

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM33280 (CHEMBL153094 | aliphatic sulfamate, 2) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 0.700 | -51.9 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 7 (Homo sapiens (Human)) | BDBM10880 (AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM33279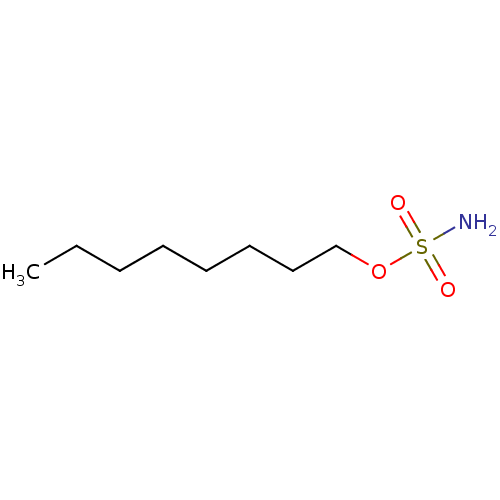 (CHEMBL24261 | aliphatic sulfamate, 1) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 2.60 | -48.7 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 12 (Homo sapiens (Human)) | BDBM10884 ((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 7 (Homo sapiens (Human)) | BDBM10884 ((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM33279 (CHEMBL24261 | aliphatic sulfamate, 1) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 3.70 | -47.8 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM33281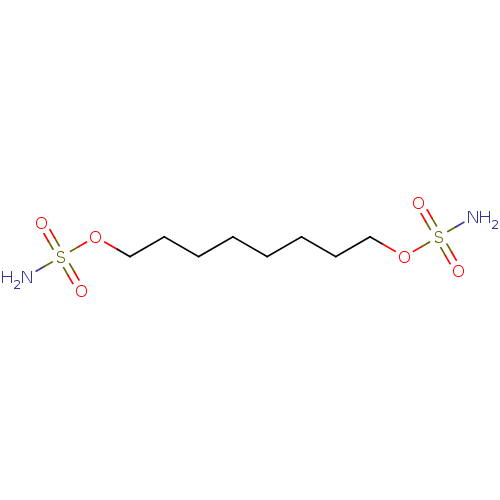 (CHEMBL182455 | bis-sulfamate, 3) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 12 (Homo sapiens (Human)) | BDBM10880 (AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM33282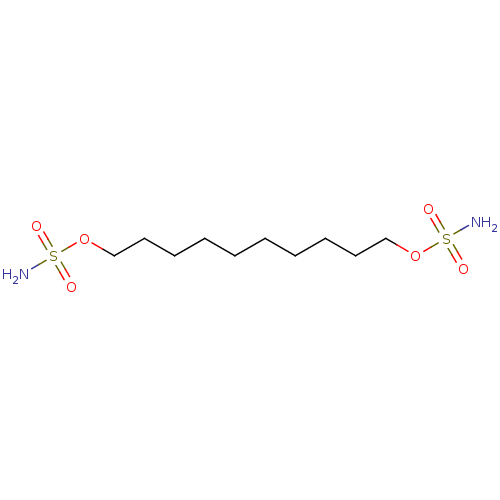 (CHEMBL171476 | bis-sulfamate, 4) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 12 (Homo sapiens (Human)) | BDBM33279 (CHEMBL24261 | aliphatic sulfamate, 1) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 12 (Homo sapiens (Human)) | BDBM33281 (CHEMBL182455 | bis-sulfamate, 3) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 12 (Homo sapiens (Human)) | BDBM33280 (CHEMBL153094 | aliphatic sulfamate, 2) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 7.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 12 (Homo sapiens (Human)) | BDBM33282 (CHEMBL171476 | bis-sulfamate, 4) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 8.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM10884 ((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | 9 | -45.6 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 6 (Homo sapiens (Human)) | BDBM10884 ((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 6 (Homo sapiens (Human)) | BDBM10880 (AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM10880 (AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | 12 | -44.9 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM33281 (CHEMBL182455 | bis-sulfamate, 3) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | 14.6 | -44.4 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 13 (Mus musculus (mouse)) | BDBM10880 (AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 13 (Mus musculus (mouse)) | BDBM10884 ((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM33280 (CHEMBL153094 | aliphatic sulfamate, 2) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 23.1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 2 (Homo sapiens (Human)) | BDBM33282 (CHEMBL171476 | bis-sulfamate, 4) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | 24.5 | -43.1 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM33279 (CHEMBL24261 | aliphatic sulfamate, 1) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM10880 (AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 14 (Homo sapiens (Human)) | BDBM10884 ((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 5B, mitochondrial (Homo sapiens (Human)) | BDBM10884 ((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 33 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 14 (Homo sapiens (Human)) | BDBM10880 (AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | 41 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 5A, mitochondrial (Homo sapiens (Human)) | BDBM10884 ((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 42 | -41.8 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 9 (Homo sapiens (Human)) | BDBM10884 ((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 52 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 5B, mitochondrial (Homo sapiens (Human)) | BDBM10880 (AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 54 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 5A, mitochondrial (Homo sapiens (Human)) | BDBM10880 (AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 63 | -40.8 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 4 (Homo sapiens (Human)) | BDBM33280 (CHEMBL153094 | aliphatic sulfamate, 2) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 67.6 | -40.6 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 4 (Homo sapiens (Human)) | BDBM10880 (AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | 74 | -40.4 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 7 (Homo sapiens (Human)) | BDBM33279 (CHEMBL24261 | aliphatic sulfamate, 1) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 76.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 7 (Homo sapiens (Human)) | BDBM33281 (CHEMBL182455 | bis-sulfamate, 3) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 79.6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 4 (Homo sapiens (Human)) | BDBM33279 (CHEMBL24261 | aliphatic sulfamate, 1) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 79.9 | -40.2 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 4 (Homo sapiens (Human)) | BDBM33281 (CHEMBL182455 | bis-sulfamate, 3) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 84.1 | -40.1 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 7 (Homo sapiens (Human)) | BDBM33280 (CHEMBL153094 | aliphatic sulfamate, 2) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 88.9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 4 (Homo sapiens (Human)) | BDBM33282 (CHEMBL171476 | bis-sulfamate, 4) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 91.2 | -39.9 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 7 (Homo sapiens (Human)) | BDBM33282 (CHEMBL171476 | bis-sulfamate, 4) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 92.3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM10880 (AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | 250 | -37.4 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM33281 (CHEMBL182455 | bis-sulfamate, 3) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 378 | -36.4 