 Found 894 hits with Last Name = 'nicolai' and Initial = 'e'
Found 894 hits with Last Name = 'nicolai' and Initial = 'e' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328879
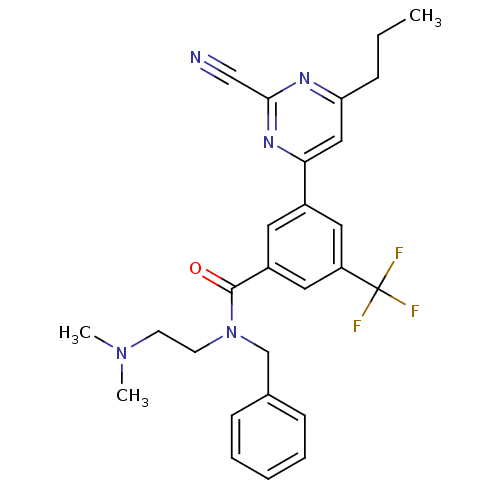
(CHEMBL1234898 | N-benzyl-3-(2-cyano-6-propylpyrimi...)Show SMILES CCCc1cc(nc(n1)C#N)-c1cc(cc(c1)C(F)(F)F)C(=O)N(CCN(C)C)Cc1ccccc1 Show InChI InChI=1S/C27H28F3N5O/c1-4-8-23-16-24(33-25(17-31)32-23)20-13-21(15-22(14-20)27(28,29)30)26(36)35(12-11-34(2)3)18-19-9-6-5-7-10-19/h5-7,9-10,13-16H,4,8,11-12,18H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50313476

(4-(3-(piperidin-1-yl)propyl)-6-(3-(trifluoromethyl...)Show SMILES FC(F)(F)c1cccc(c1)-c1cc(CCCN2CCCCC2)nc(n1)C#N Show InChI InChI=1S/C20H21F3N4/c21-20(22,23)16-7-4-6-15(12-16)18-13-17(25-19(14-24)26-18)8-5-11-27-9-2-1-3-10-27/h4,6-7,12-13H,1-3,5,8-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328887
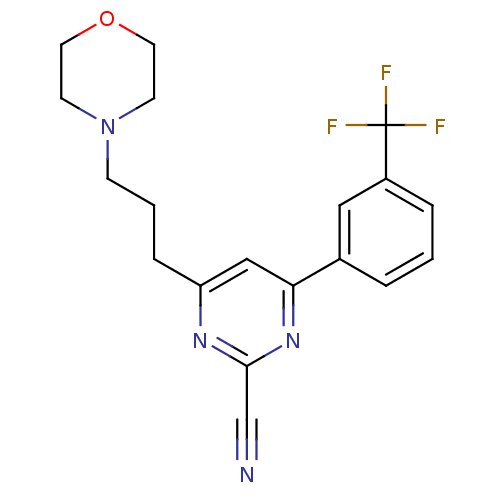
(4-(3-morpholinopropyl)-6-(3-(trifluoromethyl)pheny...)Show SMILES FC(F)(F)c1cccc(c1)-c1cc(CCCN2CCOCC2)nc(n1)C#N Show InChI InChI=1S/C19H19F3N4O/c20-19(21,22)15-4-1-3-14(11-15)17-12-16(24-18(13-23)25-17)5-2-6-26-7-9-27-10-8-26/h1,3-4,11-12H,2,5-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50313477

(4-(3-(pentan-3-ylamino)propyl)-6-(3-(trifluorometh...)Show SMILES CCC(CC)NCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C20H23F3N4/c1-3-16(4-2)25-10-6-9-17-12-18(27-19(13-24)26-17)14-7-5-8-15(11-14)20(21,22)23/h5,7-8,11-12,16,25H,3-4,6,9-10H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328880
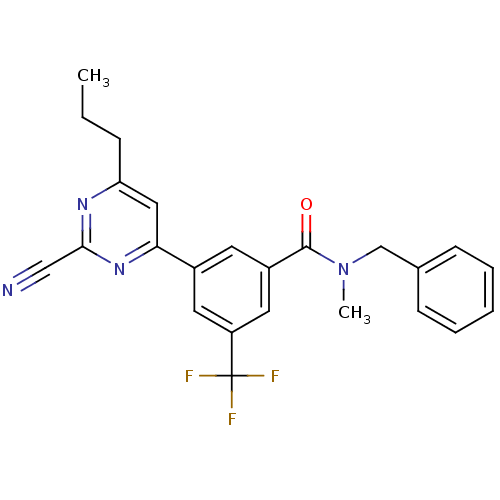
(CHEMBL1271174 | N-benzyl-3-(2-cyano-6-propylpyrimi...)Show SMILES CCCc1cc(nc(n1)C#N)-c1cc(cc(c1)C(F)(F)F)C(=O)N(C)Cc1ccccc1 Show InChI InChI=1S/C24H21F3N4O/c1-3-7-20-13-21(30-22(14-28)29-20)17-10-18(12-19(11-17)24(25,26)27)23(32)31(2)15-16-8-5-4-6-9-16/h4-6,8-13H,3,7,15H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328890
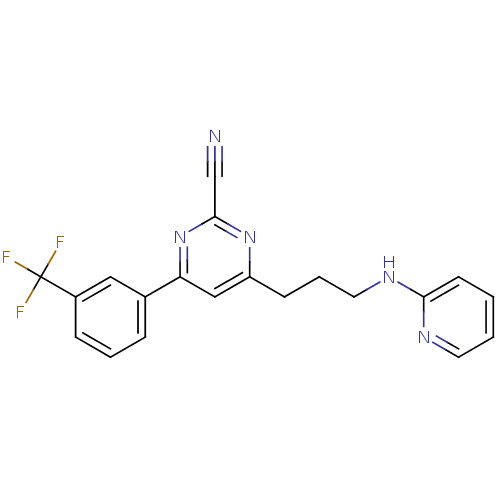
(4-(3-(pyridin-2-ylamino)propyl)-6-(3-(trifluoromet...)Show SMILES FC(F)(F)c1cccc(c1)-c1cc(CCCNc2ccccn2)nc(n1)C#N Show InChI InChI=1S/C20H16F3N5/c21-20(22,23)15-6-3-5-14(11-15)17-12-16(27-19(13-24)28-17)7-4-10-26-18-8-1-2-9-25-18/h1-3,5-6,8-9,11-12H,4,7,10H2,(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328885

