 Found 108 hits with Last Name = 'fujimura' and Initial = 'k'
Found 108 hits with Last Name = 'fujimura' and Initial = 'k' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272062
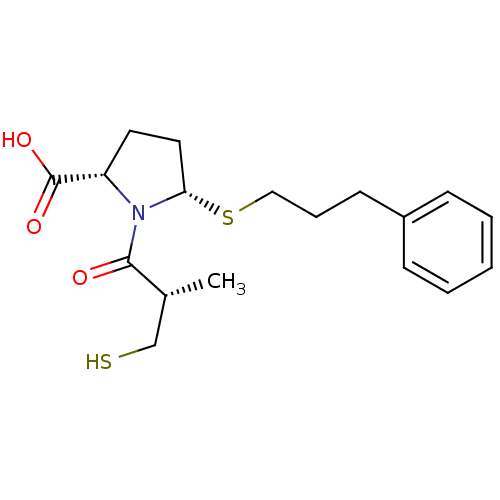
((2S,5S)-1-((S)-3-mercapto-2-methylpropanoyl)-5-(3-...)Show SMILES C[C@H](CS)C(=O)N1[C@H](CC[C@H]1C(O)=O)SCCCc1ccccc1 |r| Show InChI InChI=1S/C18H25NO3S2/c1-13(12-23)17(20)19-15(18(21)22)9-10-16(19)24-11-5-8-14-6-3-2-4-7-14/h2-4,6-7,13,15-16,23H,5,8-12H2,1H3,(H,21,22)/t13-,15+,16+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.0290 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272061
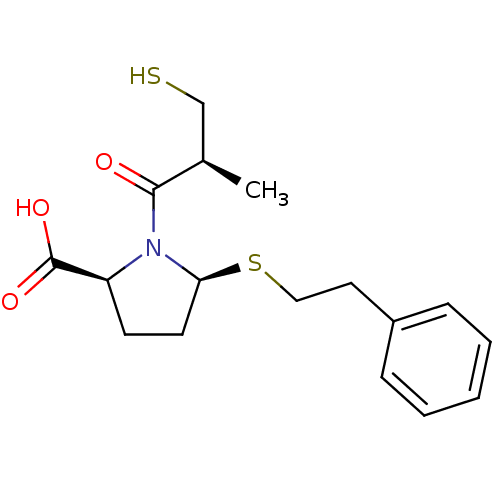
((2S,5S)-1-((S)-3-mercapto-2-methylpropanoyl)-5-(ph...)Show SMILES C[C@H](CS)C(=O)N1[C@H](CC[C@H]1C(O)=O)SCCc1ccccc1 |r| Show InChI InChI=1S/C17H23NO3S2/c1-12(11-22)16(19)18-14(17(20)21)7-8-15(18)23-10-9-13-5-3-2-4-6-13/h2-6,12,14-15,22H,7-11H2,1H3,(H,20,21)/t12-,14+,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272145

((2S,5S)-1-((S)-3-Mercapto-2-methyl-propionyl)-5-(3...)Show SMILES C[C@H](CS)C(=O)N1[C@H](CC[C@H]1C(O)=O)SCc1cccc(C)c1 |r| Show InChI InChI=1S/C17H23NO3S2/c1-11-4-3-5-13(8-11)10-23-15-7-6-14(17(20)21)18(15)16(19)12(2)9-22/h3-5,8,12,14-15,22H,6-7,9-10H2,1-2H3,(H,20,21)/t12-,14+,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272199
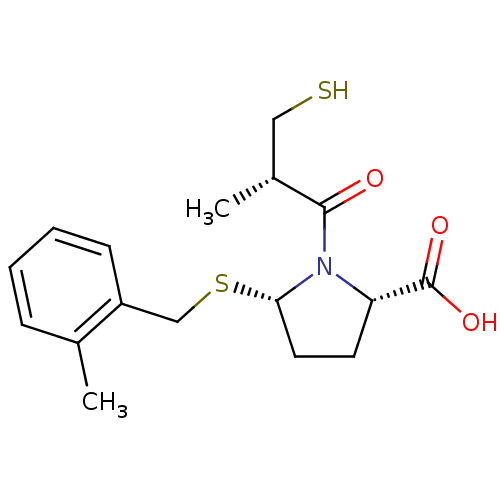
((2S,5S)-1-((S)-3-Mercapto-2-methyl-propionyl)-5-(2...)Show SMILES C[C@H](CS)C(=O)N1[C@H](CC[C@H]1C(O)=O)SCc1ccccc1C |r| Show InChI InChI=1S/C17H23NO3S2/c1-11-5-3-4-6-13(11)10-23-15-8-7-14(17(20)21)18(15)16(19)12(2)9-22/h3-6,12,14-15,22H,7-10H2,1-2H3,(H,20,21)/t12-,14+,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272060
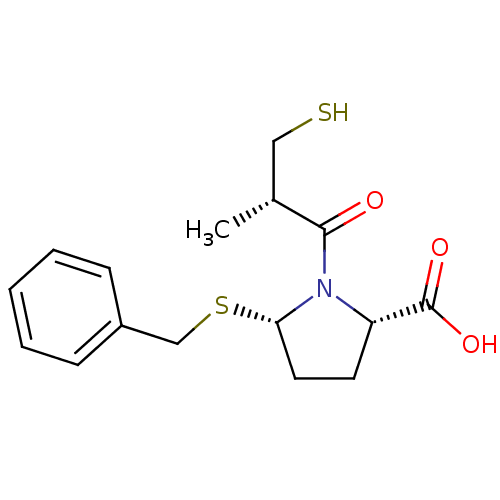
((2S,5S)-5-(benzylthio)-1-((S)-3-mercapto-2-methylp...)Show SMILES C[C@H](CS)C(=O)N1[C@H](CC[C@H]1C(O)=O)SCc1ccccc1 |r| Show InChI InChI=1S/C16H21NO3S2/c1-11(9-21)15(18)17-13(16(19)20)7-8-14(17)22-10-12-5-3-2-4-6-12/h2-6,11,13-14,21H,7-10H2,1H3,(H,19,20)/t11-,13+,14+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272099
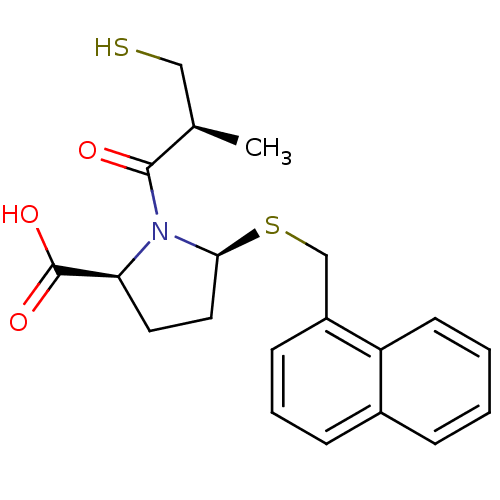
((2S,5S)-1-((S)-3-mercapto-2-methylpropanoyl)-5-(na...)Show SMILES C[C@H](CS)C(=O)N1[C@H](CC[C@H]1C(O)=O)SCc1cccc2ccccc12 |r| Show InChI InChI=1S/C20H23NO3S2/c1-13(11-25)19(22)21-17(20(23)24)9-10-18(21)26-12-15-7-4-6-14-5-2-3-8-16(14)15/h2-8,13,17-18,25H,9-12H2,1H3,(H,23,24)/t13-,17+,18+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272203

