

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lethal factor (Bacillus anthracis) | BDBM50379543 (CHEMBL2012752) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of N-terminal 6-histine tagged Bacillus anthracis LF 263-C terminal catalytic domain using MCA-KKVYPYPME-Dap(Dnp)-NH2 as substrate after 4... | Bioorg Med Chem Lett 22: 2242-6 (2012) Article DOI: 10.1016/j.bmcl.2012.01.095 BindingDB Entry DOI: 10.7270/Q2MC912Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50329261 ((R)-2-(4-fluoro-3,5-dimethylphenylamino)-6-(4-fluo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate after 4 hrs by FRET assay | Bioorg Med Chem Lett 21: 2030-3 (2011) Article DOI: 10.1016/j.bmcl.2011.02.010 BindingDB Entry DOI: 10.7270/Q24M94VB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50329261 ((R)-2-(4-fluoro-3,5-dimethylphenylamino)-6-(4-fluo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate by FRET assay | Bioorg Med Chem Lett 20: 6850-3 (2010) Article DOI: 10.1016/j.bmcl.2010.08.058 BindingDB Entry DOI: 10.7270/Q2222V01 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50218667 (APC-1390 | CHEMBL46809) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Binding affinity to 5-HT3 serotonin receptor in NG 108-15 neuroblastoma glioma cells using [3H]-GR-65,630 radioligand. | Bioorg Med Chem Lett 11: 1629-33 (2001) BindingDB Entry DOI: 10.7270/Q2R78DHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor VII/Tissue factor (Homo sapiens (Human)) | BDBM50103651 (2-(3'-Amino-5-chloro-2-hydroxy-biphenyl-3-yl)-1H-b...) | PDB KEGG UniProtKB/SwissProt DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.0780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Binding affinity for factor VIIa/TF | Bioorg Med Chem Lett 11: 2253-6 (2001) BindingDB Entry DOI: 10.7270/Q2XG9QD9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50093157 (CHEMBL431969 | Derivative of piperazine-1-carboxyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Evaluated for its inhibitory potency against tryptase | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50093175 (CHEMBL311655 | Derivative of APC-2059) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Evaluated for its inhibitory potency against tryptase | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50379542 (CHEMBL2012753) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of N-terminal 6-histine tagged Bacillus anthracis LF 263-C terminal catalytic domain using MCA-KKVYPYPME-Dap(Dnp)-NH2 as substrate after 4... | Bioorg Med Chem Lett 22: 2242-6 (2012) Article DOI: 10.1016/j.bmcl.2012.01.095 BindingDB Entry DOI: 10.7270/Q2MC912Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50340754 ((S)-2-(4-fluoro-3,5-dimethylbenzyl)-6-(4-fluoroben...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of N-terminal 6-histine tagged Bacillus anthracis LF 263-C terminal catalytic domain using MCA-KKVYPYPME-Dap(Dnp)-NH2 as substrate after 4... | Bioorg Med Chem Lett 22: 2242-6 (2012) Article DOI: 10.1016/j.bmcl.2012.01.095 BindingDB Entry DOI: 10.7270/Q2MC912Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50340754 ((S)-2-(4-fluoro-3,5-dimethylbenzyl)-6-(4-fluoroben...