

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Gastrin-releasing peptide receptor (RAT) | BDBM50005601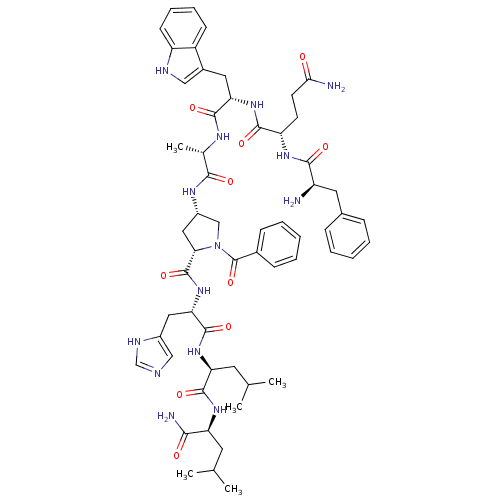 (CHEMBL2369819) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Patents Similars | PubMed | >0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS-Universités Montpellier I & II Curated by ChEMBL | Assay Description In vitro binding affinity against bombesin / GRP receptors on rat pancreatic acini. | J Med Chem 43: 2356-61 (2000) BindingDB Entry DOI: 10.7270/Q21J990K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Gastrin-releasing peptide receptor (RAT) | BDBM50408923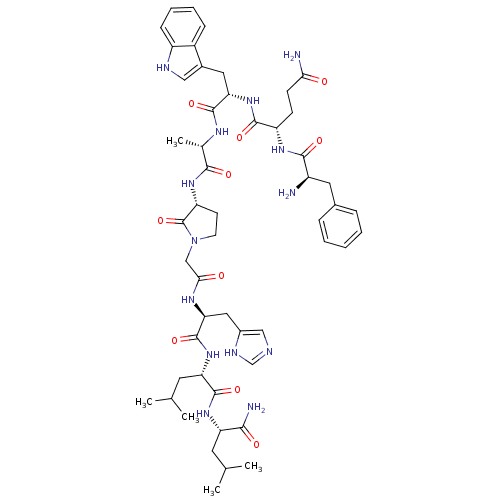 (CHEMBL2114155) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | >0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNRS-Universités Montpellier I & II Curated by ChEMBL | Assay Description In vitro binding affinity against bombesin / GRP receptors on rat pancreatic acini. | J Med Chem 43: 2356-61 (2000) BindingDB Entry DOI: 10.7270/Q21J990K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine deaminase (Bos taurus (bovine)) | BDBM22925 ((8R)-3-[(2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)oxol...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase DrugBank KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PubMed | 0.00100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ di Camerino Curated by ChEMBL | Assay Description Compound was tested for the inhibition of adenosine deaminase from calf intestine; Range of 0.01-0.001 nM | J Med Chem 37: 201-5 (1994) BindingDB Entry DOI: 10.7270/Q2KD1ZHQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Homo sapiens (Human)) | BDBM50434903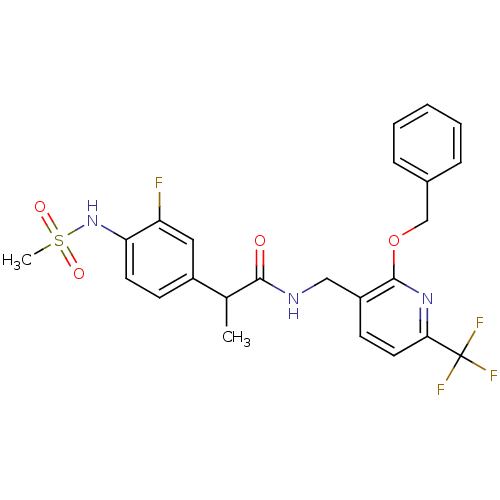 (CHEMBL2385223) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.00100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University Curated by ChEMBL | Assay Description Antagonist activity at human TRPV1 expressed in CHOK1 cells assessed as inhibition of N-arachidonoyldopamine-induced activity after 5 mins by FLIPR a... | Eur J Med Chem 64: 589-602 (2013) Article DOI: 10.1016/j.ejmech.2013.04.003 BindingDB Entry DOI: 10.7270/Q2BZ67FC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50016460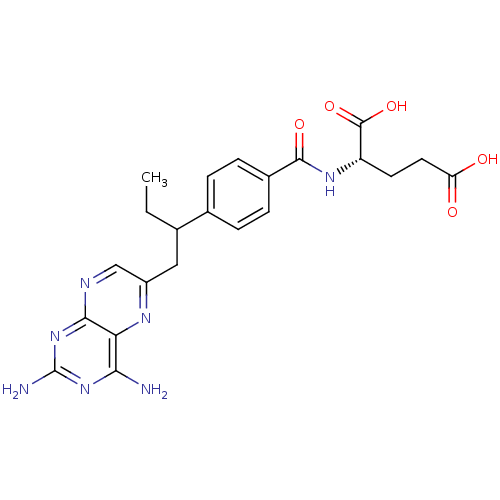 ((S)-2-{4-[1-(2,4-Diamino-pteridin-6-ylmethyl)-prop...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | PubMed | 0.00280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International Curated by ChEMBL | Assay Description Concentration of the compound inhibiting dihydrofolate reductase derived from L1210 cells | J Med Chem 33: 212-5 (1990) BindingDB Entry DOI: 10.7270/Q2FX78FR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50405400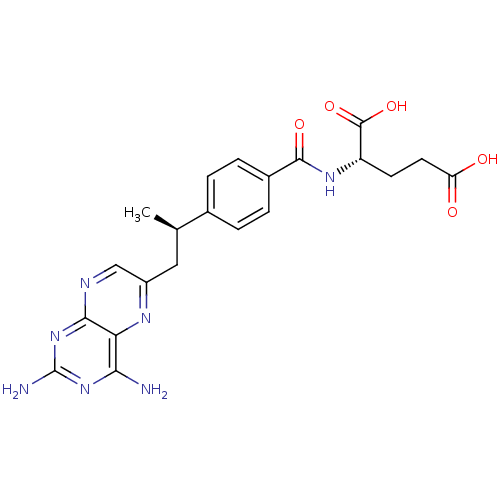 (CHEMBL2051987) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | PubMed | 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was evaluated for binding affinity against