((2S,5R)-5-(4-isopropylbenzylthio)-1-((S)-3-mercapt...)Show SMILES CC(C)c1ccc(CS[C@@H]2CC[C@H](N2C(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C19H27NO3S2/c1-12(2)15-6-4-14(5-7-15)11-25-17-9-8-16(19(22)23)20(17)18(21)13(3)10-24/h4-7,12-13,16-17,24H,8-11H2,1-3H3,(H,22,23)/t13-,16+,17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272200
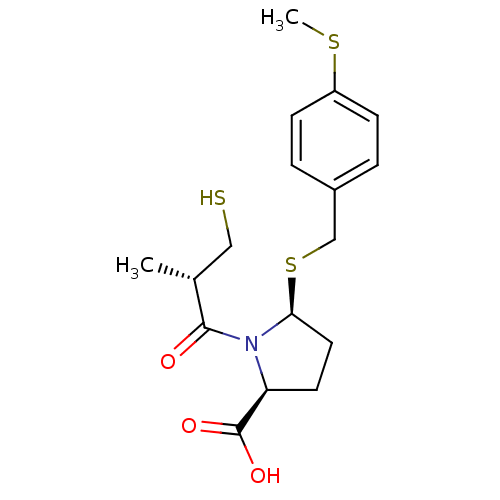
((2S,5S)-5-(4-(methylthio)benzylthio)-1-((S)-3-merc...)Show SMILES CSc1ccc(CS[C@H]2CC[C@H](N2C(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C17H23NO3S3/c1-11(9-22)16(19)18-14(17(20)21)7-8-15(18)24-10-12-3-5-13(23-2)6-4-12/h3-6,11,14-15,22H,7-10H2,1-2H3,(H,20,21)/t11-,14+,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272141
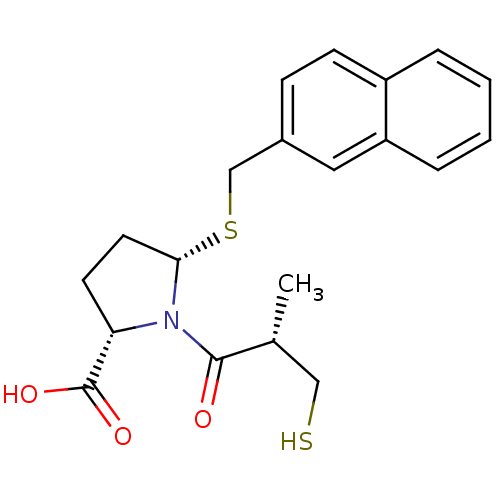
((2S,5S)-1-((S)-3-mercapto-2-methylpropanoyl)-5-(na...)Show SMILES C[C@H](CS)C(=O)N1[C@H](CC[C@H]1C(O)=O)SCc1ccc2ccccc2c1 |r| Show InChI InChI=1S/C20H23NO3S2/c1-13(11-25)19(22)21-17(20(23)24)8-9-18(21)26-12-14-6-7-15-4-2-3-5-16(15)10-14/h2-7,10,13,17-18,25H,8-9,11-12H2,1H3,(H,23,24)/t13-,17+,18+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272142
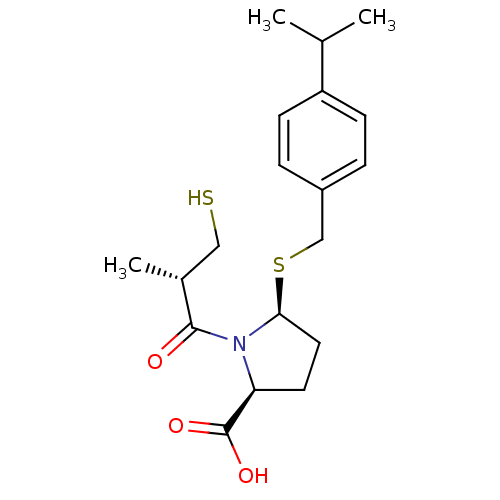
((2S,5S)-5-(4-isopropylbenzylthio)-1-((S)-3-mercapt...)Show SMILES CC(C)c1ccc(CS[C@H]2CC[C@H](N2C(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C19H27NO3S2/c1-12(2)15-6-4-14(5-7-15)11-25-17-9-8-16(19(22)23)20(17)18(21)13(3)10-24/h4-7,12-13,16-17,24H,8-11H2,1-3H3,(H,22,23)/t13-,16+,17+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266136
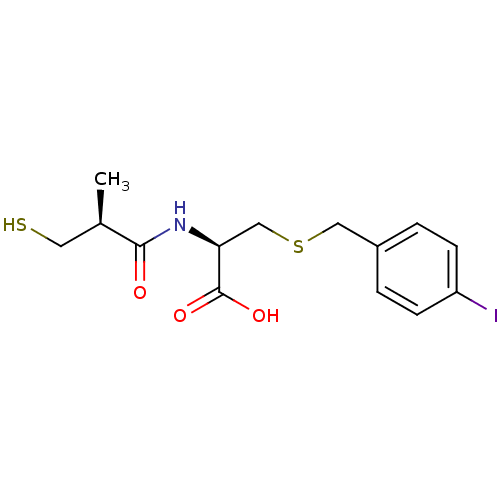
((R)-3-(4-iodobenzylthio)-2-((S)-3-mercapto-2-methy...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(I)cc1)C(O)=O |r| Show InChI InChI=1S/C14H18INO3S2/c1-9(6-20)13(17)16-12(14(18)19)8-21-7-10-2-4-11(15)5-3-10/h2-5,9,12,20H,6-8H2,1H3,(H,16,17)(H,18,19)/t9-,12+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272144
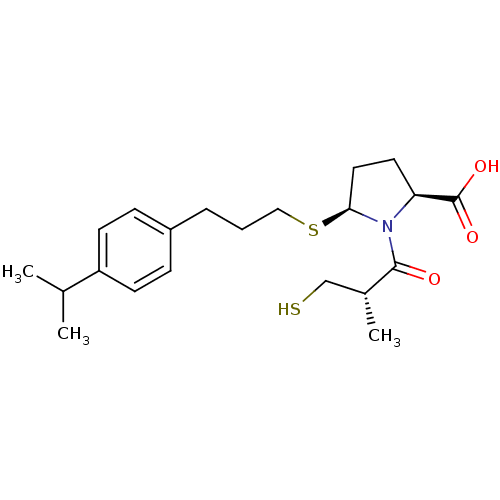
((2S,5S)-5-(3-(4-isopropylphenyl)propylthio)-1-((S)...)Show SMILES CC(C)c1ccc(CCCS[C@H]2CC[C@H](N2C(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C21H31NO3S2/c1-14(2)17-8-6-16(7-9-17)5-4-12-27-19-11-10-18(21(24)25)22(19)20(23)15(3)13-26/h6-9,14-15,18-19,26H,4-5,10-13H2,1-3H3,(H,24,25)/t15-,18+,19+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272143
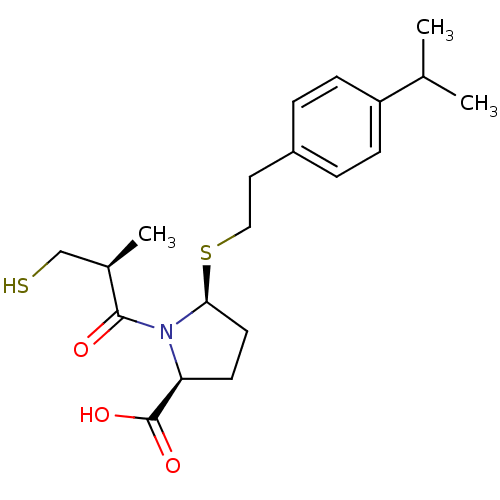
((2S,5S)-5-(4-isopropylphenethylthio)-1-((S)-3-merc...)Show SMILES CC(C)c1ccc(CCS[C@H]2CC[C@H](N2C(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C20H29NO3S2/c1-13(2)16-6-4-15(5-7-16)10-11-26-18-9-8-17(20(23)24)21(18)19(22)14(3)12-25/h4-7,13-14,17-18,25H,8-12H2,1-3H3,(H,23,24)/t14-,17+,18+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266170
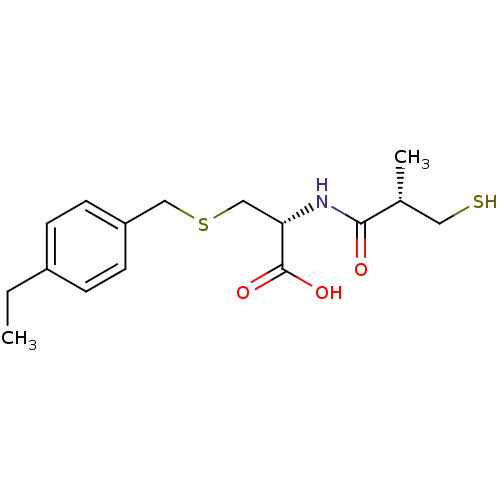