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate after 4 hrs by FRET assay | Bioorg Med Chem Lett 21: 2030-3 (2011) Article DOI: 10.1016/j.bmcl.2011.02.010 BindingDB Entry DOI: 10.7270/Q24M94VB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50379536 (CHEMBL2012836) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of N-terminal 6-histine tagged Bacillus anthracis LF 263-C terminal catalytic domain using MCA-KKVYPYPME-Dap(Dnp)-NH2 as substrate after 4... | Bioorg Med Chem Lett 22: 2242-6 (2012) Article DOI: 10.1016/j.bmcl.2012.01.095 BindingDB Entry DOI: 10.7270/Q2MC912Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50093192 (CHEMBL311482 | Derivative of piperazine-1-carboxyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Evaluated for its inhibitory potency against tryptase | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50379541 (CHEMBL2012832) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of N-terminal 6-histine tagged Bacillus anthracis LF 263-C terminal catalytic domain using MCA-KKVYPYPME-Dap(Dnp)-NH2 as substrate after 4... | Bioorg Med Chem Lett 22: 2242-6 (2012) Article DOI: 10.1016/j.bmcl.2012.01.095 BindingDB Entry DOI: 10.7270/Q2MC912Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50329265 ((R)-2-(4-fluoro-3,5-dimethylphenylamino)-6-(4-fluo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of N-terminal 6-histine tagged Bacillus anthracis LF 263-C terminal catalytic domain using MCA-KKVYPYPME-Dap(Dnp)-NH2 as substrate after 4... | Bioorg Med Chem Lett 22: 2242-6 (2012) Article DOI: 10.1016/j.bmcl.2012.01.095 BindingDB Entry DOI: 10.7270/Q2MC912Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50329265 ((R)-2-(4-fluoro-3,5-dimethylphenylamino)-6-(4-fluo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate by FRET assay | Bioorg Med Chem Lett 20: 6850-3 (2010) Article DOI: 10.1016/j.bmcl.2010.08.058 BindingDB Entry DOI: 10.7270/Q2222V01 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50329265 ((R)-2-(4-fluoro-3,5-dimethylphenylamino)-6-(4-fluo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate after 4 hrs by FRET assay | Bioorg Med Chem Lett 21: 2030-3 (2011) Article DOI: 10.1016/j.bmcl.2011.02.010 BindingDB Entry DOI: 10.7270/Q24M94VB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50340755 ((S)-2-(4-fluoro-3-methylbenzyl)-6-(4-fluorobenzyla...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate after 4 hrs by FRET assay | Bioorg Med Chem Lett 21: 2030-3 (2011) Article DOI: 10.1016/j.bmcl.2011.02.010 BindingDB Entry DOI: 10.7270/Q24M94VB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50379533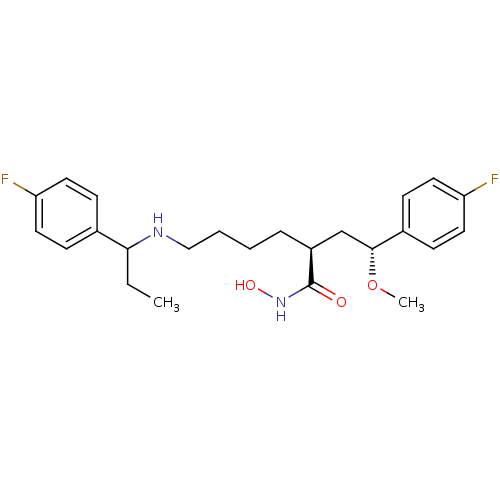 (CHEMBL2012838) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of N-terminal 6-histine tagged Bacillus anthracis LF 263-C terminal catalytic domain using MCA-KKVYPYPME-Dap(Dnp)-NH2 as substrate after 4... | Bioorg Med Chem Lett 22: 2242-6 (2012) Article DOI: 10.1016/j.bmcl.2012.01.095 BindingDB Entry DOI: 10.7270/Q2MC912Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50329270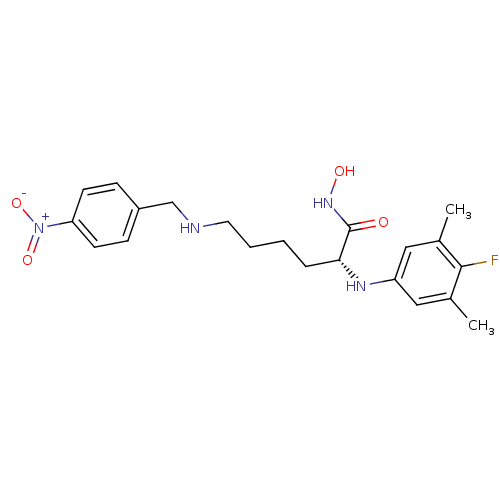 ((R)-2-(4-fluoro-3,5-dimethylphenylamino)-N-hydroxy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate by FRET assay | Bioorg Med Chem Lett 20: 6850-3 (2010) Article DOI: 10.1016/j.bmcl.2010.08.058 BindingDB Entry DOI: 10.7270/Q2222V01 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50379540 (CHEMBL2012750) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of N-terminal 6-histine tagged Bacillus anthracis LF 263-C terminal catalytic domain using MCA-KKVYPYPME-Dap(Dnp)-NH2 as substrate after 