Dihydrofolate reductase of L1210 cells | J Med Chem 29: 1056-61 (1986) BindingDB Entry DOI: 10.7270/Q2P55MHM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50405400 (CHEMBL2051987) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | PubMed | 0.00300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was evaluated for binding affinity against Dihydrofolate reductase of L1210 cells | J Med Chem 29: 1056-61 (1986) BindingDB Entry DOI: 10.7270/Q2P55MHM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50010932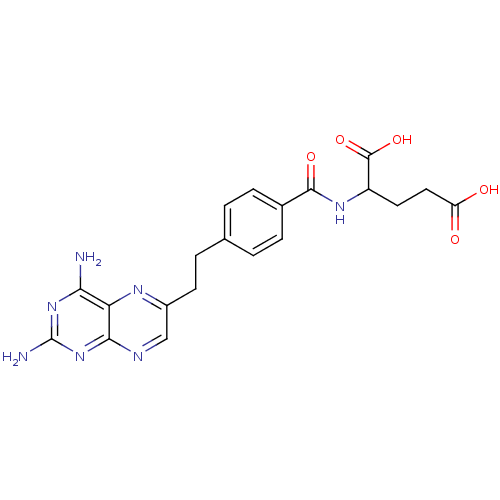 ((10-DA, 10-Deazaminopterin)2-{4-[2-(2,4-Diamino-pt...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.00340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International Curated by ChEMBL | Assay Description Concentration of the compound inhibiting dihydrofolate reductase derived from L1210 cells | J Med Chem 33: 212-5 (1990) BindingDB Entry DOI: 10.7270/Q2FX78FR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50226274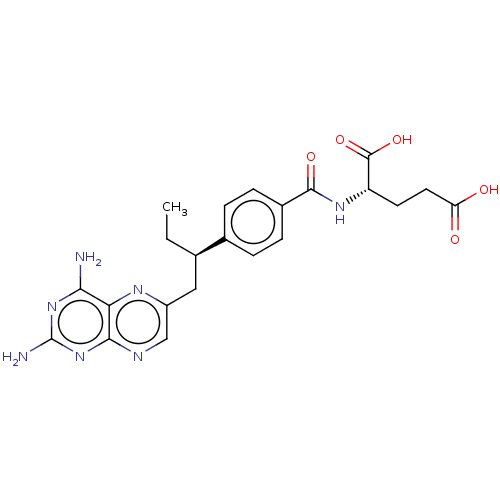 (CHEMBL3349020) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem | PubMed | 0.00400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was evaluated for binding affinity against Dihydrofolate reductase of L1210 cells | J Med Chem 29: 1056-61 (1986) BindingDB Entry DOI: 10.7270/Q2P55MHM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM18050 (2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)a...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB PubMed | 0.00400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was evaluated for binding affinity against Dihydrofolate reductase of L1210 cells | J Med Chem 29: 1056-61 (1986) BindingDB Entry DOI: 10.7270/Q2P55MHM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50014707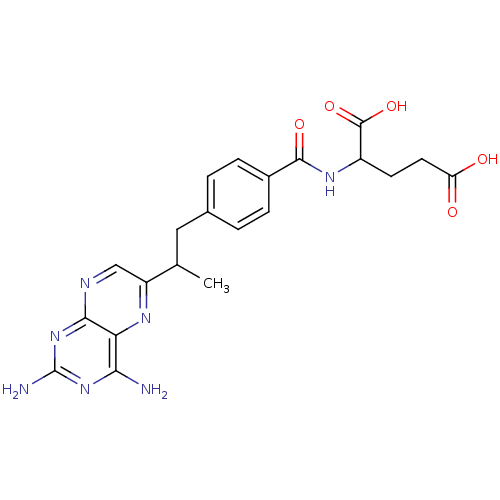 (2-{4-[2-(2,4-Diamino-pteridin-6-yl)-propyl]-benzoy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.00520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International Curated by ChEMBL | Assay Description Concentration of the compound inhibiting dihydrofolate reductase derived from L1210 cells | J Med Chem 33: 212-5 (1990) BindingDB Entry DOI: 10.7270/Q2FX78FR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50014706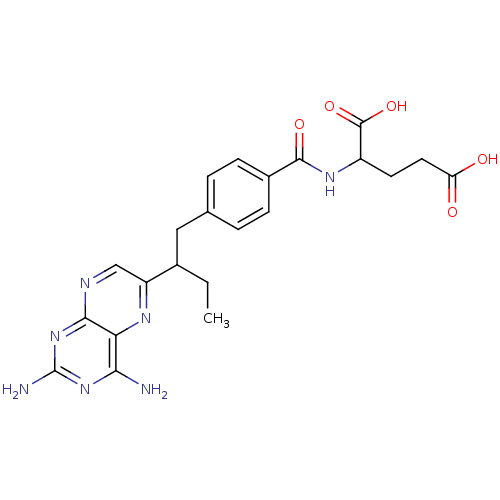 (2-{4-[2-(2,4-Diamino-pteridin-6-yl)-butyl]-benzoyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.00550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International Curated by ChEMBL | Assay Description Concentration of the compound inhibiting Dihydrofolate reductase derived from L1210 cells | J Med Chem 33: 212-5 (1990) BindingDB Entry DOI: 10.7270/Q2FX78FR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM18050 (2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)a...