 Found 156 hits with Last Name = 'neel' and Initial = 'da'
Found 156 hits with Last Name = 'neel' and Initial = 'da' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50146331
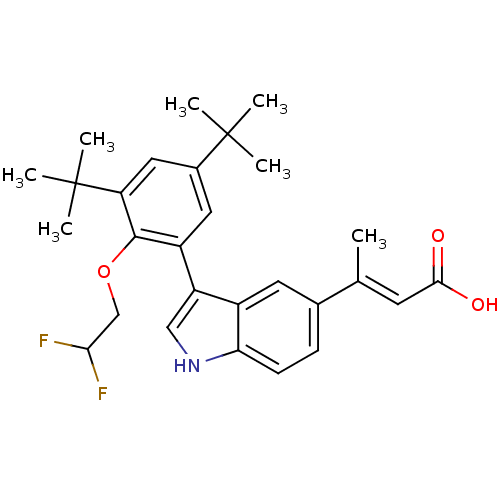
((E)-3-{3-[3,5-Di-tert-butyl-2-(2,2-difluoro-ethoxy...)Show SMILES C\C(=C/C(O)=O)c1ccc2[nH]cc(-c3cc(cc(c3OCC(F)F)C(C)(C)C)C(C)(C)C)c2c1 Show InChI InChI=1S/C28H33F2NO3/c1-16(10-25(32)33)17-8-9-23-19(11-17)21(14-31-23)20-12-18(27(2,3)4)13-22(28(5,6)7)26(20)34-15-24(29)30/h8-14,24,31H,15H2,1-7H3,(H,32,33)/b16-10+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-9-cis-RA from Retinoid X receptor alpha in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50146327

