683
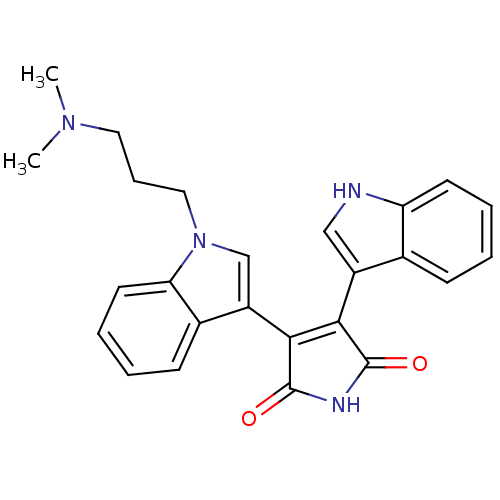
(2-[1-(3-dimethylaminopropyl)-indol-3-yl]-3-(indol-...)Show SMILES CN(C)CCCn1cc(C2=C(C(=O)NC2=O)c2c[nH]c3ccccc23)c2ccccc12 |t:9| Show InChI InChI=1S/C25H24N4O2/c1-28(2)12-7-13-29-15-19(17-9-4-6-11-21(17)29)23-22(24(30)27-25(23)31)18-14-26-20-10-5-3-8-16(18)20/h3-6,8-11,14-15,26H,7,12-13H2,1-2H3,(H,27,30,31) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 700 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratoires Glaxo
| Assay Description
The activity of PK is measured by its ability to transfer from [gamma-32P]ATP to phosphorylase b. |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
(Oryctolagus cuniculus (rabbit)) | BDBM2694
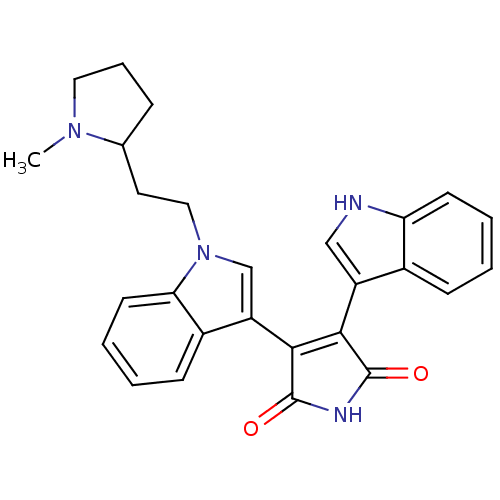
(3-(1H-indol-3-yl)-4-{1-[2-(1-methylpyrrolidin-2-yl...)Show SMILES CN1CCCC1CCn1cc(C2=C(C(=O)NC2=O)c2c[nH]c3ccccc23)c2ccccc12 |t:12| Show InChI InChI=1S/C27H26N4O2/c1-30-13-6-7-17(30)12-14-31-16-21(19-9-3-5-11-23(19)31)25-24(26(32)29-27(25)33)20-15-28-22-10-4-2-8-18(20)22/h2-5,8-11,15-17,28H,6-7,12-14H2,1H3,(H,29,32,33) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| n/a | n/a | 750 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratoires Glaxo
| Assay Description
The activity of PK is measured by its ability to transfer from [gamma-32P]ATP to phosphorylase b. |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM50462709
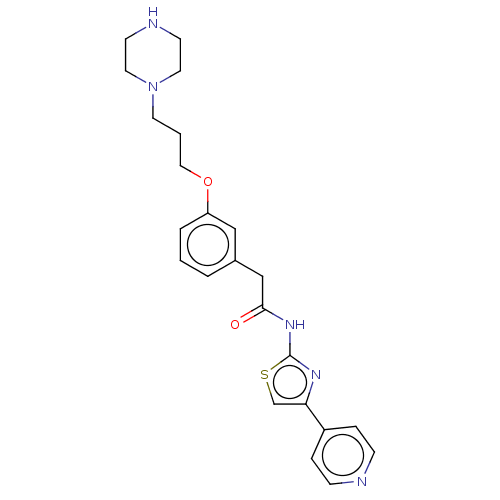
(CHEMBL4245507)Show SMILES O=C(Cc1cccc(OCCCN2CCNCC2)c1)Nc1nc(cs1)-c1ccncc1 Show InChI InChI=1S/C23H27N5O2S/c29-22(27-23-26-21(17-31-23)19-5-7-24-8-6-19)16-18-3-1-4-20(15-18)30-14-2-11-28-12-9-25-10-13-28/h1,3-8,15,17,25H,2,9-14,16H2,(H,26,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Vertex Pharmaceuticals, Inc
Curated by ChEMBL
| Assay Description
Inhibition of PHK-gamma2 (unknown origin) |
Bioorg Med Chem Lett 28: 2616-2621 (2018)
Article DOI: 10.1016/j.bmcl.2018.06.043
BindingDB Entry DOI: 10.7270/Q2XP77MH |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
(Oryctolagus cuniculus (rabbit)) | BDBM2692
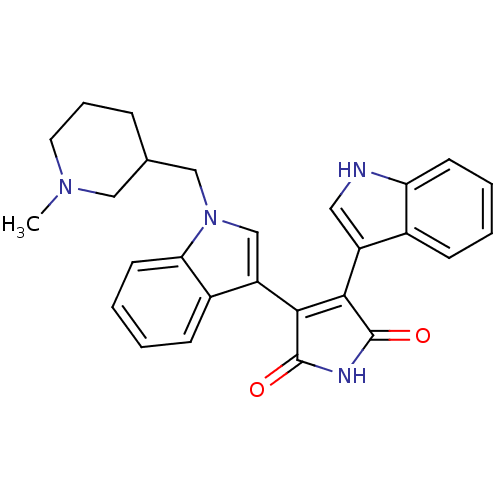
(3-(1H-indol-3-yl)-4-{1-[(1-methylpiperidin-3-yl)me...)Show SMILES CN1CCCC(Cn2cc(C3=C(C(=O)NC3=O)c3c[nH]c4ccccc34)c3ccccc23)C1 |t:10| Show InChI InChI=1S/C27H26N4O2/c1-30-12-6-7-17(14-30)15-31-16-21(19-9-3-5-11-23(19)31)25-24(26(32)29-27(25)33)20-13-28-22-10-4-2-8-18(20)22/h2-5,8-11,13,16-17,28H,6-7,12,14-15H2,1H3,(H,29,32,33) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratoires Glaxo
| Assay Description