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB PubMed | 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International Curated by ChEMBL | Assay Description Inhibitory activity of Dihydrofolate reductase derived from L1210 cell line. | J Med Chem 33: 673-7 (1990) BindingDB Entry DOI: 10.7270/Q2T43S3F | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM18050 (2-[(4-{[(2,4-diaminopteridin-6-yl)methyl](methyl)a...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB PubMed | 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International Curated by ChEMBL | Assay Description Inhibition of the dihydrofolate reductase enzyme(DHFR) derived from L1210 murine leukemia cells. | J Med Chem 35: 320-4 (1992) BindingDB Entry DOI: 10.7270/Q2WQ02RM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Dimer of Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM554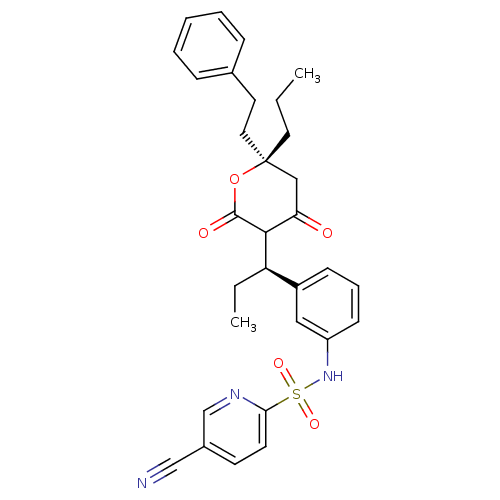 (5-cyano-N-{3-[(1R)-1-[(6R)-4-hydroxy-2-oxo-6-(2-ph...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 0.00700 | -63.0 | n/a | n/a | n/a | n/a | n/a | 5.0 | 22 |
Upjohn | Assay Description HIV-1 protease was purified and refolded from E. coli inclusion bodies. The substrate used spans the p17-p24 processing site (R-V-S-Q-N-Y-P-I-V-Q-N-K... | J Med Chem 39: 4349-53 (1996) Article DOI: 10.1021/jm960541s BindingDB Entry DOI: 10.7270/Q2HQ3X35 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50008287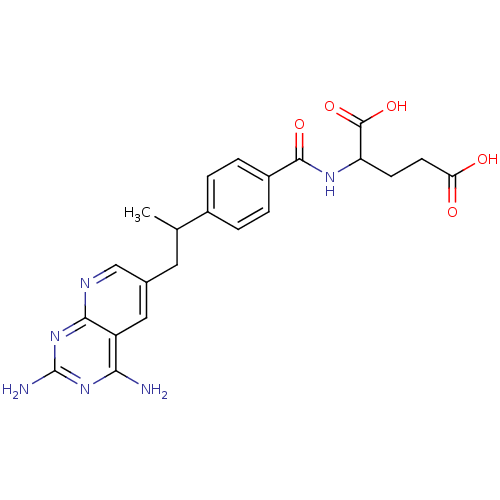 (2-{4-[2-(2,4-Diamino-pyrido[2,3-d]pyrimidin-6-yl)-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International Curated by ChEMBL | Assay Description Inhibition of the dihydrofolate reductase enzyme(DHFR) derived from L1210 murine leukemia cells. | J Med Chem 35: 320-4 (1992) BindingDB Entry DOI: 10.7270/Q2WQ02RM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50008287 (2-{4-[2-(2,4-Diamino-pyrido[2,3-d]pyrimidin-6-yl)-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.00700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International Curated by ChEMBL | Assay Description Inhibitory activity of Dihydrofolate reductase derived from L1210 cell line. | J Med Chem 33: 673-7 (1990) BindingDB Entry DOI: 10.7270/Q2T43S3F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dimer of Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM558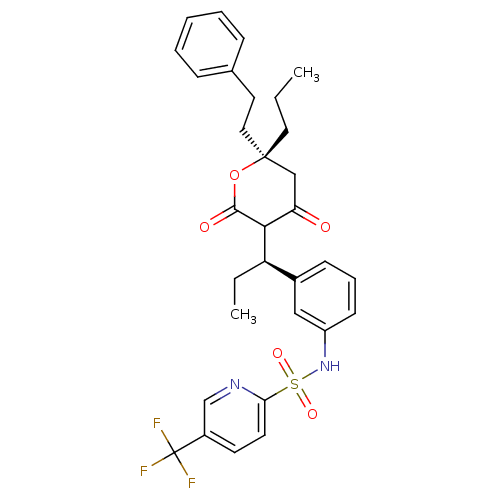 (N-{3-[(1R)-1-[(6R)-4-hydroxy-2-oxo-6-(2-phenylethy...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 0.00800 | -62.7 | n/a | n/a | n/a | n/a | n/a | 5.0 | 22 |
Upjohn | Assay Description HIV-1 protease was purified and refolded from E. coli inclusion bodies. The substrate used spans the p17-p24 processing site (R-V-S-Q-N-Y-P-I-V-Q-N-K... | J Med Chem 39: 4349-53 (1996) Article DOI: 10.1021/jm960541s BindingDB Entry DOI: 10.7270/Q2HQ3X35 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Alpha-1B adrenergic receptor (C.H.O.) | BDBM50007568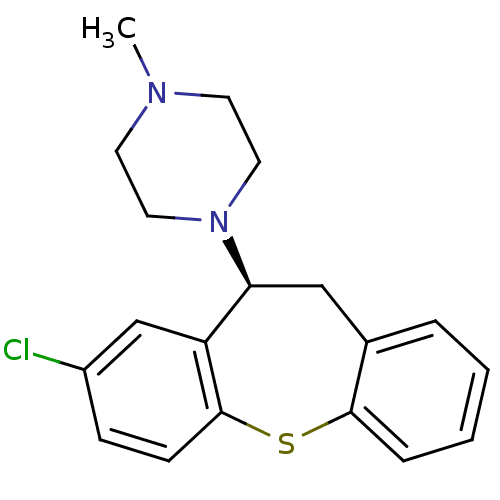 (1-((S)-8-Chloro-10,11-dihydro-dibenzo[b,f]thiepin-...) | Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen Curated by ChEMBL | Assay