 Found 630 hits with Last Name = 'cottom' and Initial = 'j'
Found 630 hits with Last Name = 'cottom' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
5-hydroxytryptamine receptor 1D
(Homo sapiens (Human)) | BDBM50243699
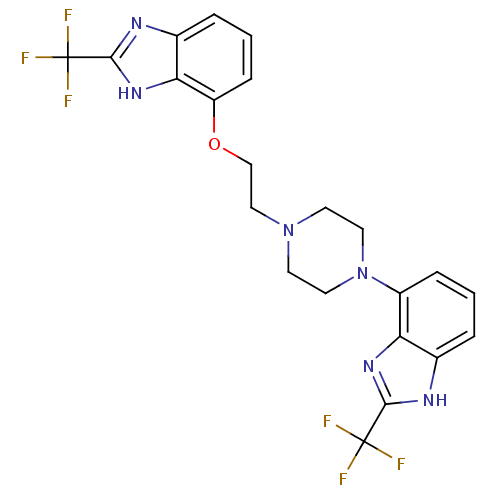
(2-(trifluoromethyl)-7-(2-(4-(2-(trifluoromethyl)-1...)Show SMILES FC(F)(F)c1nc2cccc(OCCN3CCN(CC3)c3cccc4[nH]c(nc34)C(F)(F)F)c2[nH]1 Show InChI InChI=1S/C22H20F6N6O/c23-21(24,25)19-29-13-3-1-5-15(17(13)31-19)34-9-7-33(8-10-34)11-12-35-16-6-2-4-14-18(16)32-20(30-14)22(26,27)28/h1-6H,7-12H2,(H,29,31)(H,30,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Binding affinity to human 5HT1D receptor |
Bioorg Med Chem 16: 6617-40 (2008)
Article DOI: 10.1016/j.bmc.2008.05.024
BindingDB Entry DOI: 10.7270/Q2FB52Q7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50243700
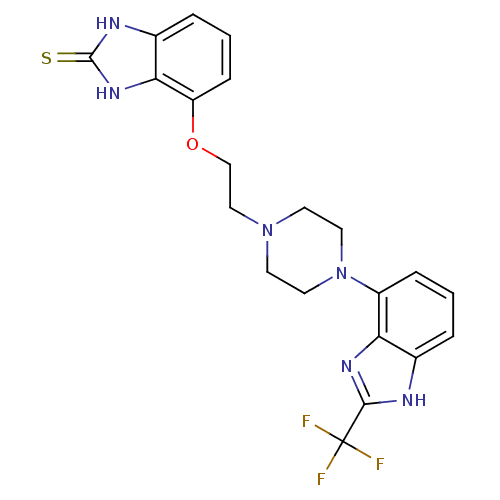
(4-(2-(4-(2-(trifluoromethyl)-1H-benzo[d]imidazol-4...)Show SMILES FC(F)(F)c1nc2c(cccc2[nH]1)N1CCN(CCOc2cccc3[nH]c(=S)[nH]c23)CC1 Show InChI InChI=1S/C21H21F3N6OS/c22-21(23,24)19-25-13-3-1-5-15(17(13)27-19)30-9-7-29(8-10-30)11-12-31-16-6-2-4-14-18(16)28-20(32)26-14/h1-6H,7-12H2,(H,25,27)(H2,26,28,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Binding affinity to human 5HT1A receptor |
Bioorg Med Chem 16: 6617-40 (2008)
Article DOI: 10.1016/j.bmc.2008.05.024
BindingDB Entry DOI: 10.7270/Q2FB52Q7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50243699

(2-(trifluoromethyl)-7-(2-(4-(2-(trifluoromethyl)-1...)Show SMILES FC(F)(F)c1nc2cccc(OCCN3CCN(CC3)c3cccc4[nH]c(nc34)C(F)(F)F)c2[nH]1 Show InChI InChI=1S/C22H20F6N6O/c23-21(24,25)19-29-13-3-1-5-15(17(13)31-19)34-9-7-33(8-10-34)11-12-35-16-6-2-4-14-18(16)32-20(30-14)22(26,27)28/h1-6H,7-12H2,(H,29,31)(H,30,32) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Binding affinity to human 5HT1A receptor |
Bioorg Med Chem 16: 6617-40 (2008)
Article DOI: 10.1016/j.bmc.2008.05.024
BindingDB Entry DOI: 10.7270/Q2FB52Q7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1B
(Homo sapiens (Human)) | BDBM50243699

(2-(trifluoromethyl)-7-(2-(4-(2-(trifluoromethyl)-1...)Show SMILES FC(F)(F)c1nc2cccc(OCCN3CCN(CC3)c3cccc4[nH]c(nc34)C(F)(F)F)c2[nH]1 Show InChI InChI=1S/C22H20F6N6O/c23-21(24,25)19-29-13-3-1-5-15(17(13)31-19)34-9-7-33(8-10-34)11-12-35-16-6-2-4-14-18(16)32-20(30-14)22(26,27)28/h1-6H,7-12H2,(H,29,31)(H,30,32) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Binding affinity to human 5HT1B receptor |
Bioorg Med Chem 16: 6617-40 (2008)
Article DOI: 10.1016/j.bmc.2008.05.024
BindingDB Entry DOI: 10.7270/Q2FB52Q7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM50244213
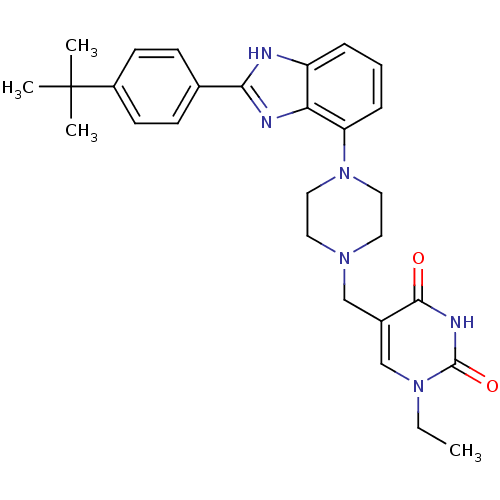
(5-((4-(2-(4-tert-butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES CCn1cc(CN2CCN(CC2)c2cccc3[nH]c(nc23)-c2ccc(cc2)C(C)(C)C)c(=O)[nH]c1=O Show InChI InChI=1S/C28H34N6O2/c1-5-33-18-20(26(35)31-27(33)36)17-32-13-15-34(16-14-32)23-8-6-7-22-24(23)30-25(29-22)19-9-11-21(12-10-19)28(2,3)4/h6-12,18H,5,13-17H2,1-4H3,(H,29,30)(H,31,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Binding affinity to 5HT2A receptor (unknown origin) |
Bioorg Med Chem 16: 6617-40 (2008)
Article DOI: 10.1016/j.bmc.2008.05.024
BindingDB Entry DOI: 10.7270/Q2FB52Q7 |
More data for this
Ligand-Target Pair | |
Substance-K receptor
(Homo sapiens (Human)) | BDBM50256882
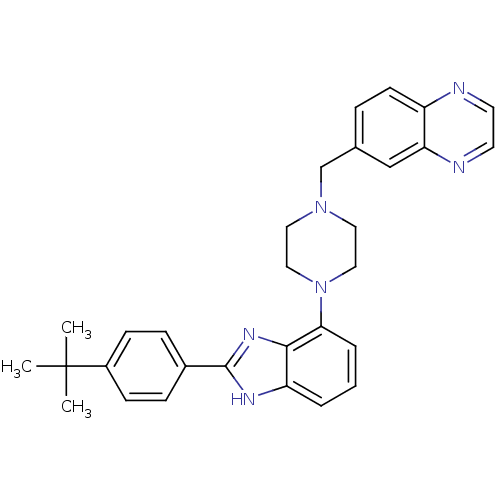
(6-((4-(2-(4-tert-butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES CC(C)(C)c1ccc(cc1)-c1nc2c(cccc2[nH]1)N1CCN(Cc2ccc3nccnc3c2)CC1 Show InChI InChI=1S/C30H32N6/c1-30(2,3)23-10-8-22(9-11-23)29-33-25-5-4-6-27(28(25)34-29)36-17-15-35(16-18-36)20-21-7-12-24-26(19-21)32-14-13-31-24/h4-14,19H,15-18,20H2,1-3H3,(H,33,34) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human neurokinin NK2 receptor |
J Med Chem 52: 2148-52 (2009)
Article DOI: 10.1021/jm801572m
BindingDB Entry DOI: 10.7270/Q2ZC82RT |
More data for this
Ligand-Target Pair | |
Histamine H2 receptor
(Homo sapiens (Human)) | BDBM50256882

