 Found 60 hits of Enzyme Inhibition Constant Data
Found 60 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM21625
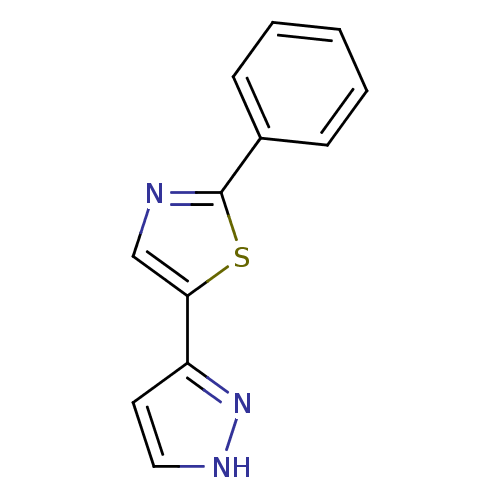
(2-phenyl-5-(1H-pyrazol-3-yl)-1,3-thiazole | 2-phen...)Show InChI InChI=1S/C12H9N3S/c1-2-4-9(5-3-1)12-13-8-11(16-12)10-6-7-14-15-10/h1-8H,(H,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084155
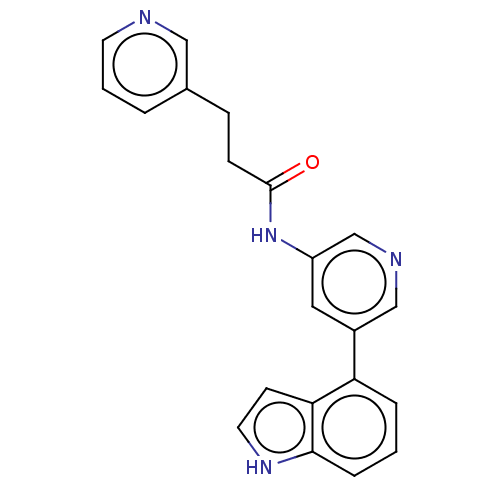
(CHEMBL3425953)Show SMILES O=C(CCc1cccnc1)Nc1cncc(c1)-c1cccc2[nH]ccc12 Show InChI InChI=1S/C21H18N4O/c26-21(7-6-15-3-2-9-22-12-15)25-17-11-16(13-23-14-17)18-4-1-5-20-19(18)8-10-24-20/h1-5,8-14,24H,6-7H2,(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084258
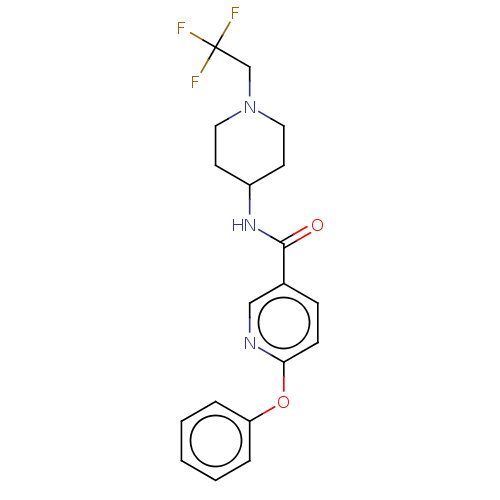
(CHEMBL3425959)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(Oc2ccccc2)nc1 Show InChI InChI=1S/C19H20F3N3O2/c20-19(21,22)13-25-10-8-15(9-11-25)24-18(26)14-6-7-17(23-12-14)27-16-4-2-1-3-5-16/h1-7,12,15H,8-11,13H2,(H,24,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084156
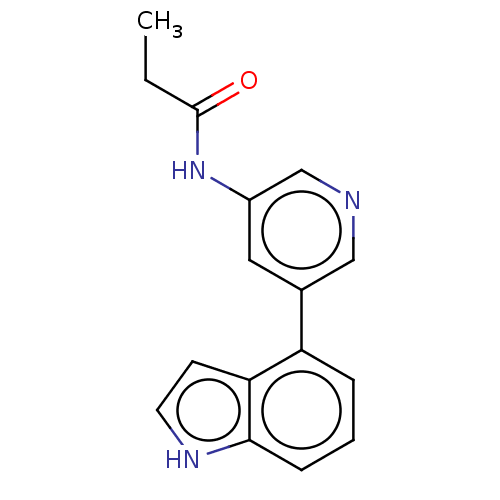
(CHEMBL3425954)Show InChI InChI=1S/C16H15N3O/c1-2-16(20)19-12-8-11(9-17-10-12)13-4-3-5-15-14(13)6-7-18-15/h3-10,18H,2H2,1H3,(H,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084157
