 Found 1067 hits with Last Name = 'hall' and Initial = 'se'
Found 1067 hits with Last Name = 'hall' and Initial = 'se' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Prothrombin
(Homo sapiens (Human)) | BDBM50107460
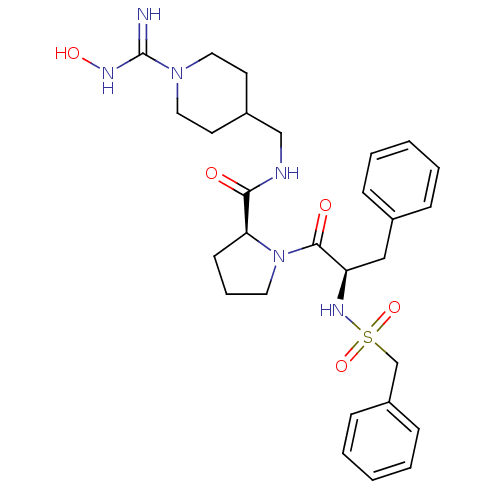
((S)-1-((R)-3-Phenyl-2-phenylmethanesulfonylamino-p...)Show SMILES ONC(=N)N1CCC(CNC(=O)[C@@H]2CCCN2C(=O)[C@@H](Cc2ccccc2)NS(=O)(=O)Cc2ccccc2)CC1 Show InChI InChI=1S/C28H38N6O5S/c29-28(31-37)33-16-13-22(14-17-33)19-30-26(35)25-12-7-15-34(25)27(36)24(18-21-8-3-1-4-9-21)32-40(38,39)20-23-10-5-2-6-11-23/h1-6,8-11,22,24-25,32,37H,7,12-20H2,(H2,29,31)(H,30,35)/t24-,25+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Competitive kinetic for thrombin inhibition Ki was determined |
Bioorg Med Chem Lett 12: 45-9 (2001)
BindingDB Entry DOI: 10.7270/Q2GX4C3P |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM50018531
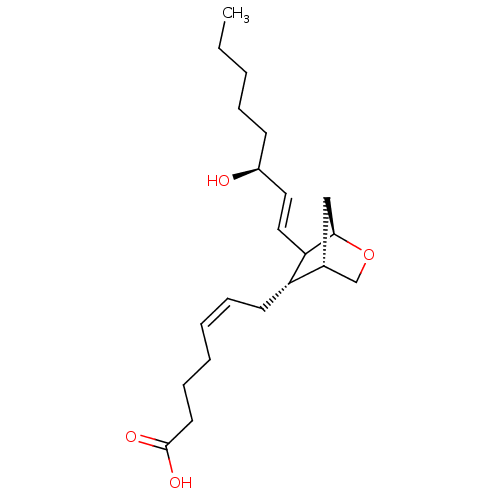
((Z)-7-[(S)-6-((E)-(S)-3-Hydroxy-oct-1-enyl)-2-oxa-...)Show SMILES CCCCC[C@H](O)\C=C\C1[C@H]2C[C@H](CO2)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C21H34O4/c1-2-3-6-9-17(22)12-13-19-18(16-14-20(19)25-15-16)10-7-4-5-8-11-21(23)24/h4,7,12-13,16-20,22H,2-3,5-6,8-11,14-15H2,1H3,(H,23,24)/b7-4-,13-12+/t16-,17+,18+,19?,20-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM81997

(CAS_114865 | NSC_114865 | U44069)Show SMILES CCCCCC(O)C=CC1C2COC(C2)C1CC=CCCCC(O)=O |w:7.6,17.18,TLB:16:15:14:12.11,THB:8:9:14:12.11| Show InChI InChI=1S/C21H34O4/c1-2-3-6-9-17(22)12-13-18-16-14-20(25-15-16)19(18)10-7-4-5-8-11-21(23)24/h4,7,12-13,16-20,22H,2-3,5-6,8-11,14-15H2,1H3,(H,23,24) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 4.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM50008781
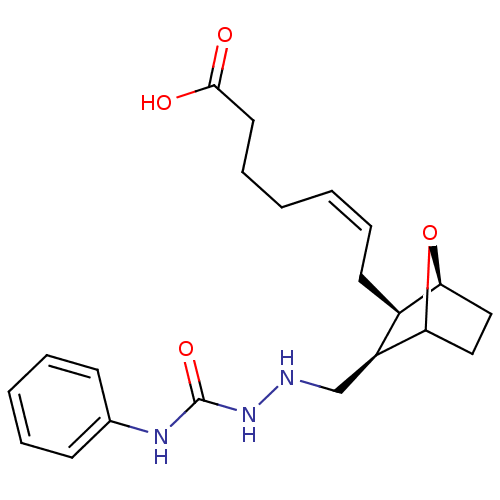
(7-(3-(2-ethyl-N-phenylhydrazinecarboxamide)-7-oxa-...)Show SMILES OC(=O)CCC\C=C/C[C@H]1[C@@H]2CCC(O2)[C@H]1CNNC(=O)Nc1ccccc1 Show InChI InChI=1S/C21H29N3O4/c25-20(26)11-7-2-1-6-10-16-17(19-13-12-18(16)28-19)14-22-24-21(27)23-15-8-4-3-5-9-15/h1,3-6,8-9,16-19,22H,2,7,10-14H2,(H,25,26)(H2,23,24,27)/b6-1-/t16-,17+,18+,19?/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 5.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM50002805

((daltroban){4-[2-(4-Chloro-benzenesulfonylamino)-e...)Show InChI InChI=1S/C16H16ClNO4S/c17-14-5-7-15(8-6-14)23(21,22)18-10-9-12-1-3-13(4-2-12)11-16(19)20/h1-8,18H,9-11H2,(H,19,20) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
| PubMed
| 5.43 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM81997

