 Found 10127 hits with Last Name = 'jo' and Initial = 'h'
Found 10127 hits with Last Name = 'jo' and Initial = 'h' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50095105
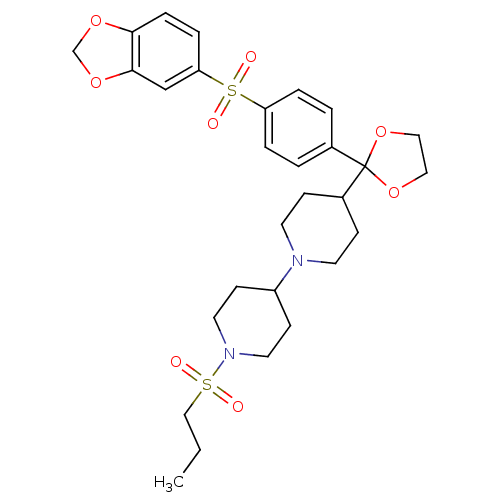
(4-{2-[4-(Benzo[1,3]dioxole-5-sulfonyl)-phenyl]-[1,...)Show SMILES CCCS(=O)(=O)N1CCC(CC1)N1CCC(CC1)C1(OCCO1)c1ccc(cc1)S(=O)(=O)c1ccc2OCOc2c1 Show InChI InChI=1S/C29H38N2O8S2/c1-2-19-40(32,33)31-15-11-24(12-16-31)30-13-9-23(10-14-30)29(38-17-18-39-29)22-3-5-25(6-4-22)41(34,35)26-7-8-27-28(20-26)37-21-36-27/h3-8,20,23-24H,2,9-19,21H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Antagonistic activity of the compound against Muscarinic acetylcholine receptor M2 |
Bioorg Med Chem Lett 11: 2311-4 (2001)
BindingDB Entry DOI: 10.7270/Q28S4P7Z |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50343152
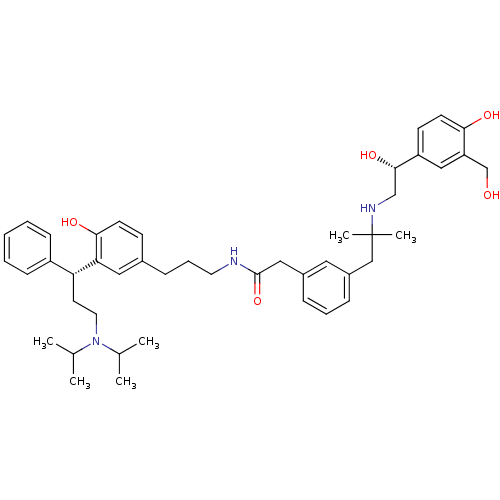
(CHEMBL1773196 | N-(3-(3-((R)-3-(diisopropylamino)-...)Show SMILES CC(C)N(CC[C@H](c1ccccc1)c1cc(CCCNC(=O)Cc2cccc(CC(C)(C)NC[C@H](O)c3ccc(O)c(CO)c3)c2)ccc1O)C(C)C |r| Show InChI InChI=1S/C45H61N3O5/c1-31(2)48(32(3)4)23-21-39(36-15-8-7-9-16-36)40-25-33(17-19-42(40)51)14-11-22-46-44(53)26-34-12-10-13-35(24-34)28-45(5,6)47-29-43(52)37-18-20-41(50)38(27-37)30-49/h7-10,12-13,15-20,24-25,27,31-32,39,43,47,49-52H,11,14,21-23,26,28-30H2,1-6H3,(H,46,53)/t39-,43+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0910 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl Scopolamine from human muscarinic M3 receptor expressed in CHO cells by scintillation proximity assay |
Bioorg Med Chem Lett 21: 2759-63 (2011)
Article DOI: 10.1016/j.bmcl.2010.10.132
BindingDB Entry DOI: 10.7270/Q2G73F1B |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50291847

(7-[3-(Quinolin-2-ylmethoxy)-benzyloxy]-2-(1H-tetra...)Show SMILES O=C1CC(Oc2cc(OCc3cccc(OCc4ccc5ccccc5n4)c3)ccc12)c1nnn[nH]1 Show InChI InChI=1S/C27H21N5O4/c33-24-14-26(27-29-31-32-30-27)36-25-13-21(10-11-22(24)25)34-15-17-4-3-6-20(12-17)35-16-19-9-8-18-5-1-2-7-23(18)28-19/h1-13,26H,14-16H2,(H,29,30,31,32) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhone-Poulenc Rorer Central Research
Curated by ChEMBL
| Assay Description
Binding affinity against LTD4 receptor in guinea pig lung membranes. |
J Med Chem 34: 1704-7 (1991)
BindingDB Entry DOI: 10.7270/Q2FJ2FR5 |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50291855

(7-[3-(Quinolin-2-ylmethoxy)-phenoxymethyl]-2-(1H-t...)Show SMILES O=C1CC(Oc2cc(COc3cccc(OCc4ccc5ccccc5n4)c3)ccc12)c1nnn[nH]1 Show InChI InChI=1S/C27H21N5O4/c33-24-14-26(27-29-31-32-30-27)36-25-12-17(8-11-22(24)25)15-34-20-5-3-6-21(13-20)35-16-19-10-9-18-4-1-2-7-23(18)28-19/h1-13,26H,14-16H2,(H,29,30,31,32) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhone-Poulenc Rorer Central Research
Curated by ChEMBL
| Assay Description
Binding affinity against LTD4 receptor in guinea pig lung membranes. |
J Med Chem 34: 1704-7 (1991)
BindingDB Entry DOI: 10.7270/Q2FJ2FR5 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50128835
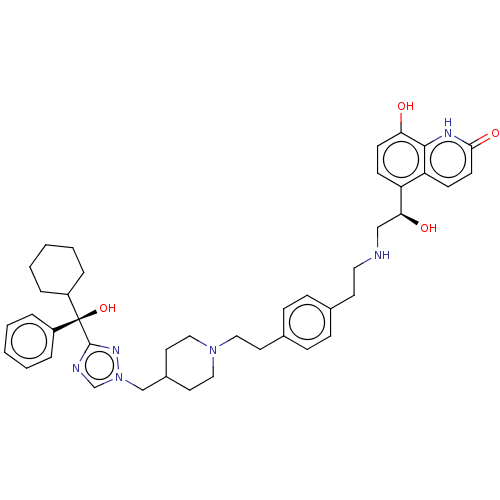
(CHEMBL3629360)Show SMILES O[C@@H](CNCCc1ccc(CCN2CCC(Cn3cnc(n3)[C@@](O)(C3CCCCC3)c3ccccc3)CC2)cc1)c1ccc(O)c2[nH]c(=O)ccc12 |r| Show InChI InChI=1S/C42H52N6O4/c49-37-17-15-35(36-16-18-39(51)45-40(36)37)38(50)27-43-23-19-30-11-13-31(14-12-30)20-24-47-25-21-32(22-26-47)28-48-29-44-41(46-48)42(52,33-7-3-1-4-8-33)34-9-5-2-6-10-34/h1,3-4,7-8,11-18,29,32,34,38,43,49-50,52H,2,5-6,9-10,19-28H2,(H,45,51)/t38-,42-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
Curated by ChEMBL
| Assay Description
Antagonist activity at muscarinic M2 receptor (unknown origin) |
Bioorg Med Chem Lett 25: 5121-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.10.008
BindingDB Entry DOI: 10.7270/Q2JQ12V7 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50103771
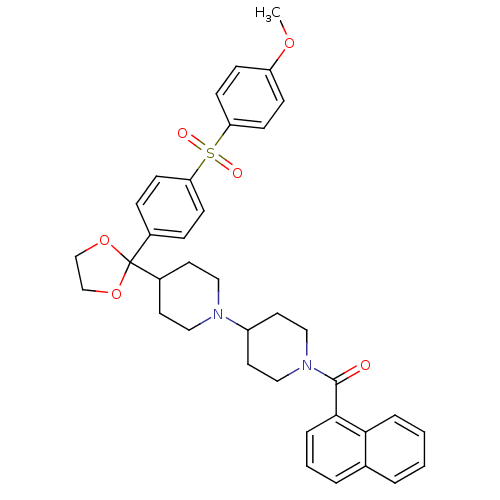
((4-{2-[4-(4-Methoxy-benzenesulfonyl)-phenyl]-[1,3]...)Show SMILES COc1ccc(cc1)S(=O)(=O)c1ccc(cc1)C1(OCCO1)C1CCN(CC1)C1CCN(CC1)C(=O)c1cccc2ccccc12 Show InChI InChI=1S/C37H40N2O6S/c1-43-31-11-15-33(16-12-31)46(41,42)32-13-9-28(10-14-32)37(44-25-26-45-37)29-17-21-38(22-18-29)30-19-23-39(24-20-30)36(40)35-8-4-6-27-5-2-3-7-34(27)35/h2-16,29-30H,17-26H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Antagonistic activity of the compound against muscarinic acetylcholine receptor M2 |
Bioorg Med Chem Lett 11: 2311-4 (2001)
BindingDB Entry DOI: 10.7270/Q28S4P7Z |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M1
(Homo sapiens (Human)) | BDBM50128835