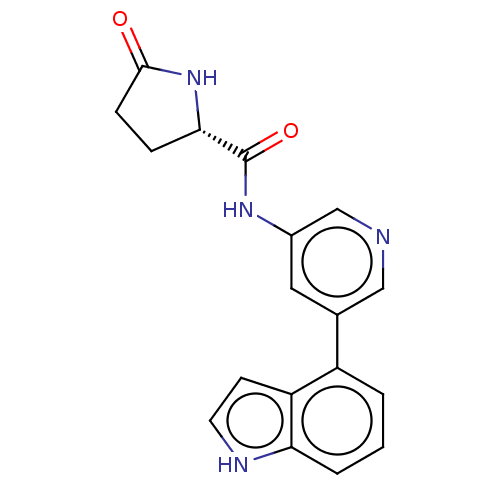
(CHEMBL3425955)Show SMILES O=C(Nc1cncc(c1)-c1cccc2[nH]ccc12)[C@@H]1CCC(=O)N1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084255
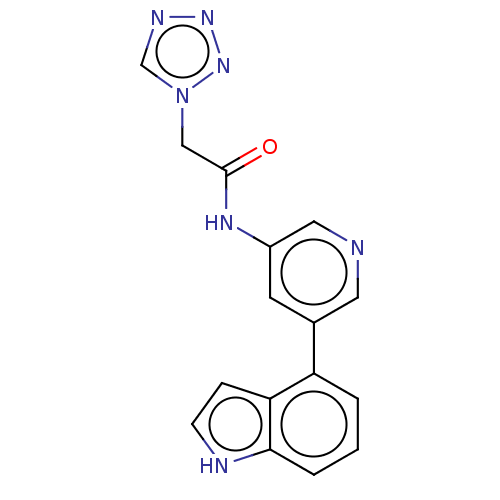
(CHEMBL3425956)Show InChI InChI=1S/C16H13N7O/c24-16(9-23-10-19-21-22-23)20-12-6-11(7-17-8-12)13-2-1-3-15-14(13)4-5-18-15/h1-8,10,18H,9H2,(H,20,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084259
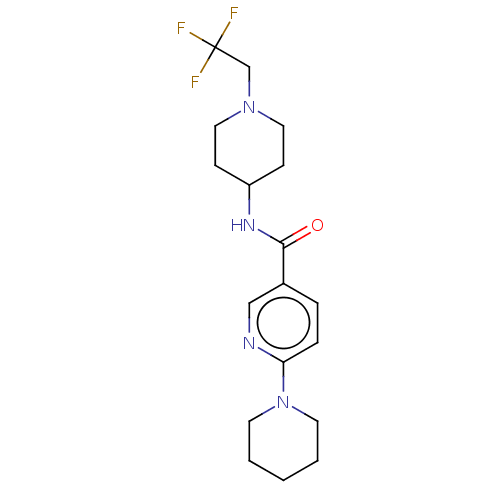
(CHEMBL3425960)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(nc1)N1CCCCC1 Show InChI InChI=1S/C18H25F3N4O/c19-18(20,21)13-24-10-6-15(7-11-24)23-17(26)14-4-5-16(22-12-14)25-8-2-1-3-9-25/h4-5,12,15H,1-3,6-11,13H2,(H,23,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084153
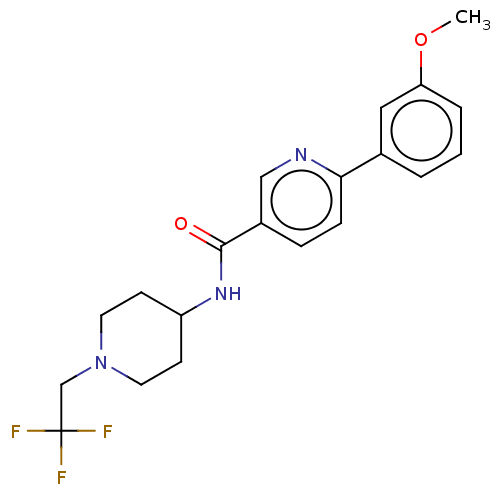
(CHEMBL3425948)Show SMILES COc1cccc(c1)-c1ccc(cn1)C(=O)NC1CCN(CC(F)(F)F)CC1 Show InChI InChI=1S/C20H22F3N3O2/c1-28-17-4-2-3-14(11-17)18-6-5-15(12-24-18)19(27)25-16-7-9-26(10-8-16)13-20(21,22)23/h2-6,11-12,16H,7-10,13H2,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084152

