 Found 128 hits with Last Name = 'hammer' and Initial = 'j'
Found 128 hits with Last Name = 'hammer' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260806
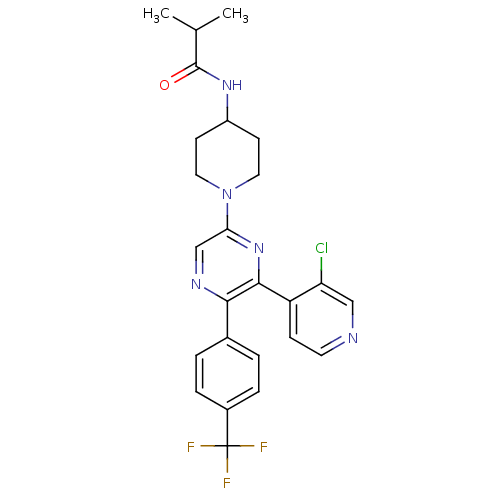
(CHEMBL497557 | N-(1-(6-(3-chloropyridin-4-yl)-5-(4...)Show SMILES CC(C)C(=O)NC1CCN(CC1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccncc1Cl Show InChI InChI=1S/C25H25ClF3N5O/c1-15(2)24(35)32-18-8-11-34(12-9-18)21-14-31-22(16-3-5-17(6-4-16)25(27,28)29)23(33-21)19-7-10-30-13-20(19)26/h3-7,10,13-15,18H,8-9,11-12H2,1-2H3,(H,32,35) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Rattus norvegicus (rat)) | BDBM21278

(5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl...)Show SMILES Cc1c(nn(c1-c1ccc(Cl)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C22H21Cl3N4O/c1-14-20(22(30)27-28-11-3-2-4-12-28)26-29(19-10-9-17(24)13-18(19)25)21(14)15-5-7-16(23)8-6-15/h5-10,13H,2-4,11-12H2,1H3,(H,27,30) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at rat CB1 receptor in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Rattus norvegicus (rat)) | BDBM50260806

(CHEMBL497557 | N-(1-(6-(3-chloropyridin-4-yl)-5-(4...)Show SMILES CC(C)C(=O)NC1CCN(CC1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccncc1Cl Show InChI InChI=1S/C25H25ClF3N5O/c1-15(2)24(35)32-18-8-11-34(12-9-18)21-14-31-22(16-3-5-17(6-4-16)25(27,28)29)23(33-21)19-7-10-30-13-20(19)26/h3-7,10,13-15,18H,8-9,11-12H2,1-2H3,(H,32,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at rat CB1 receptor in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM21278

(5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl...)Show SMILES Cc1c(nn(c1-c1ccc(Cl)cc1)-c1ccc(Cl)cc1Cl)C(=O)NN1CCCCC1 Show InChI InChI=1S/C22H21Cl3N4O/c1-14-20(22(30)27-28-11-3-2-4-12-28)26-29(19-10-9-17(24)13-18(19)25)21(14)15-5-7-16(23)8-6-15/h5-10,13H,2-4,11-12H2,1H3,(H,27,30) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260767
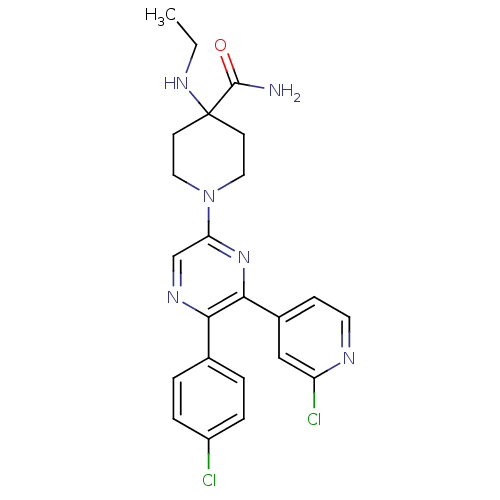
(1-(5-(4-chlorophenyl)-6-(2-chloropyridin-4-yl)pyra...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(Cl)cc2)c(n1)-c1ccnc(Cl)c1)C(N)=O Show InChI InChI=1S/C23H24Cl2N6O/c1-2-29-23(22(26)32)8-11-31(12-9-23)19-14-28-20(15-3-5-17(24)6-4-15)21(30-19)16-7-10-27-18(25)13-16/h3-7,10,13-14,29H,2,8-9,11-12H2,1H3,(H2,26,32) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.670 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260805
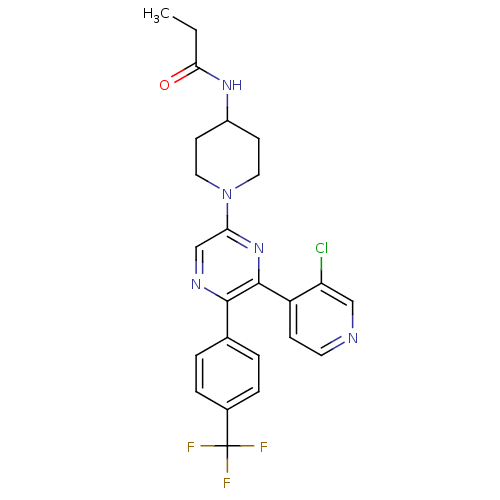
(CHEMBL524804 | N-(1-(6-(3-chloropyridin-4-yl)-5-(4...)Show SMILES CCC(=O)NC1CCN(CC1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccncc1Cl Show InChI InChI=1S/C24H23ClF3N5O/c1-2-21(34)31-17-8-11-33(12-9-17)20-14-30-22(15-3-5-16(6-4-15)24(26,27)28)23(32-20)18-7-10-29-13-19(18)25/h3-7,10,13-14,17H,2,8-9,11-12H2,1H3,(H,31,34) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Rattus norvegicus (rat)) | BDBM50260681
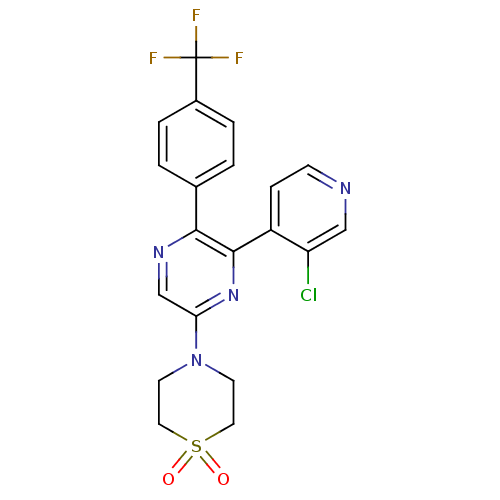
(4-[6-(3-Chloro-pyridin-4-yl)-5-(4-trifluoromethyl-...)Show SMILES FC(F)(F)c1ccc(cc1)-c1ncc(nc1-c1ccncc1Cl)N1CCS(=O)(=O)CC1 Show InChI InChI=1S/C20H16ClF3N4O2S/c21-16-11-25-6-5-15(16)19-18(13-1-3-14(4-2-13)20(22,23)24)26-12-17(27-19)28-7-9-31(29,30)10-8-28/h1-6,11-12H,7-10H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.830 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at rat CB1 receptor in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260681

