 Found 3202 hits with Last Name = 'sawa' and Initial = 'm'
Found 3202 hits with Last Name = 'sawa' and Initial = 'm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50325534
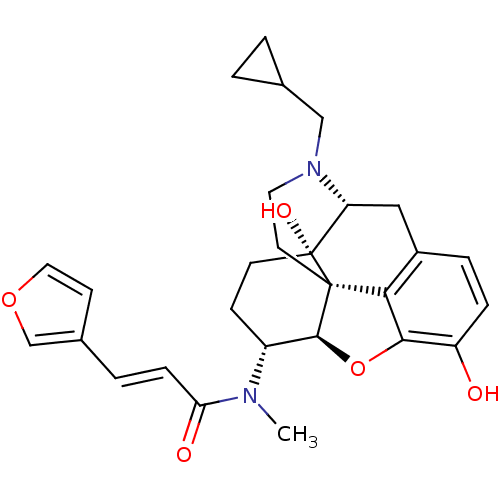
(CHEMBL267495 | nalfurafine)Show SMILES CN([C@@H]1CC[C@@]2(O)[C@H]3Cc4ccc(O)c5O[C@@H]1[C@]2(CCN3CC1CC1)c45)C(=O)\C=C\c1ccoc1 Show InChI InChI=1S/C28H32N2O5/c1-29(23(32)7-4-18-9-13-34-16-18)20-8-10-28(33)22-14-19-5-6-21(31)25-24(19)27(28,26(20)35-25)11-12-30(22)15-17-2-3-17/h4-7,9,13,16-17,20,22,26,31,33H,2-3,8,10-12,14-15H2,1H3/b7-4+/t20-,22-,26+,27+,28-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.0190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.128527
BindingDB Entry DOI: 10.7270/Q2HT2TDM |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50596292

(CHEMBL5185211)Show SMILES [H][C@]12Oc3c4c(C[C@]5([H])C(CC[C@H]1N(C)C(=O)\C=C\c1ccoc1)[C@]24CCN5CC1CC1)ccc3O |r,THB:28:27:9:4.5.6| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0272 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.128527
BindingDB Entry DOI: 10.7270/Q2HT2TDM |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50596293
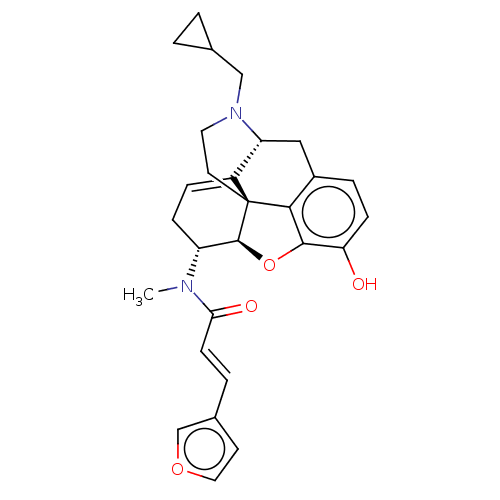
(CHEMBL5188658)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC6CC6)CC[C@@]14C5=CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)ccc3O |r,t:21,THB:10:9:17:4.5.6| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.0356 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.128527
BindingDB Entry DOI: 10.7270/Q2HT2TDM |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| MMDB
Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tokyo
Curated by ChEMBL
| Assay Description
Agonistic activity of the compound towards retinoic acid receptor-gamma |
J Med Chem 40: 4222-34 (1998)
Article DOI: 10.1021/jm9704309
BindingDB Entry DOI: 10.7270/Q21J98VC |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50109635
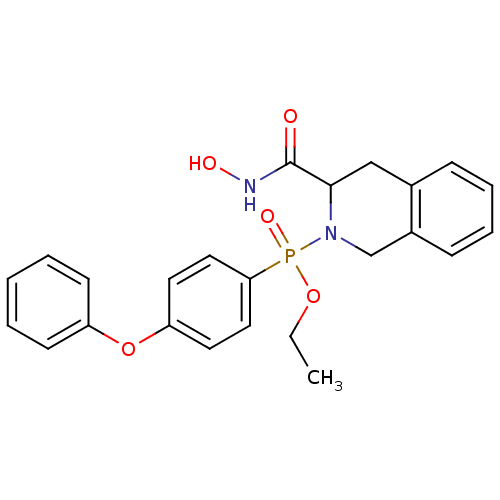
((3-Hydroxycarbamoyl-3,4-dihydro-1H-isoquinolin-2-y...)Show SMILES CCOP(=O)(N1Cc2ccccc2CC1C(=O)NO)c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C24H25N2O5P/c1-2-30-32(29,22-14-12-21(13-15-22)31-20-10-4-3-5-11-20)26-17-19-9-7-6-8-18(19)16-23(26)24(27)25-28/h3-15,23,28H,2,16-17H2,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-9 (MMP-9)(gelatinase-B). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50109621
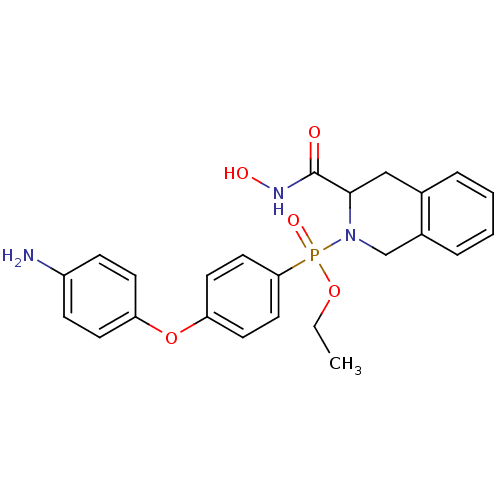
(CHEMBL425316 | [4-(4-Amino-phenoxy)-phenyl]-(3-hyd...)Show SMILES CCOP(=O)(N1Cc2ccccc2CC1C(=O)NO)c1ccc(Oc2ccc(N)cc2)cc1 Show InChI InChI=1S/C24H26N3O5P/c1-2-31-33(30,22-13-11-21(12-14-22)32-20-9-7-19(25)8-10-20)27-16-18-6-4-3-5-17(18)15-23(27)24(28)26-29/h3-14,23,29H,2,15-16,25H2,1H3,(H,26,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-9 (MMP-9)(gelatinase-B). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50596292