(CHEMBL3425949)Show SMILES CC(C)(O)c1nc(no1)-c1ccc(CNC(=O)c2cnc(nc2)-c2ccccn2)cc1 Show InChI InChI=1S/C22H20N6O3/c1-22(2,30)21-27-18(28-31-21)15-8-6-14(7-9-15)11-26-20(29)16-12-24-19(25-13-16)17-5-3-4-10-23-17/h3-10,12-13,30H,11H2,1-2H3,(H,26,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084265
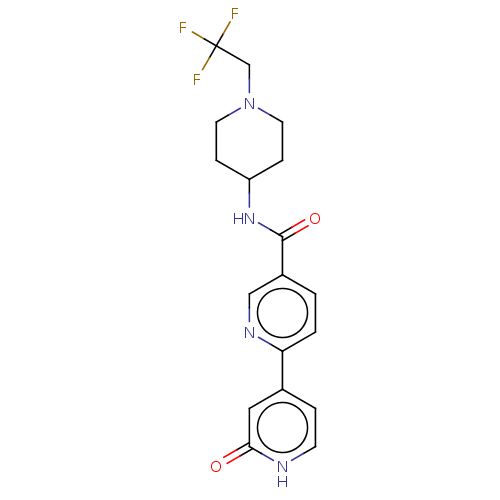
(CHEMBL3425966)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(nc1)-c1cc[nH]c(=O)c1 Show InChI InChI=1S/C18H19F3N4O2/c19-18(20,21)11-25-7-4-14(5-8-25)24-17(27)13-1-2-15(23-10-13)12-3-6-22-16(26)9-12/h1-3,6,9-10,14H,4-5,7-8,11H2,(H,22,26)(H,24,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084267
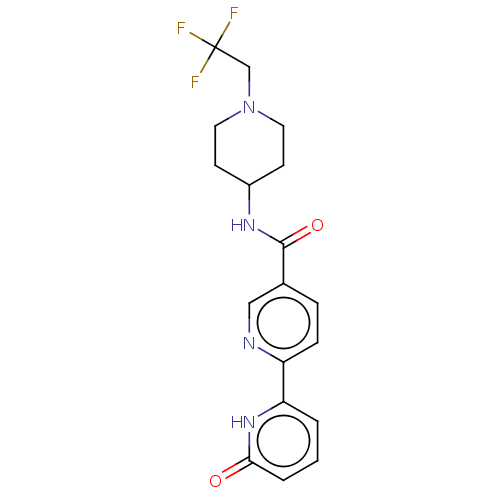
(CHEMBL3425968)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(nc1)-c1cccc(=O)[nH]1 Show InChI InChI=1S/C18H19F3N4O2/c19-18(20,21)11-25-8-6-13(7-9-25)23-17(27)12-4-5-14(22-10-12)15-2-1-3-16(26)24-15/h1-5,10,13H,6-9,11H2,(H,23,27)(H,24,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 71 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084256
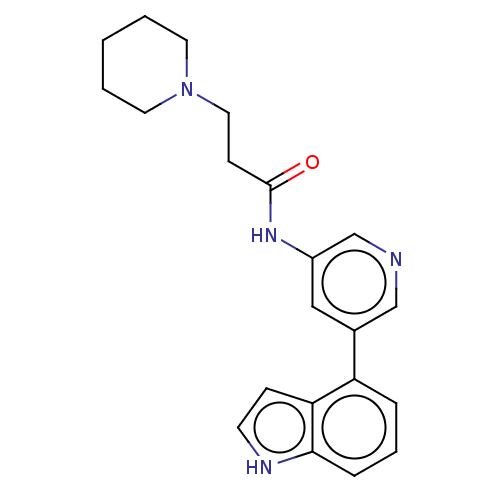
(CHEMBL3425957)Show SMILES O=C(CCN1CCCCC1)Nc1cncc(c1)-c1cccc2[nH]ccc12 Show InChI InChI=1S/C21H24N4O/c26-21(8-12-25-10-2-1-3-11-25)24-17-13-16(14-22-15-17)18-5-4-6-20-19(18)7-9-23-20/h4-7,9,13-15,23H,1-3,8,10-12H2,(H,24,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084149
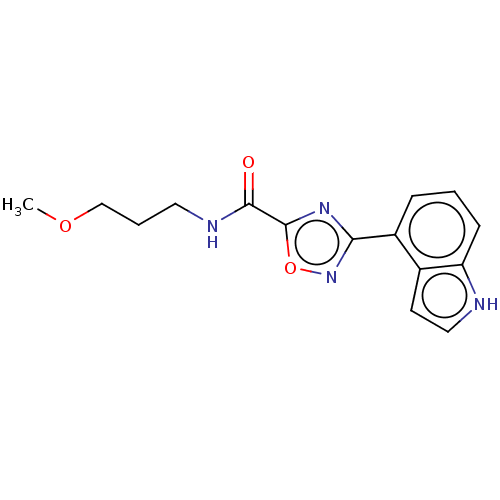
(CHEMBL3425952)Show InChI InChI=1S/C15H16N4O3/c1-21-9-3-7-17-14(20)15-18-13(19-22-15)11-4-2-5-12-10(11)6-8-16-12/h2,4-6,8,16H,3,7,9H2,1H3,(H,17,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084151
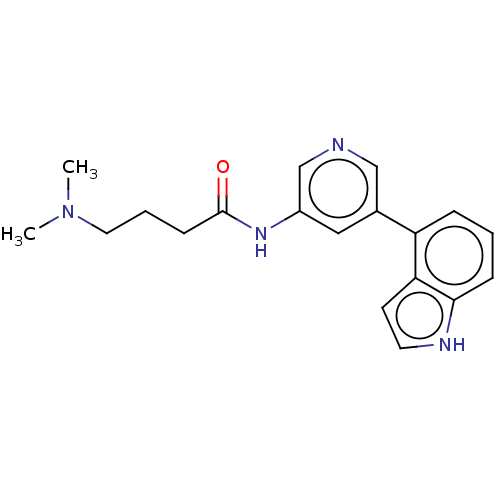
(CHEMBL3425950)Show InChI InChI=1S/C19H22N4O/c1-23(2)10-4-7-19(24)22-15-11-14(12-20-13-15)16-5-3-6-18-17(16)8-9-21-18/h3,5-6,8-9,11-13,21H,4,7,10H2,1-2H3,(H,22,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084154
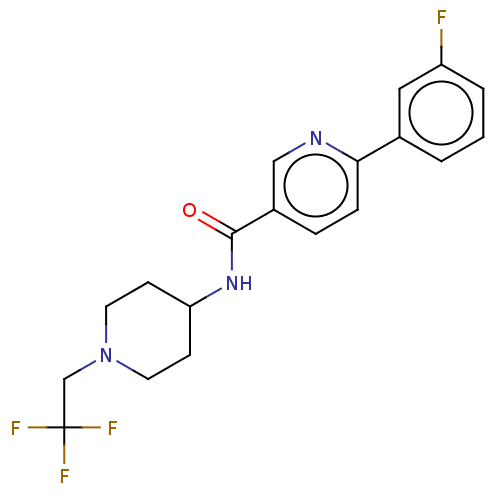
(CHEMBL1233897)Show SMILES Fc1cccc(c1)-c1ccc(cn1)C(=O)NC1CCN(CC(F)(F)F)CC1 Show InChI InChI=1S/C19H19F4N3O/c20-15-3-1-2-13(10-15)17-5-4-14(11-24-17)18(27)25-16-6-8-26(9-7-16)12-19(21,22)23/h1-5,10-11,16H,6-9,12H2,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084153

