 Found 852 hits with Last Name = 'takaya' and Initial = 'k'
Found 852 hits with Last Name = 'takaya' and Initial = 'k' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50095997
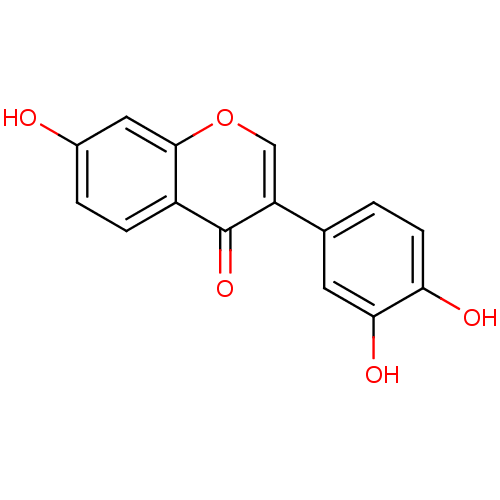
(3',4',7-trihydroxyisoflavone | CHEMBL13486)Show InChI InChI=1S/C15H10O5/c16-9-2-3-10-14(6-9)20-7-11(15(10)19)8-1-4-12(17)13(18)5-8/h1-7,16-18H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50096004
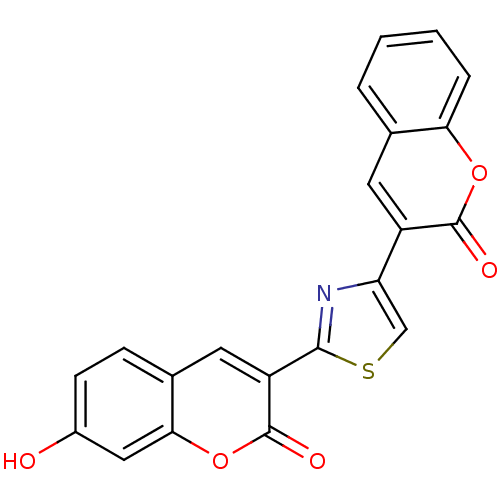
(7-Hydroxy-3-[4-(2-oxo-2H-chromen-3-yl)-thiazol-2-y...)Show SMILES Oc1ccc2cc(-c3nc(cs3)-c3cc4ccccc4oc3=O)c(=O)oc2c1 Show InChI InChI=1S/C21H11NO5S/c23-13-6-5-12-8-15(21(25)27-18(12)9-13)19-22-16(10-28-19)14-7-11-3-1-2-4-17(11)26-20(14)24/h1-10,23H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50096003
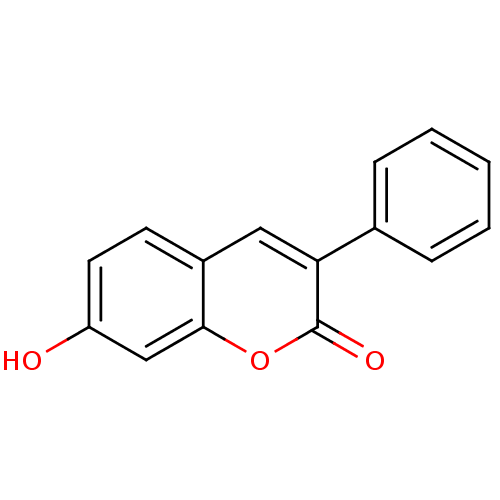
(7-Hydroxy-3-phenyl-chromen-2-one | 7-hydroxy-3-phe...)Show InChI InChI=1S/C15H10O3/c16-12-7-6-11-8-13(10-4-2-1-3-5-10)15(17)18-14(11)9-12/h1-9,16H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50096001
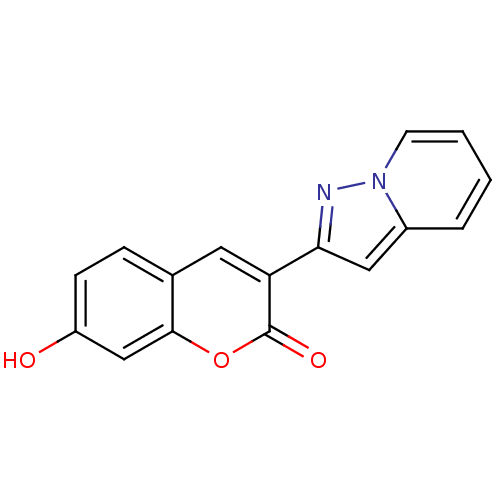
(7-Hydroxy-3-pyrazolo[1,5-a]pyridin-2-yl-chromen-2-...)Show InChI InChI=1S/C16H10N2O3/c19-12-5-4-10-7-13(16(20)21-15(10)9-12)14-8-11-3-1-2-6-18(11)17-14/h1-9,19H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50095993
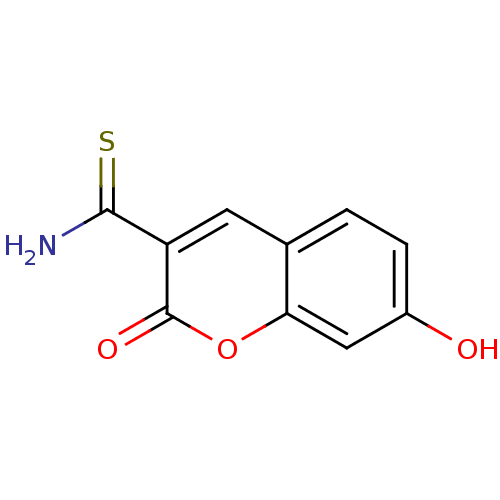
(7-Hydroxy-2-oxo-2H-chromene-3-carbothioic acid ami...)Show InChI InChI=1S/C10H7NO3S/c11-9(15)7-3-5-1-2-6(12)4-8(5)14-10(7)13/h1-4,12H,(H2,11,15) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50096002
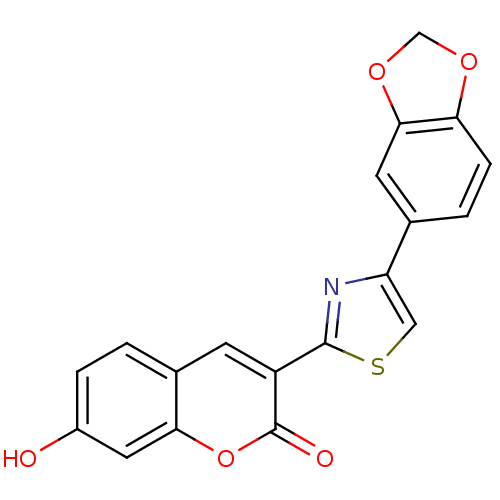
(3-(4-Benzo[1,3]dioxol-5-yl-thiazol-2-yl)-7-hydroxy...)Show SMILES Oc1ccc2cc(-c3nc(cs3)-c3ccc4OCOc4c3)c(=O)oc2c1 Show InChI InChI=1S/C19H11NO5S/c21-12-3-1-11-5-13(19(22)25-16(11)7-12)18-20-14(8-26-18)10-2-4-15-17(6-10)24-9-23-15/h1-8,21H,9H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50096007
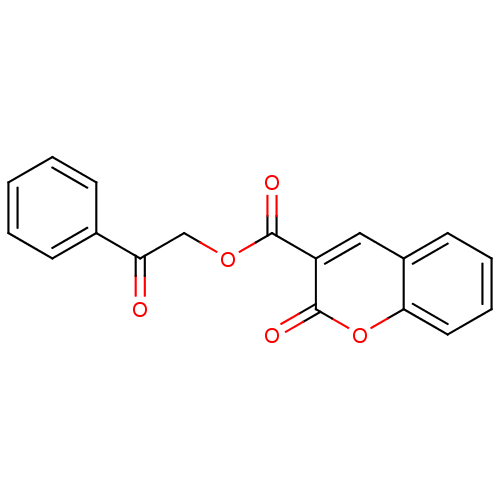
(2-Oxo-2H-chromene-3-carboxylic acid 2-oxo-2-phenyl...)Show InChI InChI=1S/C18H12O5/c19-15(12-6-2-1-3-7-12)11-22-17(20)14-10-13-8-4-5-9-16(13)23-18(14)21/h1-10H,11H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Beta-1,4-galactosyltransferase 1
(Homo sapiens (Human)) | BDBM50370676

(CHEMBL607907)Show SMILES O[C@@H]1[C@@H](COP(O)(=O)OP(O)(=O)O[C@H]2O[C@@H](COCCOCCOCCOCc3ccc4ccccc4c3)[C@H](O)[C@H](O)[C@@H]2O)OC([C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C32H44N2O20P2/c35-24-7-8-34(32(41)33-24)30-28(39)26(37)23(51-30)18-50-55(42,43)54-56(44,45)53-31-29(40)27(38)25(36)22(52-31)17-49-14-12-47-10-9-46-11-13-48-16-19-5-6-20-3-1-2-4-21(20)15-19/h1-8,15,22-23,25-31,36-40H,9-14,16-18H2,(H,42,43)(H,44,45)(H,33,35,41)/t22-,23+,25-,26+,27-,28+,29-,30?,31+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.86E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido University
Curated by ChEMBL
| Assay Description
Inhibitory constant against human galactosyltransferase using UDP-Gal |
J Med Chem 48: 6054-65 (2005)
Article DOI: 10.1021/jm0504297
BindingDB Entry DOI: 10.7270/Q2JW8FPX |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50096006
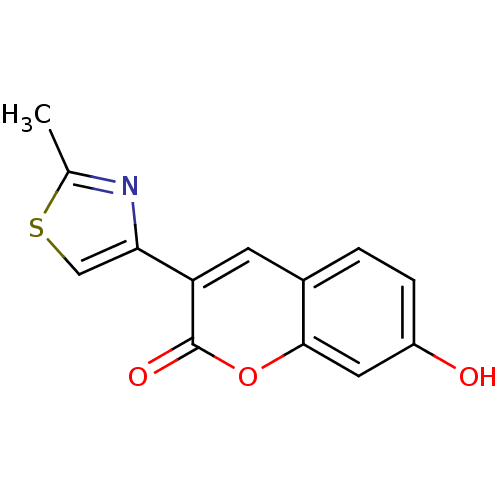
(7-Hydroxy-3-(2-methyl-thiazol-4-yl)-chromen-2-one ...)Show InChI InChI=1S/C13H9NO3S/c1-7-14-11(6-18-7)10-4-8-2-3-9(15)5-12(8)17-13(10)16/h2-6,15H,1H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50096000
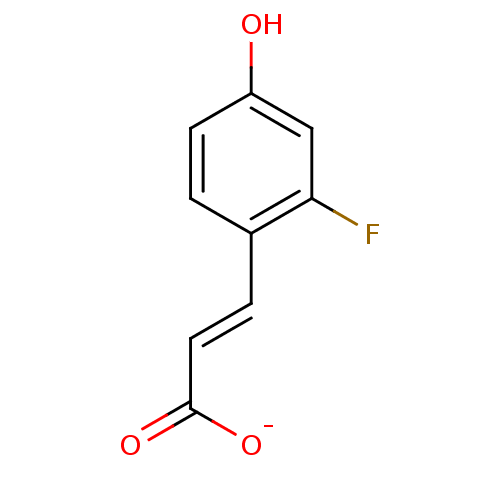