(6-((4-(2-(4-tert-butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES CC(C)(C)c1ccc(cc1)-c1nc2c(cccc2[nH]1)N1CCN(Cc2ccc3nccnc3c2)CC1 Show InChI InChI=1S/C30H32N6/c1-30(2,3)23-10-8-22(9-11-23)29-33-25-5-4-6-27(28(25)34-29)36-17-15-35(16-18-36)20-21-7-12-24-26(19-21)32-14-13-31-24/h4-14,19H,15-18,20H2,1-3H3,(H,33,34) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of human histamine H2 receptor |
J Med Chem 52: 2148-52 (2009)
Article DOI: 10.1021/jm801572m
BindingDB Entry DOI: 10.7270/Q2ZC82RT |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118382
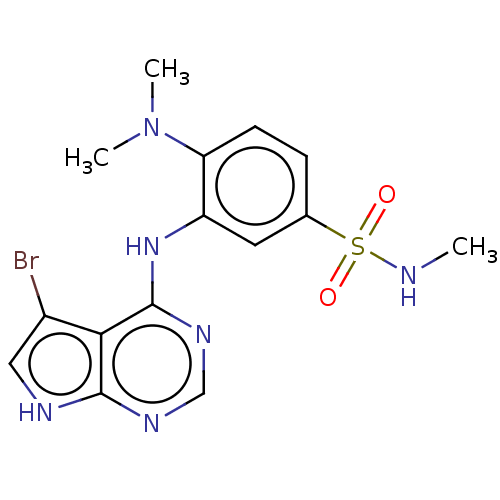
(CHEMBL3613198)Show SMILES CNS(=O)(=O)c1ccc(N(C)C)c(Nc2ncnc3[nH]cc(Br)c23)c1 Show InChI InChI=1S/C15H17BrN6O2S/c1-17-25(23,24)9-4-5-12(22(2)3)11(6-9)21-15-13-10(16)7-18-14(13)19-8-20-15/h4-8,17H,1-3H3,(H2,18,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118381

(CHEMBL3613197)Show SMILES CNS(=O)(=O)c1ccc(N(C)C)c(Nc2ncnc3[nH]cc(C)c23)c1 Show InChI InChI=1S/C16H20N6O2S/c1-10-8-18-15-14(10)16(20-9-19-15)21-12-7-11(25(23,24)17-2)5-6-13(12)22(3)4/h5-9,17H,1-4H3,(H2,18,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118384

(CHEMBL3613305)Show SMILES CNS(=O)(=O)c1ccc(N2CCOCC2)c(Nc2ncnc3[nH]cc(Br)c23)c1 Show InChI InChI=1S/C17H19BrN6O3S/c1-19-28(25,26)11-2-3-14(24-4-6-27-7-5-24)13(8-11)23-17-15-12(18)9-20-16(15)21-10-22-17/h2-3,8-10,19H,4-7H2,1H3,(H2,20,21,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118386
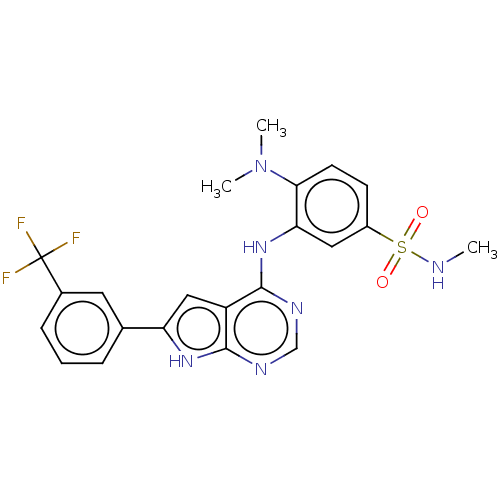
(CHEMBL3613307)Show SMILES CNS(=O)(=O)c1ccc(N(C)C)c(Nc2ncnc3[nH]c(cc23)-c2cccc(c2)C(F)(F)F)c1 Show InChI InChI=1S/C22H21F3N6O2S/c1-26-34(32,33)15-7-8-19(31(2)3)18(10-15)30-21-16-11-17(29-20(16)27-12-28-21)13-5-4-6-14(9-13)22(23,24)25/h4-12,26H,1-3H3,(H2,27,28,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase TNNI3K
(Homo sapiens (Human)) | BDBM50118382