(CHEMBL3425948)Show SMILES COc1cccc(c1)-c1ccc(cn1)C(=O)NC1CCN(CC(F)(F)F)CC1 Show InChI InChI=1S/C20H22F3N3O2/c1-28-17-4-2-3-14(11-17)18-6-5-15(12-24-18)19(27)25-16-7-9-26(10-8-16)13-20(21,22)23/h2-6,11-12,16H,7-10,13H2,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition fluorescein-conjugated ligand binding to human HPGDS expressed in Escherichia coli by fluorescence polarization assay |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084260

(CHEMBL3425961)Show InChI InChI=1S/C17H23F3N4O/c18-17(19,20)12-23-9-5-14(6-10-23)22-16(25)13-3-4-15(21-11-13)24-7-1-2-8-24/h3-4,11,14H,1-2,5-10,12H2,(H,22,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084150
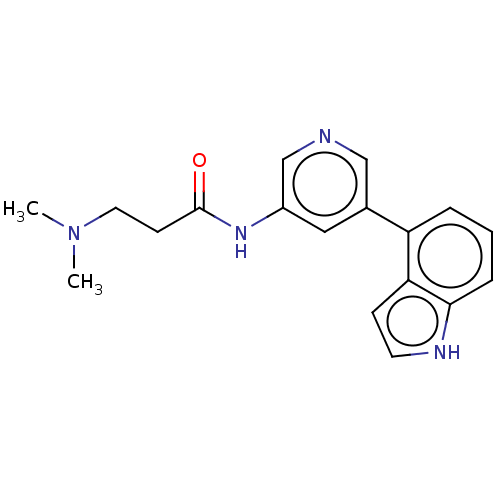
(CHEMBL3425951)Show InChI InChI=1S/C18H20N4O/c1-22(2)9-7-18(23)21-14-10-13(11-19-12-14)15-4-3-5-17-16(15)6-8-20-17/h3-6,8,10-12,20H,7,9H2,1-2H3,(H,21,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084262
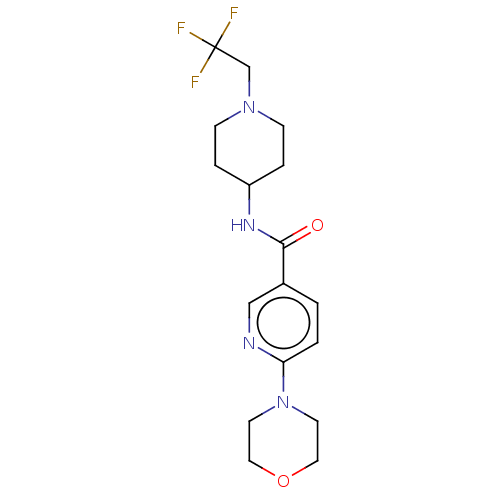
(CHEMBL3425963)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(nc1)N1CCOCC1 Show InChI InChI=1S/C17H23F3N4O2/c18-17(19,20)12-23-5-3-14(4-6-23)22-16(25)13-1-2-15(21-11-13)24-7-9-26-10-8-24/h1-2,11,14H,3-10,12H2,(H,22,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084150

(CHEMBL3425951)Show InChI InChI=1S/C18H20N4O/c1-22(2)9-7-18(23)21-14-10-13(11-19-12-14)15-4-3-5-17-16(15)6-8-20-17/h3-6,8,10-12,20H,7,9H2,1-2H3,(H,21,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 460 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition fluorescein-conjugated ligand binding to human HPGDS expressed in Escherichia coli by fluorescence polarization assay |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084264
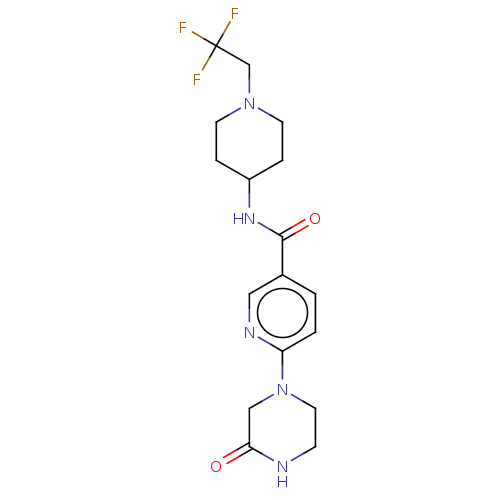
(CHEMBL3425965)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(nc1)N1CCNC(=O)C1 Show InChI InChI=1S/C17H22F3N5O2/c18-17(19,20)11-24-6-3-13(4-7-24)23-16(27)12-1-2-14(22-9-12)25-8-5-21-15(26)10-25/h1-2,9,13H,3-8,10-11H2,(H,21,26)(H,23,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 590 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084151