((E)-3-[3-(3,5-Di-tert-butyl-2-propoxy-phenyl)-1H-i...)Show SMILES CCCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)-c1c[nH]c2ccc(cc12)C(\C)=C\C(O)=O Show InChI InChI=1S/C29H37NO3/c1-9-12-33-27-22(15-20(28(3,4)5)16-24(27)29(6,7)8)23-17-30-25-11-10-19(14-21(23)25)18(2)13-26(31)32/h10-11,13-17,30H,9,12H2,1-8H3,(H,31,32)/b18-13+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-9-cis-RA from Retinoid X receptor alpha in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50129726
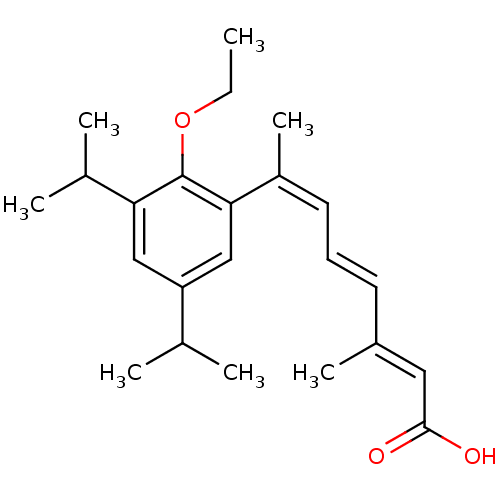
((2E,4E,6Z)-7-(2-Ethoxy-3,5-diisopropyl-phenyl)-3-m...)Show SMILES CCOc1c(cc(cc1\C(C)=C/C=C/C(/C)=C/C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C23H32O3/c1-8-26-23-20(16(4)5)13-19(15(2)3)14-21(23)18(7)11-9-10-17(6)12-22(24)25/h9-16H,8H2,1-7H3,(H,24,25)/b10-9+,17-12+,18-11- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-9-cis-RA from Retinoid X receptor alpha in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50146328
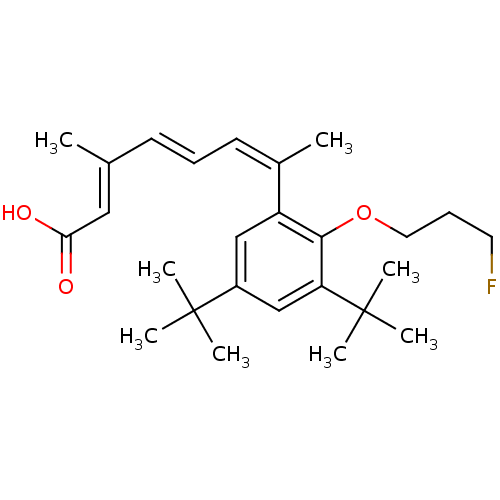
((2E,4E,6Z)-7-[3,5-Di-tert-butyl-2-(3-fluoro-propox...)Show SMILES C\C(\C=C\C=C(\C)c1cc(cc(c1OCCCF)C(C)(C)C)C(C)(C)C)=C/C(O)=O Show InChI InChI=1S/C26H37FO3/c1-18(15-23(28)29)11-9-12-19(2)21-16-20(25(3,4)5)17-22(26(6,7)8)24(21)30-14-10-13-27/h9,11-12,15-17H,10,13-14H2,1-8H3,(H,28,29)/b11-9+,18-15+,19-12- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Effective concentration for antagonistic activity against RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50146337
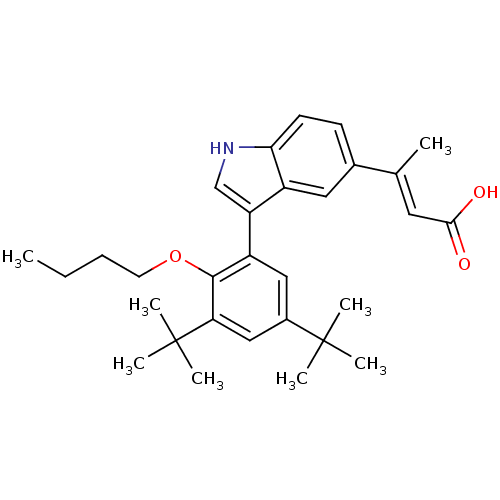
((E)-3-[3-(2-Butoxy-3,5-di-tert-butyl-phenyl)-1H-in...)Show SMILES CCCCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)-c1c[nH]c2ccc(cc12)C(\C)=C\C(O)=O Show InChI InChI=1S/C30H39NO3/c1-9-10-13-34-28-23(16-21(29(3,4)5)17-25(28)30(6,7)8)24-18-31-26-12-11-20(15-22(24)26)19(2)14-27(32)33/h11-12,14-18,31H,9-10,13H2,1-8H3,(H,32,33)/b19-14+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-9-cis-RA from Retinoid X receptor alpha in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50129725
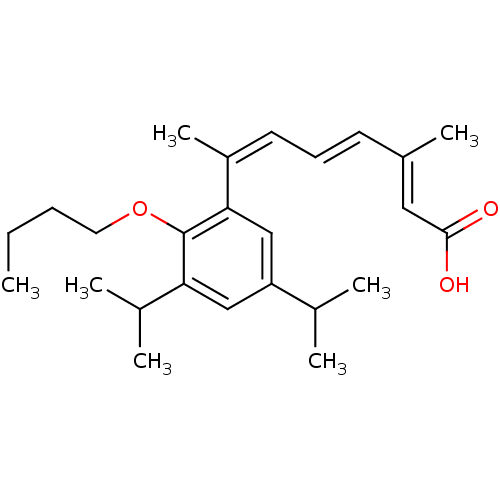
((2E,4E,6Z)-7-(2-Butoxy-3,5-diisopropyl-phenyl)-3-m...)Show SMILES CCCCOc1c(cc(cc1\C(C)=C/C=C/C(/C)=C/C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C25H36O3/c1-8-9-13-28-25-22(18(4)5)15-21(17(2)3)16-23(25)20(7)12-10-11-19(6)14-24(26)27/h10-12,14-18H,8-9,13H2,1-7H3,(H,26,27)/b11-10+,19-14+,20-12- | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Effective concentration for antagonistic activity against RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50146334
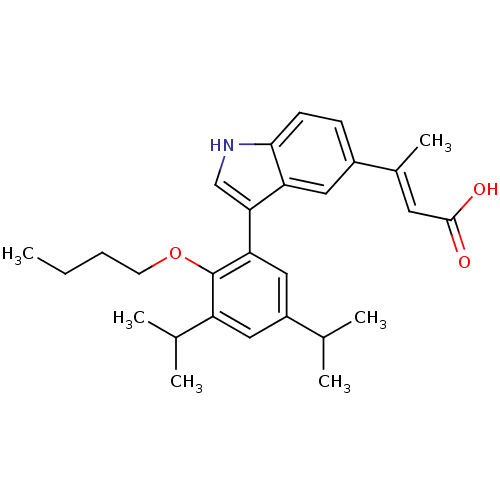
((E)-3-[3-(2-Butoxy-3,5-diisopropyl-phenyl)-1H-indo...)Show SMILES CCCCOc1c(cc(cc1-c1c[nH]c2ccc(cc12)C(\C)=C\C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C28H35NO3/c1-7-8-11-32-28-22(18(4)5)14-21(17(2)3)15-24(28)25-16-29-26-10-9-20(13-23(25)26)19(6)12-27(30)31/h9-10,12-18,29H,7-8,11H2,1-6H3,(H,30,31)/b19-12+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 7.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-9-cis-RA from Retinoid X receptor alpha in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50146330
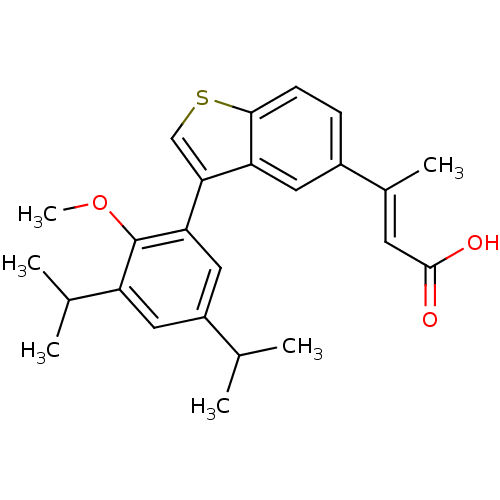
((E)-3-[3-(3,5-Diisopropyl-2-methoxy-phenyl)-benzo[...)Show SMILES COc1c(cc(cc1-c1csc2ccc(cc12)C(\C)=C\C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C25H28O3S/c1-14(2)18-11-19(15(3)4)25(28-6)21(12-18)22-13-29-23-8-7-17(10-20(22)23)16(5)9-24(26)27/h7-15H,1-6H3,(H,26,27)/b16-9+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-9-cis-RA from Retinoid X receptor alpha in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50146326