(4-[6-(3-Chloro-pyridin-4-yl)-5-(4-trifluoromethyl-...)Show SMILES FC(F)(F)c1ccc(cc1)-c1ncc(nc1-c1ccncc1Cl)N1CCS(=O)(=O)CC1 Show InChI InChI=1S/C20H16ClF3N4O2S/c21-16-11-25-6-5-15(16)19-18(13-1-3-14(4-2-13)20(22,23)24)26-12-17(27-19)28-7-9-31(29,30)10-8-28/h1-6,11-12H,7-10H2 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.830 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260768
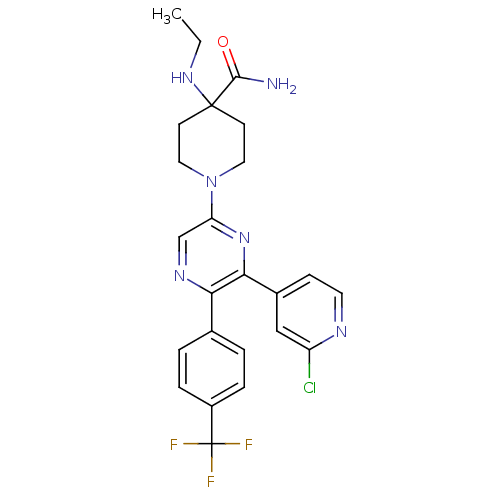
(1-(6-(2-chloropyridin-4-yl)-5-(4-(trifluoromethyl)...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccnc(Cl)c1)C(N)=O Show InChI InChI=1S/C24H24ClF3N6O/c1-2-32-23(22(29)35)8-11-34(12-9-23)19-14-31-20(15-3-5-17(6-4-15)24(26,27)28)21(33-19)16-7-10-30-18(25)13-16/h3-7,10,13-14,32H,2,8-9,11-12H2,1H3,(H2,29,35) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260682
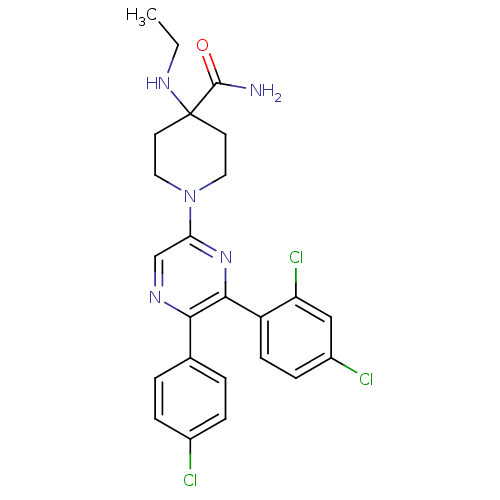
(1-(5-(4-chlorophenyl)-6-(2,4-dichlorophenyl)pyrazi...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(Cl)cc2)c(n1)-c1ccc(Cl)cc1Cl)C(N)=O Show InChI InChI=1S/C24H24Cl3N5O/c1-2-30-24(23(28)33)9-11-32(12-10-24)20-14-29-21(15-3-5-16(25)6-4-15)22(31-20)18-8-7-17(26)13-19(18)27/h3-8,13-14,30H,2,9-12H2,1H3,(H2,28,33) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.910 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260803
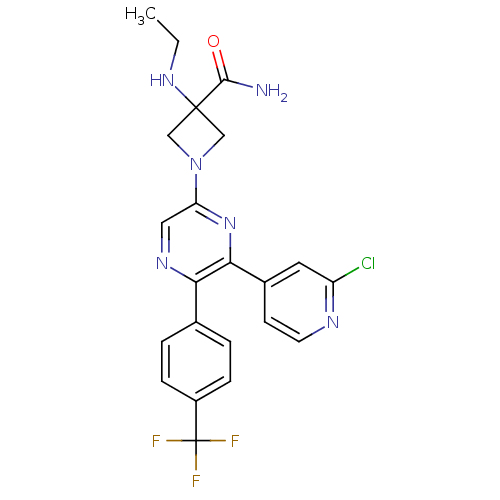
(1-(6-(2-chloropyridin-4-yl)-5-(4-(trifluoromethyl)...)Show SMILES CCNC1(CN(C1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccnc(Cl)c1)C(N)=O Show InChI InChI=1S/C22H20ClF3N6O/c1-2-30-21(20(27)33)11-32(12-21)17-10-29-18(13-3-5-15(6-4-13)22(24,25)26)19(31-17)14-7-8-28-16(23)9-14/h3-10,30H,2,11-12H2,1H3,(H2,27,33) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50092053
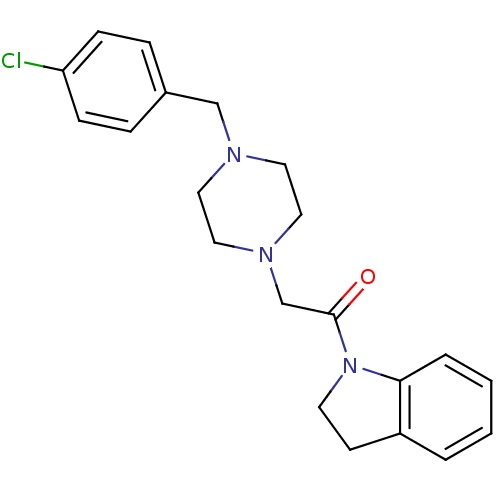
(2-[4-(4-Chloro-benzyl)-piperazin-1-yl]-1-(2,3-dihy...)Show InChI InChI=1S/C21H24ClN3O/c22-19-7-5-17(6-8-19)15-23-11-13-24(14-12-23)16-21(26)25-10-9-18-3-1-2-4-20(18)25/h1-8H,9-16H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D4 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260718