The activity of PK is measured by its ability to transfer from [gamma-32P]ATP to phosphorylase b. |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
(Oryctolagus cuniculus (rabbit)) | BDBM2681
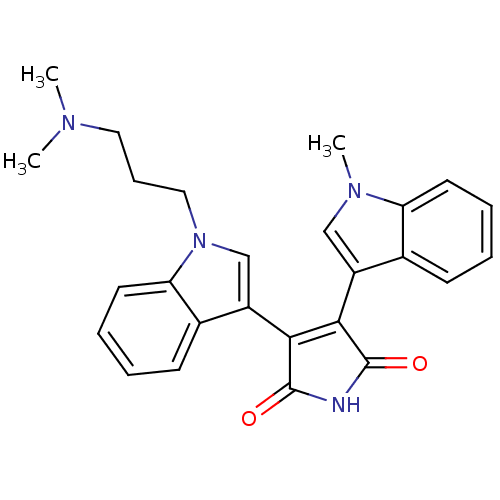
(3-{1-[3-(dimethylamino)propyl]-1H-indol-3-yl}-4-(1...)Show SMILES CN(C)CCCn1cc(C2=C(C(=O)NC2=O)c2cn(C)c3ccccc23)c2ccccc12 |t:9| Show InChI InChI=1S/C26H26N4O2/c1-28(2)13-8-14-30-16-20(18-10-5-7-12-22(18)30)24-23(25(31)27-26(24)32)19-15-29(3)21-11-6-4-9-17(19)21/h4-7,9-12,15-16H,8,13-14H2,1-3H3,(H,27,31,32) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.70E+3 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Laboratoires Glaxo
| Assay Description
IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
(Oryctolagus cuniculus (rabbit)) | BDBM2682
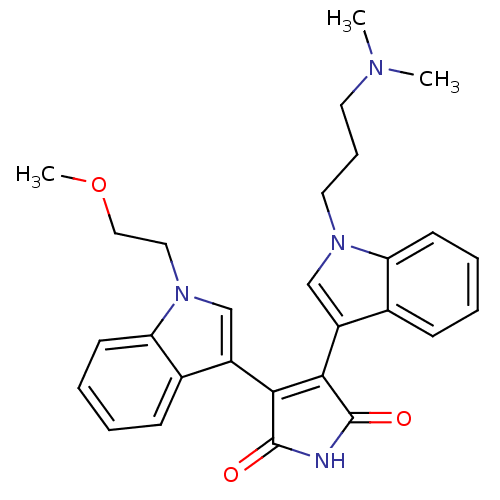
(3-{1-[3-(dimethylamino)propyl]-1H-indol-3-yl}-4-[1...)Show SMILES COCCn1cc(C2=C(C(=O)NC2=O)c2cn(CCCN(C)C)c3ccccc23)c2ccccc12 |t:7| Show InChI InChI=1S/C28H30N4O3/c1-30(2)13-8-14-31-17-21(19-9-4-6-11-23(19)31)25-26(28(34)29-27(25)33)22-18-32(15-16-35-3)24-12-7-5-10-20(22)24/h4-7,9-12,17-18H,8,13-16H2,1-3H3,(H,29,33,34) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.80E+3 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Laboratoires Glaxo
| Assay Description
IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
(Oryctolagus cuniculus (rabbit)) | BDBM2693
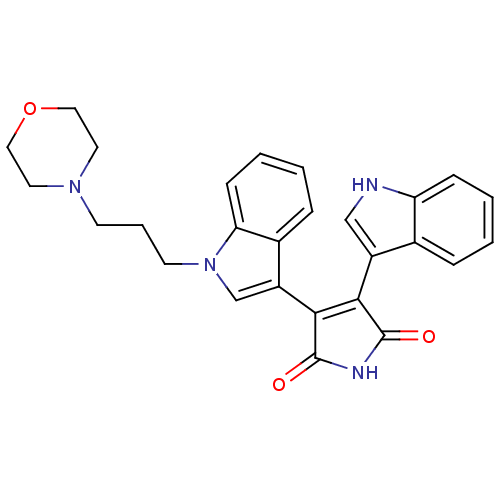
(3-(1H-indol-3-yl)-4-{1-[3-(morpholin-4-yl)propyl]-...)Show SMILES O=C1NC(=O)C(=C1c1c[nH]c2ccccc12)c1cn(CCCN2CCOCC2)c2ccccc12 |c:5| Show InChI InChI=1S/C27H26N4O3/c32-26-24(20-16-28-22-8-3-1-6-18(20)22)25(27(33)29-26)21-17-31(23-9-4-2-7-19(21)23)11-5-10-30-12-14-34-15-13-30/h1-4,6-9,16-17,28H,5,10-15H2,(H,29,32,33) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratoires Glaxo
| Assay Description
The activity of PK is measured by its ability to transfer from [gamma-32P]ATP to phosphorylase b. |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
(Oryctolagus cuniculus (rabbit)) | BDBM2690
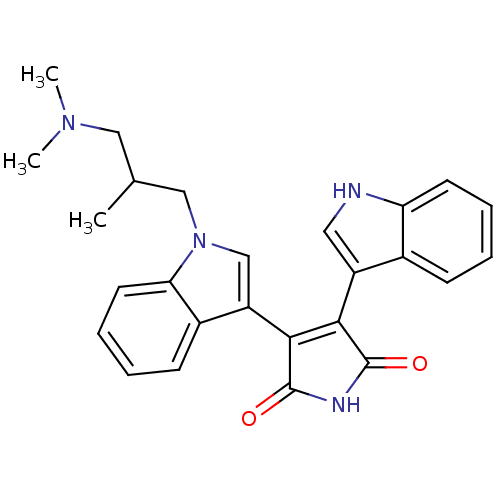
(3-(1-{2-[(dimethylamino)methyl]propyl}-1H-indol-3-...)Show SMILES CC(CN(C)C)Cn1cc(C2=C(C(=O)NC2=O)c2c[nH]c3ccccc23)c2ccccc12 |t:10| Show InChI InChI=1S/C26H26N4O2/c1-16(13-29(2)3)14-30-15-20(18-9-5-7-11-22(18)30)24-23(25(31)28-26(24)32)19-12-27-21-10-6-4-8-17(19)21/h4-12,15-16,27H,13-14H2,1-3H3,(H,28,31,32) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratoires Glaxo
| Assay Description