((E)-3-[3-(2-Ethoxy-3,5-diisopropyl-phenyl)-1H-inda...)Show SMILES CCOc1c(cc(cc1-c1[nH]nc2ccc(cc12)C(C)=CC(O)=O)C(C)C)C(C)C |w:20.23| Show InChI InChI=1S/C25H30N2O3/c1-7-30-25-19(15(4)5)12-18(14(2)3)13-21(25)24-20-11-17(16(6)10-23(28)29)8-9-22(20)26-27-24/h8-15H,7H2,1-6H3,(H,26,27)(H,28,29) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 69 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Effective concentration for antagonistic activity against RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50146325
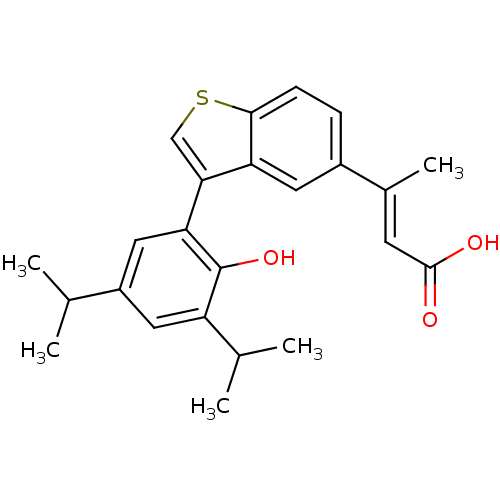
((E)-3-[3-(2-Hydroxy-3,5-diisopropyl-phenyl)-benzo[...)Show SMILES CC(C)c1cc(C(C)C)c(O)c(c1)-c1csc2ccc(cc12)C(\C)=C\C(O)=O Show InChI InChI=1S/C24H26O3S/c1-13(2)17-10-18(14(3)4)24(27)20(11-17)21-12-28-22-7-6-16(9-19(21)22)15(5)8-23(25)26/h6-14,27H,1-5H3,(H,25,26)/b15-8+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 131 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-9-cis-RA from Retinoid X receptor alpha in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50146335
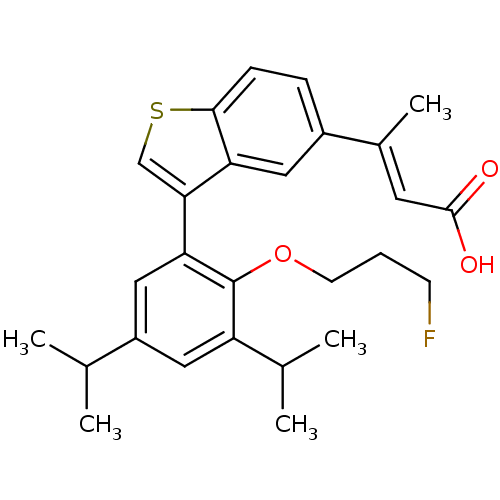
((E)-3-{3-[2-(3-Fluoro-propoxy)-3,5-diisopropyl-phe...)Show SMILES CC(C)c1cc(C(C)C)c(OCCCF)c(c1)-c1csc2ccc(cc12)C(\C)=C\C(O)=O Show InChI InChI=1S/C27H31FO3S/c1-16(2)20-13-21(17(3)4)27(31-10-6-9-28)23(14-20)24-15-32-25-8-7-19(12-22(24)25)18(5)11-26(29)30/h7-8,11-17H,6,9-10H2,1-5H3,(H,29,30)/b18-11+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 395 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-9-cis-RA from Retinoid X receptor alpha in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50146329
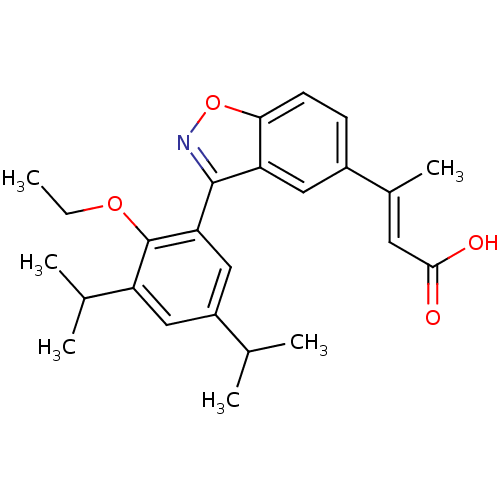
((E)-3-[3-(2-Ethoxy-3,5-diisopropyl-phenyl)-benzo[d...)Show SMILES CCOc1c(cc(cc1-c1noc2ccc(cc12)C(\C)=C\C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C25H29NO4/c1-7-29-25-19(15(4)5)12-18(14(2)3)13-21(25)24-20-11-17(16(6)10-23(27)28)8-9-22(20)30-26-24/h8-15H,7H2,1-6H3,(H,27,28)/b16-10+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 648 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Effective concentration for antagonistic activity against RXR-alpha expressed in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50146332
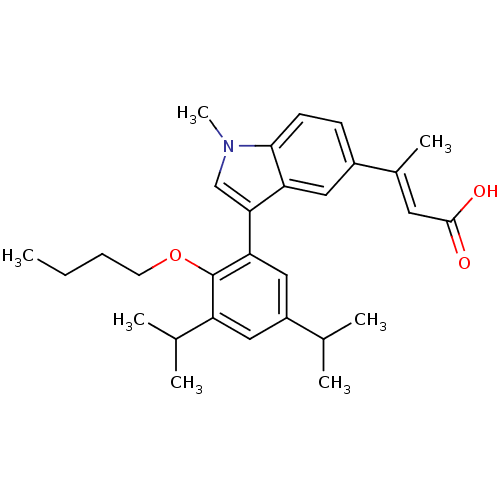
((E)-3-[3-(2-Butoxy-3,5-diisopropyl-phenyl)-1-methy...)Show SMILES CCCCOc1c(cc(cc1-c1cn(C)c2ccc(cc12)C(\C)=C\C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C29H37NO3/c1-8-9-12-33-29-23(19(4)5)15-22(18(2)3)16-25(29)26-17-30(7)27-11-10-21(14-24(26)27)20(6)13-28(31)32/h10-11,13-19H,8-9,12H2,1-7H3,(H,31,32)/b20-13+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-9-cis-RA from Retinoid X receptor alpha in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor RXR-alpha
(Homo sapiens (Human)) | BDBM50146336