(CHEMBL3629360)Show SMILES O[C@@H](CNCCc1ccc(CCN2CCC(Cn3cnc(n3)[C@@](O)(C3CCCCC3)c3ccccc3)CC2)cc1)c1ccc(O)c2[nH]c(=O)ccc12 |r| Show InChI InChI=1S/C42H52N6O4/c49-37-17-15-35(36-16-18-39(51)45-40(36)37)38(50)27-43-23-19-30-11-13-31(14-12-30)20-24-47-25-21-32(22-26-47)28-48-29-44-41(46-48)42(52,33-7-3-1-4-8-33)34-9-5-2-6-10-34/h1,3-4,7-8,11-18,29,32,34,38,43,49-50,52H,2,5-6,9-10,19-28H2,(H,45,51)/t38-,42-/m0/s1 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
Curated by ChEMBL
| Assay Description
Antagonist activity at muscarinic M1 receptor (unknown origin) |
Bioorg Med Chem Lett 25: 5121-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.10.008
BindingDB Entry DOI: 10.7270/Q2JQ12V7 |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1/2
(Homo sapiens (Human)) | BDBM50009075
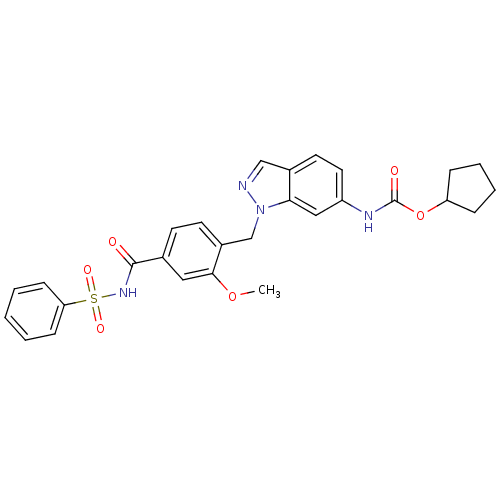
(CHEMBL22033 | ICI 198615 | ICI-198615 | [1-(4-Benz...)Show SMILES COc1cc(ccc1Cn1ncc2ccc(NC(=O)OC3CCCC3)cc12)C(=O)NS(=O)(=O)c1ccccc1 Show InChI InChI=1S/C28H28N4O6S/c1-37-26-15-19(27(33)31-39(35,36)24-9-3-2-4-10-24)11-12-21(26)18-32-25-16-22(14-13-20(25)17-29-32)30-28(34)38-23-7-5-6-8-23/h2-4,9-17,23H,5-8,18H2,1H3,(H,30,34)(H,31,33) | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rorer Central Research
Curated by ChEMBL
| Assay Description
In vitro binding affinity against cysteinyl leukotriene D4 receptor from guinea pig lung membrane |
J Med Chem 33: 2828-41 (1990)
BindingDB Entry DOI: 10.7270/Q29C6WDM |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50103774
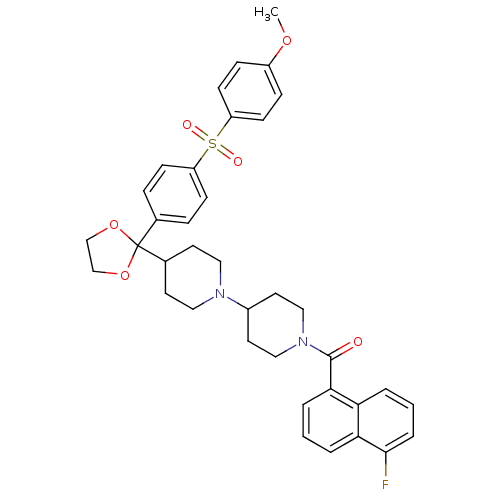
((5-Fluoro-naphthalen-1-yl)-(4-{2-[4-(4-methoxy-ben...)Show SMILES COc1ccc(cc1)S(=O)(=O)c1ccc(cc1)C1(OCCO1)C1CCN(CC1)C1CCN(CC1)C(=O)c1cccc2c(F)cccc12 Show InChI InChI=1S/C37H39FN2O6S/c1-44-29-10-14-31(15-11-29)47(42,43)30-12-8-26(9-13-30)37(45-24-25-46-37)27-16-20-39(21-17-27)28-18-22-40(23-19-28)36(41)34-6-2-5-33-32(34)4-3-7-35(33)38/h2-15,27-28H,16-25H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Antagonistic activity of the compound against muscarinic acetylcholine receptor M2 |
Bioorg Med Chem Lett 11: 2311-4 (2001)
BindingDB Entry DOI: 10.7270/Q28S4P7Z |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50103772
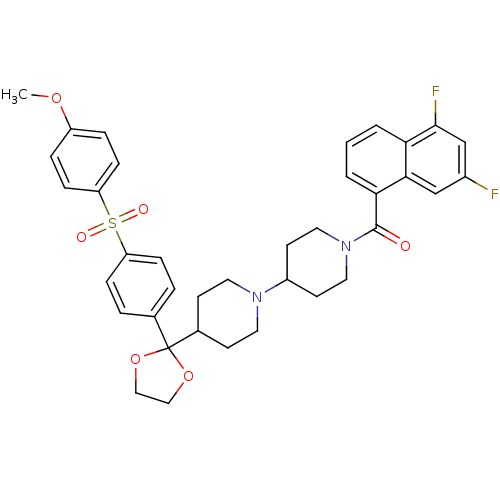
((5,7-Difluoro-naphthalen-1-yl)-(4-{2-[4-(4-methoxy...)Show SMILES COc1ccc(cc1)S(=O)(=O)c1ccc(cc1)C1(OCCO1)C1CCN(CC1)C1CCN(CC1)C(=O)c1cccc2c(F)cc(F)cc12 Show InChI InChI=1S/C37H38F2N2O6S/c1-45-29-7-11-31(12-8-29)48(43,44)30-9-5-25(6-10-30)37(46-21-22-47-37)26-13-17-40(18-14-26)28-15-19-41(20-16-28)36(42)33-4-2-3-32-34(33)23-27(38)24-35(32)39/h2-12,23-24,26,28H,13-22H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Antagonistic activity of the compound against Muscarinic acetylcholine receptor M2 |
Bioorg Med Chem Lett 11: 2311-4 (2001)
BindingDB Entry DOI: 10.7270/Q28S4P7Z |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50000287
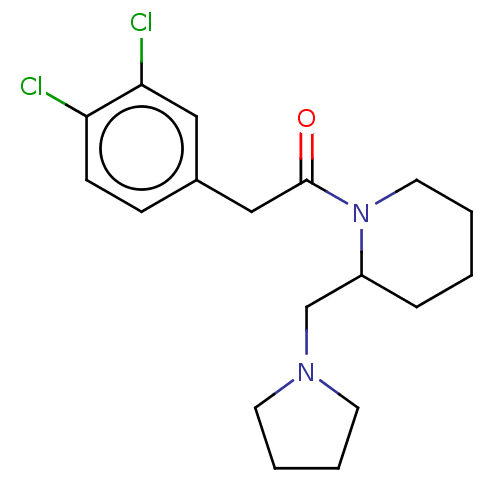
(2-(3,4-Dichloro-phenyl)-1-(2-pyrrolidin-1-ylmethyl...)Show InChI InChI=1S/C18H24Cl2N2O/c19-16-7-6-14(11-17(16)20)12-18(23)22-10-2-1-5-15(22)13-21-8-3-4-9-21/h6-7,11,15H,1-5,8-10,12-13H2 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.114079
BindingDB Entry DOI: 10.7270/Q2XK8KNF |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50343153
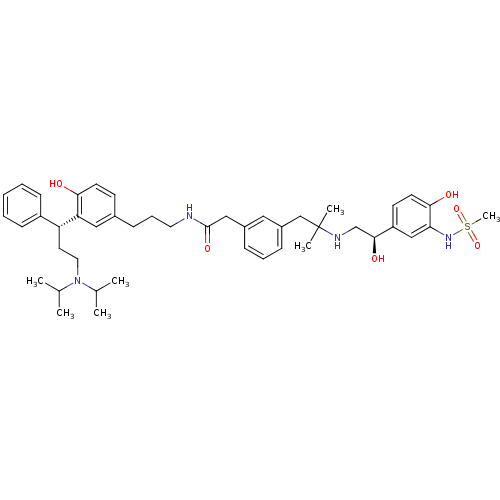
(CHEMBL1773197 | N-(3-(3-((R)-3-(diisopropylamino)-...)Show SMILES CC(C)N(CC[C@H](c1ccccc1)c1cc(CCCNC(=O)Cc2cccc(CC(C)(C)NC[C@H](O)c3ccc(O)c(NS(C)(=O)=O)c3)c2)ccc1O)C(C)C |r| Show InChI InChI=1S/C45H62N4O6S/c1-31(2)49(32(3)4)24-22-38(36-16-9-8-10-17-36)39-26-33(18-20-41(39)50)15-12-23-46-44(53)27-34-13-11-14-35(25-34)29-45(5,6)47-30-43(52)37-19-21-42(51)40(28-37)48-56(7,54)55/h8-11,13-14,16-21,25-26,28,31-32,38,43,47-48,50-52H,12,15,22-24,27,29-30H2,1-7H3,(H,46,53)/t38-,43+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.276 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl Scopolamine from human muscarinic M3 receptor expressed in CHO cells by scintillation proximity assay |
Bioorg Med Chem Lett 21: 2759-63 (2011)
Article DOI: 10.1016/j.bmcl.2010.10.132
BindingDB Entry DOI: 10.7270/Q2G73F1B |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50103767
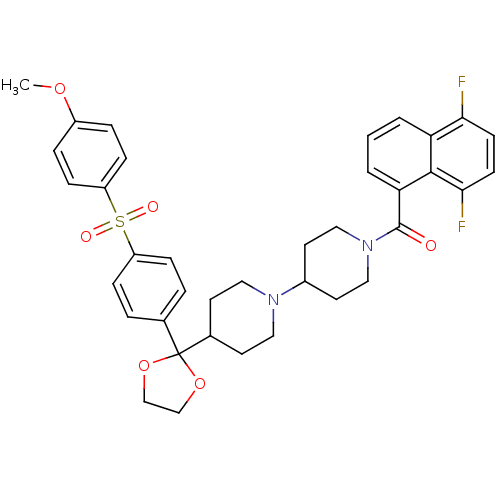
((5,8-Difluoro-naphthalen-1-yl)-(4-{2-[4-(4-methoxy...)Show SMILES COc1ccc(cc1)S(=O)(=O)c1ccc(cc1)C1(OCCO1)C1CCN(CC1)C1CCN(CC1)C(=O)c1cccc2c(F)ccc(F)c12 Show InChI InChI=1S/C37H38F2N2O6S/c1-45-28-7-11-30(12-8-28)48(43,44)29-9-5-25(6-10-29)37(46-23-24-47-37)26-15-19-40(20-16-26)27-17-21-41(22-18-27)36(42)32-4-2-3-31-33(38)13-14-34(39)35(31)32/h2-14,26-27H,15-24H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Antagonistic activity of the compound against Muscarinic acetylcholine receptor M2 |
Bioorg Med Chem Lett 11: 2311-4 (2001)
BindingDB Entry DOI: 10.7270/Q28S4P7Z |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50128835