((R)-3-(4-ethylbenzylthio)-2-((S)-3-mercapto-2-meth...)Show SMILES CCc1ccc(CSC[C@H](NC(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C16H23NO3S2/c1-3-12-4-6-13(7-5-12)9-22-10-14(16(19)20)17-15(18)11(2)8-21/h4-7,11,14,21H,3,8-10H2,1-2H3,(H,17,18)(H,19,20)/t11-,14+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM21642

((2S)-1-[(2S)-2-methyl-3-sulfanylpropanoyl]pyrrolid...)Show InChI InChI=1S/C9H15NO3S/c1-6(5-14)8(11)10-4-2-3-7(10)9(12)13/h6-7,14H,2-5H2,1H3,(H,12,13)/t6-,7+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266173
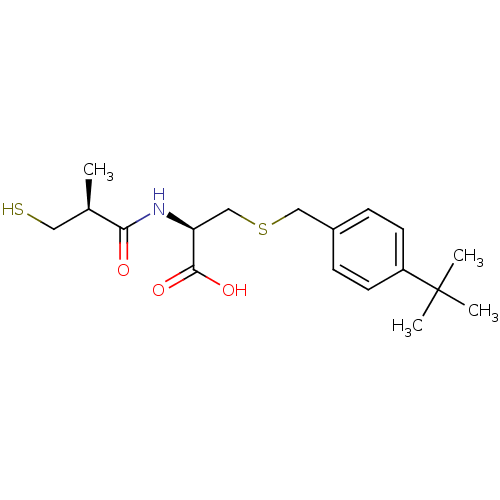
((R)-3-(4-tert-butylbenzylthio)-2-((S)-3-mercapto-2...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(cc1)C(C)(C)C)C(O)=O |r| Show InChI InChI=1S/C18H27NO3S2/c1-12(9-23)16(20)19-15(17(21)22)11-24-10-13-5-7-14(8-6-13)18(2,3)4/h5-8,12,15,23H,9-11H2,1-4H3,(H,19,20)(H,21,22)/t12-,15+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50272201
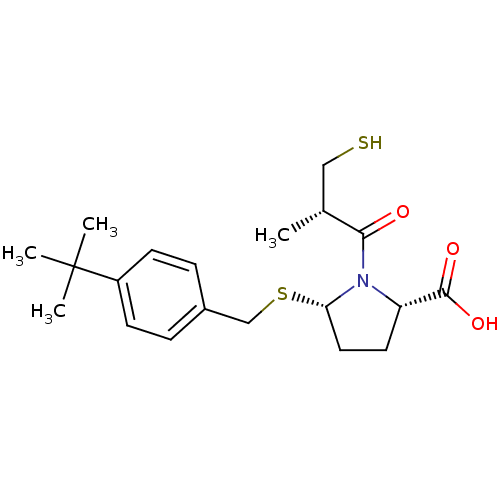
((2S,5S)-5-(4-tert-butylbenzylthio)-1-((S)-3-mercap...)Show SMILES C[C@H](CS)C(=O)N1[C@H](CC[C@H]1C(O)=O)SCc1ccc(cc1)C(C)(C)C |r| Show InChI InChI=1S/C20H29NO3S2/c1-13(11-25)18(22)21-16(19(23)24)9-10-17(21)26-12-14-5-7-15(8-6-14)20(2,3)4/h5-8,13,16-17,25H,9-12H2,1-4H3,(H,23,24)/t13-,16+,17+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of guinea pig lung leukotriene A4 hydrolase |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50272202
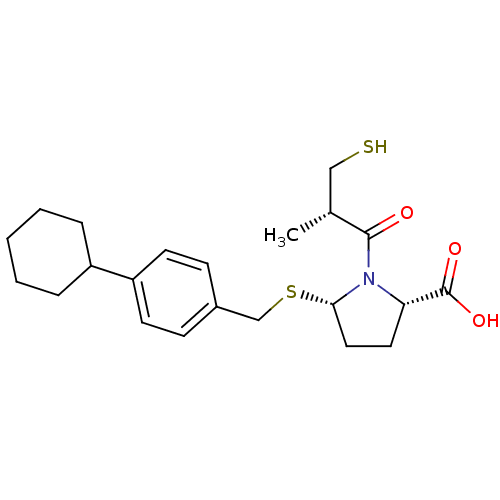
((2S,5S)-5-(4-cyclohexylbenzylthio)-1-((S)-3-mercap...)Show SMILES C[C@H](CS)C(=O)N1[C@H](CC[C@H]1C(O)=O)SCc1ccc(cc1)C1CCCCC1 |r| Show InChI InChI=1S/C22H31NO3S2/c1-15(13-27)21(24)23-19(22(25)26)11-12-20(23)28-14-16-7-9-18(10-8-16)17-5-3-2-4-6-17/h7-10,15,17,19-20,27H,2-6,11-14H2,1H3,(H,25,26)/t15-,19+,20+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of guinea pig lung leukotriene A4 hydrolase |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266075
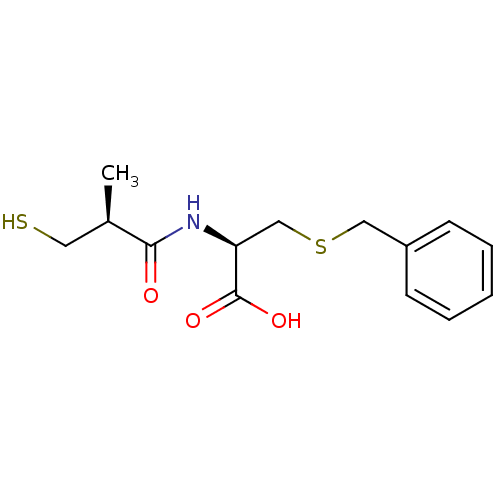
((R)-3-(benzylthio)-2-((S)-3-mercapto-2-methylpropa...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccccc1)C(O)=O |r| Show InChI InChI=1S/C14H19NO3S2/c1-10(7-19)13(16)15-12(14(17)18)9-20-8-11-5-3-2-4-6-11/h2-6,10,12,19H,7-9H2,1H3,(H,15,16)(H,17,18)/t10-,12+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266244
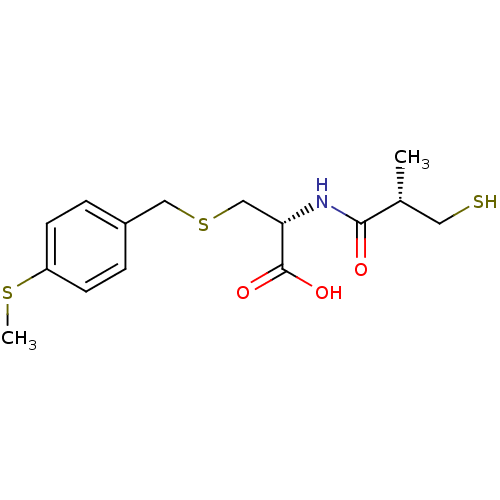
((R)-2-((S)-3-mercapto-2-methylpropanamido)-3-(4-(m...)Show SMILES CSc1ccc(CSC[C@H](NC(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C15H21NO3S3/c1-10(7-20)14(17)16-13(15(18)19)9-22-8-11-3-5-12(21-2)6-4-11/h3-6,10,13,20H,7-9H2,1-2H3,(H,16,17)(H,18,19)/t10-,13+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 46 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50272142