(4-(3-(2,2,2-trifluoroethylamino)propyl)-6-(3-(trif...)Show SMILES FC(F)(F)CNCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C17H14F6N4/c18-16(19,20)10-25-6-2-5-13-8-14(27-15(9-24)26-13)11-3-1-4-12(7-11)17(21,22)23/h1,3-4,7-8,25H,2,5-6,10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328888
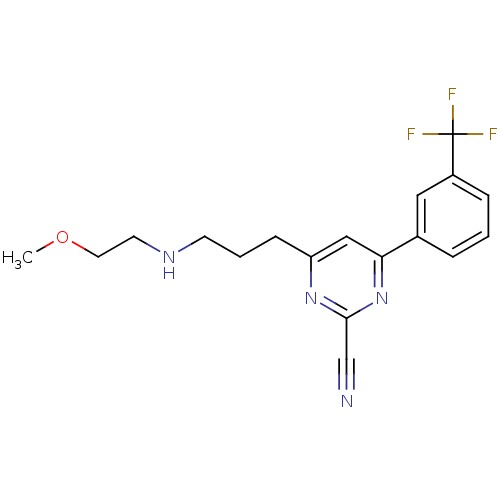
(4-(3-(2-methoxyethylamino)propyl)-6-(3-(trifluorom...)Show InChI InChI=1S/C18H19F3N4O/c1-26-9-8-23-7-3-6-15-11-16(25-17(12-22)24-15)13-4-2-5-14(10-13)18(19,20)21/h2,4-5,10-11,23H,3,6-9H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 560 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328884
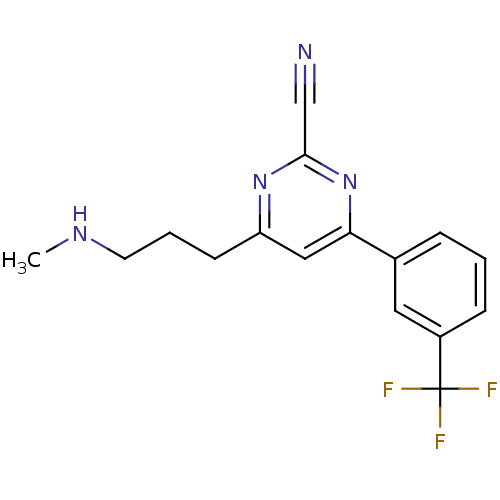
(4-(3-(methylamino)propyl)-6-(3-(trifluoromethyl)ph...)Show InChI InChI=1S/C16H15F3N4/c1-21-7-3-6-13-9-14(23-15(10-20)22-13)11-4-2-5-12(8-11)16(17,18)19/h2,4-5,8-9,21H,3,6-7H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 630 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50313479

(4-cycloheptyl-6-(3-(piperidin-1-yl)propyl)pyrimidi...)Show InChI InChI=1S/C20H30N4/c21-16-20-22-18(11-8-14-24-12-6-3-7-13-24)15-19(23-20)17-9-4-1-2-5-10-17/h15,17H,1-14H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328878
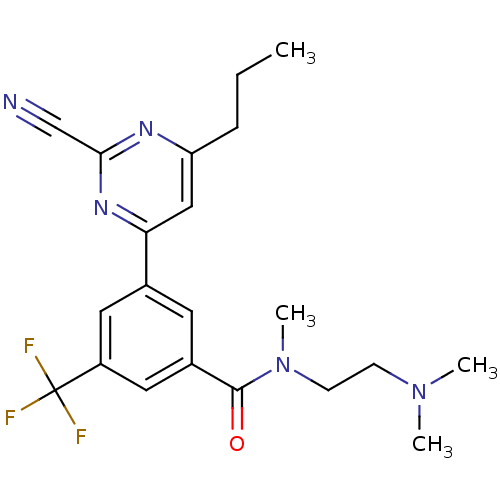
(3-(2-cyano-6-propylpyrimidin-4-yl)-N-(2-(dimethyla...)Show SMILES CCCc1cc(nc(n1)C#N)-c1cc(cc(c1)C(F)(F)F)C(=O)N(C)CCN(C)C Show InChI InChI=1S/C21H24F3N5O/c1-5-6-17-12-18(27-19(13-25)26-17)14-9-15(11-16(10-14)21(22,23)24)20(30)29(4)8-7-28(2)3/h9-12H,5-8H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 710 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328886
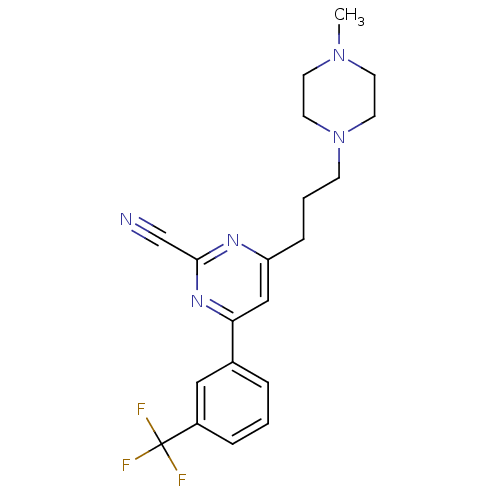
(4-(3-(4-methylpiperazin-1-yl)propyl)-6-(3-(trifluo...)Show SMILES CN1CCN(CCCc2cc(nc(n2)C#N)-c2cccc(c2)C(F)(F)F)CC1 Show InChI InChI=1S/C20H22F3N5/c1-27-8-10-28(11-9-27)7-3-6-17-13-18(26-19(14-24)25-17)15-4-2-5-16(12-15)20(21,22)23/h2,4-5,12-13H,3,6-11H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328889
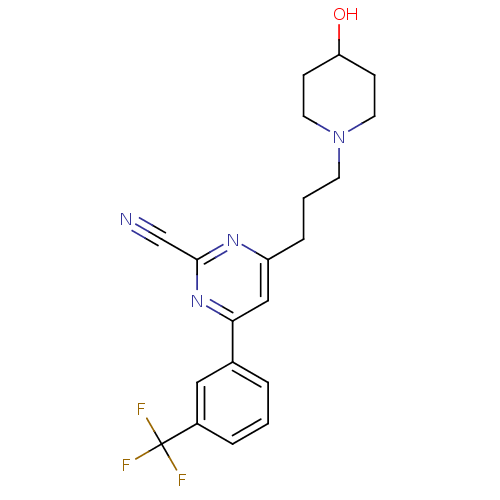
(4-(3-(4-hydroxypiperidin-1-yl)propyl)-6-(3-(triflu...)Show SMILES OC1CCN(CCCc2cc(nc(n2)C#N)-c2cccc(c2)C(F)(F)F)CC1 Show InChI InChI=1S/C20H21F3N4O/c21-20(22,23)15-4-1-3-14(11-15)18-12-16(25-19(13-24)26-18)5-2-8-27-9-6-17(28)7-10-27/h1,3-4,11-12,17,28H,2,5-10H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.12E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328895