((E)-3-[3-(2-Ethoxy-3,5-diisopropyl-phenyl)-imidazo...)Show SMILES CCOc1c(cc(cc1-c1cnc2ccc(cn12)C(\C)=C\C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C25H30N2O3/c1-7-30-25-20(16(4)5)11-19(15(2)3)12-21(25)22-13-26-23-9-8-18(14-27(22)23)17(6)10-24(28)29/h8-16H,7H2,1-6H3,(H,28,29)/b17-10+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 2.08E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-9-cis-RA from Retinoid X receptor alpha in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50146335

((E)-3-{3-[2-(3-Fluoro-propoxy)-3,5-diisopropyl-phe...)Show SMILES CC(C)c1cc(C(C)C)c(OCCCF)c(c1)-c1csc2ccc(cc12)C(\C)=C\C(O)=O Show InChI InChI=1S/C27H31FO3S/c1-16(2)20-13-21(17(3)4)27(31-10-6-9-28)23(14-20)24-15-32-25-8-7-19(12-22(24)25)18(5)11-26(29)30/h7-8,11-17H,6,9-10H2,1-5H3,(H,29,30)/b18-11+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50146336

((E)-3-[3-(2-Ethoxy-3,5-diisopropyl-phenyl)-imidazo...)Show SMILES CCOc1c(cc(cc1-c1cnc2ccc(cn12)C(\C)=C\C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C25H30N2O3/c1-7-30-25-20(16(4)5)11-19(15(2)3)12-21(25)22-13-26-23-9-8-18(14-27(22)23)17(6)10-24(28)29/h8-16H,7H2,1-6H3,(H,28,29)/b17-10+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50146332

((E)-3-[3-(2-Butoxy-3,5-diisopropyl-phenyl)-1-methy...)Show SMILES CCCCOc1c(cc(cc1-c1cn(C)c2ccc(cc12)C(\C)=C\C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C29H37NO3/c1-8-9-12-33-29-23(19(4)5)15-22(18(2)3)16-25(29)26-17-30(7)27-11-10-21(14-24(26)27)20(6)13-28(31)32/h10-11,13-19H,8-9,12H2,1-7H3,(H,31,32)/b20-13+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50146334

((E)-3-[3-(2-Butoxy-3,5-diisopropyl-phenyl)-1H-indo...)Show SMILES CCCCOc1c(cc(cc1-c1c[nH]c2ccc(cc12)C(\C)=C\C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C28H35NO3/c1-7-8-11-32-28-22(18(4)5)14-21(17(2)3)15-24(28)25-16-29-26-10-9-20(13-23(25)26)19(6)12-27(30)31/h9-10,12-18,29H,7-8,11H2,1-6H3,(H,30,31)/b19-12+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50146337

((E)-3-[3-(2-Butoxy-3,5-di-tert-butyl-phenyl)-1H-in...)Show SMILES CCCCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)-c1c[nH]c2ccc(cc12)C(\C)=C\C(O)=O Show InChI InChI=1S/C30H39NO3/c1-9-10-13-34-28-23(16-21(29(3,4)5)17-25(28)30(6,7)8)24-18-31-26-12-11-20(15-22(24)26)19(2)14-27(32)33/h11-12,14-18,31H,9-10,13H2,1-8H3,(H,32,33)/b19-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50146328

((2E,4E,6Z)-7-[3,5-Di-tert-butyl-2-(3-fluoro-propox...)Show SMILES C\C(\C=C\C=C(\C)c1cc(cc(c1OCCCF)C(C)(C)C)C(C)(C)C)=C/C(O)=O Show InChI InChI=1S/C26H37FO3/c1-18(15-23(28)29)11-9-12-19(2)21-16-20(25(3,4)5)17-22(26(6,7)8)24(21)30-14-10-13-27/h9,11-12,15-17H,10,13-14H2,1-8H3,(H,28,29)/b11-9+,18-15+,19-12- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50146327

