| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM50604026 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2246311 (CHEMBL5160521) |
|---|
| IC50 | 8643±n/a nM |
|---|
| Citation |  Li, D; Sloman, DL; Achab, A; Zhou, H; McGowan, MA; White, C; Gibeau, C; Zhang, H; Pu, Q; Bharathan, I; Hopkins, B; Liu, K; Ferguson, H; Fradera, X; Lesburg, CA; Martinot, TA; Qi, J; Song, ZJ; Yin, J; Zhang, H; Song, L; Wan, B; DAddio, S; Solban, N; Miller, JR; Zamlynny, B; Bass, A; Freeland, E; Ykoruk, B; Hilliard, C; Ferraro, J; Zhai, J; Knemeyer, I; Otte, KM; Vincent, S; Sciammetta, N; Pasternak, A; Bennett, DJ; Han, Y Oxetane Promise Delivered: Discovery of Long-Acting IDO1 Inhibitors Suitable for Q3W Oral or Parenteral Dosing. J Med Chem65:6001-6016 (2022) [PubMed] Article Li, D; Sloman, DL; Achab, A; Zhou, H; McGowan, MA; White, C; Gibeau, C; Zhang, H; Pu, Q; Bharathan, I; Hopkins, B; Liu, K; Ferguson, H; Fradera, X; Lesburg, CA; Martinot, TA; Qi, J; Song, ZJ; Yin, J; Zhang, H; Song, L; Wan, B; DAddio, S; Solban, N; Miller, JR; Zamlynny, B; Bass, A; Freeland, E; Ykoruk, B; Hilliard, C; Ferraro, J; Zhai, J; Knemeyer, I; Otte, KM; Vincent, S; Sciammetta, N; Pasternak, A; Bennett, DJ; Han, Y Oxetane Promise Delivered: Discovery of Long-Acting IDO1 Inhibitors Suitable for Q3W Oral or Parenteral Dosing. J Med Chem65:6001-6016 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50604026 |
|---|
| n/a |
|---|
| Name | BDBM50604026 |
|---|
| Synonyms: | CHEMBL5192384 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H22F4N2O3 |
|---|
| Mol. Mass. | 474.4474 |
|---|
| SMILES | CC(C)(O)c1cc(ncc1-c1ccc(cc1)C1(COC1)C(=O)Nc1ccc(F)cc1)C(F)(F)F |
|---|
| Structure | 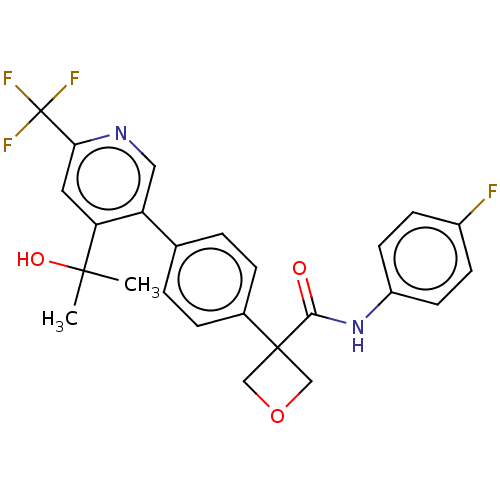
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Li, D; Sloman, DL; Achab, A; Zhou, H; McGowan, MA; White, C; Gibeau, C; Zhang, H; Pu, Q; Bharathan, I; Hopkins, B; Liu, K; Ferguson, H; Fradera, X; Lesburg, CA; Martinot, TA; Qi, J; Song, ZJ; Yin, J; Zhang, H; Song, L; Wan, B; DAddio, S; Solban, N; Miller, JR; Zamlynny, B; Bass, A; Freeland, E; Ykoruk, B; Hilliard, C; Ferraro, J; Zhai, J; Knemeyer, I; Otte, KM; Vincent, S; Sciammetta, N; Pasternak, A; Bennett, DJ; Han, Y Oxetane Promise Delivered: Discovery of Long-Acting IDO1 Inhibitors Suitable for Q3W Oral or Parenteral Dosing. J Med Chem65:6001-6016 (2022) [PubMed] Article
Li, D; Sloman, DL; Achab, A; Zhou, H; McGowan, MA; White, C; Gibeau, C; Zhang, H; Pu, Q; Bharathan, I; Hopkins, B; Liu, K; Ferguson, H; Fradera, X; Lesburg, CA; Martinot, TA; Qi, J; Song, ZJ; Yin, J; Zhang, H; Song, L; Wan, B; DAddio, S; Solban, N; Miller, JR; Zamlynny, B; Bass, A; Freeland, E; Ykoruk, B; Hilliard, C; Ferraro, J; Zhai, J; Knemeyer, I; Otte, KM; Vincent, S; Sciammetta, N; Pasternak, A; Bennett, DJ; Han, Y Oxetane Promise Delivered: Discovery of Long-Acting IDO1 Inhibitors Suitable for Q3W Oral or Parenteral Dosing. J Med Chem65:6001-6016 (2022) [PubMed] Article