(1-(6-(2-chlorophenyl)-5-(4-chlorophenyl)pyrazin-2-...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(Cl)cc2)c(n1)-c1ccccc1Cl)C(N)=O Show InChI InChI=1S/C24H25Cl2N5O/c1-2-29-24(23(27)32)11-13-31(14-12-24)20-15-28-21(16-7-9-17(25)10-8-16)22(30-20)18-5-3-4-6-19(18)26/h3-10,15,29H,2,11-14H2,1H3,(H2,27,32) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.66 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260717
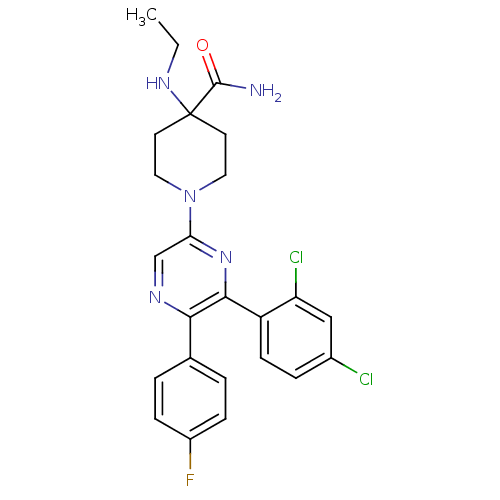
(1-(6-(2,4-dichlorophenyl)-5-(4-fluorophenyl)pyrazi...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(F)cc2)c(n1)-c1ccc(Cl)cc1Cl)C(N)=O Show InChI InChI=1S/C24H24Cl2FN5O/c1-2-30-24(23(28)33)9-11-32(12-10-24)20-14-29-21(15-3-6-17(27)7-4-15)22(31-20)18-8-5-16(25)13-19(18)26/h3-8,13-14,30H,2,9-12H2,1H3,(H2,28,33) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260804
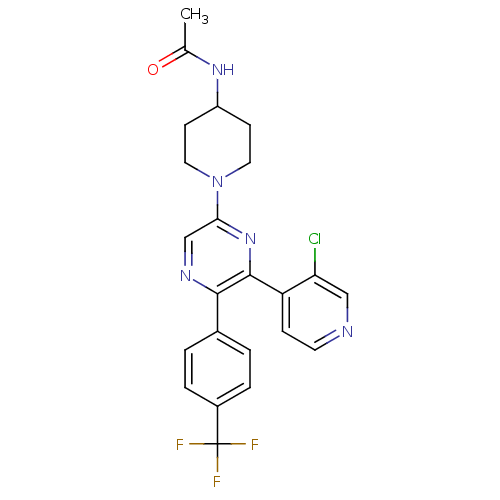
(CHEMBL497556 | N-(1-(6-(3-chloropyridin-4-yl)-5-(4...)Show SMILES CC(=O)NC1CCN(CC1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccncc1Cl Show InChI InChI=1S/C23H21ClF3N5O/c1-14(33)30-17-7-10-32(11-8-17)20-13-29-21(15-2-4-16(5-3-15)23(25,26)27)22(31-20)18-6-9-28-12-19(18)24/h2-6,9,12-13,17H,7-8,10-11H2,1H3,(H,30,33) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.97 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Rattus norvegicus (rat)) | BDBM50260803

