 Found 16142 hits with Last Name = 'lu' and Initial = 'w'
Found 16142 hits with Last Name = 'lu' and Initial = 'w' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
neuronal acetylcholine receptor subunit alpha-2
(Xenopus) | BDBM86041
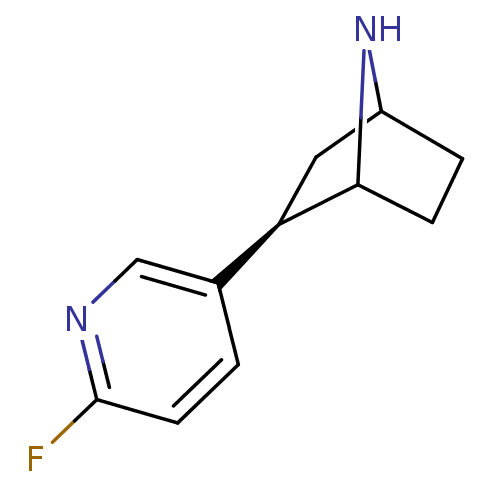
(NFEP)Show SMILES Fc1ccc(cn1)[C@H]1CC2CCC1N2 |r,TLB:4:7:10.11:13| Show InChI InChI=1S/C11H13FN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2/t8?,9-,10?/m1/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50056769
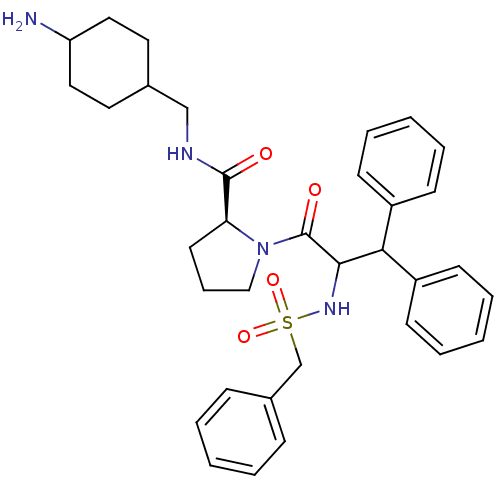
((S)-1-(3,3-Diphenyl-2-phenylmethanesulfonylamino-p...)Show SMILES NC1CCC(CNC(=O)[C@@H]2CCCN2C(=O)C(NS(=O)(=O)Cc2ccccc2)C(c2ccccc2)c2ccccc2)CC1 |wU:9.8,(16.05,-4.09,;16.82,-5.42,;16.81,-7.08,;17.34,-8.54,;16.12,-9.36,;16.57,-10.83,;15.52,-11.95,;14.02,-11.6,;12.97,-12.72,;13.56,-10.13,;14.5,-8.9,;13.63,-7.64,;12.11,-8.1,;12.12,-9.64,;10.86,-10.51,;10.97,-12.05,;9.46,-9.85,;8.2,-10.74,;6.87,-11.5,;6.09,-10.16,;7.64,-12.86,;5.51,-12.28,;4.18,-11.51,;4.18,-9.97,;2.85,-9.2,;1.52,-9.97,;1.52,-11.53,;2.85,-12.28,;9.33,-8.33,;7.93,-7.68,;7.8,-6.14,;6.4,-5.49,;5.14,-6.38,;5.28,-7.92,;6.68,-8.56,;10.59,-7.44,;9.81,-6.11,;10.59,-4.77,;12.13,-4.77,;12.89,-6.11,;12.13,-7.44,;16.29,-7.89,;15.73,-6.52,)| Show InChI InChI=1S/C34H42N4O4S/c35-29-20-18-25(19-21-29)23-36-33(39)30-17-10-22-38(30)34(40)32(37-43(41,42)24-26-11-4-1-5-12-26)31(27-13-6-2-7-14-27)28-15-8-3-9-16-28/h1-9,11-16,25,29-32,37H,10,17-24,35H2,(H,36,39)/t25?,29?,30-,32?/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards thrombin |
J Med Chem 40: 830-2 (1997)
Article DOI: 10.1021/jm960762y
BindingDB Entry DOI: 10.7270/Q25H7GXW |
More data for this
Ligand-Target Pair | |
cholinergic receptor, nicotinic, alpha 1 (Muscle) isoform X1
(Xenopus) | BDBM86042

(NEP)Show InChI InChI=1S/C11H14N2/c1-2-8(7-12-5-1)10-6-9-3-4-11(10)13-9/h1-2,5,7,9-11,13H,3-4,6H2/t9?,10-,11?/m1/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
cholinergic receptor, nicotinic, alpha 1 (Muscle) isoform X1
(Xenopus) | BDBM86041

(NFEP)Show SMILES Fc1ccc(cn1)[C@H]1CC2CCC1N2 |r,TLB:4:7:10.11:13| Show InChI InChI=1S/C11H13FN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2/t8?,9-,10?/m1/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
cholinergic receptor, nicotinic, alpha 1 (Muscle) isoform X1
(Xenopus) | BDBM86040
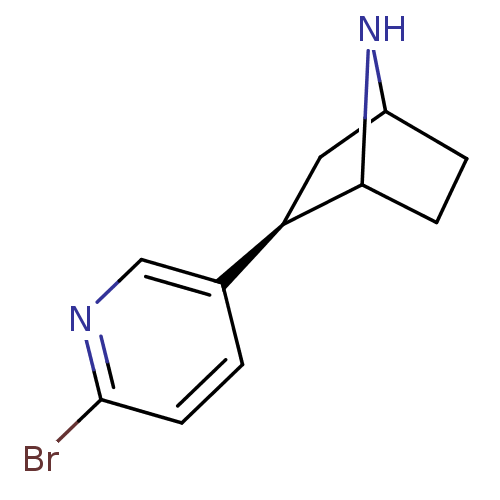
(NBEP)Show SMILES Brc1ccc(cn1)[C@H]1CC2CCC1N2 |r,TLB:4:7:10.11:13| Show InChI InChI=1S/C11H13BrN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2/t8?,9-,10?/m1/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
neuronal acetylcholine receptor subunit alpha-4
(Xenopus) | BDBM86042

(NEP)Show InChI InChI=1S/C11H14N2/c1-2-8(7-12-5-1)10-6-9-3-4-11(10)13-9/h1-2,5,7,9-11,13H,3-4,6H2/t9?,10-,11?/m1/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
neuronal acetylcholine receptor subunit alpha-4
(Xenopus) | BDBM86041

(NFEP)Show SMILES Fc1ccc(cn1)[C@H]1CC2CCC1N2 |r,TLB:4:7:10.11:13| Show InChI InChI=1S/C11H13FN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2/t8?,9-,10?/m1/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00920 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM25761
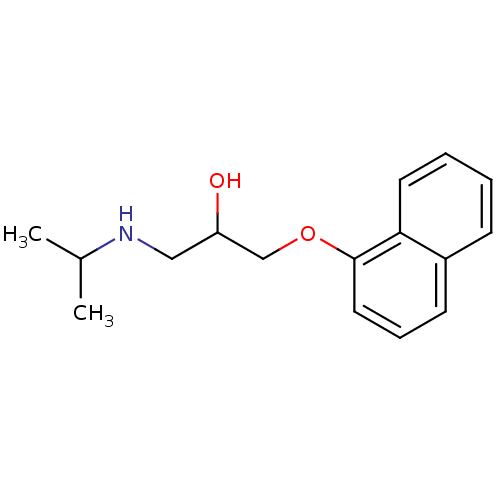
(Anapriline | Avlocardyl | CHEMBL27 | PROPANOLOL(-)...)Show InChI InChI=1S/C16H21NO2/c1-12(2)17-10-14(18)11-19-16-9-5-7-13-6-3-4-8-15(13)16/h3-9,12,14,17-18H,10-11H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
neuronal acetylcholine receptor subunit alpha-4
(Xenopus) | BDBM86040

(NBEP)Show SMILES Brc1ccc(cn1)[C@H]1CC2CCC1N2 |r,TLB:4:7:10.11:13| Show InChI InChI=1S/C11H13BrN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2/t8?,9-,10?/m1/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
neuronal acetylcholine receptor subunit alpha-2
(Xenopus) | BDBM86040

(NBEP)Show SMILES Brc1ccc(cn1)[C@H]1CC2CCC1N2 |r,TLB:4:7:10.11:13| Show InChI InChI=1S/C11H13BrN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2/t8?,9-,10?/m1/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM25761