The activity of PK is measured by its ability to transfer from [gamma-32P]ATP to phosphorylase b. |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
(Oryctolagus cuniculus (rabbit)) | BDBM2689
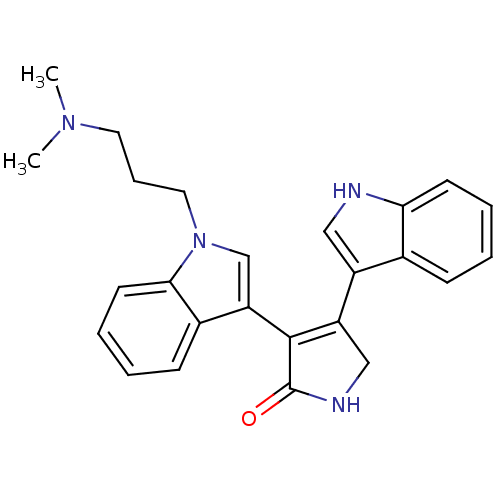
(3-[1-(3-dymethylaminopropyl)-indol-3-yl]-4-(indol-...)Show SMILES CN(C)CCCn1cc(C2=C(CNC2=O)c2c[nH]c3ccccc23)c2ccccc12 |t:9| Show InChI InChI=1S/C25H26N4O/c1-28(2)12-7-13-29-16-21(18-9-4-6-11-23(18)29)24-20(15-27-25(24)30)19-14-26-22-10-5-3-8-17(19)22/h3-6,8-11,14,16,26H,7,12-13,15H2,1-2H3,(H,27,30) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2.75E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratoires Glaxo
| Assay Description
The activity of PK is measured by its ability to transfer from [gamma-32P]ATP to phosphorylase b. |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
(Oryctolagus cuniculus (rabbit)) | BDBM2583
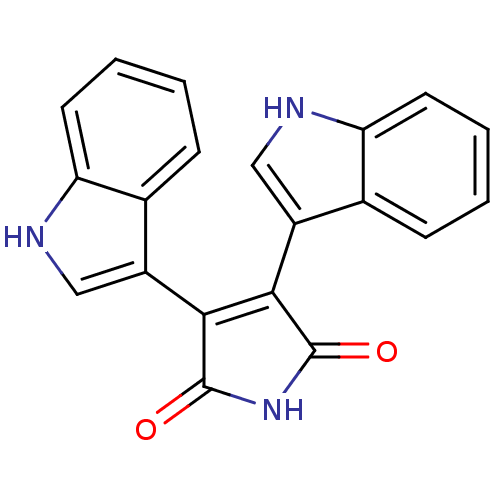
(3,4-Bis(3-indolyl)-1H-pyrrole-2,5-dione | 3,4-bis(...)Show SMILES O=C1NC(=O)C(=C1c1c[nH]c2ccccc12)c1c[nH]c2ccccc12 |c:5| Show InChI InChI=1S/C20H13N3O2/c24-19-17(13-9-21-15-7-3-1-5-11(13)15)18(20(25)23-19)14-10-22-16-8-4-2-6-12(14)16/h1-10,21-22H,(H,23,24,25) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a | 7.0 | 30 |
Laboratoires Glaxo
| Assay Description
IC50 is the inhibitor concentration which inhibits 50% of kinase activity that catalyzes the transfer of the terminal phosphate from [gamma-32P] labe... |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase regulatory subunit alpha, skeletal muscle isoform
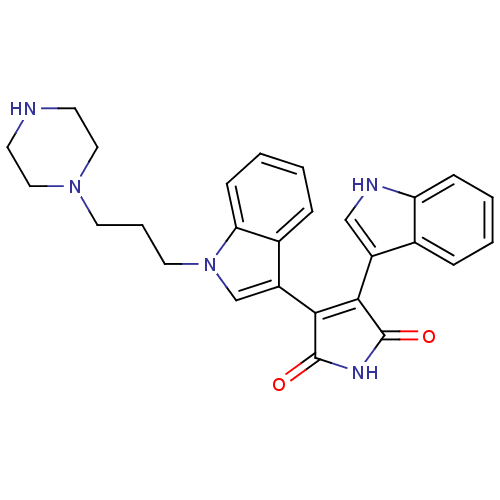
(Oryctolagus cuniculus (rabbit)) | BDBM2686
(3-(1H-indol-3-yl)-4-{1-[3-(piperazin-1-yl)propyl]-...)Show SMILES O=C1NC(=O)C(=C1c1c[nH]c2ccccc12)c1cn(CCCN2CCNCC2)c2ccccc12 |c:5| Show InChI InChI=1S/C27H27N5O2/c33-26-24(20-16-29-22-8-3-1-6-18(20)22)25(27(34)30-26)21-17-32(23-9-4-2-7-19(21)23)13-5-12-31-14-10-28-11-15-31/h1-4,6-9,16-17,28-29H,5,10-15H2,(H,30,33,34) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratoires Glaxo
| Assay Description
The activity of PK is measured by its ability to transfer from [gamma-32P]ATP to phosphorylase b. |
J Biol Chem 266: 15771-81 (1991)
BindingDB Entry DOI: 10.7270/Q2FF3QJ4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM50135286
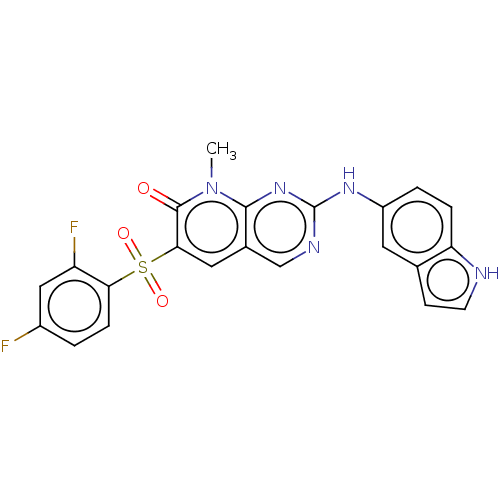
(CHEMBL3745885)Show SMILES Cn1c2nc(Nc3ccc4[nH]ccc4c3)ncc2cc(c1=O)S(=O)(=O)c1ccc(F)cc1F Show InChI InChI=1S/C22H15F2N5O3S/c1-29-20-13(9-19(21(29)30)33(31,32)18-5-2-14(23)10-16(18)24)11-26-22(28-20)27-15-3-4-17-12(8-15)6-7-25-17/h2-11,25H,1H3,(H,26,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Icahn School of Medicine at Mount Sinai