(CHEMBL5185211)Show SMILES [H][C@]12Oc3c4c(C[C@]5([H])C(CC[C@H]1N(C)C(=O)\C=C\c1ccoc1)[C@]24CCN5CC1CC1)ccc3O |r,THB:28:27:9:4.5.6| | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.158 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.128527
BindingDB Entry DOI: 10.7270/Q2HT2TDM |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Homo sapiens (Human)) | BDBM50274347
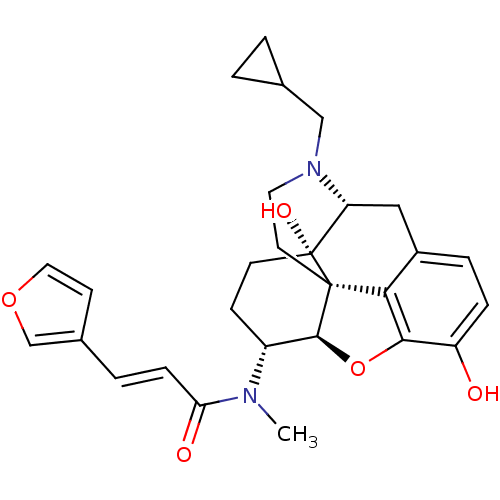
((2E)-N-[(5R,6R)-17-(cyclopropylmethyl)-4,5-epoxy-3...)Show SMILES CN([C@@H]1CC[C@@]2(O)[C@H]3Cc4ccc(O)c5O[C@@H]1[C@]2(CCN3CC1CC1)c45)C(=O)\C=C\c1ccoc1 |r,TLB:4:5:9.25.8:18.20.19,THB:6:5:9.25.8:18.20.19| Show InChI InChI=1S/C28H32N2O5/c1-29(23(32)7-4-18-9-13-34-16-18)20-8-10-28(33)22-14-19-5-6-21(31)25-24(19)27(28,26(20)35-25)11-12-30(22)15-17-2-3-17/h4-7,9,13,16-17,20,22,26,31,33H,2-3,8,10-12,14-15H2,1H3/b7-4+/t20-,22-,26+,27+,28-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.238 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Displacement of [3H]U69,593 from human kappa opioid receptor expressed in CHO cell membranes by micro-beta scintillation counting method |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50109626
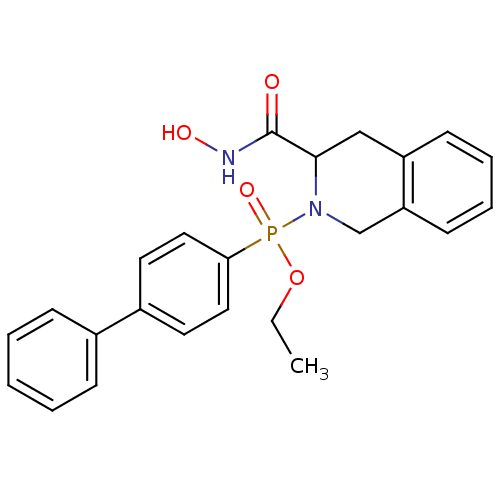
(Biphenyl-4-yl-(3-hydroxycarbamoyl-3,4-dihydro-1H-i...)Show SMILES CCOP(=O)(N1Cc2ccccc2CC1C(=O)NO)c1ccc(cc1)-c1ccccc1 Show InChI InChI=1S/C24H25N2O4P/c1-2-30-31(29,22-14-12-19(13-15-22)18-8-4-3-5-9-18)26-17-21-11-7-6-10-20(21)16-23(26)24(27)25-28/h3-15,23,28H,2,16-17H2,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-9 (MMP-9)(gelatinase-B). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50109621

(CHEMBL425316 | [4-(4-Amino-phenoxy)-phenyl]-(3-hyd...)Show SMILES CCOP(=O)(N1Cc2ccccc2CC1C(=O)NO)c1ccc(Oc2ccc(N)cc2)cc1 Show InChI InChI=1S/C24H26N3O5P/c1-2-31-33(30,22-13-11-21(12-14-22)32-20-9-7-19(25)8-10-20)27-16-18-6-4-3-5-17(18)15-23(27)24(28)26-29/h3-14,23,29H,2,15-16,25H2,1H3,(H,26,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-3 (MMP-3)(stromelysin-1). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50109635

((3-Hydroxycarbamoyl-3,4-dihydro-1H-isoquinolin-2-y...)Show SMILES CCOP(=O)(N1Cc2ccccc2CC1C(=O)NO)c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C24H25N2O5P/c1-2-30-32(29,22-14-12-21(13-15-22)31-20-10-4-3-5-11-20)26-17-19-9-7-6-8-18(19)16-23(26)24(27)25-28/h3-15,23,28H,2,16-17H2,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-3 (MMP-3)(stromelysin-1). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50109625
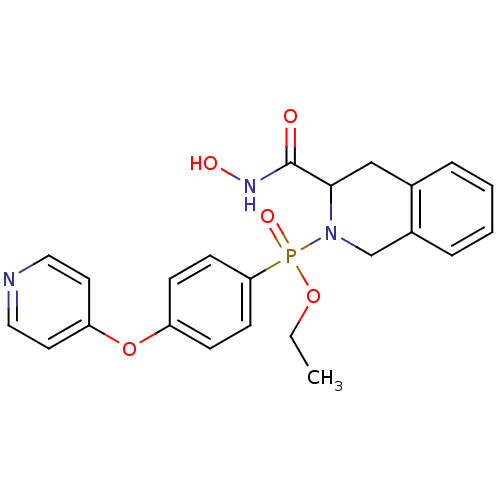
((3-Hydroxycarbamoyl-3,4-dihydro-1H-isoquinolin-2-y...)Show SMILES CCOP(=O)(N1Cc2ccccc2CC1C(=O)NO)c1ccc(Oc2ccncc2)cc1 Show InChI InChI=1S/C23H24N3O5P/c1-2-30-32(29,21-9-7-19(8-10-21)31-20-11-13-24-14-12-20)26-16-18-6-4-3-5-17(18)15-22(26)23(27)25-28/h3-14,22,28H,2,15-16H2,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-9 (MMP-9)(gelatinase-B). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Homo sapiens (Human)) | BDBM50325534

(CHEMBL267495 | nalfurafine)Show SMILES CN([C@@H]1CC[C@@]2(O)[C@H]3Cc4ccc(O)c5O[C@@H]1[C@]2(CCN3CC1CC1)c45)C(=O)\C=C\c1ccoc1 Show InChI InChI=1S/C28H32N2O5/c1-29(23(32)7-4-18-9-13-34-16-18)20-8-10-28(33)22-14-19-5-6-21(31)25-24(19)27(28,26(20)35-25)11-12-30(22)15-17-2-3-17/h4-7,9,13,16-17,20,22,26,31,33H,2-3,8,10-12,14-15H2,1H3/b7-4+/t20-,22-,26+,27+,28-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.128527
BindingDB Entry DOI: 10.7270/Q2HT2TDM |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50109629
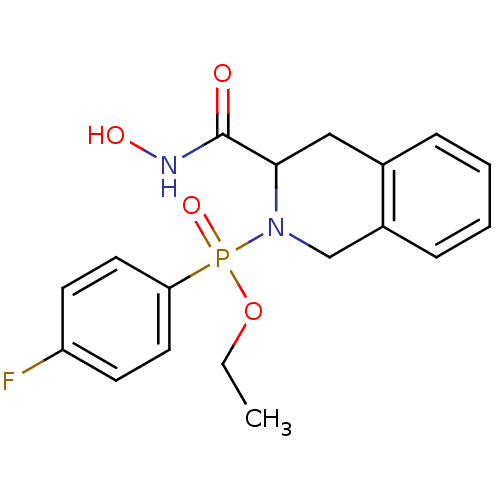
((4-Fluoro-phenyl)-(3-hydroxycarbamoyl-3,4-dihydro-...)Show InChI InChI=1S/C18H20FN2O4P/c1-2-25-26(24,16-9-7-15(19)8-10-16)21-12-14-6-4-3-5-13(14)11-17(21)18(22)20-23/h3-10,17,23H,2,11-12H2,1H3,(H,20,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.970 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-1 (MMP-1)(recombinant human collagenase-1). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM31892

