 Found 115 hits with Last Name = 'scott' and Initial = 'mp'
Found 115 hits with Last Name = 'scott' and Initial = 'mp' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM92649
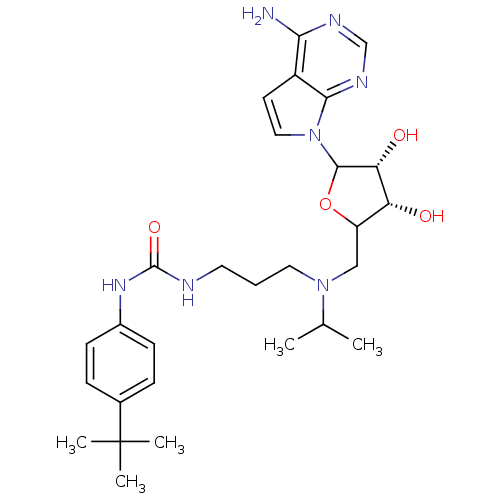
(EPZ004777)Show SMILES CC(C)N(CCCNC(=O)Nc1ccc(cc1)C(C)(C)C)CC1OC([C@H](O)[C@@H]1O)n1ccc2c(N)ncnc12 |r| Show InChI InChI=1S/C28H41N7O4/c1-17(2)34(13-6-12-30-27(38)33-19-9-7-18(8-10-19)28(3,4)5)15-21-22(36)23(37)26(39-21)35-14-11-20-24(29)31-16-32-25(20)35/h7-11,14,16-17,21-23,26,36-37H,6,12-13,15H2,1-5H3,(H2,29,31,32)(H2,30,33,38)/t21?,22-,23-,26?/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
| Assay Description
Assay of DOT1L enzymatic activity were performed under balanced conditions using a radiometric assay. |
Chem Biol Drug Des 80: 971-80 (2012)
Article DOI: 10.1111/cbdd.12050
BindingDB Entry DOI: 10.7270/Q2Z89B12 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378462
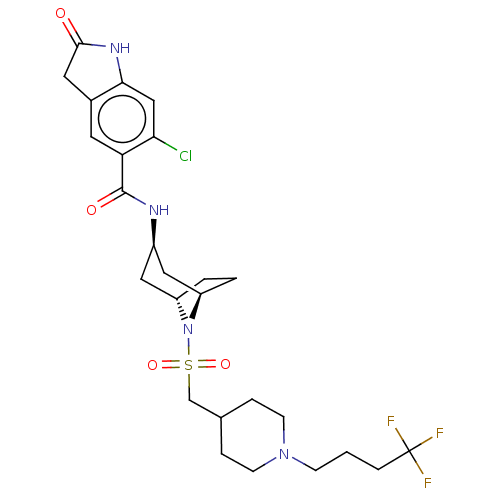
(6-chloro-2-oxo-N-((1R,3r,5S)-8-(((1-(4,4,4-trifluo...)Show SMILES FC(F)(F)CCCN1CCC(CS(=O)(=O)N2[C@H]3CC[C@@H]2C[C@@H](C3)NC(=O)c2cc3CC(=O)Nc3cc2Cl)CC1 |r,TLB:12:15:18.17:22.21.20| Show InChI InChI=1S/C26H34ClF3N4O4S/c27-22-14-23-17(11-24(35)32-23)10-21(22)25(36)31-18-12-19-2-3-20(13-18)34(19)39(37,38)15-16-4-8-33(9-5-16)7-1-6-26(28,29)30/h10,14,16,18-20H,1-9,11-13,15H2,(H,31,36)(H,32,35)/t18-,19+,20- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Non-competitive inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using varyin... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378462

(6-chloro-2-oxo-N-((1R,3r,5S)-8-(((1-(4,4,4-trifluo...)Show SMILES FC(F)(F)CCCN1CCC(CS(=O)(=O)N2[C@H]3CC[C@@H]2C[C@@H](C3)NC(=O)c2cc3CC(=O)Nc3cc2Cl)CC1 |r,TLB:12:15:18.17:22.21.20| Show InChI InChI=1S/C26H34ClF3N4O4S/c27-22-14-23-17(11-24(35)32-23)10-21(22)25(36)31-18-12-19-2-3-20(13-18)34(19)39(37,38)15-16-4-8-33(9-5-16)7-1-6-26(28,29)30/h10,14,16,18-20H,1-9,11-13,15H2,(H,31,36)(H,32,35)/t18-,19+,20- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Non-competitive inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using fixed ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378459

(N-((1R,3r,5S)-8-((4-(benzylamino)piperidin-1-yl)su...)Show SMILES Clc1cc2NC(=O)Cc2cc1C(=O)N[C@@H]1C[C@@H]2CC[C@H](C1)N2S(=O)(=O)N1CCC(CC1)NCc1ccccc1 |r,TLB:22:21:15.14.20:18.17| Show InChI InChI=1S/C28H34ClN5O4S/c29-25-16-26-19(13-27(35)32-26)12-24(25)28(36)31-21-14-22-6-7-23(15-21)34(22)39(37,38)33-10-8-20(9-11-33)30-17-18-4-2-1-3-5-18/h1-5,12,16,20-23,30H,6-11,13-15,17H2,(H,31,36)(H,32,35)/t21-,22+,23- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Non-competitive inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using varyin... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM92648
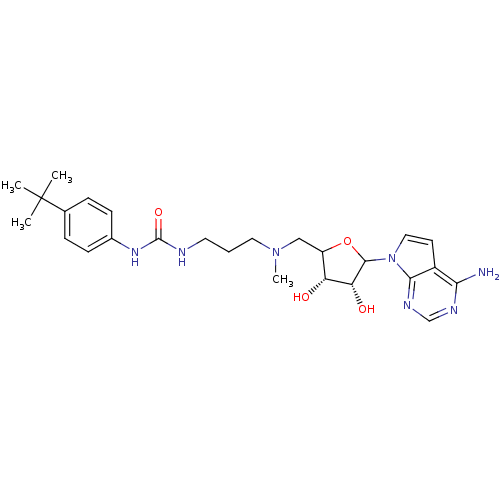
(EPZ004450)Show SMILES CN(CCCNC(=O)Nc1ccc(cc1)C(C)(C)C)CC1OC([C@H](O)[C@@H]1O)n1ccc2c(N)ncnc12 |r| Show InChI InChI=1S/C26H37N7O4/c1-26(2,3)16-6-8-17(9-7-16)31-25(36)28-11-5-12-32(4)14-19-20(34)21(35)24(37-19)33-13-10-18-22(27)29-15-30-23(18)33/h6-10,13,15,19-21,24,34-35H,5,11-12,14H2,1-4H3,(H2,27,29,30)(H2,28,31,36)/t19?,20-,21-,24?/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
| Assay Description
Assay of DOT1L enzymatic activity were performed under balanced conditions using a radiometric assay. |
Chem Biol Drug Des 80: 971-80 (2012)
Article DOI: 10.1111/cbdd.12050
BindingDB Entry DOI: 10.7270/Q2Z89B12 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378459

