 Found 130 hits with Last Name = 'olsson' and Initial = 't'
Found 130 hits with Last Name = 'olsson' and Initial = 't' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50262998
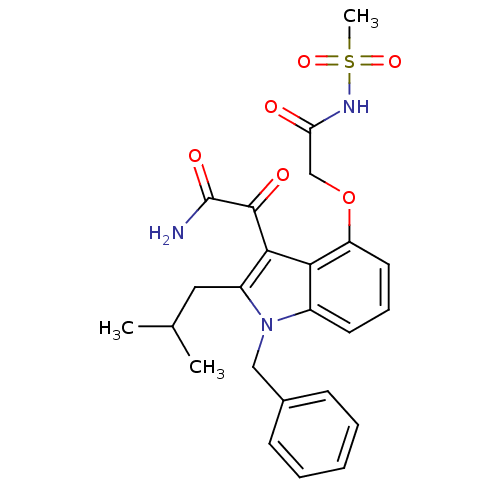
(CHEMBL477548 | mesyl-2-(3-(2-amino-2-oxoacetyl)-1-...)Show SMILES CC(C)Cc1c(C(=O)C(N)=O)c2c(OCC(=O)NS(C)(=O)=O)cccc2n1Cc1ccccc1 Show InChI InChI=1S/C24H27N3O6S/c1-15(2)12-18-22(23(29)24(25)30)21-17(27(18)13-16-8-5-4-6-9-16)10-7-11-19(21)33-14-20(28)26-34(3,31)32/h4-11,15H,12-14H2,1-3H3,(H2,25,30)(H,26,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to sPLA2X (unknown origin) |
Bioorg Med Chem Lett 24: 5251-5 (2014)
Article DOI: 10.1016/j.bmcl.2014.09.058
BindingDB Entry DOI: 10.7270/Q2668FS8 |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008271
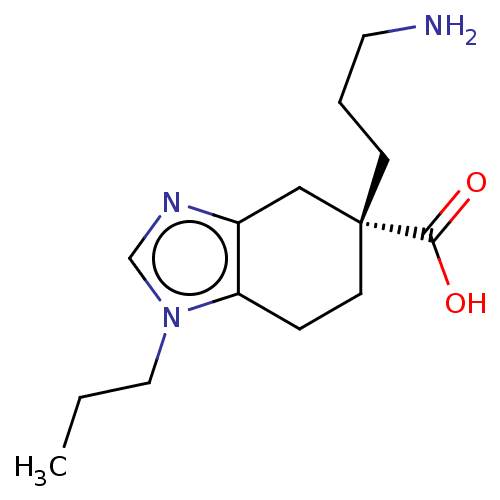
(CHEMBL3235132)Show InChI InChI=1S/C14H23N3O2/c1-2-8-17-10-16-11-9-14(13(18)19,5-3-7-15)6-4-12(11)17/h10H,2-9,15H2,1H3,(H,18,19)/t14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human TAF1a using hippuryl-L-arginine/hippuryl-L-lysine as substrate by liquid chromatographic analysis |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50055366
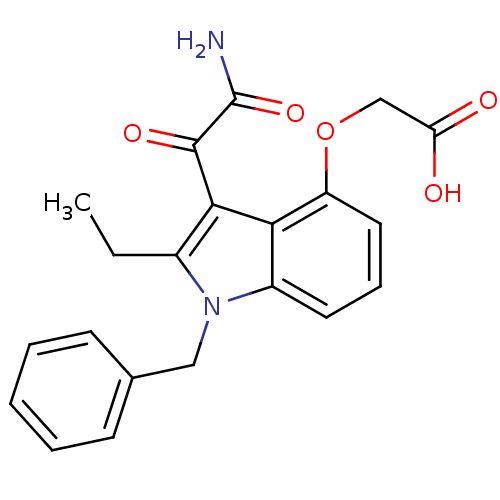
((3-Aminooxalyl-1-benzyl-2-ethyl-1H-indol-4-yloxy)-...)Show SMILES CCc1c(C(=O)C(N)=O)c2c(OCC(O)=O)cccc2n1Cc1ccccc1 Show InChI InChI=1S/C21H20N2O5/c1-2-14-19(20(26)21(22)27)18-15(9-6-10-16(18)28-12-17(24)25)23(14)11-13-7-4-3-5-8-13/h3-10H,2,11-12H2,1H3,(H2,22,27)(H,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-2A expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50422375
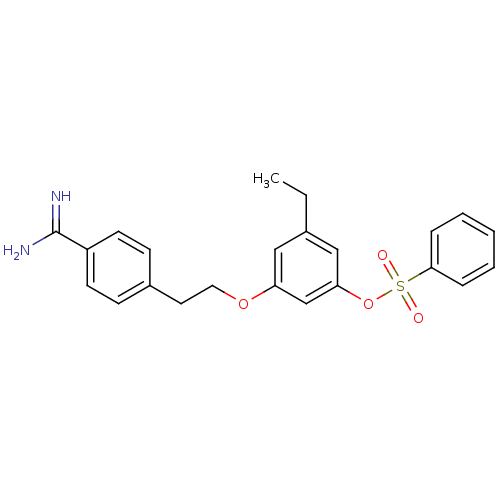
(CHEMBL116616)Show SMILES CCc1cc(OCCc2ccc(cc2)C(N)=N)cc(OS(=O)(=O)c2ccccc2)c1 Show InChI InChI=1S/C23H24N2O4S/c1-2-17-14-20(28-13-12-18-8-10-19(11-9-18)23(24)25)16-21(15-17)29-30(26,27)22-6-4-3-5-7-22/h3-11,14-16H,2,12-13H2,1H3,(H3,24,25) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibitory activity against thrombin |
J Med Chem 44: 3424-39 (2001)
BindingDB Entry DOI: 10.7270/Q2D21ZWN |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366784
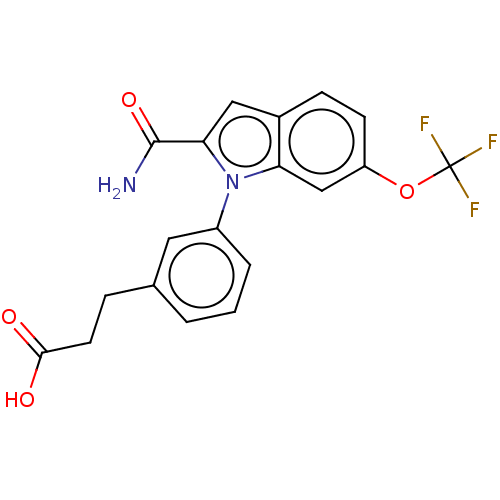
(CHEMBL4171084)Show SMILES NC(=O)c1cc2ccc(OC(F)(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C19H15F3N2O4/c20-19(21,22)28-14-6-5-12-9-16(18(23)27)24(15(12)10-14)13-3-1-2-11(8-13)4-7-17(25)26/h1-3,5-6,8-10H,4,7H2,(H2,23,27)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50055366