(1-(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyrimid...)Show SMILES NC(=O)C1(CC1)NCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C19H18F3N5O/c20-19(21,22)13-4-1-3-12(9-13)15-10-14(26-16(11-23)27-15)5-2-8-25-18(6-7-18)17(24)28/h1,3-4,9-10,25H,2,5-8H2,(H2,24,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.58E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328896
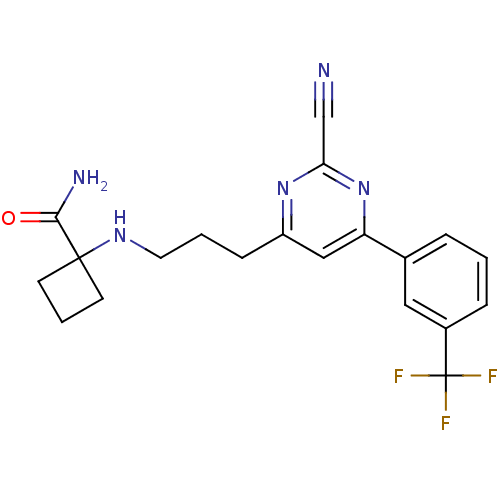
(1-(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyrimid...)Show SMILES NC(=O)C1(CCC1)NCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C20H20F3N5O/c21-20(22,23)14-5-1-4-13(10-14)16-11-15(27-17(12-24)28-16)6-2-9-26-19(18(25)29)7-3-8-19/h1,4-5,10-11,26H,2-3,6-9H2,(H2,25,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.51E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328894
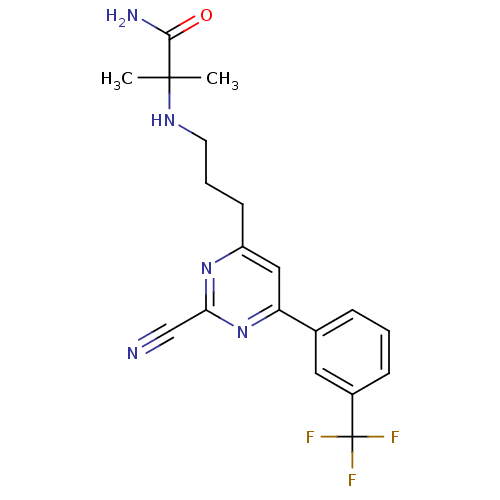
(2-(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyrimid...)Show SMILES CC(C)(NCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F)C(N)=O Show InChI InChI=1S/C19H20F3N5O/c1-18(2,17(24)28)25-8-4-7-14-10-15(27-16(11-23)26-14)12-5-3-6-13(9-12)19(20,21)22/h3,5-6,9-10,25H,4,7-8H2,1-2H3,(H2,24,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human ERG channel expressed in HEK293 cells by patch clamp assay |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328894

(2-(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyrimid...)Show SMILES CC(C)(NCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F)C(N)=O Show InChI InChI=1S/C19H20F3N5O/c1-18(2,17(24)28)25-8-4-7-14-10-15(27-16(11-23)26-14)12-5-3-6-13(9-12)19(20,21)22/h3,5-6,9-10,25H,4,7-8H2,1-2H3,(H2,24,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.16E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328883

(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyrimidin-...)Show SMILES CCC(CC)NC(=O)CCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C20H21F3N4O/c1-3-15(4-2)26-19(28)9-8-16-11-17(27-18(12-24)25-16)13-6-5-7-14(10-13)20(21,22)23/h5-7,10-11,15H,3-4,8-9H2,1-2H3,(H,26,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.16E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328893
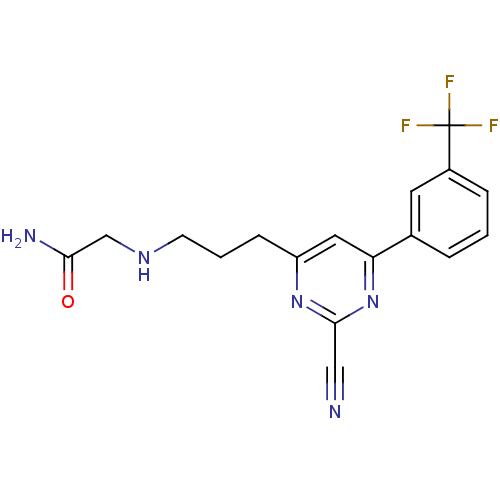
(2-(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyrimid...)Show SMILES NC(=O)CNCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C17H16F3N5O/c18-17(19,20)12-4-1-3-11(7-12)14-8-13(24-16(9-21)25-14)5-2-6-23-10-15(22)26/h1,3-4,7-8,23H,2,5-6,10H2,(H2,22,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.98E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328892
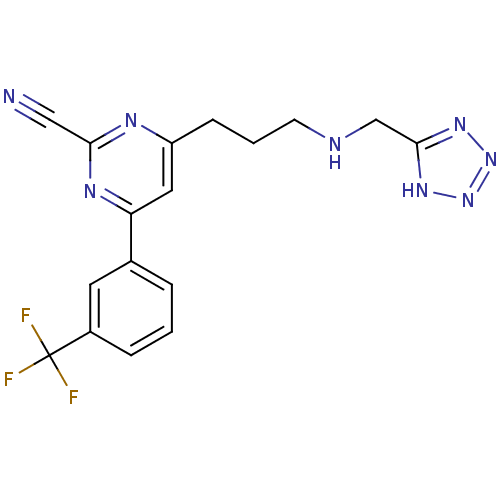
(4-(3-((1H-tetrazol-5-yl)methylamino)propyl)-6-(3-(...)Show SMILES FC(F)(F)c1cccc(c1)-c1cc(CCCNCc2nnn[nH]2)nc(n1)C#N Show InChI InChI=1S/C17H15F3N8/c18-17(19,20)12-4-1-3-11(7-12)14-8-13(23-15(9-21)24-14)5-2-6-22-10-16-25-27-28-26-16/h1,3-4,7-8,22H,2,5-6,10H2,(H,25,26,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328891
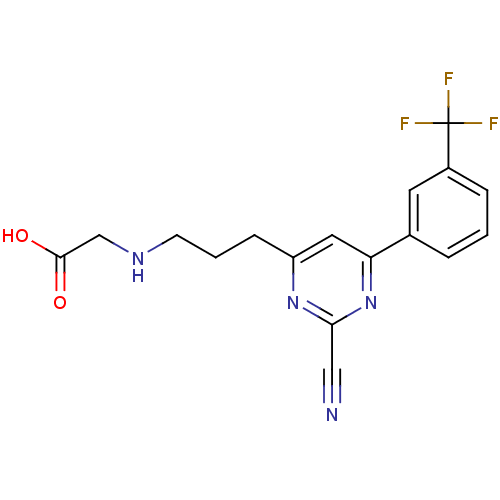
(2-(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyrimid...)Show SMILES OC(=O)CNCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C17H15F3N4O2/c18-17(19,20)12-4-1-3-11(7-12)14-8-13(23-15(9-21)24-14)5-2-6-22-10-16(25)26/h1,3-4,7-8,22H,2,5-6,10H2,(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328877
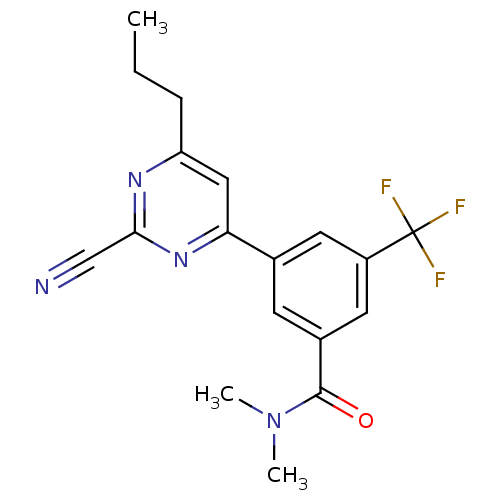
(3-(2-cyano-6-propylpyrimidin-4-yl)-N,N-dimethyl-5-...)Show SMILES CCCc1cc(nc(n1)C#N)-c1cc(cc(c1)C(F)(F)F)C(=O)N(C)C Show InChI InChI=1S/C18H17F3N4O/c1-4-5-14-9-15(24-16(10-22)23-14)11-6-12(17(26)25(2)3)8-13(7-11)18(19,20)21/h6-9H,4-5H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328882