(N-((1R,3r,5S)-8-((4-(benzylamino)piperidin-1-yl)su...)Show SMILES Clc1cc2NC(=O)Cc2cc1C(=O)N[C@@H]1C[C@@H]2CC[C@H](C1)N2S(=O)(=O)N1CCC(CC1)NCc1ccccc1 |r,TLB:22:21:15.14.20:18.17| Show InChI InChI=1S/C28H34ClN5O4S/c29-25-16-26-19(13-27(35)32-26)12-24(25)28(36)31-21-14-22-6-7-23(15-21)34(22)39(37,38)33-10-8-20(9-11-33)30-17-18-4-2-1-3-5-18/h1-5,12,16,20-23,30H,6-11,13-15,17H2,(H,31,36)(H,32,35)/t21-,22+,23- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 4.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Mixed type inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using fixed N-ter... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM92647
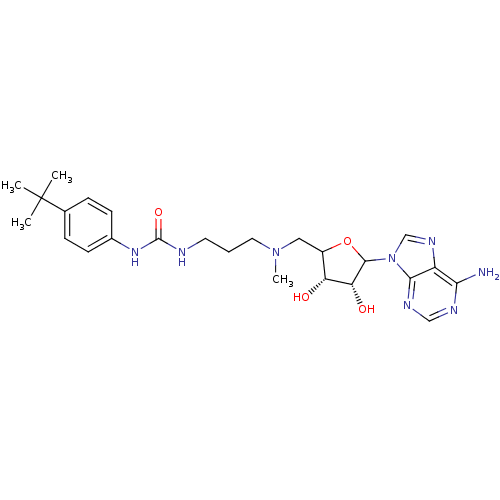
(EPZ003696)Show SMILES CN(CCCNC(=O)Nc1ccc(cc1)C(C)(C)C)CC1OC([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C25H36N8O4/c1-25(2,3)15-6-8-16(9-7-15)31-24(36)27-10-5-11-32(4)12-17-19(34)20(35)23(37-17)33-14-30-18-21(26)28-13-29-22(18)33/h6-9,13-14,17,19-20,23,34-35H,5,10-12H2,1-4H3,(H2,26,28,29)(H2,27,31,36)/t17?,19-,20-,23?/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
| Assay Description
Assay of DOT1L enzymatic activity were performed under balanced conditions using a radiometric assay. |
Chem Biol Drug Des 80: 971-80 (2012)
Article DOI: 10.1111/cbdd.12050
BindingDB Entry DOI: 10.7270/Q2Z89B12 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM92642
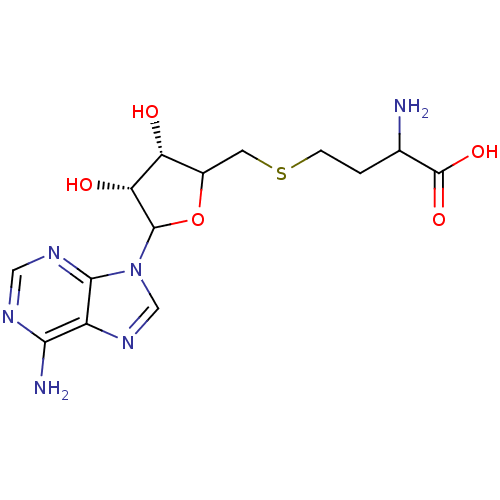
(SAH)Show SMILES NC(CCSCC1OC([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12)C(O)=O |r| Show InChI InChI=1S/C14H20N6O5S/c15-6(14(23)24)1-2-26-3-7-9(21)10(22)13(25-7)20-5-19-8-11(16)17-4-18-12(8)20/h4-7,9-10,13,21-22H,1-3,15H2,(H,23,24)(H2,16,17,18)/t6?,7?,9-,10-,13?/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 320 | n/a | n/a | 71 | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
| Assay Description
Assay of DOT1L enzymatic activity were performed under balanced conditions using a radiometric assay. |
Chem Biol Drug Des 80: 971-80 (2012)
Article DOI: 10.1111/cbdd.12050
BindingDB Entry DOI: 10.7270/Q2Z89B12 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM92646
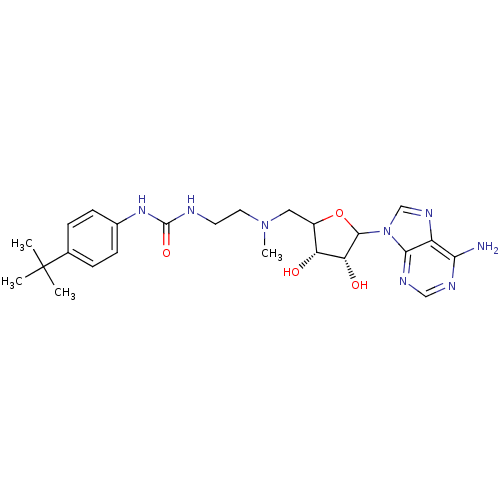
(EPZ003647)Show SMILES CN(CCNC(=O)Nc1ccc(cc1)C(C)(C)C)CC1OC([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C24H34N8O4/c1-24(2,3)14-5-7-15(8-6-14)30-23(35)26-9-10-31(4)11-16-18(33)19(34)22(36-16)32-13-29-17-20(25)27-12-28-21(17)32/h5-8,12-13,16,18-19,22,33-34H,9-11H2,1-4H3,(H2,25,27,28)(H2,26,30,35)/t16?,18-,19-,22?/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 845 | n/a | n/a | 167 | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
| Assay Description
Assay of DOT1L enzymatic activity were performed under balanced conditions using a radiometric assay. |
Chem Biol Drug Des 80: 971-80 (2012)
Article DOI: 10.1111/cbdd.12050
BindingDB Entry DOI: 10.7270/Q2Z89B12 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM92644
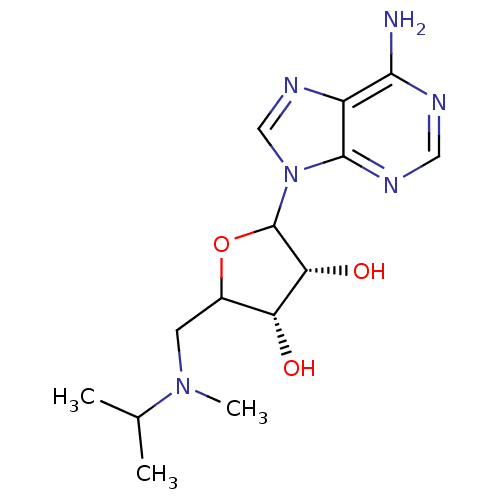
(EPZ002446)Show SMILES CC(C)N(C)CC1OC([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C14H22N6O3/c1-7(2)19(3)4-8-10(21)11(22)14(23-8)20-6-18-9-12(15)16-5-17-13(9)20/h5-8,10-11,14,21-22H,4H2,1-3H3,(H2,15,16,17)/t8?,10-,11-,14?/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
| Assay Description
Assay of DOT1L enzymatic activity were performed under balanced conditions using a radiometric assay. |
Chem Biol Drug Des 80: 971-80 (2012)
Article DOI: 10.1111/cbdd.12050
BindingDB Entry DOI: 10.7270/Q2Z89B12 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM92645
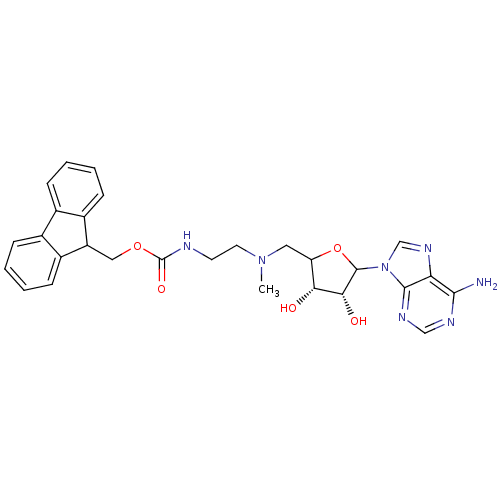
(EPZ003144)Show SMILES CN(CCNC(=O)OCC1c2ccccc2-c2ccccc12)CC1OC([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C28H31N7O5/c1-34(12-21-23(36)24(37)27(40-21)35-15-33-22-25(29)31-14-32-26(22)35)11-10-30-28(38)39-13-20-18-8-4-2-6-16(18)17-7-3-5-9-19(17)20/h2-9,14-15,20-21,23-24,27,36-37H,10-13H2,1H3,(H,30,38)(H2,29,31,32)/t21?,23-,24-,27?/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
| Assay Description
Assay of DOT1L enzymatic activity were performed under balanced conditions using a radiometric assay. |
Chem Biol Drug Des 80: 971-80 (2012)
Article DOI: 10.1111/cbdd.12050
BindingDB Entry DOI: 10.7270/Q2Z89B12 |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase, H3 lysine-79 specific
(Homo sapiens (Human)) | BDBM92643
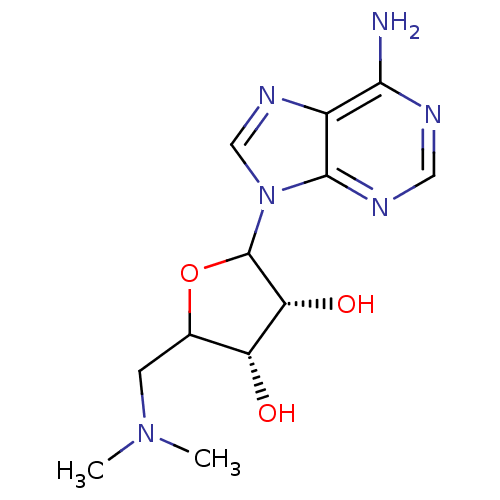
(EPZ000004)Show SMILES CN(C)CC1OC([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12 |r| Show InChI InChI=1S/C12H18N6O3/c1-17(2)3-6-8(19)9(20)12(21-6)18-5-16-7-10(13)14-4-15-11(7)18/h4-6,8-9,12,19-20H,3H2,1-2H3,(H2,13,14,15)/t6?,8-,9-,12?/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
| Assay Description
Assay of DOT1L enzymatic activity were performed under balanced conditions using a radiometric assay. |
Chem Biol Drug Des 80: 971-80 (2012)
Article DOI: 10.1111/cbdd.12050
BindingDB Entry DOI: 10.7270/Q2Z89B12 |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50362985