Curated by ChEMBL
| Assay Description
Inhibition of human PHKg2 using [KKLNRTLSFAEPG] as substrate |
Bioorg Med Chem 24: 521-44 (2016)
Article DOI: 10.1016/j.bmc.2015.11.045
BindingDB Entry DOI: 10.7270/Q24Q7WT8 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM50401152
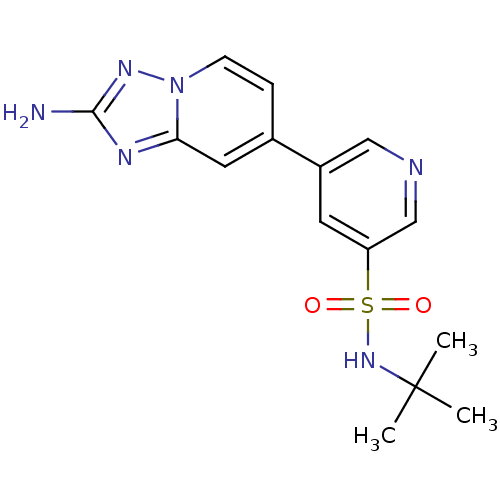
(CHEMBL2205766)Show SMILES CC(C)(C)NS(=O)(=O)c1cncc(c1)-c1ccn2nc(N)nc2c1 Show InChI InChI=1S/C15H18N6O2S/c1-15(2,3)20-24(22,23)12-6-11(8-17-9-12)10-4-5-21-13(7-10)18-14(16)19-21/h4-9,20H,1-3H3,(H2,16,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Cellzome Ltd
Curated by ChEMBL
| Assay Description
Inhibition of PHKG2 |
Bioorg Med Chem Lett 22: 4613-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.05.090
BindingDB Entry DOI: 10.7270/Q2HQ412B |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM50499634
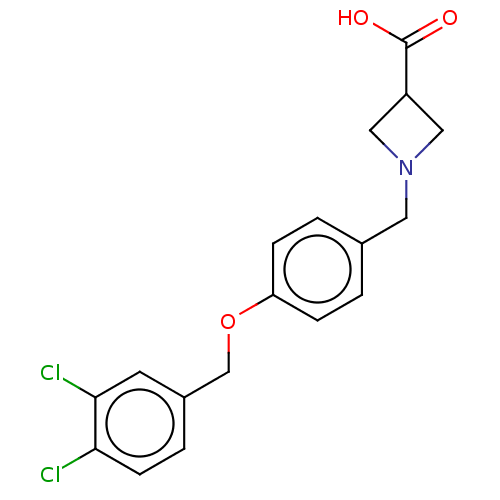
(CHEMBL3741589)Show SMILES OC(=O)C1CN(Cc2ccc(OCc3ccc(Cl)c(Cl)c3)cc2)C1 Show InChI InChI=1S/C18H17Cl2NO3/c19-16-6-3-13(7-17(16)20)11-24-15-4-1-12(2-5-15)8-21-9-14(10-21)18(22)23/h1-7,14H,8-11H2,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Bioresearch Center
Curated by ChEMBL
| Assay Description
Inhibition of PHKG2 (unknown origin) |
J Med Chem 58: 9154-70 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00928
BindingDB Entry DOI: 10.7270/Q2PZ5CV4 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM50135286

(CHEMBL3745885)Show SMILES Cn1c2nc(Nc3ccc4[nH]ccc4c3)ncc2cc(c1=O)S(=O)(=O)c1ccc(F)cc1F Show InChI InChI=1S/C22H15F2N5O3S/c1-29-20-13(9-19(21(29)30)33(31,32)18-5-2-14(23)10-16(18)24)11-26-22(28-20)27-15-3-4-17-12(8-15)6-7-25-17/h2-11,25H,1H3,(H,26,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Icahn School of Medicine at Mount Sinai
Curated by ChEMBL
| Assay Description
Inhibition of human PHKg1 using [KKLNRTLSFAEPG] as substrate |
Bioorg Med Chem 24: 521-44 (2016)
Article DOI: 10.1016/j.bmc.2015.11.045
BindingDB Entry DOI: 10.7270/Q24Q7WT8 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM50519662
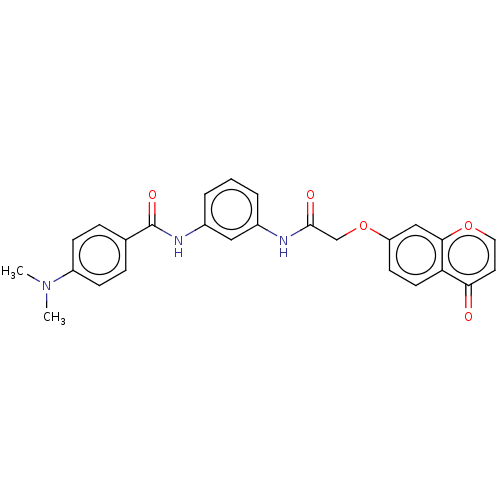
(CHEMBL4438748)Show SMILES CN(C)c1ccc(cc1)C(=O)Nc1cccc(NC(=O)COc2ccc3c(c2)occc3=O)c1 Show InChI InChI=1S/C26H23N3O5/c1-29(2)20-8-6-17(7-9-20)26(32)28-19-5-3-4-18(14-19)27-25(31)16-34-21-10-11-22-23(30)12-13-33-24(22)15-21/h3-15H,16H2,1-2H3,(H,27,31)(H,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant full-length human PhKgamma1 using KKLNRTLSFAEPG as substrate incubated for 40 mins in presence of [gamma33P]ATP by radiomet... |