(CHEMBL3629360)Show SMILES O[C@@H](CNCCc1ccc(CCN2CCC(Cn3cnc(n3)[C@@](O)(C3CCCCC3)c3ccccc3)CC2)cc1)c1ccc(O)c2[nH]c(=O)ccc12 |r| Show InChI InChI=1S/C42H52N6O4/c49-37-17-15-35(36-16-18-39(51)45-40(36)37)38(50)27-43-23-19-30-11-13-31(14-12-30)20-24-47-25-21-32(22-26-47)28-48-29-44-41(46-48)42(52,33-7-3-1-4-8-33)34-9-5-2-6-10-34/h1,3-4,7-8,11-18,29,32,34,38,43,49-50,52H,2,5-6,9-10,19-28H2,(H,45,51)/t38-,42-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl scopolamine from human cloned muscarinic M3 receptor by dilution method |
Bioorg Med Chem Lett 25: 5121-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.10.008
BindingDB Entry DOI: 10.7270/Q2JQ12V7 |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50009075

(CHEMBL22033 | ICI 198615 | ICI-198615 | [1-(4-Benz...)Show SMILES COc1cc(ccc1Cn1ncc2ccc(NC(=O)OC3CCCC3)cc12)C(=O)NS(=O)(=O)c1ccccc1 Show InChI InChI=1S/C28H28N4O6S/c1-37-26-15-19(27(33)31-39(35,36)24-9-3-2-4-10-24)11-12-21(26)18-32-25-16-22(14-13-20(25)17-29-32)30-28(34)38-23-7-5-6-8-23/h2-4,9-17,23H,5-8,18H2,1H3,(H,30,34)(H,31,33) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhone-Poulenc Rorer Central Research
Curated by ChEMBL
| Assay Description
Binding affinity against LTD4 receptor in guinea pig lung membranes. |
J Med Chem 34: 1704-7 (1991)
BindingDB Entry DOI: 10.7270/Q2FJ2FR5 |
More data for this
Ligand-Target Pair | |
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM35226
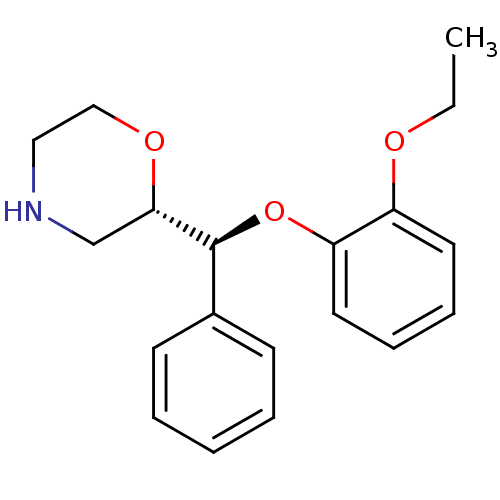
((S,S)-reboxetine | Reboxetine | Vestra)Show SMILES CCOc1ccccc1O[C@H]([C@@H]1CNCCO1)c1ccccc1 |r| Show InChI InChI=1S/C19H23NO3/c1-2-21-16-10-6-7-11-17(16)23-19(15-8-4-3-5-9-15)18-14-20-12-13-22-18/h3-11,18-20H,2,12-14H2,1H3/t18-,19-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.300 | -56.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Wyeth Research
| Assay Description
Compounds were evaluated the inhibition of [3H] nisoxetine binding to MDCK-Net6 cells, stably transfected with the human norepinephrine transporter (... |
Bioorg Med Chem 17: 7802-15 (2009)
Article DOI: 10.1016/j.bmc.2009.09.023
BindingDB Entry DOI: 10.7270/Q26Q1VK2 |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50291848
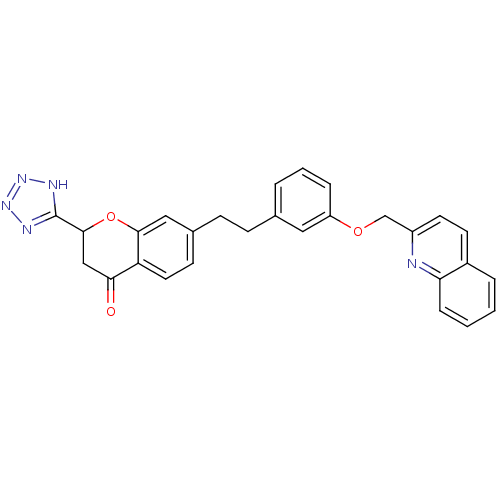
(7-{2-[3-(Quinolin-2-ylmethoxy)-phenyl]-ethyl}-2-(1...)Show SMILES O=C1CC(Oc2cc(CCc3cccc(OCc4ccc5ccccc5n4)c3)ccc12)c1nnn[nH]1 Show InChI InChI=1S/C28H23N5O3/c34-25-16-27(28-30-32-33-31-28)36-26-15-19(10-13-23(25)26)9-8-18-4-3-6-22(14-18)35-17-21-12-11-20-5-1-2-7-24(20)29-21/h1-7,10-15,27H,8-9,16-17H2,(H,30,31,32,33) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhone-Poulenc Rorer Central Research
Curated by ChEMBL
| Assay Description
Binding affinity against LTD4 receptor in guinea pig lung membranes. |
J Med Chem 34: 1704-7 (1991)
BindingDB Entry DOI: 10.7270/Q2FJ2FR5 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50343161
(5-((R)-2-(4-(4-(3-((R)-3-(diisopropylamino)-1-phen...)Show SMILES CC(C)N(CC[C@H](c1ccccc1)c1cc(CCCCOc2ccc(CCNC[C@H](O)c3ccc(O)c4[nH]c(=O)ccc34)cc2)ccc1O)C(C)C |r| Show InChI InChI=1S/C44H55N3O5/c1-30(2)47(31(3)4)26-24-36(34-11-6-5-7-12-34)39-28-33(15-20-40(39)48)10-8-9-27-52-35-16-13-32(14-17-35)23-25-45-29-42(50)37-18-21-41(49)44-38(37)19-22-43(51)46-44/h5-7,11-22,28,30-31,36,42,45,48-50H,8-10,23-27,29H2,1-4H3,(H,46,51)/t36-,42+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.305 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl Scopolamine from human muscarinic M3 receptor expressed in CHO cells by scintillation proximity assay |
Bioorg Med Chem Lett 21: 2759-63 (2011)
Article DOI: 10.1016/j.bmcl.2010.10.132
BindingDB Entry DOI: 10.7270/Q2G73F1B |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50343161