(3-(2-Fluoro-4-hydroxy-phenyl)-acrylic acid anion)Show InChI InChI=1S/C9H7FO3/c10-8-5-7(11)3-1-6(8)2-4-9(12)13/h1-5,11H,(H,12,13)/p-1/b4-2+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| PubMed
| 2.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50095994
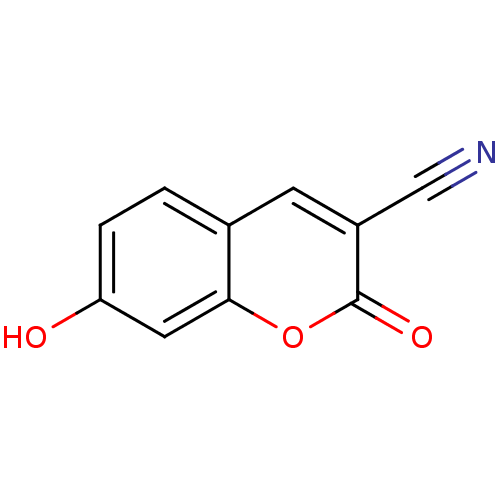
(3-Cyano-7-hydroxycoumarin (2) | 7-Hydroxy-2-oxo-2H...)Show InChI InChI=1S/C10H5NO3/c11-5-7-3-6-1-2-8(12)4-9(6)14-10(7)13/h1-4,12H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50096005

(7-Hydroxy-3-(4-methyl-thiazol-2-yl)-chromen-2-one ...)Show InChI InChI=1S/C13H9NO3S/c1-7-6-18-12(14-7)10-4-8-2-3-9(15)5-11(8)17-13(10)16/h2-6,15H,1H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50096008
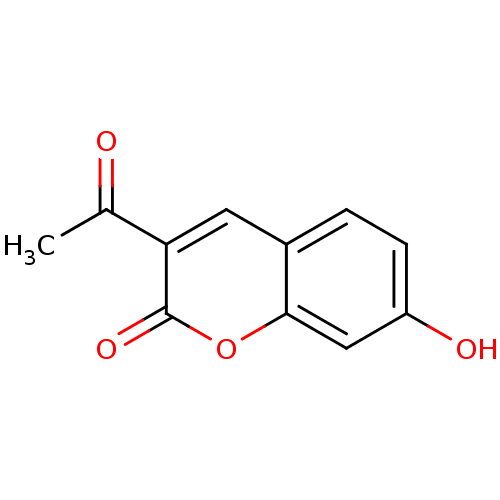
(3-Acetyl-7-hydroxy-chromen-2-one | 3-acetyl-7-hydr...)Show InChI InChI=1S/C11H8O4/c1-6(12)9-4-7-2-3-8(13)5-10(7)15-11(9)14/h2-5,13H,1H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 4.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Beta-1,4-galactosyltransferase 1
(Homo sapiens (Human)) | BDBM50370674
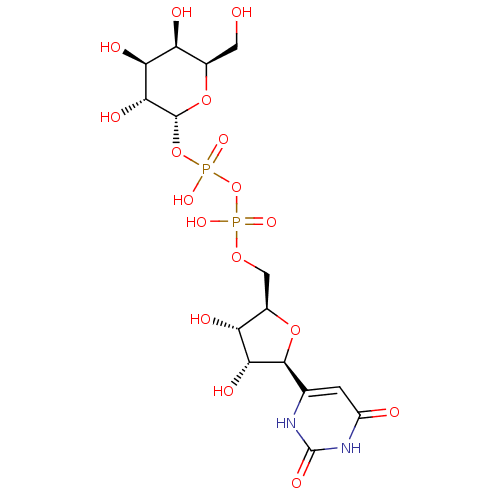
(UDP-GALACTOSE)Show SMILES OC[C@H]1O[C@H](OP(O)(=O)OP(O)(=O)OC[C@H]2O[C@H]([C@H](O)[C@@H]2O)c2cc(=O)[nH]c(=O)[nH]2)[C@H](O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C15H24N2O17P2/c18-2-5-8(20)10(22)12(24)14(32-5)33-36(28,29)34-35(26,27)30-3-6-9(21)11(23)13(31-6)4-1-7(19)17-15(25)16-4/h1,5-6,8-14,18,20-24H,2-3H2,(H,26,27)(H,28,29)(H2,16,17,19,25)/t5-,6-,8+,9-,10+,11-,12-,13+,14-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 4.91E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido University
Curated by ChEMBL
| Assay Description
Inhibitory constant against human recombinant Beta-1,4-galactosyltransferase I |
J Med Chem 48: 6054-65 (2005)
Article DOI: 10.1021/jm0504297
BindingDB Entry DOI: 10.7270/Q2JW8FPX |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50095996
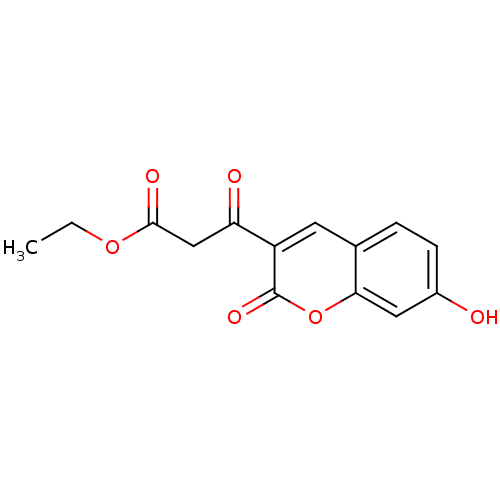
(3-(7-Hydroxy-2-oxo-2H-chromen-3-yl)-3-oxo-propioni...)Show InChI InChI=1S/C14H12O6/c1-2-19-13(17)7-11(16)10-5-8-3-4-9(15)6-12(8)20-14(10)18/h3-6,15H,2,7H2,1H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 5.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50096009
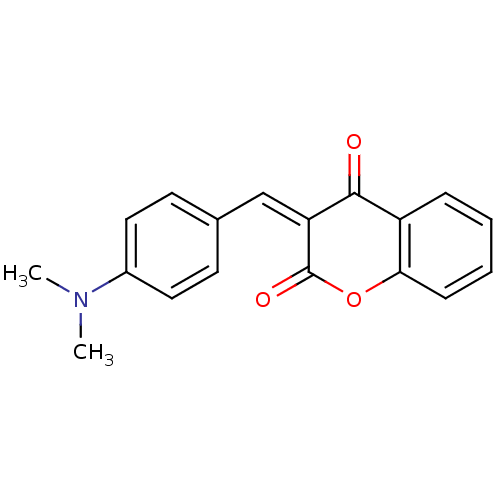
(3-(4-Dimethylamino-benzylidene)-chroman-2,4-dione ...)Show InChI InChI=1S/C18H15NO3/c1-19(2)13-9-7-12(8-10-13)11-15-17(20)14-5-3-4-6-16(14)22-18(15)21/h3-11H,1-2H3/b15-11- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 6.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | |
Macrophage migration inhibitory factor
(Homo sapiens (Human)) | BDBM50095995
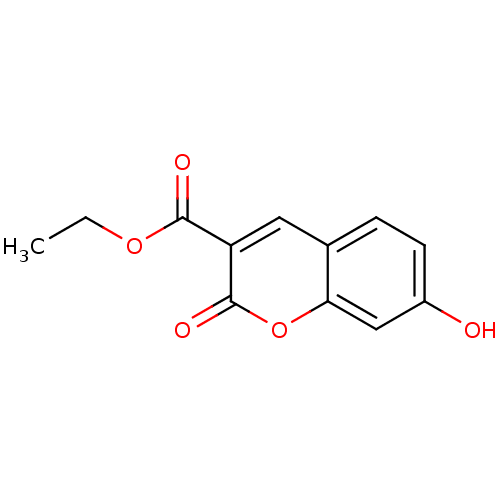
(7-HYDROXY-2-OXO-CHROMENE-3-CARBOXYLIC ACID ETHYL E...)Show InChI InChI=1S/C12H10O5/c1-2-16-11(14)9-5-7-3-4-8(13)6-10(7)17-12(9)15/h3-6,13H,2H2,1H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
PubMed
| 7.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Yamanouchi Pharmaceutical Company Ltd.
Curated by ChEMBL
| Assay Description
Inhibitory activity against tautomerase macrophage migration inhibitory factor (MIF) |
J Med Chem 44: 540-7 (2001)
BindingDB Entry DOI: 10.7270/Q2W66MGR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-1,4-galactosyltransferase 1
(Homo sapiens (Human)) | BDBM50370675
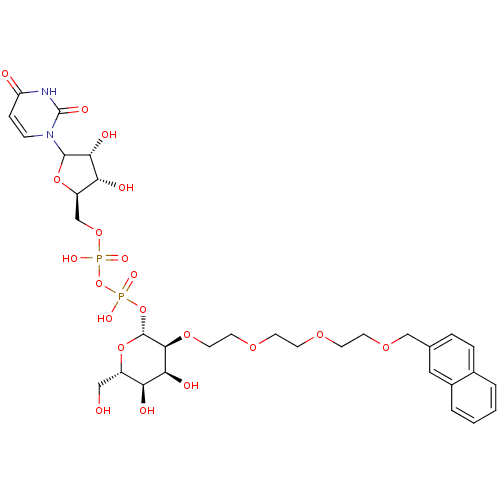