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 14 (Homo sapiens (Human)) | BDBM33279 (CHEMBL24261 | aliphatic sulfamate, 1) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM33280 (CHEMBL153094 | aliphatic sulfamate, 2) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 530 | -35.6 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 13 (Mus musculus (mouse)) | BDBM33281 (CHEMBL182455 | bis-sulfamate, 3) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 569 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 14 (Homo sapiens (Human)) | BDBM33281 (CHEMBL182455 | bis-sulfamate, 3) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 605 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 5B, mitochondrial (Homo sapiens (Human)) | BDBM33279 (CHEMBL24261 | aliphatic sulfamate, 1) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 624 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 14 (Homo sapiens (Human)) | BDBM33280 (CHEMBL153094 | aliphatic sulfamate, 2) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 711 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 6 (Homo sapiens (Human)) | BDBM33279 (CHEMBL24261 | aliphatic sulfamate, 1) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 795 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 5A, mitochondrial (Homo sapiens (Human)) | BDBM33280 (CHEMBL153094 | aliphatic sulfamate, 2) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 805 | -34.6 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 14 (Homo sapiens (Human)) | BDBM33282 (CHEMBL171476 | bis-sulfamate, 4) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 852 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 3 (Homo sapiens (Human)) | BDBM33280 (CHEMBL153094 | aliphatic sulfamate, 2) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 877 | -34.3 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 5B, mitochondrial (Homo sapiens (Human)) | BDBM33280 (CHEMBL153094 | aliphatic sulfamate, 2) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 888 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM33282 (CHEMBL171476 | bis-sulfamate, 4) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 890 | -34.3 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 13 (Mus musculus (mouse)) | BDBM33280 (CHEMBL153094 | aliphatic sulfamate, 2) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 956 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 13 (Mus musculus (mouse)) | BDBM33282 (CHEMBL171476 | bis-sulfamate, 4) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 960 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 6 (Homo sapiens (Human)) | BDBM33281 (CHEMBL182455 | bis-sulfamate, 3) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.02E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 13 (Mus musculus (mouse)) | BDBM33279 (CHEMBL24261 | aliphatic sulfamate, 1) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.13E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 3 (Homo sapiens (Human)) | BDBM33281 (CHEMBL182455 | bis-sulfamate, 3) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.13E+3 | -33.7 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 5A, mitochondrial (Homo sapiens (Human)) | BDBM33281 (CHEMBL182455 | bis-sulfamate, 3) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.20E+3 | -33.6 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 3 (Homo sapiens (Human)) | BDBM33282 (CHEMBL171476 | bis-sulfamate, 4) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 1.21E+3 | -33.6 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 5A, mitochondrial (Homo sapiens (Human)) | BDBM33279 (CHEMBL24261 | aliphatic sulfamate, 1) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.25E+3 | -33.5 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 6 (Homo sapiens (Human)) | BDBM33280 (CHEMBL153094 | aliphatic sulfamate, 2) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.33E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 5A, mitochondrial (Homo sapiens (Human)) | BDBM33282 (CHEMBL171476 | bis-sulfamate, 4) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 1.37E+3 | -33.2 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 6 (Homo sapiens (Human)) | BDBM33282 (CHEMBL171476 | bis-sulfamate, 4) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 1.41E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 5B, mitochondrial (Homo sapiens (Human)) | BDBM33281 (CHEMBL182455 | bis-sulfamate, 3) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 1.46E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 5B, mitochondrial (Homo sapiens (Human)) | BDBM33282 (CHEMBL171476 | bis-sulfamate, 4) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 1.59E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 4 (Homo sapiens (Human)) | BDBM10884 ((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | 8.50E+3 | -28.7 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Carbonic anhydrase 3 (Homo sapiens (Human)) | BDBM33279 (CHEMBL24261 | aliphatic sulfamate, 1) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 9.10E+3 | -28.6 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 1 (Homo sapiens (Human)) | BDBM10884 ((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | 5.00E+4 | -24.4 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 3 (Homo sapiens (Human)) | BDBM10880 (AZA | AZA2 | AZM acetazolamide | Acerazolamide, AA...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank Article PubMed | 2.00E+5 | -21.0 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carbonic anhydrase 3 (Homo sapiens (Human)) | BDBM10884 ((2S,4S)-4-(ethylamino)-2-methyl-1,1-dioxo-2H,3H,4H...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase DrugBank MCE KEGG MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 7.70E+5 | -17.7 | n/a | n/a | n/a | n/a | n/a | 7.5 | 23 |
CNR | Assay Description An Applied Photophysics stopped-flow instrument has been used for assaying the CA-catalyzed CO2 hydration activity. Phenol red has been used as indic... | J Med Chem 52: 5990-8 (2009) Article DOI: 10.1021/jm900641r BindingDB Entry DOI: 10.7270/Q2GF0RVR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||