(CHEMBL1945502)Show SMILES COc1ccc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)cc1CCCN(C)C Show InChI InChI=1S/C24H26ClN5OS/c1-30(2)10-4-5-15-11-18(7-9-21(15)31-3)27-24-26-14-16-12-22(32)28-20-13-17(25)6-8-19(20)23(16)29-24/h6-9,11,13-14H,4-5,10,12H2,1-3H3,(H,28,32)(H,26,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of KDR |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378446
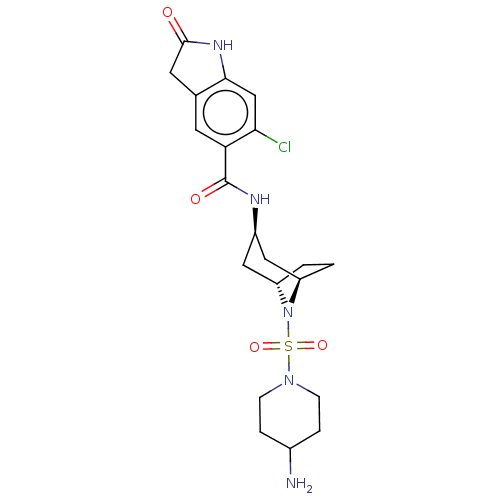
(N-((1R,3r,5S)-8-((4-aminopiperidin-1-yl)sulfonyl)-...)Show SMILES NC1CCN(CC1)S(=O)(=O)N1[C@H]2CC[C@@H]1C[C@@H](C2)NC(=O)c1cc2CC(=O)Nc2cc1Cl |r,TLB:7:10:13.12:17.16.15| Show InChI InChI=1S/C21H28ClN5O4S/c22-18-11-19-12(8-20(28)25-19)7-17(18)21(29)24-14-9-15-1-2-16(10-14)27(15)32(30,31)26-5-3-13(23)4-6-26/h7,11,13-16H,1-6,8-10,23H2,(H,24,29)(H,25,28)/t14-,15+,16- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378445