(CHEMBL607908)Show SMILES OC[C@@H]1O[C@H](OP(O)(=O)OP(O)(=O)OC[C@H]2OC([C@H](O)[C@@H]2O)n2ccc(=O)[nH]c2=O)[C@@H](OCCOCCOCCOCc2ccc3ccccc3c2)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C32H44N2O20P2/c35-16-22-25(37)27(39)29(49-14-13-47-10-9-46-11-12-48-17-19-5-6-20-3-1-2-4-21(20)15-19)31(52-22)53-56(44,45)54-55(42,43)50-18-23-26(38)28(40)30(51-23)34-8-7-24(36)33-32(34)41/h1-8,15,22-23,25-31,35,37-40H,9-14,16-18H2,(H,42,43)(H,44,45)(H,33,36,41)/t22-,23+,25-,26+,27-,28+,29-,30?,31+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.17E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido University
Curated by ChEMBL
| Assay Description
Inhibitory constant against human recombinant Beta-1,4-galactosyltransferase I |
J Med Chem 48: 6054-65 (2005)
Article DOI: 10.1021/jm0504297
BindingDB Entry DOI: 10.7270/Q2JW8FPX |
More data for this
Ligand-Target Pair | |
Beta-1,4-galactosyltransferase 1
(Homo sapiens (Human)) | BDBM50370678

(CHEMBL611116)Show SMILES O[C@@H]1[C@@H](COP(O)(=O)OP(O)(=O)O[C@H]2O[C@@H](CNNC(=O)COCCOCCOCc3ccc4ccccc4c3)[C@H](O)[C@H](O)[C@@H]2O)OC([C@@H]1O)n1ccc(=O)[nH]c1=O |r| Show InChI InChI=1S/C32H44N4O20P2/c37-23-7-8-36(32(44)34-23)30-28(42)26(40)22(53-30)16-52-57(45,46)56-58(47,48)55-31-29(43)27(41)25(39)21(54-31)14-33-35-24(38)17-51-12-10-49-9-11-50-15-18-5-6-19-3-1-2-4-20(19)13-18/h1-8,13,21-22,25-31,33,39-43H,9-12,14-17H2,(H,35,38)(H,45,46)(H,47,48)(H,34,37,44)/t21-,22+,25-,26+,27-,28+,29-,30?,31+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.06E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido University
Curated by ChEMBL
| Assay Description
Inhibitory constant against human recombinant Beta-1,4-galactosyltransferase I |
J Med Chem 48: 6054-65 (2005)
Article DOI: 10.1021/jm0504297
BindingDB Entry DOI: 10.7270/Q2JW8FPX |
More data for this
Ligand-Target Pair | |
Beta-1,4-galactosyltransferase 1
(Homo sapiens (Human)) | BDBM50370677

(CHEMBL609634)Show SMILES Cc1ccc2ccc(COC(=O)COCCOCC(=O)NC[C@@H]3O[C@H](OP(O)(=O)OP(O)(=O)OC[C@H]4OC([C@H](O)[C@@H]4O)n4ccc(=O)[nH]c4=O)[C@@H](O)[C@@H](O)[C@H]3O)cc2c1 |r| Show InChI InChI=1S/C33H43N3O21P2/c1-17-2-4-19-5-3-18(11-20(19)10-17)13-52-25(39)16-51-9-8-50-15-24(38)34-12-21-26(40)28(42)30(44)32(55-21)56-59(48,49)57-58(46,47)53-14-22-27(41)29(43)31(54-22)36-7-6-23(37)35-33(36)45/h2-7,10-11,21-22,26-32,40-44H,8-9,12-16H2,1H3,(H,34,38)(H,46,47)(H,48,49)(H,35,37,45)/t21-,22+,26-,27+,28-,29+,30-,31?,32+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.23E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido University
Curated by ChEMBL
| Assay Description
Inhibitory constant against human recombinant Beta-1,4-galactosyltransferase I |
J Med Chem 48: 6054-65 (2005)
Article DOI: 10.1021/jm0504297
BindingDB Entry DOI: 10.7270/Q2JW8FPX |
More data for this
Ligand-Target Pair | |
Beta-1,4-galactosyltransferase 1
(Homo sapiens (Human)) | BDBM50173721
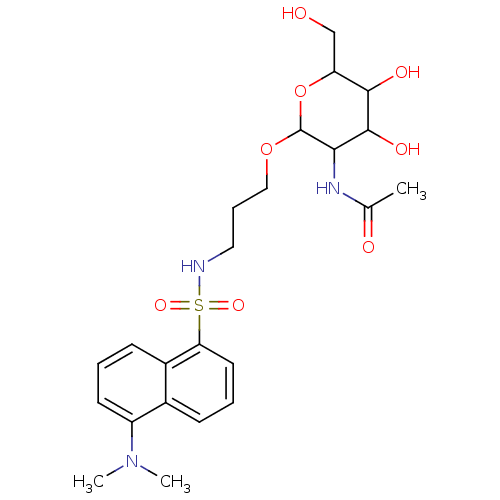
(CHEMBL196432 | Uridine-5'-diphosphogalactose deriv...)Show SMILES CN(C)c1cccc2c(cccc12)S(=O)(=O)NCCCOC1OC(CO)C(O)C(O)C1NC(C)=O Show InChI InChI=1S/C23H33N3O8S/c1-14(28)25-20-22(30)21(29)18(13-27)34-23(20)33-12-6-11-24-35(31,32)19-10-5-7-15-16(19)8-4-9-17(15)26(2)3/h4-5,7-10,18,20-24,27,29-30H,6,11-13H2,1-3H3,(H,25,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 7.67E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido University
Curated by ChEMBL
| Assay Description
Inhibitory constant against human Beta-1,4-galactosyltransferase I using UDP-Gal (0-160 uM) |
J Med Chem 48: 6054-65 (2005)
Article DOI: 10.1021/jm0504297
BindingDB Entry DOI: 10.7270/Q2JW8FPX |
More data for this
Ligand-Target Pair | |
Beta-1,4-galactosyltransferase 1
(Homo sapiens (Human)) | BDBM50370679