(CHEMBL3613198)Show SMILES CNS(=O)(=O)c1ccc(N(C)C)c(Nc2ncnc3[nH]cc(Br)c23)c1 Show InChI InChI=1S/C15H17BrN6O2S/c1-17-25(23,24)9-4-5-12(22(2)3)11(6-9)21-15-13-10(16)7-18-14(13)19-8-20-15/h4-8,17H,1-3H3,(H2,18,19,20,21) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human myc-tagged TNNI3K autophosphorylation overexpressed in HEKMSRII cells preincubated for 30 mins followed by pervanadate solution a... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118464
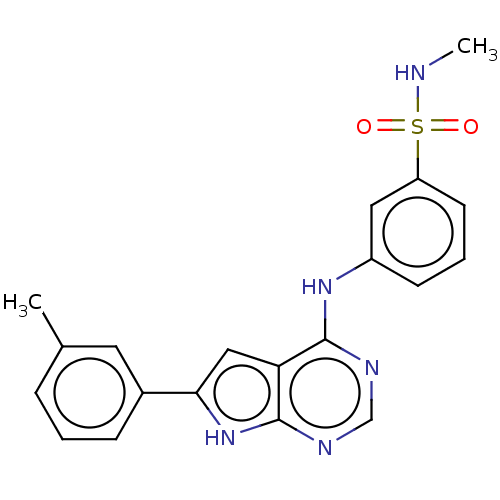
(CHEMBL3613186)Show SMILES CNS(=O)(=O)c1cccc(Nc2ncnc3[nH]c(cc23)-c2cccc(C)c2)c1 Show InChI InChI=1S/C20H19N5O2S/c1-13-5-3-6-14(9-13)18-11-17-19(22-12-23-20(17)25-18)24-15-7-4-8-16(10-15)28(26,27)21-2/h3-12,21H,1-2H3,(H2,22,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118468
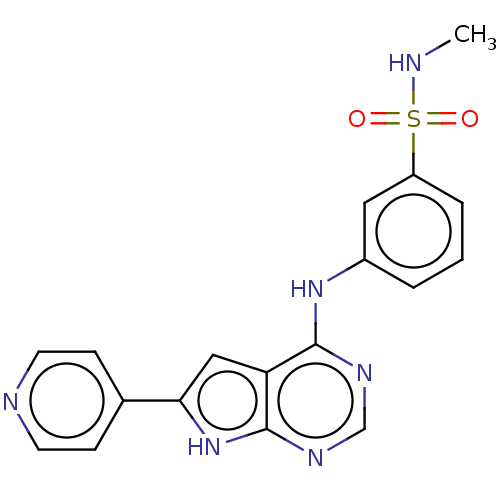
(CHEMBL3613190)Show SMILES CNS(=O)(=O)c1cccc(Nc2ncnc3[nH]c(cc23)-c2ccncc2)c1 Show InChI InChI=1S/C18H16N6O2S/c1-19-27(25,26)14-4-2-3-13(9-14)23-17-15-10-16(12-5-7-20-8-6-12)24-18(15)22-11-21-17/h2-11,19H,1H3,(H2,21,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118467
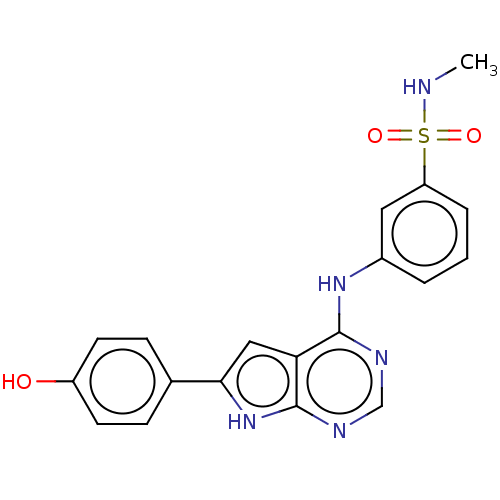
(CHEMBL3613189)Show SMILES CNS(=O)(=O)c1cccc(Nc2ncnc3[nH]c(cc23)-c2ccc(O)cc2)c1 Show InChI InChI=1S/C19H17N5O3S/c1-20-28(26,27)15-4-2-3-13(9-15)23-18-16-10-17(24-19(16)22-11-21-18)12-5-7-14(25)8-6-12/h2-11,20,25H,1H3,(H2,21,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118465
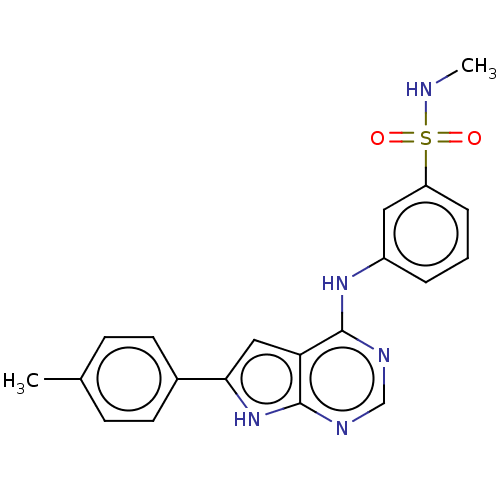
(CHEMBL3613187)Show SMILES CNS(=O)(=O)c1cccc(Nc2ncnc3[nH]c(cc23)-c2ccc(C)cc2)c1 Show InChI InChI=1S/C20H19N5O2S/c1-13-6-8-14(9-7-13)18-11-17-19(22-12-23-20(17)25-18)24-15-4-3-5-16(10-15)28(26,27)21-2/h3-12,21H,1-2H3,(H2,22,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118428

(CHEMBL3613184)Show SMILES CNS(=O)(=O)c1cccc(Nc2ncnc3[nH]c(cc23)-c2ccccc2)c1 Show InChI InChI=1S/C19H17N5O2S/c1-20-27(25,26)15-9-5-8-14(10-15)23-18-16-11-17(13-6-3-2-4-7-13)24-19(16)22-12-21-18/h2-12,20H,1H3,(H2,21,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118420
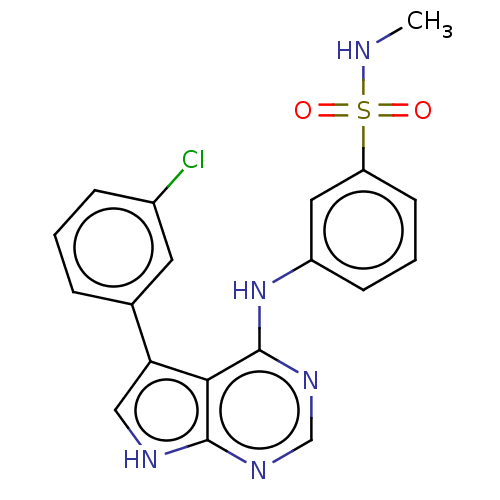
(CHEMBL3613177)Show SMILES CNS(=O)(=O)c1cccc(Nc2ncnc3[nH]cc(-c4cccc(Cl)c4)c23)c1 Show InChI InChI=1S/C19H16ClN5O2S/c1-21-28(26,27)15-7-3-6-14(9-15)25-19-17-16(10-22-18(17)23-11-24-19)12-4-2-5-13(20)8-12/h2-11,21H,1H3,(H2,22,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
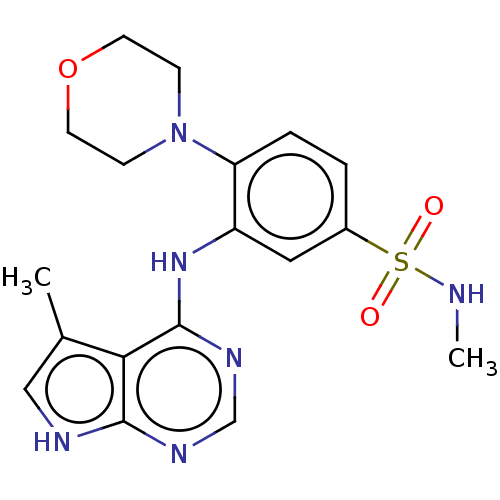
(Homo sapiens (Human)) | BDBM50118383
(CHEMBL3613199)Show SMILES CNS(=O)(=O)c1ccc(N2CCOCC2)c(Nc2ncnc3[nH]cc(C)c23)c1 Show InChI InChI=1S/C18H22N6O3S/c1-12-10-20-17-16(12)18(22-11-21-17)23-14-9-13(28(25,26)19-2)3-4-15(14)24-5-7-27-8-6-24/h3-4,9-11,19H,5-8H2,1-2H3,(H2,20,21,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118388
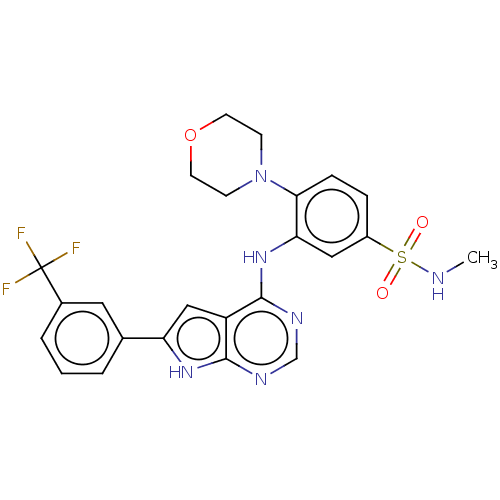
(CHEMBL3613309)Show SMILES CNS(=O)(=O)c1ccc(N2CCOCC2)c(Nc2ncnc3[nH]c(cc23)-c2cccc(c2)C(F)(F)F)c1 Show InChI InChI=1S/C24H23F3N6O3S/c1-28-37(34,35)17-5-6-21(33-7-9-36-10-8-33)20(12-17)32-23-18-13-19(31-22(18)29-14-30-23)15-3-2-4-16(11-15)24(25,26)27/h2-6,11-14,28H,7-10H2,1H3,(H2,29,30,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase TNNI3K
(Homo sapiens (Human)) | BDBM50118381