(N-((1R,3r,5S)-8-((4-aminopiperidin-1-yl)sulfonyl)-...)Show SMILES NC1CCN(CC1)S(=O)(=O)N1[C@H]2CC[C@@H]1C[C@@H](C2)NC(=O)c1ccc2NC(=O)Cc2c1 |r,TLB:7:10:13.12:17.16.15| Show InChI InChI=1S/C21H29N5O4S/c22-15-5-7-25(8-6-15)31(29,30)26-17-2-3-18(26)12-16(11-17)23-21(28)13-1-4-19-14(9-13)10-20(27)24-19/h1,4,9,15-18H,2-3,5-8,10-12,22H2,(H,23,28)(H,24,27)/t16-,17+,18- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378444
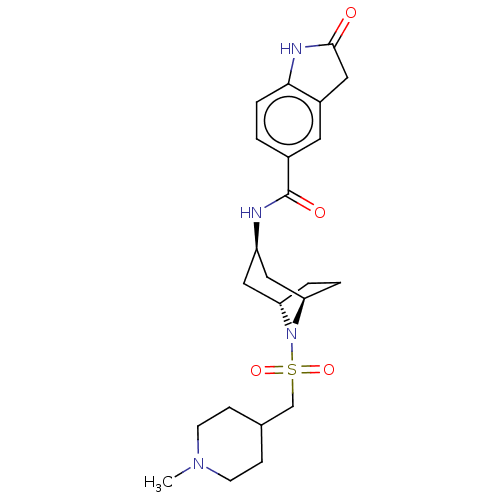
(N-((1R,3r,5S)-8-(((1-methylpiperidin-4-yl)methyl)s...)Show SMILES CN1CCC(CS(=O)(=O)N2[C@H]3CC[C@@H]2C[C@@H](C3)NC(=O)c2ccc3NC(=O)Cc3c2)CC1 |r,TLB:6:9:12.11:16.15.14| Show InChI InChI=1S/C23H32N4O4S/c1-26-8-6-15(7-9-26)14-32(30,31)27-19-3-4-20(27)13-18(12-19)24-23(29)16-2-5-21-17(10-16)11-22(28)25-21/h2,5,10,15,18-20H,3-4,6-9,11-14H2,1H3,(H,24,29)(H,25,28)/t18-,19+,20- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378453

(6-chloro-N-((1R,3r,5S)-8-((4-(methylamino)piperidi...)Show SMILES CNC1CCN(CC1)S(=O)(=O)N1[C@H]2CC[C@@H]1C[C@@H](C2)NC(=O)c1cc2CC(=O)Nc2cc1Cl |r,TLB:8:11:14.13:18.17.16| Show InChI InChI=1S/C22H30ClN5O4S/c1-24-14-4-6-27(7-5-14)33(31,32)28-16-2-3-17(28)11-15(10-16)25-22(30)18-8-13-9-21(29)26-20(13)12-19(18)23/h8,12,14-17,24H,2-7,9-11H2,1H3,(H,25,30)(H,26,29)/t15-,16+,17- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378452
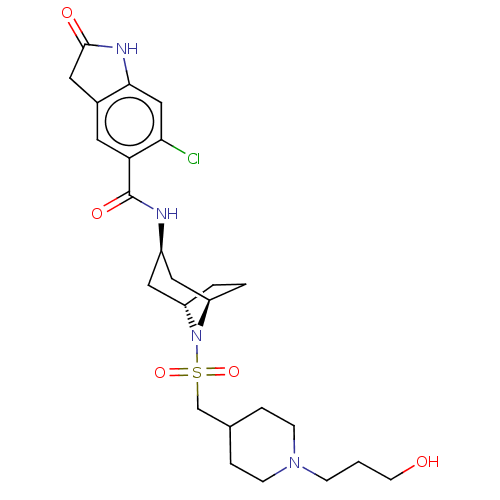
(6-chloro-N-((1R,3r,5S)-8-(((1-(3-hydroxypropyl)pip...)Show SMILES OCCCN1CCC(CS(=O)(=O)N2[C@H]3CC[C@@H]2C[C@@H](C3)NC(=O)c2cc3CC(=O)Nc3cc2Cl)CC1 |r,TLB:9:12:15.14:19.18.17| Show InChI InChI=1S/C25H35ClN4O5S/c26-22-14-23-17(11-24(32)28-23)10-21(22)25(33)27-18-12-19-2-3-20(13-18)30(19)36(34,35)15-16-4-7-29(8-5-16)6-1-9-31/h10,14,16,18-20,31H,1-9,11-13,15H2,(H,27,33)(H,28,32)/t18-,19+,20- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378462

(6-chloro-2-oxo-N-((1R,3r,5S)-8-(((1-(4,4,4-trifluo...)Show SMILES FC(F)(F)CCCN1CCC(CS(=O)(=O)N2[C@H]3CC[C@@H]2C[C@@H](C3)NC(=O)c2cc3CC(=O)Nc3cc2Cl)CC1 |r,TLB:12:15:18.17:22.21.20| Show InChI InChI=1S/C26H34ClF3N4O4S/c27-22-14-23-17(11-24(35)32-23)10-21(22)25(36)31-18-12-19-2-3-20(13-18)34(19)39(37,38)15-16-4-8-33(9-5-16)7-1-6-26(28,29)30/h10,14,16,18-20H,1-9,11-13,15H2,(H,31,36)(H,32,35)/t18-,19+,20- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378461
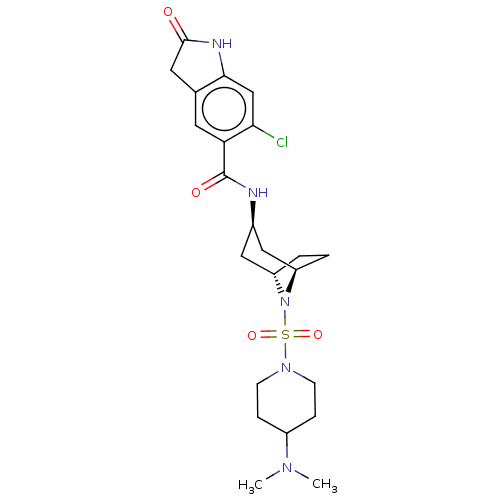
(6-chloro-N-((1R,3r,5S)-8-((4-(dimethylamino)piperi...)Show SMILES CN(C)C1CCN(CC1)S(=O)(=O)N1[C@H]2CC[C@@H]1C[C@@H](C2)NC(=O)c1cc2CC(=O)Nc2cc1Cl |r,TLB:9:12:15.14:19.18.17| Show InChI InChI=1S/C23H32ClN5O4S/c1-27(2)16-5-7-28(8-6-16)34(32,33)29-17-3-4-18(29)12-15(11-17)25-23(31)19-9-14-10-22(30)26-21(14)13-20(19)24/h9,13,15-18H,3-8,10-12H2,1-2H3,(H,25,31)(H,26,30)/t15-,17+,18- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378463
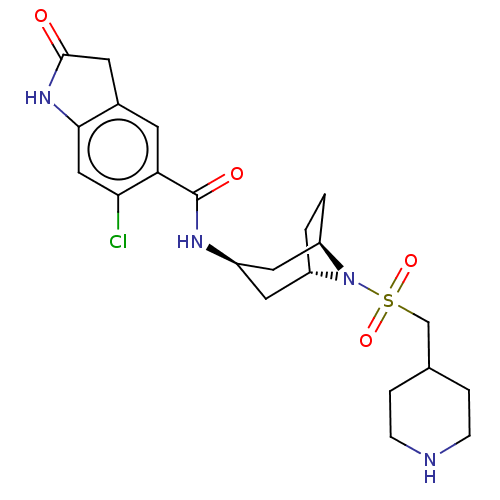
(6-chloro-2-oxo-N-((1R,3r,5S)-8-((piperidin-4-ylmet...)Show SMILES Clc1cc2NC(=O)Cc2cc1C(=O)N[C@@H]1C[C@@H]2CC[C@H](C1)N2S(=O)(=O)CC1CCNCC1 |r,TLB:22:21:15.14.20:18.17| Show InChI InChI=1S/C22H29ClN4O4S/c23-19-11-20-14(8-21(28)26-20)7-18(19)22(29)25-15-9-16-1-2-17(10-15)27(16)32(30,31)12-13-3-5-24-6-4-13/h7,11,13,15-17,24H,1-6,8-10,12H2,(H,25,29)(H,26,28)/t15-,16+,17- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM50500875
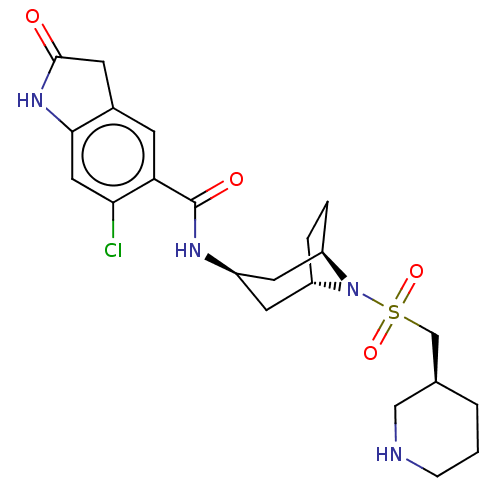
(CHEMBL3798745)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)NC(=O)c1cc3CC(=O)Nc3cc1Cl)N2S(=O)(=O)C[C@H]1CCCNC1 |r| Show InChI InChI=1S/C22H29ClN4O4S/c23-19-10-20-14(7-21(28)26-20)6-18(19)22(29)25-15-8-16-3-4-17(9-15)27(16)32(30,31)12-13-2-1-5-24-11-13/h6,10,13,15-17,24H,1-5,7-9,11-12H2,(H,25,29)(H,26,28)/t13-,15-,16+,17-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378459