((3-Aminooxalyl-1-benzyl-2-ethyl-1H-indol-4-yloxy)-...)Show SMILES CCc1c(C(=O)C(N)=O)c2c(OCC(O)=O)cccc2n1Cc1ccccc1 Show InChI InChI=1S/C21H20N2O5/c1-2-14-19(20(26)21(22)27)18-15(9-6-10-16(18)28-12-17(24)25)23(14)11-13-7-4-3-5-8-13/h3-10H,2,11-12H2,1H3,(H2,22,27)(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008272
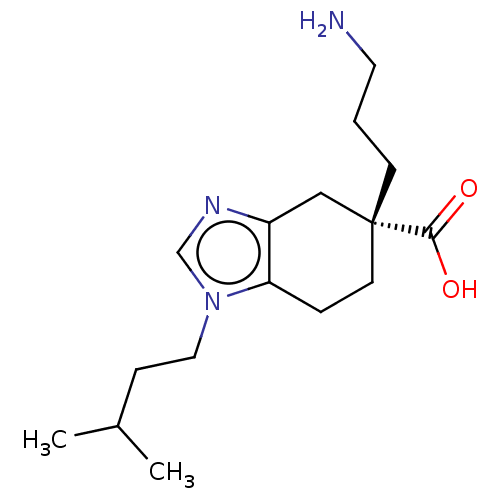
(CHEMBL3235133)Show SMILES Cl.CC(C)CCn1cnc2C[C@@](CCCN)(CCc12)C(O)=O |r| Show InChI InChI=1S/C16H27N3O2.ClH/c1-12(2)5-9-19-11-18-13-10-16(15(20)21,6-3-8-17)7-4-14(13)19;/h11-12H,3-10,17H2,1-2H3,(H,20,21);1H/t16-;/m1./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <41 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human TAF1a |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008279
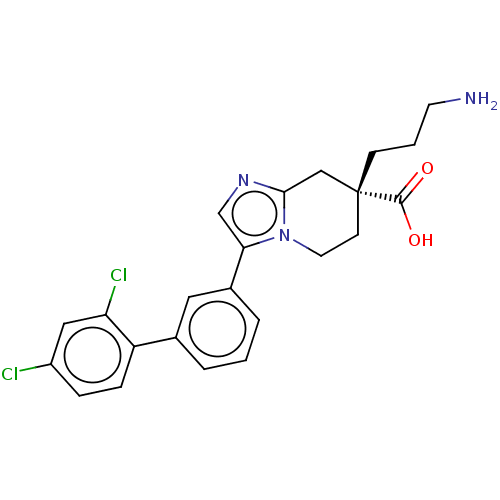
(CHEMBL3235140)Show SMILES Cl.NCCC[C@]1(CCn2c(C1)ncc2-c1cccc(c1)-c1ccc(Cl)cc1Cl)C(O)=O |r| Show InChI InChI=1S/C23H23Cl2N3O2.ClH/c24-17-5-6-18(19(25)12-17)15-3-1-4-16(11-15)20-14-27-21-13-23(22(29)30,7-2-9-26)8-10-28(20)21;/h1,3-6,11-12,14H,2,7-10,13,26H2,(H,29,30);1H/t23-;/m1./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | <41 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human TAF1a |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366811
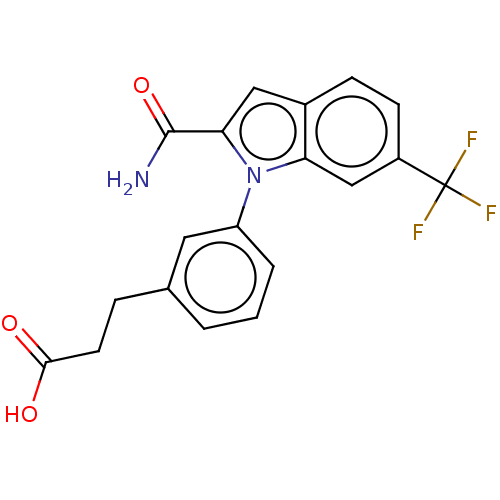
(CHEMBL4176544)Show SMILES NC(=O)c1cc2ccc(cc2n1-c1cccc(CCC(O)=O)c1)C(F)(F)F Show InChI InChI=1S/C19H15F3N2O3/c20-19(21,22)13-6-5-12-9-16(18(23)27)24(15(12)10-13)14-3-1-2-11(8-14)4-7-17(25)26/h1-3,5-6,8-10H,4,7H2,(H2,23,27)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 43 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50031106
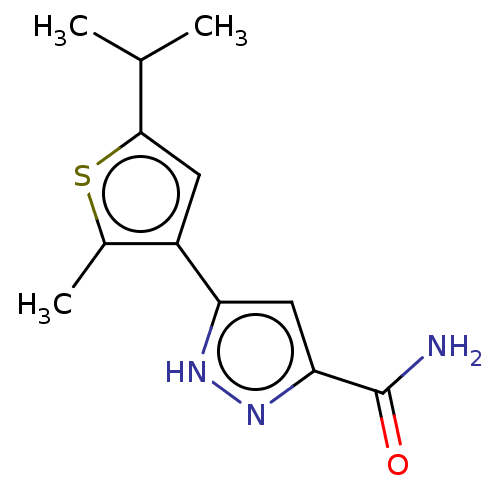
(CHEMBL3337975)Show InChI InChI=1S/C12H15N3OS/c1-6(2)11-4-8(7(3)17-11)9-5-10(12(13)16)15-14-9/h4-6H,1-3H3,(H2,13,16)(H,14,15) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >51 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human sPLA2X using 1,2-bis(heptanoylthio) glycerophosphocholine substrate incubated for 30 mins |
Bioorg Med Chem Lett 24: 5251-5 (2014)
Article DOI: 10.1016/j.bmcl.2014.09.058
BindingDB Entry DOI: 10.7270/Q2668FS8 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50422376