(CAS_114865 | NSC_114865 | U44069)Show SMILES CCCCCC(O)C=CC1C2COC(C2)C1CC=CCCCC(O)=O |w:7.6,17.18,TLB:16:15:14:12.11,THB:8:9:14:12.11| Show InChI InChI=1S/C21H34O4/c1-2-3-6-9-17(22)12-13-18-16-14-20(25-15-16)19(18)10-7-4-5-8-11-21(23)24/h4,7,12-13,16-20,22H,2-3,5-6,8-11,14-15H2,1H3,(H,23,24) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
Similars
| PubMed
| 7.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM50008781

(7-(3-(2-ethyl-N-phenylhydrazinecarboxamide)-7-oxa-...)Show SMILES OC(=O)CCC\C=C/C[C@H]1[C@@H]2CCC(O2)[C@H]1CNNC(=O)Nc1ccccc1 Show InChI InChI=1S/C21H29N3O4/c25-20(26)11-7-2-1-6-10-16-17(19-13-12-18(16)28-19)14-22-24-21(27)23-15-8-4-3-5-9-15/h1,3-6,8-9,16-19,22H,2,7,10-14H2,(H,25,26)(H2,23,24,27)/b6-1-/t16-,17+,18+,19?/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 7.34 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50107463
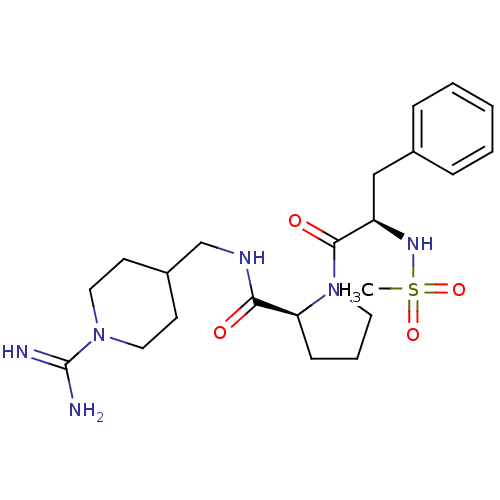
((S)-1-((R)-2-Methanesulfonylamino-3-phenyl-propion...)Show SMILES CS(=O)(=O)N[C@H](Cc1ccccc1)C(=O)N1CCC[C@H]1C(=O)NCC1CCN(CC1)C(N)=N Show InChI InChI=1S/C22H34N6O4S/c1-33(31,32)26-18(14-16-6-3-2-4-7-16)21(30)28-11-5-8-19(28)20(29)25-15-17-9-12-27(13-10-17)22(23)24/h2-4,6-7,17-19,26H,5,8-15H2,1H3,(H3,23,24)(H,25,29)/t18-,19+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 8.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Competitive kinetic for human alpha thrombin inhibition Ki was determined |
Bioorg Med Chem Lett 12: 45-9 (2001)
BindingDB Entry DOI: 10.7270/Q2GX4C3P |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM50002805

((daltroban){4-[2-(4-Chloro-benzenesulfonylamino)-e...)Show InChI InChI=1S/C16H16ClNO4S/c17-14-5-7-15(8-6-14)23(21,22)18-10-9-12-1-3-13(4-2-12)11-16(19)20/h1-8,18H,9-11H2,(H,19,20) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
| PubMed
| 11.3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM50018531

((Z)-7-[(S)-6-((E)-(S)-3-Hydroxy-oct-1-enyl)-2-oxa-...)Show SMILES CCCCC[C@H](O)\C=C\C1[C@H]2C[C@H](CO2)[C@@H]1C\C=C/CCCC(O)=O Show InChI InChI=1S/C21H34O4/c1-2-3-6-9-17(22)12-13-19-18(16-14-20(19)25-15-16)10-7-4-5-8-11-21(23)24/h4,7,12-13,16-20,22H,2-3,5-6,8-11,14-15H2,1H3,(H,23,24)/b7-4-,13-12+/t16-,17+,18+,19?,20-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 18.7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM81998

(CAS_119571 | NSC_119571 | SQ 30741)Show SMILES CCCCCCC(=O)NCC(=O)NCC1C2CCC(O2)C1CC=CCCCC(O)=O |w:22.23,TLB:13:14:16.17:19,THB:21:20:16.17:19| Show InChI InChI=1S/C23H38N2O5/c1-2-3-4-8-11-21(26)25-16-22(27)24-15-18-17(19-13-14-20(18)30-19)10-7-5-6-9-12-23(28)29/h5,7,17-20H,2-4,6,8-16H2,1H3,(H,24,27)(H,25,26)(H,28,29) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 28.3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM81999
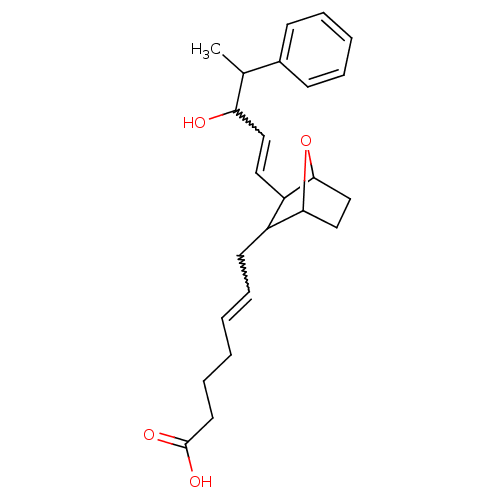
(CAS_3036123 | NSC_3036123 | SQ 28668)Show SMILES CC(C(O)C=CC1C2CCC(O2)C1CC=CCCCC(O)=O)c1ccccc1 |w:4.3,14.15,TLB:5:6:8.9:11,THB:13:12:8.9:11| Show InChI InChI=1S/C24H32O4/c1-17(18-9-5-4-6-10-18)21(25)14-13-20-19(22-15-16-23(20)28-22)11-7-2-3-8-12-24(26)27/h2,4-7,9-10,13-14,17,19-23,25H,3,8,11-12,15-16H2,1H3,(H,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 31.8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM81998