(Anapriline | Avlocardyl | CHEMBL27 | PROPANOLOL(-)...)Show InChI InChI=1S/C16H21NO2/c1-12(2)17-10-14(18)11-19-16-9-5-7-13-6-3-4-8-15(13)16/h3-9,12,14,17-18H,10-11H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase ROS
(Homo sapiens (Human)) | BDBM50448785
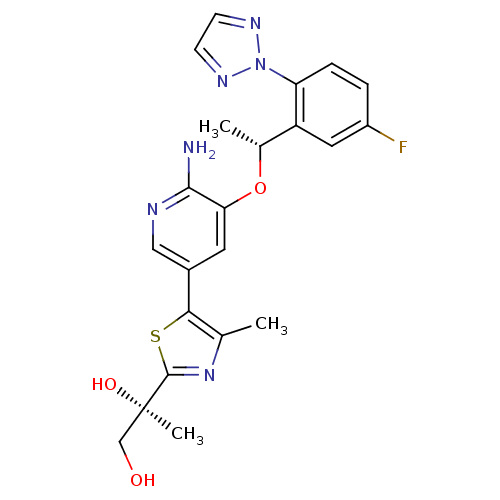
(CHEMBL3128069)Show SMILES C[C@@H](Oc1cc(cnc1N)-c1sc(nc1C)[C@](C)(O)CO)c1cc(F)ccc1-n1nccn1 |r| Show InChI InChI=1S/C22H23FN6O3S/c1-12-19(33-21(28-12)22(3,31)11-30)14-8-18(20(24)25-10-14)32-13(2)16-9-15(23)4-5-17(16)29-26-6-7-27-29/h4-10,13,30-31H,11H2,1-3H3,(H2,24,25)/t13-,22-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of ROS1 (unknown origin) by Pfizer mobility shift assay |
J Med Chem 57: 1170-87 (2014)
Article DOI: 10.1021/jm401805h
BindingDB Entry DOI: 10.7270/Q29C6ZX5 |
More data for this
Ligand-Target Pair | |
neuronal acetylcholine receptor subunit alpha-2
(Xenopus) | BDBM86042

(NEP)Show InChI InChI=1S/C11H14N2/c1-2-8(7-12-5-1)10-6-9-3-4-11(10)13-9/h1-2,5,7,9-11,13H,3-4,6H2/t9?,10-,11?/m1/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50345133
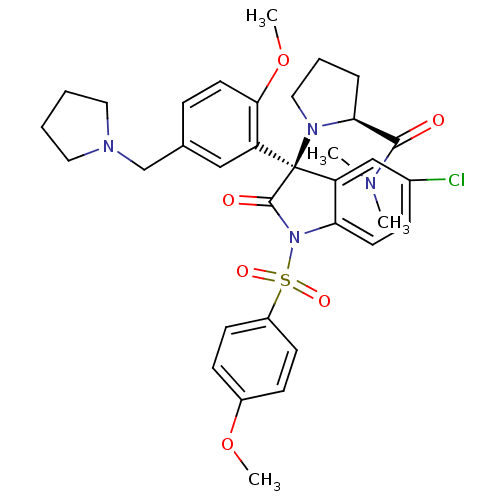
((S)-1-((R)-5-chloro-3-(2-methoxy-5-(pyrrolidin-1-y...)Show SMILES COc1ccc(cc1)S(=O)(=O)N1C(=O)[C@@](N2CCC[C@H]2C(=O)N(C)C)(c2cc(Cl)ccc12)c1cc(CN2CCCC2)ccc1OC |r| Show InChI InChI=1S/C34H39ClN4O6S/c1-36(2)32(40)30-8-7-19-38(30)34(28-20-23(9-16-31(28)45-4)22-37-17-5-6-18-37)27-21-24(35)10-15-29(27)39(33(34)41)46(42,43)26-13-11-25(44-3)12-14-26/h9-16,20-21,30H,5-8,17-19,22H2,1-4H3/t30-,34+/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from human vasopressin V1b receptor expressed in CHO cells by scintillation counting |
Bioorg Med Chem Lett 21: 3828-31 (2011)
Article DOI: 10.1016/j.bmcl.2011.03.012
BindingDB Entry DOI: 10.7270/Q20P10BC |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 4
(Homo sapiens (Human)) | BDBM82505
(CAS_121881 | NSC_121881 | SB204070)Show InChI InChI=1S/C19H27ClN2O4/c1-2-3-6-22-7-4-13(5-8-22)12-26-19(23)14-11-15(20)16(21)18-17(14)24-9-10-25-18/h11,13H,2-10,12,21H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
J Neurochem 74: 478-89 (2000)
Article DOI: 10.1046/j.1471-4159.2000.740478.x
BindingDB Entry DOI: 10.7270/Q2DJ5D5Z |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50345134
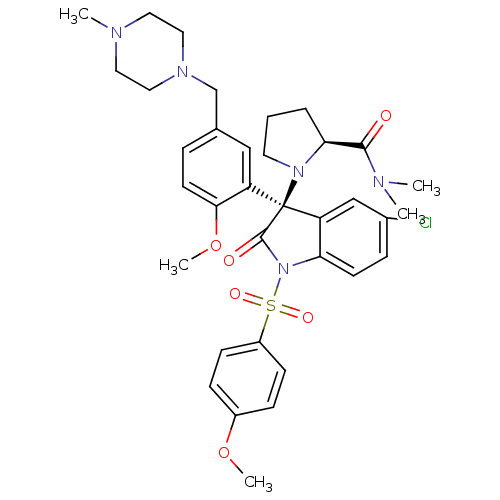
((S)-1-((R)-5-chloro-3-(2-methoxy-5-((4-methylpiper...)Show SMILES COc1ccc(cc1)S(=O)(=O)N1C(=O)[C@@](N2CCC[C@H]2C(=O)N(C)C)(c2cc(Cl)ccc12)c1cc(CN2CCN(C)CC2)ccc1OC |r| Show InChI InChI=1S/C35H42ClN5O6S/c1-37(2)33(42)31-7-6-16-40(31)35(29-21-24(8-15-32(29)47-5)23-39-19-17-38(3)18-20-39)28-22-25(36)9-14-30(28)41(34(35)43)48(44,45)27-12-10-26(46-4)11-13-27/h8-15,21-22,31H,6-7,16-20,23H2,1-5H3/t31-,35+/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from human vasopressin V1b receptor expressed in CHO cells by scintillation counting |
Bioorg Med Chem Lett 21: 3828-31 (2011)
Article DOI: 10.1016/j.bmcl.2011.03.012
BindingDB Entry DOI: 10.7270/Q20P10BC |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50345131
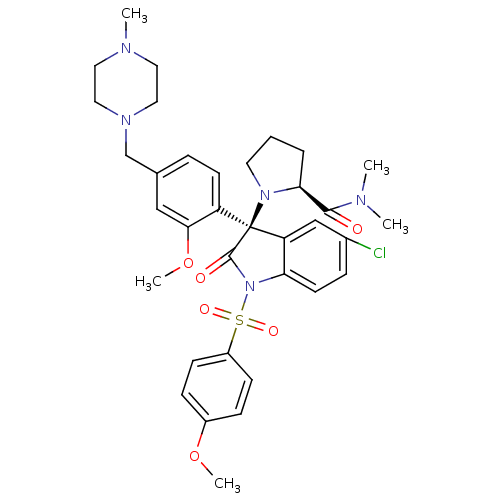
((S)-1-((R)-5-chloro-3-(2-methoxy-4-((4-methylpiper...)Show SMILES COc1ccc(cc1)S(=O)(=O)N1C(=O)[C@@](N2CCC[C@H]2C(=O)N(C)C)(c2cc(Cl)ccc12)c1ccc(CN2CCN(C)CC2)cc1OC |r| Show InChI InChI=1S/C35H42ClN5O6S/c1-37(2)33(42)31-7-6-16-40(31)35(28-14-8-24(21-32(28)47-5)23-39-19-17-38(3)18-20-39)29-22-25(36)9-15-30(29)41(34(35)43)48(44,45)27-12-10-26(46-4)11-13-27/h8-15,21-22,31H,6-7,16-20,23H2,1-5H3/t31-,35-/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from human vasopressin V1b receptor expressed in CHO cells by scintillation counting |
Bioorg Med Chem Lett 21: 3828-31 (2011)
Article DOI: 10.1016/j.bmcl.2011.03.012
BindingDB Entry DOI: 10.7270/Q20P10BC |
More data for this
Ligand-Target Pair | |
Cholinergic receptor, nicotinic alpha 1
(Xenopus) | BDBM86040