(CHEMBL611112)Show SMILES COCCOCCOCCOC[C@@H]1O[C@H](OP(O)(=O)OP(O)(=O)OC[C@H]2OC([C@H](O)[C@@H]2O)n2ccc(=O)[nH]c2=O)[C@@H](O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C22H38N2O20P2/c1-36-4-5-37-6-7-38-8-9-39-10-12-15(26)17(28)19(30)21(42-12)43-46(34,35)44-45(32,33)40-11-13-16(27)18(29)20(41-13)24-3-2-14(25)23-22(24)31/h2-3,12-13,15-21,26-30H,4-11H2,1H3,(H,32,33)(H,34,35)(H,23,25,31)/t12-,13+,15-,16+,17-,18+,19-,20?,21+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.49E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido University
Curated by ChEMBL
| Assay Description
Inhibitory constant against human recombinant Beta-1,4-galactosyltransferase I |
J Med Chem 48: 6054-65 (2005)
Article DOI: 10.1021/jm0504297
BindingDB Entry DOI: 10.7270/Q2JW8FPX |
More data for this
Ligand-Target Pair | |
Sepiapterin reductase
(Homo sapiens (Human)) | US20240092784, Example 259
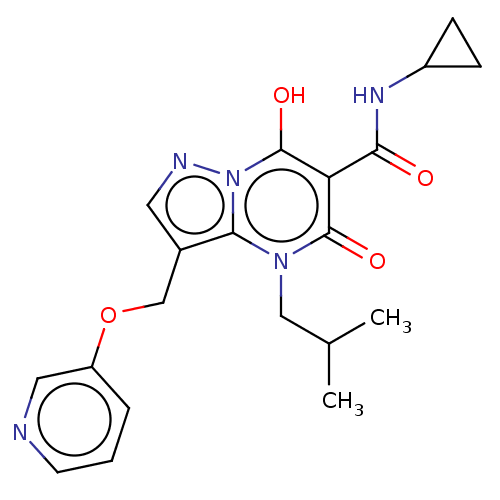
(N-Cyclopropyl-7-hydroxy-4-isobutyl-5-oxo-3-((pyrid...) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50264847
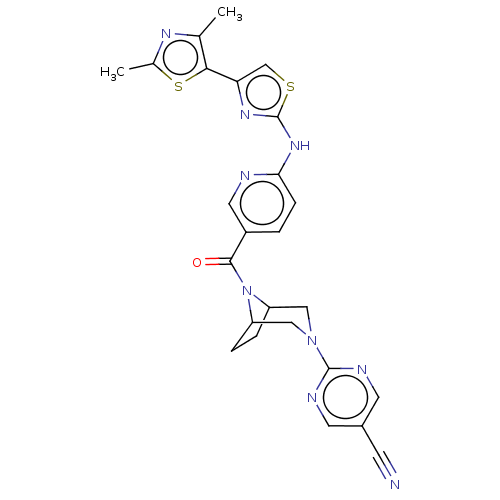
(CHEMBL4096902)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2C3CCC2CN(C3)c2ncc(cn2)C#N)n1 Show InChI InChI=1S/C25H23N9OS2/c1-14-22(37-15(2)30-14)20-13-36-25(31-20)32-21-6-3-17(10-27-21)23(35)34-18-4-5-19(34)12-33(11-18)24-28-8-16(7-26)9-29-24/h3,6,8-10,13,18-19H,4-5,11-12H2,1-2H3,(H,27,31,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human TRPV4 expressed in CHOK1 cells assessed as inhibition of 4alphaPDD-induced activation pretreated for 5 mins ... |
Bioorg Med Chem 25: 2177-2190 (2017)
Article DOI: 10.1016/j.bmc.2017.02.047
BindingDB Entry DOI: 10.7270/Q2N58PT9 |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50256209
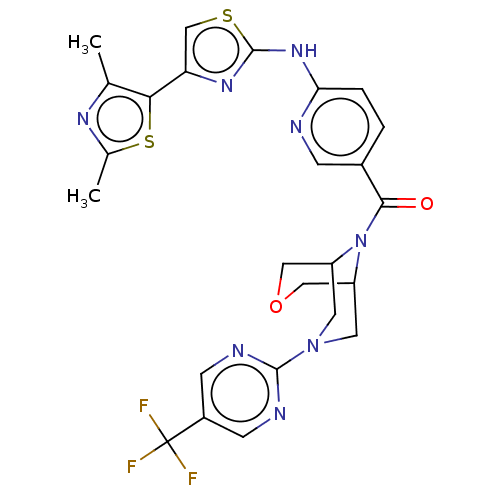
(CHEMBL4101768)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2C3COCC2CN(C3)c2ncc(cn2)C(F)(F)F)n1 Show InChI InChI=1S/C25H23F3N8O2S2/c1-13-21(40-14(2)32-13)19-12-39-24(33-19)34-20-4-3-15(5-29-20)22(37)36-17-8-35(9-18(36)11-38-10-17)23-30-6-16(7-31-23)25(26,27)28/h3-7,12,17-18H,8-11H2,1-2H3,(H,29,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 0.190 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human TRPV4 expressed in CHOK1 cells assessed as inhibition of 4alphaPDD-induced activation pretreated for 5 mins ... |
Bioorg Med Chem 25: 2177-2190 (2017)
Article DOI: 10.1016/j.bmc.2017.02.047
BindingDB Entry DOI: 10.7270/Q2N58PT9 |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50264846
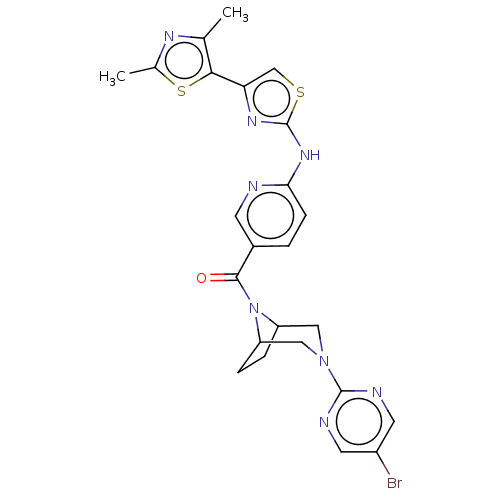
(CHEMBL4066531)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2C3CCC2CN(C3)c2ncc(Br)cn2)n1 Show InChI InChI=1S/C24H23BrN8OS2/c1-13-21(36-14(2)29-13)19-12-35-24(30-19)31-20-6-3-15(7-26-20)22(34)33-17-4-5-18(33)11-32(10-17)23-27-8-16(25)9-28-23/h3,6-9,12,17-18H,4-5,10-11H2,1-2H3,(H,26,30,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human TRPV4 expressed in CHOK1 cells assessed as inhibition of 4alphaPDD-induced activation pretreated for 5 mins ... |
Bioorg Med Chem 25: 2177-2190 (2017)
Article DOI: 10.1016/j.bmc.2017.02.047
BindingDB Entry DOI: 10.7270/Q2N58PT9 |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50192809
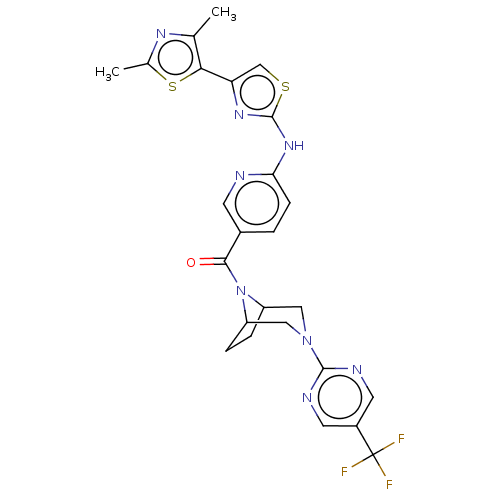
(CHEMBL3941914)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2C3CCC2CN(C3)c2ncc(cn2)C(F)(F)F)n1 Show InChI InChI=1S/C25H23F3N8OS2/c1-13-21(39-14(2)32-13)19-12-38-24(33-19)34-20-6-3-15(7-29-20)22(37)36-17-4-5-18(36)11-35(10-17)23-30-8-16(9-31-23)25(26,27)28/h3,6-9,12,17-18H,4-5,10-11H2,1-2H3,(H,29,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV4 assessed as inhibition of 4alpha-PDD-induced activation |