(CHEMBL116149)Show SMILES COc1c(Cl)cc(Cl)cc1S(=O)(=O)Oc1cccc(OCCc2ccc(cc2)C(N)=N)c1 Show InChI InChI=1S/C22H20Cl2N2O5S/c1-29-21-19(24)11-16(23)12-20(21)32(27,28)31-18-4-2-3-17(13-18)30-10-9-14-5-7-15(8-6-14)22(25)26/h2-8,11-13H,9-10H2,1H3,(H3,25,26) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 56 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibitory activity against thrombin |
J Med Chem 44: 3424-39 (2001)
BindingDB Entry DOI: 10.7270/Q2D21ZWN |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008278
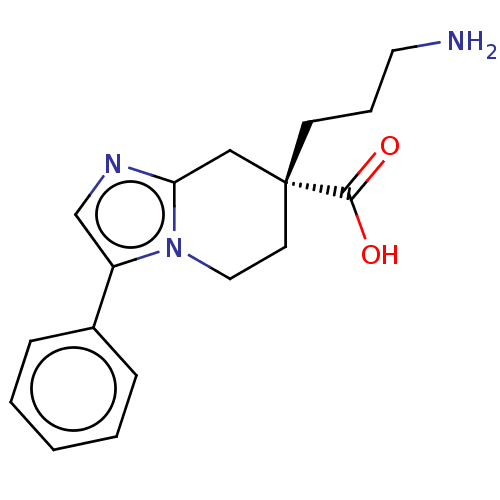
(CHEMBL3235139)Show SMILES NCCC[C@]1(CCn2c(C1)ncc2-c1ccccc1)C(O)=O |r| Show InChI InChI=1S/C17H21N3O2/c18-9-4-7-17(16(21)22)8-10-20-14(12-19-15(20)11-17)13-5-2-1-3-6-13/h1-3,5-6,12H,4,7-11,18H2,(H,21,22)/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 66 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human TAF1a |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366983
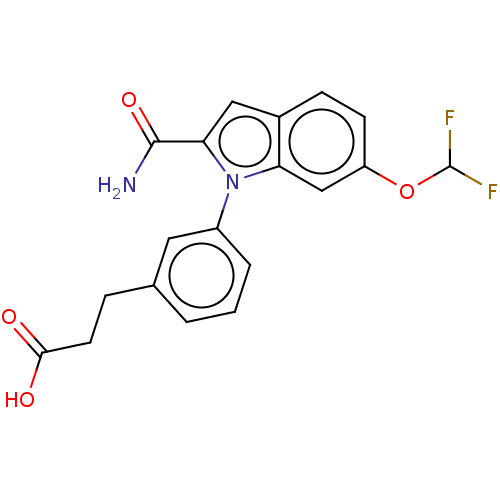
(CHEMBL4160483)Show SMILES NC(=O)c1cc2ccc(OC(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C19H16F2N2O4/c20-19(21)27-14-6-5-12-9-16(18(22)26)23(15(12)10-14)13-3-1-2-11(8-13)4-7-17(24)25/h1-3,5-6,8-10,19H,4,7H2,(H2,22,26)(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 68 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008271

(CHEMBL3235132)Show InChI InChI=1S/C14H23N3O2/c1-2-8-17-10-16-11-9-14(13(18)19,5-3-7-15)6-4-12(11)17/h10H,2-9,15H2,1H3,(H,18,19)/t14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 76 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human TAF1a |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008273
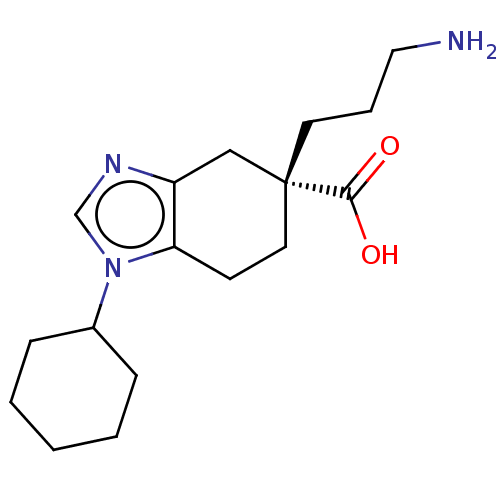
(CHEMBL3235134)Show InChI InChI=1S/C17H27N3O2/c18-10-4-8-17(16(21)22)9-7-15-14(11-17)19-12-20(15)13-5-2-1-3-6-13/h12-13H,1-11,18H2,(H,21,22)/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of TAF1a in human plasma assessed as clot lysis |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008269

(CHEMBL3235130)Show InChI InChI=1S/C10H14N2O2S/c1-6(15)8(10(13)14)4-7-2-3-9(11)12-5-7/h2-3,5-6,8,15H,4H2,1H3,(H2,11,12)(H,13,14)/t6-,8-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 87 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human TAF1a using hippuryl-L-arginine/hippuryl-L-lysine as substrate by liquid chromatographic analysis |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366807

(CHEMBL4172222)Show InChI InChI=1S/C30H47N5O15/c1-11(2)5-4-6-16(39)33-20-23(43)21(41)14(47-29(20)50-28-19(32-12(3)37)22(42)18(31)15(10-36)48-28)9-13(38)26-24(44)25(45)27(49-26)35-8-7-17(40)34-30(35)46/h4,6-8,11,13-15,18-29,36,38,41-45H,5,9-10,31H2,1-3H3,(H,32,37)(H,33,39)(H,34,40,46)/b6-4+/t13?,14-,15-,18-,19-,20-,21+,22+,23-,24+,25-,26-,27-,28-,29+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366839
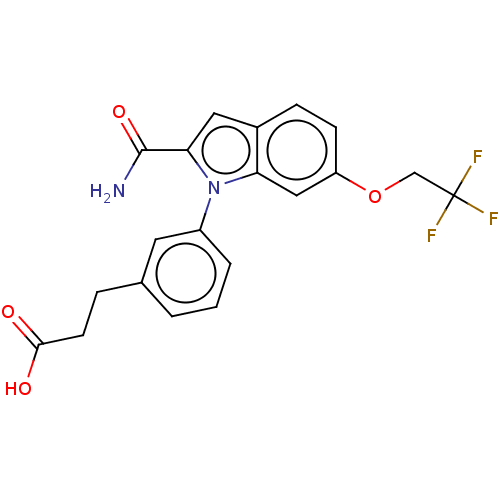
(CHEMBL4175583)Show SMILES NC(=O)c1cc2ccc(OCC(F)(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C20H17F3N2O4/c21-20(22,23)11-29-15-6-5-13-9-17(19(24)28)25(16(13)10-15)14-3-1-2-12(8-14)4-7-18(26)27/h1-3,5-6,8-10H,4,7,11H2,(H2,24,28)(H,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 93 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Mus musculus) | BDBM50366784