(9-cis retinoic acid | 9C-RA | CHEMBL705 | Panretin...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C\C=C\C(\C)=C\C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8-,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tokyo
Curated by ChEMBL
| Assay Description
Agonistic activity of the compound towards retinoic acid receptor-gamma |
J Med Chem 40: 4222-34 (1998)
Article DOI: 10.1021/jm9704309
BindingDB Entry DOI: 10.7270/Q21J98VC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50156278
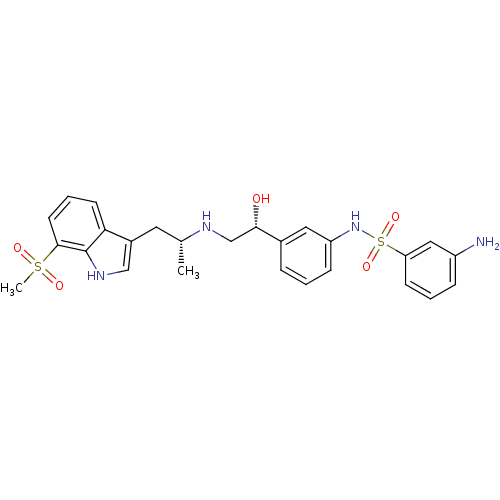
(3-Amino-N-(3-{(R)-1-hydroxy-2-[(R)-2-(7-methanesul...)Show SMILES C[C@H](Cc1c[nH]c2c(cccc12)S(C)(=O)=O)NC[C@H](O)c1cccc(NS(=O)(=O)c2cccc(N)c2)c1 Show InChI InChI=1S/C26H30N4O5S2/c1-17(12-19-15-29-26-23(19)10-5-11-25(26)36(2,32)33)28-16-24(31)18-6-3-8-21(13-18)30-37(34,35)22-9-4-7-20(27)14-22/h3-11,13-15,17,24,28-31H,12,16,27H2,1-2H3/t17-,24+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dainippon Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Binding inhibition constant was determined by inhibition of [125I]iodocyanopindolol binding to Beta-3 adrenergic receptor |
Bioorg Med Chem Lett 14: 5963-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.054
BindingDB Entry DOI: 10.7270/Q2BZ65HF |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50109633
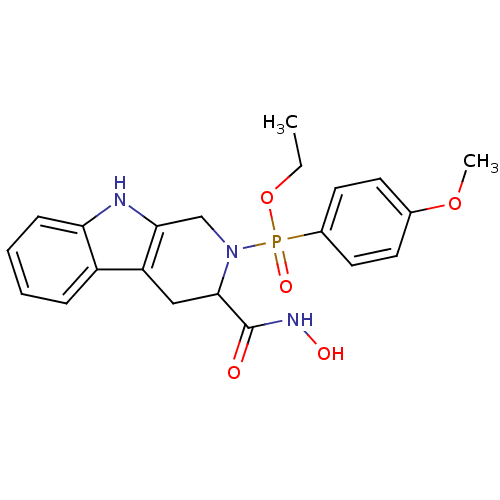
((3-Hydroxycarbamoyl-1,3,4,9-tetrahydro-beta-carbol...)Show SMILES CCOP(=O)(N1Cc2[nH]c3ccccc3c2CC1C(=O)NO)c1ccc(OC)cc1 Show InChI InChI=1S/C21H24N3O5P/c1-3-29-30(27,15-10-8-14(28-2)9-11-15)24-13-19-17(12-20(24)21(25)23-26)16-6-4-5-7-18(16)22-19/h4-11,20,22,26H,3,12-13H2,1-2H3,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 1.13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-9 (MMP-9)(gelatinase-B). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50127610
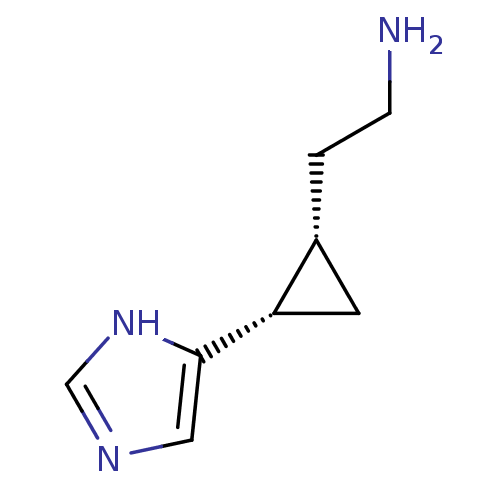
((1S,2S)-2-(2-aminoethyl)-1-(1H-imidazol-4-yl)cyclo...)Show InChI InChI=1S/C8H13N3/c9-2-1-6-3-7(6)8-4-10-5-11-8/h4-7H,1-3,9H2,(H,10,11)/t6-,7+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido University
Curated by ChEMBL
| Assay Description
Binding affinity to histamine H3 receptor |
Bioorg Med Chem 18: 1076-82 (2010)
Article DOI: 10.1016/j.bmc.2009.12.046
BindingDB Entry DOI: 10.7270/Q21V5F35 |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50230035
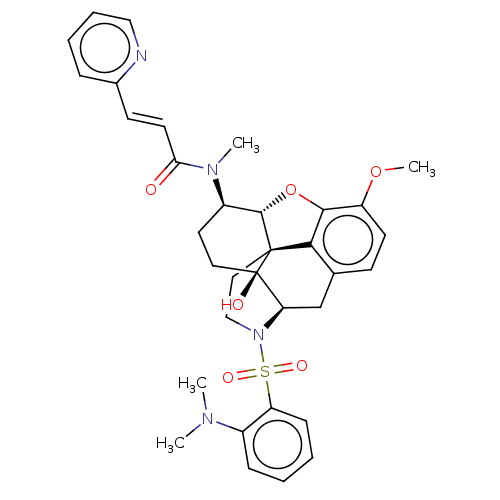
(CHEMBL4091544)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5(O)CC[C@H]2N(C)C(=O)\C=C\c1ccccn1)S(=O)(=O)c1ccccc1N(C)C)ccc3OC |r,THB:30:9:5.4.6:13| Show InChI InChI=1S/C34H38N4O6S/c1-36(2)24-10-5-6-11-27(24)45(41,42)38-20-18-33-30-22-12-14-26(43-4)31(30)44-32(33)25(16-17-34(33,40)28(38)21-22)37(3)29(39)15-13-23-9-7-8-19-35-23/h5-15,19,25,28,32,40H,16-18,20-21H2,1-4H3/b15-13+/t25-,28-,32+,33+,34-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular calcium level preincubated for 15... |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50230164
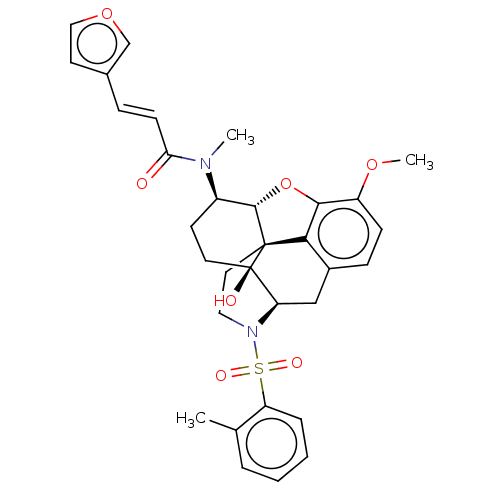
(CHEMBL4091834 | US10377763, Example 2)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5(O)CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1ccccc1C)ccc3OC |r,THB:29:9:5.4.6:13| Show InChI InChI=1S/C32H34N2O7S/c1-20-6-4-5-7-25(20)42(37,38)34-16-15-31-28-22-9-10-24(39-3)29(28)41-30(31)23(12-14-32(31,36)26(34)18-22)33(2)27(35)11-8-21-13-17-40-19-21/h4-11,13,17,19,23,26,30,36H,12,14-16,18H2,1-3H3/b11-8+/t23-,26-,30+,31+,32-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular calcium level preincubated for 15... |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50230139
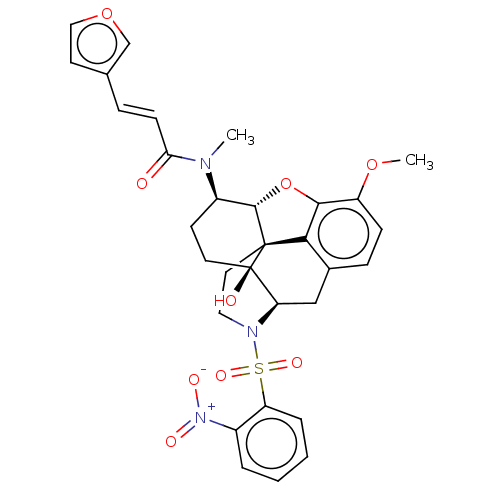
(CHEMBL4105072 | US10377763, Example 8)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5(O)CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1ccccc1[N+]([O-])=O)ccc3OC |r,THB:29:9:5.4.6:13| Show InChI InChI=1S/C31H31N3O9S/c1-32(26(35)10-7-19-12-16-42-18-19)22-11-13-31(36)25-17-20-8-9-23(41-2)28-27(20)30(31,29(22)43-28)14-15-33(25)44(39,40)24-6-4-3-5-21(24)34(37)38/h3-10,12,16,18,22,25,29,36H,11,13-15,17H2,1-2H3/b10-7+/t22-,25-,29+,30+,31-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular calcium level preincubated for 15... |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50109635