(CHEMBL3613197)Show SMILES CNS(=O)(=O)c1ccc(N(C)C)c(Nc2ncnc3[nH]cc(C)c23)c1 Show InChI InChI=1S/C16H20N6O2S/c1-10-8-18-15-14(10)16(20-9-19-15)21-12-7-11(25(23,24)17-2)5-6-13(12)22(3)4/h5-9,17H,1-4H3,(H2,18,19,20,21) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human myc-tagged TNNI3K autophosphorylation overexpressed in HEKMSRII cells preincubated for 30 mins followed by pervanadate solution a... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50244213

(5-((4-(2-(4-tert-butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES CCn1cc(CN2CCN(CC2)c2cccc3[nH]c(nc23)-c2ccc(cc2)C(C)(C)C)c(=O)[nH]c1=O Show InChI InChI=1S/C28H34N6O2/c1-5-33-18-20(26(35)31-27(33)36)17-32-13-15-34(16-14-32)23-8-6-7-22-24(23)30-25(29-22)19-9-11-21(12-10-19)28(2,3)4/h6-12,18H,5,13-17H2,1-4H3,(H,29,30)(H,31,35,36) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [125I]-(D-Trp6)-GnRH from human GnRH receptor |
Bioorg Med Chem Lett 19: 1986-90 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.043
BindingDB Entry DOI: 10.7270/Q28S4PS4 |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50244213

(5-((4-(2-(4-tert-butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES CCn1cc(CN2CCN(CC2)c2cccc3[nH]c(nc23)-c2ccc(cc2)C(C)(C)C)c(=O)[nH]c1=O Show InChI InChI=1S/C28H34N6O2/c1-5-33-18-20(26(35)31-27(33)36)17-32-13-15-34(16-14-32)23-8-6-7-22-24(23)30-25(29-22)19-9-11-21(12-10-19)28(2,3)4/h6-12,18H,5,13-17H2,1-4H3,(H,29,30)(H,31,35,36) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [125I]-(D-Trp6)LHRH from human recombinant GnRH receptor |
Bioorg Med Chem 16: 6617-40 (2008)
Article DOI: 10.1016/j.bmc.2008.05.024
BindingDB Entry DOI: 10.7270/Q2FB52Q7 |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50256836
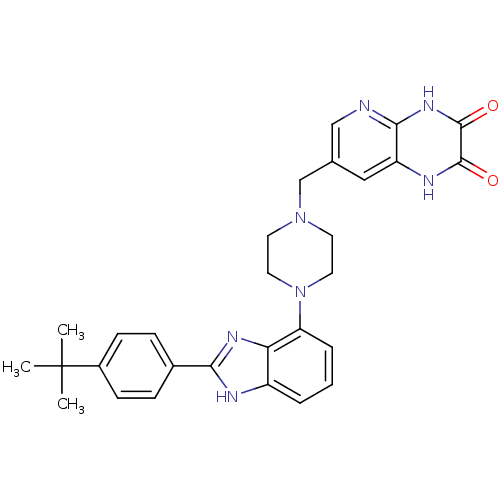
(7-((4-(2-(4-tert-butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES CC(C)(C)c1ccc(cc1)-c1nc2c(cccc2[nH]1)N1CCN(Cc2cnc3[nH]c(=O)c(=O)[nH]c3c2)CC1 Show InChI InChI=1S/C29H31N7O2/c1-29(2,3)20-9-7-19(8-10-20)25-31-21-5-4-6-23(24(21)33-25)36-13-11-35(12-14-36)17-18-15-22-26(30-16-18)34-28(38)27(37)32-22/h4-10,15-16H,11-14,17H2,1-3H3,(H,31,33)(H,32,37)(H,30,34,38) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [D-Trp6]-GnRH from human recombinant GnRH receptor |
J Med Chem 52: 2148-52 (2009)
Article DOI: 10.1021/jm801572m
BindingDB Entry DOI: 10.7270/Q2ZC82RT |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50315277