Bioorg Med Chem Lett 26: 4936-4941 (2016)
Article DOI: 10.1016/j.bmcl.2016.09.014
BindingDB Entry DOI: 10.7270/Q2154K06 |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50192809

(CHEMBL3941914)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2C3CCC2CN(C3)c2ncc(cn2)C(F)(F)F)n1 Show InChI InChI=1S/C25H23F3N8OS2/c1-13-21(39-14(2)32-13)19-12-38-24(33-19)34-20-6-3-15(7-29-20)22(37)36-17-4-5-18(36)11-35(10-17)23-30-8-16(9-31-23)25(26,27)28/h3,6-9,12,17-18H,4-5,10-11H2,1-2H3,(H,29,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.220 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human TRPV4 expressed in CHOK1 cells assessed as inhibition of 4alphaPDD-induced activation pretreated for 5 mins ... |
Bioorg Med Chem 25: 2177-2190 (2017)
Article DOI: 10.1016/j.bmc.2017.02.047
BindingDB Entry DOI: 10.7270/Q2N58PT9 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Mus musculus (Mouse)) | BDBM118179

(US8653125, Ia-40)Show SMILES Clc1ccc(cc1Cl)-c1c[nH]c(n1)[C@@H]1CC[C@@H](CC1)NC(=O)c1ccon1 |r,wU:16.21,13.14,(9.04,-4.47,;7.7,-3.7,;6.37,-4.47,;5.04,-3.7,;5.04,-2.16,;6.37,-1.39,;7.7,-2.16,;9.04,-1.39,;3.7,-1.39,;3.7,.15,;2.24,.62,;1.33,-.62,;2.24,-1.87,;,.15,;,1.69,;-1.33,2.46,;-2.67,1.69,;-2.67,.15,;-1.33,-.62,;-4,2.46,;-5.33,1.69,;-5.33,.15,;-6.67,2.46,;-8.13,1.98,;-9.04,3.23,;-8.13,4.47,;-6.67,4,)| Show InChI InChI=1S/C19H18Cl2N4O2/c20-14-6-3-12(9-15(14)21)17-10-22-18(24-17)11-1-4-13(5-2-11)23-19(26)16-7-8-27-25-16/h3,6-11,13H,1-2,4-5H2,(H,22,24)(H,23,26)/t11-,13+ | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 0.260 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Shionogi Co., Ltd.
US Patent
| Assay Description
cDNA sequence encoding mouse NPY Y5 receptor (Biochem. Biophys. Acta 1328: 83-89, 1997) was cloned in the expression vector (pME18S, Takebe et al. Mo... |
US Patent US8653125 (2014)
BindingDB Entry DOI: 10.7270/Q2VQ31BN |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50256228
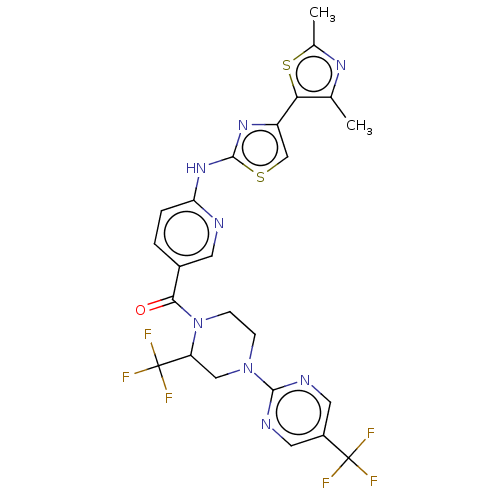
(CHEMBL4099293)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2CCN(CC2C(F)(F)F)c2ncc(cn2)C(F)(F)F)n1 Show InChI InChI=1S/C24H20F6N8OS2/c1-12-19(41-13(2)34-12)16-11-40-22(35-16)36-18-4-3-14(7-31-18)20(39)38-6-5-37(10-17(38)24(28,29)30)21-32-8-15(9-33-21)23(25,26)27/h3-4,7-9,11,17H,5-6,10H2,1-2H3,(H,31,35,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.290 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human TRPV4 expressed in CHOK1 cells assessed as inhibition of 4alphaPDD-induced activation pretreated for 5 mins ... |
Bioorg Med Chem 25: 2177-2190 (2017)
Article DOI: 10.1016/j.bmc.2017.02.047
BindingDB Entry DOI: 10.7270/Q2N58PT9 |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50192811
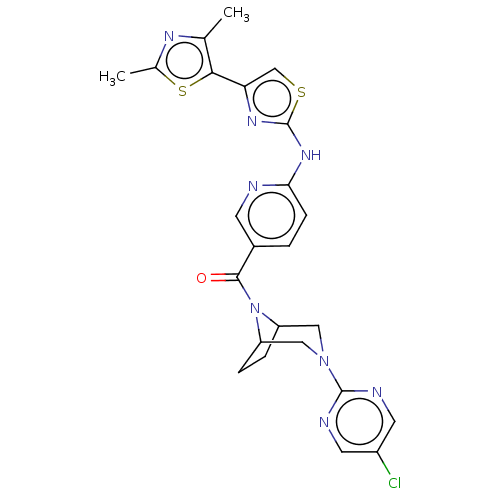
(CHEMBL3971502)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2C3CCC2CN(C3)c2ncc(Cl)cn2)n1 Show InChI InChI=1S/C24H23ClN8OS2/c1-13-21(36-14(2)29-13)19-12-35-24(30-19)31-20-6-3-15(7-26-20)22(34)33-17-4-5-18(33)11-32(10-17)23-27-8-16(25)9-28-23/h3,6-9,12,17-18H,4-5,10-11H2,1-2H3,(H,26,30,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV4 assessed as inhibition of 4alpha-PDD-induced activation |
Bioorg Med Chem Lett 26: 4936-4941 (2016)
Article DOI: 10.1016/j.bmcl.2016.09.014
BindingDB Entry DOI: 10.7270/Q2154K06 |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50192811