(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyrimidin-...)Show InChI InChI=1S/C15H10F3N3O2/c16-15(17,18)10-3-1-2-9(6-10)12-7-11(4-5-14(22)23)20-13(8-19)21-12/h1-3,6-7H,4-5H2,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328897
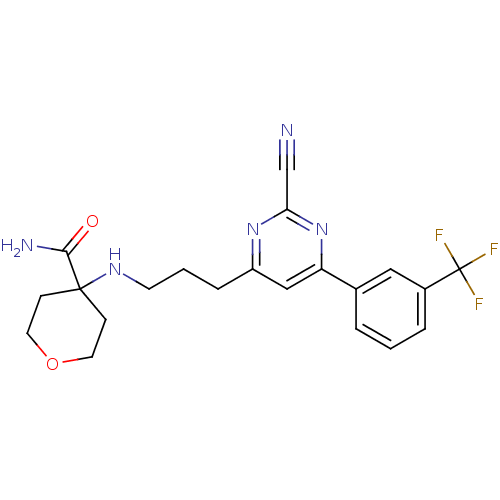
(4-(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyrimid...)Show SMILES NC(=O)C1(CCOCC1)NCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C21H22F3N5O2/c22-21(23,24)15-4-1-3-14(11-15)17-12-16(28-18(13-25)29-17)5-2-8-27-20(19(26)30)6-9-31-10-7-20/h1,3-4,11-12,27H,2,5-10H2,(H2,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328898

((S)-4-(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyr...)Show SMILES NC(=O)[C@@H]1COCCN1CCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F |r| Show InChI InChI=1S/C20H20F3N5O2/c21-20(22,23)14-4-1-3-13(9-14)16-10-15(26-18(11-24)27-16)5-2-6-28-7-8-30-12-17(28)19(25)29/h1,3-4,9-10,17H,2,5-8,12H2,(H2,25,29)/t17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50328881
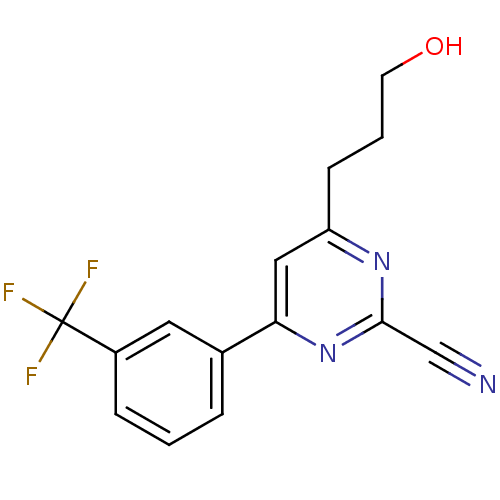
(4-(3-hydroxypropyl)-6-(3-(trifluoromethyl)phenyl)p...)Show InChI InChI=1S/C15H12F3N3O/c16-15(17,18)11-4-1-3-10(7-11)13-8-12(5-2-6-22)20-14(9-19)21-13/h1,3-4,7-8,22H,2,5-6H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H] dofetilide from human ERG channel expressed in HEK293 cells |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50313480

(4-(cyclohexylamino)-6-(piperazin-1-yl)-1,3,5-triaz...)Show InChI InChI=1S/C14H21N7/c15-10-12-18-13(17-11-4-2-1-3-5-11)20-14(19-12)21-8-6-16-7-9-21/h11,16H,1-9H2,(H,17,18,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Corporation
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin K by fluorescence assay |
Bioorg Med Chem Lett 20: 1524-7 (2010)
Article DOI: 10.1016/j.bmcl.2010.01.100
BindingDB Entry DOI: 10.7270/Q24X57ZH |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cathepsin K
(Homo sapiens (Human)) | BDBM50328879