(5-((4-(2-(4-tert-butylphenyl)imidazo[1,2-a]pyridin...)Show SMILES CCn1cc(CN2CCN(CC2)c2cccn3cc(nc23)-c2ccc(cc2)C(C)(C)C)c(=O)[nH]c1=O Show InChI InChI=1S/C28H34N6O2/c1-5-32-18-21(26(35)30-27(32)36)17-31-13-15-33(16-14-31)24-7-6-12-34-19-23(29-25(24)34)20-8-10-22(11-9-20)28(2,3)4/h6-12,18-19H,5,13-17H2,1-4H3,(H,30,35,36) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [125I]D-Trp6-GnRH from human recombinant GNRH receptor by scintillation counting |
Bioorg Med Chem Lett 20: 2512-5 (2010)
Article DOI: 10.1016/j.bmcl.2010.02.099
BindingDB Entry DOI: 10.7270/Q2W0962G |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1A
(Homo sapiens (Human)) | BDBM50243853
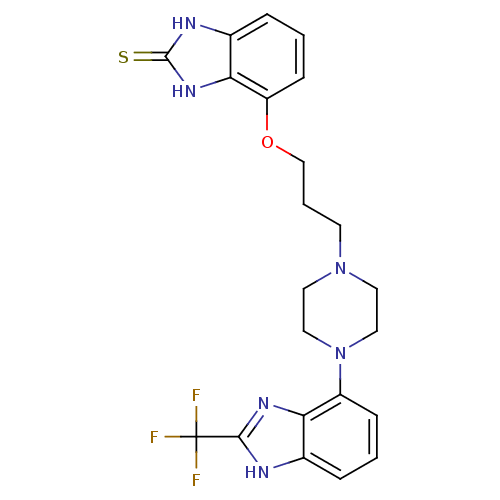
(4-(3-(4-(2-(Trifluoromethyl)-1H-benzo[d]imidazol-4...)Show SMILES FC(F)(F)c1nc2c(cccc2[nH]1)N1CCN(CCCOc2cccc3[nH]c(=S)[nH]c23)CC1 Show InChI InChI=1S/C22H23F3N6OS/c23-22(24,25)20-26-14-4-1-6-16(18(14)28-20)31-11-9-30(10-12-31)8-3-13-32-17-7-2-5-15-19(17)29-21(33)27-15/h1-2,4-7H,3,8-13H2,(H,26,28)(H2,27,29,33) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Binding affinity to human 5HT1A receptor |
Bioorg Med Chem 16: 6617-40 (2008)
Article DOI: 10.1016/j.bmc.2008.05.024
BindingDB Entry DOI: 10.7270/Q2FB52Q7 |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50256834
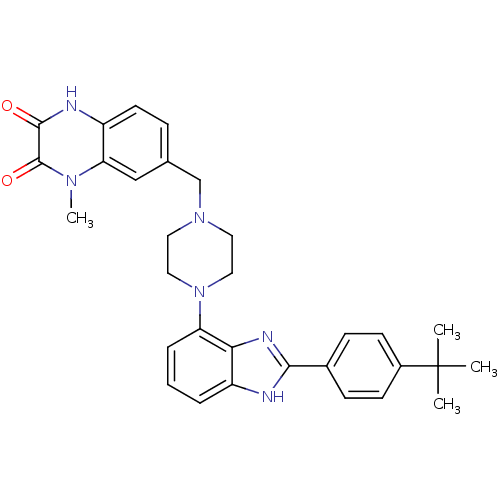
(7-((4-(2-(4-tert-butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES Cn1c2cc(CN3CCN(CC3)c3cccc4[nH]c(nc34)-c3ccc(cc3)C(C)(C)C)ccc2[nH]c(=O)c1=O Show InChI InChI=1S/C31H34N6O2/c1-31(2,3)22-11-9-21(10-12-22)28-32-24-6-5-7-25(27(24)34-28)37-16-14-36(15-17-37)19-20-8-13-23-26(18-20)35(4)30(39)29(38)33-23/h5-13,18H,14-17,19H2,1-4H3,(H,32,34)(H,33,38) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [D-Trp6]-GnRH from human recombinant GnRH receptor |
J Med Chem 52: 2148-52 (2009)
Article DOI: 10.1021/jm801572m
BindingDB Entry DOI: 10.7270/Q2ZC82RT |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118418
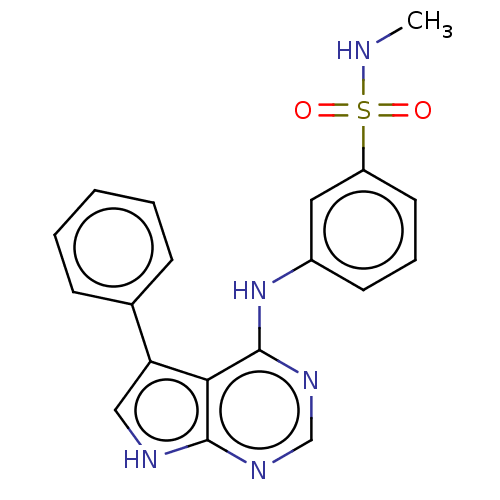
(CHEMBL3612667)Show SMILES CNS(=O)(=O)c1cccc(Nc2ncnc3[nH]cc(-c4ccccc4)c23)c1 Show InChI InChI=1S/C19H17N5O2S/c1-20-27(25,26)15-9-5-8-14(10-15)24-19-17-16(13-6-3-2-4-7-13)11-21-18(17)22-12-23-19/h2-12,20H,1H3,(H2,21,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118421

(CHEMBL3613178)Show SMILES CNS(=O)(=O)c1cccc(Nc2ncnc3[nH]cc(-c4ccc(Cl)cc4)c23)c1 Show InChI InChI=1S/C19H16ClN5O2S/c1-21-28(26,27)15-4-2-3-14(9-15)25-19-17-16(10-22-18(17)23-11-24-19)12-5-7-13(20)8-6-12/h2-11,21H,1H3,(H2,22,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118422
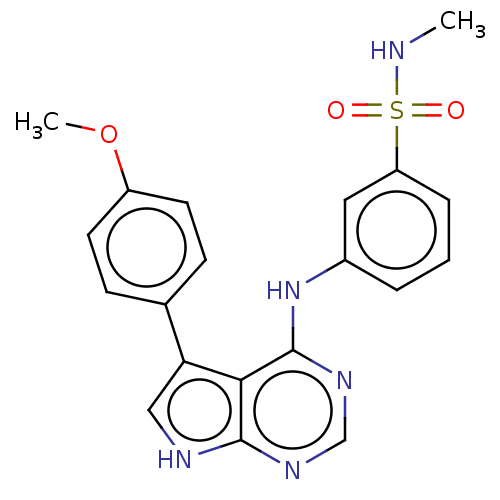
(CHEMBL3613179)Show SMILES CNS(=O)(=O)c1cccc(Nc2ncnc3[nH]cc(-c4ccc(OC)cc4)c23)c1 Show InChI InChI=1S/C20H19N5O3S/c1-21-29(26,27)16-5-3-4-14(10-16)25-20-18-17(11-22-19(18)23-12-24-20)13-6-8-15(28-2)9-7-13/h3-12,21H,1-2H3,(H2,22,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50244213

(5-((4-(2-(4-tert-butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES CCn1cc(CN2CCN(CC2)c2cccc3[nH]c(nc23)-c2ccc(cc2)C(C)(C)C)c(=O)[nH]c1=O Show InChI InChI=1S/C28H34N6O2/c1-5-33-18-20(26(35)31-27(33)36)17-32-13-15-34(16-14-32)23-8-6-7-22-24(23)30-25(29-22)19-9-11-21(12-10-19)28(2,3)4/h6-12,18H,5,13-17H2,1-4H3,(H,29,30)(H,31,35,36) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant GnRH receptor assessed as reduction in (D-Trp6)LHRH-induced myo-(1,2)-[3H]inositol production |
Bioorg Med Chem 16: 6617-40 (2008)
Article DOI: 10.1016/j.bmc.2008.05.024
BindingDB Entry DOI: 10.7270/Q2FB52Q7 |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50244213

(5-((4-(2-(4-tert-butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES CCn1cc(CN2CCN(CC2)c2cccc3[nH]c(nc23)-c2ccc(cc2)C(C)(C)C)c(=O)[nH]c1=O Show InChI InChI=1S/C28H34N6O2/c1-5-33-18-20(26(35)31-27(33)36)17-32-13-15-34(16-14-32)23-8-6-7-22-24(23)30-25(29-22)19-9-11-21(12-10-19)28(2,3)4/h6-12,18H,5,13-17H2,1-4H3,(H,29,30)(H,31,35,36) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant GnRH receptor expressed in HEK293 cells assessed as reduction in (D-Trp6)-GnRH-stimulated IP production by w... |
Bioorg Med Chem Lett 19: 1986-90 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.043
BindingDB Entry DOI: 10.7270/Q28S4PS4 |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50315276