(5-((R)-2-(4-(4-(3-((R)-3-(diisopropylamino)-1-phen...)Show SMILES CC(C)N(CC[C@H](c1ccccc1)c1cc(CCCCOc2ccc(CCNC[C@H](O)c3ccc(O)c4[nH]c(=O)ccc34)cc2)ccc1O)C(C)C |r| Show InChI InChI=1S/C44H55N3O5/c1-30(2)47(31(3)4)26-24-36(34-11-6-5-7-12-34)39-28-33(15-20-40(39)48)10-8-9-27-52-35-16-13-32(14-17-35)23-25-45-29-42(50)37-18-21-41(49)44-38(37)19-22-43(51)46-44/h5-7,11-22,28,30-31,36,42,45,48-50H,8-10,23-27,29H2,1-4H3,(H,46,51)/t36-,42+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl scopolamine from human cloned muscarinic M3 receptor by dilution method |
Bioorg Med Chem Lett 25: 5121-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.10.008
BindingDB Entry DOI: 10.7270/Q2JQ12V7 |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50000296
(CHEMBL441765 | CHEMBL482811 | U-50488H | US1149237...)Show SMILES CN([C@@H]1CCCC[C@H]1N1CCCC1)C(=O)Cc1ccc(Cl)c(Cl)c1 |r| Show InChI InChI=1S/C19H26Cl2N2O/c1-22(19(24)13-14-8-9-15(20)16(21)12-14)17-6-2-3-7-18(17)23-10-4-5-11-23/h8-9,12,17-18H,2-7,10-11,13H2,1H3/t17-,18-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.114079
BindingDB Entry DOI: 10.7270/Q2XK8KNF |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50128837
(CHEMBL3629354)Show SMILES CN1CCC(Cn2cnc(n2)C(O)(c2ccccc2)c2ccccc2)CC1 Show InChI InChI=1S/C22H26N4O/c1-25-14-12-18(13-15-25)16-26-17-23-21(24-26)22(27,19-8-4-2-5-9-19)20-10-6-3-7-11-20/h2-11,17-18,27H,12-16H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl scopolamine from human cloned muscarinic M3 receptor by dilution method |
Bioorg Med Chem Lett 25: 5121-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.10.008
BindingDB Entry DOI: 10.7270/Q2JQ12V7 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M4
(Homo sapiens (Human)) | BDBM50128835

(CHEMBL3629360)Show SMILES O[C@@H](CNCCc1ccc(CCN2CCC(Cn3cnc(n3)[C@@](O)(C3CCCCC3)c3ccccc3)CC2)cc1)c1ccc(O)c2[nH]c(=O)ccc12 |r| Show InChI InChI=1S/C42H52N6O4/c49-37-17-15-35(36-16-18-39(51)45-40(36)37)38(50)27-43-23-19-30-11-13-31(14-12-30)20-24-47-25-21-32(22-26-47)28-48-29-44-41(46-48)42(52,33-7-3-1-4-8-33)34-9-5-2-6-10-34/h1,3-4,7-8,11-18,29,32,34,38,43,49-50,52H,2,5-6,9-10,19-28H2,(H,45,51)/t38-,42-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
Curated by ChEMBL
| Assay Description
Antagonist activity at muscarinic M4 receptor (unknown origin) |
Bioorg Med Chem Lett 25: 5121-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.10.008
BindingDB Entry DOI: 10.7270/Q2JQ12V7 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50518984
(CHEMBL4564189)Show SMILES CC(C)NC[C@H](O)COc1ccccc1CCCCNC(=O)NCCCCc1ccccc1OC[C@@H](O)CNC(C)C |r| Show InChI InChI=1S/C33H54N4O5/c1-25(2)36-21-29(38)23-41-31-17-7-5-13-27(31)15-9-11-19-34-33(40)35-20-12-10-16-28-14-6-8-18-32(28)42-24-30(39)22-37-26(3)4/h5-8,13-14,17-18,25-26,29-30,36-39H,9-12,15-16,19-24H2,1-4H3,(H2,34,35,40)/t29-,30-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen
Curated by ChEMBL
| Assay Description
Displacement of [3H]DHA from beta2 adrenergic receptor (unknown origin) stably expressed in HEK293 cell membranes measured after 90 mins by scintilla... |
J Med Chem 62: 7806-7839 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00595
BindingDB Entry DOI: 10.7270/Q2PG1W4K |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50343154
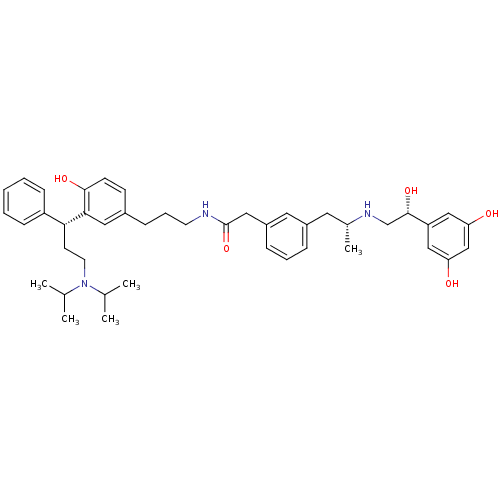
(2-(3-((R)-2-((R)-2-(3,5-dihydroxyphenyl)-2-hydroxy...)Show SMILES CC(C)N(CC[C@H](c1ccccc1)c1cc(CCCNC(=O)Cc2cccc(C[C@@H](C)NC[C@H](O)c3cc(O)cc(O)c3)c2)ccc1O)C(C)C |r| Show InChI InChI=1S/C43H57N3O5/c1-29(2)46(30(3)4)20-18-39(35-14-7-6-8-15-35)40-23-32(16-17-41(40)49)13-10-19-44-43(51)24-34-12-9-11-33(22-34)21-31(5)45-28-42(50)36-25-37(47)27-38(48)26-36/h6-9,11-12,14-17,22-23,25-27,29-31,39,42,45,47-50H,10,13,18-21,24,28H2,1-5H3,(H,44,51)/t31-,39-,42+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.397 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl Scopolamine from human muscarinic M3 receptor expressed in CHO cells by scintillation proximity assay |
Bioorg Med Chem Lett 21: 2759-63 (2011)
Article DOI: 10.1016/j.bmcl.2010.10.132
BindingDB Entry DOI: 10.7270/Q2G73F1B |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50518969
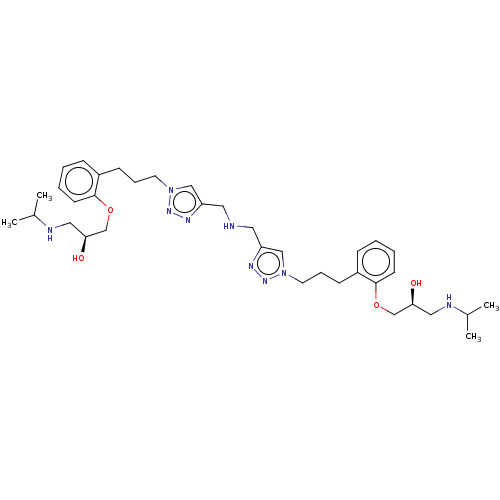
(CHEMBL4514723)Show SMILES CC(C)NC[C@H](O)COc1ccccc1CCCn1cc(CNCc2cn(CCCc3ccccc3OC[C@@H](O)CNC(C)C)nn2)nn1 |r| Show InChI InChI=1S/C36H55N9O4/c1-27(2)38-21-33(46)25-48-35-15-7-5-11-29(35)13-9-17-44-23-31(40-42-44)19-37-20-32-24-45(43-41-32)18-10-14-30-12-6-8-16-36(30)49-26-34(47)22-39-28(3)4/h5-8,11-12,15-16,23-24,27-28,33-34,37-39,46-47H,9-10,13-14,17-22,25-26H2,1-4H3/t33-,34-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.437 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen
Curated by ChEMBL
| Assay Description
Displacement of [3H]DHA from beta2 adrenergic receptor (unknown origin) stably expressed in HEK293 cell membranes measured after 90 mins by scintilla... |
J Med Chem 62: 7806-7839 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00595
BindingDB Entry DOI: 10.7270/Q2PG1W4K |
More data for this
Ligand-Target Pair | |
Kappa-type opioid receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50027433
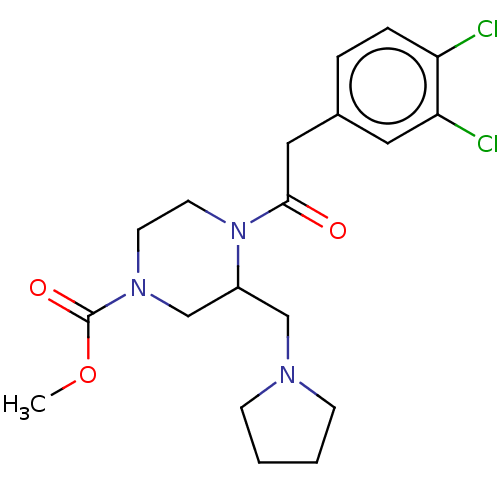
(CHEMBL603370 | GR-89696)Show SMILES COC(=O)N1CCN(C(CN2CCCC2)C1)C(=O)Cc1ccc(Cl)c(Cl)c1 Show InChI InChI=1S/C19H25Cl2N3O3/c1-27-19(26)23-8-9-24(15(13-23)12-22-6-2-3-7-22)18(25)11-14-4-5-16(20)17(21)10-14/h4-5,10,15H,2-3,6-9,11-13H2,1H3 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2021.114079
BindingDB Entry DOI: 10.7270/Q2XK8KNF |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
5-hydroxytryptamine receptor 2A
(Homo sapiens (Human)) | BDBM35255