(CHEMBL4171084)Show SMILES NC(=O)c1cc2ccc(OC(F)(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C19H15F3N2O4/c20-19(21,22)28-14-6-5-12-9-16(18(23)27)24(15(12)10-14)13-3-1-2-11(8-13)4-7-17(25)26/h1-3,5-6,8-10H,4,7H2,(H2,23,27)(H,25,26) | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 96 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of mouse sPLA2-10 using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate pretreated for 20 mins followed by substrate additio... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008272

(CHEMBL3235133)Show SMILES Cl.CC(C)CCn1cnc2C[C@@](CCCN)(CCc12)C(O)=O |r| Show InChI InChI=1S/C16H27N3O2.ClH/c1-12(2)5-9-19-11-18-13-10-16(15(20)21,6-3-8-17)7-4-14(13)19;/h11-12H,3-10,17H2,1-2H3,(H,20,21);1H/t16-;/m1./s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of TAF1a in human plasma assessed as clot lysis |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008274
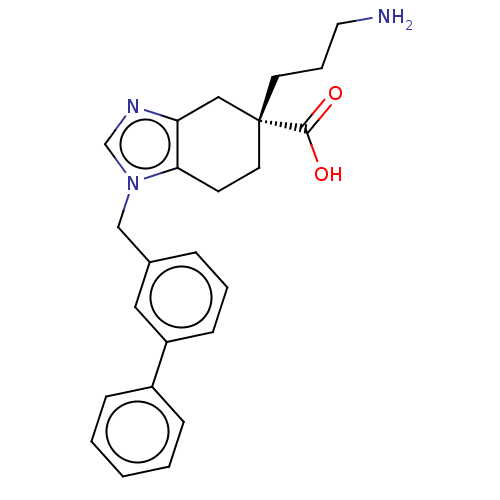
(CHEMBL3235135)Show SMILES NCCC[C@]1(CCc2c(C1)ncn2Cc1cccc(c1)-c1ccccc1)C(O)=O |r| Show InChI InChI=1S/C24H27N3O2/c25-13-5-11-24(23(28)29)12-10-22-21(15-24)26-17-27(22)16-18-6-4-9-20(14-18)19-7-2-1-3-8-19/h1-4,6-9,14,17H,5,10-13,15-16,25H2,(H,28,29)/t24-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 106 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human TAF1a |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50055366

((3-Aminooxalyl-1-benzyl-2-ethyl-1H-indol-4-yloxy)-...)Show SMILES CCc1c(C(=O)C(N)=O)c2c(OCC(O)=O)cccc2n1Cc1ccccc1 Show InChI InChI=1S/C21H20N2O5/c1-2-14-19(20(26)21(22)27)18-15(9-6-10-16(18)28-12-17(24)25)23(14)11-13-7-4-3-5-8-13/h3-10H,2,11-12H2,1H3,(H2,22,27)(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 107 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human sPLA2X using 1,2-bis(heptanoylthio) glycerophosphocholine substrate incubated for 30 mins |
Bioorg Med Chem Lett 24: 5251-5 (2014)
Article DOI: 10.1016/j.bmcl.2014.09.058
BindingDB Entry DOI: 10.7270/Q2668FS8 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366840
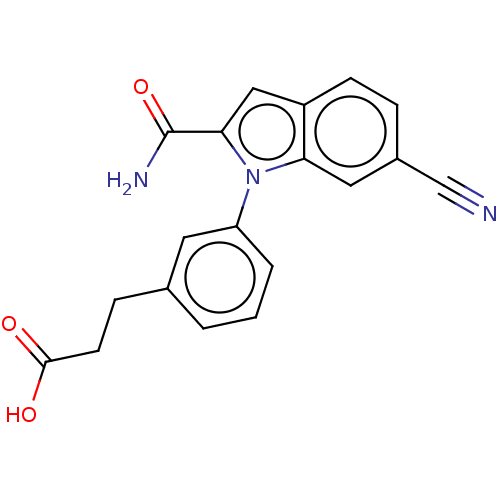
(CHEMBL4174522)Show SMILES NC(=O)c1cc2ccc(cc2n1-c1cccc(CCC(O)=O)c1)C#N Show InChI InChI=1S/C19H15N3O3/c20-11-13-4-6-14-10-17(19(21)25)22(16(14)9-13)15-3-1-2-12(8-15)5-7-18(23)24/h1-4,6,8-10H,5,7H2,(H2,21,25)(H,23,24) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008273

(CHEMBL3235134)Show InChI InChI=1S/C17H27N3O2/c18-10-4-8-17(16(21)22)9-7-15-14(11-17)19-12-20(15)13-5-2-1-3-6-13/h12-13H,1-11,18H2,(H,21,22)/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 117 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human TAF1a |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008275
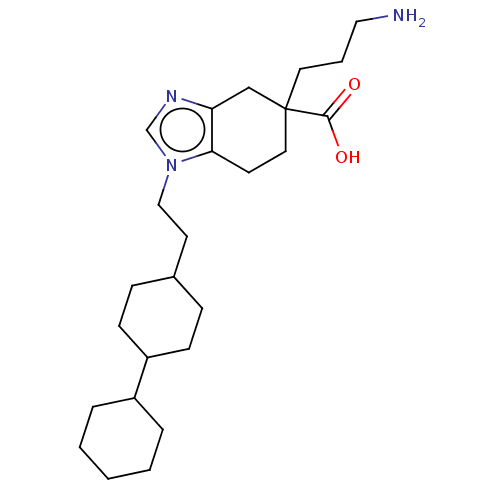
(CHEMBL3235136)Show SMILES NCCCC1(CCc2c(C1)ncn2CCC1CCC(CC1)C1CCCCC1)C(O)=O |(12.03,-18.01,;11.64,-19.5,;10.15,-19.91,;9.76,-21.4,;8.28,-21.8,;8.28,-20.26,;6.95,-19.48,;5.61,-20.25,;5.62,-21.8,;6.95,-22.56,;4.15,-22.29,;3.24,-21.04,;4.14,-19.78,;3.66,-18.32,;4.68,-17.17,;4.2,-15.71,;5.22,-14.56,;4.74,-13.1,;3.23,-12.78,;2.2,-13.93,;2.68,-15.4,;2.75,-11.32,;3.78,-10.18,;3.31,-8.72,;1.8,-8.39,;.77,-9.54,;1.25,-11.01,;9.04,-23.13,;8.26,-24.46,;10.58,-23.14,)| Show InChI InChI=1S/C25H41N3O2/c26-15-4-13-25(24(29)30)14-11-23-22(17-25)27-18-28(23)16-12-19-7-9-21(10-8-19)20-5-2-1-3-6-20/h18-21H,1-17,26H2,(H,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 124 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human TAF1a |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Phospholipase A2 group V
(Homo sapiens (Human)) | BDBM50055366