(CHEMBL3971502)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2C3CCC2CN(C3)c2ncc(Cl)cn2)n1 Show InChI InChI=1S/C24H23ClN8OS2/c1-13-21(36-14(2)29-13)19-12-35-24(30-19)31-20-6-3-15(7-26-20)22(34)33-17-4-5-18(33)11-32(10-17)23-27-8-16(25)9-28-23/h3,6-9,12,17-18H,4-5,10-11H2,1-2H3,(H,26,30,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human TRPV4 expressed in CHOK1 cells assessed as inhibition of 4alphaPDD-induced activation pretreated for 5 mins ... |
Bioorg Med Chem 25: 2177-2190 (2017)
Article DOI: 10.1016/j.bmc.2017.02.047
BindingDB Entry DOI: 10.7270/Q2N58PT9 |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50264858
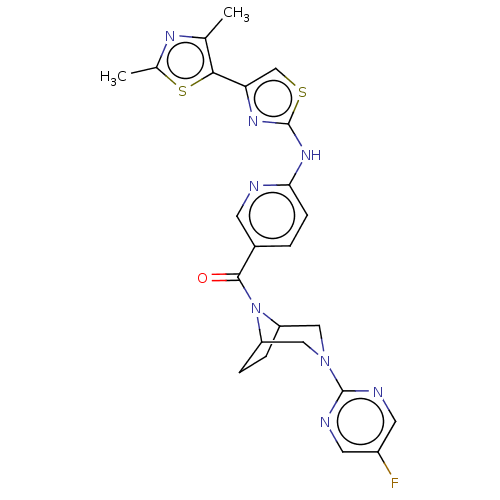
(CHEMBL4077638)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2C3CCC2CN(C3)c2ncc(F)cn2)n1 Show InChI InChI=1S/C24H23FN8OS2/c1-13-21(36-14(2)29-13)19-12-35-24(30-19)31-20-6-3-15(7-26-20)22(34)33-17-4-5-18(33)11-32(10-17)23-27-8-16(25)9-28-23/h3,6-9,12,17-18H,4-5,10-11H2,1-2H3,(H,26,30,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human TRPV4 expressed in CHOK1 cells assessed as inhibition of 4alphaPDD-induced activation pretreated for 5 mins ... |
Bioorg Med Chem 25: 2177-2190 (2017)
Article DOI: 10.1016/j.bmc.2017.02.047
BindingDB Entry DOI: 10.7270/Q2N58PT9 |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50192807
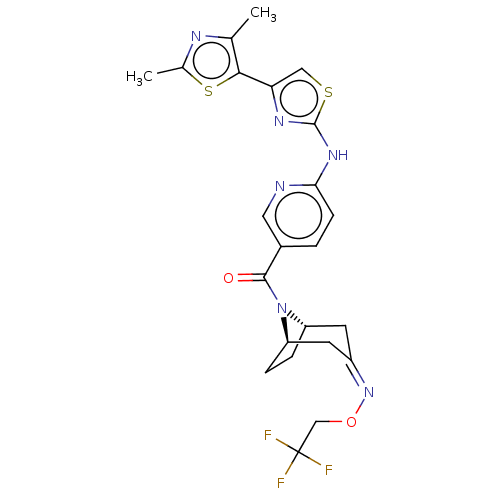
(CHEMBL3984947)Show SMILES [H][C@]12CC[C@]([H])(C\C(C1)=N/OCC(F)(F)F)N2C(=O)c1ccc(Nc2nc(cs2)-c2sc(C)nc2C)nc1 |r| Show InChI InChI=1S/C23H23F3N6O2S2/c1-12-20(36-13(2)28-12)18-10-35-22(29-18)30-19-6-3-14(9-27-19)21(33)32-16-4-5-17(32)8-15(7-16)31-34-11-23(24,25)26/h3,6,9-10,16-17H,4-5,7-8,11H2,1-2H3,(H,27,29,30)/b31-15-/t16-,17+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.360 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV4 assessed as inhibition of 4alpha-PDD-induced activation |
Bioorg Med Chem Lett 26: 4936-4941 (2016)
Article DOI: 10.1016/j.bmcl.2016.09.014
BindingDB Entry DOI: 10.7270/Q2154K06 |
More data for this
Ligand-Target Pair | |
Sepiapterin reductase
(Homo sapiens (Human)) | US20240092784, Example 394
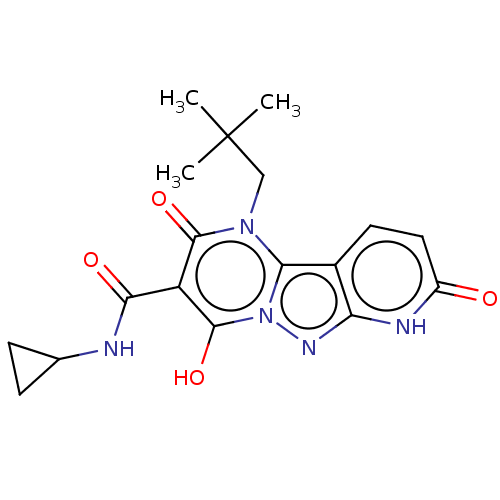
(N-Cyclopropyl-4-hydroxy-1-neopentyl-2,8-dioxo-1,2,...) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 0.390 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50264859
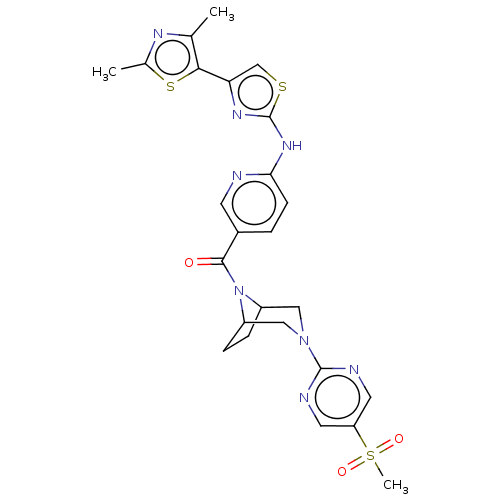
(CHEMBL4078117)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2C3CCC2CN(C3)c2ncc(cn2)S(C)(=O)=O)n1 Show InChI InChI=1S/C25H26N8O3S3/c1-14-22(38-15(2)29-14)20-13-37-25(30-20)31-21-7-4-16(8-26-21)23(34)33-17-5-6-18(33)12-32(11-17)24-27-9-19(10-28-24)39(3,35)36/h4,7-10,13,17-18H,5-6,11-12H2,1-3H3,(H,26,30,31) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.390 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human TRPV4 expressed in CHOK1 cells assessed as inhibition of 4alphaPDD-induced activation pretreated for 5 mins ... |
Bioorg Med Chem 25: 2177-2190 (2017)
Article DOI: 10.1016/j.bmc.2017.02.047
BindingDB Entry DOI: 10.7270/Q2N58PT9 |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50192809

(CHEMBL3941914)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2C3CCC2CN(C3)c2ncc(cn2)C(F)(F)F)n1 Show InChI InChI=1S/C25H23F3N8OS2/c1-13-21(39-14(2)32-13)19-12-38-24(33-19)34-20-6-3-15(7-29-20)22(37)36-17-4-5-18(36)11-35(10-17)23-30-8-16(9-31-23)25(26,27)28/h3,6-9,12,17-18H,4-5,10-11H2,1-2H3,(H,29,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV4 assessed as inhibition of hypotonicity-induced activation |