(2-Chlor-11-(2-dimethylaminoaethoxy)-dibenzo(b,f)-t...)Show InChI InChI=1S/C18H18ClNOS/c1-20(2)9-10-21-16-11-13-5-3-4-6-17(13)22-18-8-7-14(19)12-15(16)18/h3-8,11-12H,9-10H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
| Assay Description
Compounds were evaluated the inhibition of [3H] ketanserin binding to membranes from CHO cells, stably transfected with the human 5-HT2A receptor. Da... |
Bioorg Med Chem 17: 7802-15 (2009)
Article DOI: 10.1016/j.bmc.2009.09.023
BindingDB Entry DOI: 10.7270/Q26Q1VK2 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50103773
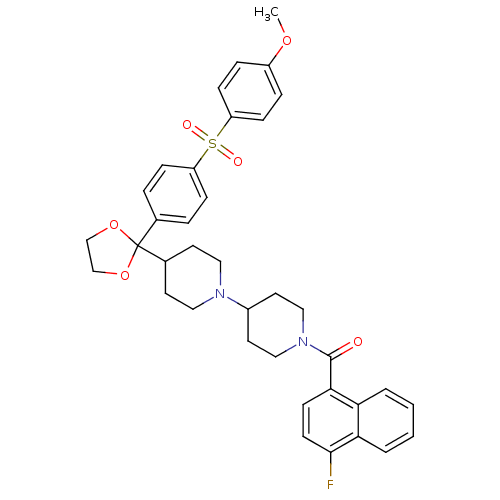
((4-Fluoro-naphthalen-1-yl)-(4-{2-[4-(4-methoxy-ben...)Show SMILES COc1ccc(cc1)S(=O)(=O)c1ccc(cc1)C1(OCCO1)C1CCN(CC1)C1CCN(CC1)C(=O)c1ccc(F)c2ccccc12 Show InChI InChI=1S/C37H39FN2O6S/c1-44-29-8-12-31(13-9-29)47(42,43)30-10-6-26(7-11-30)37(45-24-25-46-37)27-16-20-39(21-17-27)28-18-22-40(23-19-28)36(41)34-14-15-35(38)33-5-3-2-4-32(33)34/h2-15,27-28H,16-25H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Antagonistic activity of the compound against muscarinic acetylcholine receptor M2 |
Bioorg Med Chem Lett 11: 2311-4 (2001)
BindingDB Entry DOI: 10.7270/Q28S4P7Z |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50103768
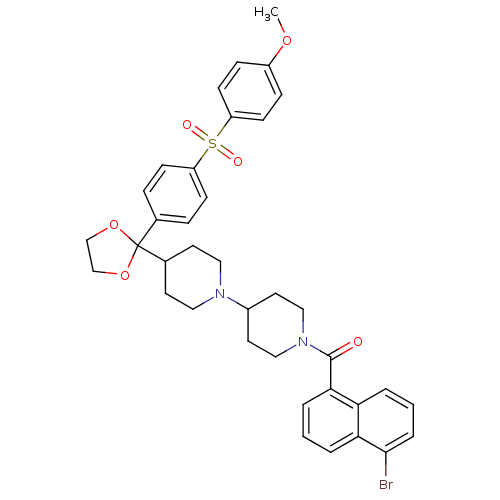
((5-Bromo-naphthalen-1-yl)-(4-{2-[4-(4-methoxy-benz...)Show SMILES COc1ccc(cc1)S(=O)(=O)c1ccc(cc1)C1(OCCO1)C1CCN(CC1)C1CCN(CC1)C(=O)c1cccc2c(Br)cccc12 Show InChI InChI=1S/C37H39BrN2O6S/c1-44-29-10-14-31(15-11-29)47(42,43)30-12-8-26(9-13-30)37(45-24-25-46-37)27-16-20-39(21-17-27)28-18-22-40(23-19-28)36(41)34-6-2-5-33-32(34)4-3-7-35(33)38/h2-15,27-28H,16-25H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Antagonistic activity of the compound against muscarinic acetylcholine receptor M2 |
Bioorg Med Chem Lett 11: 2311-4 (2001)
BindingDB Entry DOI: 10.7270/Q28S4P7Z |
More data for this
Ligand-Target Pair | |
Renin
(Homo sapiens (Human)) | BDBM50065428
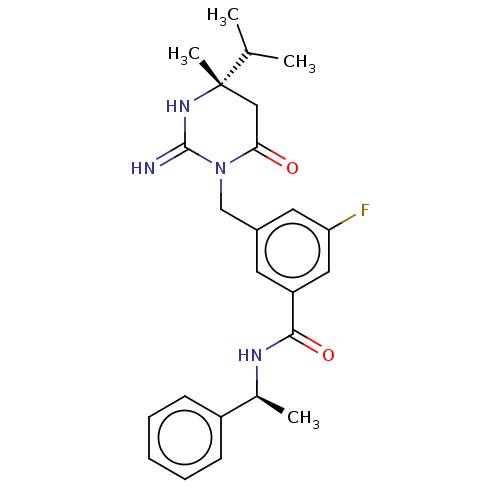
(CHEMBL3401350)Show SMILES CC(C)[C@]1(C)CC(=O)N(Cc2cc(F)cc(c2)C(=O)N[C@@H](C)c2ccccc2)C(=N)N1 |r| Show InChI InChI=1S/C24H29FN4O2/c1-15(2)24(4)13-21(30)29(23(26)28-24)14-17-10-19(12-20(25)11-17)22(31)27-16(3)18-8-6-5-7-9-18/h5-12,15-16H,13-14H2,1-4H3,(H2,26,28)(H,27,31)/t16-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of renin (unknown origin) |
Bioorg Med Chem Lett 25: 1592-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.02.003
BindingDB Entry DOI: 10.7270/Q2X63PMJ |
More data for this
Ligand-Target Pair | |
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM35229
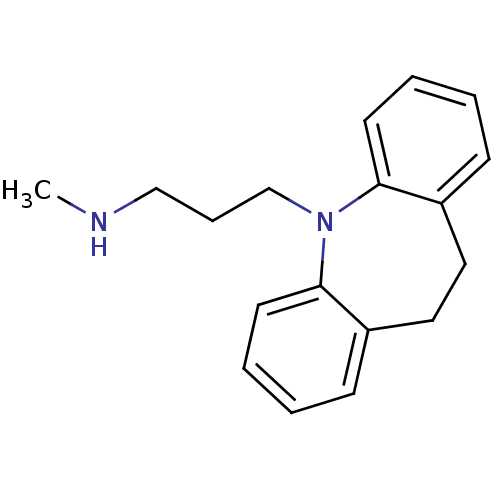
(3-(10,11-dihydro-5H-dibenzo[b,f]azepin-5-yl)-N-met...)Show InChI InChI=1S/C18H22N2/c1-19-13-6-14-20-17-9-4-2-7-15(17)11-12-16-8-3-5-10-18(16)20/h2-5,7-10,19H,6,11-14H2,1H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.600 | -54.8 | n/a | n/a | n/a | n/a | n/a | 7.4 | 37 |
Wyeth Research
| Assay Description
Compounds were evaluated the inhibition of [3H] nisoxetine binding to MDCK-Net6 cells, stably transfected with the human norepinephrine transporter (... |
Bioorg Med Chem 17: 7802-15 (2009)
Article DOI: 10.1016/j.bmc.2009.09.023
BindingDB Entry DOI: 10.7270/Q26Q1VK2 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50343157
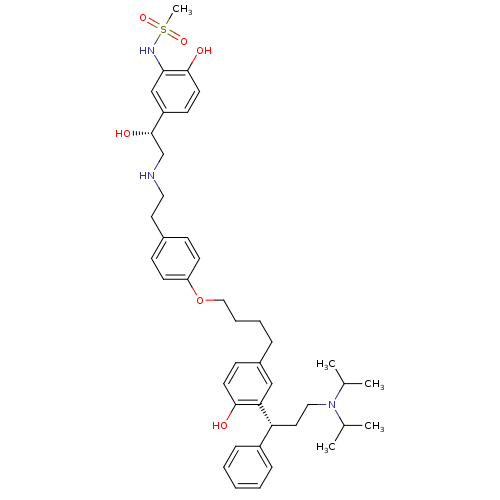
(CHEMBL1773264 | N-(5-((R)-2-(4-(4-(3-((R)-3-(diiso...)Show SMILES CC(C)N(CC[C@H](c1ccccc1)c1cc(CCCCOc2ccc(CCNC[C@H](O)c3ccc(O)c(NS(C)(=O)=O)c3)cc2)ccc1O)C(C)C |r| Show InChI InChI=1S/C42H57N3O6S/c1-30(2)45(31(3)4)25-23-37(34-12-7-6-8-13-34)38-27-33(16-20-40(38)46)11-9-10-26-51-36-18-14-32(15-19-36)22-24-43-29-42(48)35-17-21-41(47)39(28-35)44-52(5,49)50/h6-8,12-21,27-28,30-31,37,42-44,46-48H,9-11,22-26,29H2,1-5H3/t37-,42+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.634 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl Scopolamine from human muscarinic M3 receptor expressed in CHO cells by scintillation proximity assay |
Bioorg Med Chem Lett 21: 2759-63 (2011)
Article DOI: 10.1016/j.bmcl.2010.10.132
BindingDB Entry DOI: 10.7270/Q2G73F1B |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50128830