((E)-3-[3-(3,5-Di-tert-butyl-2-propoxy-phenyl)-1H-i...)Show SMILES CCCOc1c(cc(cc1C(C)(C)C)C(C)(C)C)-c1c[nH]c2ccc(cc12)C(\C)=C\C(O)=O Show InChI InChI=1S/C29H37NO3/c1-9-12-33-27-22(15-20(28(3,4)5)16-24(27)29(6,7)8)23-17-30-25-11-10-19(14-21(23)25)18(2)13-26(31)32/h10-11,13-17,30H,9,12H2,1-8H3,(H,31,32)/b18-13+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50146331

((E)-3-{3-[3,5-Di-tert-butyl-2-(2,2-difluoro-ethoxy...)Show SMILES C\C(=C/C(O)=O)c1ccc2[nH]cc(-c3cc(cc(c3OCC(F)F)C(C)(C)C)C(C)(C)C)c2c1 Show InChI InChI=1S/C28H33F2NO3/c1-16(10-25(32)33)17-8-9-23-19(11-17)21(14-31-23)20-12-18(27(2,3)4)13-22(28(5,6)7)26(20)34-15-24(29)30/h8-14,24,31H,15H2,1-7H3,(H,32,33)/b16-10+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50146330

((E)-3-[3-(3,5-Diisopropyl-2-methoxy-phenyl)-benzo[...)Show SMILES COc1c(cc(cc1-c1csc2ccc(cc12)C(\C)=C\C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C25H28O3S/c1-14(2)18-11-19(15(3)4)25(28-6)21(12-18)22-13-29-23-8-7-17(10-20(22)23)16(5)9-24(26)27/h7-15H,1-6H3,(H,26,27)/b16-9+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50146329

((E)-3-[3-(2-Ethoxy-3,5-diisopropyl-phenyl)-benzo[d...)Show SMILES CCOc1c(cc(cc1-c1noc2ccc(cc12)C(\C)=C\C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C25H29NO4/c1-7-29-25-19(15(4)5)12-18(14(2)3)13-21(25)24-20-11-17(16(6)10-23(27)28)8-9-22(20)30-26-24/h8-15H,7H2,1-6H3,(H,27,28)/b16-10+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50129725

((2E,4E,6Z)-7-(2-Butoxy-3,5-diisopropyl-phenyl)-3-m...)Show SMILES CCCCOc1c(cc(cc1\C(C)=C/C=C/C(/C)=C/C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C25H36O3/c1-8-9-13-28-25-22(18(4)5)15-21(17(2)3)16-23(25)20(7)12-10-11-19(6)14-24(26)27/h10-12,14-18H,8-9,13H2,1-7H3,(H,26,27)/b11-10+,19-14+,20-12- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50146326

((E)-3-[3-(2-Ethoxy-3,5-diisopropyl-phenyl)-1H-inda...)Show SMILES CCOc1c(cc(cc1-c1[nH]nc2ccc(cc12)C(C)=CC(O)=O)C(C)C)C(C)C |w:20.23| Show InChI InChI=1S/C25H30N2O3/c1-7-30-25-19(15(4)5)12-18(14(2)3)13-21(25)24-20-11-17(16(6)10-23(28)29)8-9-22(20)26-27-24/h8-15H,7H2,1-6H3,(H,26,27)(H,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50129726

((2E,4E,6Z)-7-(2-Ethoxy-3,5-diisopropyl-phenyl)-3-m...)Show SMILES CCOc1c(cc(cc1\C(C)=C/C=C/C(/C)=C/C(O)=O)C(C)C)C(C)C Show InChI InChI=1S/C23H32O3/c1-8-26-23-20(16(4)5)13-19(15(2)3)14-21(23)18(7)11-9-10-17(6)12-22(24)25/h9-16H,8H2,1-7H3,(H,24,25)/b10-9+,17-12+,18-11- | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50146325