Description Displacement of [3H]prozosin from hamster cloned alpha1b receptor | J Med Chem 53: 7021-34 (2010) Article DOI: 10.1021/jm100652h BindingDB Entry DOI: 10.7270/Q20C4W0P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-1B adrenergic receptor (C.H.O.) | BDBM50007568 (1-((S)-8-Chloro-10,11-dihydro-dibenzo[b,f]thiepin-...) | Reactome pathway UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen Curated by ChEMBL | Assay Description Displacement of [3H]prozosin from hamster cloned alpha1b receptor | J Med Chem 53: 7021-34 (2010) Article DOI: 10.1021/jm100652h BindingDB Entry DOI: 10.7270/Q20C4W0P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Homo sapiens (Human)) | BDBM50398494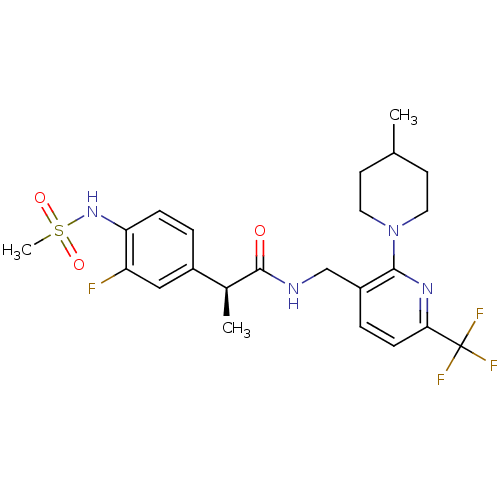 (CHEMBL2177429) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University Curated by ChEMBL | Assay Description Antagonist activity at human TRPV1 expressed in CHOK1 cells assessed as inhibition of N-acetyldopamine-induced activity after 5 mins by FLIPR assay | Bioorg Med Chem 21: 6657-64 (2013) Article DOI: 10.1016/j.bmc.2013.08.015 BindingDB Entry DOI: 10.7270/Q26Q1ZPN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Apoptosis regulator Bcl-2 (Homo sapiens (Human)) | BDBM50270877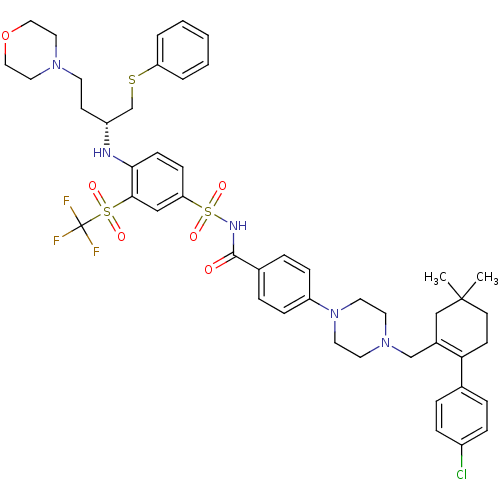 ((R)-4-(4-((2-(4-chlorophenyl)-5,5-dimethylcyclohex...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | <0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Taros Chemicals GmbH & Co. KG Curated by ChEMBL | Assay Description Inhibition of BCL-2 (unknown origin) by fluorescence polarization assay | Eur J Med Chem 167: 76-95 (2019) Article DOI: 10.1016/j.ejmech.2019.01.084 BindingDB Entry DOI: 10.7270/Q2HH6PDC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Homo sapiens (Human)) | BDBM50398494 (CHEMBL2177429) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University Curated by ChEMBL | Assay Description Antagonist activity at human TRPV1 expressed in CHOK1 cells assessed as inhibition of N-acetyldopamine-induced activity after 5 mins by FLIPR assay | J Med Chem 55: 8392-408 (2012) Article DOI: 10.1021/jm300780p BindingDB Entry DOI: 10.7270/Q2TX3GH1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50101499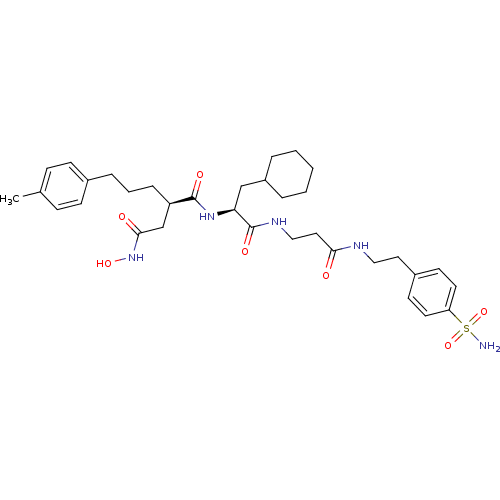 (CHEMBL74040 | N*1*-(2-Cyclohexyl-1-{2-[2-(4-sulfam...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Royal Danish School of Pharmacy Curated by ChEMBL | Assay Description Inhibitory constant against matrix metalloproteinase-2 | J Med Chem 44: 2333-43 (2001) BindingDB Entry DOI: 10.7270/Q2JH3KFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50101505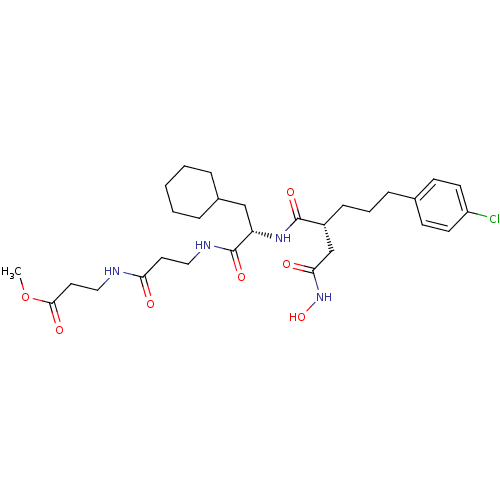 (3-(3-{2-[5-(4-Chloro-phenyl)-2-hydroxycarbamoylmet...