(CHEMBL3425950)Show InChI InChI=1S/C19H22N4O/c1-23(2)10-4-7-19(24)22-15-11-14(12-20-13-15)16-5-3-6-18-17(16)8-9-21-18/h3,5-6,8-9,11-13,21H,4,7,10H2,1-2H3,(H,22,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 820 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition fluorescein-conjugated ligand binding to human HPGDS expressed in Escherichia coli by fluorescence polarization assay |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084257
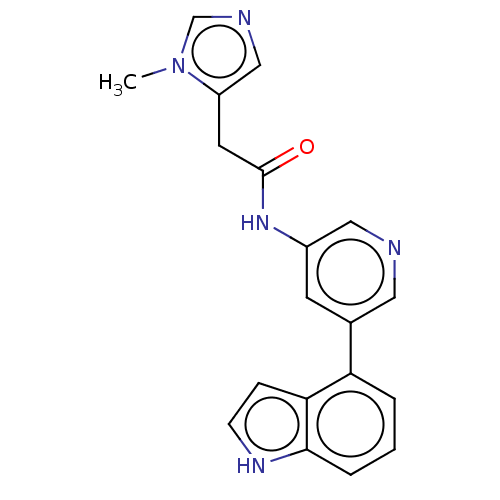
(CHEMBL3425958)Show InChI InChI=1S/C19H17N5O/c1-24-12-21-11-15(24)8-19(25)23-14-7-13(9-20-10-14)16-3-2-4-18-17(16)5-6-22-18/h2-7,9-12,22H,8H2,1H3,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition human HPGDS expressed in Escherichia coli assessed as reduction in GST enzymatic activity using MCBL and glutathione incubated for 30 mins... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084149

(CHEMBL3425952)Show InChI InChI=1S/C15H16N4O3/c1-21-9-3-7-17-14(20)15-18-13(19-22-15)11-4-2-5-12-10(11)6-8-16-12/h2,4-6,8,16H,3,7,9H2,1H3,(H,17,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition fluorescein-conjugated ligand binding to human HPGDS expressed in Escherichia coli by fluorescence polarization assay |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM21615
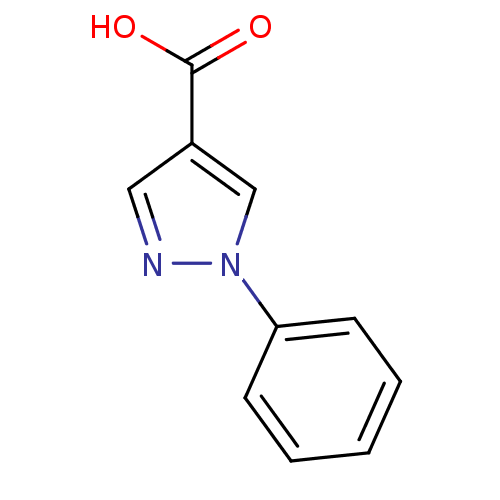
(1-phenyl-1H-pyrazole-4-carboxylic acid | Pyrazole,...)Show InChI InChI=1S/C10H8N2O2/c13-10(14)8-6-11-12(7-8)9-4-2-1-3-5-9/h1-7H,(H,13,14) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 1.40E+5 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli by isothermal titration calorimetry |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084264

(CHEMBL3425965)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(nc1)N1CCNC(=O)C1 Show InChI InChI=1S/C17H22F3N5O2/c18-17(19,20)11-24-6-3-13(4-7-24)23-16(27)12-1-2-14(22-9-12)25-8-5-21-15(26)10-25/h1-2,9,13H,3-8,10-11H2,(H,21,26)(H,23,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084154

(CHEMBL1233897)Show SMILES Fc1cccc(c1)-c1ccc(cn1)C(=O)NC1CCN(CC(F)(F)F)CC1 Show InChI InChI=1S/C19H19F4N3O/c20-15-3-1-2-13(10-15)17-5-4-14(11-24-17)18(27)25-16-6-8-26(9-7-16)12-19(21,22)23/h1-5,10-11,16H,6-9,12H2,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | 9.5 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084267

(CHEMBL3425968)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(nc1)-c1cccc(=O)[nH]1 Show InChI InChI=1S/C18H19F3N4O2/c19-18(20,21)11-25-8-6-13(7-9-25)23-17(27)12-4-5-14(22-10-12)15-2-1-3-16(26)24-15/h1-5,10,13H,6-9,11H2,(H,23,27)(H,24,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084269
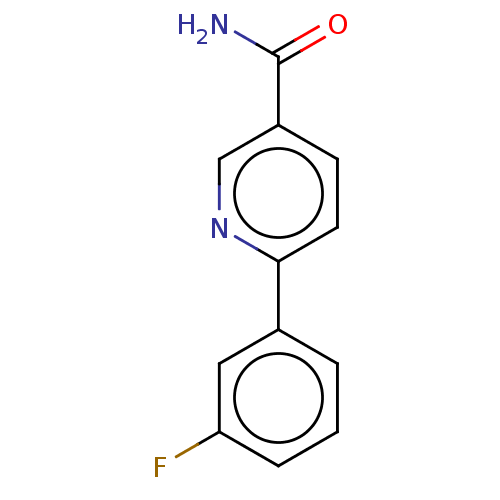
(CHEMBL3425970)Show InChI InChI=1S/C12H9FN2O/c13-10-3-1-2-8(6-10)11-5-4-9(7-15-11)12(14)16/h1-7H,(H2,14,16) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 800 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli by isothermal titration calorimetry |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM21625