((3-Hydroxycarbamoyl-3,4-dihydro-1H-isoquinolin-2-y...)Show SMILES CCOP(=O)(N1Cc2ccccc2CC1C(=O)NO)c1ccc(Oc2ccccc2)cc1 Show InChI InChI=1S/C24H25N2O5P/c1-2-30-32(29,22-14-12-21(13-15-22)31-20-10-4-3-5-11-20)26-17-19-9-7-6-8-18(19)16-23(26)24(27)25-28/h3-15,23,28H,2,16-17H2,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.81 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-1 (MMP-1)(recombinant human collagenase-1). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50592812
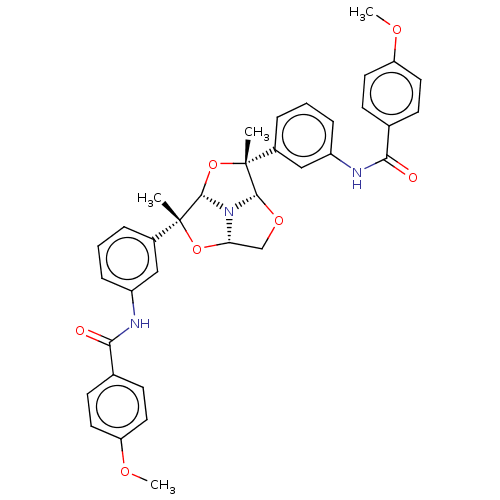
(CHEMBL5194980)Show SMILES [H][C@]12CO[C@]3([H])N1[C@@]([H])(O[C@]3(C)c1cccc(NC(=O)c3ccc(OC)cc3)c1)[C@](C)(O2)c1cccc(NC(=O)c2ccc(OC)cc2)c1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114505
BindingDB Entry DOI: 10.7270/Q2WM1JFQ |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50230161
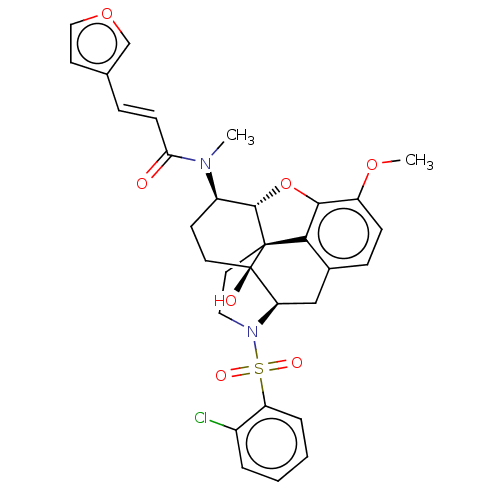
(CHEMBL4089496)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5(O)CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1ccccc1Cl)ccc3OC |r,THB:29:9:5.4.6:13| Show InChI InChI=1S/C31H31ClN2O7S/c1-33(26(35)10-7-19-12-16-40-18-19)22-11-13-31(36)25-17-20-8-9-23(39-2)28-27(20)30(31,29(22)41-28)14-15-34(25)42(37,38)24-6-4-3-5-21(24)32/h3-10,12,16,18,22,25,29,36H,11,13-15,17H2,1-2H3/b10-7+/t22-,25-,29+,30+,31-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular calcium level preincubated for 15... |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50230138
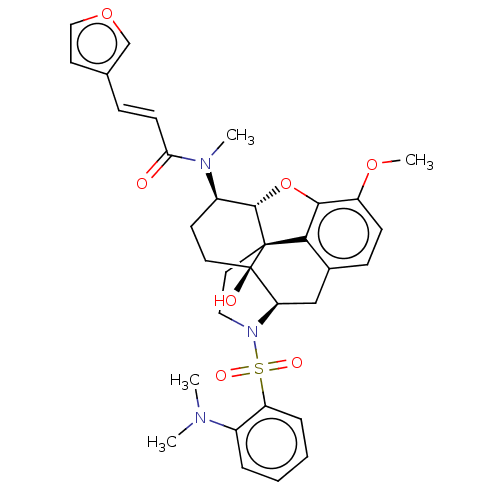
(CHEMBL4094318)Show SMILES Cl.[H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5(O)CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1ccccc1N(C)C)ccc3OC |r,THB:30:10:6.5.7:14| Show InChI InChI=1S/C33H37N3O7S.ClH/c1-34(2)23-7-5-6-8-26(23)44(39,40)36-17-16-32-29-22-10-11-25(41-4)30(29)43-31(32)24(13-15-33(32,38)27(36)19-22)35(3)28(37)12-9-21-14-18-42-20-21;/h5-12,14,18,20,24,27,31,38H,13,15-17,19H2,1-4H3;1H/b12-9+;/t24-,27-,31+,32+,33-;/m1./s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular calcium level preincubated for 15... |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50454546
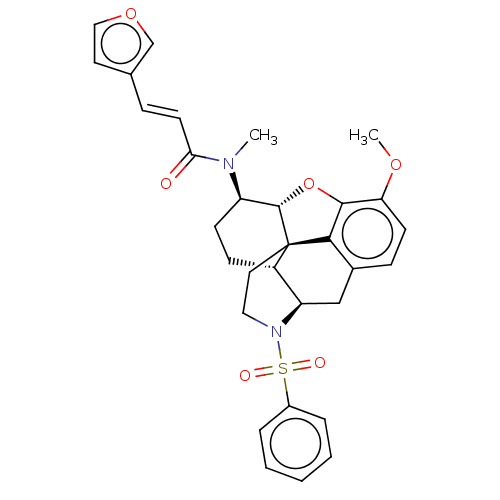
(CHEMBL4215362)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5([H])CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1ccccc1)ccc3OC |r,THB:29:9:13:6.4.5| Show InChI InChI=1S/C31H32N2O6S/c1-32(27(34)13-8-20-14-17-38-19-20)24-11-10-23-25-18-21-9-12-26(37-2)29-28(21)31(23,30(24)39-29)15-16-33(25)40(35,36)22-6-4-3-5-7-22/h3-9,12-14,17,19,23-25,30H,10-11,15-16,18H2,1-2H3/b13-8+/t23-,24+,25+,30-,31-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular Ca2+ release preincubated for 15 ... |
Bioorg Med Chem Lett 28: 774-777 (2018)
Article DOI: 10.1016/j.bmcl.2017.12.069
BindingDB Entry DOI: 10.7270/Q2BK1G0R |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50230158
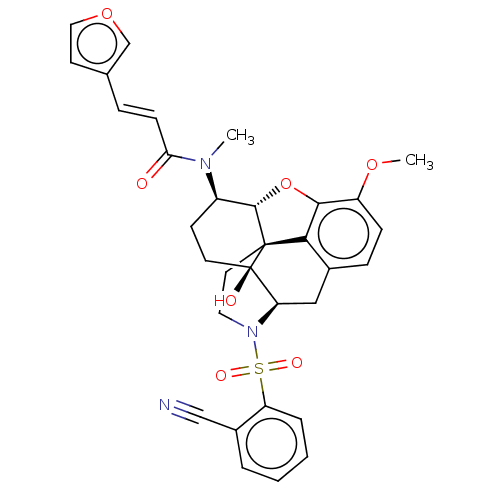
(CHEMBL4073947 | US10377763, Example 11)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5(O)CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1ccccc1C#N)ccc3OC |r,THB:29:9:5.4.6:13| Show InChI InChI=1S/C32H31N3O7S/c1-34(27(36)10-7-20-12-16-41-19-20)23-11-13-32(37)26-17-21-8-9-24(40-2)29-28(21)31(32,30(23)42-29)14-15-35(26)43(38,39)25-6-4-3-5-22(25)18-33/h3-10,12,16,19,23,26,30,37H,11,13-15,17H2,1-2H3/b10-7+/t23-,26-,30+,31+,32-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular calcium level preincubated for 15... |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50109633