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Royal Danish School of Pharmacy Curated by ChEMBL | Assay Description Inhibitory constant against matrix metalloproteinase-2 | J Med Chem 44: 2333-43 (2001) BindingDB Entry DOI: 10.7270/Q2JH3KFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50101508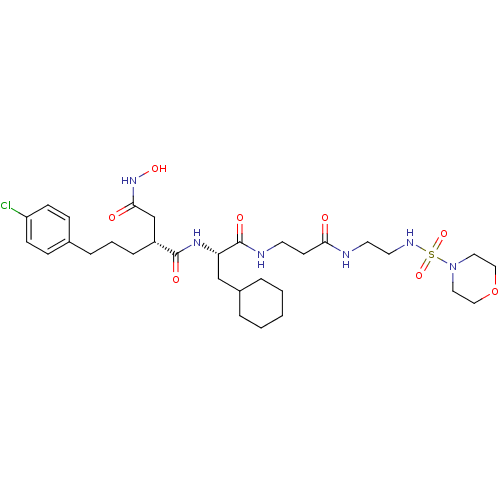 (2-[3-(4-Chloro-phenyl)-propyl]-N*1*-(2-cyclohexyl-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Royal Danish School of Pharmacy Curated by ChEMBL | Assay Description Inhibitory constant against matrix metalloproteinase-2 | J Med Chem 44: 2333-43 (2001) BindingDB Entry DOI: 10.7270/Q2JH3KFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50101516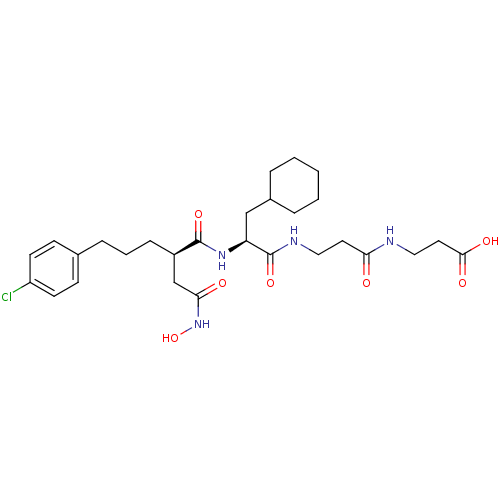 (3-(3-{2-[5-(4-Chloro-phenyl)-2-hydroxycarbamoylmet...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Royal Danish School of Pharmacy Curated by ChEMBL | Assay Description Inhibitory constant against matrix metalloproteinase-2 | J Med Chem 44: 2333-43 (2001) BindingDB Entry DOI: 10.7270/Q2JH3KFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50101512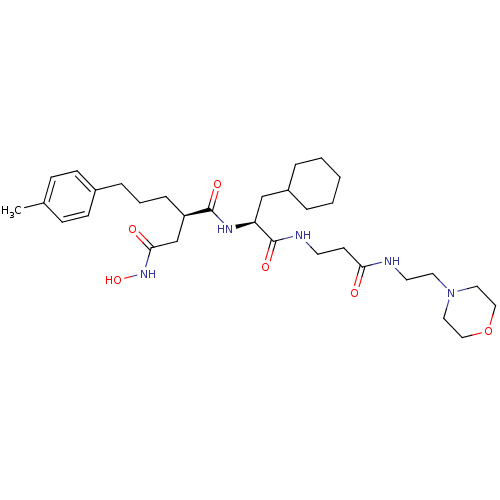 (CHEMBL306033 | N*1*-{2-Cyclohexyl-1-[2-(2-morpholi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Royal Danish School of Pharmacy Curated by ChEMBL | Assay Description Inhibitory constant against matrix metalloproteinase-2 | J Med Chem 44: 2333-43 (2001) BindingDB Entry DOI: 10.7270/Q2JH3KFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50101509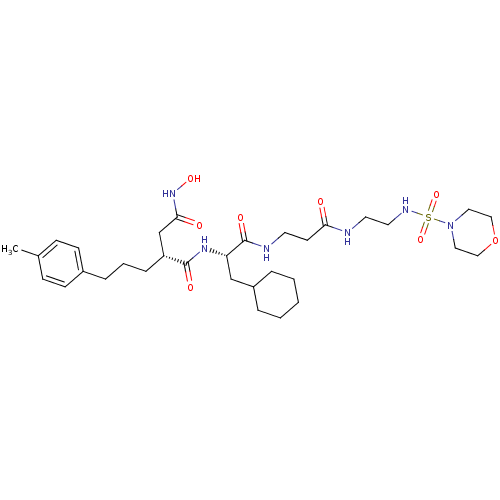 (CHEMBL306947 | N*1*-(2-Cyclohexyl-1-{2-[2-(morphol...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Royal Danish School of Pharmacy Curated by ChEMBL | Assay Description Inhibitory constant against matrix metalloproteinase-2 | J Med Chem 44: 2333-43 (2001) BindingDB Entry DOI: 10.7270/Q2JH3KFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50405401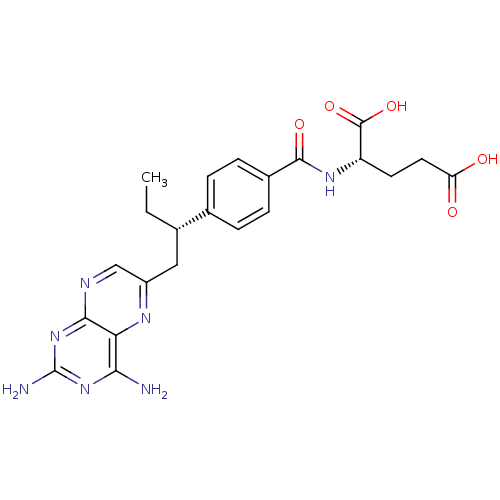 (CHEMBL2051990) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | PubMed | 0.0120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was evaluated for binding affinity against Dihydrofolate reductase of L1210 cells | J Med Chem 29: 1056-61 (1986) BindingDB Entry DOI: 10.7270/Q2P55MHM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50008285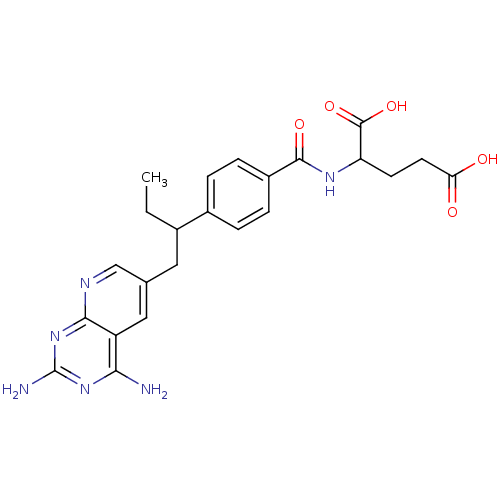 (2-{4-[1-(2,4-Diamino-pyrido[2,3-d]pyrimidin-6-ylme...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International Curated by ChEMBL | Assay Description Inhibition of the dihydrofolate reductase enzyme(DHFR) derived from L1210 murine leukemia cells. | J Med Chem 35: 320-4 (1992) BindingDB Entry DOI: 10.7270/Q2WQ02RM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50008285 (2-{4-[1-(2,4-Diamino-pyrido[2,3-d]pyrimidin-6-ylme...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International Curated by ChEMBL | Assay Description Inhibitory activity of Dihydrofolate reductase derived from L1210 cell line. | J Med Chem 33: 673-7 (1990) BindingDB Entry DOI: 10.7270/Q2T43S3F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dihydrofolate reductase (Mus musculus (Mouse)) | BDBM50008286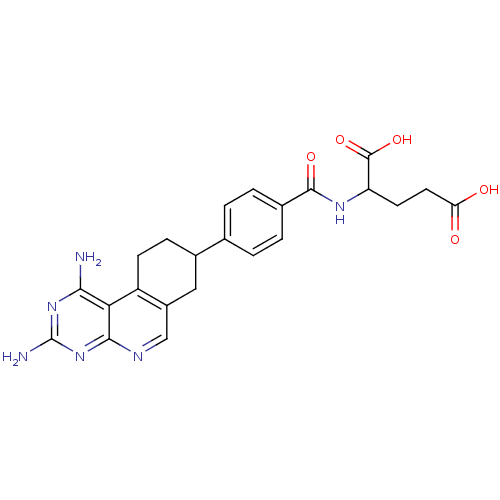 (2-[4-(1,3-Diamino-7,8,9,10-tetrahydro-pyrimido[4,5...