(N-((1R,3r,5S)-8-((4-(benzylamino)piperidin-1-yl)su...)Show SMILES Clc1cc2NC(=O)Cc2cc1C(=O)N[C@@H]1C[C@@H]2CC[C@H](C1)N2S(=O)(=O)N1CCC(CC1)NCc1ccccc1 |r,TLB:22:21:15.14.20:18.17| Show InChI InChI=1S/C28H34ClN5O4S/c29-25-16-26-19(13-27(35)32-26)12-24(25)28(36)31-21-14-22-6-7-23(15-21)34(22)39(37,38)33-10-8-20(9-11-33)30-17-18-4-2-1-3-5-18/h1-5,12,16,20-23,30H,6-11,13-15,17H2,(H,31,36)(H,32,35)/t21-,22+,23- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378464

(2-oxo-N-((1R,3r,5S)-8-((piperidin-4-ylmethyl)sulfo...)Show SMILES O=C(N[C@@H]1C[C@@H]2CC[C@H](C1)N2S(=O)(=O)CC1CCNCC1)c1ccc2NC(=O)Cc2c1 |r,TLB:11:10:7.6:4.3.9| Show InChI InChI=1S/C22H30N4O4S/c27-21-10-16-9-15(1-4-20(16)25-21)22(28)24-17-11-18-2-3-19(12-17)26(18)31(29,30)13-14-5-7-23-8-6-14/h1,4,9,14,17-19,23H,2-3,5-8,10-13H2,(H,24,28)(H,25,27)/t17-,18+,19- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378442
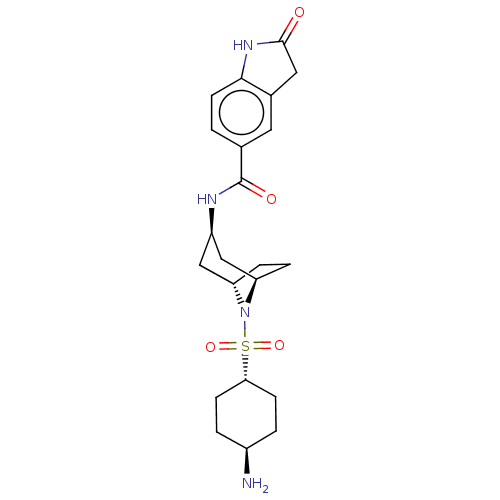
(N-((1R,3R,5S)-8-(((1r,4R)-4-aminocyclohexyl)sulfon...)Show SMILES N[C@H]1CC[C@@H](CC1)S(=O)(=O)N1[C@H]2CC[C@@H]1C[C@@H](C2)NC(=O)c1ccc2NC(=O)Cc2c1 |r,wU:1.0,14.15,11.11,wD:4.7,16.20,TLB:7:10:13.12:17.16.15,(-9.98,-1.26,;-8.44,-1.26,;-7.67,.07,;-6.13,.07,;-5.36,-1.26,;-6.13,-2.6,;-7.67,-2.6,;-3.82,-1.26,;-3.82,-2.8,;-3.82,.28,;-2.28,-1.26,;-1.88,-3,;-3.08,-2.04,;-3.08,-.5,;-1.88,.46,;-.38,.12,;.29,-1.27,;-.38,-2.66,;1.83,-1.27,;2.6,.06,;1.83,1.4,;4.14,.06,;4.91,-1.27,;6.45,-1.27,;7.22,.06,;8.73,.38,;8.89,1.91,;9.98,3,;7.48,2.54,;6.45,1.4,;4.91,1.4,)| Show InChI InChI=1S/C22H30N4O4S/c23-15-2-6-19(7-3-15)31(29,30)26-17-4-5-18(26)12-16(11-17)24-22(28)13-1-8-20-14(9-13)10-21(27)25-20/h1,8-9,15-19H,2-7,10-12,23H2,(H,24,28)(H,25,27)/t15-,16-,17+,18-,19- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM50500876
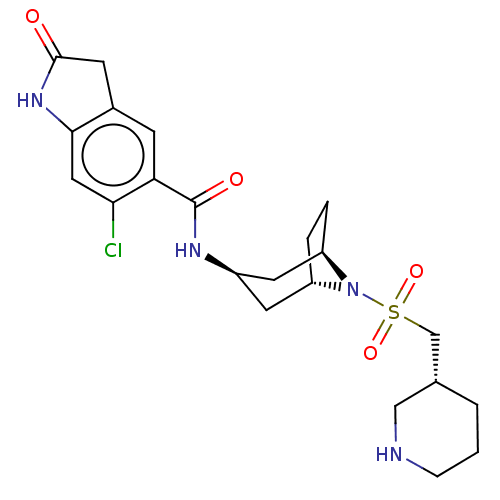
(CHEMBL3797575)Show SMILES [H][C@]12CC[C@]([H])(C[C@@H](C1)NC(=O)c1cc3CC(=O)Nc3cc1Cl)N2S(=O)(=O)C[C@@H]1CCCNC1 |r| Show InChI InChI=1S/C22H29ClN4O4S/c23-19-10-20-14(7-21(28)26-20)6-18(19)22(29)25-15-8-16-3-4-17(9-15)27(16)32(30,31)12-13-2-1-5-24-11-13/h6,10,13,15-17,24H,1-5,7-9,11-12H2,(H,25,29)(H,26,28)/t13-,15-,16+,17-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase PLK3
(Homo sapiens (Human)) | BDBM50362988