((3-Hydroxycarbamoyl-1,3,4,9-tetrahydro-beta-carbol...)Show SMILES CCOP(=O)(N1Cc2[nH]c3ccccc3c2CC1C(=O)NO)c1ccc(OC)cc1 Show InChI InChI=1S/C21H24N3O5P/c1-3-29-30(27,15-10-8-14(28-2)9-11-15)24-13-19-17(12-20(24)21(25)23-26)16-6-4-5-7-18(16)22-19/h4-11,20,22,26H,3,12-13H2,1-2H3,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 2.02 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-1 (MMP-1)(recombinant human collagenase-1). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Matrix metalloproteinase-9
(Homo sapiens (Human)) | BDBM50109630
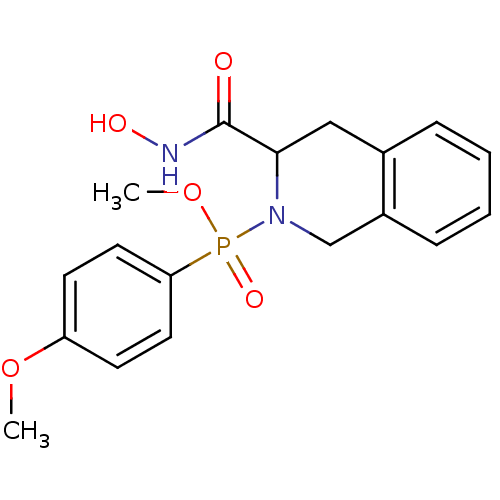
((3-Hydroxycarbamoyl-3,4-dihydro-1H-isoquinolin-2-y...)Show InChI InChI=1S/C18H21N2O5P/c1-24-15-7-9-16(10-8-15)26(23,25-2)20-12-14-6-4-3-5-13(14)11-17(20)18(21)19-22/h3-10,17,22H,11-12H2,1-2H3,(H,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.03 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-9 (MMP-9)(gelatinase-B). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50109633