((2S,5S)-5-(4-isopropylbenzylthio)-1-((S)-3-mercapt...)Show SMILES CC(C)c1ccc(CS[C@H]2CC[C@H](N2C(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C19H27NO3S2/c1-12(2)15-6-4-14(5-7-15)11-25-17-9-8-16(19(22)23)20(17)18(21)13(3)10-24/h4-7,12-13,16-17,24H,8-11H2,1-3H3,(H,22,23)/t13-,16+,17+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 52 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of guinea pig lung leukotriene A4 hydrolase |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266316
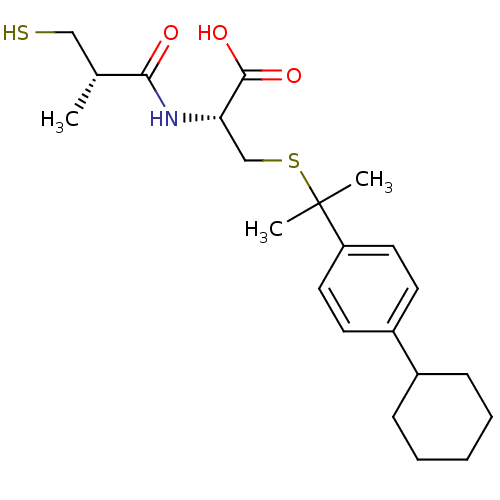
((R)-3-(2-(4-cyclohexylphenyl)propan-2-ylthio)-2-((...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSC(C)(C)c1ccc(cc1)C1CCCCC1)C(O)=O |r| Show InChI InChI=1S/C22H33NO3S2/c1-15(13-27)20(24)23-19(21(25)26)14-28-22(2,3)18-11-9-17(10-12-18)16-7-5-4-6-8-16/h9-12,15-16,19,27H,4-8,13-14H2,1-3H3,(H,23,24)(H,25,26)/t15-,19+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266247
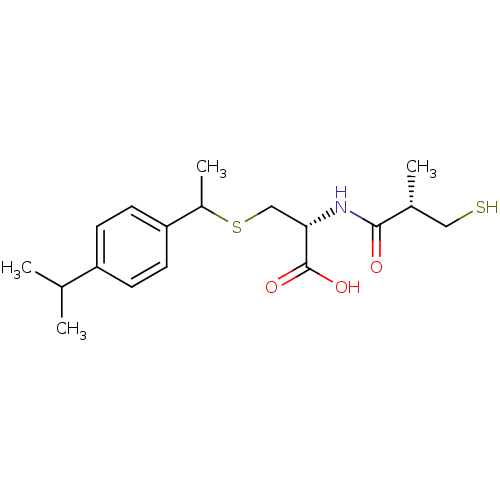
((2R)-3-(1-(4-isopropylphenyl)ethylthio)-2-((S)-3-m...)Show SMILES CC(C)c1ccc(cc1)C(C)SC[C@H](NC(=O)[C@H](C)CS)C(O)=O |r| Show InChI InChI=1S/C18H27NO3S2/c1-11(2)14-5-7-15(8-6-14)13(4)24-10-16(18(21)22)19-17(20)12(3)9-23/h5-8,11-13,16,23H,9-10H2,1-4H3,(H,19,20)(H,21,22)/t12-,13?,16+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 55 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266248
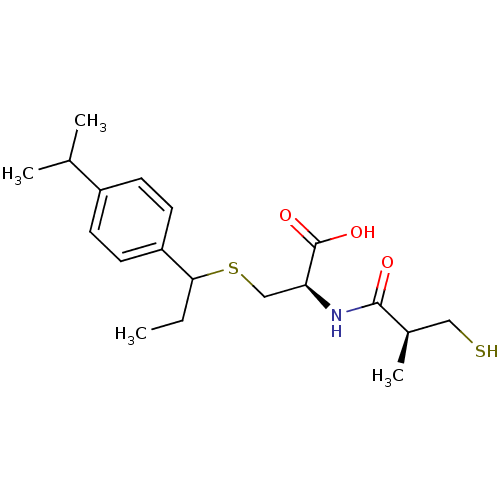
((2R)-3-(1-(4-isopropylphenyl)propylthio)-2-((S)-3-...)Show SMILES CCC(SC[C@H](NC(=O)[C@H](C)CS)C(O)=O)c1ccc(cc1)C(C)C |r| Show InChI InChI=1S/C19H29NO3S2/c1-5-17(15-8-6-14(7-9-15)12(2)3)25-11-16(19(22)23)20-18(21)13(4)10-24/h6-9,12-13,16-17,24H,5,10-11H2,1-4H3,(H,20,21)(H,22,23)/t13-,16+,17?/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 67 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266171
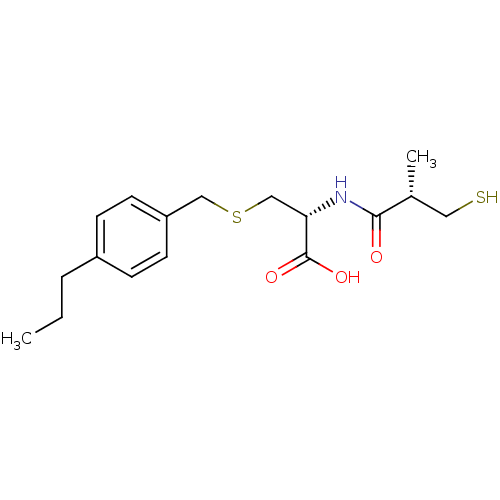
((R)-2-((S)-3-mercapto-2-methylpropanamido)-3-(4-pr...)Show SMILES CCCc1ccc(CSC[C@H](NC(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C17H25NO3S2/c1-3-4-13-5-7-14(8-6-13)10-23-11-15(17(20)21)18-16(19)12(2)9-22/h5-8,12,15,22H,3-4,9-11H2,1-2H3,(H,18,19)(H,20,21)/t12-,15+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 72 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266175
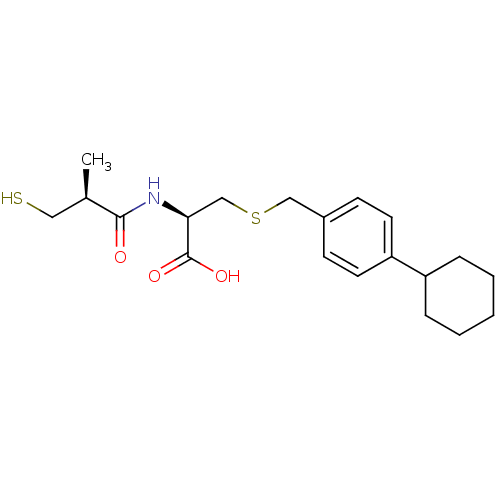
((R)-3-(4-cyclohexylbenzylthio)-2-((S)-3-mercapto-2...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(cc1)C1CCCCC1)C(O)=O |r| Show InChI InChI=1S/C20H29NO3S2/c1-14(11-25)19(22)21-18(20(23)24)13-26-12-15-7-9-17(10-8-15)16-5-3-2-4-6-16/h7-10,14,16,18,25H,2-6,11-13H2,1H3,(H,21,22)(H,23,24)/t14-,18+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 79 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266280