((3-Aminooxalyl-1-benzyl-2-ethyl-1H-indol-4-yloxy)-...)Show SMILES CCc1c(C(=O)C(N)=O)c2c(OCC(O)=O)cccc2n1Cc1ccccc1 Show InChI InChI=1S/C21H20N2O5/c1-2-14-19(20(26)21(22)27)18-15(9-6-10-16(18)28-12-17(24)25)23(14)11-13-7-4-3-5-8-13/h3-10H,2,11-12H2,1H3,(H2,22,27)(H,24,25) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 124 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-5 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate p... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50422368
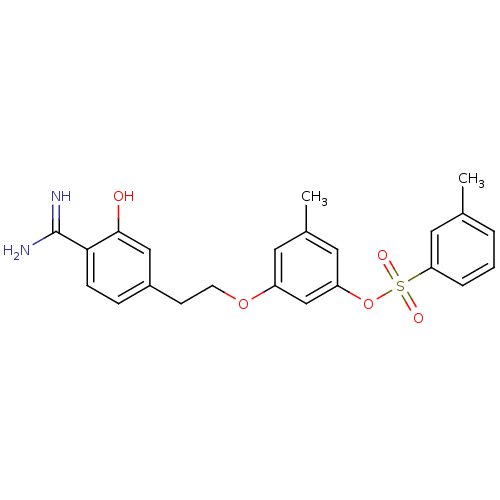
(CHEMBL117266)Show SMILES Cc1cccc(c1)S(=O)(=O)Oc1cc(C)cc(OCCc2ccc(C(N)=N)c(O)c2)c1 Show InChI InChI=1S/C23H24N2O5S/c1-15-4-3-5-20(12-15)31(27,28)30-19-11-16(2)10-18(14-19)29-9-8-17-6-7-21(23(24)25)22(26)13-17/h3-7,10-14,26H,8-9H2,1-2H3,(H3,24,25) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 135 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibitory activity against thrombin |
J Med Chem 44: 3424-39 (2001)
BindingDB Entry DOI: 10.7270/Q2D21ZWN |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50055366