((3-Hydroxycarbamoyl-1,3,4,9-tetrahydro-beta-carbol...)Show SMILES CCOP(=O)(N1Cc2[nH]c3ccccc3c2CC1C(=O)NO)c1ccc(OC)cc1 Show InChI InChI=1S/C21H24N3O5P/c1-3-29-30(27,15-10-8-14(28-2)9-11-15)24-13-19-17(12-20(24)21(25)23-26)16-6-4-5-7-18(16)22-19/h4-11,20,22,26H,3,12-13H2,1-2H3,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 2.04 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-3 (MMP-3)(stromelysin-1). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50230141
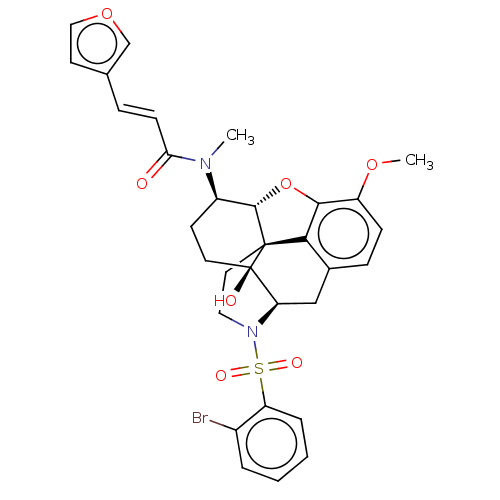
(CHEMBL4083587 | US10377763, Example 20)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5(O)CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1ccccc1Br)ccc3OC |r,THB:29:9:5.4.6:13| Show InChI InChI=1S/C31H31BrN2O7S/c1-33(26(35)10-7-19-12-16-40-18-19)22-11-13-31(36)25-17-20-8-9-23(39-2)28-27(20)30(31,29(22)41-28)14-15-34(25)42(37,38)24-6-4-3-5-21(24)32/h3-10,12,16,18,22,25,29,36H,11,13-15,17H2,1-2H3/b10-7+/t22-,25-,29+,30+,31-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular calcium level preincubated for 15... |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50230144
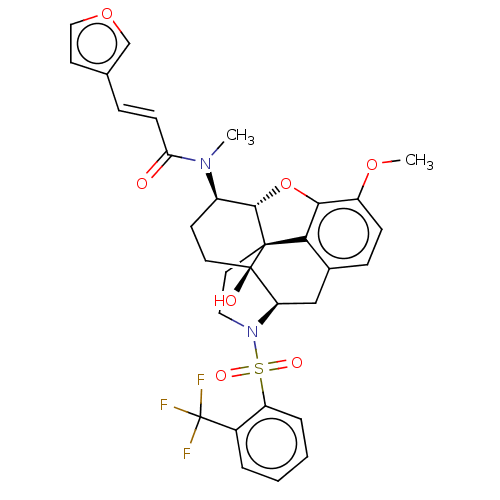
(CHEMBL4081763 | US10377763, Example 5)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5(O)CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1ccccc1C(F)(F)F)ccc3OC |r,THB:29:9:5.4.6:13| Show InChI InChI=1S/C32H31F3N2O7S/c1-36(26(38)10-7-19-12-16-43-18-19)22-11-13-31(39)25-17-20-8-9-23(42-2)28-27(20)30(31,29(22)44-28)14-15-37(25)45(40,41)24-6-4-3-5-21(24)32(33,34)35/h3-10,12,16,18,22,25,29,39H,11,13-15,17H2,1-2H3/b10-7+/t22-,25-,29+,30+,31-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular calcium level preincubated for 15... |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50230134
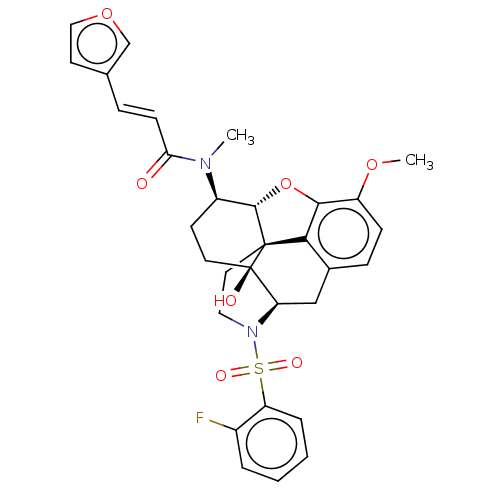
(CHEMBL4087046)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5(O)CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1ccccc1F)ccc3OC |r,THB:29:9:5.4.6:13| Show InChI InChI=1S/C31H31FN2O7S/c1-33(26(35)10-7-19-12-16-40-18-19)22-11-13-31(36)25-17-20-8-9-23(39-2)28-27(20)30(31,29(22)41-28)14-15-34(25)42(37,38)24-6-4-3-5-21(24)32/h3-10,12,16,18,22,25,29,36H,11,13-15,17H2,1-2H3/b10-7+/t22-,25-,29+,30+,31-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular calcium level preincubated for 15... |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50454547
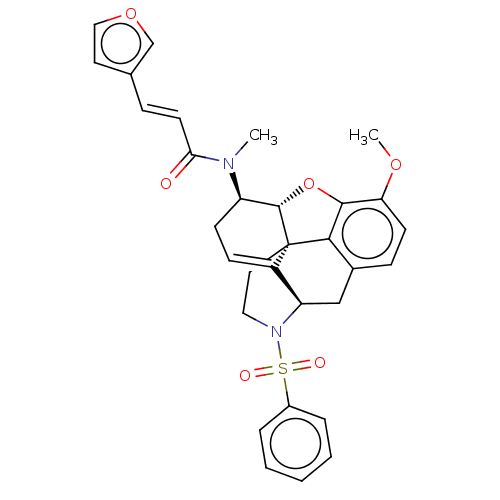
(CHEMBL4213974)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14C5=CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1ccccc1)ccc3OC |r,t:16,THB:28:9:13:6.4.5| Show InChI InChI=1S/C31H30N2O6S/c1-32(27(34)13-8-20-14-17-38-19-20)24-11-10-23-25-18-21-9-12-26(37-2)29-28(21)31(23,30(24)39-29)15-16-33(25)40(35,36)22-6-4-3-5-7-22/h3-10,12-14,17,19,24-25,30H,11,15-16,18H2,1-2H3/b13-8+/t24-,25-,30+,31+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular Ca2+ release preincubated for 15 ... |
Bioorg Med Chem Lett 28: 774-777 (2018)
Article DOI: 10.1016/j.bmcl.2017.12.069
BindingDB Entry DOI: 10.7270/Q2BK1G0R |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50592811
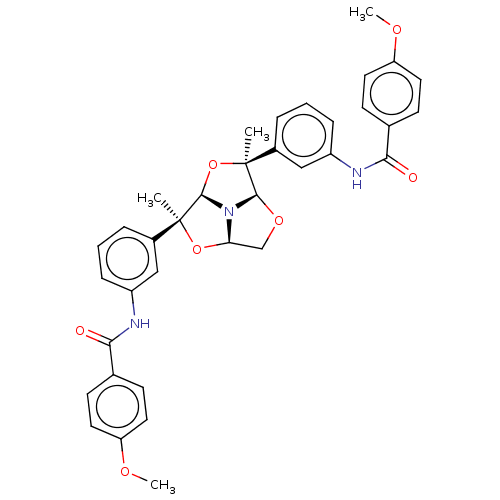
(CHEMBL5170308)Show SMILES [H][C@@]12CO[C@@]3([H])N1[C@]([H])(O[C@@]3(C)c1cccc(NC(=O)c3ccc(OC)cc3)c1)[C@@](C)(O2)c1cccc(NC(=O)c2ccc(OC)cc2)c1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114505
BindingDB Entry DOI: 10.7270/Q2WM1JFQ |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50291705
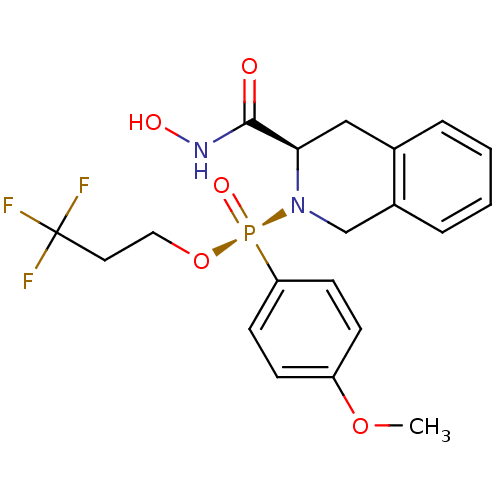
((S)-((R)-3-Hydroxycarbamoyl-3,4-dihydro-1H-isoquin...)Show SMILES COc1ccc(cc1)[P@@](=O)(OCCC(F)(F)F)N1Cc2ccccc2C[C@@H]1C(=O)NO Show InChI InChI=1S/C20H22F3N2O5P/c1-29-16-6-8-17(9-7-16)31(28,30-11-10-20(21,22)23)25-13-15-5-3-2-4-14(15)12-18(25)19(26)24-27/h2-9,18,27H,10-13H2,1H3,(H,24,26)/t18-,31-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibition of Matrix metalloprotease-1 (MMP-1) |
J Med Chem 45: 930-6 (2002)
BindingDB Entry DOI: 10.7270/Q2QV3KS7 |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50592810
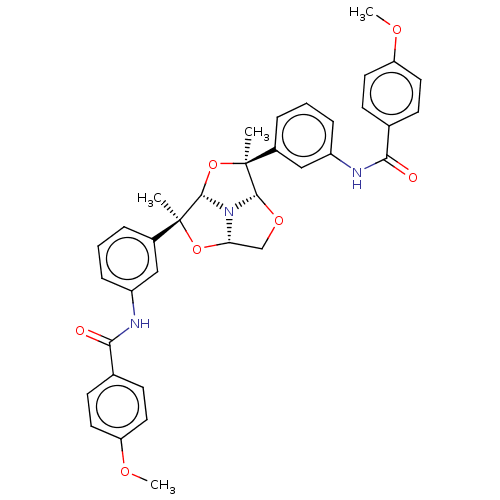
(CHEMBL5174113)Show SMILES [H][C@]12CO[C@]3([H])N1[C@@]([H])(O[C@@]3(C)c1cccc(NC(=O)c3ccc(OC)cc3)c1)[C@@](C)(O2)c1cccc(NC(=O)c2ccc(OC)cc2)c1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.128530
BindingDB Entry DOI: 10.7270/Q28919WZ |
More data for this
Ligand-Target Pair | |
Disintegrin and metalloproteinase domain-containing protein 17
(Homo sapiens (Human)) | BDBM50109633