(2-phenyl-5-(1H-pyrazol-3-yl)-1,3-thiazole | 2-phen...)Show InChI InChI=1S/C12H9N3S/c1-2-4-9(5-3-1)12-13-8-11(16-12)10-6-7-14-15-10/h1-8H,(H,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 110 | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition HPGDS in human MEG01 cells assessed as reduction in PGD2 production incubated for 30 mins followed by stimulation with 5 uM ionomycin for ... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084154

(CHEMBL1233897)Show SMILES Fc1cccc(c1)-c1ccc(cn1)C(=O)NC1CCN(CC(F)(F)F)CC1 Show InChI InChI=1S/C19H19F4N3O/c20-15-3-1-2-13(10-15)17-5-4-14(11-24-17)18(27)25-16-6-8-26(9-7-16)12-19(21,22)23/h1-5,10-11,16H,6-9,12H2,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 35 | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition HPGDS in human MEG01 cells assessed as reduction in PGD2 production incubated for 30 mins followed by stimulation with 5 uM ionomycin for ... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM21625

(2-phenyl-5-(1H-pyrazol-3-yl)-1,3-thiazole | 2-phen...)Show InChI InChI=1S/C12H9N3S/c1-2-4-9(5-3-1)12-13-8-11(16-12)10-6-7-14-15-10/h1-8H,(H,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 51 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084153

(CHEMBL3425948)Show SMILES COc1cccc(c1)-c1ccc(cn1)C(=O)NC1CCN(CC(F)(F)F)CC1 Show InChI InChI=1S/C20H22F3N3O2/c1-28-17-4-2-3-14(11-17)18-6-5-15(12-24-18)19(27)25-16-7-9-26(10-8-16)13-20(21,22)23/h2-6,11-12,16H,7-10,13H2,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084156

(CHEMBL3425954)Show InChI InChI=1S/C16H15N3O/c1-2-16(20)19-12-8-11(9-17-10-12)13-4-3-5-15-14(13)6-7-18-15/h3-10,18H,2H2,1H3,(H,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084263
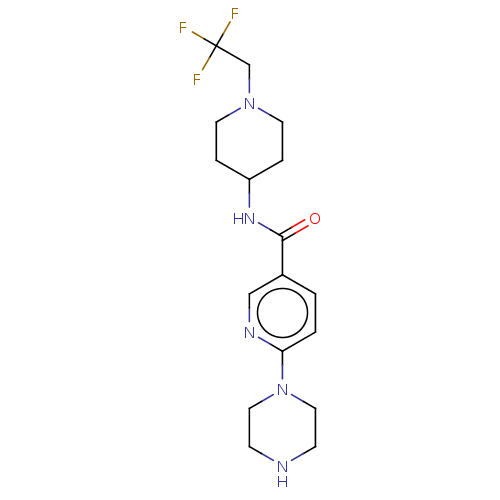
(CHEMBL3425964)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(nc1)N1CCNCC1 Show InChI InChI=1S/C17H24F3N5O/c18-17(19,20)12-24-7-3-14(4-8-24)23-16(26)13-1-2-15(22-11-13)25-9-5-21-6-10-25/h1-2,11,14,21H,3-10,12H2,(H,23,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.30E+4 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084265

(CHEMBL3425966)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(nc1)-c1cc[nH]c(=O)c1 Show InChI InChI=1S/C18H19F3N4O2/c19-18(20,21)11-25-7-4-14(5-8-25)24-17(27)13-1-2-15(23-10-13)12-3-6-22-16(26)9-12/h1-3,6,9-10,14H,4-5,7-8,11H2,(H,22,26)(H,24,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084157

(CHEMBL3425955)Show SMILES O=C(Nc1cncc(c1)-c1cccc2[nH]ccc12)[C@@H]1CCC(=O)N1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084151

(CHEMBL3425950)Show InChI InChI=1S/C19H22N4O/c1-23(2)10-4-7-19(24)22-15-11-14(12-20-13-15)16-5-3-6-18-17(16)8-9-21-18/h3,5-6,8-9,11-13,21H,4,7,10H2,1-2H3,(H,22,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | 570 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084153

(CHEMBL3425948)Show SMILES COc1cccc(c1)-c1ccc(cn1)C(=O)NC1CCN(CC(F)(F)F)CC1 Show InChI InChI=1S/C20H22F3N3O2/c1-28-17-4-2-3-14(11-17)18-6-5-15(12-24-18)19(27)25-16-7-9-26(10-8-16)13-20(21,22)23/h2-6,11-12,16H,7-10,13H2,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli by isothermal titration calorimetry |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084150