(NBEP)Show SMILES Brc1ccc(cn1)[C@H]1CC2CCC1N2 |r,TLB:4:7:10.11:13| Show InChI InChI=1S/C11H13BrN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2/t8?,9-,10?/m1/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepacivirus C) | BDBM50485494
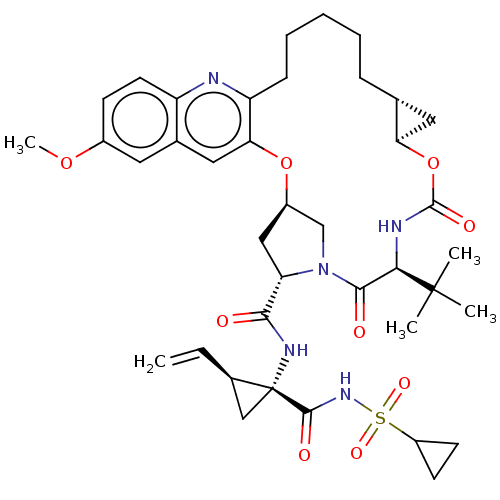
(CHEMBL2063089)Show SMILES [H][C@@]12C[C@@]1([H])OC(=O)N[C@H](C(=O)N1C[C@@]([H])(C[C@H]1C(=O)N[C@@]1(C[C@@]1([H])C=C)C(=O)NS(=O)(=O)C1CC1)Oc1cc3cc(OC)ccc3nc1CCCCC2)C(C)(C)C |r| Show InChI InChI=1S/C39H51N5O9S/c1-6-24-20-39(24,36(47)43-54(49,50)27-13-14-27)42-34(45)30-19-26-21-44(30)35(46)33(38(2,3)4)41-37(48)53-31-17-22(31)10-8-7-9-11-29-32(52-26)18-23-16-25(51-5)12-15-28(23)40-29/h6,12,15-16,18,22,24,26-27,30-31,33H,1,7-11,13-14,17,19-21H2,2-5H3,(H,41,48)(H,42,45)(H,43,47)/t22-,24-,26-,30+,31-,33-,39-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/TrEMBL
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus (isolate BK) genotype 1b NS3/4a protease D168V mutant expressed in Escherichia coli by time-resolved fluorescence ana... |
ACS Med Chem Lett 3: 332-6 (2012)
Article DOI: 10.1021/ml300017p
BindingDB Entry DOI: 10.7270/Q2KH0R6V |
More data for this
Ligand-Target Pair | |
neuronal acetylcholine receptor subunit alpha-2
(Xenopus) | BDBM86040

(NBEP)Show SMILES Brc1ccc(cn1)[C@H]1CC2CCC1N2 |r,TLB:4:7:10.11:13| Show InChI InChI=1S/C11H13BrN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2/t8?,9-,10?/m1/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50057826

((S)-1-((R)-2-Amino-3,3-dicyclohexyl-propionyl)-pyr...)Show SMILES N[C@H](C(C1CCCCC1)C1CCCCC1)C(=O)N1CCC[C@H]1C(=O)NCC1CCC(N)CC1 |wU:21.24,wD:1.0,(8.57,-5.29,;9.89,-4.53,;9.89,-2.99,;11.24,-2.22,;12.55,-2.99,;13.9,-2.22,;13.9,-.68,;12.58,.09,;11.23,-.68,;8.57,-2.21,;7.23,-2.98,;5.9,-2.21,;5.9,-.67,;7.23,.1,;8.57,-.67,;11.24,-5.3,;11.24,-6.84,;12.72,-4.89,;13.26,-3.44,;14.8,-3.51,;15.22,-5,;13.92,-5.85,;13.86,-7.39,;12.49,-8.1,;15.15,-8.21,;16.52,-7.51,;17.82,-8.32,;17.78,-9.66,;18.86,-11.05,;17.78,-12.17,;18.86,-13.25,;17.85,-10.92,;16.76,-9.44,)| Show InChI InChI=1S/C27H48N4O2/c28-22-15-13-19(14-16-22)18-30-26(32)23-12-7-17-31(23)27(33)25(29)24(20-8-3-1-4-9-20)21-10-5-2-6-11-21/h19-25H,1-18,28-29H2,(H,30,32)/t19?,22?,23-,25+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory activity against human thrombin |
J Med Chem 40: 1565-9 (1997)
Article DOI: 10.1021/jm970140s
BindingDB Entry DOI: 10.7270/Q2PR7V29 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 4
(Homo sapiens (Human)) | BDBM82505