(CHEMBL1945500)Show SMILES CN(C)CCCc1cc(Cl)cc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C23H23Cl2N5S/c1-30(2)7-3-4-14-8-17(25)11-18(9-14)27-23-26-13-15-10-21(31)28-20-12-16(24)5-6-19(20)22(15)29-23/h5-6,8-9,11-13H,3-4,7,10H2,1-2H3,(H,28,31)(H,26,27,29) | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PLK3 |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase PLK3
(Homo sapiens (Human)) | BDBM50362985

(CHEMBL1945502)Show SMILES COc1ccc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)cc1CCCN(C)C Show InChI InChI=1S/C24H26ClN5OS/c1-30(2)10-4-5-15-11-18(7-9-21(15)31-3)27-24-26-14-16-12-22(32)28-20-13-17(25)6-8-19(20)23(16)29-24/h6-9,11,13-14H,4-5,10,12H2,1-3H3,(H,28,32)(H,26,27,29) | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PLK3 |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
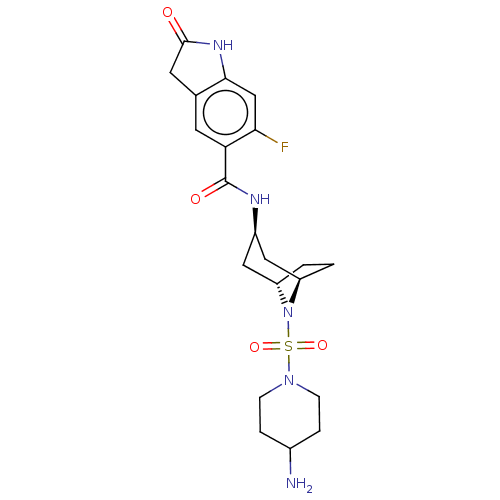
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378443
(N-((1R,3r,5S)-8-((4-aminopiperidin-1-yl)sulfonyl)-...)Show SMILES NC1CCN(CC1)S(=O)(=O)N1[C@H]2CC[C@@H]1C[C@@H](C2)NC(=O)c1cc2CC(=O)Nc2cc1F |r,TLB:7:10:13.12:17.16.15| Show InChI InChI=1S/C21H28FN5O4S/c22-18-11-19-12(8-20(28)25-19)7-17(18)21(29)24-14-9-15-1-2-16(10-14)27(15)32(30,31)26-5-3-13(23)4-6-26/h7,11,13-16H,1-6,8-10,23H2,(H,24,29)(H,25,28)/t14-,15+,16- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase PLK3
(Homo sapiens (Human)) | BDBM50362986
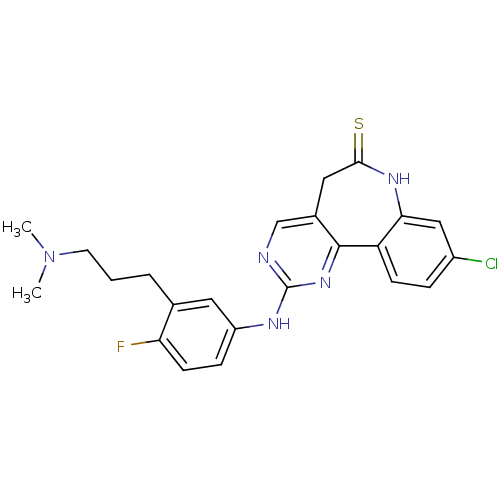
(CHEMBL1945501)Show SMILES CN(C)CCCc1cc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)ccc1F Show InChI InChI=1S/C23H23ClFN5S/c1-30(2)9-3-4-14-10-17(6-8-19(14)25)27-23-26-13-15-11-21(31)28-20-12-16(24)5-7-18(20)22(15)29-23/h5-8,10,12-13H,3-4,9,11H2,1-2H3,(H,28,31)(H,26,27,29) | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PLK3 |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase PLK3
(Homo sapiens (Human)) | BDBM50362984
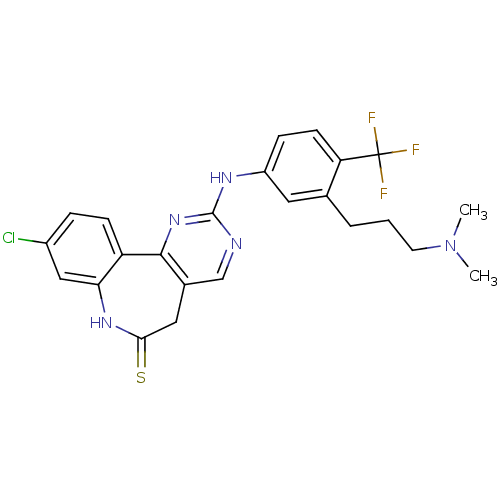
(CHEMBL1945801)Show SMILES CN(C)CCCc1cc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)ccc1C(F)(F)F Show InChI InChI=1S/C24H23ClF3N5S/c1-33(2)9-3-4-14-10-17(6-8-19(14)24(26,27)28)30-23-29-13-15-11-21(34)31-20-12-16(25)5-7-18(20)22(15)32-23/h5-8,10,12-13H,3-4,9,11H2,1-2H3,(H,31,34)(H,29,30,32) | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PLK3 |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50362981
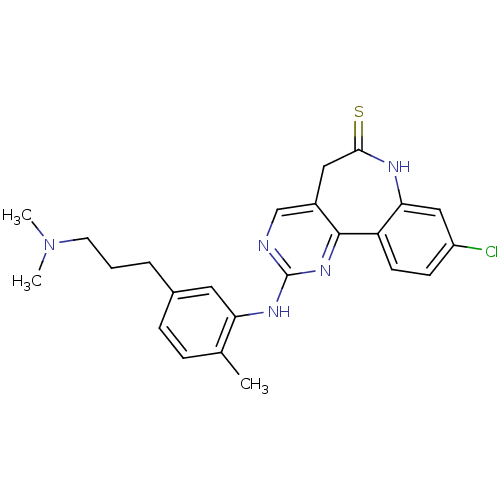
(CHEMBL1945171)Show SMILES CN(C)CCCc1ccc(C)c(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C24H26ClN5S/c1-15-6-7-16(5-4-10-30(2)3)11-20(15)28-24-26-14-17-12-22(31)27-21-13-18(25)8-9-19(21)23(17)29-24/h6-9,11,13-14H,4-5,10,12H2,1-3H3,(H,27,31)(H,26,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of KDR |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50362982
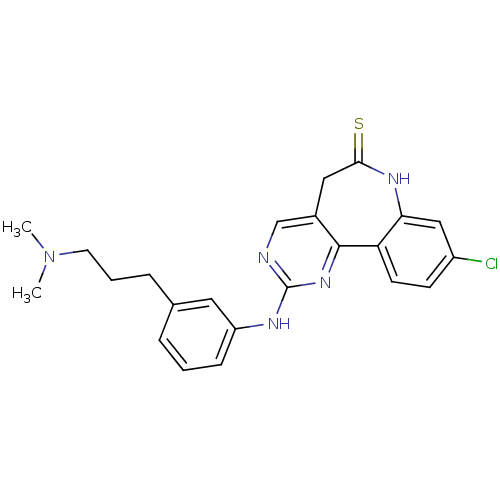
(CHEMBL1945170)Show SMILES CN(C)CCCc1cccc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C23H24ClN5S/c1-29(2)10-4-6-15-5-3-7-18(11-15)26-23-25-14-16-12-21(30)27-20-13-17(24)8-9-19(20)22(16)28-23/h3,5,7-9,11,13-14H,4,6,10,12H2,1-2H3,(H,27,30)(H,25,26,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of KDR |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM50362976
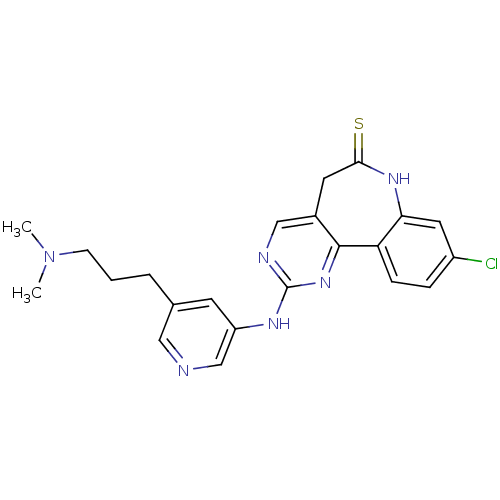
(CHEMBL1945803)Show SMILES CN(C)CCCc1cncc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C22H23ClN6S/c1-29(2)7-3-4-14-8-17(13-24-11-14)26-22-25-12-15-9-20(30)27-19-10-16(23)5-6-18(19)21(15)28-22/h5-6,8,10-13H,3-4,7,9H2,1-2H3,(H,27,30)(H,25,26,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of FGFR1 |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50362976