(CHEMBL3425951)Show InChI InChI=1S/C18H20N4O/c1-22(2)9-7-18(23)21-14-10-13(11-19-12-14)15-4-3-5-17-16(15)6-8-20-17/h3-6,8,10-12,20H,7,9H2,1-2H3,(H,21,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli by isothermal titration calorimetry |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084261

(CHEMBL3425962)Show InChI InChI=1S/C15H21F3N4O/c1-21(2)13-4-3-11(9-19-13)14(23)20-12-5-7-22(8-6-12)10-15(16,17)18/h3-4,9,12H,5-8,10H2,1-2H3,(H,20,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.40E+4 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084260

(CHEMBL3425961)Show InChI InChI=1S/C17H23F3N4O/c18-17(19,20)12-23-9-5-14(6-10-23)22-16(25)13-3-4-15(21-11-13)24-7-1-2-8-24/h3-4,11,14H,1-2,5-10,12H2,(H,22,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084266
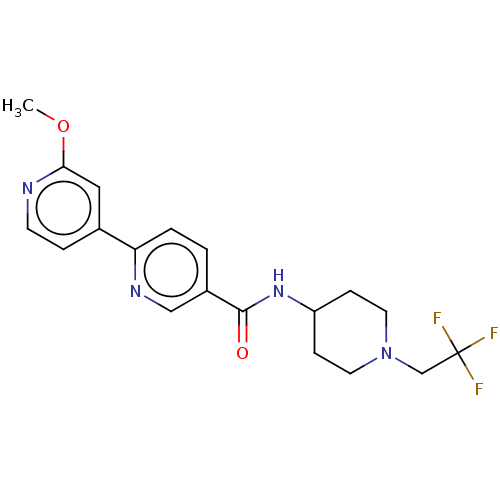
(CHEMBL3425967)Show SMILES COc1cc(ccn1)-c1ccc(cn1)C(=O)NC1CCN(CC(F)(F)F)CC1 Show InChI InChI=1S/C19H21F3N4O2/c1-28-17-10-13(4-7-23-17)16-3-2-14(11-24-16)18(27)25-15-5-8-26(9-6-15)12-19(20,21)22/h2-4,7,10-11,15H,5-6,8-9,12H2,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 2.00E+3 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084259

(CHEMBL3425960)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(nc1)N1CCCCC1 Show InChI InChI=1S/C18H25F3N4O/c19-18(20,21)13-24-10-6-15(7-11-24)23-17(26)14-4-5-16(22-12-14)25-8-2-1-3-9-25/h4-5,12,15H,1-3,6-11,13H2,(H,23,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | <100 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084257

(CHEMBL3425958)Show InChI InChI=1S/C19H17N5O/c1-24-12-21-11-15(24)8-19(25)23-14-7-13(9-20-10-14)16-3-2-4-18-17(16)5-6-22-18/h2-7,9-12,22H,8H2,1H3,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | <2.00E+4 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084256

(CHEMBL3425957)Show SMILES O=C(CCN1CCCCC1)Nc1cncc(c1)-c1cccc2[nH]ccc12 Show InChI InChI=1S/C21H24N4O/c26-21(8-12-25-10-2-1-3-11-25)24-17-13-16(14-22-15-17)18-5-4-6-20-19(18)7-9-23-20/h4-7,9,13-15,23H,1-3,8,10-12H2,(H,24,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084152

(CHEMBL3425949)Show SMILES CC(C)(O)c1nc(no1)-c1ccc(CNC(=O)c2cnc(nc2)-c2ccccn2)cc1 Show InChI InChI=1S/C22H20N6O3/c1-22(2,30)21-27-18(28-31-21)15-8-6-14(7-9-15)11-26-20(29)16-12-24-19(25-13-16)17-5-3-4-10-23-17/h3-10,12-13,30H,11H2,1-2H3,(H,26,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli by isothermal titration calorimetry |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM21625

(2-phenyl-5-(1H-pyrazol-3-yl)-1,3-thiazole | 2-phen...)Show InChI InChI=1S/C12H9N3S/c1-2-4-9(5-3-1)12-13-8-11(16-12)10-6-7-14-15-10/h1-8H,(H,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli by isothermal titration calorimetry |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084258

(CHEMBL3425959)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(Oc2ccccc2)nc1 Show InChI InChI=1S/C19H20F3N3O2/c20-19(21,22)13-25-10-8-15(9-11-25)24-18(26)14-6-7-17(23-12-14)27-16-4-2-1-3-5-16/h1-7,12,15H,8-11,13H2,(H,24,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084155

(CHEMBL3425953)Show SMILES O=C(CCc1cccnc1)Nc1cncc(c1)-c1cccc2[nH]ccc12 Show InChI InChI=1S/C21H18N4O/c26-21(7-6-15-3-2-9-22-12-15)25-17-11-16(13-23-14-17)18-4-1-5-20-19(18)8-10-24-20/h1-5,8-14,24H,6-7H2,(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084153

(CHEMBL3425948)Show SMILES COc1cccc(c1)-c1ccc(cn1)C(=O)NC1CCN(CC(F)(F)F)CC1 Show InChI InChI=1S/C20H22F3N3O2/c1-28-17-4-2-3-14(11-17)18-6-5-15(12-24-18)19(27)25-16-7-9-26(10-8-16)13-20(21,22)23/h2-6,11-12,16H,7-10,13H2,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | n/a | 130 | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition HPGDS in human MEG01 cells assessed as reduction in PGD2 production incubated for 30 mins followed by stimulation with 5 uM ionomycin for ... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084262