(CHEMBL1234898 | N-benzyl-3-(2-cyano-6-propylpyrimi...)Show SMILES CCCc1cc(nc(n1)C#N)-c1cc(cc(c1)C(F)(F)F)C(=O)N(CCN(C)C)Cc1ccccc1 Show InChI InChI=1S/C27H28F3N5O/c1-4-8-23-16-24(33-25(17-31)32-23)20-13-21(15-22(14-20)27(28,29)30)26(36)35(12-11-34(2)3)18-19-9-6-5-7-10-19/h5-7,9-10,13-16H,4,8,11-12,18H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | <1 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin K using Z-Phe-Arg-MCA substrate by fluorimetric assay |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50027142
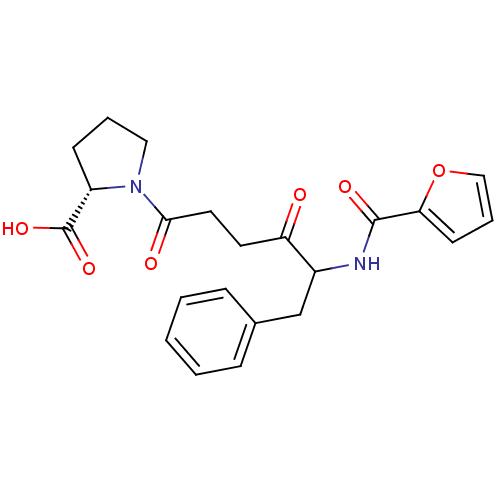
(1-{5-[(Furan-2-carbonyl)-amino]-4-oxo-6-phenyl-hex...)Show SMILES OC(=O)[C@@H]1CCCN1C(=O)CCC(=O)C(Cc1ccccc1)NC(=O)c1ccco1 Show InChI InChI=1S/C22H24N2O6/c25-18(10-11-20(26)24-12-4-8-17(24)22(28)29)16(14-15-6-2-1-3-7-15)23-21(27)19-9-5-13-30-19/h1-3,5-7,9,13,16-17H,4,8,10-12,14H2,(H,23,27)(H,28,29)/t16?,17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50% |
J Med Chem 24: 964-9 (1982)
BindingDB Entry DOI: 10.7270/Q2959J4Z |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50027132
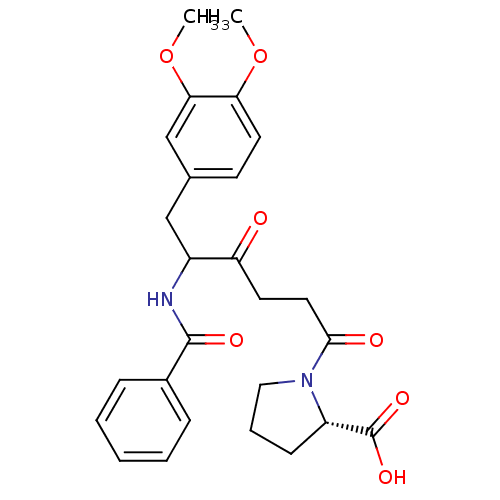
(1-[5-Benzoylamino-6-(3,4-dimethoxy-phenyl)-4-oxo-h...)Show SMILES COc1ccc(CC(NC(=O)c2ccccc2)C(=O)CCC(=O)N2CCC[C@H]2C(O)=O)cc1OC Show InChI InChI=1S/C26H30N2O7/c1-34-22-12-10-17(16-23(22)35-2)15-19(27-25(31)18-7-4-3-5-8-18)21(29)11-13-24(30)28-14-6-9-20(28)26(32)33/h3-5,7-8,10,12,16,19-20H,6,9,11,13-15H2,1-2H3,(H,27,31)(H,32,33)/t19?,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50% |
J Med Chem 24: 964-9 (1982)
BindingDB Entry DOI: 10.7270/Q2959J4Z |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50499970
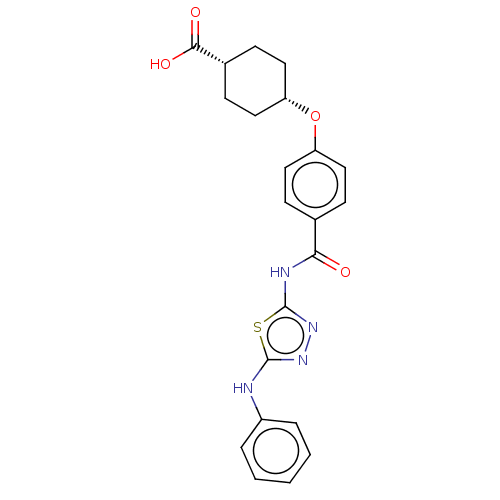
(CHEMBL3741505)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)Oc1ccc(cc1)C(=O)Nc1nnc(Nc2ccccc2)s1 |r,wU:6.9,3.2,(-9.07,-.95,;-8,-1.56,;-7.99,-2.79,;-6.67,-.78,;-5.33,-1.55,;-4,-.77,;-4,.77,;-5.34,1.53,;-6.67,.76,;-2.67,1.54,;-1.33,.77,;,1.54,;1.33,.77,;1.33,-.77,;,-1.54,;-1.33,-.77,;2.67,-1.54,;3.73,-.93,;2.66,-3.08,;4,-3.86,;4.14,-5.38,;5.64,-5.7,;6.41,-4.36,;7.94,-4.2,;8.57,-2.79,;10.1,-2.63,;10.72,-1.22,;9.81,.03,;8.28,-.14,;7.66,-1.55,;5.38,-3.22,)| Show InChI InChI=1S/C22H22N4O4S/c27-19(24-22-26-25-21(31-22)23-16-4-2-1-3-5-16)14-6-10-17(11-7-14)30-18-12-8-15(9-13-18)20(28)29/h1-7,10-11,15,18H,8-9,12-13H2,(H,23,25)(H,28,29)(H,24,26,27)/t15-,18+ | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-aventis R&D
Curated by ChEMBL
| Assay Description
Inhibition of DGAT-1 in human Chang cells assessed as lipid level after 6 hrs in presence of substrate [14C]glycerol by HPLC analysis |
Bioorg Med Chem Lett 26: 25-32 (2016)
Article DOI: 10.1016/j.bmcl.2015.11.046
BindingDB Entry DOI: 10.7270/Q2Z89GDN |
More data for this
Ligand-Target Pair | |
Integrin alpha-L/Integrin beta-2/Intercellular adhesion molecule 1
(Homo sapiens (Human)) | BDBM50199036
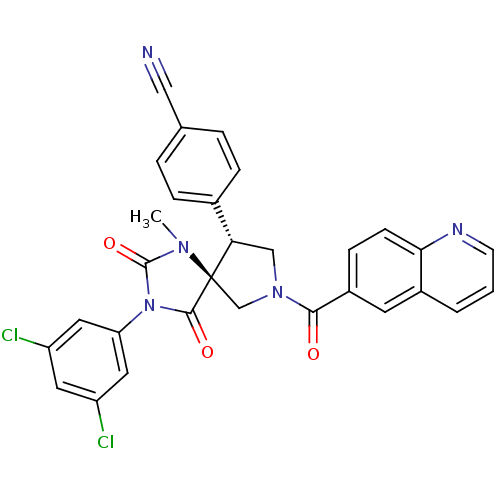
(4-[(5S,9R)-3-(3,5-dichloro-phenyl)-1-methyl-2,4-di...)Show SMILES CN1C(=O)N(C(=O)[C@]11CN(C[C@H]1c1ccc(cc1)C#N)C(=O)c1ccc2ncccc2c1)c1cc(Cl)cc(Cl)c1 Show InChI InChI=1S/C30H21Cl2N5O3/c1-35-29(40)37(24-13-22(31)12-23(32)14-24)28(39)30(35)17-36(16-25(30)19-6-4-18(15-33)5-7-19)27(38)21-8-9-26-20(11-21)3-2-10-34-26/h2-14,25H,16-17H2,1H3/t25-,30+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Cerep
Curated by ChEMBL
| Assay Description
Inhibition of LFA1-mediated adhesion of T cell to HUVEC |
J Med Chem 49: 6946-9 (2006)
Article DOI: 10.1021/jm0610806
BindingDB Entry DOI: 10.7270/Q2SF2XC3 |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50328890