(CHEMBL1945803)Show SMILES CN(C)CCCc1cncc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C22H23ClN6S/c1-29(2)7-3-4-14-8-17(13-24-11-14)26-22-25-12-15-9-20(30)27-19-10-16(23)5-6-18(19)21(15)28-22/h5-6,8,10-13H,3-4,7,9H2,1-2H3,(H,27,30)(H,25,26,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of KDR |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase PLK3
(Homo sapiens (Human)) | BDBM50362982

(CHEMBL1945170)Show SMILES CN(C)CCCc1cccc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C23H24ClN5S/c1-29(2)10-4-6-15-5-3-7-18(11-15)26-23-25-14-16-12-21(30)27-20-13-17(24)8-9-19(20)22(16)28-23/h3,5,7-9,11,13-14H,4,6,10,12H2,1-2H3,(H,27,30)(H,25,26,28) | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PLK3 |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM50362982

(CHEMBL1945170)Show SMILES CN(C)CCCc1cccc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C23H24ClN5S/c1-29(2)10-4-6-15-5-3-7-18(11-15)26-23-25-14-16-12-21(30)27-20-13-17(24)8-9-19(20)22(16)28-23/h3,5,7-9,11,13-14H,4,6,10,12H2,1-2H3,(H,27,30)(H,25,26,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of FGFR1 |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378473
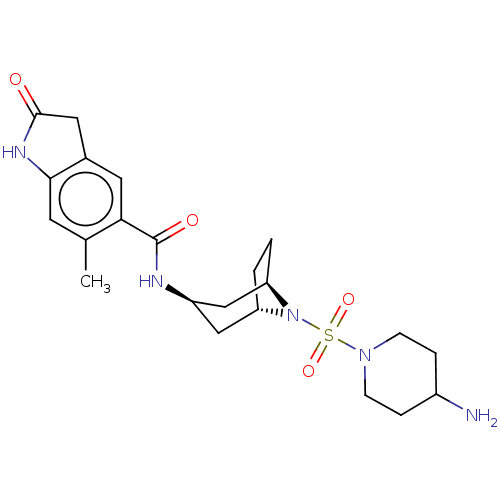
(N-((1R,3r,5S)-8-((4-aminopiperidin-1-yl)sulfonyl)-...)Show SMILES Cc1cc2NC(=O)Cc2cc1C(=O)N[C@@H]1C[C@@H]2CC[C@H](C1)N2S(=O)(=O)N1CCC(N)CC1 |r,TLB:22:21:15.14.20:18.17| Show InChI InChI=1S/C22H31N5O4S/c1-13-8-20-14(10-21(28)25-20)9-19(13)22(29)24-16-11-17-2-3-18(12-16)27(17)32(30,31)26-6-4-15(23)5-7-26/h8-9,15-18H,2-7,10-12,23H2,1H3,(H,24,29)(H,25,28)/t16-,17+,18- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase PLK3
(Homo sapiens (Human)) | BDBM50362981

(CHEMBL1945171)Show SMILES CN(C)CCCc1ccc(C)c(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C24H26ClN5S/c1-15-6-7-16(5-4-10-30(2)3)11-20(15)28-24-26-14-17-12-22(31)27-21-13-18(25)8-9-19(21)23(17)29-24/h6-9,11,13-14H,4-5,10,12H2,1-3H3,(H,27,31)(H,26,28,29) | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PLK3 |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50362982

(CHEMBL1945170)Show SMILES CN(C)CCCc1cccc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C23H24ClN5S/c1-29(2)10-4-6-15-5-3-7-18(11-15)26-23-25-14-16-12-21(30)27-20-13-17(24)8-9-19(20)22(16)28-23/h3,5,7-9,11,13-14H,4,6,10,12H2,1-2H3,(H,27,30)(H,25,26,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PDGFRb |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Mast/stem cell growth factor receptor Kit
(Homo sapiens (Human)) | BDBM50362976