(1-(6-(2-chloropyridin-4-yl)-5-(4-(trifluoromethyl)...)Show SMILES CCNC1(CN(C1)c1cnc(-c2ccc(cc2)C(F)(F)F)c(n1)-c1ccnc(Cl)c1)C(N)=O Show InChI InChI=1S/C22H20ClF3N6O/c1-2-30-21(20(27)33)11-32(12-21)17-10-29-18(13-3-5-15(6-4-13)22(24,25)26)19(31-17)14-7-8-28-16(23)9-14/h3-10,30H,2,11-12H2,1H3,(H2,27,33) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.06 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at rat CB1 receptor in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260720
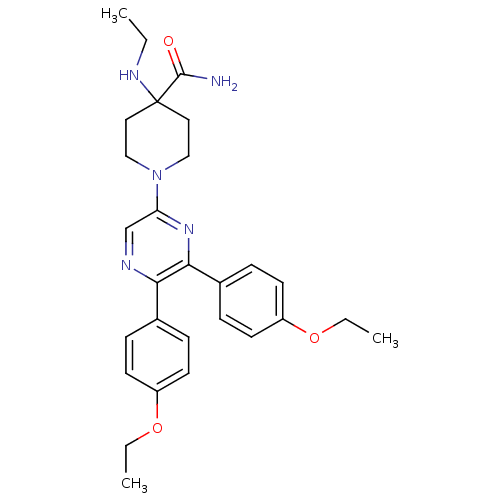
(1-(5,6-bis(4-ethoxyphenyl)pyrazin-2-yl)-4-(ethylam...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(OCC)cc2)c(n1)-c1ccc(OCC)cc1)C(N)=O Show InChI InChI=1S/C28H35N5O3/c1-4-31-28(27(29)34)15-17-33(18-16-28)24-19-30-25(20-7-11-22(12-8-20)35-5-2)26(32-24)21-9-13-23(14-10-21)36-6-3/h7-14,19,31H,4-6,15-18H2,1-3H3,(H2,29,34) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.84 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50124931
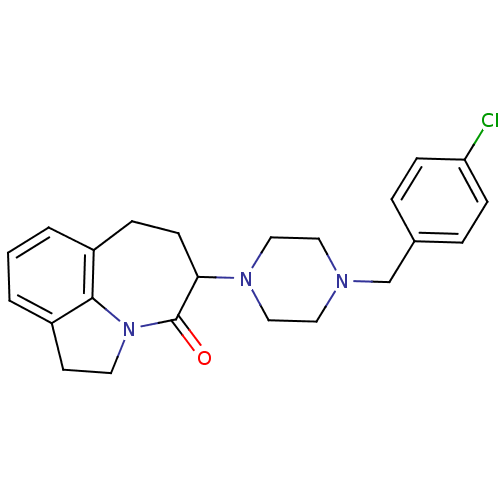
(5-[4-(4-Chloro-benzyl)-piperazin-1-yl]-1,2,6,7-tet...)Show SMILES Clc1ccc(CN2CCN(CC2)C2CCc3cccc4CCN(c34)C2=O)cc1 Show InChI InChI=1S/C23H26ClN3O/c24-20-7-4-17(5-8-20)16-25-12-14-26(15-13-25)21-9-6-18-2-1-3-19-10-11-27(22(18)19)23(21)28/h1-5,7-8,21H,6,9-16H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
D4 receptor functional activity was measured inhibition of quinpirole stimulated [35S]GTP-gamma-S binding from cell membranes. |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50092046
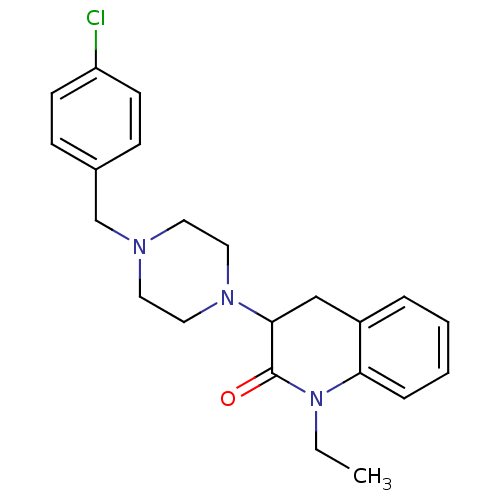
(3-[4-(4-Chloro-benzyl)-piperazin-1-yl]-1-ethyl-3,4...)Show SMILES CCN1C(=O)C(Cc2ccccc12)N1CCN(Cc2ccc(Cl)cc2)CC1 Show InChI InChI=1S/C22H26ClN3O/c1-2-26-20-6-4-3-5-18(20)15-21(22(26)27)25-13-11-24(12-14-25)16-17-7-9-19(23)10-8-17/h3-10,21H,2,11-16H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D4 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50124932

(5-[4-(4-Methyl-benzyl)-piperazin-1-yl]-1,2,6,7-tet...)Show SMILES Cc1ccc(CN2CCN(CC2)C2CCc3cccc4CCN(c34)C2=O)cc1 Show InChI InChI=1S/C24H29N3O/c1-18-5-7-19(8-6-18)17-25-13-15-26(16-14-25)22-10-9-20-3-2-4-21-11-12-27(23(20)21)24(22)28/h2-8,22H,9-17H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D4 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50001884
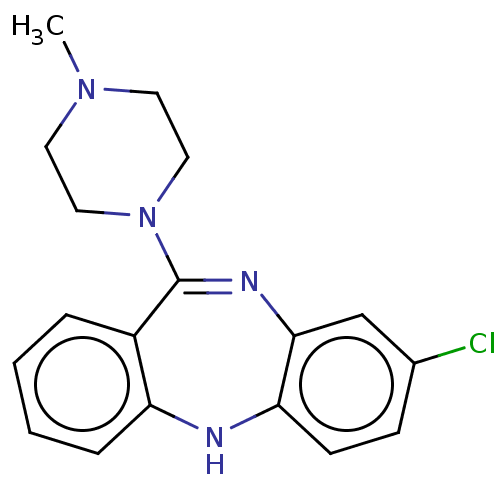
(2-[4-(4-Methyl-benzyl)-piperazin-1-yl]-1-(2-methyl...)Show SMILES CN1CCN(CC1)C1=Nc2cc(Cl)ccc2Nc2ccccc12 |t:8| Show InChI InChI=1S/C18H19ClN4/c1-22-8-10-23(11-9-22)18-14-4-2-3-5-15(14)20-16-7-6-13(19)12-17(16)21-18/h2-7,12,20H,8-11H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]prazosin from alpha-1 adrenergic receptor of rat brain homogenate |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50124931