(CAS_119571 | NSC_119571 | SQ 30741)Show SMILES CCCCCCC(=O)NCC(=O)NCC1C2CCC(O2)C1CC=CCCCC(O)=O |w:22.23,TLB:13:14:16.17:19,THB:21:20:16.17:19| Show InChI InChI=1S/C23H38N2O5/c1-2-3-4-8-11-21(26)25-16-22(27)24-15-18-17(19-13-14-20(18)30-19)10-7-5-6-9-12-23(28)29/h5,7,17-20H,2-4,6,8-16H2,1H3,(H,24,27)(H,25,26)(H,28,29) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 49.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM81999

(CAS_3036123 | NSC_3036123 | SQ 28668)Show SMILES CC(C(O)C=CC1C2CCC(O2)C1CC=CCCCC(O)=O)c1ccccc1 |w:4.3,14.15,TLB:5:6:8.9:11,THB:13:12:8.9:11| Show InChI InChI=1S/C24H32O4/c1-17(18-9-5-4-6-10-18)21(25)14-13-20-19(22-15-16-23(20)28-22)11-7-2-3-8-12-24(26)27/h2,4-7,9-10,13-14,17,19-23,25H,3,8,11-12,15-16H2,1H3,(H,26,27) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 72.7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Thromboxane A2 receptor
(Homo sapiens (Human)) | BDBM50002765
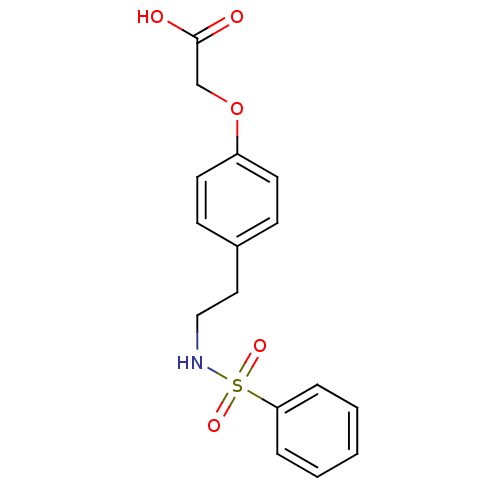
(BM 13177 | BM-13177 | CHEMBL8273 | Sulotroban | [4...)Show InChI InChI=1S/C16H17NO5S/c18-16(19)12-22-14-8-6-13(7-9-14)10-11-17-23(20,21)15-4-2-1-3-5-15/h1-9,17H,10-12H2,(H,18,19) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Squibb Institute for Medical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 245: 786-92 (1988)
BindingDB Entry DOI: 10.7270/Q2D21W4Z |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3153
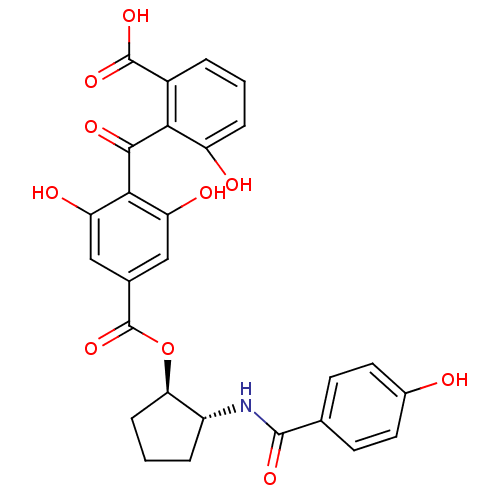
(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of protein kinase C eta |
Bioorg Med Chem Lett 6: 973-978 (1996)
Article DOI: 10.1016/0960-894X(96)00151-5
BindingDB Entry DOI: 10.7270/Q23B603M |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
Bioorg Med Chem Lett 5: 2151-4 (1995)
Article DOI: 10.1016/0960-894X(95)00365-Z
BindingDB Entry DOI: 10.7270/Q2VH5M1B |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3183

(2-({2,6-dihydroxy-4-[({2-[(4-hydroxybenzene)amido]...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)OC1CCCC1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | 7.5 | 30 |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 39: 5215-27 (1996)
Article DOI: 10.1021/jm960581w
BindingDB Entry DOI: 10.7270/Q2G73BVV |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM3183