((3-Aminooxalyl-1-benzyl-2-ethyl-1H-indol-4-yloxy)-...)Show SMILES CCc1c(C(=O)C(N)=O)c2c(OCC(O)=O)cccc2n1Cc1ccccc1 Show InChI InChI=1S/C21H20N2O5/c1-2-14-19(20(26)21(22)27)18-15(9-6-10-16(18)28-12-17(24)25)23(14)11-13-7-4-3-5-8-13/h3-10H,2,11-12H2,1H3,(H2,22,27)(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using HDL as substrate pretreated for 20 mins followed by substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366905
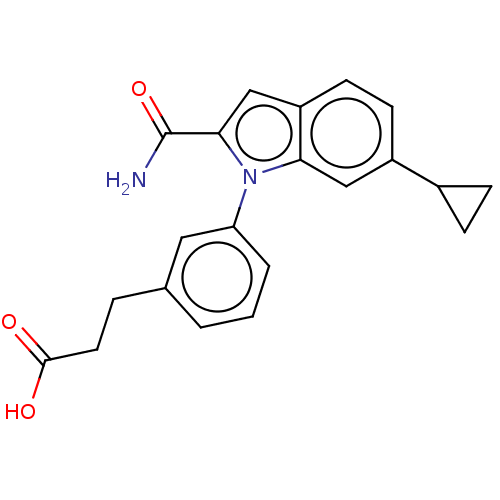
(CHEMBL4173359)Show SMILES NC(=O)c1cc2ccc(cc2n1-c1cccc(CCC(O)=O)c1)C1CC1 Show InChI InChI=1S/C21H20N2O3/c22-21(26)19-12-16-8-7-15(14-5-6-14)11-18(16)23(19)17-3-1-2-13(10-17)4-9-20(24)25/h1-3,7-8,10-12,14H,4-6,9H2,(H2,22,26)(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 160 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366889
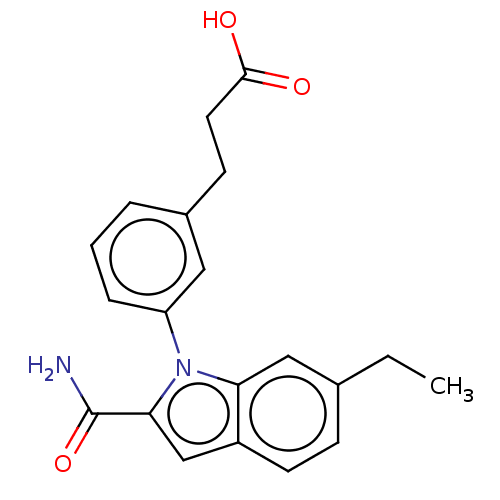
(CHEMBL4175643)Show SMILES CCc1ccc2cc(C(N)=O)n(-c3cccc(CCC(O)=O)c3)c2c1 Show InChI InChI=1S/C20H22F2N4O/c1-19-9-8-14-13-5-3-12(20(21,22)18-23-25-26-24-18)10-11(13)2-4-15(14)16(19)6-7-17(19)27/h3,5,10,14-16H,2,4,6-9H2,1H3,(H,23,24,25,26)/t14-,15-,16+,19+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50031105
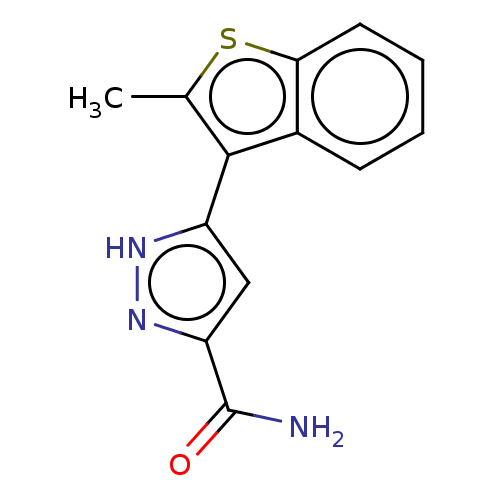
(CHEMBL3337976)Show InChI InChI=1S/C13H11N3OS/c1-7-12(8-4-2-3-5-11(8)18-7)9-6-10(13(14)17)16-15-9/h2-6H,1H3,(H2,14,17)(H,15,16) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 174 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human sPLA2X using 1,2-bis(heptanoylthio) glycerophosphocholine substrate incubated for 30 mins |
Bioorg Med Chem Lett 24: 5251-5 (2014)
Article DOI: 10.1016/j.bmcl.2014.09.058
BindingDB Entry DOI: 10.7270/Q2668FS8 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366876
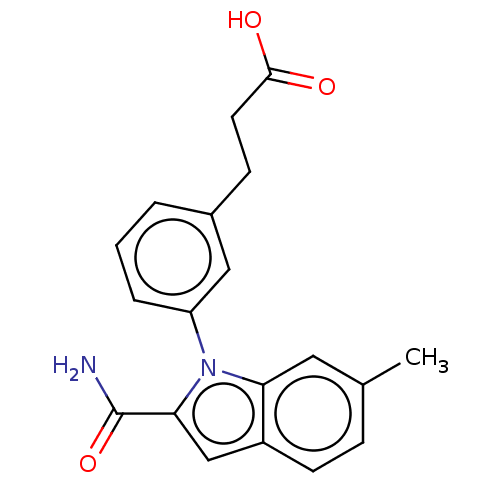
(CHEMBL4171797)Show SMILES Cc1ccc2cc(C(N)=O)n(-c3cccc(CCC(O)=O)c3)c2c1 Show InChI InChI=1S/C19H18N2O3/c1-12-5-7-14-11-17(19(20)24)21(16(14)9-12)15-4-2-3-13(10-15)6-8-18(22)23/h2-5,7,9-11H,6,8H2,1H3,(H2,20,24)(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Pantothenate synthetase
(Mycobacterium tuberculosis (strain ATCC 25618 / H3...) | BDBM50180855
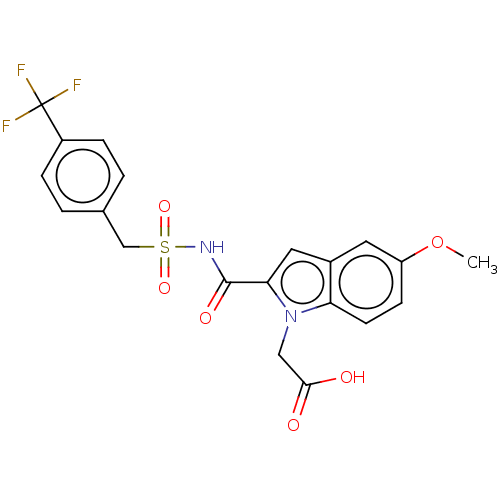
(CHEMBL3818314)Show SMILES COc1ccc2n(CC(O)=O)c(cc2c1)C(=O)NS(=O)(=O)Cc1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C20H17F3N2O6S/c1-31-15-6-7-16-13(8-15)9-17(25(16)10-18(26)27)19(28)24-32(29,30)11-12-2-4-14(5-3-12)20(21,22)23/h2-9H,10-11H2,1H3,(H,24,28)(H,26,27) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
Cambridge Crystallographic Data Centre
Curated by ChEMBL
| Assay Description
Inhibition of Mycobacterium tuberculosis pantothenate synthetase expressed in Escherichia coli |
J Med Chem 59: 4314-25 (2016)
Article DOI: 10.1021/acs.jmedchem.5b01980
BindingDB Entry DOI: 10.7270/Q2222WQV |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366968
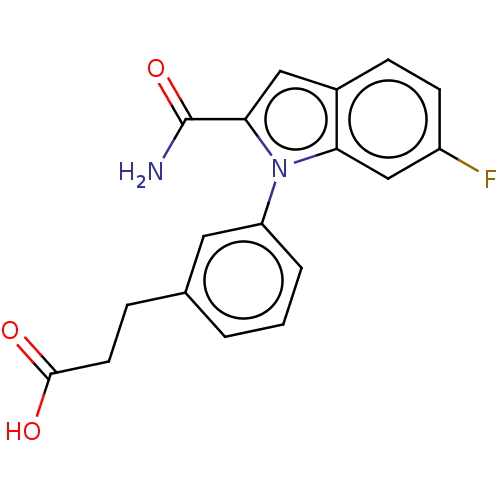
(CHEMBL4161605)Show InChI InChI=1S/C18H15FN2O3/c19-13-6-5-12-9-16(18(20)24)21(15(12)10-13)14-3-1-2-11(8-14)4-7-17(22)23/h1-3,5-6,8-10H,4,7H2,(H2,20,24)(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366784

(CHEMBL4171084)Show SMILES NC(=O)c1cc2ccc(OC(F)(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C19H15F3N2O4/c20-19(21,22)28-14-6-5-12-9-16(18(23)27)24(15(12)10-14)13-3-1-2-11(8-13)4-7-17(25)26/h1-3,5-6,8-10H,4,7H2,(H2,23,27)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using HDL as substrate pretreated for 20 mins followed by substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Prothrombin
(Homo sapiens (Human)) | BDBM50422377
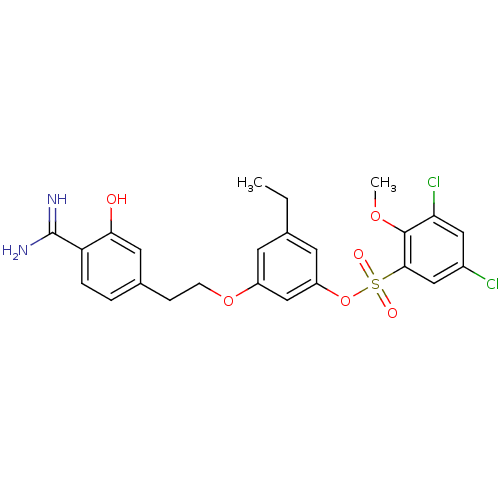
(CHEMBL116971)Show SMILES CCc1cc(OCCc2ccc(C(N)=N)c(O)c2)cc(OS(=O)(=O)c2cc(Cl)cc(Cl)c2OC)c1 Show InChI InChI=1S/C24H24Cl2N2O6S/c1-3-14-8-17(33-7-6-15-4-5-19(24(27)28)21(29)10-15)13-18(9-14)34-35(30,31)22-12-16(25)11-20(26)23(22)32-2/h4-5,8-13,29H,3,6-7H2,1-2H3,(H3,27,28) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 288 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibitory activity against thrombin |
J Med Chem 44: 3424-39 (2001)
BindingDB Entry DOI: 10.7270/Q2D21ZWN |
More data for this
Ligand-Target Pair | |
Phospholipase A2, membrane associated
(Homo sapiens (Human)) | BDBM50366784