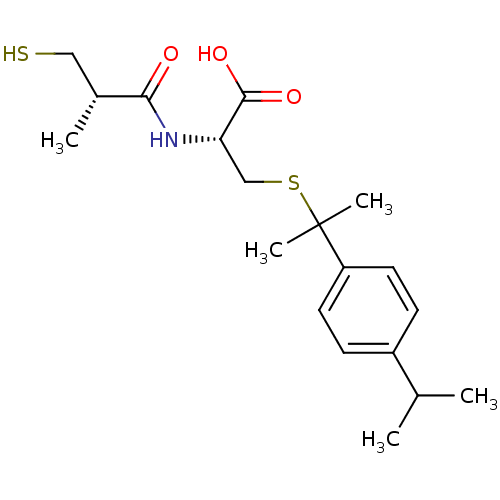
((R)-3-(2-(4-isopropylphenyl)propan-2-ylthio)-2-((S...)Show SMILES CC(C)c1ccc(cc1)C(C)(C)SC[C@H](NC(=O)[C@H](C)CS)C(O)=O |r| Show InChI InChI=1S/C19H29NO3S2/c1-12(2)14-6-8-15(9-7-14)19(4,5)25-11-16(18(22)23)20-17(21)13(3)10-24/h6-9,12-13,16,24H,10-11H2,1-5H3,(H,20,21)(H,22,23)/t13-,16+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 79 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266279
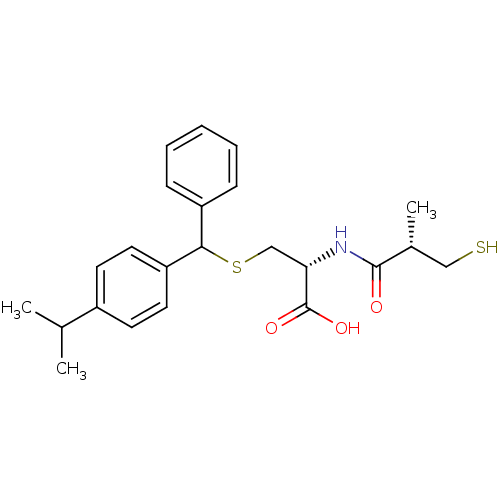
((2R)-3-((4-isopropylphenyl)(phenyl)methylthio)-2-(...)Show SMILES CC(C)c1ccc(cc1)C(SC[C@H](NC(=O)[C@H](C)CS)C(O)=O)c1ccccc1 |r| Show InChI InChI=1S/C23H29NO3S2/c1-15(2)17-9-11-19(12-10-17)21(18-7-5-4-6-8-18)29-14-20(23(26)27)24-22(25)16(3)13-28/h4-12,15-16,20-21,28H,13-14H2,1-3H3,(H,24,25)(H,26,27)/t16-,20+,21?/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 91 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266208
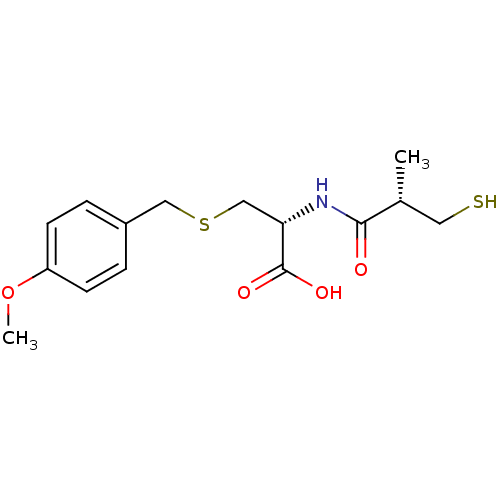
((R)-2-((S)-3-mercapto-2-methylpropanamido)-3-(4-me...)Show SMILES COc1ccc(CSC[C@H](NC(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C15H21NO4S2/c1-10(7-21)14(17)16-13(15(18)19)9-22-8-11-3-5-12(20-2)6-4-11/h3-6,10,13,21H,7-9H2,1-2H3,(H,16,17)(H,18,19)/t10-,13+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266133
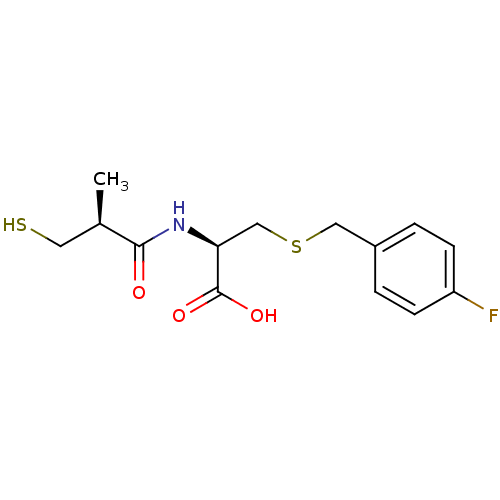
((R)-3-(4-fluorobenzylthio)-2-((S)-3-mercapto-2-met...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(F)cc1)C(O)=O |r| Show InChI InChI=1S/C14H18FNO3S2/c1-9(6-20)13(17)16-12(14(18)19)8-21-7-10-2-4-11(15)5-3-10/h2-5,9,12,20H,6-8H2,1H3,(H,16,17)(H,18,19)/t9-,12+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50272200

((2S,5S)-5-(4-(methylthio)benzylthio)-1-((S)-3-merc...)Show SMILES CSc1ccc(CS[C@H]2CC[C@H](N2C(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C17H23NO3S3/c1-11(9-22)16(19)18-14(17(20)21)7-8-15(18)24-10-12-3-5-13(23-2)6-4-12/h3-6,11,14-15,22H,7-10H2,1-2H3,(H,20,21)/t11-,14+,15+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of guinea pig lung leukotriene A4 hydrolase |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266173

((R)-3-(4-tert-butylbenzylthio)-2-((S)-3-mercapto-2...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(cc1)C(C)(C)C)C(O)=O |r| Show InChI InChI=1S/C18H27NO3S2/c1-12(9-23)16(20)19-15(17(21)22)11-24-10-13-5-7-14(8-6-13)18(2,3)4/h5-8,12,15,23H,9-11H2,1-4H3,(H,19,20)(H,21,22)/t12-,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 130 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266138