(2-({2,6-dihydroxy-4-[({2-[(4-hydroxybenzene)amido]...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)OC1CCCC1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | 7.5 | 30 |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 39: 5215-27 (1996)
Article DOI: 10.1021/jm960581w
BindingDB Entry DOI: 10.7270/Q2G73BVV |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of protein kinase C delta |
Bioorg Med Chem Lett 6: 973-978 (1996)
Article DOI: 10.1016/0960-894X(96)00151-5
BindingDB Entry DOI: 10.7270/Q23B603M |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
Bioorg Med Chem Lett 5: 2151-4 (1995)
Article DOI: 10.1016/0960-894X(95)00365-Z
BindingDB Entry DOI: 10.7270/Q2VH5M1B |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
A Division of Eli Lilly & Company
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant human Protein kinase C delta isozyme |
J Med Chem 40: 226-35 (1997)
Article DOI: 10.1021/jm960497g
BindingDB Entry DOI: 10.7270/Q2J965HR |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
A Division of Eli Lilly & Company
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant human Protein kinase C eta isozyme |
J Med Chem 40: 226-35 (1997)
Article DOI: 10.1021/jm960497g
BindingDB Entry DOI: 10.7270/Q2J965HR |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C delta |
Bioorg Med Chem Lett 5: 2015-2020 (1995)
Article DOI: 10.1016/0960-894X(95)00344-S
BindingDB Entry DOI: 10.7270/Q29C6XD1 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of src kinase |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50450704
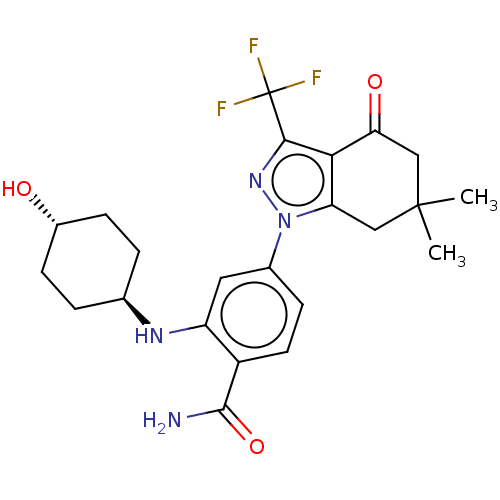
(CHEMBL560895 | SNX-2112)Show SMILES CC1(C)Cc2c(c(nn2-c2ccc(C(N)=O)c(N[C@H]3CC[C@H](O)CC3)c2)C(F)(F)F)C(=O)C1 |r,wU:18.18,wD:21.22,(-2.35,-2,;-2.38,-.77,;-3.43,-.12,;-1.03,-1.55,;.3,-.77,;.3,.77,;1.76,1.24,;2.66,.02,;1.76,-1.24,;2.24,-2.7,;1.13,-3.76,;1.5,-5.26,;2.98,-5.68,;3.35,-7.18,;4.53,-7.52,;2.46,-8.03,;4.09,-4.62,;5.57,-5.04,;6.68,-3.97,;6.3,-2.47,;7.41,-1.4,;8.89,-1.82,;9.77,-.96,;9.27,-3.32,;8.16,-4.39,;3.72,-3.12,;2.24,2.7,;3.44,2.95,;1.41,3.61,;2.62,3.87,;-1.03,1.55,;-1.03,2.79,;-2.38,.77,)| Show InChI InChI=1S/C23H27F3N4O3/c1-22(2)10-17-19(18(32)11-22)20(23(24,25)26)29-30(17)13-5-8-15(21(27)33)16(9-13)28-12-3-6-14(31)7-4-12/h5,8-9,12,14,28,31H,3-4,6-7,10-11H2,1-2H3,(H2,27,33)/t12-,14- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Serenex Inc
Curated by ChEMBL
| Assay Description
Inhibition of Hsp90 in human A375 cells assessed as pS6 degradation after 24 hrs by high content screening |
J Med Chem 52: 4288-305 (2009)
Article DOI: 10.1021/jm900230j
BindingDB Entry DOI: 10.7270/Q2MK6GQJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3153

(2-{[2,6-dihydroxy-4-({[(1R,2R)-2-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C27H23NO10/c29-15-9-7-13(8-10-15)25(34)28-17-4-2-6-21(17)38-27(37)14-11-19(31)23(20(32)12-14)24(33)22-16(26(35)36)3-1-5-18(22)30/h1,3,5,7-12,17,21,29-32H,2,4,6H2,(H,28,34)(H,35,36)/t17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C eta |
Bioorg Med Chem Lett 5: 2015-2020 (1995)
Article DOI: 10.1016/0960-894X(95)00344-S
BindingDB Entry DOI: 10.7270/Q29C6XD1 |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50055672
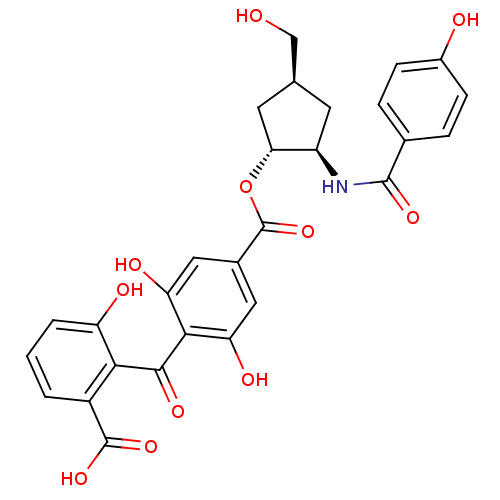
(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC[C@H]1C[C@@H](NC(=O)c2ccc(O)cc2)[C@@H](C1)OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2C(O)=O)c(O)c1 Show InChI InChI=1S/C28H25NO11/c30-12-13-8-18(29-26(36)14-4-6-16(31)7-5-14)22(9-13)40-28(39)15-10-20(33)24(21(34)11-15)25(35)23-17(27(37)38)2-1-3-19(23)32/h1-7,10-11,13,18,22,30-34H,8-9,12H2,(H,29,36)(H,37,38)/t13-,18+,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
A Division of Eli Lilly & Company
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant human Protein kinase C eta isozyme |
J Med Chem 40: 226-35 (1997)
Article DOI: 10.1021/jm960497g
BindingDB Entry DOI: 10.7270/Q2J965HR |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50055668
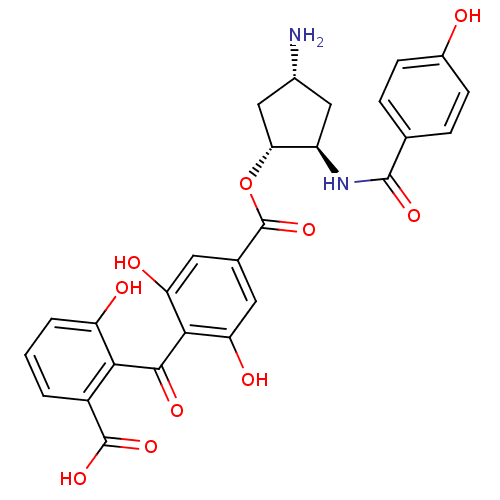
(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES N[C@@H]1C[C@@H](NC(=O)c2ccc(O)cc2)[C@@H](C1)OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2C(O)=O)c(O)c1 Show InChI InChI=1S/C27H24N2O10/c28-14-10-17(29-25(35)12-4-6-15(30)7-5-12)21(11-14)39-27(38)13-8-19(32)23(20(33)9-13)24(34)22-16(26(36)37)2-1-3-18(22)31/h1-9,14,17,21,30-33H,10-11,28H2,(H,29,35)(H,36,37)/t14-,17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
A Division of Eli Lilly & Company
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant human Protein kinase C beta 2 isozyme |
J Med Chem 40: 226-35 (1997)
Article DOI: 10.1021/jm960497g
BindingDB Entry DOI: 10.7270/Q2J965HR |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50107460