J Med Chem 62: 10691-10710 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01143
BindingDB Entry DOI: 10.7270/Q2MC93FG |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM13533

(1-[2-(4-methylphenyl)-5-tert-butyl-pyrazol-3-yl]-3...)Show SMILES Cc1ccc(cc1)-n1nc(cc1NC(=O)Nc1ccc(OCCN2CCOCC2)c2ccccc12)C(C)(C)C Show InChI InChI=1S/C31H37N5O3/c1-22-9-11-23(12-10-22)36-29(21-28(34-36)31(2,3)4)33-30(37)32-26-13-14-27(25-8-6-5-7-24(25)26)39-20-17-35-15-18-38-19-16-35/h5-14,21H,15-20H2,1-4H3,(H2,32,33,37) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Ansaris
Curated by ChEMBL
| Assay Description
Inhibition of PHKgamma2 |
Bioorg Med Chem Lett 21: 7155-65 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.078
BindingDB Entry DOI: 10.7270/Q2NC61NH |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM50359359

(CHEMBL1929238)Show SMILES CN(C)CCN1CCN(CCC1=O)C(=O)c1cc(sc1NC(=O)Nc1cccc(Cl)c1Cl)C(C)(C)C Show InChI InChI=1S/C25H33Cl2N5O3S/c1-25(2,3)19-15-16(22(36-19)29-24(35)28-18-8-6-7-17(26)21(18)27)23(34)32-10-9-20(33)31(13-14-32)12-11-30(4)5/h6-8,15H,9-14H2,1-5H3,(H2,28,29,35) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Ansaris
Curated by ChEMBL
| Assay Description
Inhibition of PHKgamma2 |
Bioorg Med Chem Lett 21: 7155-65 (2011)
Article DOI: 10.1016/j.bmcl.2011.09.078
BindingDB Entry DOI: 10.7270/Q2NC61NH |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM50537742
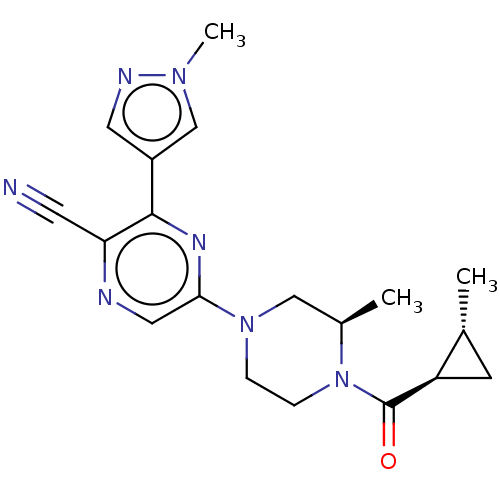
(CHEMBL4634634 | US11179389, Compound 1-14)Show SMILES C[C@@H]1C[C@H]1C(=O)N1CCN(C[C@H]1C)c1cnc(C#N)c(n1)-c1cnn(C)c1 |r| Show InChI InChI=1S/C19H23N7O/c1-12-6-15(12)19(27)26-5-4-25(10-13(26)2)17-9-21-16(7-20)18(23-17)14-8-22-24(3)11-14/h8-9,11-13,15H,4-6,10H2,1-3H3/t12-,13-,15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant full length human GST-tagged PHKG1 expressed in baculovirus expression system using serine/threonine-7 peptide as substrate... |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2019.126715
BindingDB Entry DOI: 10.7270/Q2HM5CZG |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM50537742

(CHEMBL4634634 | US11179389, Compound 1-14)Show SMILES C[C@@H]1C[C@H]1C(=O)N1CCN(C[C@H]1C)c1cnc(C#N)c(n1)-c1cnn(C)c1 |r| Show InChI InChI=1S/C19H23N7O/c1-12-6-15(12)19(27)26-5-4-25(10-13(26)2)17-9-21-16(7-20)18(23-17)14-8-22-24(3)11-14/h8-9,11-13,15H,4-6,10H2,1-3H3/t12-,13-,15-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | >1.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant full length human N-terminal GST-tagged PHKG2 expressed in baculovirus expression system using serine/threonine-7 peptide a... |