((E)-3-[3-(2-Hydroxy-3,5-diisopropyl-phenyl)-benzo[...)Show SMILES CC(C)c1cc(C(C)C)c(O)c(c1)-c1csc2ccc(cc12)C(\C)=C\C(O)=O Show InChI InChI=1S/C24H26O3S/c1-13(2)17-10-18(14(3)4)24(27)20(11-17)21-12-28-22-7-6-16(9-19(21)22)15(5)8-23(25)26/h6-14,27H,1-5H3,(H,25,26)/b15-8+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Ability to displace [3H]-ATRA from Retinoic acid receptor gamma in CV-1 cells |
Bioorg Med Chem Lett 14: 2759-63 (2004)
Article DOI: 10.1016/j.bmcl.2004.03.073
BindingDB Entry DOI: 10.7270/Q2513XNB |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of src kinase |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM17055
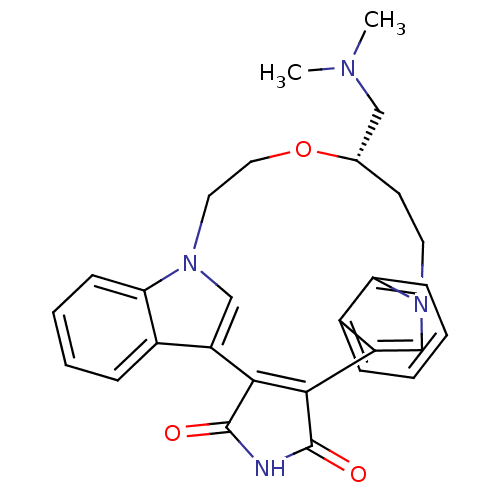
((18S)-18-[(dimethylamino)methyl]-17-oxa-4,14,21-tr...)Show SMILES CN(C)C[C@@H]1CCn2cc(C3=C(C(=O)NC3=O)c3cn(CCO1)c1ccccc31)c1ccccc21 |r,t:10| Show InChI InChI=1S/C28H28N4O3/c1-30(2)15-18-11-12-31-16-21(19-7-3-5-9-23(19)31)25-26(28(34)29-27(25)33)22-17-32(13-14-35-18)24-10-6-4-8-20(22)24/h3-10,16-18H,11-15H2,1-2H3,(H,29,33,34)/t18-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 1 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C eta |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50052034
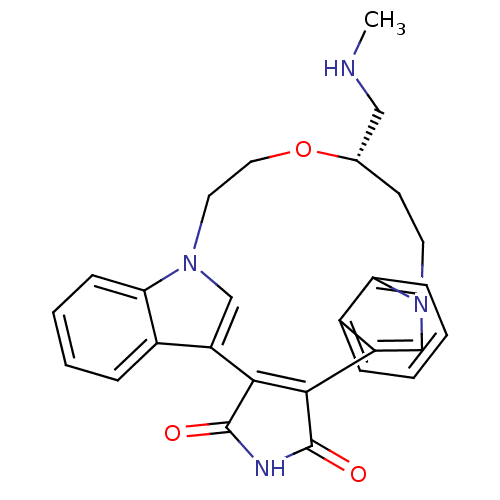
(18-methylaminomethyl-(18S)-17-oxa-4,14,21-triazahe...)Show SMILES CNC[C@@H]1CCn2cc(C3=C(C(=O)NC3=O)c3cn(CCO1)c1ccccc31)c1ccccc21 |t:9| Show InChI InChI=1S/C27H26N4O3/c1-28-14-17-10-11-30-15-20(18-6-2-4-8-22(18)30)24-25(27(33)29-26(24)32)21-16-31(12-13-34-17)23-9-5-3-7-19(21)23/h2-9,15-17,28H,10-14H2,1H3,(H,29,32,33)/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 2 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50052034

(18-methylaminomethyl-(18S)-17-oxa-4,14,21-triazahe...)Show SMILES CNC[C@@H]1CCn2cc(C3=C(C(=O)NC3=O)c3cn(CCO1)c1ccccc31)c1ccccc21 |t:9| Show InChI InChI=1S/C27H26N4O3/c1-28-14-17-10-11-30-15-20(18-6-2-4-8-22(18)30)24-25(27(33)29-26(24)32)21-16-31(12-13-34-17)23-9-5-3-7-19(21)23/h2-9,15-17,28H,10-14H2,1H3,(H,29,32,33)/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 1 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM17055

((18S)-18-[(dimethylamino)methyl]-17-oxa-4,14,21-tr...)Show SMILES CN(C)C[C@@H]1CCn2cc(C3=C(C(=O)NC3=O)c3cn(CCO1)c1ccccc31)c1ccccc21 |r,t:10| Show InChI InChI=1S/C28H28N4O3/c1-30(2)15-18-11-12-31-16-21(19-7-3-5-9-23(19)31)25-26(28(34)29-27(25)33)22-17-32(13-14-35-18)24-10-6-4-8-20(22)24/h3-10,16-18H,11-15H2,1-2H3,(H,29,33,34)/t18-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 2 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C epsilon type
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C epsilon |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 2 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 1 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50052041
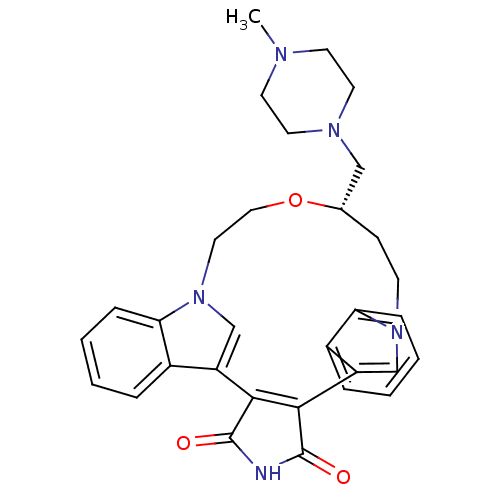
(18-(4-methylhexahydro-1-pyrazinylmethyl)-(18S)-17-...)Show SMILES CN1CCN(C[C@@H]2CCn3cc(C4=C(C(=O)NC4=O)c4cn(CCO2)c2ccccc42)c2ccccc32)CC1 |t:12| Show InChI InChI=1S/C31H33N5O3/c1-33-12-14-34(15-13-33)18-21-10-11-35-19-24(22-6-2-4-8-26(22)35)28-29(31(38)32-30(28)37)25-20-36(16-17-39-21)27-9-5-3-7-23(25)27/h2-9,19-21H,10-18H2,1H3,(H,32,37,38)/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 2 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50052039
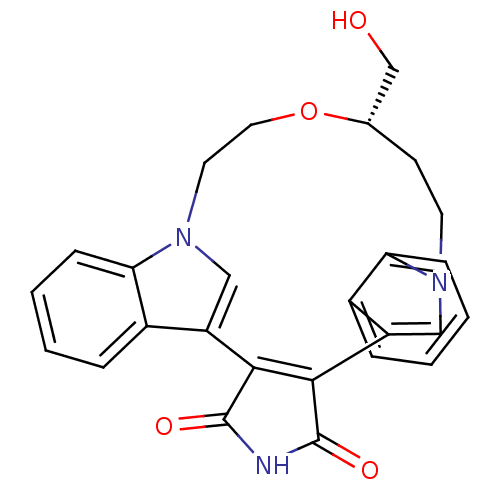
(18-hydroxymethyl-(18S)-17-oxa-4,14,21-triazahexacy...)Show SMILES OC[C@@H]1CCn2cc(C3=C(C(=O)NC3=O)c3cn(CCO1)c1ccccc31)c1ccccc21 |t:8| Show InChI InChI=1S/C26H23N3O4/c30-15-16-9-10-28-13-19(17-5-1-3-7-21(17)28)23-24(26(32)27-25(23)31)20-14-29(11-12-33-16)22-8-4-2-6-18(20)22/h1-8,13-14,16,30H,9-12,15H2,(H,27,31,32)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 2 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C delta type
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C delta |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50052041