(CAS_121881 | NSC_121881 | SB204070)Show InChI InChI=1S/C19H27ClN2O4/c1-2-3-6-22-7-4-13(5-8-22)12-26-19(23)14-11-15(20)16(21)18-17(14)24-9-10-25-18/h11,13H,2-10,12,21H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
J Neurochem 74: 478-89 (2000)
Article DOI: 10.1046/j.1471-4159.2000.740478.x
BindingDB Entry DOI: 10.7270/Q2DJ5D5Z |
More data for this
Ligand-Target Pair | |
Genome polyprotein
(Hepacivirus C) | BDBM50485491
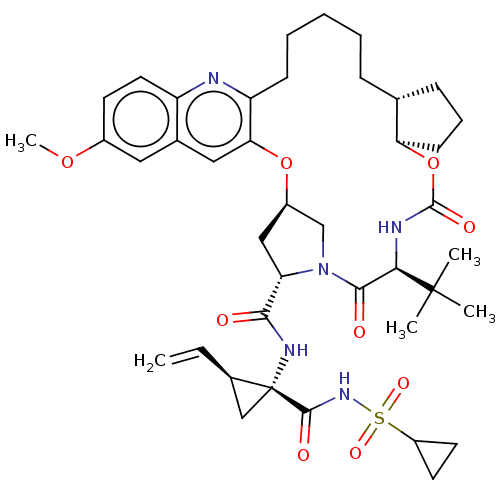
(CHEMBL2063088)Show SMILES [H][C@@]12C[C@H](N(C1)C(=O)[C@@H](NC(=O)O[C@]1([H])CCC[C@@]1([H])CCCCCc1nc3ccc(OC)cc3cc1O2)C(C)(C)C)C(=O)N[C@@]1(C[C@H]1C=C)C(=O)NS(=O)(=O)C1CC1 |r| Show InChI InChI=1S/C41H55N5O9S/c1-6-26-22-41(26,38(49)45-56(51,52)29-16-17-29)44-36(47)32-21-28-23-46(32)37(48)35(40(2,3)4)43-39(50)55-33-14-10-12-24(33)11-8-7-9-13-31-34(54-28)20-25-19-27(53-5)15-18-30(25)42-31/h6,15,18-20,24,26,28-29,32-33,35H,1,7-14,16-17,21-23H2,2-5H3,(H,43,50)(H,44,47)(H,45,49)/t24-,26-,28-,32+,33-,35-,41-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/TrEMBL
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus (isolate BK) genotype 1b NS3/4a protease D168V mutant expressed in Escherichia coli by time-resolved fluorescence ana... |
ACS Med Chem Lett 3: 332-6 (2012)
Article DOI: 10.1021/ml300017p
BindingDB Entry DOI: 10.7270/Q2KH0R6V |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50451823
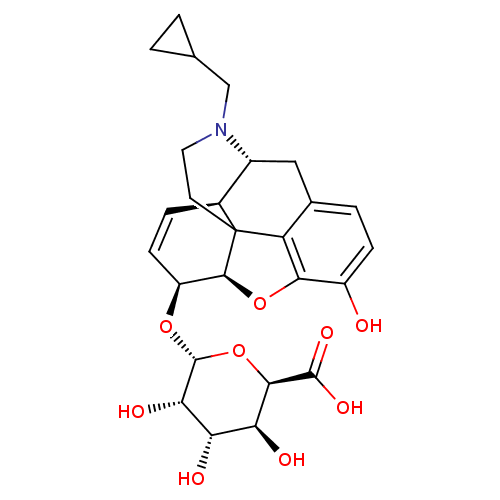
(CHEMBL2112840)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC6CC6)CCC14[C@@]5([H])C=C[C@@H]2O[C@@H]1O[C@H]([C@@H](O)[C@H](O)[C@@H]1O)C(O)=O)ccc3O |c:23,THB:3:4:17:9.15.14,10:9:17:4.5.6| Show InChI InChI=1S/C26H31NO9/c28-15-5-3-12-9-14-13-4-6-16(34-25-20(31)18(29)19(30)22(36-25)24(32)33)23-26(13,17(12)21(15)35-23)7-8-27(14)10-11-1-2-11/h3-6,11,13-14,16,18-20,22-23,25,28-31H,1-2,7-10H2,(H,32,33)/t13-,14+,16-,18-,19-,20-,22+,23-,25+,26?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Ultrafine UFC Ltd
Curated by ChEMBL
| Assay Description
mu-1 receptor binding affinity in rat brain by 3H [d-Ala2, d-Leu5] enkephalin displacement. |
Bioorg Med Chem Lett 13: 1207-14 (2003)
BindingDB Entry DOI: 10.7270/Q2251JQC |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50192770
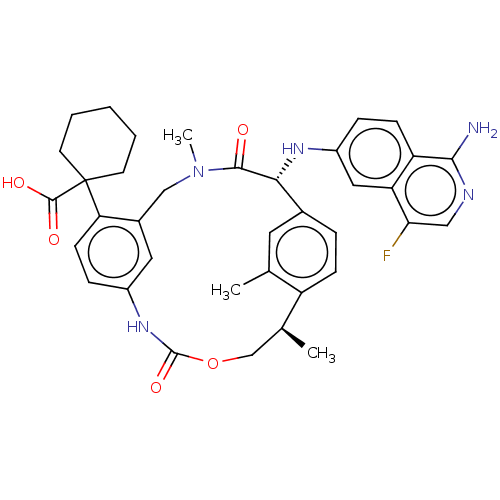
(CHEMBL3956096)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCCCC1)C(O)=O |r| Show InChI InChI=1S/C37H40FN5O5/c1-21-15-23-7-10-27(21)22(2)20-48-36(47)42-25-9-12-30(37(35(45)46)13-5-4-6-14-37)24(16-25)19-43(3)34(44)32(23)41-26-8-11-28-29(17-26)31(38)18-40-33(28)39/h7-12,15-18,22,32,41H,4-6,13-14,19-20H2,1-3H3,(H2,39,40)(H,42,47)(H,45,46)/t22-,32+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
| PDB
Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TF-factor 7a using factor 10 as substrate at 37 degC |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
neuronal acetylcholine receptor subunit alpha-2
(Xenopus) | BDBM86045
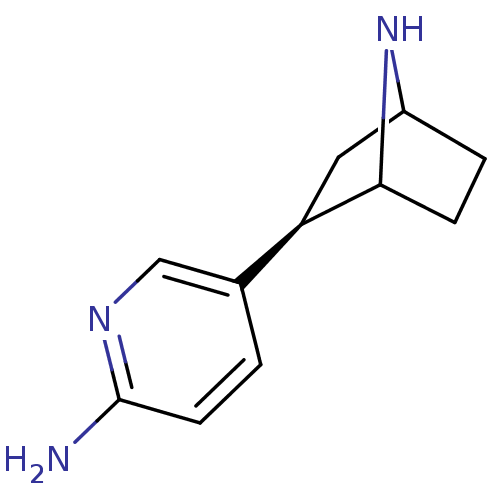
(NNEP)Show SMILES Nc1ccc(cn1)[C@H]1CC2CCC1N2 |r,TLB:4:7:10.11:13| Show InChI InChI=1S/C11H15N3/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2,(H2,12,13)/t8?,9-,10?/m1/s1 | PDB
Reactome pathway
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM85034

(BW-501 | BW-501C | BW501)Show InChI InChI=1S/C17H20ClN3O/c18-15-9-4-5-10-16(15)22-12-6-11-20-17(19)13-21-14-7-2-1-3-8-14/h1-5,7-10,21H,6,11-13H2,(H2,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 4
(GUINEA PIG) | BDBM82505

(CAS_121881 | NSC_121881 | SB204070)Show InChI InChI=1S/C19H27ClN2O4/c1-2-3-6-22-7-4-13(5-8-22)12-26-19(23)14-11-15(20)16(21)18-17(14)24-9-10-25-18/h11,13H,2-10,12,21H2,1H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
J Neurochem 69: 1810-9 (1997)
Article DOI: 10.1046/j.1471-4159.1997.69051810.x
BindingDB Entry DOI: 10.7270/Q2Z60MMW |
More data for this
Ligand-Target Pair | |
cholinergic receptor, nicotinic, alpha 1 (Muscle) isoform X1
(Xenopus) | BDBM86045

(NNEP)Show SMILES Nc1ccc(cn1)[C@H]1CC2CCC1N2 |r,TLB:4:7:10.11:13| Show InChI InChI=1S/C11H15N3/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2,(H2,12,13)/t8?,9-,10?/m1/s1 | PDB
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0770 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Miami
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 1246-52 (2002)
Article DOI: 10.1124/jpet.102.035899
BindingDB Entry DOI: 10.7270/Q2HQ3XG3 |
More data for this
Ligand-Target Pair | |
Vasopressin V1b receptor
(Homo sapiens (Human)) | BDBM50345130
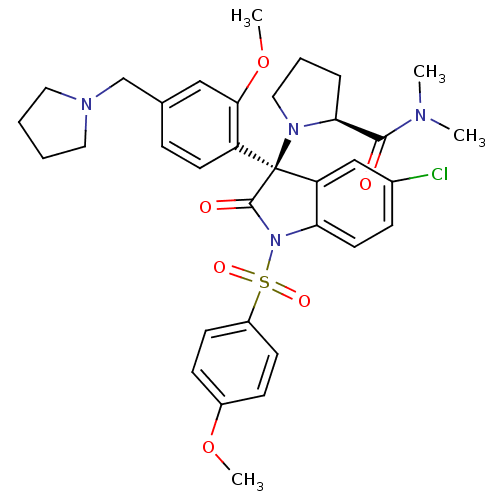
((S)-1-((R)-5-chloro-3-(2-methoxy-4-(pyrrolidin-1-y...)Show SMILES COc1ccc(cc1)S(=O)(=O)N1C(=O)[C@@](N2CCC[C@H]2C(=O)N(C)C)(c2cc(Cl)ccc12)c1ccc(CN2CCCC2)cc1OC |r| Show InChI InChI=1S/C34H39ClN4O6S/c1-36(2)32(40)30-8-7-19-38(30)34(27-15-9-23(20-31(27)45-4)22-37-17-5-6-18-37)28-21-24(35)10-16-29(28)39(33(34)41)46(42,43)26-13-11-25(44-3)12-14-26/h9-16,20-21,30H,5-8,17-19,22H2,1-4H3/t30-,34-/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from human vasopressin V1b receptor expressed in CHO cells by scintillation counting |
Bioorg Med Chem Lett 21: 3828-31 (2011)
Article DOI: 10.1016/j.bmcl.2011.03.012
BindingDB Entry DOI: 10.7270/Q20P10BC |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50454822
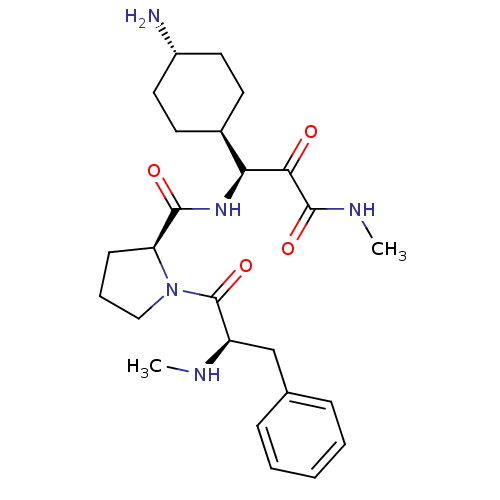
(CHEMBL2062141 | L-370518)Show SMILES [H][C@@](NC(=O)[C@@H]1CCCN1C(=O)[C@@H](Cc1ccccc1)NC)(C(=O)C(=O)NC)[C@@]1([H])CC[C@H](N)CC1 |wU:1.0,wD:12.13,5.4,32.34,28.30,(9.54,-15.25,;8.45,-16.34,;7.42,-17.49,;5.92,-17.17,;5.44,-15.7,;4.89,-18.31,;5.21,-19.82,;3.87,-20.59,;2.73,-19.56,;3.35,-18.15,;2.58,-16.82,;3.35,-15.48,;1.04,-16.82,;.27,-15.48,;1.04,-14.15,;2.58,-14.15,;3.35,-12.82,;2.58,-11.48,;1.04,-11.48,;.27,-12.82,;.27,-18.15,;-1.27,-18.15,;7.98,-14.88,;6.47,-14.56,;9.01,-13.73,;10.51,-14.05,;8.53,-12.27,;9.56,-11.12,;9.96,-16.66,;8.87,-17.75,;10.99,-15.52,;12.5,-15.84,;12.97,-17.3,;14.48,-17.62,;11.94,-18.45,;10.44,-18.13,)| Show InChI InChI=1S/C25H37N5O4/c1-27-19(15-16-7-4-3-5-8-16)25(34)30-14-6-9-20(30)23(32)29-21(22(31)24(33)28-2)17-10-12-18(26)13-11-17/h3-5,7-8,17-21,27H,6,9-15,26H2,1-2H3,(H,28,33)(H,29,32)/t17-,18-,19-,20+,21+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards thrombin |
J Med Chem 40: 830-2 (1997)
Article DOI: 10.1021/jm960762y
BindingDB Entry DOI: 10.7270/Q25H7GXW |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50008735
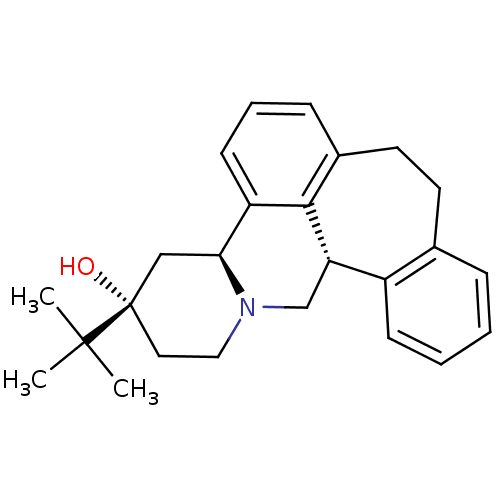
((+)-3-(tert-butyl)-(3S,4aS,13bS)-2,3,4,4a,8,9,13b,...)Show SMILES CC(C)(C)[C@]1(O)CCN2C[C@H]3c4ccccc4CCc4cccc([C@@H]2C1)c34 Show InChI InChI=1S/C25H31NO/c1-24(2,3)25(27)13-14-26-16-21-19-9-5-4-7-17(19)11-12-18-8-6-10-20(23(18)21)22(26)15-25/h4-10,21-22,27H,11-16H2,1-3H3/t21-,22-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50008735