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SRI International Curated by ChEMBL | Assay Description Inhibition of the dihydrofolate reductase enzyme(DHFR) derived from L1210 murine leukemia cells. | J Med Chem 35: 320-4 (1992) BindingDB Entry DOI: 10.7270/Q2WQ02RM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Dimer of Gag-Pol polyprotein [489-587] (Human immunodeficiency virus type 1) | BDBM557 (N-{3-[(1R)-1-[(6S)-4-hydroxy-2-oxo-6-(2-phenylethy...) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 0.0180 | -60.7 | n/a | n/a | n/a | n/a | n/a | 5.0 | 22 |
Upjohn | Assay Description HIV-1 protease was purified and refolded from E. coli inclusion bodies. The substrate used spans the p17-p24 processing site (R-V-S-Q-N-Y-P-I-V-Q-N-K... | J Med Chem 39: 4349-53 (1996) Article DOI: 10.1021/jm960541s BindingDB Entry DOI: 10.7270/Q2HQ3X35 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Calcitonin gene-related peptide type 1 receptor/Receptor activity-modifying protein 1 (Homo sapiens (Human)) | BDBM391716 (N-[(2R)-3-(7-methyl-1H- | US10300056, Example 1 | ...) | PDB KEGG UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sosei Heptares Curated by ChEMBL | Assay Description Displacement of [3H]telcagepant from recombinant human CLR/RAMP1 expressed in Sf21 insect cell membranes measured after 60 mins by microbeta scintill... | J Med Chem 63: 7906-7920 (2020) Article DOI: 10.1021/acs.jmedchem.0c01003 BindingDB Entry DOI: 10.7270/Q2NK3JKR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50101518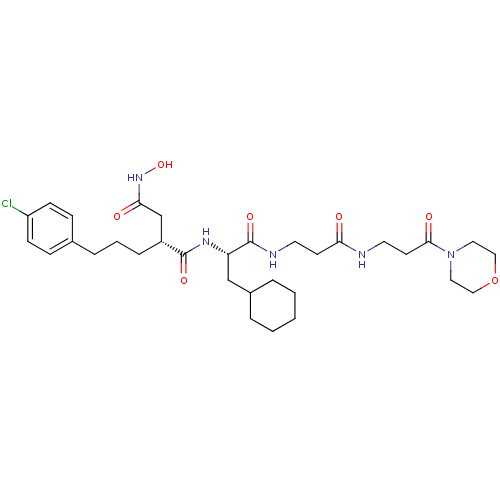 (2-[3-(4-Chloro-phenyl)-propyl]-N*1*-{2-cyclohexyl-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Royal Danish School of Pharmacy Curated by ChEMBL | Assay Description Inhibitory constant against matrix metalloproteinase-2 | J Med Chem 44: 2333-43 (2001) BindingDB Entry DOI: 10.7270/Q2JH3KFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50101520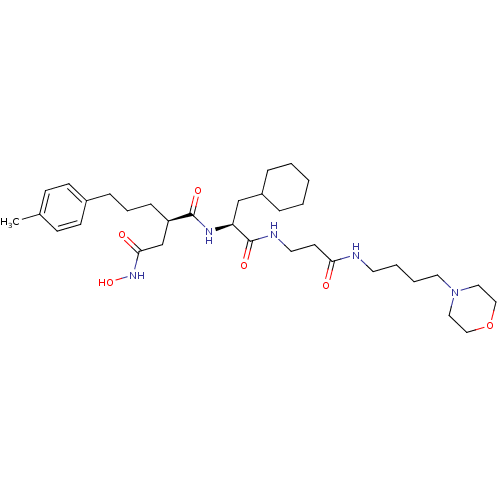 (CHEMBL77663 | N*1*-{2-Cyclohexyl-1-[2-(4-morpholin...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Royal Danish School of Pharmacy Curated by ChEMBL | Assay Description Inhibitory constant against matrix metalloproteinase-2 | J Med Chem 44: 2333-43 (2001) BindingDB Entry DOI: 10.7270/Q2JH3KFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50101504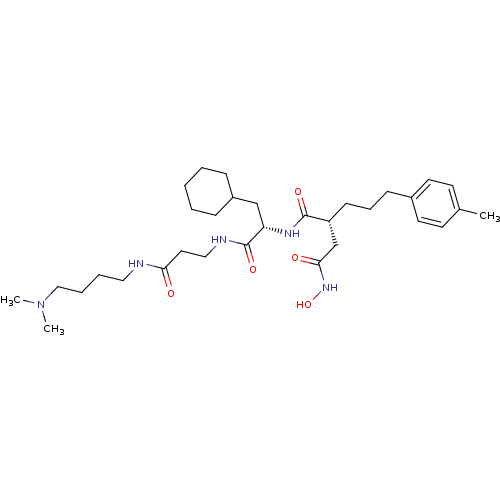 (CHEMBL77057 | N*1*-{2-Cyclohexyl-1-[2-(4-dimethyla...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Royal Danish School of Pharmacy Curated by ChEMBL | Assay Description Inhibitory constant against matrix metalloproteinase-2 | J Med Chem 44: 2333-43 (2001) BindingDB Entry DOI: 10.7270/Q2JH3KFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prolyl endopeptidase (Sus scrofa) | BDBM50155838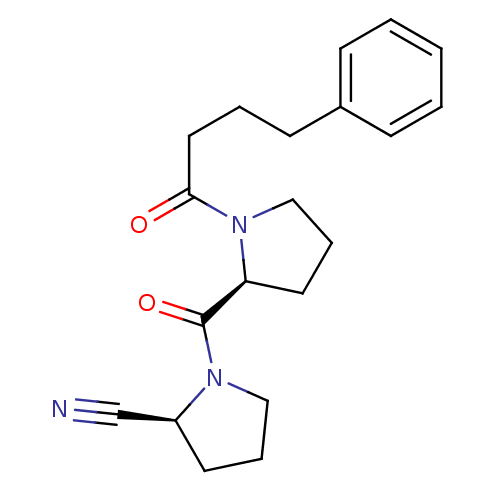 ((S)-1-((S)-1-(4-phenylbutanoyl)pyrrolidine-2-carbo...) | PDB KEGG