(CHEMBL3425963)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(nc1)N1CCOCC1 Show InChI InChI=1S/C17H23F3N4O2/c18-17(19,20)12-23-5-3-14(4-6-23)22-16(25)13-1-2-15(21-11-13)24-7-9-26-10-8-24/h1-2,11,14H,3-10,12H2,(H,22,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084255

(CHEMBL3425956)Show InChI InChI=1S/C16H13N7O/c24-16(9-23-10-19-21-22-23)20-12-6-11(7-17-8-12)13-2-1-3-15-14(13)4-5-18-15/h1-8,10,18H,9H2,(H,20,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084152

(CHEMBL3425949)Show SMILES CC(C)(O)c1nc(no1)-c1ccc(CNC(=O)c2cnc(nc2)-c2ccccn2)cc1 Show InChI InChI=1S/C22H20N6O3/c1-22(2,30)21-27-18(28-31-21)15-8-6-14(7-9-15)11-26-20(29)16-12-24-19(25-13-16)17-5-3-4-10-23-17/h3-10,12-13,30H,11H2,1-2H3,(H,26,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084150

(CHEMBL3425951)Show InChI InChI=1S/C18H20N4O/c1-22(2)9-7-18(23)21-14-10-13(11-19-12-14)15-4-3-5-17-16(15)6-8-20-17/h3-6,8,10-12,20H,7,9H2,1-2H3,(H,21,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084149

(CHEMBL3425952)Show InChI InChI=1S/C15H16N4O3/c1-21-9-3-7-17-14(20)15-18-13(19-22-15)11-4-2-5-12-10(11)6-8-16-12/h2,4-6,8,16H,3,7,9H2,1H3,(H,17,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | 650 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084154

(CHEMBL1233897)Show SMILES Fc1cccc(c1)-c1ccc(cn1)C(=O)NC1CCN(CC(F)(F)F)CC1 Show InChI InChI=1S/C19H19F4N3O/c20-15-3-1-2-13(10-15)17-5-4-14(11-24-17)18(27)25-16-6-8-26(9-7-16)12-19(21,22)23/h1-5,10-11,16H,6-9,12H2,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli by isothermal titration calorimetry |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084268
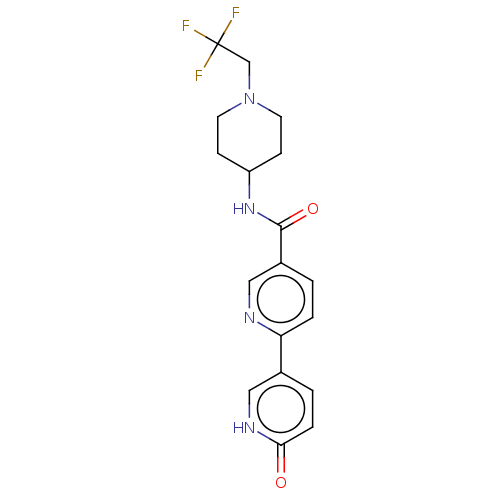
(CHEMBL3425969)Show SMILES FC(F)(F)CN1CCC(CC1)NC(=O)c1ccc(nc1)-c1ccc(=O)[nH]c1 Show InChI InChI=1S/C18H19F3N4O2/c19-18(20,21)11-25-7-5-14(6-8-25)24-17(27)13-1-3-15(22-10-13)12-2-4-16(26)23-9-12/h1-4,9-10,14H,5-8,11H2,(H,23,26)(H,24,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.00E+4 | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to human HPGDS expressed in Escherichia coli using 1-phenylpyrazole-4-carboxylic acid/6-(3-fluorophenyl)pyridine-3-carboxamide as re... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084150

(CHEMBL3425951)Show InChI InChI=1S/C18H20N4O/c1-22(2)9-7-18(23)21-14-10-13(11-19-12-14)15-4-3-5-17-16(15)6-8-20-17/h3-6,8,10-12,20H,7,9H2,1-2H3,(H,21,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 350 | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition HPGDS in human MEG01 cells assessed as reduction in PGD2 production incubated for 30 mins followed by stimulation with 5 uM ionomycin for ... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |
Hematopoietic prostaglandin D synthase
(Homo sapiens (Human)) | BDBM50084152

(CHEMBL3425949)Show SMILES CC(C)(O)c1nc(no1)-c1ccc(CNC(=O)c2cnc(nc2)-c2ccccn2)cc1 Show InChI InChI=1S/C22H20N6O3/c1-22(2,30)21-27-18(28-31-21)15-8-6-14(7-9-15)11-26-20(29)16-12-24-19(25-13-16)17-5-3-4-10-23-17/h3-10,12-13,30H,11H2,1-2H3,(H,26,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | n/a | 74 | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition HPGDS in human MEG01 cells assessed as reduction in PGD2 production incubated for 30 mins followed by stimulation with 5 uM ionomycin for ... |
Bioorg Med Chem Lett 25: 2496-500 (2015)
Article DOI: 10.1016/j.bmcl.2015.04.065
BindingDB Entry DOI: 10.7270/Q2J9683M |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data