(CHEMBL4171084)Show SMILES NC(=O)c1cc2ccc(OC(F)(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C19H15F3N2O4/c20-19(21,22)28-14-6-5-12-9-16(18(23)27)24(15(12)10-14)13-3-1-2-11(8-13)4-7-17(25)26/h1-3,5-6,8-10H,4,7H2,(H2,23,27)(H,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 310 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-2A expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50031107

(CHEMBL3337972)Show InChI InChI=1S/C10H11N3OS/c1-5-3-7(6(2)15-5)8-4-9(10(11)14)13-12-8/h3-4H,1-2H3,(H2,11,14)(H,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 316 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human sPLA2X using 1,2-bis(heptanoylthio) glycerophosphocholine substrate incubated for 30 mins |
Bioorg Med Chem Lett 24: 5251-5 (2014)
Article DOI: 10.1016/j.bmcl.2014.09.058
BindingDB Entry DOI: 10.7270/Q2668FS8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50031104
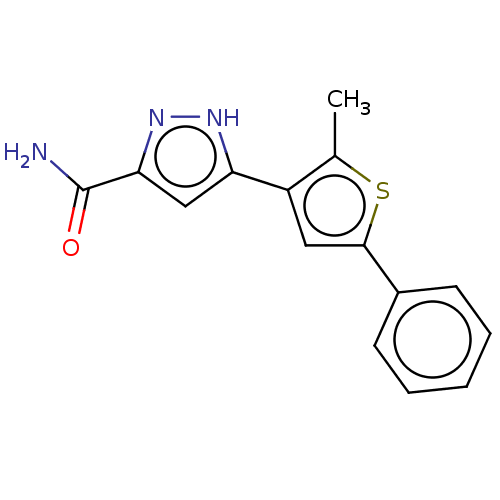
(CHEMBL3337977)Show InChI InChI=1S/C15H13N3OS/c1-9-11(12-8-13(15(16)19)18-17-12)7-14(20-9)10-5-3-2-4-6-10/h2-8H,1H3,(H2,16,19)(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 347 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human sPLA2X using 1,2-bis(heptanoylthio) glycerophosphocholine substrate incubated for 30 mins |
Bioorg Med Chem Lett 24: 5251-5 (2014)
Article DOI: 10.1016/j.bmcl.2014.09.058
BindingDB Entry DOI: 10.7270/Q2668FS8 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366983

(CHEMBL4160483)Show SMILES NC(=O)c1cc2ccc(OC(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C19H16F2N2O4/c20-19(21)27-14-6-5-12-9-16(18(22)26)23(15(12)10-14)13-3-1-2-11(8-13)4-7-17(24)25/h1-3,5-6,8-10,19H,4,7H2,(H2,22,26)(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 370 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using HDL as substrate pretreated for 20 mins followed by substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50031103

(CHEMBL3337978)Show InChI InChI=1S/C8H5Cl2N3OS/c9-6-1-3(7(10)15-6)4-2-5(8(11)14)13-12-4/h1-2H,(H2,11,14)(H,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 398 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human sPLA2X using 1,2-bis(heptanoylthio) glycerophosphocholine substrate incubated for 30 mins |
Bioorg Med Chem Lett 24: 5251-5 (2014)
Article DOI: 10.1016/j.bmcl.2014.09.058
BindingDB Entry DOI: 10.7270/Q2668FS8 |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008269

(CHEMBL3235130)Show InChI InChI=1S/C10H14N2O2S/c1-6(15)8(10(13)14)4-7-2-3-9(11)12-5-7/h2-3,5-6,8,15H,4H2,1H3,(H2,11,12)(H,13,14)/t6-,8-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of TAF1a in human plasma assessed as clot lysis |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50226610
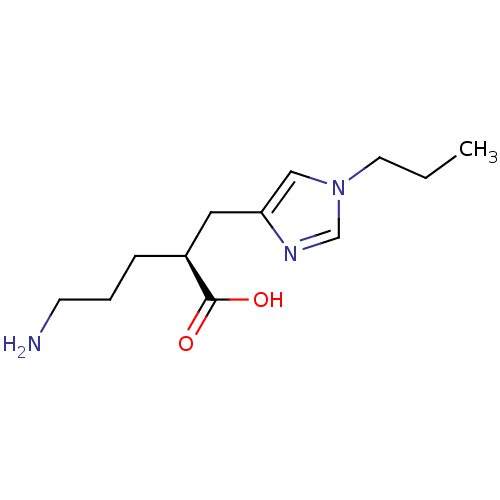
((2S)-5-AMINO-2-[(1-PROPYL-1H-IMIDAZOL-4-YL)METHYL]...)Show InChI InChI=1S/C12H21N3O2/c1-2-6-15-8-11(14-9-15)7-10(12(16)17)4-3-5-13/h8-10H,2-7,13H2,1H3,(H,16,17)/t10-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 450 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human TAF1a using hippuryl-L-arginine/hippuryl-L-lysine as substrate by liquid chromatographic analysis |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366811