(5-[4-(4-Chloro-benzyl)-piperazin-1-yl]-1,2,6,7-tet...)Show SMILES Clc1ccc(CN2CCN(CC2)C2CCc3cccc4CCN(c34)C2=O)cc1 Show InChI InChI=1S/C23H26ClN3O/c24-20-7-4-17(5-8-20)16-25-12-14-26(15-13-25)21-9-6-18-2-1-3-19-10-11-27(22(18)19)23(21)28/h1-5,7-8,21H,6,9-16H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D4 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50124934
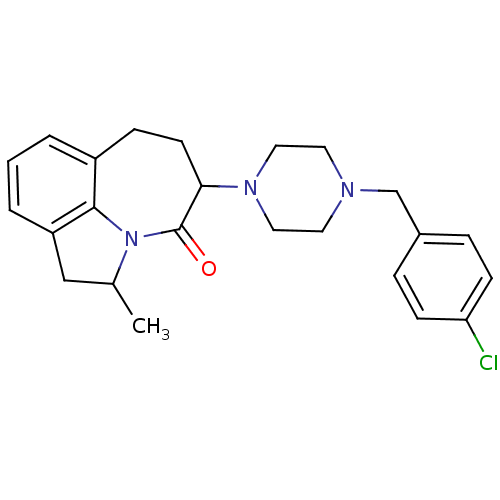
(5-[4-(4-Chloro-benzyl)-piperazin-1-yl]-2-methyl-1,...)Show SMILES CC1Cc2cccc3CCC(N4CCN(Cc5ccc(Cl)cc5)CC4)C(=O)N1c23 Show InChI InChI=1S/C24H28ClN3O/c1-17-15-20-4-2-3-19-7-10-22(24(29)28(17)23(19)20)27-13-11-26(12-14-27)16-18-5-8-21(25)9-6-18/h2-6,8-9,17,22H,7,10-16H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D4 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260766
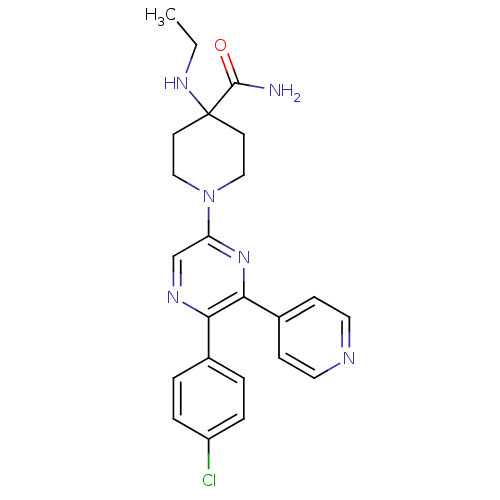
(1-(5-(4-chlorophenyl)-6-(pyridin-4-yl)pyrazin-2-yl...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(Cl)cc2)c(n1)-c1ccncc1)C(N)=O Show InChI InChI=1S/C23H25ClN6O/c1-2-28-23(22(25)31)9-13-30(14-10-23)19-15-27-20(16-3-5-18(24)6-4-16)21(29-19)17-7-11-26-12-8-17/h3-8,11-12,15,28H,2,9-10,13-14H2,1H3,(H2,25,31) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.81 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50124935
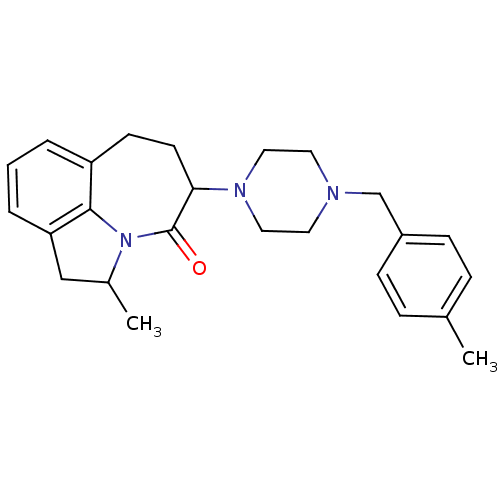
(2-Methyl-5-[4-(4-methyl-benzyl)-piperazin-1-yl]-1,...)Show SMILES CC1Cc2cccc3CCC(N4CCN(Cc5ccc(C)cc5)CC4)C(=O)N1c23 Show InChI InChI=1S/C25H31N3O/c1-18-6-8-20(9-7-18)17-26-12-14-27(15-13-26)23-11-10-21-4-3-5-22-16-19(2)28(24(21)22)25(23)29/h3-9,19,23H,10-17H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D4 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50124936
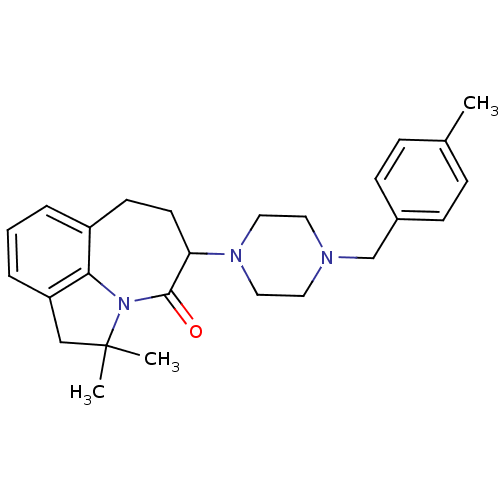
(2,2-Dimethyl-5-[4-(4-methyl-benzyl)-piperazin-1-yl...)Show SMILES Cc1ccc(CN2CCN(CC2)C2CCc3cccc4CC(C)(C)N(c34)C2=O)cc1 Show InChI InChI=1S/C26H33N3O/c1-19-7-9-20(10-8-19)18-27-13-15-28(16-14-27)23-12-11-21-5-4-6-22-17-26(2,3)29(24(21)22)25(23)30/h4-10,23H,11-18H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D4 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260719
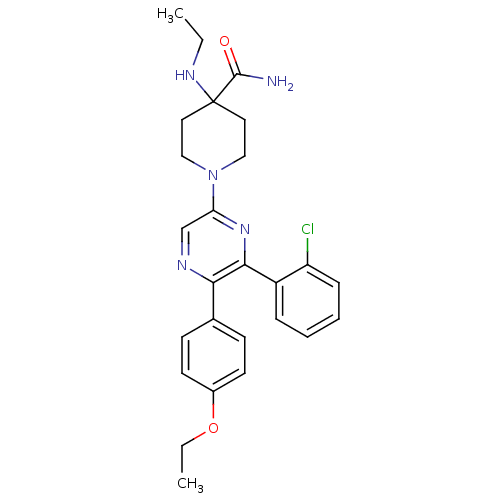
(1-(6-(2-chlorophenyl)-5-(4-ethoxyphenyl)pyrazin-2-...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(OCC)cc2)c(n1)-c1ccccc1Cl)C(N)=O Show InChI InChI=1S/C26H30ClN5O2/c1-3-30-26(25(28)33)13-15-32(16-14-26)22-17-29-23(18-9-11-19(12-10-18)34-4-2)24(31-22)20-7-5-6-8-21(20)27/h5-12,17,30H,3-4,13-16H2,1-2H3,(H2,28,33) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 15.0 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50001884