((+)-3-(tert-butyl)-(3S,4aS,13bS)-2,3,4,4a,8,9,13b,...)Show SMILES CC(C)(C)[C@]1(O)CCN2C[C@H]3c4ccccc4CCc4cccc([C@@H]2C1)c34 Show InChI InChI=1S/C25H31NO/c1-24(2,3)25(27)13-14-26-16-21-19-9-5-4-7-17(19)11-12-18-8-6-10-20(23(18)21)22(26)15-25/h4-10,21-22,27H,11-16H2,1-3H3/t21-,22-,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50454822

(CHEMBL2062141 | L-370518)Show SMILES [H][C@@](NC(=O)[C@@H]1CCCN1C(=O)[C@@H](Cc1ccccc1)NC)(C(=O)C(=O)NC)[C@@]1([H])CC[C@H](N)CC1 |wU:1.0,wD:12.13,5.4,32.34,28.30,(9.54,-15.25,;8.45,-16.34,;7.42,-17.49,;5.92,-17.17,;5.44,-15.7,;4.89,-18.31,;5.21,-19.82,;3.87,-20.59,;2.73,-19.56,;3.35,-18.15,;2.58,-16.82,;3.35,-15.48,;1.04,-16.82,;.27,-15.48,;1.04,-14.15,;2.58,-14.15,;3.35,-12.82,;2.58,-11.48,;1.04,-11.48,;.27,-12.82,;.27,-18.15,;-1.27,-18.15,;7.98,-14.88,;6.47,-14.56,;9.01,-13.73,;10.51,-14.05,;8.53,-12.27,;9.56,-11.12,;9.96,-16.66,;8.87,-17.75,;10.99,-15.52,;12.5,-15.84,;12.97,-17.3,;14.48,-17.62,;11.94,-18.45,;10.44,-18.13,)| Show InChI InChI=1S/C25H37N5O4/c1-27-19(15-16-7-4-3-5-8-16)25(34)30-14-6-9-20(30)23(32)29-21(22(31)24(33)28-2)17-10-12-18(26)13-11-17/h3-5,7-8,17-21,27H,6,9-15,26H2,1-2H3,(H,28,33)(H,29,32)/t17-,18-,19-,20+,21+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards thrombin was determined |
J Med Chem 41: 401-6 (1998)
Article DOI: 10.1021/jm9705014
BindingDB Entry DOI: 10.7270/Q2H995V2 |
More data for this
Ligand-Target Pair | |
Nociceptin receptor
(Homo sapiens (Human)) | BDBM50004178
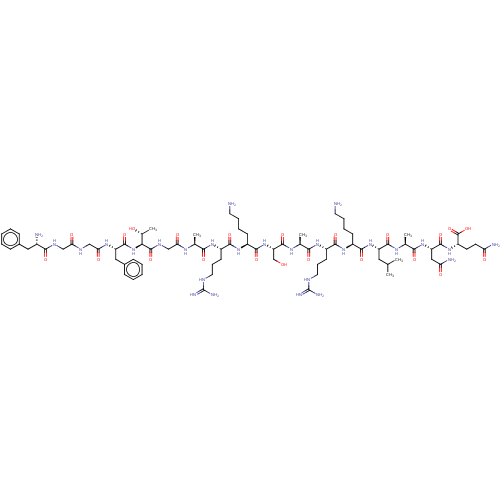
(Nociceptin | Nociceptin/orphanin FQ | ORPHANIN FQ)Show SMILES CC(C)C[C@H](NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccccc1)[C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O |r| Show InChI InChI=1S/C79H129N27O22/c1-41(2)33-54(72(122)95-44(5)66(116)103-56(36-59(84)110)73(123)102-53(77(127)128)27-28-58(83)109)104-70(120)49(23-13-15-29-80)100-69(119)52(26-18-32-90-79(87)88)99-65(115)43(4)96-75(125)57(40-107)105-71(121)50(24-14-16-30-81)101-68(118)51(25-17-31-89-78(85)86)98-64(114)42(3)94-61(112)39-93-76(126)63(45(6)108)106-74(124)55(35-47-21-11-8-12-22-47)97-62(113)38-91-60(111)37-92-67(117)48(82)34-46-19-9-7-10-20-46/h7-12,19-22,41-45,48-57,63,107-108H,13-18,23-40,80-82H2,1-6H3,(H2,83,109)(H2,84,110)(H,91,111)(H,92,117)(H,93,126)(H,94,112)(H,95,122)(H,96,125)(H,97,113)(H,98,114)(H,99,115)(H,100,119)(H,101,118)(H,102,123)(H,103,116)(H,104,120)(H,105,121)(H,106,124)(H,127,128)(H4,85,86,89)(H4,87,88,90)/t42-,43-,44-,45+,48-,49-,50-,51-,52-,53-,54-,55-,56-,57-,63-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ de Montr£al
Curated by ChEMBL
| Assay Description
Binding affinity for opioid receptor like type, human Opioid receptor like 1 expressed in membrane homogenates of COS-1 or CHO cells |
J Med Chem 45: 5353-7 (2002)
BindingDB Entry DOI: 10.7270/Q2VT1ST9 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50057827
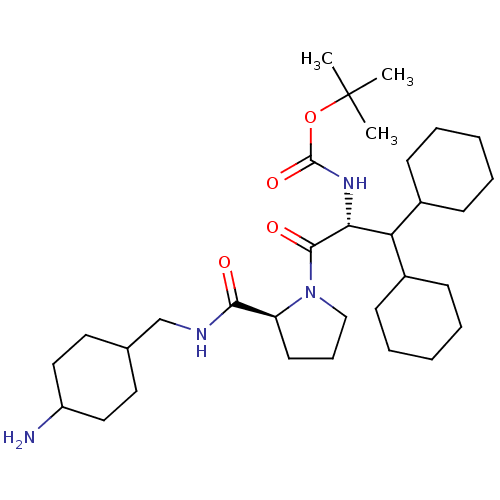
(((R)-2-{(S)-2-[(4-Amino-cyclohexylmethyl)-carbamoy...)Show SMILES CC(C)(C)OC(=O)N[C@H](C(C1CCCCC1)C1CCCCC1)C(=O)N1CCC[C@H]1C(=O)NCC1CCC(N)CC1 |wU:28.31,wD:8.7,(4.89,-7.9,;5.69,-6.58,;4.94,-5.24,;6.75,-7.68,;7.76,-6.72,;8.02,-5.27,;7.26,-3.92,;9.56,-5.29,;10.89,-4.53,;10.89,-2.99,;9.56,-2.21,;9.56,-.67,;8.22,.1,;6.9,-.67,;6.9,-2.21,;8.22,-2.98,;12.23,-2.22,;13.55,-2.99,;14.9,-2.22,;14.9,-.68,;13.57,.09,;12.22,-.68,;12.23,-5.3,;12.23,-6.84,;13.71,-4.89,;14.26,-3.44,;15.79,-3.51,;16.21,-5,;14.92,-5.85,;14.85,-7.39,;13.49,-8.1,;16.15,-8.21,;17.52,-7.51,;18.81,-8.32,;17.75,-9.44,;18.84,-10.92,;18.78,-12.17,;19.86,-13.25,;19.86,-11.05,;18.78,-9.66,)| Show InChI InChI=1S/C32H56N4O4/c1-32(2,3)40-31(39)35-28(27(23-11-6-4-7-12-23)24-13-8-5-9-14-24)30(38)36-20-10-15-26(36)29(37)34-21-22-16-18-25(33)19-17-22/h22-28H,4-21,33H2,1-3H3,(H,34,37)(H,35,39)/t22?,25?,26-,28+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory activity against human thrombin |
J Med Chem 40: 1565-9 (1997)
Article DOI: 10.1021/jm970140s
BindingDB Entry DOI: 10.7270/Q2PR7V29 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50057828