UniProtKB/SwissProt DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Kuopio Curated by ChEMBL | Assay Description Inhibitory activity against prolyl oligopeptidase of porcine brain homogenate | J Med Chem 47: 5605-7 (2004) Article DOI: 10.1021/jm049503w BindingDB Entry DOI: 10.7270/Q25D8R9G | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Homo sapiens (Human)) | BDBM50061298 (CHEMBL3393837) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Shenyang Pharmaceutical University Curated by ChEMBL | Assay Description Antagonist activity at human TRPV1 assessed as inhibition of NADA-induced effect at 1 uM by FLIPR assay | Bioorg Med Chem Lett 25: 803-6 (2015) Article DOI: 10.1016/j.bmcl.2014.12.086 BindingDB Entry DOI: 10.7270/Q2JD4ZG3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Homo sapiens (Human)) | BDBM50049553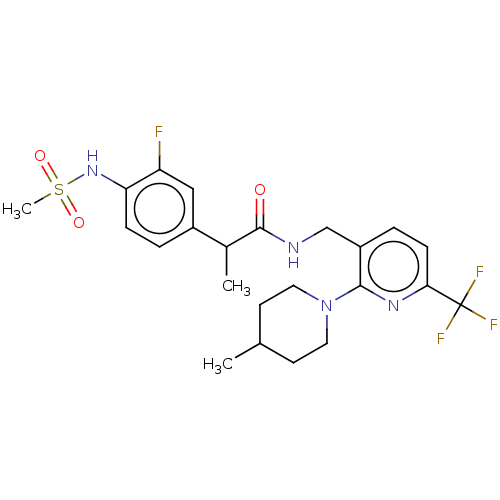 (CHEMBL2177428) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University Curated by ChEMBL | Assay Description Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of N-arachidonoyl dopamine-induced activation by FLIPR assay | Bioorg Med Chem Lett 25: 2326-30 (2015) Article DOI: 10.1016/j.bmcl.2015.04.024 BindingDB Entry DOI: 10.7270/Q2Z60QSC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor activity-modifying protein 1 (Homo sapiens (Human)) | BDBM391716 (N-[(2R)-3-(7-methyl-1H- | US10300056, Example 1 | ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Heptares Therapeutics Limited US Patent | Assay Description Radioligand binding assay. Human CGRP receptors expressed (consisting of CRLR and RAMP1) in insect Sf21 cell membrane homogenates were re-suspended i... | US Patent US10888561 (2021) BindingDB Entry DOI: 10.7270/Q28S4T1X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Homo sapiens (Human)) | BDBM50073160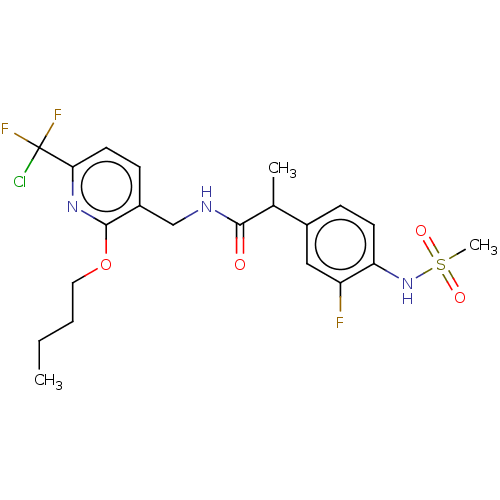 (CHEMBL3407762) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University Curated by ChEMBL | Assay Description Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of N-arachidonoyl dopamine-induced activity by FLIPR assay | Eur J Med Chem 93: 101-8 (2015) Article DOI: 10.1016/j.ejmech.2015.02.001 BindingDB Entry DOI: 10.7270/Q2N0188S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor activity-modifying protein 1 (Homo sapiens (Human)) | BDBM391716 (N-[(2R)-3-(7-methyl-1H- | US10300056, Example 1 | ...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research | Assay Description Human CGRP receptors expressed (consisting of CRLR and RAMP1) in insect Sf21 cell membrane homogenates were re-suspended in the binding buffer (10 mM... | Bioorg Med Chem Lett 18: 3641-5 (2008) BindingDB Entry DOI: 10.7270/Q2T155ZX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Transient receptor potential cation channel subfamily V member 1 (Homo sapiens (Human)) | BDBM50049553 (CHEMBL2177428) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Seoul National University Curated by