((S)-1-((R)-3-Phenyl-2-phenylmethanesulfonylamino-p...)Show SMILES ONC(=N)N1CCC(CNC(=O)[C@@H]2CCCN2C(=O)[C@@H](Cc2ccccc2)NS(=O)(=O)Cc2ccccc2)CC1 Show InChI InChI=1S/C28H38N6O5S/c29-28(31-37)33-16-13-22(14-17-33)19-30-26(35)25-12-7-15-34(25)27(36)24(18-21-8-3-1-4-9-21)32-40(38,39)20-23-10-5-2-6-11-23/h1-6,8-11,22,24-25,32,37H,7,12-20H2,(H2,29,31)(H,30,35)/t24-,25+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against hydrolysis of human alpha thrombin |
Bioorg Med Chem Lett 12: 45-9 (2001)
BindingDB Entry DOI: 10.7270/Q2GX4C3P |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50450704

(CHEMBL560895 | SNX-2112)Show SMILES CC1(C)Cc2c(c(nn2-c2ccc(C(N)=O)c(N[C@H]3CC[C@H](O)CC3)c2)C(F)(F)F)C(=O)C1 |r,wU:18.18,wD:21.22,(-2.35,-2,;-2.38,-.77,;-3.43,-.12,;-1.03,-1.55,;.3,-.77,;.3,.77,;1.76,1.24,;2.66,.02,;1.76,-1.24,;2.24,-2.7,;1.13,-3.76,;1.5,-5.26,;2.98,-5.68,;3.35,-7.18,;4.53,-7.52,;2.46,-8.03,;4.09,-4.62,;5.57,-5.04,;6.68,-3.97,;6.3,-2.47,;7.41,-1.4,;8.89,-1.82,;9.77,-.96,;9.27,-3.32,;8.16,-4.39,;3.72,-3.12,;2.24,2.7,;3.44,2.95,;1.41,3.61,;2.62,3.87,;-1.03,1.55,;-1.03,2.79,;-2.38,.77,)| Show InChI InChI=1S/C23H27F3N4O3/c1-22(2)10-17-19(18(32)11-22)20(23(24,25)26)29-30(17)13-5-8-15(21(27)33)16(9-13)28-12-3-6-14(31)7-4-12/h5,8-9,12,14,28,31H,3-4,6-7,10-11H2,1-2H3,(H2,27,33)/t12-,14- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Serenex Inc
Curated by ChEMBL
| Assay Description
Inhibition of Hsp90 in human A375 cells assessed as Hsp70 induction after 24 hrs by high content screening |
J Med Chem 52: 4288-305 (2009)
Article DOI: 10.1021/jm900230j
BindingDB Entry DOI: 10.7270/Q2MK6GQJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prothrombin
(Homo sapiens (Human)) | BDBM50107460

((S)-1-((R)-3-Phenyl-2-phenylmethanesulfonylamino-p...)Show SMILES ONC(=N)N1CCC(CNC(=O)[C@@H]2CCCN2C(=O)[C@@H](Cc2ccccc2)NS(=O)(=O)Cc2ccccc2)CC1 Show InChI InChI=1S/C28H38N6O5S/c29-28(31-37)33-16-13-22(14-17-33)19-30-26(35)25-12-7-15-34(25)27(36)24(18-21-8-3-1-4-9-21)32-40(38,39)20-23-10-5-2-6-11-23/h1-6,8-11,22,24-25,32,37H,7,12-20H2,(H2,29,31)(H,30,35)/t24-,25+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
In vitro inhibitory activity against hydrolysis of thrombin was determined |
Bioorg Med Chem Lett 12: 45-9 (2001)
BindingDB Entry DOI: 10.7270/Q2GX4C3P |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50285087

((R)-2-{2,6-Dihydroxy-4-[(3R,4R)-4-(4-hydroxy-benzo...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)N[C@@H]1CNC[C@H]1NC(=O)c1ccc(O)cc1 Show InChI InChI=1S/C26H23N3O9/c30-14-6-4-12(5-7-14)24(35)28-16-10-27-11-17(16)29-25(36)13-8-19(32)22(20(33)9-13)23(34)21-15(26(37)38)2-1-3-18(21)31/h1-9,16-17,27,30-33H,10-11H2,(H,28,35)(H,29,36)(H,37,38)/t16-,17-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C eta |
Bioorg Med Chem Lett 5: 2015-2020 (1995)
Article DOI: 10.1016/0960-894X(95)00344-S
BindingDB Entry DOI: 10.7270/Q29C6XD1 |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50055672