(5-((4-(2-(4-tert-butylphenyl)imidazo[1,2-a]pyridin...)Show SMILES CC(C)(C)c1ccc(cc1)-c1cn2cccc(N3CCN(Cc4c[nH]c(=O)[nH]c4=O)CC3)c2n1 Show InChI InChI=1S/C26H30N6O2/c1-26(2,3)20-8-6-18(7-9-20)21-17-32-10-4-5-22(23(32)28-21)31-13-11-30(12-14-31)16-19-15-27-25(34)29-24(19)33/h4-10,15,17H,11-14,16H2,1-3H3,(H2,27,29,33,34) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [125I]D-Trp6-GnRH from human recombinant GNRH receptor by scintillation counting |
Bioorg Med Chem Lett 20: 2512-5 (2010)
Article DOI: 10.1016/j.bmcl.2010.02.099
BindingDB Entry DOI: 10.7270/Q2W0962G |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50244211

(6-((4-(2-(4-tert-butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES CC(C)(C)c1ccc(cc1)-c1nc2c(cccc2[nH]1)N1CCN(Cc2ccc3[nH]c(=O)c(=O)[nH]c3c2)CC1 Show InChI InChI=1S/C30H32N6O2/c1-30(2,3)21-10-8-20(9-11-21)27-31-23-5-4-6-25(26(23)34-27)36-15-13-35(14-16-36)18-19-7-12-22-24(17-19)33-29(38)28(37)32-22/h4-12,17H,13-16,18H2,1-3H3,(H,31,34)(H,32,37)(H,33,38) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [D-Trp6]-GnRH from human recombinant GnRH receptor |
J Med Chem 52: 2148-52 (2009)
Article DOI: 10.1021/jm801572m
BindingDB Entry DOI: 10.7270/Q2ZC82RT |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50244211

(6-((4-(2-(4-tert-butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES CC(C)(C)c1ccc(cc1)-c1nc2c(cccc2[nH]1)N1CCN(Cc2ccc3[nH]c(=O)c(=O)[nH]c3c2)CC1 Show InChI InChI=1S/C30H32N6O2/c1-30(2,3)21-10-8-20(9-11-21)27-31-23-5-4-6-25(26(23)34-27)36-15-13-35(14-16-36)18-19-7-12-22-24(17-19)33-29(38)28(37)32-22/h4-12,17H,13-16,18H2,1-3H3,(H,31,34)(H,32,37)(H,33,38) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [125I]-(D-Trp6)LHRH from human recombinant GnRH receptor |
Bioorg Med Chem 16: 6617-40 (2008)
Article DOI: 10.1016/j.bmc.2008.05.024
BindingDB Entry DOI: 10.7270/Q2FB52Q7 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118417
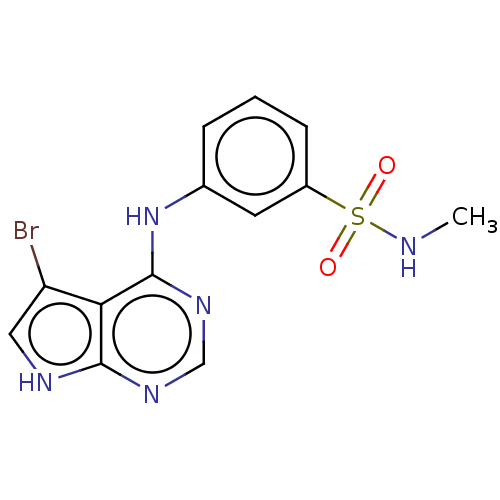
(CHEMBL3612666)Show InChI InChI=1S/C13H12BrN5O2S/c1-15-22(20,21)9-4-2-3-8(5-9)19-13-11-10(14)6-16-12(11)17-7-18-13/h2-7,15H,1H3,(H2,16,17,18,19) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50315277

(5-((4-(2-(4-tert-butylphenyl)imidazo[1,2-a]pyridin...)Show SMILES CCn1cc(CN2CCN(CC2)c2cccn3cc(nc23)-c2ccc(cc2)C(C)(C)C)c(=O)[nH]c1=O Show InChI InChI=1S/C28H34N6O2/c1-5-32-18-21(26(35)30-27(32)36)17-31-13-15-33(16-14-31)24-7-6-12-34-19-23(29-25(24)34)20-8-10-22(11-9-20)28(2,3)4/h6-12,18-19H,5,13-17H2,1-4H3,(H,30,35,36) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Antagonist activity at human recombinant GNRH receptor assessed as inhibition of D-Trp6-GNRH-induced IP accumulation after 1 hr by rapid filtration a... |
Bioorg Med Chem Lett 20: 2512-5 (2010)
Article DOI: 10.1016/j.bmcl.2010.02.099
BindingDB Entry DOI: 10.7270/Q2W0962G |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50244212

(5-((4-(2-(4-tert-Butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES CC(C)(C)c1ccc(cc1)-c1nc2c(cccc2[nH]1)N1CCN(Cc2c[nH]c(=O)[nH]c2=O)CC1 Show InChI InChI=1S/C26H30N6O2/c1-26(2,3)19-9-7-17(8-10-19)23-28-20-5-4-6-21(22(20)29-23)32-13-11-31(12-14-32)16-18-15-27-25(34)30-24(18)33/h4-10,15H,11-14,16H2,1-3H3,(H,28,29)(H2,27,30,33,34) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [125I]-(D-Trp6)LHRH from human recombinant GnRH receptor |
Bioorg Med Chem 16: 6617-40 (2008)
Article DOI: 10.1016/j.bmc.2008.05.024
BindingDB Entry DOI: 10.7270/Q2FB52Q7 |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50256835
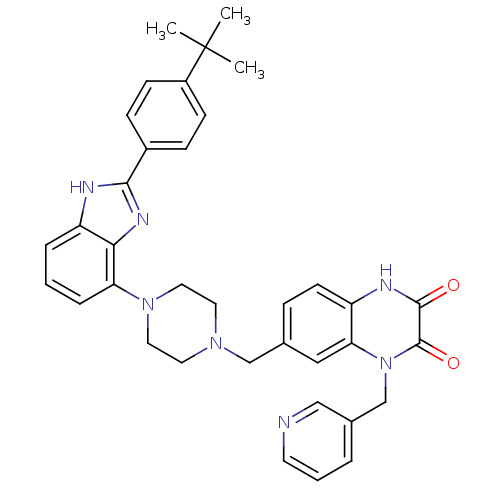
(7-((4-(2-(4-tert-butylphenyl)-1H-benzo[d]imidazol-...)Show SMILES CC(C)(C)c1ccc(cc1)-c1nc2c(cccc2[nH]1)N1CCN(Cc2ccc3[nH]c(=O)c(=O)n(Cc4cccnc4)c3c2)CC1 Show InChI InChI=1S/C36H37N7O2/c1-36(2,3)27-12-10-26(11-13-27)33-38-29-7-4-8-30(32(29)40-33)42-18-16-41(17-19-42)22-24-9-14-28-31(20-24)43(35(45)34(44)39-28)23-25-6-5-15-37-21-25/h4-15,20-21H,16-19,22-23H2,1-3H3,(H,38,40)(H,39,44) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [D-Trp6]-GnRH from human recombinant GnRH receptor |
J Med Chem 52: 2148-52 (2009)
Article DOI: 10.1021/jm801572m
BindingDB Entry DOI: 10.7270/Q2ZC82RT |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50244176
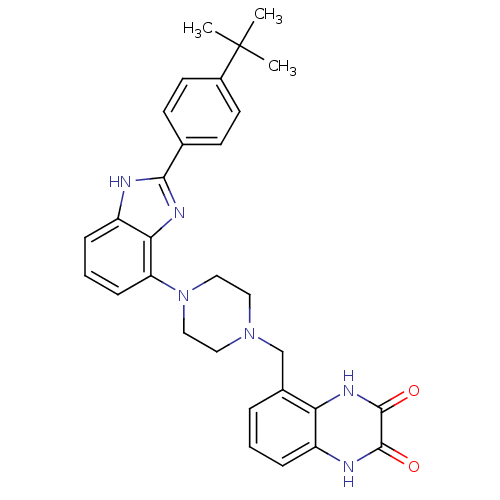
(5-({4-[2-(4-tert-Butylphenyl)-1H-benzimidazol-4-yl...)Show SMILES CC(C)(C)c1ccc(cc1)-c1nc2c(cccc2[nH]1)N1CCN(Cc2cccc3[nH]c(=O)c(=O)[nH]c23)CC1 Show InChI InChI=1S/C30H32N6O2/c1-30(2,3)21-12-10-19(11-13-21)27-31-23-8-5-9-24(26(23)33-27)36-16-14-35(15-17-36)18-20-6-4-7-22-25(20)34-29(38)28(37)32-22/h4-13H,14-18H2,1-3H3,(H,31,33)(H,32,37)(H,34,38) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [125I]-(D-Trp6)LHRH from human recombinant GnRH receptor |
Bioorg Med Chem 16: 6617-40 (2008)
Article DOI: 10.1016/j.bmc.2008.05.024
BindingDB Entry DOI: 10.7270/Q2FB52Q7 |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Rattus norvegicus) | BDBM50315277