(2-[4-(4-Methyl-benzyl)-piperazin-1-yl]-1-(2-methyl...)Show SMILES CN1CCN(CC1)C1=Nc2cc(Cl)ccc2Nc2ccccc12 |t:8| Show InChI InChI=1S/C18H19ClN4/c1-22-8-10-23(11-9-22)18-14-4-2-3-5-15(14)20-16-7-6-13(19)12-17(16)21-18/h2-7,12,20H,8-11H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PubMed
| 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D4 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50124937
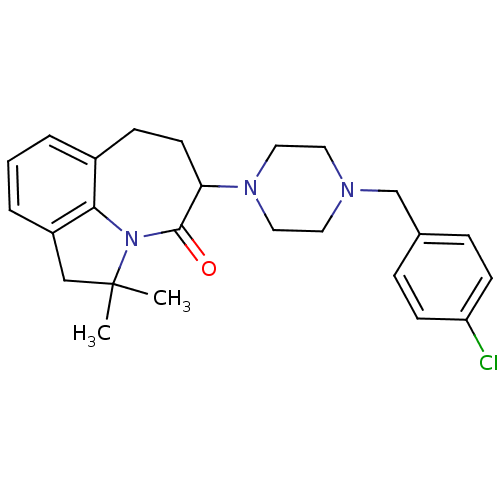
(5-[4-(4-Chloro-benzyl)-piperazin-1-yl]-2,2-dimethy...)Show SMILES CC1(C)Cc2cccc3CCC(N4CCN(Cc5ccc(Cl)cc5)CC4)C(=O)N1c23 Show InChI InChI=1S/C25H30ClN3O/c1-25(2)16-20-5-3-4-19-8-11-22(24(30)29(25)23(19)20)28-14-12-27(13-15-28)17-18-6-9-21(26)10-7-18/h3-7,9-10,22H,8,11-17H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D4 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50092046

(3-[4-(4-Chloro-benzyl)-piperazin-1-yl]-1-ethyl-3,4...)Show SMILES CCN1C(=O)C(Cc2ccccc12)N1CCN(Cc2ccc(Cl)cc2)CC1 Show InChI InChI=1S/C22H26ClN3O/c1-2-26-20-6-4-3-5-18(20)15-21(22(26)27)25-13-11-24(12-14-25)16-17-7-9-19(23)10-8-17/h3-10,21H,2,11-16H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D2 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50124935

(2-Methyl-5-[4-(4-methyl-benzyl)-piperazin-1-yl]-1,...)Show SMILES CC1Cc2cccc3CCC(N4CCN(Cc5ccc(C)cc5)CC4)C(=O)N1c23 Show InChI InChI=1S/C25H31N3O/c1-18-6-8-20(9-7-18)17-26-12-14-27(15-13-26)23-11-10-21-4-3-5-22-16-19(2)28(24(21)22)25(23)29/h3-9,19,23H,10-17H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 26 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D2 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50124938
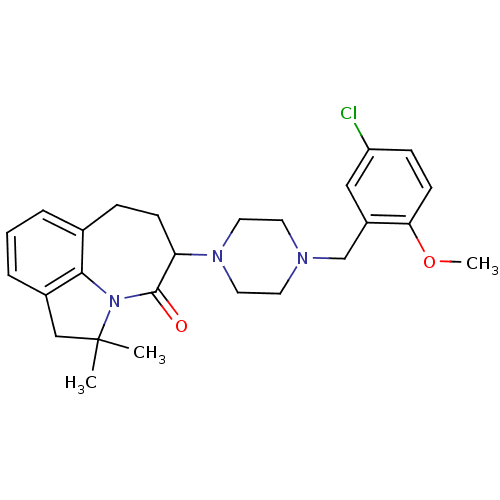
(5-[4-(5-Chloro-2-methoxy-benzyl)-piperazin-1-yl]-2...)Show SMILES COc1ccc(Cl)cc1CN1CCN(CC1)C1CCc2cccc3CC(C)(C)N(c23)C1=O Show InChI InChI=1S/C26H32ClN3O2/c1-26(2)16-19-6-4-5-18-7-9-22(25(31)30(26)24(18)19)29-13-11-28(12-14-29)17-20-15-21(27)8-10-23(20)32-3/h4-6,8,10,15,22H,7,9,11-14,16-17H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 29 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D4 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50260765
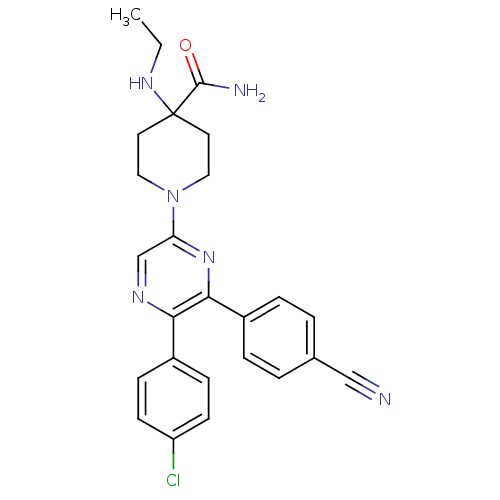
(1-(5-(4-chlorophenyl)-6-(4-cyanophenyl)pyrazin-2-y...)Show SMILES CCNC1(CCN(CC1)c1cnc(-c2ccc(Cl)cc2)c(n1)-c1ccc(cc1)C#N)C(N)=O Show InChI InChI=1S/C25H25ClN6O/c1-2-30-25(24(28)33)11-13-32(14-12-25)21-16-29-22(18-7-9-20(26)10-8-18)23(31-21)19-5-3-17(15-27)4-6-19/h3-10,16,30H,2,11-14H2,1H3,(H2,28,33) | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 32.3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Antagonist activity at human CB1 receptor expressed in SF9 cells assessed as inhibition of CP-55940-stimulated GTPgammaS binding |
Bioorg Med Chem Lett 18: 3376-81 (2008)
Article DOI: 10.1016/j.bmcl.2008.04.022
BindingDB Entry DOI: 10.7270/Q2QR4Z0T |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50124933
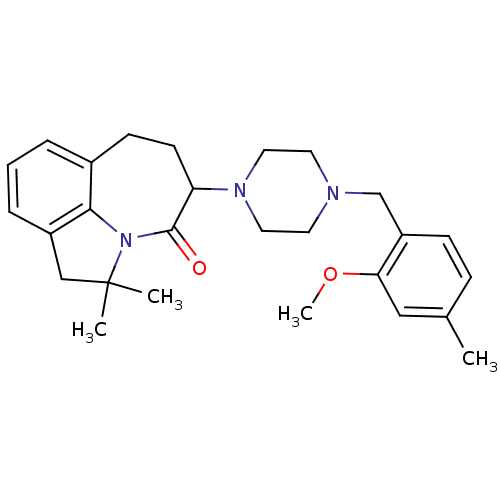
(5-[4-(2-Methoxy-4-methyl-benzyl)-piperazin-1-yl]-2...)Show SMILES COc1cc(C)ccc1CN1CCN(CC1)C1CCc2cccc3CC(C)(C)N(c23)C1=O Show InChI InChI=1S/C27H35N3O2/c1-19-8-9-22(24(16-19)32-4)18-28-12-14-29(15-13-28)23-11-10-20-6-5-7-21-17-27(2,3)30(25(20)21)26(23)31/h5-9,16,23H,10-15,17-18H2,1-4H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 34 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D4 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50124931