(4-(3-(pyridin-2-ylamino)propyl)-6-(3-(trifluoromet...)Show SMILES FC(F)(F)c1cccc(c1)-c1cc(CCCNc2ccccn2)nc(n1)C#N Show InChI InChI=1S/C20H16F3N5/c21-20(22,23)15-6-3-5-14(11-15)17-12-16(27-19(13-24)28-17)7-4-10-26-18-8-1-2-9-25-18/h1-3,5-6,8-9,11-12H,4,7,10H2,(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin K using Z-Phe-Arg-MCA substrate by fluorimetric assay |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50328894

(2-(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyrimid...)Show SMILES CC(C)(NCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F)C(N)=O Show InChI InChI=1S/C19H20F3N5O/c1-18(2,17(24)28)25-8-4-7-14-10-15(27-16(11-23)26-14)12-5-3-6-13(9-12)19(20,21)22/h3,5-6,9-10,25H,4,7-8H2,1-2H3,(H2,24,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin K using Z-Phe-Arg-MCA substrate by fluorimetric assay |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50027344
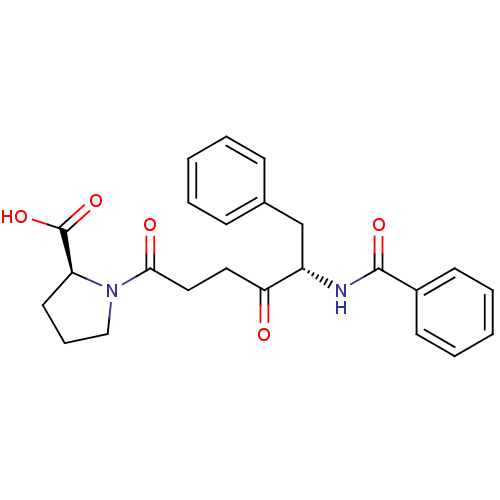
((S)-1-((S)-5-benzamido-4-oxo-6-phenylhexanoyl)pyrr...)Show SMILES OC(=O)[C@@H]1CCCN1C(=O)CCC(=O)[C@H](Cc1ccccc1)NC(=O)c1ccccc1 Show InChI InChI=1S/C24H26N2O5/c27-21(13-14-22(28)26-15-7-12-20(26)24(30)31)19(16-17-8-3-1-4-9-17)25-23(29)18-10-5-2-6-11-18/h1-6,8-11,19-20H,7,12-16H2,(H,25,29)(H,30,31)/t19-,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50% |
J Med Chem 24: 964-9 (1982)
BindingDB Entry DOI: 10.7270/Q2959J4Z |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50323087
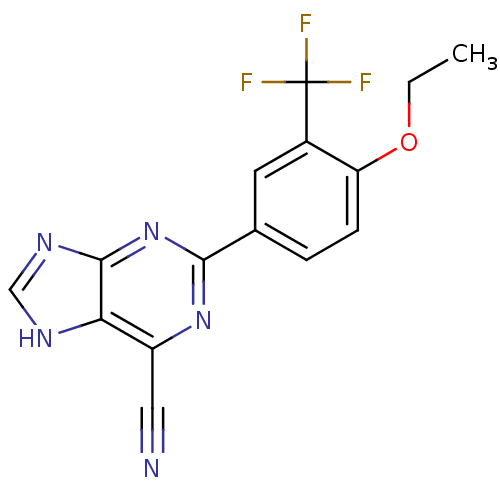
(2-(4-ethoxy-3-(trifluoromethyl)phenyl)-9H-purine-6...)Show SMILES CCOc1ccc(cc1C(F)(F)F)-c1nc(C#N)c2[nH]cnc2n1 Show InChI InChI=1S/C15H10F3N5O/c1-2-24-11-4-3-8(5-9(11)15(16,17)18)13-22-10(6-19)12-14(23-13)21-7-20-12/h3-5,7H,2H2,1H3,(H,20,21,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin S |
Bioorg Med Chem Lett 20: 4447-50 (2010)
Article DOI: 10.1016/j.bmcl.2010.06.049
BindingDB Entry DOI: 10.7270/Q2RJ4JP2 |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50499970

(CHEMBL3741505)Show SMILES OC(=O)[C@@H]1CC[C@@H](CC1)Oc1ccc(cc1)C(=O)Nc1nnc(Nc2ccccc2)s1 |r,wU:6.9,3.2,(-9.07,-.95,;-8,-1.56,;-7.99,-2.79,;-6.67,-.78,;-5.33,-1.55,;-4,-.77,;-4,.77,;-5.34,1.53,;-6.67,.76,;-2.67,1.54,;-1.33,.77,;,1.54,;1.33,.77,;1.33,-.77,;,-1.54,;-1.33,-.77,;2.67,-1.54,;3.73,-.93,;2.66,-3.08,;4,-3.86,;4.14,-5.38,;5.64,-5.7,;6.41,-4.36,;7.94,-4.2,;8.57,-2.79,;10.1,-2.63,;10.72,-1.22,;9.81,.03,;8.28,-.14,;7.66,-1.55,;5.38,-3.22,)| Show InChI InChI=1S/C22H22N4O4S/c27-19(24-22-26-25-21(31-22)23-16-4-2-1-3-5-16)14-6-10-17(11-7-14)30-18-12-8-15(9-13-18)20(28)29/h1-7,10-11,15,18H,8-9,12-13H2,(H,23,25)(H,28,29)(H,24,26,27)/t15-,18+ | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Sanofi-aventis R&D
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human DGAT-1 using 1,2,di(cis-9-octadecenoyl)-sn-glycerol as substrate by Microbeta counter |
Bioorg Med Chem Lett 26: 25-32 (2016)
Article DOI: 10.1016/j.bmcl.2015.11.046
BindingDB Entry DOI: 10.7270/Q2Z89GDN |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50313476