(CHEMBL3629353)Show SMILES O[C@@H](CNCCCCCCCCCN1CCC(CC1)OC(=O)Nc1ccccc1-c1ccc(O)c(Cl)c1)c1ccc(O)c2[nH]c(=O)ccc12 |r| Show InChI InChI=1S/C38H47ClN4O6/c39-31-24-26(12-15-33(31)44)28-10-6-7-11-32(28)41-38(48)49-27-18-22-43(23-19-27)21-9-5-3-1-2-4-8-20-40-25-35(46)29-13-16-34(45)37-30(29)14-17-36(47)42-37/h6-7,10-17,24,27,35,40,44-46H,1-5,8-9,18-23,25H2,(H,41,48)(H,42,47)/t35-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl scopolamine from human cloned muscarinic M3 receptor by dilution method |
Bioorg Med Chem Lett 25: 5121-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.10.008
BindingDB Entry DOI: 10.7270/Q2JQ12V7 |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50518981

(CHEMBL4594173)Show SMILES CC(C)NC[C@H](O)COc1ccccc1CCCC(=O)NCCNC(=O)CCCc1ccccc1OC[C@@H](O)CNC(C)C |r| Show InChI InChI=1S/C34H54N4O6/c1-25(2)37-21-29(39)23-43-31-15-7-5-11-27(31)13-9-17-33(41)35-19-20-36-34(42)18-10-14-28-12-6-8-16-32(28)44-24-30(40)22-38-26(3)4/h5-8,11-12,15-16,25-26,29-30,37-40H,9-10,13-14,17-24H2,1-4H3,(H,35,41)(H,36,42)/t29-,30-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.759 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen
Curated by ChEMBL
| Assay Description
Displacement of [3H]DHA from beta2 adrenergic receptor (unknown origin) stably expressed in HEK293 cell membranes measured after 90 mins by scintilla... |
J Med Chem 62: 7806-7839 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00595
BindingDB Entry DOI: 10.7270/Q2PG1W4K |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50343158
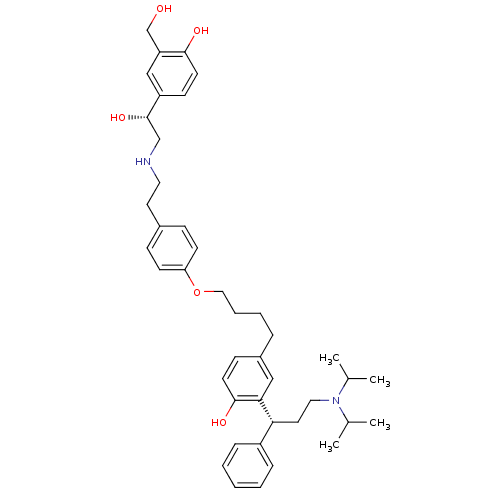
(2-((R)-3-(diisopropylamino)-1-phenylpropyl)-4-(4-(...)Show SMILES CC(C)N(CC[C@H](c1ccccc1)c1cc(CCCCOc2ccc(CCNC[C@H](O)c3ccc(O)c(CO)c3)cc2)ccc1O)C(C)C |r| Show InChI InChI=1S/C42H56N2O5/c1-30(2)44(31(3)4)24-22-38(34-11-6-5-7-12-34)39-26-33(15-19-41(39)47)10-8-9-25-49-37-17-13-32(14-18-37)21-23-43-28-42(48)35-16-20-40(46)36(27-35)29-45/h5-7,11-20,26-27,30-31,38,42-43,45-48H,8-10,21-25,28-29H2,1-4H3/t38-,42+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.765 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl Scopolamine from human muscarinic M3 receptor expressed in CHO cells by scintillation proximity assay |
Bioorg Med Chem Lett 21: 2759-63 (2011)
Article DOI: 10.1016/j.bmcl.2010.10.132
BindingDB Entry DOI: 10.7270/Q2G73F1B |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50518993

(CHEMBL4527821)Show SMILES CC(C)NC[C@H](O)COc1ccccc1CCCCCCCCc1ccccc1OC[C@@H](O)CNC(C)C |r| Show InChI InChI=1S/C32H52N2O4/c1-25(2)33-21-29(35)23-37-31-19-13-11-17-27(31)15-9-7-5-6-8-10-16-28-18-12-14-20-32(28)38-24-30(36)22-34-26(3)4/h11-14,17-20,25-26,29-30,33-36H,5-10,15-16,21-24H2,1-4H3/t29-,30-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.794 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen
Curated by ChEMBL
| Assay Description
Displacement of [3H]DHA from beta2 adrenergic receptor (unknown origin) stably expressed in HEK293 cell membranes measured after 90 mins by scintilla... |
J Med Chem 62: 7806-7839 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00595
BindingDB Entry DOI: 10.7270/Q2PG1W4K |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50103775