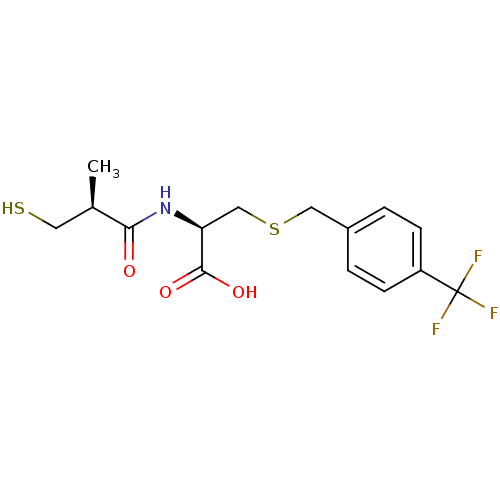
((R)-2-((S)-3-mercapto-2-methylpropanamido)-3-(4-(t...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(cc1)C(F)(F)F)C(O)=O |r| Show InChI InChI=1S/C15H18F3NO3S2/c1-9(6-23)13(20)19-12(14(21)22)8-24-7-10-2-4-11(5-3-10)15(16,17)18/h2-5,9,12,23H,6-8H2,1H3,(H,19,20)(H,21,22)/t9-,12+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266136

((R)-3-(4-iodobenzylthio)-2-((S)-3-mercapto-2-methy...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(I)cc1)C(O)=O |r| Show InChI InChI=1S/C14H18INO3S2/c1-9(6-20)13(17)16-12(14(18)19)8-21-7-10-2-4-11(15)5-3-10/h2-5,9,12,20H,6-8H2,1H3,(H,16,17)(H,18,19)/t9-,12+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266245
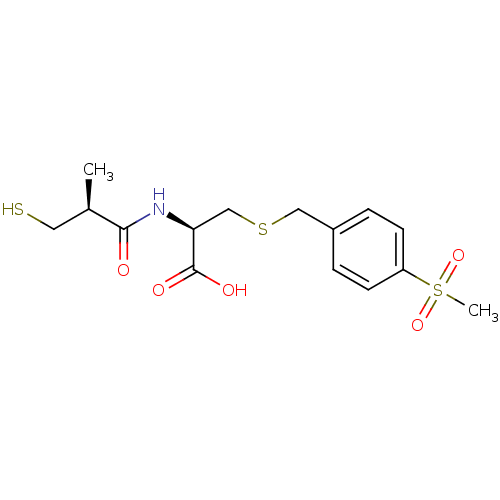
((R)-2-((S)-3-mercapto-2-methylpropanamido)-3-(4-(m...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(cc1)S(C)(=O)=O)C(O)=O |r| Show InChI InChI=1S/C15H21NO5S3/c1-10(7-22)14(17)16-13(15(18)19)9-23-8-11-3-5-12(6-4-11)24(2,20)21/h3-6,10,13,22H,7-9H2,1-2H3,(H,16,17)(H,18,19)/t10-,13+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 158 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266276
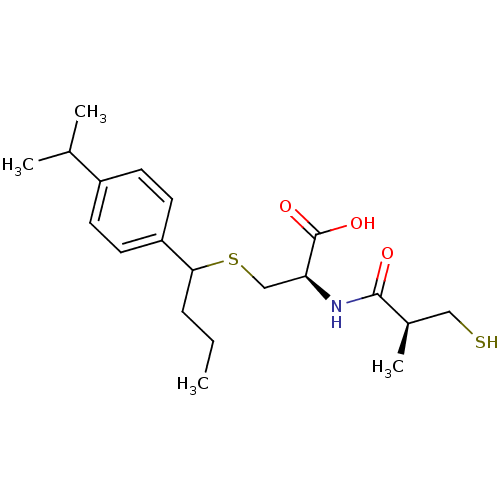
((2R)-3-(1-(4-isopropylphenyl)butylthio)-2-((S)-3-m...)Show SMILES CCCC(SC[C@H](NC(=O)[C@H](C)CS)C(O)=O)c1ccc(cc1)C(C)C |r| Show InChI InChI=1S/C20H31NO3S2/c1-5-6-18(16-9-7-15(8-10-16)13(2)3)26-12-17(20(23)24)21-19(22)14(4)11-25/h7-10,13-14,17-18,25H,5-6,11-12H2,1-4H3,(H,21,22)(H,23,24)/t14-,17+,18?/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266172
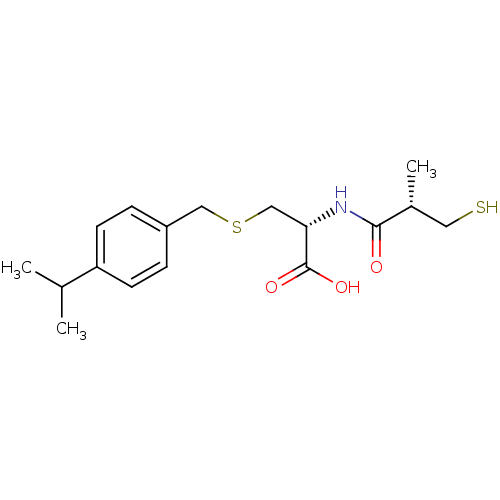
((R)-3-(4-isopropylbenzylthio)-2-((S)-3-mercapto-2-...)Show SMILES CC(C)c1ccc(CSC[C@H](NC(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C17H25NO3S2/c1-11(2)14-6-4-13(5-7-14)9-23-10-15(17(20)21)18-16(19)12(3)8-22/h4-7,11-12,15,22H,8-10H2,1-3H3,(H,18,19)(H,20,21)/t12-,15+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266247

((2R)-3-(1-(4-isopropylphenyl)ethylthio)-2-((S)-3-m...)Show SMILES CC(C)c1ccc(cc1)C(C)SC[C@H](NC(=O)[C@H](C)CS)C(O)=O |r| Show InChI InChI=1S/C18H27NO3S2/c1-11(2)14-5-7-15(8-6-14)13(4)24-10-16(18(21)22)19-17(20)12(3)9-23/h5-8,11-13,16,23H,9-10H2,1-4H3,(H,19,20)(H,21,22)/t12-,13?,16+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266315
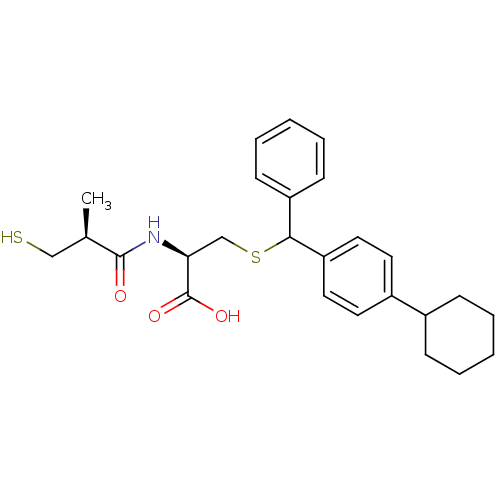
((2R)-3-((4-cyclohexylphenyl)(phenyl)methylthio)-2-...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSC(c1ccccc1)c1ccc(cc1)C1CCCCC1)C(O)=O |r| Show InChI InChI=1S/C26H33NO3S2/c1-18(16-31)25(28)27-23(26(29)30)17-32-24(21-10-6-3-7-11-21)22-14-12-20(13-15-22)19-8-4-2-5-9-19/h3,6-7,10-15,18-19,23-24,31H,2,4-5,8-9,16-17H2,1H3,(H,27,28)(H,29,30)/t18-,23+,24?/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266171