(5-[4-(4-Chloro-benzyl)-piperazin-1-yl]-1,2,6,7-tet...)Show SMILES Clc1ccc(CN2CCN(CC2)C2CCc3cccc4CCN(c34)C2=O)cc1 Show InChI InChI=1S/C23H26ClN3O/c24-20-7-4-17(5-8-20)16-25-12-14-26(15-13-25)21-9-6-18-2-1-3-19-10-11-27(22(18)19)23(21)28/h1-5,7-8,21H,6,9-16H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 62 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
D2 receptor functional activity was measured through reversal of quinpirole inhibited, forskolin stimulated cAMP production from whole cells |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(4) dopamine receptor
(Homo sapiens (Human)) | BDBM50124939
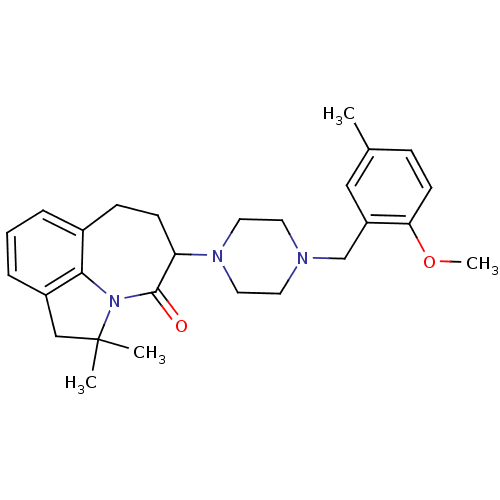
(5-[4-(2-Methoxy-5-methyl-benzyl)-piperazin-1-yl]-2...)Show SMILES COc1ccc(C)cc1CN1CCN(CC1)C1CCc2cccc3CC(C)(C)N(c23)C1=O Show InChI InChI=1S/C27H35N3O2/c1-19-8-11-24(32-4)22(16-19)18-28-12-14-29(15-13-28)23-10-9-20-6-5-7-21-17-27(2,3)30(25(20)21)26(23)31/h5-8,11,16,23H,9-10,12-15,17-18H2,1-4H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D4 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50092053

(2-[4-(4-Chloro-benzyl)-piperazin-1-yl]-1-(2,3-dihy...)Show InChI InChI=1S/C21H24ClN3O/c22-19-7-5-17(6-8-19)15-23-11-13-24(14-12-23)16-21(26)25-10-9-18-3-1-2-4-20(18)25/h1-8H,9-16H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 88 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]prazosin from alpha-1 adrenergic receptor of rat brain homogenate |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50001884

(2-[4-(4-Methyl-benzyl)-piperazin-1-yl]-1-(2-methyl...)Show SMILES CN1CCN(CC1)C1=Nc2cc(Cl)ccc2Nc2ccccc12 |t:8| Show InChI InChI=1S/C18H19ClN4/c1-22-8-10-23(11-9-22)18-14-4-2-3-5-15(14)20-16-7-6-13(19)12-17(16)21-18/h2-7,12,20H,8-11H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PubMed
| 113 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D2 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50124931

(5-[4-(4-Chloro-benzyl)-piperazin-1-yl]-1,2,6,7-tet...)Show SMILES Clc1ccc(CN2CCN(CC2)C2CCc3cccc4CCN(c34)C2=O)cc1 Show InChI InChI=1S/C23H26ClN3O/c24-20-7-4-17(5-8-20)16-25-12-14-26(15-13-25)21-9-6-18-2-1-3-19-10-11-27(22(18)19)23(21)28/h1-5,7-8,21H,6,9-16H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 116 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D2 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50124934

(5-[4-(4-Chloro-benzyl)-piperazin-1-yl]-2-methyl-1,...)Show SMILES CC1Cc2cccc3CCC(N4CCN(Cc5ccc(Cl)cc5)CC4)C(=O)N1c23 Show InChI InChI=1S/C24H28ClN3O/c1-17-15-20-4-2-3-19-7-10-22(24(29)28(17)23(19)20)27-13-11-26(12-14-27)16-18-5-8-21(25)9-6-18/h2-6,8-9,17,22H,7,10-16H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 139 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D2 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50124936

(2,2-Dimethyl-5-[4-(4-methyl-benzyl)-piperazin-1-yl...)Show SMILES Cc1ccc(CN2CCN(CC2)C2CCc3cccc4CC(C)(C)N(c34)C2=O)cc1 Show InChI InChI=1S/C26H33N3O/c1-19-7-9-20(10-8-19)18-27-13-15-28(16-14-27)23-12-11-21-5-4-6-22-17-26(2,3)29(24(21)22)25(23)30/h4-10,23H,11-18H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 165 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D2 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50124937

(5-[4-(4-Chloro-benzyl)-piperazin-1-yl]-2,2-dimethy...)Show SMILES CC1(C)Cc2cccc3CCC(N4CCN(Cc5ccc(Cl)cc5)CC4)C(=O)N1c23 Show InChI InChI=1S/C25H30ClN3O/c1-25(2)16-20-5-3-4-19-8-11-22(24(30)29(25)23(19)20)28-14-12-27(13-15-28)17-18-6-9-21(26)10-7-18/h3-7,9-10,22H,8,11-17H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 201 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D2 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50124932