Bioorg Med Chem Lett 26: 4936-4941 (2016)
Article DOI: 10.1016/j.bmcl.2016.09.014
BindingDB Entry DOI: 10.7270/Q2154K06 |
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50192809

(CHEMBL3941914)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2C3CCC2CN(C3)c2ncc(cn2)C(F)(F)F)n1 Show InChI InChI=1S/C25H23F3N8OS2/c1-13-21(39-14(2)32-13)19-12-38-24(33-19)34-20-6-3-15(7-29-20)22(37)36-17-4-5-18(36)11-35(10-17)23-30-8-16(9-31-23)25(26,27)28/h3,6-9,12,17-18H,4-5,10-11H2,1-2H3,(H,29,33,34) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human TRPV4 expressed in CHOK1 cells assessed as inhibition of hypotonicity-induced activation pretreated for 5 mi... |
Bioorg Med Chem 25: 2177-2190 (2017)
Article DOI: 10.1016/j.bmc.2017.02.047
BindingDB Entry DOI: 10.7270/Q2N58PT9 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Mus musculus (Mouse)) | BDBM118190

(US8653125, Ic-16)Show SMILES CC(C)n1c2ccc(NC(=O)[C@@H]3CC[C@@H](CC3)Oc3cc(ccn3)C(F)(F)F)cc2oc1=O |r,wU:14.17,11.10,(6.26,-3.84,;7.35,-2.75,;8.83,-3.15,;6.95,-1.26,;5.48,-.78,;4.15,-1.55,;2.82,-.78,;2.82,.76,;1.48,1.53,;.15,.76,;.15,-.78,;-1.18,1.53,;-1.18,3.07,;-2.52,3.84,;-3.85,3.07,;-3.85,1.53,;-2.52,.76,;-5.19,3.84,;-6.52,3.07,;-6.52,1.53,;-7.85,.76,;-9.19,1.53,;-9.19,3.07,;-7.85,3.84,;-7.85,-.78,;-7.85,-2.32,;-9.39,-.78,;-6.31,-.78,;4.15,1.53,;5.48,.76,;6.95,1.23,;7.85,-.01,;9.39,-.01,)| Show InChI InChI=1S/C23H24F3N3O4/c1-13(2)29-18-8-5-16(12-19(18)33-22(29)31)28-21(30)14-3-6-17(7-4-14)32-20-11-15(9-10-27-20)23(24,25)26/h5,8-14,17H,3-4,6-7H2,1-2H3,(H,28,30)/t14-,17+ | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.410 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Shionogi Co., Ltd.
US Patent
| Assay Description
cDNA sequence encoding mouse NPY Y5 receptor (Biochem. Biophys. Acta 1328: 83-89, 1997) was cloned in the expression vector (pME18S, Takebe et al. Mo... |
US Patent US8653125 (2014)
BindingDB Entry DOI: 10.7270/Q2VQ31BN |
More data for this
Ligand-Target Pair | |
Sepiapterin reductase
(Homo sapiens (Human)) | US20240092784, Example 753
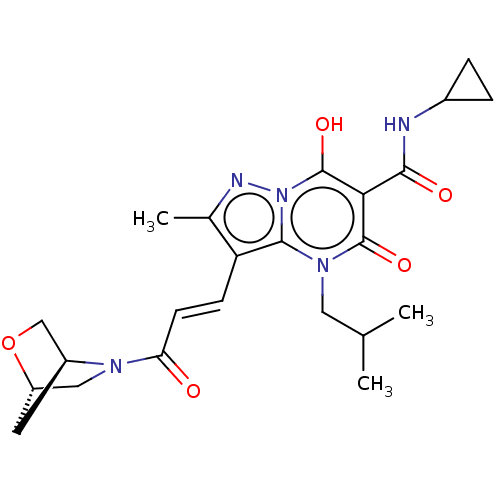
(3-((E)-3-((1S,4S)-2-Oxa-5-azabicyclo[2.2.1]heptan-...) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 0.480 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50264845
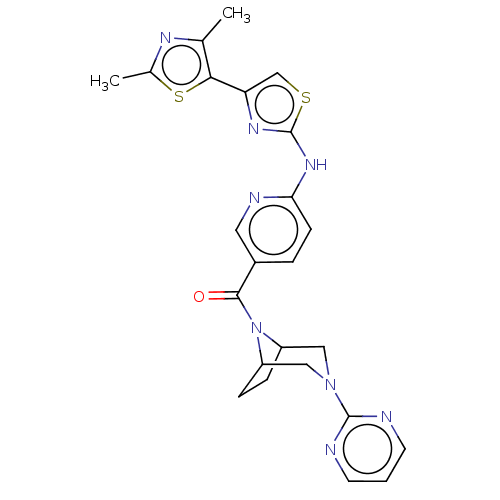
(CHEMBL4095552)Show SMILES Cc1nc(C)c(s1)-c1csc(Nc2ccc(cn2)C(=O)N2C3CCC2CN(C3)c2ncccn2)n1 Show InChI InChI=1S/C24H24N8OS2/c1-14-21(35-15(2)28-14)19-13-34-24(29-19)30-20-7-4-16(10-27-20)22(33)32-17-5-6-18(32)12-31(11-17)23-25-8-3-9-26-23/h3-4,7-10,13,17-18H,5-6,11-12H2,1-2H3,(H,27,29,30) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at recombinant human TRPV4 expressed in CHOK1 cells assessed as inhibition of 4alphaPDD-induced activation pretreated for 5 mins ... |
Bioorg Med Chem 25: 2177-2190 (2017)
Article DOI: 10.1016/j.bmc.2017.02.047
BindingDB Entry DOI: 10.7270/Q2N58PT9 |
More data for this
Ligand-Target Pair | |
Sepiapterin reductase
(Homo sapiens (Human)) | US20240092784, Example 370
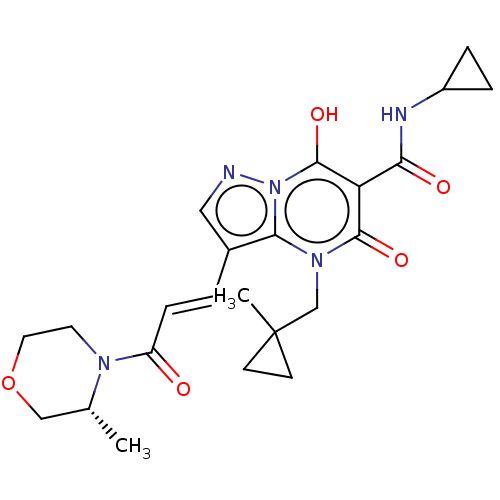
((R,E)-N-Cyclopropyl-7-hydroxy-4-((1-methylcyclopro...) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 0.540 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Sepiapterin reductase
(Homo sapiens (Human)) | US20240092784, Example 532
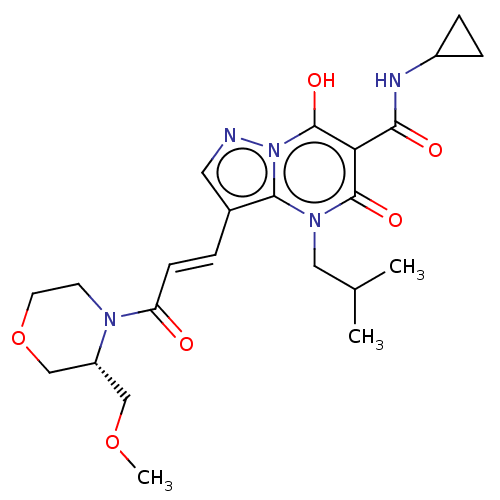