Bioorg Med Chem Lett 30: (2020)
Article DOI: 10.1016/j.bmcl.2019.126715
BindingDB Entry DOI: 10.7270/Q2HM5CZG |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM5931
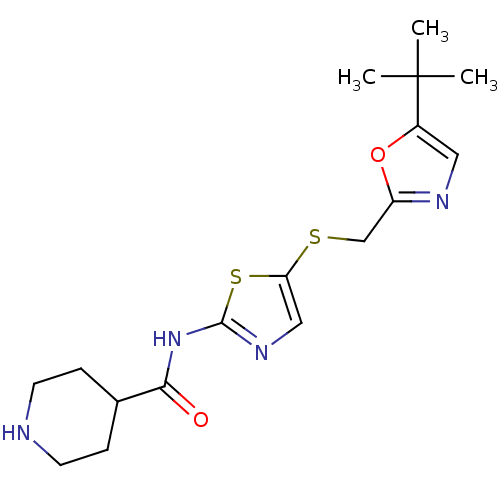
(BMS-387072 | CHEMBL296468 | N-(5-{[(5-tert-butyl-1...)Show InChI InChI=1S/C17H24N4O2S2/c1-17(2,3)12-8-19-13(23-12)10-24-14-9-20-16(25-14)21-15(22)11-4-6-18-7-5-11/h8-9,11,18H,4-7,10H2,1-3H3,(H,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 3.80E+3 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q22R3Q13 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM25118
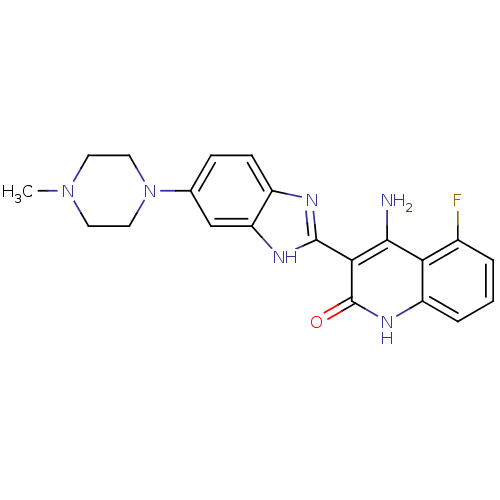
((3Z)-4-amino-5-fluoro-3-[5-(4-methylpiperazino)-1,...)Show SMILES CN1CCN(CC1)c1ccc2nc([nH]c2c1)-c1c(N)c2c(F)cccc2[nH]c1=O Show InChI InChI=1S/C21H21FN6O/c1-27-7-9-28(10-8-27)12-5-6-14-16(11-12)25-20(24-14)18-19(23)17-13(22)3-2-4-15(17)26-21(18)29/h2-6,11H,7-10H2,1H3,(H,24,25)(H3,23,26,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 640 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q22R3Q13 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM31090
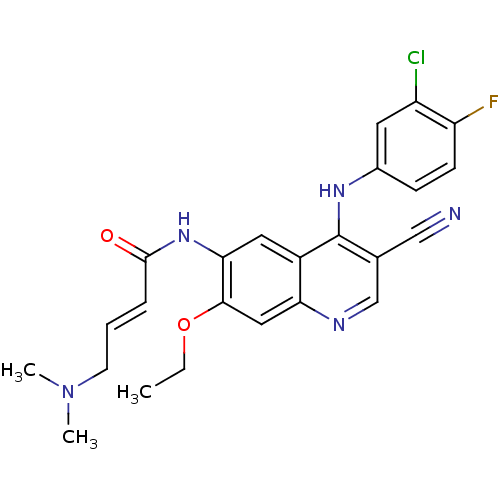
((E)-N-[4-(3-chloro-4-fluoro-anilino)-3-cyano-7-eth...)Show SMILES CCOc1cc2ncc(C#N)c(Nc3ccc(F)c(Cl)c3)c2cc1NC(=O)\C=C\CN(C)C Show InChI InChI=1S/C24H23ClFN5O2/c1-4-33-22-12-20-17(11-21(22)30-23(32)6-5-9-31(2)3)24(15(13-27)14-28-20)29-16-7-8-19(26)18(25)10-16/h5-8,10-12,14H,4,9H2,1-3H3,(H,28,29)(H,30,32)/b6-5+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 3.50E+3 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q22R3Q13 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM31099
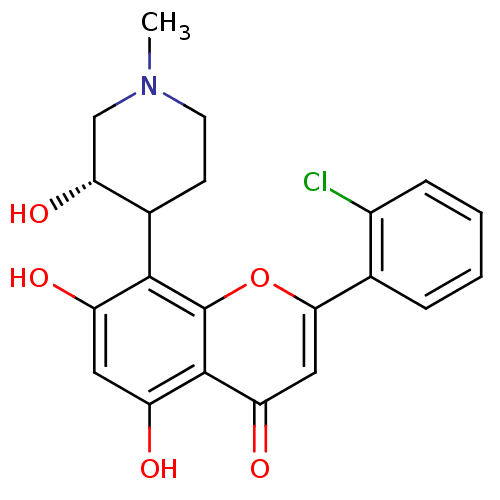
(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S)-3-hydroxy...)Show SMILES CN1CCC([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12?,17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q22R3Q13 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM17055
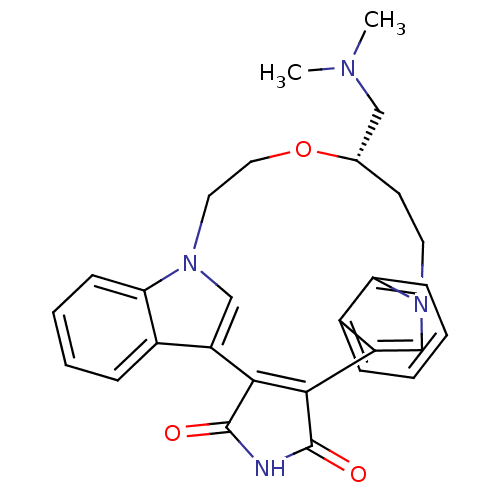
((18S)-18-[(dimethylamino)methyl]-17-oxa-4,14,21-tr...)Show SMILES CN(C)C[C@@H]1CCn2cc(C3=C(C(=O)NC3=O)c3cn(CCO1)c1ccccc31)c1ccccc21 |r,t:10| Show InChI InChI=1S/C28H28N4O3/c1-30(2)15-18-11-12-31-16-21(19-7-3-5-9-23(19)31)25-26(28(34)29-27(25)33)22-17-32(13-14-35-18)24-10-6-4-8-20(22)24/h3-10,16-18H,11-15H2,1-2H3,(H,29,33,34)/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 3.70E+3 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q22R3Q13 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM31094