4... | Bioorg Med Chem Lett 22: 2242-6 (2012) Article DOI: 10.1016/j.bmcl.2012.01.095 BindingDB Entry DOI: 10.7270/Q2MC912Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50379535 (CHEMBL2010824) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of N-terminal 6-histine tagged Bacillus anthracis LF 263-C terminal catalytic domain using MCA-KKVYPYPME-Dap(Dnp)-NH2 as substrate after 4... | Bioorg Med Chem Lett 22: 2242-6 (2012) Article DOI: 10.1016/j.bmcl.2012.01.095 BindingDB Entry DOI: 10.7270/Q2MC912Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50340756 ((S)-6-(4-chlorobenzylamino)-2-(4-fluoro-3-methylbe...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate after 4 hrs by FRET assay | Bioorg Med Chem Lett 21: 2030-3 (2011) Article DOI: 10.1016/j.bmcl.2011.02.010 BindingDB Entry DOI: 10.7270/Q24M94VB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50340757 ((S)-2-(4-fluoro-3,5-dimethylbenzyl)-6-(4-fluoro-3-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate after 4 hrs by FRET assay | Bioorg Med Chem Lett 21: 2030-3 (2011) Article DOI: 10.1016/j.bmcl.2011.02.010 BindingDB Entry DOI: 10.7270/Q24M94VB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50093156 (CHEMBL432172 | Derivative of APC-2059) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Evaluated for its inhibitory potency against tryptase | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50101018 (1,4-di{4-[4-amino(imino)methylphenylcarboxamidomet...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Binding affinity to 5-HT3 serotonin receptor in NG 108-15 neuroblastoma glioma cells using [3H]-GR-65,630 radioligand. | Bioorg Med Chem Lett 11: 1629-33 (2001) BindingDB Entry DOI: 10.7270/Q2R78DHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50093167 (CHEMBL75750 | Derivative of APC-2059) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Evaluated for its inhibitory potency against tryptase | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50093154 (CHEMBL448786 | Derivative of piperazine-1-carboxyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | PubMed | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Evaluated for its inhibitory potency against tryptase | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50329267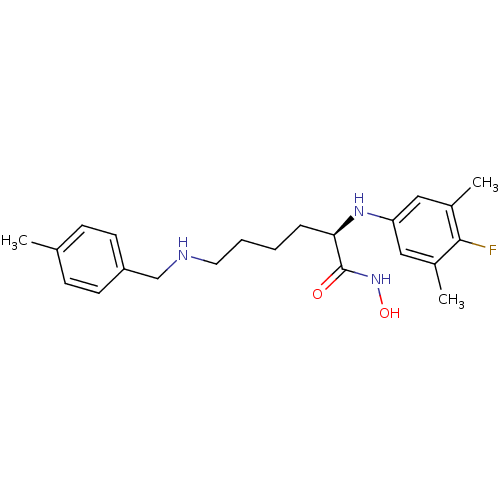 ((R)-2-(4-fluoro-3,5-dimethylphenylamino)-N-hydroxy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate by FRET assay | Bioorg Med Chem Lett 20: 6850-3 (2010) Article DOI: 10.1016/j.bmcl.2010.08.058 BindingDB Entry DOI: 10.7270/Q2222V01 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50329268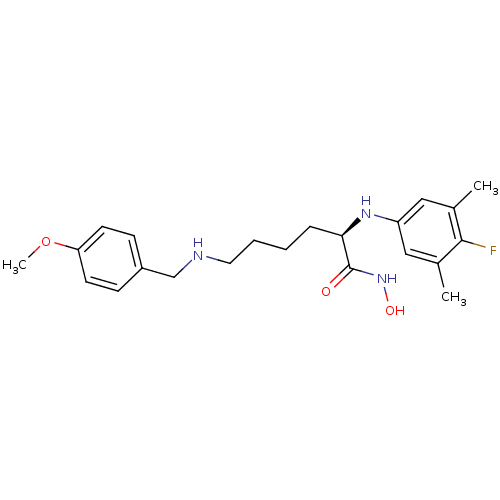 ((R)-2-(4-fluoro-3,5-dimethylphenylamino)-N-hydroxy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate by FRET assay | Bioorg Med Chem Lett 20: 6850-3 (2010) Article DOI: 10.1016/j.bmcl.2010.08.058 BindingDB Entry DOI: 10.7270/Q2222V01 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Urokinase-type plasminogen activator (Homo sapiens (Human)) | BDBM50103651 (2-(3'-Amino-5-chloro-2-hydroxy-biphenyl-3-yl)-1H-b...