(5-((4-(2-(4-tert-butylphenyl)imidazo[1,2-a]pyridin...)Show SMILES CCn1cc(CN2CCN(CC2)c2cccn3cc(nc23)-c2ccc(cc2)C(C)(C)C)c(=O)[nH]c1=O Show InChI InChI=1S/C28H34N6O2/c1-5-32-18-21(26(35)30-27(32)36)17-31-13-15-33(16-14-31)24-7-6-12-34-19-23(29-25(24)34)20-8-10-22(11-9-20)28(2,3)4/h6-12,18-19H,5,13-17H2,1-4H3,(H,30,35,36) | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [125I]D-Trp6-GnRH from rat recombinant GNRH receptor by scintillation counting |
Bioorg Med Chem Lett 20: 2512-5 (2010)
Article DOI: 10.1016/j.bmcl.2010.02.099
BindingDB Entry DOI: 10.7270/Q2W0962G |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50258205

(2-(4-tert-butylphenyl)-4-(4-((1-ethyl-5-methyl-1H-...)Show SMILES CCn1cnc(CN2CCN(CC2)c2cccc3[nH]c(nc23)-c2ccc(cc2)C(C)(C)C)c1C Show InChI InChI=1S/C28H36N6/c1-6-33-19-29-24(20(33)2)18-32-14-16-34(17-15-32)25-9-7-8-23-26(25)31-27(30-23)21-10-12-22(13-11-21)28(3,4)5/h7-13,19H,6,14-18H2,1-5H3,(H,30,31) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [125I]D-Trp6-GnRH from human recombinant GNRH receptor by scintillation counting |
Bioorg Med Chem Lett 20: 2512-5 (2010)
Article DOI: 10.1016/j.bmcl.2010.02.099
BindingDB Entry DOI: 10.7270/Q2W0962G |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118466
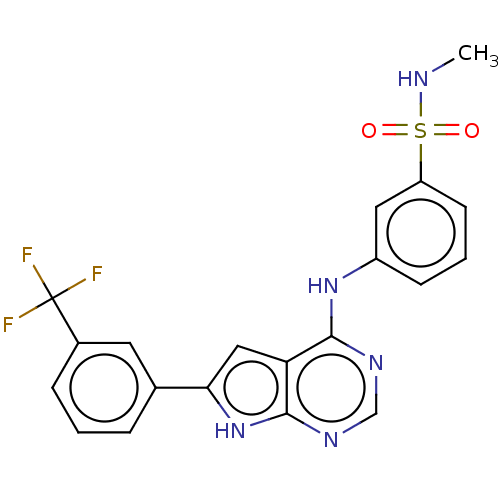
(CHEMBL3613188)Show SMILES CNS(=O)(=O)c1cccc(Nc2ncnc3[nH]c(cc23)-c2cccc(c2)C(F)(F)F)c1 Show InChI InChI=1S/C20H16F3N5O2S/c1-24-31(29,30)15-7-3-6-14(9-15)27-18-16-10-17(28-19(16)26-11-25-18)12-4-2-5-13(8-12)20(21,22)23/h2-11,24H,1H3,(H2,25,26,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118379
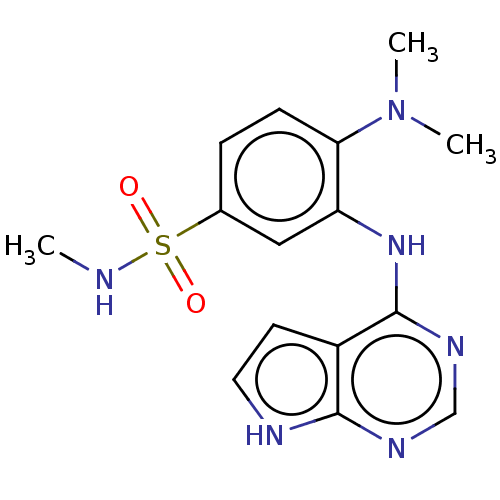
(CHEMBL3613195)Show SMILES CNS(=O)(=O)c1ccc(N(C)C)c(Nc2ncnc3[nH]ccc23)c1 Show InChI InChI=1S/C15H18N6O2S/c1-16-24(22,23)10-4-5-13(21(2)3)12(8-10)20-15-11-6-7-17-14(11)18-9-19-15/h4-9,16H,1-3H3,(H2,17,18,19,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118429
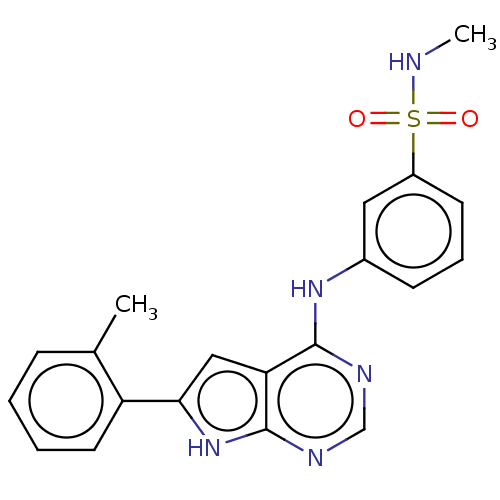
(CHEMBL3613185)Show SMILES CNS(=O)(=O)c1cccc(Nc2ncnc3[nH]c(cc23)-c2ccccc2C)c1 Show InChI InChI=1S/C20H19N5O2S/c1-13-6-3-4-9-16(13)18-11-17-19(22-12-23-20(17)25-18)24-14-7-5-8-15(10-14)28(26,27)21-2/h3-12,21H,1-2H3,(H2,22,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118424
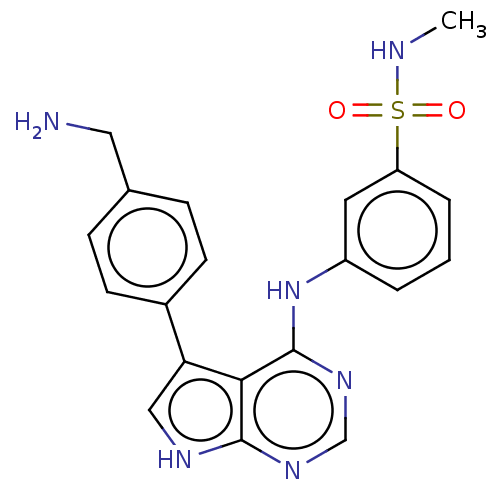
(CHEMBL3613181)Show SMILES CNS(=O)(=O)c1cccc(Nc2ncnc3[nH]cc(-c4ccc(CN)cc4)c23)c1 Show InChI InChI=1S/C20H20N6O2S/c1-22-29(27,28)16-4-2-3-15(9-16)26-20-18-17(11-23-19(18)24-12-25-20)14-7-5-13(10-21)6-8-14/h2-9,11-12,22H,10,21H2,1H3,(H2,23,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50244135
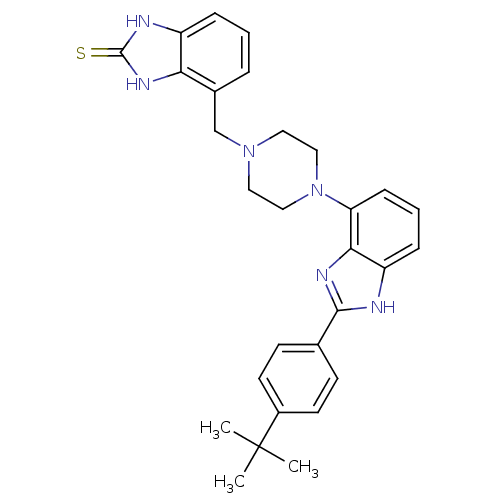