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC[C@H]1C[C@@H](NC(=O)c2ccc(O)cc2)[C@@H](C1)OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2C(O)=O)c(O)c1 Show InChI InChI=1S/C28H25NO11/c30-12-13-8-18(29-26(36)14-4-6-16(31)7-5-14)22(9-13)40-28(39)15-10-20(33)24(21(34)11-15)25(35)23-17(27(37)38)2-1-3-19(23)32/h1-7,10-11,13,18,22,30-34H,8-9,12H2,(H,29,36)(H,37,38)/t13-,18+,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
A Division of Eli Lilly & Company
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant human Protein kinase C beta 2 isozyme |
J Med Chem 40: 226-35 (1997)
Article DOI: 10.1021/jm960497g
BindingDB Entry DOI: 10.7270/Q2J965HR |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3149
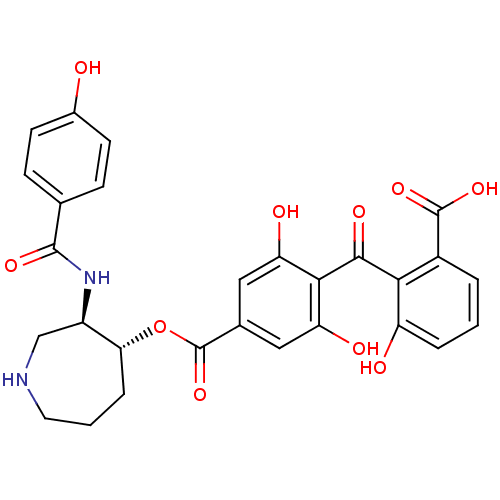
(2-{[2,6-dihydroxy-4-({[(3R,4R)-3-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 39: 5215-27 (1996)
Article DOI: 10.1021/jm960581w
BindingDB Entry DOI: 10.7270/Q2G73BVV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM3199
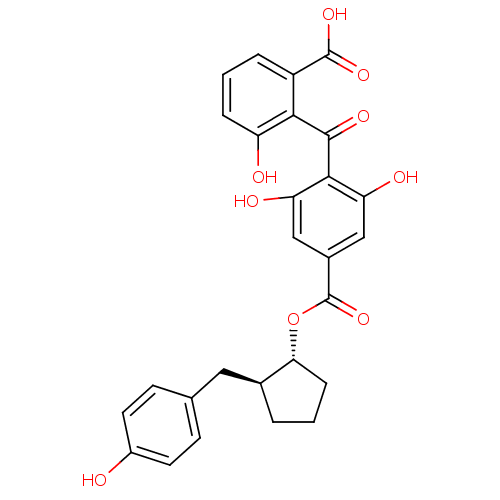
((+-)-anti-2-[[2,6-Dihydroxy-4-[[[2-(4-hydroxybenzy...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1Cc1ccc(O)cc1 |r| Show InChI InChI=1S/C27H24O9/c28-17-9-7-14(8-10-17)11-15-3-1-6-22(15)36-27(35)16-12-20(30)24(21(31)13-16)25(32)23-18(26(33)34)4-2-5-19(23)29/h2,4-5,7-10,12-13,15,22,28-31H,1,3,6,11H2,(H,33,34)/t15-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 39: 5215-27 (1996)
Article DOI: 10.1021/jm960581w
BindingDB Entry DOI: 10.7270/Q2G73BVV |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3199

((+-)-anti-2-[[2,6-Dihydroxy-4-[[[2-(4-hydroxybenzy...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCC[C@H]1Cc1ccc(O)cc1 |r| Show InChI InChI=1S/C27H24O9/c28-17-9-7-14(8-10-17)11-15-3-1-6-22(15)36-27(35)16-12-20(30)24(21(31)13-16)25(32)23-18(26(33)34)4-2-5-19(23)29/h2,4-5,7-10,12-13,15,22,28-31H,1,3,6,11H2,(H,33,34)/t15-,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 39: 5215-27 (1996)
Article DOI: 10.1021/jm960581w
BindingDB Entry DOI: 10.7270/Q2G73BVV |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM3186
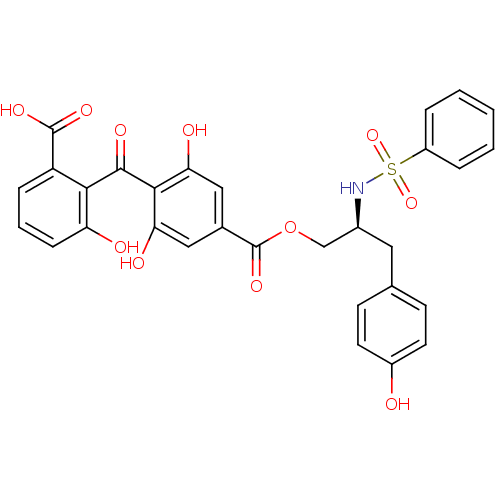
((S)-2-[[2,6-Dihydroxy-4-[[[3-(4-hydroxyphenyl)-2-[...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)OC[C@H](Cc1ccc(O)cc1)NS(=O)(=O)c1ccccc1 |r| Show InChI InChI=1S/C30H25NO11S/c32-20-11-9-17(10-12-20)13-19(31-43(40,41)21-5-2-1-3-6-21)16-42-30(39)18-14-24(34)27(25(35)15-18)28(36)26-22(29(37)38)7-4-8-23(26)33/h1-12,14-15,19,31-35H,13,16H2,(H,37,38)/t19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 39: 5215-27 (1996)
Article DOI: 10.1021/jm960581w
BindingDB Entry DOI: 10.7270/Q2G73BVV |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50055685
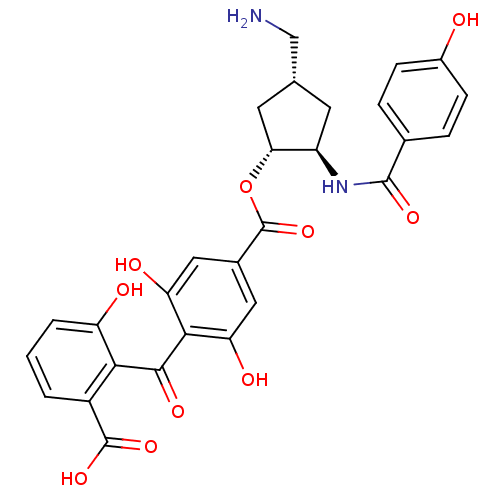
(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES NC[C@@H]1C[C@@H](NC(=O)c2ccc(O)cc2)[C@@H](C1)OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2C(O)=O)c(O)c1 Show InChI InChI=1S/C28H26N2O10/c29-12-13-8-18(30-26(36)14-4-6-16(31)7-5-14)22(9-13)40-28(39)15-10-20(33)24(21(34)11-15)25(35)23-17(27(37)38)2-1-3-19(23)32/h1-7,10-11,13,18,22,31-34H,8-9,12,29H2,(H,30,36)(H,37,38)/t13-,18-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
A Division of Eli Lilly & Company
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant human Protein kinase C eta isozyme |
J Med Chem 40: 226-35 (1997)
Article DOI: 10.1021/jm960497g
BindingDB Entry DOI: 10.7270/Q2J965HR |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3149