((R)-2-((S)-3-mercapto-2-methylpropanamido)-3-(4-pr...)Show SMILES CCCc1ccc(CSC[C@H](NC(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C17H25NO3S2/c1-3-4-13-5-7-14(8-6-13)10-23-11-15(17(20)21)18-16(19)12(2)9-22/h5-8,12,15,22H,3-4,9-11H2,1-2H3,(H,18,19)(H,20,21)/t12-,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266137
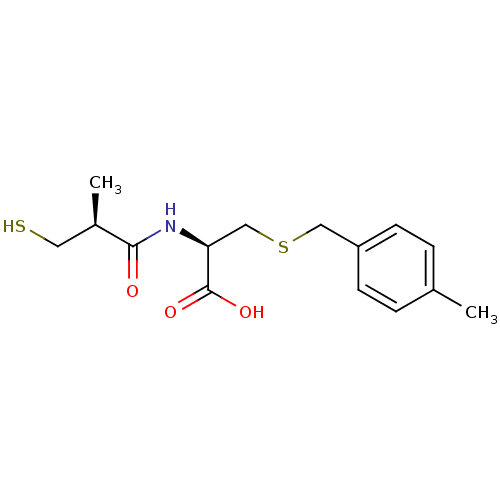
((R)-2-((S)-3-mercapto-2-methylpropanamido)-3-(4-me...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(C)cc1)C(O)=O |r| Show InChI InChI=1S/C15H21NO3S2/c1-10-3-5-12(6-4-10)8-21-9-13(15(18)19)16-14(17)11(2)7-20/h3-6,11,13,20H,7-9H2,1-2H3,(H,16,17)(H,18,19)/t11-,13+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 240 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266134
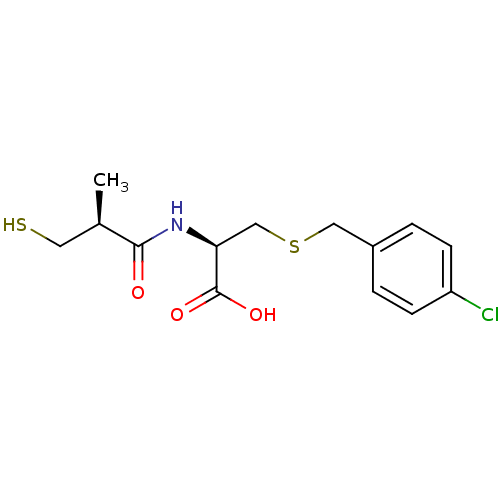
((R)-3-(4-chlorobenzylthio)-2-((S)-3-mercapto-2-met...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(Cl)cc1)C(O)=O |r| Show InChI InChI=1S/C14H18ClNO3S2/c1-9(6-20)13(17)16-12(14(18)19)8-21-7-10-2-4-11(15)5-3-10/h2-5,9,12,20H,6-8H2,1H3,(H,16,17)(H,18,19)/t9-,12+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266280

((R)-3-(2-(4-isopropylphenyl)propan-2-ylthio)-2-((S...)Show SMILES CC(C)c1ccc(cc1)C(C)(C)SC[C@H](NC(=O)[C@H](C)CS)C(O)=O |r| Show InChI InChI=1S/C19H29NO3S2/c1-12(2)14-6-8-15(9-7-14)19(4,5)25-11-16(18(22)23)20-17(21)13(3)10-24/h6-9,12-13,16,24H,10-11H2,1-5H3,(H,20,21)(H,22,23)/t13-,16+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266209
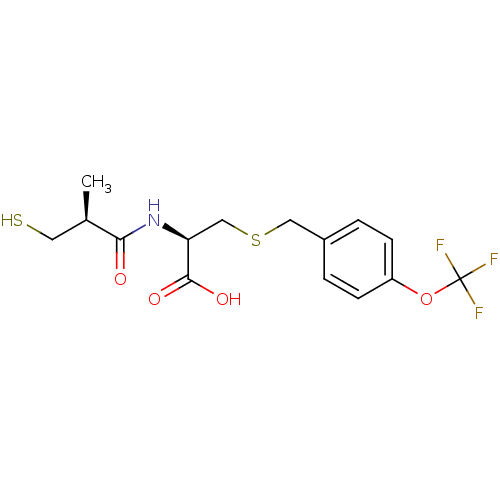
((R)-2-((S)-3-mercapto-2-methylpropanamido)-3-(4-(t...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(OC(F)(F)F)cc1)C(O)=O |r| Show InChI InChI=1S/C15H18F3NO4S2/c1-9(6-24)13(20)19-12(14(21)22)8-25-7-10-2-4-11(5-3-10)23-15(16,17)18/h2-5,9,12,24H,6-8H2,1H3,(H,19,20)(H,21,22)/t9-,12+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50272202

((2S,5S)-5-(4-cyclohexylbenzylthio)-1-((S)-3-mercap...)Show SMILES C[C@H](CS)C(=O)N1[C@H](CC[C@H]1C(O)=O)SCc1ccc(cc1)C1CCCCC1 |r| Show InChI InChI=1S/C22H31NO3S2/c1-15(13-27)21(24)23-19(22(25)26)11-12-20(23)28-14-16-7-9-18(10-8-16)17-5-3-2-4-6-17/h7-10,15,17,19-20,27H,2-6,11-14H2,1H3,(H,25,26)/t15-,19+,20+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 280 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of ACE (unknown origin) |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266135
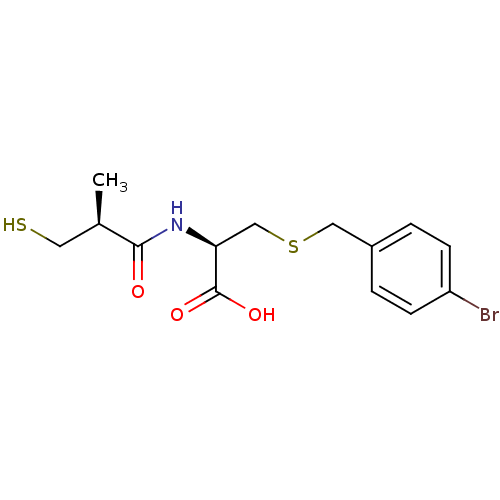