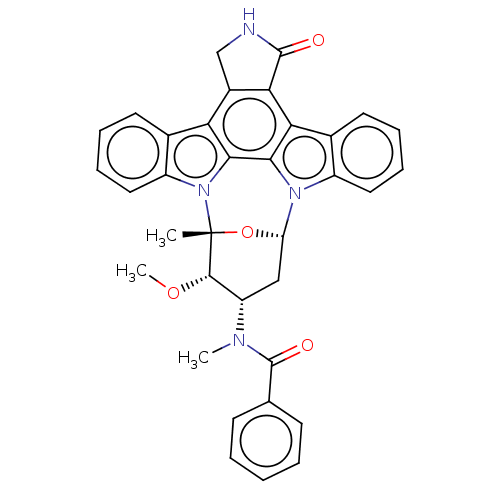
(PKC-412 | cid_24202429)Show SMILES [H][C@@]12C[C@@H]([C@H](OC)[C@@](C)(O1)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13)N(C)C(=O)c1ccccc1 |TLB:11:10:9:4.3.2,17:33:9:4.3.2,THB:24:32:9:4.3.2,5:4:9:10.31.33.32,30:31:9:4.3.2| Show InChI InChI=1S/C35H30N4O4/c1-35-32(42-3)25(37(2)34(41)19-11-5-4-6-12-19)17-26(43-35)38-23-15-9-7-13-20(23)28-29-22(18-36-33(29)40)27-21-14-8-10-16-24(21)39(35)31(27)30(28)38/h4-16,25-26,32H,17-18H2,1-3H3,(H,36,40)/t25-,26-,32-,35+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 900 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q22R3Q13 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM31095
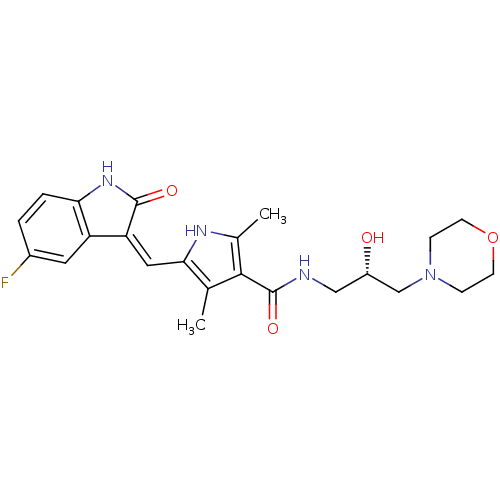
(5-[(Z)-(5-fluoranyl-2-oxidanylidene-1H-indol-3-yli...)Show SMILES Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1C(=O)NC[C@H](O)CN1CCOCC1 Show InChI InChI=1S/C23H27FN4O4/c1-13-20(10-18-17-9-15(24)3-4-19(17)27-22(18)30)26-14(2)21(13)23(31)25-11-16(29)12-28-5-7-32-8-6-28/h3-4,9-10,16,26,29H,5-8,11-12H2,1-2H3,(H,25,31)(H,27,30)/b18-10-/t16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 83 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q22R3Q13 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM31096
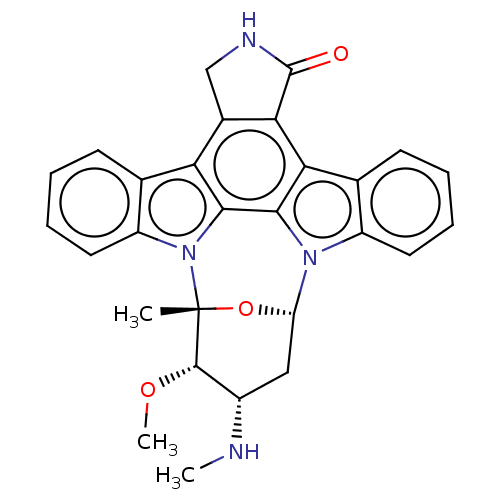
(CHEMBL290084 | Staurosporine | cid_451705)Show SMILES CN[C@H]1C[C@@H]2O[C@](C)([C@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 0.650 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q22R3Q13 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM4814
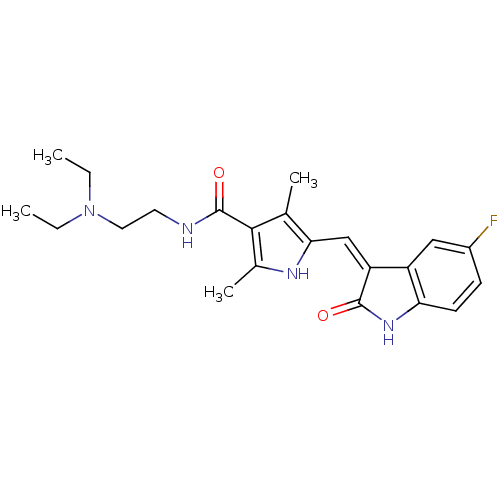
(CHEMBL535 | N-[2-(diethylamino)ethyl]-5-[(Z)-(5-fl...)Show SMILES CCN(CC)CCNC(=O)c1c(C)[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c1C Show InChI InChI=1S/C22H27FN4O2/c1-5-27(6-2)10-9-24-22(29)20-13(3)19(25-14(20)4)12-17-16-11-15(23)7-8-18(16)26-21(17)28/h7-8,11-12,25H,5-6,9-10H2,1-4H3,(H,24,29)(H,26,28)/b17-12- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q22R3Q13 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM13534