((S)-1-((R)-2-Amino-3,3-diphenyl-propionyl)-pyrroli...)Show SMILES N[C@H](C(c1ccccc1)c1ccccc1)C(=O)N1CCC[C@H]1C(=O)NCC1CCC(N)CC1 |wU:21.24,wD:1.0,(8.37,-6.36,;9.7,-5.59,;9.7,-4.05,;11.04,-3.28,;12.58,-3.28,;13.35,-1.94,;12.56,-.61,;11.02,-.62,;10.27,-1.96,;8.35,-3.28,;8.35,-1.74,;7.03,-.97,;5.69,-1.74,;5.69,-3.28,;7.03,-4.05,;11.05,-6.36,;11.05,-7.9,;12.36,-5.59,;12.52,-4.05,;14.03,-3.73,;14.8,-5.05,;13.77,-6.2,;14.1,-7.71,;12.96,-8.74,;15.57,-8.18,;16.72,-7.15,;18.17,-7.61,;18.09,-8.95,;19.1,-10.37,;17.98,-11.46,;19.06,-12.54,;18.09,-10.21,;17.07,-8.71,)| Show InChI InChI=1S/C27H36N4O2/c28-22-15-13-19(14-16-22)18-30-26(32)23-12-7-17-31(23)27(33)25(29)24(20-8-3-1-4-9-20)21-10-5-2-6-11-21/h1-6,8-11,19,22-25H,7,12-18,28-29H2,(H,30,32)/t19?,22?,23-,25+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory activity against human thrombin |
J Med Chem 40: 1565-9 (1997)
Article DOI: 10.1021/jm970140s
BindingDB Entry DOI: 10.7270/Q2PR7V29 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50063555
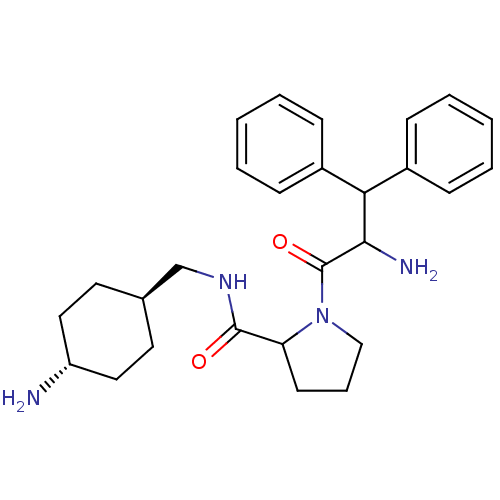
(1-(2-Amino-3,3-diphenyl-propionyl)-pyrrolidine-2-c...)Show SMILES NC(C(c1ccccc1)c1ccccc1)C(=O)N1CCCC1C(=O)NC[C@H]1CC[C@H](N)CC1 |wU:26.28,wD:29.32,(6.26,-10.67,;4.93,-9.9,;3.6,-10.69,;3.6,-12.23,;2.27,-13,;2.27,-14.52,;3.6,-15.29,;4.95,-14.51,;4.93,-12.98,;2.27,-9.92,;2.27,-8.36,;.92,-7.59,;-.41,-8.36,;-.41,-9.92,;.92,-10.69,;4.93,-8.36,;3.6,-7.59,;6.26,-7.59,;5.79,-6.14,;7.03,-5.23,;8.27,-6.14,;7.8,-7.59,;8.69,-8.85,;8.69,-10.39,;10.02,-8.06,;11.36,-8.83,;12.68,-8.05,;12.65,-6.51,;14,-5.72,;15.34,-6.47,;16.67,-5.7,;15.36,-8.01,;14.05,-8.8,)| Show InChI InChI=1S/C27H36N4O2/c28-22-15-13-19(14-16-22)18-30-26(32)23-12-7-17-31(23)27(33)25(29)24(20-8-3-1-4-9-20)21-10-5-2-6-11-21/h1-6,8-11,19,22-25H,7,12-18,28-29H2,(H,30,32)/t19-,22-,23?,25? | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards thrombin |
J Med Chem 41: 1011-3 (1998)
Article DOI: 10.1021/jm9706933
BindingDB Entry DOI: 10.7270/Q2ZG6RCF |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50057828

((S)-1-((R)-2-Amino-3,3-diphenyl-propionyl)-pyrroli...)Show SMILES N[C@H](C(c1ccccc1)c1ccccc1)C(=O)N1CCC[C@H]1C(=O)NCC1CCC(N)CC1 |wU:21.24,wD:1.0,(8.37,-6.36,;9.7,-5.59,;9.7,-4.05,;11.04,-3.28,;12.58,-3.28,;13.35,-1.94,;12.56,-.61,;11.02,-.62,;10.27,-1.96,;8.35,-3.28,;8.35,-1.74,;7.03,-.97,;5.69,-1.74,;5.69,-3.28,;7.03,-4.05,;11.05,-6.36,;11.05,-7.9,;12.36,-5.59,;12.52,-4.05,;14.03,-3.73,;14.8,-5.05,;13.77,-6.2,;14.1,-7.71,;12.96,-8.74,;15.57,-8.18,;16.72,-7.15,;18.17,-7.61,;18.09,-8.95,;19.1,-10.37,;17.98,-11.46,;19.06,-12.54,;18.09,-10.21,;17.07,-8.71,)| Show InChI InChI=1S/C27H36N4O2/c28-22-15-13-19(14-16-22)18-30-26(32)23-12-7-17-31(23)27(33)25(29)24(20-8-3-1-4-9-20)21-10-5-2-6-11-21/h1-6,8-11,19,22-25H,7,12-18,28-29H2,(H,30,32)/t19?,22?,23-,25+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards thrombin was determined |
J Med Chem 41: 401-6 (1998)
Article DOI: 10.1021/jm9705014
BindingDB Entry DOI: 10.7270/Q2H995V2 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 4
(Homo sapiens (Human)) | BDBM82505