(4-{4-[2-(4-tert-Butyl-phenyl)-1H-benzoimidazol-4-y...)Show SMILES CC(C)(C)c1ccc(cc1)-c1nc2c(cccc2[nH]1)N1CCN(Cc2cccc3[nH]c(=S)[nH]c23)CC1 Show InChI InChI=1S/C29H32N6S/c1-29(2,3)21-12-10-19(11-13-21)27-30-23-8-5-9-24(26(23)32-27)35-16-14-34(15-17-35)18-20-6-4-7-22-25(20)33-28(36)31-22/h4-13H,14-18H2,1-3H3,(H,30,32)(H2,31,33,36) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [125I]-(D-Trp6)LHRH from human recombinant GnRH receptor |
Bioorg Med Chem 16: 6617-40 (2008)
Article DOI: 10.1016/j.bmc.2008.05.024
BindingDB Entry DOI: 10.7270/Q2FB52Q7 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50118385
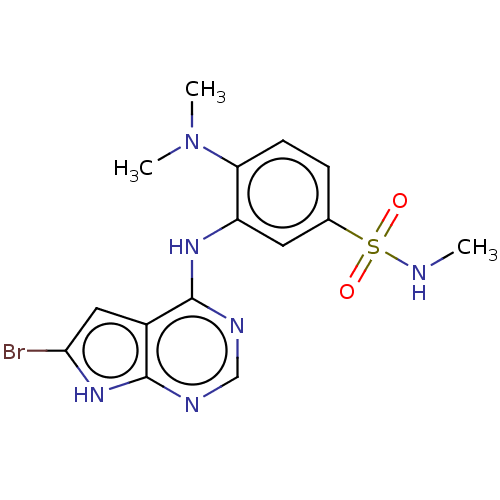
(CHEMBL3613306)Show SMILES CNS(=O)(=O)c1ccc(N(C)C)c(Nc2ncnc3[nH]c(Br)cc23)c1 Show InChI InChI=1S/C15H17BrN6O2S/c1-17-25(23,24)9-4-5-12(22(2)3)11(6-9)20-14-10-7-13(16)21-15(10)19-8-18-14/h4-8,17H,1-3H3,(H2,18,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of His6-tagged full length BRAF V600E mutant (2 to 766 residues) (unknown origin) expressed in Baculovirus expression system by BRAMA meth... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase TNNI3K
(Homo sapiens (Human)) | BDBM50118384

(CHEMBL3613305)Show SMILES CNS(=O)(=O)c1ccc(N2CCOCC2)c(Nc2ncnc3[nH]cc(Br)c23)c1 Show InChI InChI=1S/C17H19BrN6O3S/c1-19-28(25,26)11-2-3-14(24-4-6-27-7-5-24)13(8-11)23-17-15-12(18)9-20-16(15)21-10-22-17/h2-3,8-10,19H,4-7H2,1H3,(H2,20,21,22,23) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 6.30 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
Curated by ChEMBL
| Assay Description
Inhibition of human myc-tagged TNNI3K autophosphorylation overexpressed in HEKMSRII cells preincubated for 30 mins followed by pervanadate solution a... |
J Med Chem 58: 7431-48 (2015)
Article DOI: 10.1021/acs.jmedchem.5b00931
BindingDB Entry DOI: 10.7270/Q2FT8NV1 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Gonadotropin-releasing hormone receptor
(Homo sapiens (Human)) | BDBM50315275
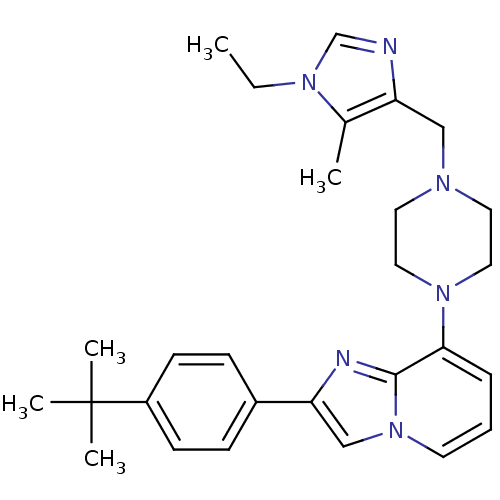
(2-(4-tert-butylphenyl)-8-(4-((1-ethyl-5-methyl-1H-...)Show SMILES CCn1cnc(CN2CCN(CC2)c2cccn3cc(nc23)-c2ccc(cc2)C(C)(C)C)c1C Show InChI InChI=1S/C28H36N6/c1-6-32-20-29-24(21(32)2)18-31-14-16-33(17-15-31)26-8-7-13-34-19-25(30-27(26)34)22-9-11-23(12-10-22)28(3,4)5/h7-13,19-20H,6,14-18H2,1-5H3 | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Displacement of [125I]D-Trp6-GnRH from human recombinant GNRH receptor by scintillation counting |
Bioorg Med Chem Lett 20: 2512-5 (2010)
Article DOI: 10.1016/j.bmcl.2010.02.099
BindingDB Entry DOI: 10.7270/Q2W0962G |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data