(18-(4-methylhexahydro-1-pyrazinylmethyl)-(18S)-17-...)Show SMILES CN1CCN(C[C@@H]2CCn3cc(C4=C(C(=O)NC4=O)c4cn(CCO2)c2ccccc42)c2ccccc32)CC1 |t:12| Show InChI InChI=1S/C31H33N5O3/c1-33-12-14-34(15-13-33)18-21-10-11-35-19-24(22-6-2-4-8-26(22)35)28-29(31(38)32-30(28)37)25-20-36(16-17-39-21)27-9-5-3-7-23(25)27/h2-9,19-21H,10-18H2,1H3,(H,32,37,38)/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 1 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C eta type
(Homo sapiens (Human)) | BDBM50052034

(18-methylaminomethyl-(18S)-17-oxa-4,14,21-triazahe...)Show SMILES CNC[C@@H]1CCn2cc(C3=C(C(=O)NC3=O)c3cn(CCO1)c1ccccc31)c1ccccc21 |t:9| Show InChI InChI=1S/C27H26N4O3/c1-28-14-17-10-11-30-15-20(18-6-2-4-8-22(18)30)24-25(27(33)29-26(24)32)21-16-31(12-13-34-17)23-9-5-3-7-19(21)23/h2-9,15-17,28H,10-14H2,1H3,(H,29,32,33)/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C eta |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50052039

(18-hydroxymethyl-(18S)-17-oxa-4,14,21-triazahexacy...)Show SMILES OC[C@@H]1CCn2cc(C3=C(C(=O)NC3=O)c3cn(CCO1)c1ccccc31)c1ccccc21 |t:8| Show InChI InChI=1S/C26H23N3O4/c30-15-16-9-10-28-13-19(17-5-1-3-7-21(17)28)23-24(26(32)27-25(23)31)20-14-29(11-12-33-16)22-8-4-2-6-18(20)22/h1-8,13-14,16,30H,9-12,15H2,(H,27,31,32)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 32 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 2 |
Bioorg Med Chem Lett 5: 2093-2096 (1995)
Article DOI: 10.1016/0960-894X(95)00350-3
BindingDB Entry DOI: 10.7270/Q2D50MXM |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50052037
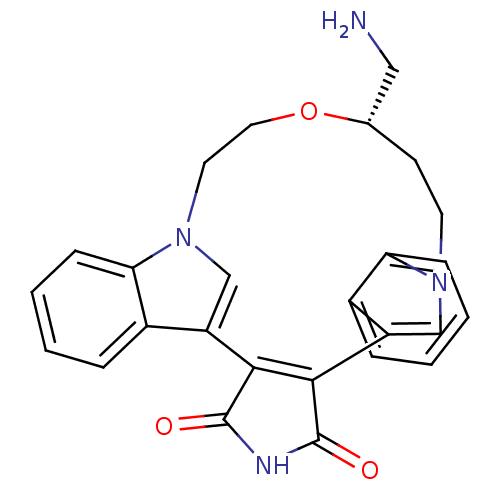
(18-aminomethyl-(18S)-17-oxa-4,14,21-triazahexacycl...)Show SMILES NC[C@@H]1CCn2cc(C3=C(C(=O)NC3=O)c3cn(CCO1)c1ccccc31)c1ccccc21 |t:8| Show InChI InChI=1S/C26H24N4O3/c27-13-16-9-10-29-14-19(17-5-1-3-7-21(17)29)23-24(26(32)28-25(23)31)20-15-30(11-12-33-16)22-8-4-2-6-18(20)22/h1-8,14-16H,9-13,27H2,(H,28,31,32)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 2 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50052035
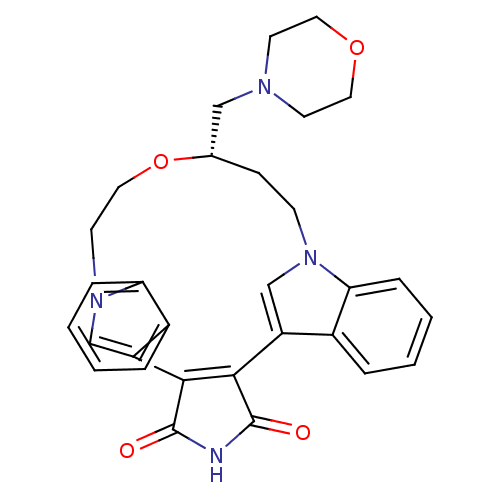
(18-(1,4-oxazinan-4-ylmethyl)-(18S)-17-oxa-4,14,21-...)Show SMILES O=C1NC(=O)C2=C1c1cn(CCO[C@H](CN3CCOCC3)CCn3cc2c2ccccc32)c2ccccc12 |c:5| Show InChI InChI=1S/C30H30N4O4/c35-29-27-23-18-33(25-7-3-1-5-21(23)25)10-9-20(17-32-11-14-37-15-12-32)38-16-13-34-19-24(28(27)30(36)31-29)22-6-2-4-8-26(22)34/h1-8,18-20H,9-17H2,(H,31,35,36)/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 2 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50052036