(CAS_121881 | NSC_121881 | SB204070)Show InChI InChI=1S/C19H27ClN2O4/c1-2-3-6-22-7-4-13(5-8-22)12-26-19(23)14-11-15(20)16(21)18-17(14)24-9-10-25-18/h11,13H,2-10,12,21H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
J Neurochem 69: 1810-9 (1997)
Article DOI: 10.1046/j.1471-4159.1997.69051810.x
BindingDB Entry DOI: 10.7270/Q2Z60MMW |
More data for this
Ligand-Target Pair | |
Protachykinin-1
(Homo sapiens (Human)) | BDBM50001450
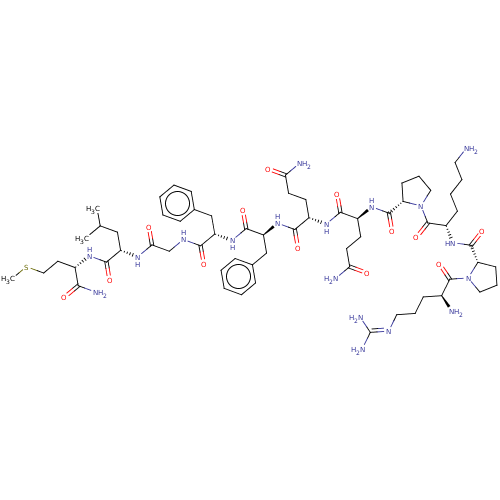
((SP)Arg-Pro-Lys-Pro-Gln-Gln-Phe-Phe-Gly-Leu-Met-NH...)Show SMILES [#6]-[#16]-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#6])-[#6])-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@H](-[#6]-[#6]-[#6](-[#7])=O)-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@@H](-[#7])-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](-[#7])=O |r| Show InChI InChI=1S/C63H98N18O13S/c1-37(2)33-45(57(89)74-41(53(68)85)27-32-95-3)73-52(84)36-72-54(86)46(34-38-15-6-4-7-16-38)78-58(90)47(35-39-17-8-5-9-18-39)79-56(88)42(23-25-50(66)82)75-55(87)43(24-26-51(67)83)76-59(91)49-22-14-31-81(49)62(94)44(20-10-11-28-64)77-60(92)48-21-13-30-80(48)61(93)40(65)19-12-29-71-63(69)70/h4-9,15-18,37,40-49H,10-14,19-36,64-65H2,1-3H3,(H2,66,82)(H2,67,83)(H2,68,85)(H,72,86)(H,73,84)(H,74,89)(H,75,87)(H,76,91)(H,77,92)(H,78,90)(H,79,88)(H4,69,70,71)/t40-,41-,42-,43-,44-,45-,46-,47-,48-,49-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
Prothrombin
(Homo sapiens (Human)) | BDBM50056773
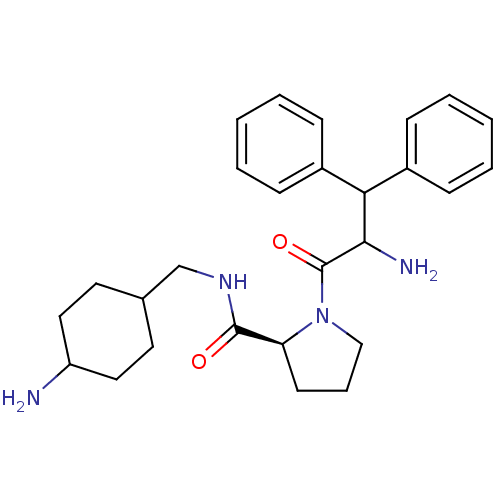
((S)-1-(2-Amino-3,3-diphenyl-propionyl)-pyrrolidine...)Show SMILES NC(C(c1ccccc1)c1ccccc1)C(=O)N1CCC[C@H]1C(=O)NCC1CCC(N)CC1 |wU:21.24,(4.23,-10.4,;5.49,-9.52,;5.36,-7.98,;6.62,-7.1,;5.84,-5.77,;6.62,-4.44,;8.16,-4.44,;8.92,-5.77,;8.16,-7.1,;3.97,-7.33,;3.85,-5.81,;2.46,-5.14,;1.19,-6.03,;1.31,-7.56,;2.71,-8.22,;6.89,-10.18,;7,-11.71,;8.15,-9.29,;8.14,-7.75,;9.66,-7.31,;10.53,-8.57,;9.59,-9.8,;10.05,-11.26,;9,-12.39,;11.55,-11.61,;12.6,-10.48,;12.15,-9.01,;13.36,-8.19,;12.84,-6.74,;12.85,-5.08,;12.08,-3.75,;11.76,-6.18,;12.31,-7.56,)| Show InChI InChI=1S/C27H36N4O2/c28-22-15-13-19(14-16-22)18-30-26(32)23-12-7-17-31(23)27(33)25(29)24(20-8-3-1-4-9-20)21-10-5-2-6-11-21/h1-6,8-11,19,22-25H,7,12-18,28-29H2,(H,30,32)/t19?,22?,23-,25?/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity towards thrombin |
J Med Chem 40: 830-2 (1997)
Article DOI: 10.1021/jm960762y
BindingDB Entry DOI: 10.7270/Q25H7GXW |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 4
(Homo sapiens (Human)) | BDBM82505