((3-Hydroxycarbamoyl-1,3,4,9-tetrahydro-beta-carbol...)Show SMILES CCOP(=O)(N1Cc2[nH]c3ccccc3c2CC1C(=O)NO)c1ccc(OC)cc1 Show InChI InChI=1S/C21H24N3O5P/c1-3-29-30(27,15-10-8-14(28-2)9-11-15)24-13-19-17(12-20(24)21(25)23-26)16-6-4-5-7-18(16)22-19/h4-11,20,22,26H,3,12-13H2,1-2H3,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tumor necrosis factor alpha converting enzyme (TACE) from human acute monocytic leukemia cell line. |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50291705

((S)-((R)-3-Hydroxycarbamoyl-3,4-dihydro-1H-isoquin...)Show SMILES COc1ccc(cc1)[P@@](=O)(OCCC(F)(F)F)N1Cc2ccccc2C[C@@H]1C(=O)NO Show InChI InChI=1S/C20H22F3N2O5P/c1-29-16-6-8-17(9-7-16)31(28,30-11-10-20(21,22)23)25-13-15-5-3-2-4-14(15)12-18(25)19(26)24-27/h2-9,18,27H,10-13H2,1H3,(H,24,26)/t18-,31-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory constant against recombinant human Matrix metalloproteinase-1 |
Bioorg Med Chem Lett 12: 581-4 (2002)
BindingDB Entry DOI: 10.7270/Q2K64HCM |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50592809
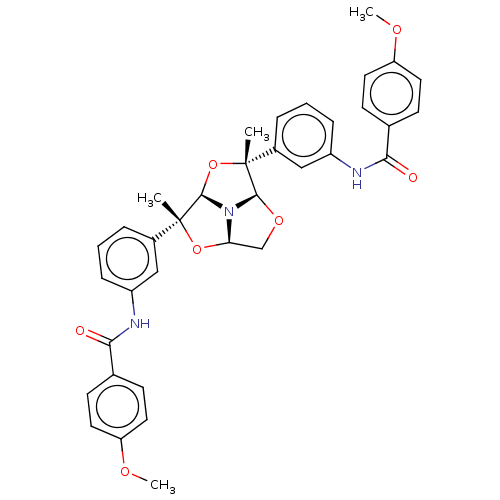
(CHEMBL5172765)Show SMILES [H][C@@]12CO[C@@]3([H])N1[C@]([H])(O[C@]3(C)c1cccc(NC(=O)c3ccc(OC)cc3)c1)[C@](C)(O2)c1cccc(NC(=O)c2ccc(OC)cc2)c1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50230167
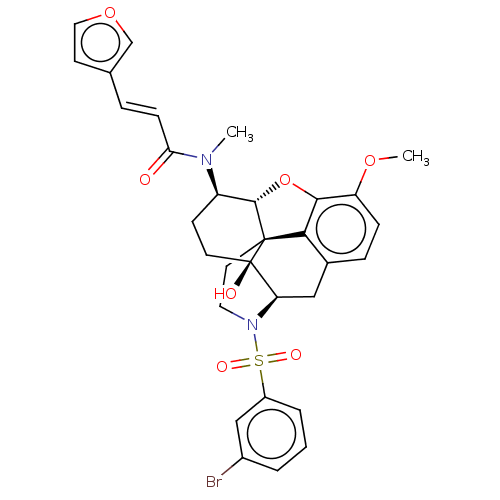
(CHEMBL4071220 | US10377763, Example 21)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5(O)CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1cccc(Br)c1)ccc3OC |r,THB:29:9:5.4.6:13| Show InChI InChI=1S/C31H31BrN2O7S/c1-33(26(35)9-6-19-11-15-40-18-19)23-10-12-31(36)25-16-20-7-8-24(39-2)28-27(20)30(31,29(23)41-28)13-14-34(25)42(37,38)22-5-3-4-21(32)17-22/h3-9,11,15,17-18,23,25,29,36H,10,12-14,16H2,1-2H3/b9-6+/t23-,25-,29+,30+,31-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular calcium level preincubated for 15... |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50454547

(CHEMBL4213974)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14C5=CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1ccccc1)ccc3OC |r,t:16,THB:28:9:13:6.4.5| Show InChI InChI=1S/C31H30N2O6S/c1-32(27(34)13-8-20-14-17-38-19-20)24-11-10-23-25-18-21-9-12-26(37-2)29-28(21)31(23,30(24)39-29)15-16-33(25)40(35,36)22-6-4-3-5-7-22/h3-10,12-14,17,19,24-25,30H,11,15-16,18H2,1-2H3/b13-8+/t24-,25-,30+,31+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmcl.2022.128550
BindingDB Entry DOI: 10.7270/Q2GT5S6B |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50230160
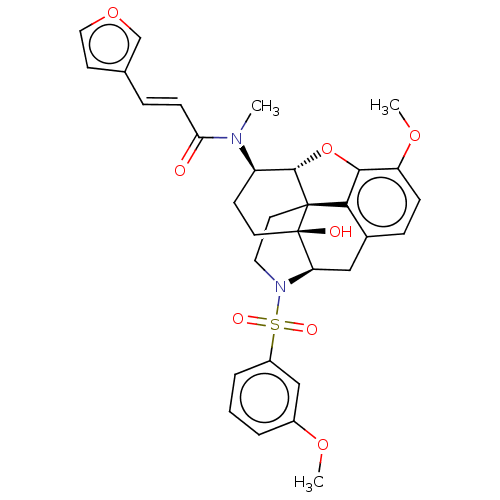
(CHEMBL4065120)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5(O)CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1cccc(OC)c1)ccc3OC |r,THB:29:9:5.4.6:13| Show InChI InChI=1S/C32H34N2O8S/c1-33(27(35)10-7-20-12-16-41-19-20)24-11-13-32(36)26-17-21-8-9-25(40-3)29-28(21)31(32,30(24)42-29)14-15-34(26)43(37,38)23-6-4-5-22(18-23)39-2/h4-10,12,16,18-19,24,26,30,36H,11,13-15,17H2,1-3H3/b10-7+/t24-,26-,30+,31+,32-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular calcium level preincubated for 15... |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50109622