(2-{[2,6-dihydroxy-4-({[(3R,4R)-3-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
A Division of Eli Lilly & Company
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant human Protein kinase C eta isozyme |
J Med Chem 40: 226-35 (1997)
Article DOI: 10.1021/jm960497g
BindingDB Entry DOI: 10.7270/Q2J965HR |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3149

(2-{[2,6-dihydroxy-4-({[(3R,4R)-3-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C eta |
Bioorg Med Chem Lett 6: 973-978 (1996)
Article DOI: 10.1016/0960-894X(96)00151-5
BindingDB Entry DOI: 10.7270/Q23B603M |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50055684
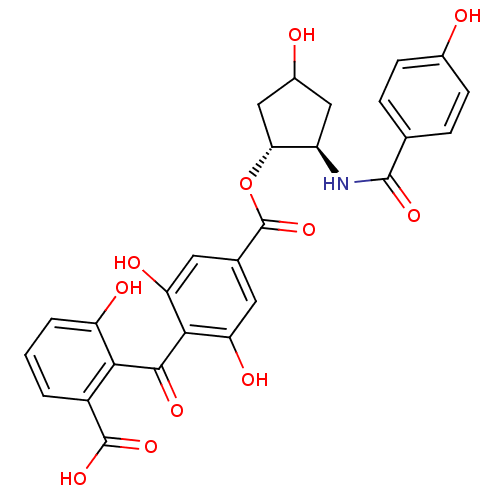
(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES OC1C[C@@H](NC(=O)c2ccc(O)cc2)[C@@H](C1)OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2C(O)=O)c(O)c1 Show InChI InChI=1S/C27H23NO11/c29-14-6-4-12(5-7-14)25(35)28-17-10-15(30)11-21(17)39-27(38)13-8-19(32)23(20(33)9-13)24(34)22-16(26(36)37)2-1-3-18(22)31/h1-9,15,17,21,29-33H,10-11H2,(H,28,35)(H,36,37)/t15?,17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
A Division of Eli Lilly & Company
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant human Protein kinase C eta isozyme |
J Med Chem 40: 226-35 (1997)
Article DOI: 10.1021/jm960497g
BindingDB Entry DOI: 10.7270/Q2J965HR |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50055668

(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES N[C@@H]1C[C@@H](NC(=O)c2ccc(O)cc2)[C@@H](C1)OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2C(O)=O)c(O)c1 Show InChI InChI=1S/C27H24N2O10/c28-14-10-17(29-25(35)12-4-6-15(30)7-5-12)21(11-14)39-27(38)13-8-19(32)23(20(33)9-13)24(34)22-16(26(36)37)2-1-3-18(22)31/h1-9,14,17,21,30-33H,10-11,28H2,(H,29,35)(H,36,37)/t14-,17-,21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
A Division of Eli Lilly & Company
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant human Protein kinase C eta isozyme |
J Med Chem 40: 226-35 (1997)
Article DOI: 10.1021/jm960497g
BindingDB Entry DOI: 10.7270/Q2J965HR |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50055677
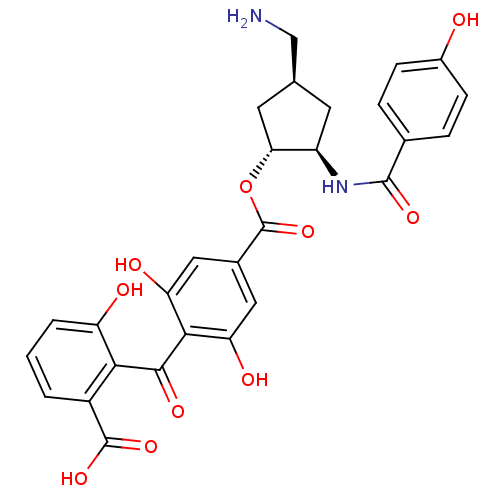
(4-(2-Carboxy-6-hydroxy-benzoyl)-3,5-dihydroxy-benz...)Show SMILES NC[C@H]1C[C@@H](NC(=O)c2ccc(O)cc2)[C@@H](C1)OC(=O)c1cc(O)c(C(=O)c2c(O)cccc2C(O)=O)c(O)c1 Show InChI InChI=1S/C28H26N2O10/c29-12-13-8-18(30-26(36)14-4-6-16(31)7-5-14)22(9-13)40-28(39)15-10-20(33)24(21(34)11-15)25(35)23-17(27(37)38)2-1-3-19(23)32/h1-7,10-11,13,18,22,31-34H,8-9,12,29H2,(H,30,36)(H,37,38)/t13-,18+,22+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
A Division of Eli Lilly & Company
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant human Protein kinase C eta isozyme |
J Med Chem 40: 226-35 (1997)
Article DOI: 10.1021/jm960497g
BindingDB Entry DOI: 10.7270/Q2J965HR |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3149

(2-{[2,6-dihydroxy-4-({[(3R,4R)-3-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C zeta |
Bioorg Med Chem Lett 5: 2015-2020 (1995)
Article DOI: 10.1016/0960-894X(95)00344-S
BindingDB Entry DOI: 10.7270/Q29C6XD1 |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3149