((R,E)-N-Cyclopropyl-7-hydroxy-4-isobutyl-3-(3-(3-(...) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 0.550 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Sepiapterin reductase
(Homo sapiens (Human)) | US20240092784, Example 372
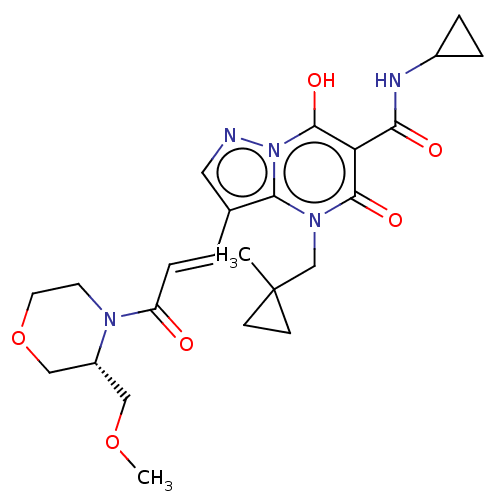
((R,E)-N-Cyclopropyl-7-hydroxy-3-(3-(3-(methoxymeth...) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 0.550 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Sepiapterin reductase
(Homo sapiens (Human)) | US20240092784, Example 401
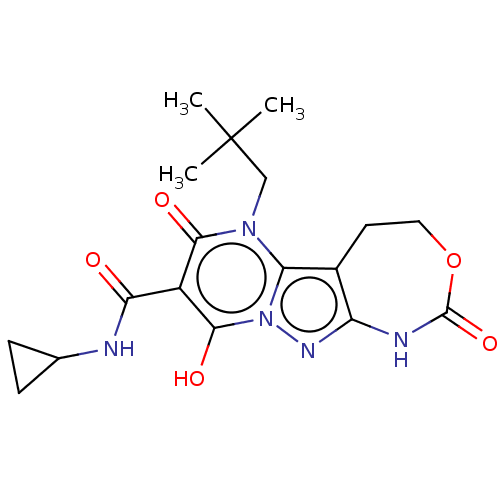
(N-Cyclopropyl-9-hydroxy-6-neopentyl-2,7-dioxo-1,2,...) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 0.560 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Mus musculus (Mouse)) | BDBM118188

(US8653125, Ic-13)Show SMILES CC(C)n1c2ccc(NC(=O)[C@@H]3CC[C@@H](CC3)Oc3cc(Cl)ccc3C#N)cc2oc1=O |r,wU:14.17,11.10,(6.15,-5.38,;7.24,-4.29,;8.73,-4.69,;6.84,-2.8,;5.38,-2.32,;4.05,-3.09,;2.71,-2.32,;2.71,-.78,;1.38,-.01,;.05,-.78,;.05,-2.32,;-1.29,-.01,;-1.29,1.53,;-2.62,2.3,;-3.96,1.53,;-3.96,-.01,;-2.62,-.78,;-5.29,2.3,;-6.62,1.53,;-6.62,-.01,;-7.96,-.78,;-7.96,-2.32,;-9.29,-.01,;-9.29,1.53,;-7.96,2.3,;-7.96,3.84,;-7.96,5.38,;4.05,-.01,;5.38,-.78,;6.84,-.31,;7.75,-1.55,;9.29,-1.55,)| Show InChI InChI=1S/C24H24ClN3O4/c1-14(2)28-20-10-7-18(12-22(20)32-24(28)30)27-23(29)15-4-8-19(9-5-15)31-21-11-17(25)6-3-16(21)13-26/h3,6-7,10-12,14-15,19H,4-5,8-9H2,1-2H3,(H,27,29)/t15-,19+ | Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 0.560 | n/a | n/a | n/a | n/a | 7.4 | 25 |
Shionogi Co., Ltd.
US Patent
| Assay Description
cDNA sequence encoding mouse NPY Y5 receptor (Biochem. Biophys. Acta 1328: 83-89, 1997) was cloned in the expression vector (pME18S, Takebe et al. Mo... |
US Patent US8653125 (2014)
BindingDB Entry DOI: 10.7270/Q2VQ31BN |
More data for this
Ligand-Target Pair | |
Sepiapterin reductase
(Homo sapiens (Human)) | US20240092784, Example 517
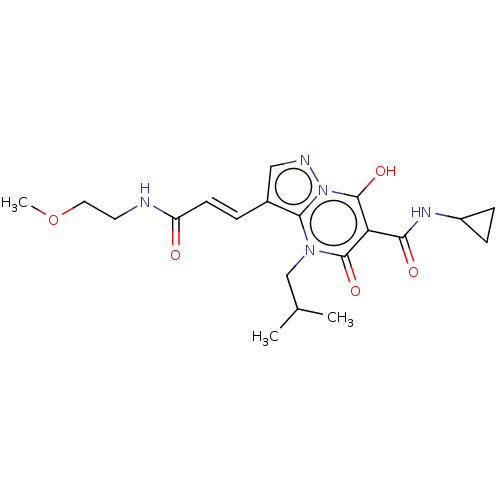
((E)-N-Cyclopropyl-7-hydroxy-4-isobutyl-3-(3-((2-me...) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 0.570 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Transient receptor potential cation channel subfamily V member 4
(Homo sapiens (Human)) | BDBM50192804
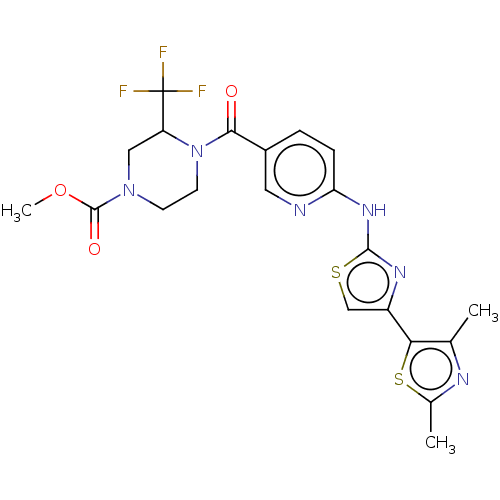
(CHEMBL3933401)Show SMILES COC(=O)N1CCN(C(C1)C(F)(F)F)C(=O)c1ccc(Nc2nc(cs2)-c2sc(C)nc2C)nc1 Show InChI InChI=1S/C21H21F3N6O3S2/c1-11-17(35-12(2)26-11)14-10-34-19(27-14)28-16-5-4-13(8-25-16)18(31)30-7-6-29(20(32)33-3)9-15(30)21(22,23)24/h4-5,8,10,15H,6-7,9H2,1-3H3,(H,25,27,28) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.570 | n/a | n/a | n/a | n/a | n/a | n/a |
Shionogi& Co., Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at human TRPV4 assessed as inhibition of 4alpha-PDD-induced activation |
Bioorg Med Chem Lett 26: 4936-4941 (2016)
Article DOI: 10.1016/j.bmcl.2016.09.014
BindingDB Entry DOI: 10.7270/Q2154K06 |
More data for this
Ligand-Target Pair | |
Sepiapterin reductase
(Homo sapiens (Human)) | US20240092784, Example 514
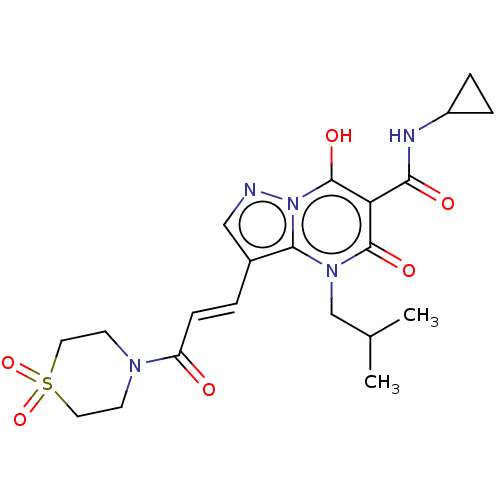
((E)-N-Cyclopropyl-3-(3-(1,1-dioxidothiomorpholino)...) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 0.580 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Sepiapterin reductase
(Homo sapiens (Human)) | US20240092784, Example 456

((E)-3-(3-(8-Oxa-3-azabicyclo[3.2.1]octan-3-yl)-3-o...) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| UniChem
| | n/a | n/a | 0.580 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data