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Binding affinity of the compound towards urokinase-type plasminogen activator (microPa) | Bioorg Med Chem Lett 11: 2253-6 (2001) BindingDB Entry DOI: 10.7270/Q2XG9QD9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50329269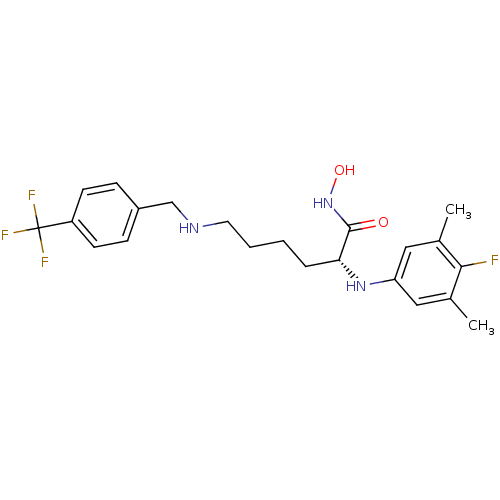 ((R)-2-(4-fluoro-3,5-dimethylphenylamino)-N-hydroxy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate by FRET assay | Bioorg Med Chem Lett 20: 6850-3 (2010) Article DOI: 10.1016/j.bmcl.2010.08.058 BindingDB Entry DOI: 10.7270/Q2222V01 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50379544 (CHEMBL2012751) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of N-terminal 6-histine tagged Bacillus anthracis LF 263-C terminal catalytic domain using MCA-KKVYPYPME-Dap(Dnp)-NH2 as substrate after 4... | Bioorg Med Chem Lett 22: 2242-6 (2012) Article DOI: 10.1016/j.bmcl.2012.01.095 BindingDB Entry DOI: 10.7270/Q2MC912Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50093173 (CHEMBL309830 | Derivative of piperazine-1-carboxyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Evaluated for its inhibitory potency against tryptase | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50093158 (1-{4-[4-amino(imino)methylaminobenzylcarbamoyl]hex...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Evaluated for its inhibitory potency against tryptase | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50101882 (CHEMBL53829 | Phosphoric acid mono-{2-[3-(3-benzyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Inhibitory activity against Coagulation factor X in human plasma | Bioorg Med Chem Lett 11: 1797-800 (2001) BindingDB Entry DOI: 10.7270/Q20K27TW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50101871 (3-[3-(3-Benzyl-5-carbamimidoyl-1H-indol-2-yl)-5-br...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Inhibitory activity against Coagulation factor X in human plasma | Bioorg Med Chem Lett 11: 1797-800 (2001) BindingDB Entry DOI: 10.7270/Q20K27TW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50103651 (2-(3'-Amino-5-chloro-2-hydroxy-biphenyl-3-yl)-1H-b...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Binding affinity of the compound towards Coagulation factor X | Bioorg Med Chem Lett 11: 2253-6 (2001) BindingDB Entry DOI: 10.7270/Q2XG9QD9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50093199 (CHEMBL75972 | Derivative of APC-2059) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Evaluated for its inhibitory potency against tryptase | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50218668 (CHEMBL42900) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Binding affinity to 5-HT3 serotonin receptor in NG 108-15 neuroblastoma glioma cells using [3H]-GR-65,630 radioligand. | Bioorg Med Chem Lett 11: 1629-33 (2001) BindingDB Entry DOI: 10.7270/Q2R78DHK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50340758 ((S)-6-(4-fluorobenzylamino)-2-((R)-2-(4-fluorophen...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 0.580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate after 4 hrs by FRET assay | Bioorg Med Chem Lett 21: 2030-3 (2011) Article DOI: 10.1016/j.bmcl.2011.02.010 BindingDB Entry DOI: 10.7270/Q24M94VB | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50340758 ((S)-6-(4-fluorobenzylamino)-2-((R)-2-(4-fluorophen...