(CHEMBL4176544)Show SMILES NC(=O)c1cc2ccc(cc2n1-c1cccc(CCC(O)=O)c1)C(F)(F)F Show InChI InChI=1S/C19H15F3N2O3/c20-19(21,22)13-6-5-12-9-16(18(23)27)24(15(12)10-13)14-3-1-2-11(8-14)4-7-17(25)26/h1-3,5-6,8-10H,4,7H2,(H2,23,27)(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 480 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using HDL as substrate pretreated for 20 mins followed by substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50422369
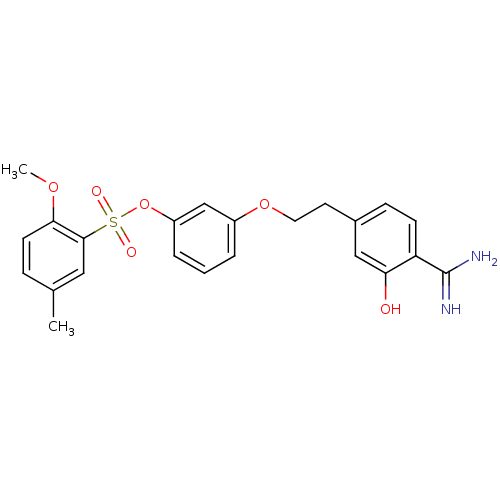
(CHEMBL114124)Show SMILES COc1ccc(C)cc1S(=O)(=O)Oc1cccc(OCCc2ccc(C(N)=N)c(O)c2)c1 Show InChI InChI=1S/C23H24N2O6S/c1-15-6-9-21(29-2)22(12-15)32(27,28)31-18-5-3-4-17(14-18)30-11-10-16-7-8-19(23(24)25)20(26)13-16/h3-9,12-14,26H,10-11H2,1-2H3,(H3,24,25) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 575 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibitory activity against thrombin |
J Med Chem 44: 3424-39 (2001)
BindingDB Entry DOI: 10.7270/Q2D21ZWN |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008276
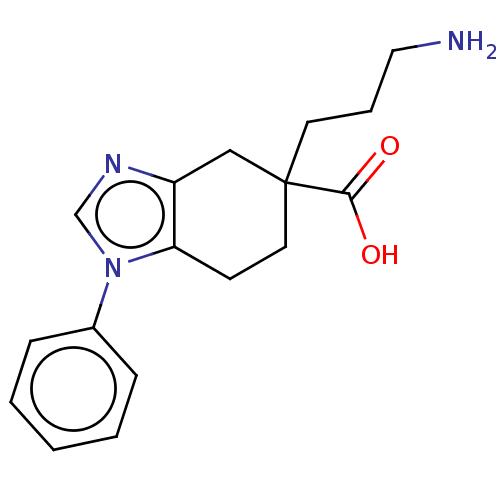
(CHEMBL3235137)Show InChI InChI=1S/C17H21N3O2/c18-10-4-8-17(16(21)22)9-7-15-14(11-17)19-12-20(15)13-5-2-1-3-6-13/h1-3,5-6,12H,4,7-11,18H2,(H,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 585 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human TAF1a |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Carboxypeptidase B2
(Homo sapiens (Human)) | BDBM50008270
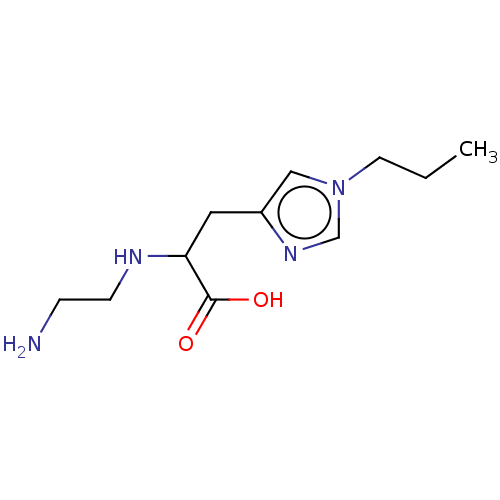
(CHEMBL3235131)Show InChI InChI=1S/C11H20N4O2/c1-2-5-15-7-9(14-8-15)6-10(11(16)17)13-4-3-12/h7-8,10,13H,2-6,12H2,1H3,(H,16,17) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 620 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human TAF1a using hippuryl-L-arginine/hippuryl-L-lysine as substrate by liquid chromatographic analysis |
Bioorg Med Chem 22: 2261-8 (2014)
Article DOI: 10.1016/j.bmc.2014.02.010
BindingDB Entry DOI: 10.7270/Q2DZ09TG |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366970
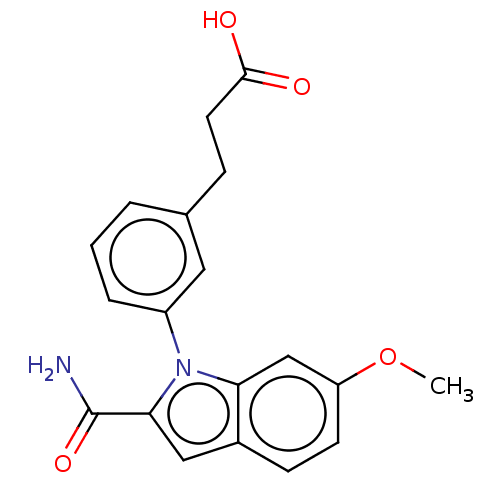
(CHEMBL4160502)Show SMILES COc1ccc2cc(C(N)=O)n(-c3cccc(CCC(O)=O)c3)c2c1 Show InChI InChI=1S/C19H18N2O4/c1-25-15-7-6-13-10-17(19(20)24)21(16(13)11-15)14-4-2-3-12(9-14)5-8-18(22)23/h2-4,6-7,9-11H,5,8H2,1H3,(H2,20,24)(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 620 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine as substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50031102
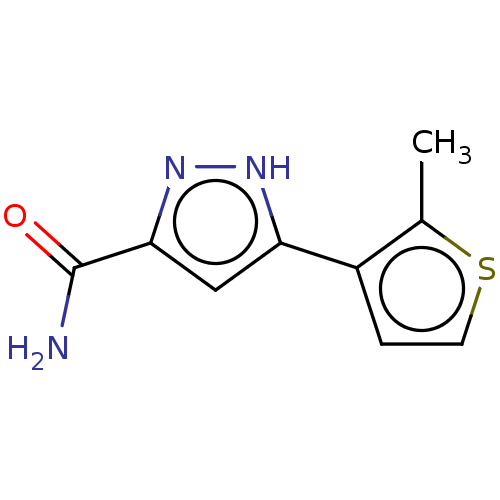
(CHEMBL3337979)Show InChI InChI=1S/C9H9N3OS/c1-5-6(2-3-14-5)7-4-8(9(10)13)12-11-7/h2-4H,1H3,(H2,10,13)(H,11,12) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 631 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human sPLA2X using 1,2-bis(heptanoylthio) glycerophosphocholine substrate incubated for 30 mins |
Bioorg Med Chem Lett 24: 5251-5 (2014)
Article DOI: 10.1016/j.bmcl.2014.09.058
BindingDB Entry DOI: 10.7270/Q2668FS8 |
More data for this
Ligand-Target Pair | |
Group 10 secretory phospholipase A2
(Homo sapiens (Human)) | BDBM50366839

(CHEMBL4175583)Show SMILES NC(=O)c1cc2ccc(OCC(F)(F)F)cc2n1-c1cccc(CCC(O)=O)c1 Show InChI InChI=1S/C20H17F3N2O4/c21-20(22,23)11-29-15-6-5-13-9-17(19(24)28)25(16(13)10-15)14-3-1-2-12(8-14)4-7-18(26)27/h1-3,5-6,8-10H,4,7,11H2,(H2,24,28)(H,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 740 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human sPLA2-10 expressed in Escherichia coli BL21(DE3) using HDL as substrate pretreated for 20 mins followed by substrate ... |
ACS Med Chem Lett 9: 594-599 (2018)
Article DOI: 10.1021/acsmedchemlett.7b00505
BindingDB Entry DOI: 10.7270/Q2Z60RMJ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data