(4-(3-(piperidin-1-yl)propyl)-6-(3-(trifluoromethyl...)Show SMILES FC(F)(F)c1cccc(c1)-c1cc(CCCN2CCCCC2)nc(n1)C#N Show InChI InChI=1S/C20H21F3N4/c21-20(22,23)16-7-4-6-15(12-16)18-13-17(25-19(14-24)26-18)8-5-11-27-9-2-1-3-10-27/h4,6-7,12-13H,1-3,5,8-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Corporation
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin K by fluorescence assay |
Bioorg Med Chem Lett 20: 1524-7 (2010)
Article DOI: 10.1016/j.bmcl.2010.01.100
BindingDB Entry DOI: 10.7270/Q24X57ZH |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cathepsin K
(Homo sapiens (Human)) | BDBM50313477

(4-(3-(pentan-3-ylamino)propyl)-6-(3-(trifluorometh...)Show SMILES CCC(CC)NCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C20H23F3N4/c1-3-16(4-2)25-10-6-9-17-12-18(27-19(13-24)26-17)14-7-5-8-15(11-14)20(21,22)23/h5,7-8,11-12,16,25H,3-4,6,9-10H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Schering-Plough Corporation
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin K by fluorescence assay |
Bioorg Med Chem Lett 20: 1524-7 (2010)
Article DOI: 10.1016/j.bmcl.2010.01.100
BindingDB Entry DOI: 10.7270/Q24X57ZH |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Cathepsin K
(Homo sapiens (Human)) | BDBM50313476

(4-(3-(piperidin-1-yl)propyl)-6-(3-(trifluoromethyl...)Show SMILES FC(F)(F)c1cccc(c1)-c1cc(CCCN2CCCCC2)nc(n1)C#N Show InChI InChI=1S/C20H21F3N4/c21-20(22,23)16-7-4-6-15(12-16)18-13-17(25-19(14-24)26-18)8-5-11-27-9-2-1-3-10-27/h4,6-7,12-13H,1-3,5,8-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin K using Z-Phe-Arg-MCA substrate by fluorimetric assay |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cathepsin K
(Homo sapiens (Human)) | BDBM50328878

(3-(2-cyano-6-propylpyrimidin-4-yl)-N-(2-(dimethyla...)Show SMILES CCCc1cc(nc(n1)C#N)-c1cc(cc(c1)C(F)(F)F)C(=O)N(C)CCN(C)C Show InChI InChI=1S/C21H24F3N5O/c1-5-6-17-12-18(27-19(13-25)26-17)14-9-15(11-16(10-14)21(22,23)24)20(30)29(4)8-7-28(2)3/h9-12H,5-8H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin K using Z-Phe-Arg-MCA substrate by fluorimetric assay |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50313477

(4-(3-(pentan-3-ylamino)propyl)-6-(3-(trifluorometh...)Show SMILES CCC(CC)NCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C20H23F3N4/c1-3-16(4-2)25-10-6-9-17-12-18(27-19(13-24)26-17)14-7-5-8-15(11-14)20(21,22)23/h5,7-8,11-12,16,25H,3-4,6,9-10H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin K using Z-Phe-Arg-MCA substrate by fluorimetric assay |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Cathepsin K
(Homo sapiens (Human)) | BDBM50328896

(1-(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyrimid...)Show SMILES NC(=O)C1(CCC1)NCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C20H20F3N5O/c21-20(22,23)14-5-1-4-13(10-14)16-11-15(27-17(12-24)28-16)6-2-9-26-19(18(25)29)7-3-8-19/h1,4-5,10-11,26H,2-3,6-9H2,(H2,25,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin K using Z-Phe-Arg-MCA substrate by fluorimetric assay |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Cathepsin S
(Homo sapiens (Human)) | BDBM50323088
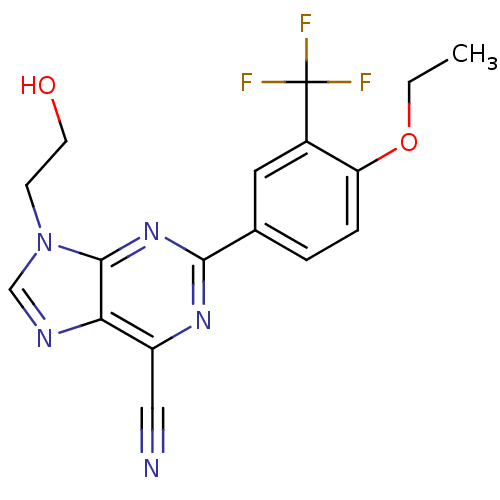
(2-(4-ethoxy-3-(trifluoromethyl)phenyl)-9-(2-hydrox...)Show SMILES CCOc1ccc(cc1C(F)(F)F)-c1nc(C#N)c2ncn(CCO)c2n1 Show InChI InChI=1S/C17H14F3N5O2/c1-2-27-13-4-3-10(7-11(13)17(18,19)20)15-23-12(8-21)14-16(24-15)25(5-6-26)9-22-14/h3-4,7,9,26H,2,5-6H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin S |
Bioorg Med Chem Lett 20: 4447-50 (2010)
Article DOI: 10.1016/j.bmcl.2010.06.049
BindingDB Entry DOI: 10.7270/Q2RJ4JP2 |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50027147

(1-[5-Benzoylamino-6-(4-hydroxy-phenyl)-4-oxo-hexan...)Show SMILES OC(=O)[C@@H]1CCCN1C(=O)CCC(=O)C(Cc1ccc(O)cc1)NC(=O)c1ccccc1 Show InChI InChI=1S/C24H26N2O6/c27-18-10-8-16(9-11-18)15-19(25-23(30)17-5-2-1-3-6-17)21(28)12-13-22(29)26-14-4-7-20(26)24(31)32/h1-3,5-6,8-11,19-20,27H,4,7,12-15H2,(H,25,30)(H,31,32)/t19?,20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50% |
J Med Chem 24: 964-9 (1982)
BindingDB Entry DOI: 10.7270/Q2959J4Z |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50328897