ChEMBL | Assay Description Antagonist activity at human TRPV1 expressed in CHO cells assessed as inhibition of N-arachidonoyl dopamine-induced activity by FLIPR assay | Eur J Med Chem 93: 101-8 (2015) Article DOI: 10.1016/j.ejmech.2015.02.001 BindingDB Entry DOI: 10.7270/Q2N0188S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor activity-modifying protein 1 (Homo sapiens (Human)) | BDBM391727 (N-[(2R)-1-({(2S)-3-{1-[2- | US10300056, Example 12...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.0250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Heptares Therapeutics Limited US Patent | Assay Description Radioligand binding assay. Human CGRP receptors expressed (consisting of CRLR and RAMP1) in insect Sf21 cell membrane homogenates were re-suspended i... | US Patent US10888561 (2021) BindingDB Entry DOI: 10.7270/Q28S4T1X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Receptor activity-modifying protein 1 (Homo sapiens (Human)) | BDBM391727 (N-[(2R)-1-({(2S)-3-{1-[2- | US10300056, Example 12...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | 0.0250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research | Assay Description Human CGRP receptors expressed (consisting of CRLR and RAMP1) in insect Sf21 cell membrane homogenates were re-suspended in the binding buffer (10 mM... | Bioorg Med Chem Lett 18: 3641-5 (2008) BindingDB Entry DOI: 10.7270/Q2T155ZX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Calcitonin gene-related peptide type 1 receptor/Receptor activity-modifying protein 1 (Homo sapiens (Human)) | BDBM50539955 (CHEMBL4638938) | PDB KEGG UniProtKB/SwissProt antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 0.0251 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sosei Heptares Curated by ChEMBL | Assay Description Displacement of [3H]telcagepant from recombinant human CLR/RAMP1 expressed in Sf21 insect cell membranes measured after 60 mins by microbeta scintill... | J Med Chem 63: 7906-7920 (2020) Article DOI: 10.1021/acs.jmedchem.0c01003 BindingDB Entry DOI: 10.7270/Q2NK3JKR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50101511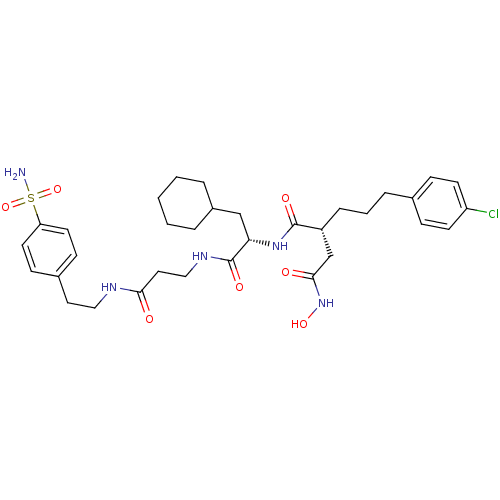 (2-[3-(4-Chloro-phenyl)-propyl]-N*1*-(2-cyclohexyl-...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Royal Danish School of Pharmacy Curated by ChEMBL | Assay Description Inhibitory constant against matrix metalloproteinase-2 | J Med Chem 44: 2333-43 (2001) BindingDB Entry DOI: 10.7270/Q2JH3KFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50101495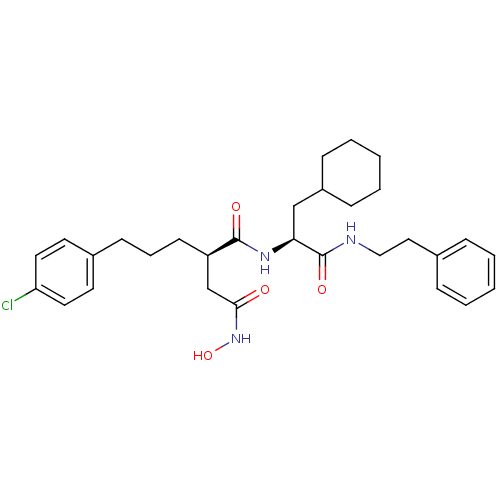 ((R)-2-[3-(4-Chloro-phenyl)-propyl]-N*1*-((S)-2-cyc...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Royal Danish School of Pharmacy Curated by ChEMBL | Assay Description Inhibitory constant against matrix metalloproteinase-2 | J Med Chem 44: 2333-43 (2001) BindingDB Entry DOI: 10.7270/Q2JH3KFR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 47184 total ) | Next | Last >> |