(5-[4-(4-Methyl-benzyl)-piperazin-1-yl]-1,2,6,7-tet...)Show SMILES Cc1ccc(CN2CCN(CC2)C2CCc3cccc4CCN(c34)C2=O)cc1 Show InChI InChI=1S/C24H29N3O/c1-18-5-7-19(8-6-18)17-25-13-15-26(16-14-25)22-10-9-20-3-2-4-21-11-12-27(23(20)21)24(22)28/h2-8,22H,9-17H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 209 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D2 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50124938

(5-[4-(5-Chloro-2-methoxy-benzyl)-piperazin-1-yl]-2...)Show SMILES COc1ccc(Cl)cc1CN1CCN(CC1)C1CCc2cccc3CC(C)(C)N(c23)C1=O Show InChI InChI=1S/C26H32ClN3O2/c1-26(2)16-19-6-4-5-18-7-9-22(25(31)30(26)24(18)19)29-13-11-28(12-14-29)17-20-15-21(27)8-10-23(20)32-3/h4-6,8,10,15,22H,7,9,11-14,16-17H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D2 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
D(2) dopamine receptor
(Homo sapiens (Human)) | BDBM50124939

(5-[4-(2-Methoxy-5-methyl-benzyl)-piperazin-1-yl]-2...)Show SMILES COc1ccc(C)cc1CN1CCN(CC1)C1CCc2cccc3CC(C)(C)N(c23)C1=O Show InChI InChI=1S/C27H35N3O2/c1-19-8-11-24(32-4)22(16-19)18-28-12-14-29(15-13-28)23-10-9-20-6-5-7-21-17-27(2,3)30(25(20)21)26(23)31/h5-8,11,16,23H,9-10,12-15,17-18H2,1-4H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 313 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]-YM 09151 from D2 receptor |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
Suppressor of tumorigenicity 14 protein
(Homo sapiens (Human)) | BDBM50615186
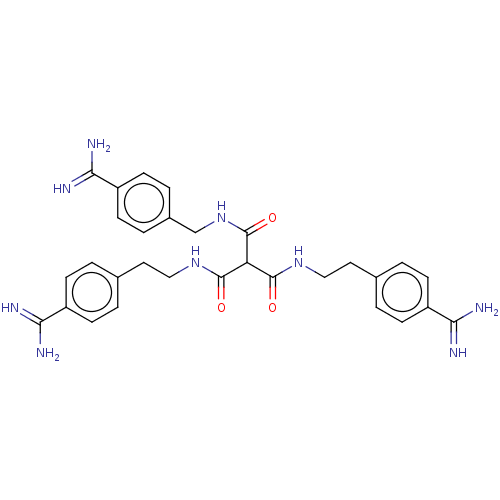
(CHEMBL5282514)Show SMILES NC(=N)c1ccc(CCNC(=O)C(C(=O)NCCc2ccc(cc2)C(N)=N)C(=O)NCc2ccc(cc2)C(N)=N)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 393 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Suppressor of tumorigenicity 14 protein
(Homo sapiens (Human)) | BDBM50615190
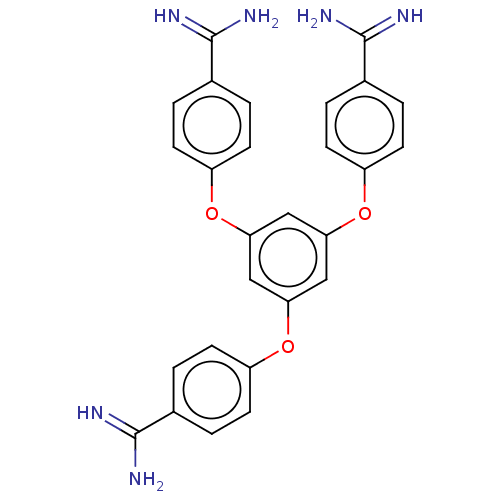
(CHEMBL5269464)Show SMILES NC(=N)c1ccc(Oc2cc(Oc3ccc(cc3)C(N)=N)cc(Oc3ccc(cc3)C(N)=N)c2)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 398 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Suppressor of tumorigenicity 14 protein
(Homo sapiens (Human)) | BDBM50615189

(CHEMBL5279198)Show SMILES NCc1ccc(Oc2cc(Oc3ccc(cc3)C(N)=N)cc(Oc3ccc(cc3)C(N)=N)c2)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 459 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50124936

(2,2-Dimethyl-5-[4-(4-methyl-benzyl)-piperazin-1-yl...)Show SMILES Cc1ccc(CN2CCN(CC2)C2CCc3cccc4CC(C)(C)N(c34)C2=O)cc1 Show InChI InChI=1S/C26H33N3O/c1-19-7-9-20(10-8-19)18-27-13-15-28(16-14-27)23-12-11-21-5-4-6-22-17-26(2,3)29(24(21)22)25(23)30/h4-10,23H,11-18H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]prazosin from alpha-1 adrenergic receptor of rat brain homogenate |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |
Alpha-1A/Alpha-1B/Alpha-1D adrenergic receptor
(Rattus norvegicus (rat)-Rattus norvegicus (Rat)) | BDBM50124933

(5-[4-(2-Methoxy-4-methyl-benzyl)-piperazin-1-yl]-2...)Show SMILES COc1cc(C)ccc1CN1CCN(CC1)C1CCc2cccc3CC(C)(C)N(c23)C1=O Show InChI InChI=1S/C27H35N3O2/c1-19-8-9-22(24(16-19)32-4)18-28-12-14-29(15-13-28)23-11-10-20-6-5-7-21-17-27(2,3)30(25(20)21)26(23)31/h5-9,16,23H,10-15,17-18H2,1-4H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 653 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Neurogen Corporation
Curated by ChEMBL
| Assay Description
Displacement of [3H]prazosin from alpha-1 adrenergic receptor of rat brain homogenate |
Bioorg Med Chem Lett 13: 701-4 (2003)
BindingDB Entry DOI: 10.7270/Q2B56J44 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data