((3-Hydroxycarbamoyl-3,4-dihydro-1H-isoquinolin-2-y...)Show SMILES COc1ccc(cc1)P(=O)(OCC1CCCCC1)N1Cc2ccccc2CC1C(=O)NO Show InChI InChI=1S/C24H31N2O5P/c1-30-21-11-13-22(14-12-21)32(29,31-17-18-7-3-2-4-8-18)26-16-20-10-6-5-9-19(20)15-23(26)24(27)25-28/h5-6,9-14,18,23,28H,2-4,7-8,15-17H2,1H3,(H,25,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.43 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-3 (MMP-3)(stromelysin-1). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Beta-3 adrenergic receptor
(Homo sapiens (Human)) | BDBM50156267
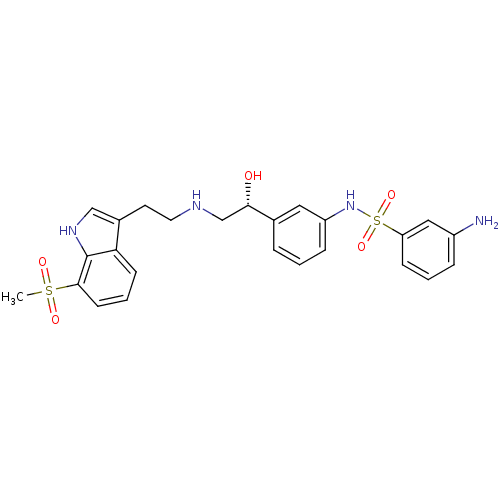
(3-Amino-N-(3-{(R)-1-hydroxy-2-[2-(7-methanesulfony...)Show SMILES CS(=O)(=O)c1cccc2c(CCNC[C@H](O)c3cccc(NS(=O)(=O)c4cccc(N)c4)c3)c[nH]c12 Show InChI InChI=1S/C25H28N4O5S2/c1-35(31,32)24-10-4-9-22-18(15-28-25(22)24)11-12-27-16-23(30)17-5-2-7-20(13-17)29-36(33,34)21-8-3-6-19(26)14-21/h2-10,13-15,23,27-30H,11-12,16,26H2,1H3/t23-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Dainippon Pharmaceutical Co., Ltd
Curated by ChEMBL
| Assay Description
Binding inhibition constant was determined by inhibition of [125I]iodocyanopindolol binding to Beta-3 adrenergic receptor |
Bioorg Med Chem Lett 14: 5963-6 (2004)
Article DOI: 10.1016/j.bmcl.2004.09.054
BindingDB Entry DOI: 10.7270/Q2BZ65HF |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50592795
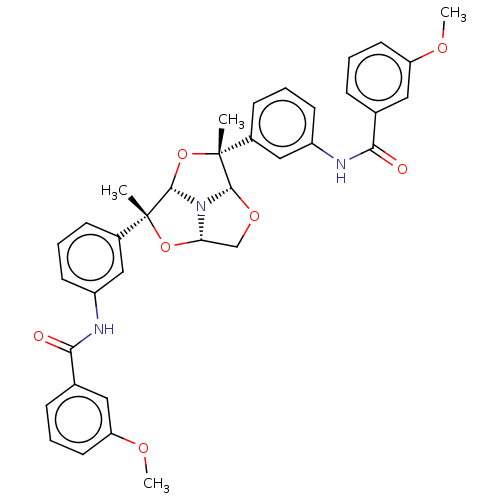
(CHEMBL5207082)Show SMILES [H][C@]12CO[C@]3([H])N1[C@@]([H])(O[C@]3(C)c1cccc(NC(=O)c3cccc(OC)c3)c1)[C@](C)(O2)c1cccc(NC(=O)c2cccc(OC)c2)c1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114505
BindingDB Entry DOI: 10.7270/Q2WM1JFQ |
More data for this
Ligand-Target Pair | |
Stromelysin-1
(Homo sapiens (Human)) | BDBM50109630

((3-Hydroxycarbamoyl-3,4-dihydro-1H-isoquinolin-2-y...)Show InChI InChI=1S/C18H21N2O5P/c1-24-15-7-9-16(10-8-15)26(23,25-2)20-12-14-6-4-3-5-13(14)11-17(20)18(21)19-22/h3-10,17,22H,11-12H2,1-2H3,(H,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.95 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-3 (MMP-3)(stromelysin-1). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Disintegrin and metalloproteinase domain-containing protein 17
(Homo sapiens (Human)) | BDBM50109614

(CHEMBL353536 | [4-(2-Ethoxy-ethoxy)-phenyl]-(3-hyd...)Show SMILES CCOCCOc1ccc(cc1)P(=O)(OCC)N1Cc2ccccc2CC1C(=O)NO Show InChI InChI=1S/C22H29N2O6P/c1-3-28-13-14-29-19-9-11-20(12-10-19)31(27,30-4-2)24-16-18-8-6-5-7-17(18)15-21(24)22(25)23-26/h5-12,21,26H,3-4,13-16H2,1-2H3,(H,23,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.98 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tumor necrosis factor alpha converting enzyme (TACE) from human acute monocytic leukemia cell line. |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |
Orexin/Hypocretin receptor type 1
(Homo sapiens (Human)) | BDBM50230156
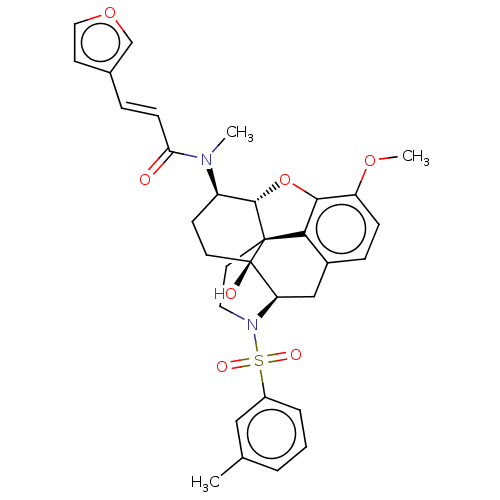
(CHEMBL4097697 | US10377763, Example 3)Show SMILES [H][C@@]12Oc3c4c(C[C@@]5([H])N(CC[C@@]14[C@@]5(O)CC[C@H]2N(C)C(=O)\C=C\c1ccoc1)S(=O)(=O)c1cccc(C)c1)ccc3OC |r,THB:29:9:5.4.6:13| Show InChI InChI=1S/C32H34N2O7S/c1-20-5-4-6-23(17-20)42(37,38)34-15-14-31-28-22-8-9-25(39-3)29(28)41-30(31)24(11-13-32(31,36)26(34)18-22)33(2)27(35)10-7-21-12-16-40-19-21/h4-10,12,16-17,19,24,26,30,36H,11,13-15,18H2,1-3H3/b10-7+/t24-,26-,30+,31+,32-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Tsukuba
Curated by ChEMBL
| Assay Description
Antagonist activity at human OX1R expressed in CHOK1 cells assessed as inhibition of orexin A-induced intracellular calcium level preincubated for 15... |
J Med Chem 60: 1018-1040 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01418
BindingDB Entry DOI: 10.7270/Q22N54JH |
More data for this
Ligand-Target Pair | |
Interstitial collagenase
(Homo sapiens (Human)) | BDBM50109631
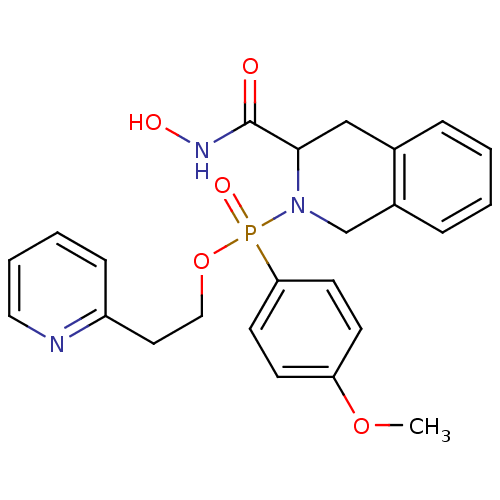
((3-Hydroxycarbamoyl-3,4-dihydro-1H-isoquinolin-2-y...)Show SMILES COc1ccc(cc1)P(=O)(OCCc1ccccn1)N1Cc2ccccc2CC1C(=O)NO Show InChI InChI=1S/C24H26N3O5P/c1-31-21-9-11-22(12-10-21)33(30,32-15-13-20-8-4-5-14-25-20)27-17-19-7-3-2-6-18(19)16-23(27)24(28)26-29/h2-12,14,23,29H,13,15-17H2,1H3,(H,26,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nippon Organon K.K.
Curated by ChEMBL
| Assay Description
Inhibitory activity against matrix metalloprotease-1 (MMP-1)(recombinant human collagenase-1). |
J Med Chem 45: 919-29 (2002)
BindingDB Entry DOI: 10.7270/Q2XK8G9Q |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data