(4-(3-(2-cyano-6-(3-(trifluoromethyl)phenyl)pyrimid...)Show SMILES NC(=O)C1(CCOCC1)NCCCc1cc(nc(n1)C#N)-c1cccc(c1)C(F)(F)F Show InChI InChI=1S/C21H22F3N5O2/c22-21(23,24)15-4-1-3-14(11-15)17-12-16(28-18(13-25)29-17)5-2-8-27-20(19(26)30)6-9-31-10-7-20/h1,3-4,11-12,27H,2,5-10H2,(H2,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin K using Z-Phe-Arg-MCA substrate by fluorimetric assay |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM50027144
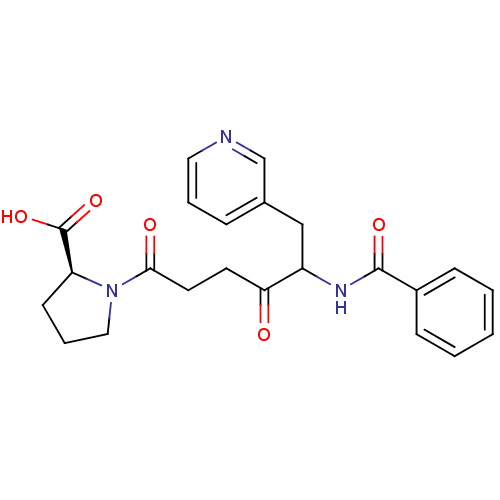
(1-(5-Benzoylamino-4-oxo-6-pyridin-3-yl-hexanoyl)-p...)Show SMILES OC(=O)[C@@H]1CCCN1C(=O)CCC(=O)C(Cc1cccnc1)NC(=O)c1ccccc1 Show InChI InChI=1S/C23H25N3O5/c27-20(10-11-21(28)26-13-5-9-19(26)23(30)31)18(14-16-6-4-12-24-15-16)25-22(29)17-7-2-1-3-8-17/h1-4,6-8,12,15,18-19H,5,9-11,13-14H2,(H,25,29)(H,30,31)/t18?,19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Concentration required to inhibit the activity of Angiotensin I converting enzyme by 50% |
J Med Chem 24: 964-9 (1982)
BindingDB Entry DOI: 10.7270/Q2959J4Z |
More data for this
Ligand-Target Pair | |
Renin
(Homo sapiens (Human)) | BDBM50016033

(CHEMBL3085574 | {1-[1-{1-[2-(1-Benzylcarbamoyl-3-m...)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1c[nH]cn1)NC(=O)[C@H](Cc1ccccc1)NC(=O)OC(C)(C)C)[C@@H](O)CC(=O)N[C@@H](CC(C)C)C(=O)NCc1ccccc1 Show InChI InChI=1S/C41H59N7O7/c1-26(2)18-31(35(49)22-36(50)45-32(19-27(3)4)37(51)43-23-29-16-12-9-13-17-29)46-39(53)34(21-30-24-42-25-44-30)47-38(52)33(20-28-14-10-8-11-15-28)48-40(54)55-41(5,6)7/h8-17,24-27,31-35,49H,18-23H2,1-7H3,(H,42,44)(H,43,51)(H,45,50)(H,46,53)(H,47,52)(H,48,54)/t31-,32-,33-,34-,35-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Warner-Lambert Company
Curated by ChEMBL
| Assay Description
Inhibitory activity against human renin |
J Med Chem 33: 838-45 (1990)
BindingDB Entry DOI: 10.7270/Q2T152M6 |
More data for this
Ligand-Target Pair | |
Integrin alpha-L/Integrin beta-2/Intercellular adhesion molecule 1
(Homo sapiens (Human)) | BDBM50199031
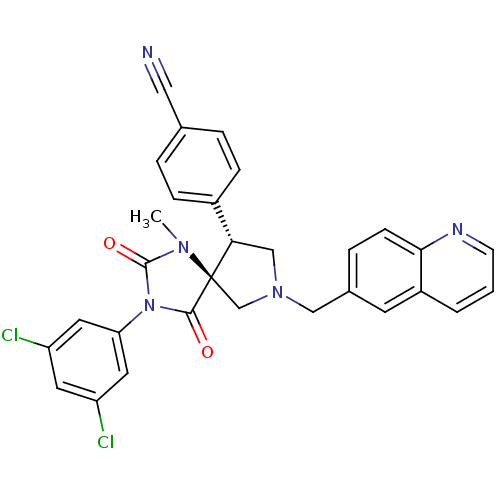
(4-[(5S,9R)-3-(3,5-dichloro-phenyl)-1-methyl-2,4-di...)Show SMILES CN1C(=O)N(C(=O)[C@]11CN(Cc2ccc3ncccc3c2)C[C@H]1c1ccc(cc1)C#N)c1cc(Cl)cc(Cl)c1 Show InChI InChI=1S/C30H23Cl2N5O2/c1-35-29(39)37(25-13-23(31)12-24(32)14-25)28(38)30(35)18-36(17-26(30)21-7-4-19(15-33)5-8-21)16-20-6-9-27-22(11-20)3-2-10-34-27/h2-14,26H,16-18H2,1H3/t26-,30+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Cerep
Curated by ChEMBL
| Assay Description
Inhibition of LFA1-mediated adhesion of T cell to HUVEC |
J Med Chem 49: 6946-9 (2006)
Article DOI: 10.1021/jm0610806
BindingDB Entry DOI: 10.7270/Q2SF2XC3 |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50328884

(4-(3-(methylamino)propyl)-6-(3-(trifluoromethyl)ph...)Show InChI InChI=1S/C16H15F3N4/c1-21-7-3-6-13-9-14(23-15(10-20)22-13)11-4-2-5-12(8-11)16(17,18)19/h2,4-5,8-9,21H,3,6-7H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant cathepsin K using Z-Phe-Arg-MCA substrate by fluorimetric assay |
Bioorg Med Chem Lett 20: 6237-41 (2010)
Article DOI: 10.1016/j.bmcl.2010.08.101
BindingDB Entry DOI: 10.7270/Q2RR1ZFT |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data