(CAS_121881 | NSC_121881 | SB204070)Show InChI InChI=1S/C19H27ClN2O4/c1-2-3-6-22-7-4-13(5-8-22)12-26-19(23)14-11-15(20)16(21)18-17(14)24-9-10-25-18/h11,13H,2-10,12,21H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
J Neurochem 74: 478-89 (2000)
Article DOI: 10.1046/j.1471-4159.2000.740478.x
BindingDB Entry DOI: 10.7270/Q2DJ5D5Z |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Rattus norvegicus (rat)) | BDBM84994
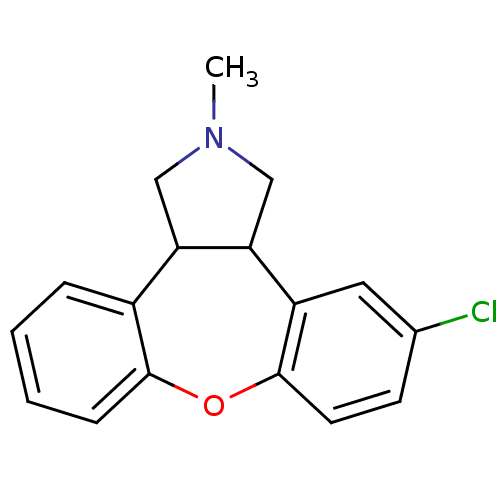
(CAS_163091 | NSC_163091 | ORG-5222)Show InChI InChI=1S/C17H16ClNO/c1-19-9-14-12-4-2-3-5-16(12)20-17-7-6-11(18)8-13(17)15(14)10-19/h2-8,14-15H,9-10H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM85033
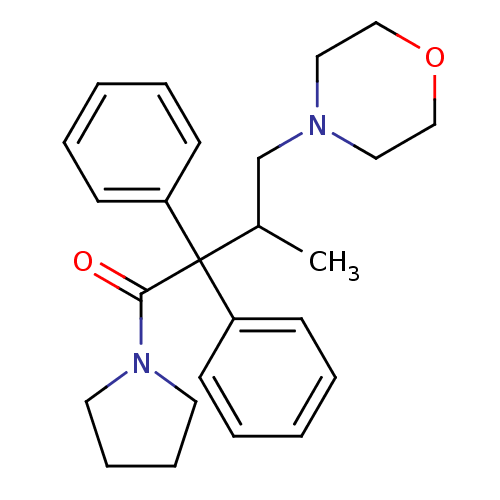
(CAS_357-56-2 | Dextromoramide | NSC_9648)Show SMILES CC(CN1CCOCC1)C(C(=O)N1CCCC1)(c1ccccc1)c1ccccc1 Show InChI InChI=1S/C25H32N2O2/c1-21(20-26-16-18-29-19-17-26)25(22-10-4-2-5-11-22,23-12-6-3-7-13-23)24(28)27-14-8-9-15-27/h2-7,10-13,21H,8-9,14-20H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | |
Coagulation factor VII/Tissue factor
(Homo sapiens (Human)) | BDBM50192768
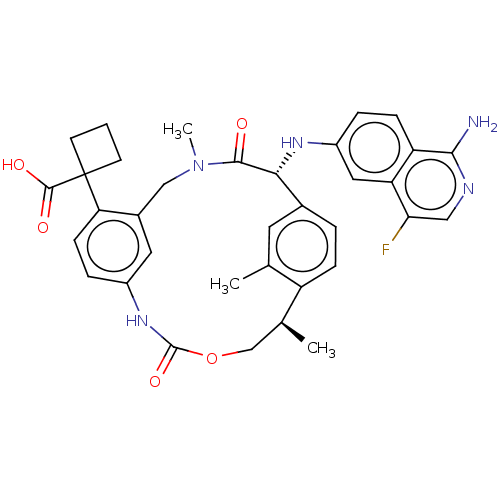
(CHEMBL3900166)Show SMILES C[C@H]1COC(=O)Nc2ccc(c(CN(C)C(=O)[C@H](Nc3ccc4c(N)ncc(F)c4c3)c3ccc1c(C)c3)c2)C1(CCC1)C(O)=O |r| Show InChI InChI=1S/C35H36FN5O5/c1-19-13-21-5-8-25(19)20(2)18-46-34(45)40-23-7-10-28(35(33(43)44)11-4-12-35)22(14-23)17-41(3)32(42)30(21)39-24-6-9-26-27(15-24)29(36)16-38-31(26)37/h5-10,13-16,20,30,39H,4,11-12,17-18H2,1-3H3,(H2,37,38)(H,40,45)(H,43,44)/t20-,30+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Bristol-Myers Squibb Research& Development
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TF-factor 7a using factor 10 as substrate at 37 degC |
Bioorg Med Chem Lett 26: 5051-5057 (2016)
Article DOI: 10.1016/j.bmcl.2016.08.088
BindingDB Entry DOI: 10.7270/Q24X59Q6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Genome polyprotein
(Hepacivirus C) | BDBM50485492
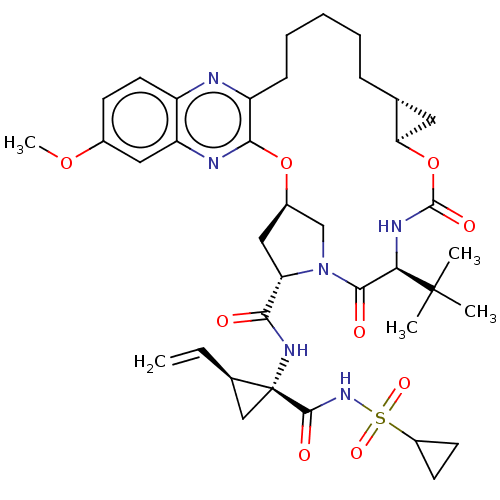
(Grazoprevir | Grazoprevir monohydrate | MK-5172 | ...)Show SMILES [H][C@@]12C[C@@]1([H])OC(=O)N[C@H](C(=O)N1C[C@@]([H])(C[C@H]1C(=O)N[C@@]1(C[C@H]1C=C)C(=O)NS(=O)(=O)C1CC1)Oc1nc3cc(OC)ccc3nc1CCCCC2)C(C)(C)C |r| Show InChI InChI=1S/C38H50N6O9S/c1-6-22-19-38(22,35(47)43-54(49,50)25-13-14-25)42-32(45)29-18-24-20-44(29)34(46)31(37(2,3)4)41-36(48)53-30-16-21(30)10-8-7-9-11-27-33(52-24)40-28-17-23(51-5)12-15-26(28)39-27/h6,12,15,17,21-22,24-25,29-31H,1,7-11,13-14,16,18-20H2,2-5H3,(H,41,48)(H,42,45)(H,43,47)/t21-,22-,24-,29+,30-,31-,38-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/TrEMBL
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Hepatitis C virus (isolate BK) genotype 1b NS3/4a protease D168V mutant expressed in Escherichia coli by time-resolved fluorescence ana... |
ACS Med Chem Lett 3: 332-6 (2012)
Article DOI: 10.1021/ml300017p
BindingDB Entry DOI: 10.7270/Q2KH0R6V |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
5-hydroxytryptamine receptor 4
(Homo sapiens (Human)) | BDBM29525
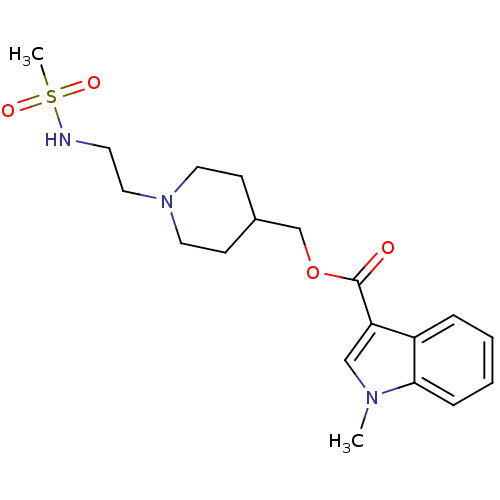
(3H-GR113808 | CHEMBL518682 | GR 113808 | [3H] GR 1...)Show SMILES Cn1cc(C(=O)OCC2CCN(CCNS(C)(=O)=O)CC2)c2ccccc12 Show InChI InChI=1S/C19H27N3O4S/c1-21-13-17(16-5-3-4-6-18(16)21)19(23)26-14-15-7-10-22(11-8-15)12-9-20-27(2,24)25/h3-6,13,15,20H,7-12,14H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
J Neurochem 69: 1810-9 (1997)
Article DOI: 10.1046/j.1471-4159.1997.69051810.x
BindingDB Entry DOI: 10.7270/Q2Z60MMW |
More data for this
Ligand-Target Pair | |
Sodium-dependent serotonin transporter
(Homo sapiens (Human)) | BDBM50010859
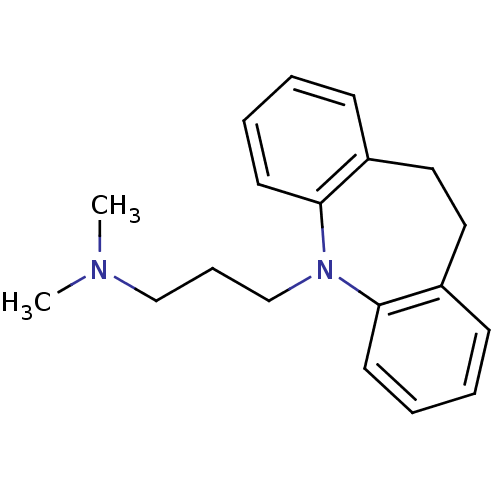
(CHEMBL11 | IMIPRAMINE HYDROCHLORIDE | IMIPRAMINE P...)Show InChI InChI=1S/C19H24N2/c1-20(2)14-7-15-21-18-10-5-3-8-16(18)12-13-17-9-4-6-11-19(17)21/h3-6,8-11H,7,12-15H2,1-2H3 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
Psychopharmacology (Berl) 124: 57-73 (1996)
Article DOI: 10.1007/bf02245606
BindingDB Entry DOI: 10.7270/Q2610XV6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
5-hydroxytryptamine receptor 4
(Homo sapiens (Human)) | BDBM85020
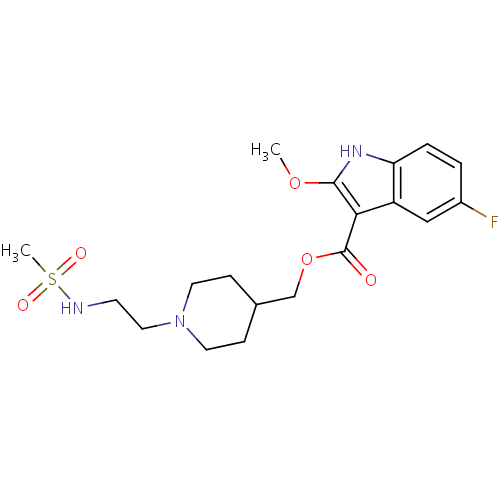
(5-Fluoro-2-methoxy-1H-indole-3-carboxylic acid [1-...)Show SMILES COc1[nH]c2ccc(F)cc2c1C(=O)OCC1CCN(CCN[S](C)(=O)=O)CC1 Show InChI InChI=1S/C19H26FN3O5S/c1-27-18-17(15-11-14(20)3-4-16(15)22-18)19(24)28-12-13-5-8-23(9-6-13)10-7-21-29(2,25)26/h3-4,11,13,21-22H,5-10,12H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| MCE
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Janssen Research Foundation
Curated by PDSP Ki Database
| |
J Neurochem 69: 1810-9 (1997)
Article DOI: 10.1046/j.1471-4159.1997.69051810.x
BindingDB Entry DOI: 10.7270/Q2Z60MMW |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data