(2-{[2,6-dihydroxy-4-({[(3R,4R)-3-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CCCNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C28H26N2O10/c31-16-8-6-14(7-9-16)26(36)30-18-13-29-10-2-5-22(18)40-28(39)15-11-20(33)24(21(34)12-15)25(35)23-17(27(37)38)3-1-4-19(23)32/h1,3-4,6-9,11-12,18,22,29,31-34H,2,5,10,13H2,(H,30,36)(H,37,38)/t18-,22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 2 |
Bioorg Med Chem Lett 5: 2015-2020 (1995)
Article DOI: 10.1016/0960-894X(95)00344-S
BindingDB Entry DOI: 10.7270/Q29C6XD1 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50008057
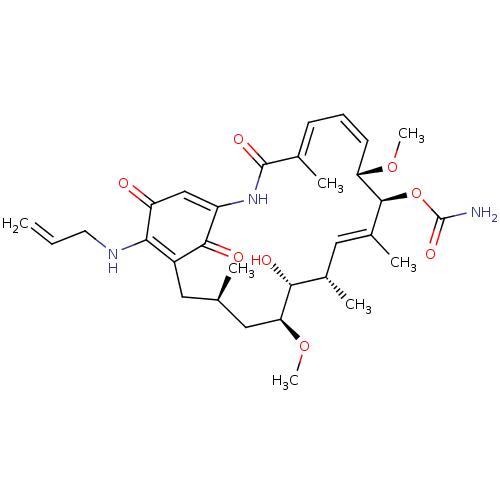
(BMS-722782 | CHEBI:64153 | TANESPIMYCIN)Show SMILES CO[C@H]1C[C@H](C)CC2=C(NCC=C)C(=O)C=C(NC(=O)\C(C)=C\C=C/[C@H](OC)[C@@H](OC(N)=O)\C(C)=C\[C@H](C)[C@H]1O)C2=O |r,c:7,23,t:15,21,34| Show InChI InChI=1S/C31H43N3O8/c1-8-12-33-26-21-13-17(2)14-25(41-7)27(36)19(4)15-20(5)29(42-31(32)39)24(40-6)11-9-10-18(3)30(38)34-22(28(21)37)16-23(26)35/h8-11,15-17,19,24-25,27,29,33,36H,1,12-14H2,2-7H3,(H2,32,39)(H,34,38)/b11-9-,18-10+,20-15+/t17-,19+,24+,25+,27-,29+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Serenex Inc
Curated by ChEMBL
| Assay Description
Inhibition of Hsp90 in human AU565 cells assessed as Her2 degradation after 24 hrs by high content screening |
J Med Chem 52: 4288-305 (2009)
Article DOI: 10.1021/jm900230j
BindingDB Entry DOI: 10.7270/Q2MK6GQJ |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3186

((S)-2-[[2,6-Dihydroxy-4-[[[3-(4-hydroxyphenyl)-2-[...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)OC[C@H](Cc1ccc(O)cc1)NS(=O)(=O)c1ccccc1 |r| Show InChI InChI=1S/C30H25NO11S/c32-20-11-9-17(10-12-20)13-19(31-43(40,41)21-5-2-1-3-6-21)16-42-30(39)18-14-24(34)27(25(35)15-18)28(36)26-22(29(37)38)7-4-8-23(26)33/h1-12,14-15,19,31-35H,13,16H2,(H,37,38)/t19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sphinx Laboratories
| Assay Description
PKC was assayed by quantitating the incorporation of 32P from [gamma-32P]ATP into histone type IIIs. |
J Med Chem 39: 5215-27 (1996)
Article DOI: 10.1021/jm960581w
BindingDB Entry DOI: 10.7270/Q2G73BVV |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM3152
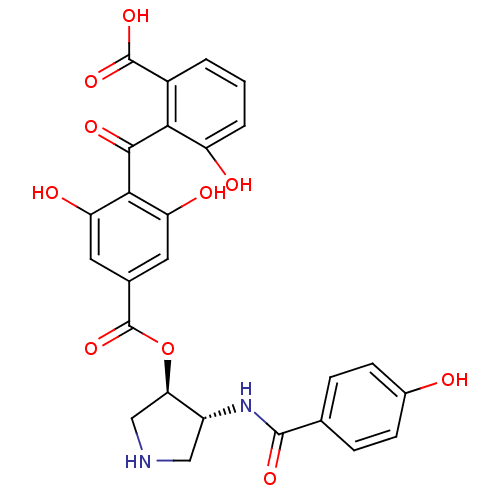
(2-{[2,6-dihydroxy-4-({[(3R,4R)-4-[(4-hydroxybenzen...)Show SMILES OC(=O)c1cccc(O)c1C(=O)c1c(O)cc(cc1O)C(=O)O[C@@H]1CNC[C@H]1NC(=O)c1ccc(O)cc1 |r| Show InChI InChI=1S/C26H22N2O10/c29-14-6-4-12(5-7-14)24(34)28-16-10-27-11-20(16)38-26(37)13-8-18(31)22(19(32)9-13)23(33)21-15(25(35)36)2-1-3-17(21)30/h1-9,16,20,27,29-32H,10-11H2,(H,28,34)(H,35,36)/t16-,20-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C eta |
Bioorg Med Chem Lett 5: 2015-2020 (1995)
Article DOI: 10.1016/0960-894X(95)00344-S
BindingDB Entry DOI: 10.7270/Q29C6XD1 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50008057

(BMS-722782 | CHEBI:64153 | TANESPIMYCIN)Show SMILES CO[C@H]1C[C@H](C)CC2=C(NCC=C)C(=O)C=C(NC(=O)\C(C)=C\C=C/[C@H](OC)[C@@H](OC(N)=O)\C(C)=C\[C@H](C)[C@H]1O)C2=O |r,c:7,23,t:15,21,34| Show InChI InChI=1S/C31H43N3O8/c1-8-12-33-26-21-13-17(2)14-25(41-7)27(36)19(4)15-20(5)29(42-31(32)39)24(40-6)11-9-10-18(3)30(38)34-22(28(21)37)16-23(26)35/h8-11,15-17,19,24-25,27,29,33,36H,1,12-14H2,2-7H3,(H2,32,39)(H,34,38)/b11-9-,18-10+,20-15+/t17-,19+,24+,25+,27-,29+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Serenex Inc
Curated by ChEMBL
| Assay Description
Inhibition of Hsp90 in human A375 cells assessed as Hsp70 induction after 24 hrs by high content screening |
J Med Chem 52: 4288-305 (2009)
Article DOI: 10.1021/jm900230j
BindingDB Entry DOI: 10.7270/Q2MK6GQJ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data