(CHEMBL572878 | N-[4-({4-[(3-methyl-1H-pyrazol-5-yl...)Show SMILES CN1CCN(CC1)c1cc(Nc2cc(C)n[nH]2)nc(Sc2ccc(NC(=O)C3CC3)cc2)n1 Show InChI InChI=1S/C23H28N8OS/c1-15-13-20(29-28-15)25-19-14-21(31-11-9-30(2)10-12-31)27-23(26-19)33-18-7-5-17(6-8-18)24-22(32)16-3-4-16/h5-8,13-14,16H,3-4,9-12H2,1-2H3,(H,24,32)(H2,25,26,27,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 2.20E+3 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q22R3Q13 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, skeletal muscle/heart isoform
(Homo sapiens (Human)) | BDBM21
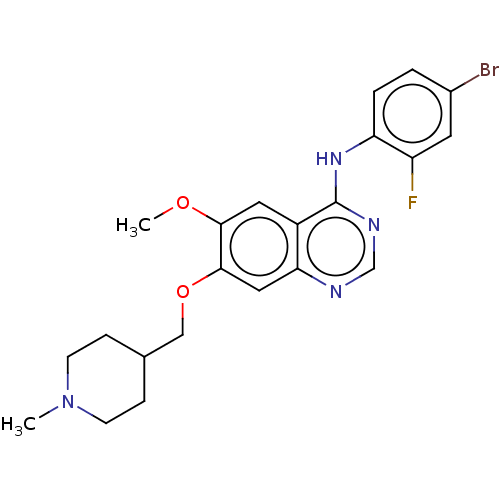
(CHEMBL24828 | N-(4-bromo-2-fluorophenyl)-6-methoxy...)Show SMILES COc1cc2c(Nc3ccc(Br)cc3F)ncnc2cc1OCC1CCN(C)CC1 Show InChI InChI=1S/C22H24BrFN4O2/c1-28-7-5-14(6-8-28)12-30-21-11-19-16(10-20(21)29-2)22(26-13-25-19)27-18-4-3-15(23)9-17(18)24/h3-4,9-11,13-14H,5-8,12H2,1-2H3,(H,25,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PCBioAssay
| n/a | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q22R3Q13 |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM25118

((3Z)-4-amino-5-fluoro-3-[5-(4-methylpiperazino)-1,...)Show SMILES CN1CCN(CC1)c1ccc2nc([nH]c2c1)-c1c(N)c2c(F)cccc2[nH]c1=O Show InChI InChI=1S/C21H21FN6O/c1-27-7-9-28(10-8-27)12-5-6-14-16(11-12)25-20(24-14)18-19(23)17-13(22)3-2-4-15(17)26-21(18)29/h2-6,11H,7-10H2,1H3,(H,24,25)(H3,23,26,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 9.00E+3 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q26D5RCJ |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM31099

(2-(2-chlorophenyl)-5,7-dihydroxy-8-[(3S)-3-hydroxy...)Show SMILES CN1CCC([C@H](O)C1)c1c(O)cc(O)c2c1oc(cc2=O)-c1ccccc1Cl Show InChI InChI=1S/C21H20ClNO5/c1-23-7-6-12(17(27)10-23)19-14(24)8-15(25)20-16(26)9-18(28-21(19)20)11-4-2-3-5-13(11)22/h2-5,8-9,12,17,24-25,27H,6-7,10H2,1H3/t12?,17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q26D5RCJ |
More data for this
Ligand-Target Pair | |
Phosphorylase b kinase gamma catalytic chain, liver/testis isoform
(Homo sapiens (Human)) | BDBM31095

(5-[(Z)-(5-fluoranyl-2-oxidanylidene-1H-indol-3-yli...)Show SMILES Cc1[nH]c(\C=C2/C(=O)Nc3ccc(F)cc23)c(C)c1C(=O)NC[C@H](O)CN1CCOCC1 Show InChI InChI=1S/C23H27FN4O4/c1-13-20(10-18-17-9-15(24)3-4-19(17)27-22(18)30)26-14(2)21(13)23(31)25-11-16(29)12-28-5-7-32-8-6-28/h3-4,9-10,16,26,29H,5-8,11-12H2,1-2H3,(H,25,31)(H,27,30)/b18-10-/t16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PCBioAssay
| n/a | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a |
Ambit Biosciences
Curated by PubChem BioAssay
| Assay Description
Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity... |
PubChem Bioassay (2008)
BindingDB Entry DOI: 10.7270/Q26D5RCJ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data