((6-Fluoro-naphthalen-1-yl)-(4-{2-[4-(4-methoxy-ben...)Show SMILES COc1ccc(cc1)S(=O)(=O)c1ccc(cc1)C1(OCCO1)C1CCN(CC1)C1CCN(CC1)C(=O)c1cccc2cc(F)ccc12 Show InChI InChI=1S/C37H39FN2O6S/c1-44-31-8-12-33(13-9-31)47(42,43)32-10-5-27(6-11-32)37(45-23-24-46-37)28-15-19-39(20-16-28)30-17-21-40(22-18-30)36(41)35-4-2-3-26-25-29(38)7-14-34(26)35/h2-14,25,28,30H,15-24H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.810 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Antagonistic activity of the compound against muscarinic acetylcholine receptor M2 |
Bioorg Med Chem Lett 11: 2311-4 (2001)
BindingDB Entry DOI: 10.7270/Q28S4P7Z |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50518982
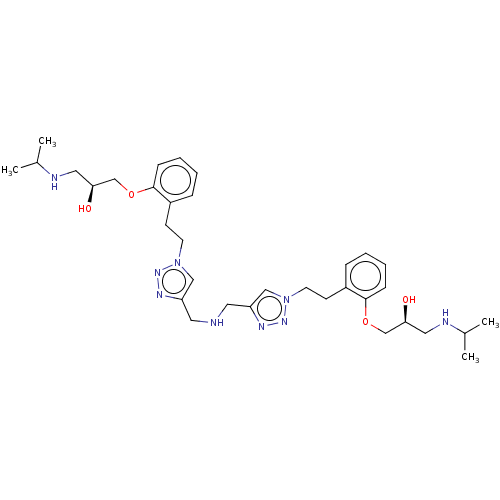
(CHEMBL4462252)Show SMILES CC(C)NC[C@H](O)COc1ccccc1CCn1cc(CNCc2cn(CCc3ccccc3OC[C@@H](O)CNC(C)C)nn2)nn1 |r| Show InChI InChI=1S/C34H51N9O4/c1-25(2)36-19-31(44)23-46-33-11-7-5-9-27(33)13-15-42-21-29(38-40-42)17-35-18-30-22-43(41-39-30)16-14-28-10-6-8-12-34(28)47-24-32(45)20-37-26(3)4/h5-12,21-22,25-26,31-32,35-37,44-45H,13-20,23-24H2,1-4H3/t31-,32-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.891 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen
Curated by ChEMBL
| Assay Description
Displacement of [3H]DHA from beta2 adrenergic receptor (unknown origin) stably expressed in HEK293 cell membranes measured after 90 mins by scintilla... |
J Med Chem 62: 7806-7839 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00595
BindingDB Entry DOI: 10.7270/Q2PG1W4K |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50518977
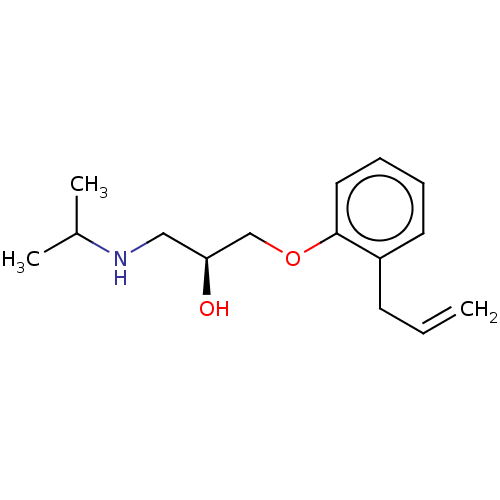
(CHEMBL1160734)Show InChI InChI=1S/C15H23NO2/c1-4-7-13-8-5-6-9-15(13)18-11-14(17)10-16-12(2)3/h4-6,8-9,12,14,16-17H,1,7,10-11H2,2-3H3/t14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.933 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen
Curated by ChEMBL
| Assay Description
Displacement of [3H]DHA from beta2 adrenergic receptor (unknown origin) stably expressed in HEK293 cell membranes measured after 90 mins by scintilla... |
J Med Chem 62: 7806-7839 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00595
BindingDB Entry DOI: 10.7270/Q2PG1W4K |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50518973

(CHEMBL4473518)Show SMILES CC(C)NC[C@H](O)COc1ccccc1CCCCCCc1ccccc1OC[C@@H](O)CNC(C)C |r| Show InChI InChI=1S/C30H48N2O4/c1-23(2)31-19-27(33)21-35-29-17-11-9-15-25(29)13-7-5-6-8-14-26-16-10-12-18-30(26)36-22-28(34)20-32-24(3)4/h9-12,15-18,23-24,27-28,31-34H,5-8,13-14,19-22H2,1-4H3/t27-,28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.955 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen
Curated by ChEMBL
| Assay Description
Displacement of [3H]DHA from beta2 adrenergic receptor (unknown origin) stably expressed in HEK293 cell membranes measured after 90 mins by scintilla... |
J Med Chem 62: 7806-7839 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00595
BindingDB Entry DOI: 10.7270/Q2PG1W4K |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M2
(Homo sapiens (Human)) | BDBM50103769
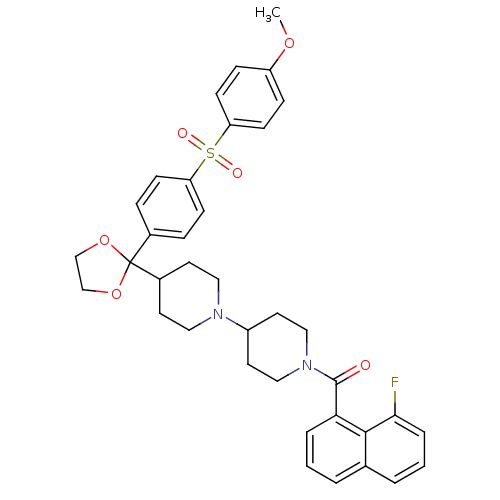
((8-Fluoro-naphthalen-1-yl)-(4-{2-[4-(4-methoxy-ben...)Show SMILES COc1ccc(cc1)S(=O)(=O)c1ccc(cc1)C1(OCCO1)C1CCN(CC1)C1CCN(CC1)C(=O)c1cccc2cccc(F)c12 Show InChI InChI=1S/C37H39FN2O6S/c1-44-30-10-14-32(15-11-30)47(42,43)31-12-8-27(9-13-31)37(45-24-25-46-37)28-16-20-39(21-17-28)29-18-22-40(23-19-29)36(41)33-6-2-4-26-5-3-7-34(38)35(26)33/h2-15,28-29H,16-25H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.990 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Antagonistic activity of the compound against Muscarinic acetylcholine receptor M2 |
Bioorg Med Chem Lett 11: 2311-4 (2001)
BindingDB Entry DOI: 10.7270/Q28S4P7Z |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50343160
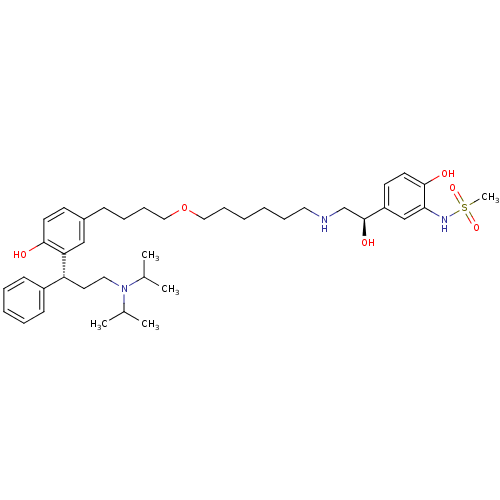
(CHEMBL1773266 | N-(5-((R)-2-(6-(4-(3-((R)-3-(diiso...)Show SMILES CC(C)N(CC[C@H](c1ccccc1)c1cc(CCCCOCCCCCCNC[C@H](O)c2ccc(O)c(NS(C)(=O)=O)c2)ccc1O)C(C)C |r| Show InChI InChI=1S/C40H61N3O6S/c1-30(2)43(31(3)4)24-22-35(33-16-9-8-10-17-33)36-27-32(18-20-38(36)44)15-11-14-26-49-25-13-7-6-12-23-41-29-40(46)34-19-21-39(45)37(28-34)42-50(5,47)48/h8-10,16-21,27-28,30-31,35,40-42,44-46H,6-7,11-15,22-26,29H2,1-5H3/t35-,40+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.990 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl Scopolamine from human muscarinic M3 receptor expressed in CHO cells by scintillation proximity assay |
Bioorg Med Chem Lett 21: 2759-63 (2011)
Article DOI: 10.1016/j.bmcl.2010.10.132
BindingDB Entry DOI: 10.7270/Q2G73F1B |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Mus musculus) | BDBM50104940
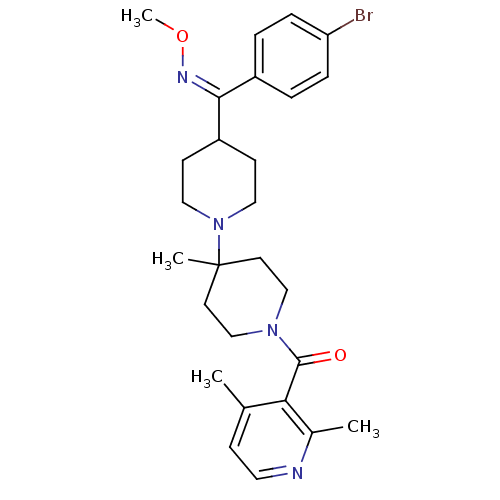
(CHEMBL323172 | {4-[(4-Bromo-phenyl)-methoxyimino-m...)Show SMILES CO\N=C(\C1CCN(CC1)C1(C)CCN(CC1)C(=O)c1c(C)ccnc1C)c1ccc(Br)cc1 Show InChI InChI=1S/C27H35BrN4O2/c1-19-9-14-29-20(2)24(19)26(33)31-17-12-27(3,13-18-31)32-15-10-22(11-16-32)25(30-34-4)21-5-7-23(28)8-6-21/h5-9,14,22H,10-13,15-18H2,1-4H3/b30-25+ | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of RANTES binding to Chemokine receptor type 5 receptor from NIH 3T3 cells |
J Med Chem 45: 3143-60 (2002)
BindingDB Entry DOI: 10.7270/Q2HD7TZJ |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50291849
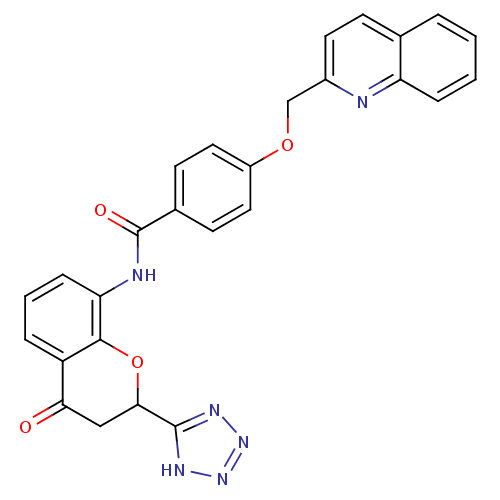
(CHEMBL47662 | N-[4-Oxo-2-(1H-tetrazol-5-yl)-chroma...)Show SMILES O=C(Nc1cccc2C(=O)CC(Oc12)c1nnn[nH]1)c1ccc(OCc2ccc3ccccc3n2)cc1 Show InChI InChI=1S/C27H20N6O4/c34-23-14-24(26-30-32-33-31-26)37-25-20(23)5-3-7-22(25)29-27(35)17-9-12-19(13-10-17)36-15-18-11-8-16-4-1-2-6-21(16)28-18/h1-13,24H,14-15H2,(H,29,35)(H,30,31,32,33) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Rhone-Poulenc Rorer Central Research
Curated by ChEMBL
| Assay Description
Binding affinity against LTD4 receptor in guinea pig lung membranes. |
J Med Chem 34: 1704-7 (1991)
BindingDB Entry DOI: 10.7270/Q2FJ2FR5 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Mus musculus) | BDBM50104933