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | 0.580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of N-terminal 6-histine tagged Bacillus anthracis LF 263-C terminal catalytic domain using MCA-KKVYPYPME-Dap(Dnp)-NH2 as substrate after 4... | Bioorg Med Chem Lett 22: 2242-6 (2012) Article DOI: 10.1016/j.bmcl.2012.01.095 BindingDB Entry DOI: 10.7270/Q2MC912Q | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50379539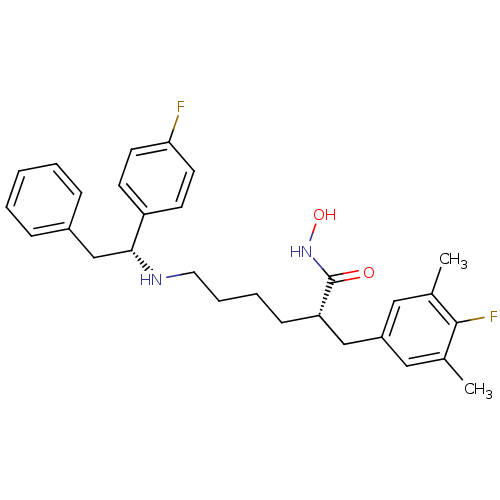 (CHEMBL2012834) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of N-terminal 6-histine tagged Bacillus anthracis LF 263-C terminal catalytic domain using MCA-KKVYPYPME-Dap(Dnp)-NH2 as substrate after 4... | Bioorg Med Chem Lett 22: 2242-6 (2012) Article DOI: 10.1016/j.bmcl.2012.01.095 BindingDB Entry DOI: 10.7270/Q2MC912Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2 (Homo sapiens (Human)) | BDBM50084616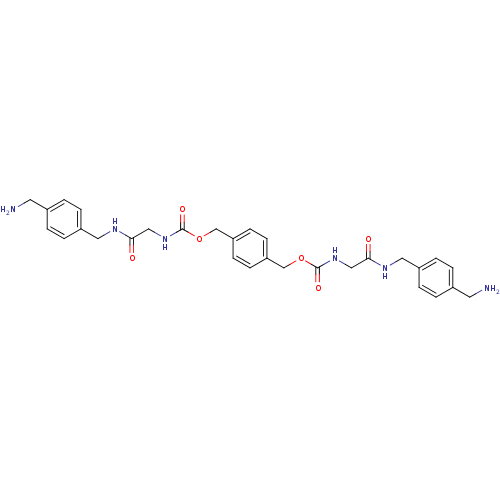 (CHEMBL310290 | [(4-Aminomethyl-benzylcarbamoyl)-me...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Inhibitory potency against tryptase | Bioorg Med Chem Lett 10: 2357-60 (2001) BindingDB Entry DOI: 10.7270/Q2NP23NX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50340759 ((R)-6-(4-fluoro-3-methylbenzylamino)-2-(4-fluoro-3...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate after 4 hrs by FRET assay | Bioorg Med Chem Lett 21: 2030-3 (2011) Article DOI: 10.1016/j.bmcl.2011.02.010 BindingDB Entry DOI: 10.7270/Q24M94VB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50340760 ((R)-6-(4-chlorobenzylamino)-2-(4-fluoro-3-methylph...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate after 4 hrs by FRET assay | Bioorg Med Chem Lett 21: 2030-3 (2011) Article DOI: 10.1016/j.bmcl.2011.02.010 BindingDB Entry DOI: 10.7270/Q24M94VB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50329263 ((R)-2-(4-fluoro-3,5-dimethylphenylamino)-6-(2-fluo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.690 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate by FRET assay | Bioorg Med Chem Lett 20: 6850-3 (2010) Article DOI: 10.1016/j.bmcl.2010.08.058 BindingDB Entry DOI: 10.7270/Q2222V01 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50101881 (4-[3-(3-Benzyl-5-carbamimidoyl-1H-indol-2-yl)-5-br...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Inhibitory activity against Coagulation factor X in human plasma | Bioorg Med Chem Lett 11: 1797-800 (2001) BindingDB Entry DOI: 10.7270/Q20K27TW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase beta-2/delta/gamma (Homo sapiens (Human)) | BDBM50093178 (CHEMBL76883 | Derivative of APC-2059) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Evaluated for its inhibitory potency against tryptase | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50340761 ((S)-6-(4-chlorobenzylamino)-2-(4-fluoro-3,5-dimeth...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.710 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate after 4 hrs by FRET assay | Bioorg Med Chem Lett 21: 2030-3 (2011) Article DOI: 10.1016/j.bmcl.2011.02.010 BindingDB Entry DOI: 10.7270/Q24M94VB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Lethal factor (Bacillus anthracis) | BDBM50340762 ((R)-6-(4-chlorobenzylamino)-2-(4-fluoro-3-methylph...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
PanThera Biopharma, LLC Curated by ChEMBL | Assay Description Inhibition of Bacillus anthracis lethal factor assessed as proteolysis using MCA-KKVYPYPME-Dap(Dnp)-NH2 peptide substrate after 4 hrs by FRET assay | Bioorg Med Chem Lett 21: 2030-3 (2011) Article DOI: 10.1016/j.bmcl.2011.02.010 BindingDB Entry DOI: 10.7270/Q24M94VB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 750 total ) | Next | Last >> |