(3,5-dioxo-17-phenylsulfonamidomethyl-(17S)-18-oxa-...)Show SMILES O=C1NC(=O)C2=C1c1cn(CCO[C@H](CNS(=O)(=O)c3ccccc3)CCn3cc2c2ccccc32)c2ccccc12 |c:5| Show InChI InChI=1S/C32H28N4O5S/c37-31-29-25-19-35(27-12-6-4-10-23(25)27)15-14-21(18-33-42(39,40)22-8-2-1-3-9-22)41-17-16-36-20-26(30(29)32(38)34-31)24-11-5-7-13-28(24)36/h1-13,19-21,33H,14-18H2,(H,34,37,38)/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 2 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50052038

(18-tetrahydro-1H-1-pyrrolylmethyl-(18S)-17-oxa-4,1...)Show SMILES O=C1NC(=O)C2=C1c1cn(CCO[C@H](CN3CCCC3)CCn3cc2c2ccccc32)c2ccccc12 |c:5| Show InChI InChI=1S/C30H30N4O3/c35-29-27-23-18-33(25-9-3-1-7-21(23)25)14-11-20(17-32-12-5-6-13-32)37-16-15-34-19-24(28(27)30(36)31-29)22-8-2-4-10-26(22)34/h1-4,7-10,18-20H,5-6,11-17H2,(H,31,35,36)/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 2 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50052038

(18-tetrahydro-1H-1-pyrrolylmethyl-(18S)-17-oxa-4,1...)Show SMILES O=C1NC(=O)C2=C1c1cn(CCO[C@H](CN3CCCC3)CCn3cc2c2ccccc32)c2ccccc12 |c:5| Show InChI InChI=1S/C30H30N4O3/c35-29-27-23-18-33(25-9-3-1-7-21(23)25)14-11-20(17-32-12-5-6-13-32)37-16-15-34-19-24(28(27)30(36)31-29)22-8-2-4-10-26(22)34/h1-4,7-10,18-20H,5-6,11-17H2,(H,31,35,36)/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 44 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 2 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM2579

((2S,3R,4R,6R)-3-methoxy-2-methyl-4-(methylamino)-2...)Show SMILES CN[C@@H]1C[C@H]2O[C@@](C)([C@@H]1OC)n1c3ccccc3c3c4CNC(=O)c4c4c5ccccc5n2c4c13 |r| Show InChI InChI=1S/C28H26N4O3/c1-28-26(34-3)17(29-2)12-20(35-28)31-18-10-6-4-8-14(18)22-23-16(13-30-27(23)33)21-15-9-5-7-11-19(15)32(28)25(21)24(22)31/h4-11,17,20,26,29H,12-13H2,1-3H3,(H,30,33)/t17-,20-,26-,28+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 45 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C alpha |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |
Protein kinase C beta type
(Homo sapiens (Human)) | BDBM50052037

(18-aminomethyl-(18S)-17-oxa-4,14,21-triazahexacycl...)Show SMILES NC[C@@H]1CCn2cc(C3=C(C(=O)NC3=O)c3cn(CCO1)c1ccccc31)c1ccccc21 |t:8| Show InChI InChI=1S/C26H24N4O3/c27-13-16-9-10-29-14-19(17-5-1-3-7-21(17)29)23-24(26(32)28-25(23)31)20-15-30(11-12-33-16)22-8-4-2-6-18(20)22/h1-8,14-16H,9-13,27H2,(H,28,31,32)/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of Protein kinase C beta 1 |
J Med Chem 39: 2664-71 (1996)
Article DOI: 10.1021/jm950588y
BindingDB Entry DOI: 10.7270/Q25H7FBV |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data