(CHEMBL1945803)Show SMILES CN(C)CCCc1cncc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C22H23ClN6S/c1-29(2)7-3-4-14-8-17(13-24-11-14)26-22-25-12-15-9-20(30)27-19-10-16(23)5-6-18(19)21(15)28-22/h5-6,8,10-13H,3-4,7,9H2,1-2H3,(H,27,30)(H,25,26,28) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of KIT |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50362986

(CHEMBL1945501)Show SMILES CN(C)CCCc1cc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)ccc1F Show InChI InChI=1S/C23H23ClFN5S/c1-30(2)9-3-4-14-10-17(6-8-19(14)25)27-23-26-13-15-11-21(31)28-20-12-16(24)5-7-18(20)22(15)29-23/h5-8,10,12-13H,3-4,9,11H2,1-2H3,(H,28,31)(H,26,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of KDR |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50362986

(CHEMBL1945501)Show SMILES CN(C)CCCc1cc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)ccc1F Show InChI InChI=1S/C23H23ClFN5S/c1-30(2)9-3-4-14-10-17(6-8-19(14)25)27-23-26-13-15-11-21(31)28-20-12-16(24)5-7-18(20)22(15)29-23/h5-8,10,12-13H,3-4,9,11H2,1-2H3,(H,28,31)(H,26,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PDGFRb |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378478

(1-methyl-2-oxo-N-((1R,3r,5S)-8-((piperidin-4-ylmet...)Show SMILES CN1C(=O)Cc2cc(ccc12)C(=O)N[C@@H]1C[C@@H]2CC[C@H](C1)N2S(=O)(=O)CC1CCNCC1 |r,TLB:22:21:15.14.20:18.17| Show InChI InChI=1S/C23H32N4O4S/c1-26-21-5-2-16(10-17(21)11-22(26)28)23(29)25-18-12-19-3-4-20(13-18)27(19)32(30,31)14-15-6-8-24-9-7-15/h2,5,10,15,18-20,24H,3-4,6-9,11-14H2,1H3,(H,25,29)/t18-,19+,20- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of full length N-terminal His-tagged SMYD3 (1 to 428 residues) (unknown origin) expressed in Escherichia coli using N-terminal GST-tagged ... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50362976

(CHEMBL1945803)Show SMILES CN(C)CCCc1cncc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C22H23ClN6S/c1-29(2)7-3-4-14-8-17(13-24-11-14)26-22-25-12-15-9-20(30)27-19-10-16(23)5-6-18(19)21(15)28-22/h5-6,8,10-13H,3-4,7,9H2,1-2H3,(H,27,30)(H,25,26,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PDGFRb |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50362985

(CHEMBL1945502)Show SMILES COc1ccc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)cc1CCCN(C)C Show InChI InChI=1S/C24H26ClN5OS/c1-30(2)10-4-5-15-11-18(7-9-21(15)31-3)27-24-26-14-16-12-22(32)28-20-13-17(25)6-8-19(20)23(16)29-24/h6-9,11,13-14H,4-5,10,12H2,1-3H3,(H,28,32)(H,26,27,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PDGFRb |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Receptor-type tyrosine-protein kinase FLT3
(Homo sapiens (Human)) | BDBM50362982

(CHEMBL1945170)Show SMILES CN(C)CCCc1cccc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C23H24ClN5S/c1-29(2)10-4-6-15-5-3-7-18(11-15)26-23-25-14-16-12-21(30)27-20-13-17(24)8-9-19(20)22(16)28-23/h3,5,7-9,11,13-14H,4,6,10,12H2,1-2H3,(H,27,30)(H,25,26,28) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of FLT3 |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase PLK3
(Homo sapiens (Human)) | BDBM50362976

(CHEMBL1945803)Show SMILES CN(C)CCCc1cncc(Nc2ncc3CC(=S)Nc4cc(Cl)ccc4-c3n2)c1 Show InChI InChI=1S/C22H23ClN6S/c1-29(2)7-3-4-14-8-17(13-24-11-14)26-22-25-12-15-9-20(30)27-19-10-16(23)5-6-18(19)21(15)28-22/h5-6,8,10-13H,3-4,7,9H2,1-2H3,(H,27,30)(H,25,26,28) | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of PLK3 |
J Med Chem 55: 197-208 (2012)
Article DOI: 10.1021/jm2011172
BindingDB Entry DOI: 10.7270/Q21C1XBR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378462

(6-chloro-2-oxo-N-((1R,3r,5S)-8-(((1-(4,4,4-trifluo...)Show SMILES FC(F)(F)CCCN1CCC(CS(=O)(=O)N2[C@H]3CC[C@@H]2C[C@@H](C3)NC(=O)c2cc3CC(=O)Nc3cc2Cl)CC1 |r,TLB:12:15:18.17:22.21.20| Show InChI InChI=1S/C26H34ClF3N4O4S/c27-22-14-23-17(11-24(35)32-23)10-21(22)25(36)31-18-12-19-2-3-20(13-18)34(19)39(37,38)15-16-4-8-33(9-5-16)7-1-6-26(28,29)30/h10,14,16,18-20H,1-9,11-13,15H2,(H,31,36)(H,32,35)/t18-,19+,20- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human SMYD3 expressed in HEK293T/17 cells using FLAG-tagged MEKK2 as substrate incubated for 30 mins in low air flow area followed by i... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | |
Histone-lysine N-methyltransferase SMYD3
(Homo sapiens (Human)) | BDBM378459

(N-((1R,3r,5S)-8-((4-(benzylamino)piperidin-1-yl)su...)Show SMILES Clc1cc2NC(=O)Cc2cc1C(=O)N[C@@H]1C[C@@H]2CC[C@H](C1)N2S(=O)(=O)N1CCC(CC1)NCc1ccccc1 |r,TLB:22:21:15.14.20:18.17| Show InChI InChI=1S/C28H34ClN5O4S/c29-25-16-26-19(13-27(35)32-26)12-24(25)28(36)31-21-14-22-6-7-23(15-21)34(22)39(37,38)33-10-8-20(9-11-33)30-17-18-4-2-1-3-5-18/h1-5,12,16,20-23,30H,6-11,13-15,17H2,(H,31,36)(H,32,35)/t21-,22+,23- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
Epizyme Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human SMYD3 expressed in HEK293T/17 cells using FLAG-tagged MEKK2 as substrate incubated for 30 mins in low air flow area followed by i... |
ACS Med Chem Lett 7: 134-8 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00272
BindingDB Entry DOI: 10.7270/Q2NG4TPR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data