(CHEMBL105821 | {4-[(4-Bromo-phenyl)-ethoxyimino-me...)Show SMILES CCO\N=C(\C1CCN(CC1)C1(C)CCN(CC1)C(=O)c1c(C)ccnc1C)c1ccc(Br)cc1 Show InChI InChI=1S/C28H37BrN4O2/c1-5-35-31-26(22-6-8-24(29)9-7-22)23-11-16-33(17-12-23)28(4)13-18-32(19-14-28)27(34)25-20(2)10-15-30-21(25)3/h6-10,15,23H,5,11-14,16-19H2,1-4H3/b31-26+ | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of RANTES binding to Chemokine receptor type 5 receptor from NIH 3T3 cells |
J Med Chem 45: 3143-60 (2002)
BindingDB Entry DOI: 10.7270/Q2HD7TZJ |
More data for this
Ligand-Target Pair | |
Beta-2 adrenergic receptor
(Homo sapiens (Human)) | BDBM50518968
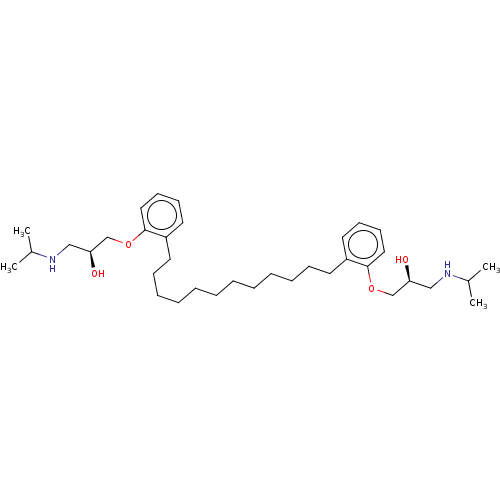
(CHEMBL4441975)Show SMILES CC(C)NC[C@H](O)COc1ccccc1CCCCCCCCCCCCc1ccccc1OC[C@@H](O)CNC(C)C |r| Show InChI InChI=1S/C36H60N2O4/c1-29(2)37-25-33(39)27-41-35-23-17-15-21-31(35)19-13-11-9-7-5-6-8-10-12-14-20-32-22-16-18-24-36(32)42-28-34(40)26-38-30(3)4/h15-18,21-24,29-30,33-34,37-40H,5-14,19-20,25-28H2,1-4H3/t33-,34-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen
Curated by ChEMBL
| Assay Description
Displacement of [3H]DHA from beta2 adrenergic receptor (unknown origin) stably expressed in HEK293 cell membranes measured after 90 mins by scintilla... |
J Med Chem 62: 7806-7839 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00595
BindingDB Entry DOI: 10.7270/Q2PG1W4K |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50128838
(CHEMBL3629355)Show SMILES CN1CCC(Cn2cnc(n2)C(O)(C2CCCCC2)c2ccccc2)CC1 Show InChI InChI=1S/C22H32N4O/c1-25-14-12-18(13-15-25)16-26-17-23-21(24-26)22(27,19-8-4-2-5-9-19)20-10-6-3-7-11-20/h2,4-5,8-9,17-18,20,27H,3,6-7,10-16H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl scopolamine from human cloned muscarinic M3 receptor by dilution method |
Bioorg Med Chem Lett 25: 5121-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.10.008
BindingDB Entry DOI: 10.7270/Q2JQ12V7 |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M3
(Homo sapiens (Human)) | BDBM50128836
(CHEMBL143228)Show InChI InChI=1S/C20H23NO3/c1-21-14-12-18(13-15-21)24-19(22)20(23,16-8-4-2-5-9-16)17-10-6-3-7-11-17/h2-11,18,23H,12-15H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-methyl scopolamine from human cloned muscarinic M3 receptor by dilution method |
Bioorg Med Chem Lett 25: 5121-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.10.008
BindingDB Entry DOI: 10.7270/Q2JQ12V7 |
More data for this
Ligand-Target Pair | |
Beta-1 adrenergic receptor
(Homo sapiens (Human)) | BDBM50518969

(CHEMBL4514723)Show SMILES CC(C)NC[C@H](O)COc1ccccc1CCCn1cc(CNCc2cn(CCCc3ccccc3OC[C@@H](O)CNC(C)C)nn2)nn1 |r| Show InChI InChI=1S/C36H55N9O4/c1-27(2)38-21-33(46)25-48-35-15-7-5-11-29(35)13-9-17-44-23-31(40-42-44)19-37-20-32-24-45(43-41-32)18-10-14-30-12-6-8-16-36(30)49-26-34(47)22-39-28(3)4/h5-8,11-12,15-16,23-24,27-28,33-34,37-39,46-47H,9-10,13-14,17-22,25-26H2,1-4H3/t33-,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Copenhagen
Curated by ChEMBL
| Assay Description
Displacement of [3H]DHA from beta1 adrenergic receptor (unknown origin) stably expressed in HEK293 cell membranes measured after 90 mins by scintilla... |
J Med Chem 62: 7806-7839 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00595
BindingDB Entry DOI: 10.7270/Q2PG1W4K |
More data for this
Ligand-Target Pair | |
Muscarinic acetylcholine receptor M5
(Homo sapiens (Human)) | BDBM50128835

(CHEMBL3629360)Show SMILES O[C@@H](CNCCc1ccc(CCN2CCC(Cn3cnc(n3)[C@@](O)(C3CCCCC3)c3ccccc3)CC2)cc1)c1ccc(O)c2[nH]c(=O)ccc12 |r| Show InChI InChI=1S/C42H52N6O4/c49-37-17-15-35(36-16-18-39(51)45-40(36)37)38(50)27-43-23-19-30-11-13-31(14-12-30)20-24-47-25-21-32(22-26-47)28-48-29-44-41(46-48)42(52,33-7-3-1-4-8-33)34-9-5-2-6-10-34/h1,3-4,7-8,11-18,29,32,34,38,43,49-50,52H,2,5-6,9-10,19-28H2,(H,45,51)/t38-,42-/m0/s1 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer
Curated by ChEMBL
| Assay Description
Antagonist activity at muscarinic M5 receptor (unknown origin) |
Bioorg Med Chem Lett 25: 5121-6 (2015)
Article DOI: 10.1016/j.bmcl.2015.10.008
BindingDB Entry DOI: 10.7270/Q2JQ12V7 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data