((R)-3-(4-bromobenzylthio)-2-((S)-3-mercapto-2-meth...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(Br)cc1)C(O)=O |r| Show InChI InChI=1S/C14H18BrNO3S2/c1-9(6-20)13(17)16-12(14(18)19)8-21-7-10-2-4-11(15)5-3-10/h2-5,9,12,20H,6-8H2,1H3,(H,16,17)(H,18,19)/t9-,12+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 280 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266174
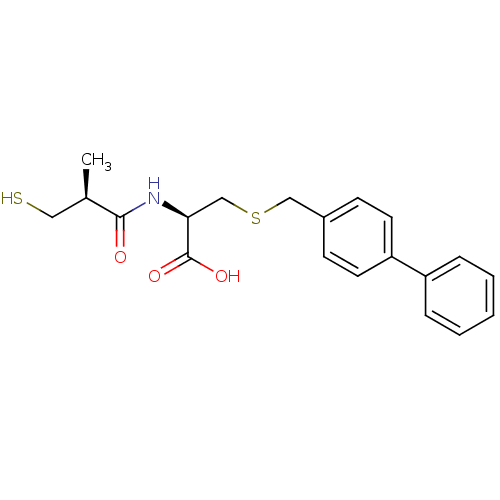
((R)-3-(biphenyl-4-ylmethylthio)-2-((S)-3-mercapto-...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(cc1)-c1ccccc1)C(O)=O |r| Show InChI InChI=1S/C20H23NO3S2/c1-14(11-25)19(22)21-18(20(23)24)13-26-12-15-7-9-17(10-8-15)16-5-3-2-4-6-16/h2-10,14,18,25H,11-13H2,1H3,(H,21,22)(H,23,24)/t14-,18+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 280 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50266170

((R)-3-(4-ethylbenzylthio)-2-((S)-3-mercapto-2-meth...)Show SMILES CCc1ccc(CSC[C@H](NC(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C16H23NO3S2/c1-3-12-4-6-13(7-5-12)9-22-10-14(16(19)20)17-15(18)11(2)8-21/h4-7,11,14,21H,3,8-10H2,1-2H3,(H,17,18)(H,19,20)/t11-,14+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 280 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of LTA4 hydrolase in guinea pig lung assessed as inhibition of LTB4 production |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |
Leukotriene A-4 hydrolase
(Cavia porcellus) | BDBM50272203

((2S,5R)-5-(4-isopropylbenzylthio)-1-((S)-3-mercapt...)Show SMILES CC(C)c1ccc(CS[C@@H]2CC[C@H](N2C(=O)[C@H](C)CS)C(O)=O)cc1 |r| Show InChI InChI=1S/C19H27NO3S2/c1-12(2)15-6-4-14(5-7-15)11-25-17-9-8-16(19(22)23)20(17)18(21)13(3)10-24/h4-7,12-13,16-17,24H,8-11H2,1-3H3,(H,22,23)/t13-,16+,17-/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of guinea pig lung leukotriene A4 hydrolase |
Bioorg Med Chem Lett 18: 4529-32 (2008)
Article DOI: 10.1016/j.bmcl.2008.07.043
BindingDB Entry DOI: 10.7270/Q2PR7VSQ |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme
(Homo sapiens (Human)) | BDBM50266213
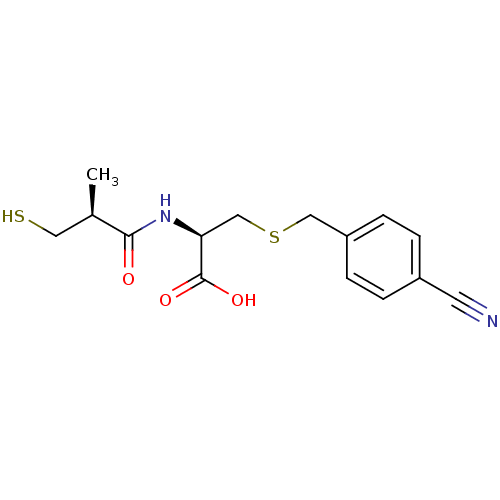
((R)-3-(4-cyanobenzylthio)-2-((S)-3-mercapto-2-meth...)Show SMILES C[C@H](CS)C(=O)N[C@@H](CSCc1ccc(cc1)C#N)C(O)=O |r| Show InChI InChI=1S/C15H18N2O3S2/c1-10(7-21)14(18)17-13(15(19)20)9-22-8-12-4-2-11(6-16)3-5-12/h2-5,10,13,21H,7-9H2,1H3,(H,17,18)(H,19,20)/t10-,13+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Santen Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Inhibition of angiotensin-converting enzyme (unknown origin) in whole blood by HPLC |
Bioorg Med Chem Lett 19: 442-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.11.042
BindingDB Entry DOI: 10.7270/Q20K28FC |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data