 Found 3969 hits with Last Name = 'ryan' and Initial = 'm'
Found 3969 hits with Last Name = 'ryan' and Initial = 'm' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17428
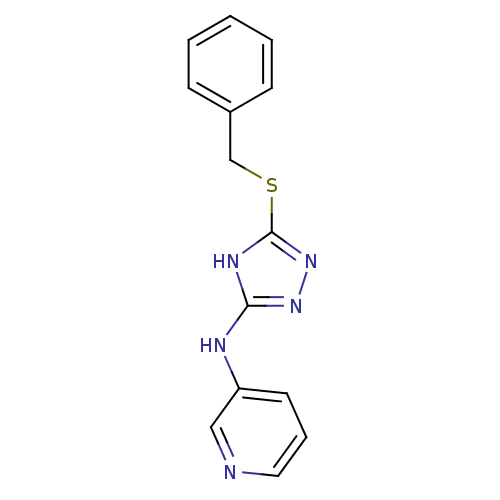
(1,2,4-Triazole Compound, 86 | N-[5-(benzylsulfanyl...)Show InChI InChI=1S/C14H13N5S/c1-2-5-11(6-3-1)10-20-14-17-13(18-19-14)16-12-7-4-8-15-9-12/h1-9H,10H2,(H2,16,17,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17355
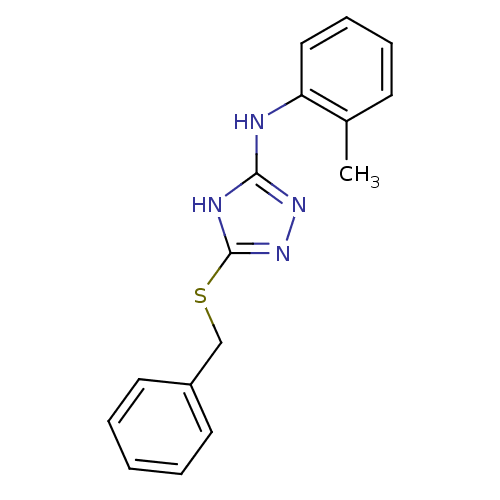
(1,2,4-Triazole Compound, 13 | 5-(benzylsulfanyl)-N...)Show InChI InChI=1S/C16H16N4S/c1-12-7-5-6-10-14(12)17-15-18-16(20-19-15)21-11-13-8-3-2-4-9-13/h2-10H,11H2,1H3,(H2,17,18,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | -58.8 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17388
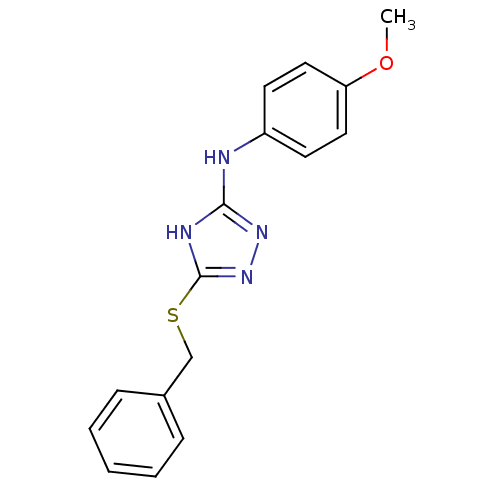
(1,2,4-Triazole Compound, 46 | 5-(benzylsulfanyl)-N...)Show InChI InChI=1S/C16H16N4OS/c1-21-14-9-7-13(8-10-14)17-15-18-16(20-19-15)22-11-12-5-3-2-4-6-12/h2-10H,11H2,1H3,(H2,17,18,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17365
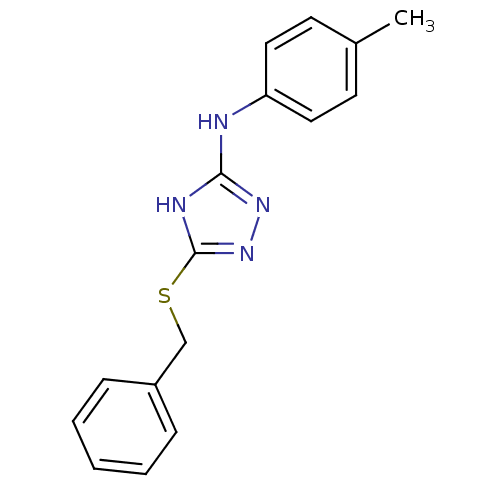
(1,2,4-Triazole Compound, 23 | 5-(benzylsulfanyl)-N...)Show InChI InChI=1S/C16H16N4S/c1-12-7-9-14(10-8-12)17-15-18-16(20-19-15)21-11-13-5-3-2-4-6-13/h2-10H,11H2,1H3,(H2,17,18,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0700 | -57.4 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50344100
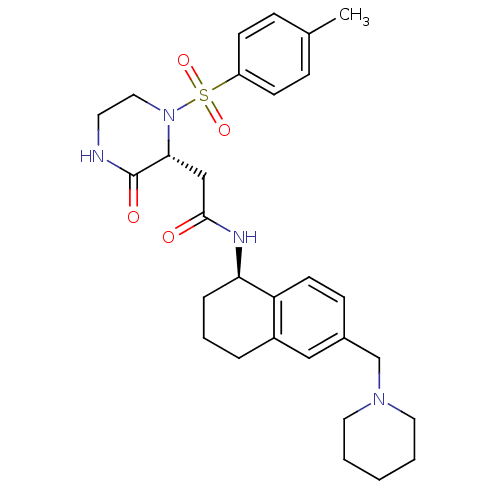
(2-((2R)-1-((4-methylphenyl)sulfonyl)-3-oxo-2-piper...)Show SMILES Cc1ccc(cc1)S(=O)(=O)N1CCNC(=O)[C@H]1CC(=O)N[C@@H]1CCCc2cc(CN3CCCCC3)ccc12 |r| Show InChI InChI=1S/C29H38N4O4S/c1-21-8-11-24(12-9-21)38(36,37)33-17-14-30-29(35)27(33)19-28(34)31-26-7-5-6-23-18-22(10-13-25(23)26)20-32-15-3-2-4-16-32/h8-13,18,26-27H,2-7,14-17,19-20H2,1H3,(H,30,35)(H,31,34)/t26-,27-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human B1 receptor |
J Med Chem 54: 7232-46 (2011)
Article DOI: 10.1021/jm200808v
BindingDB Entry DOI: 10.7270/Q28C9WP1 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech Inc.
Curated by ChEMBL
| Assay Description
Inhibition of EGFR T790M/L858R mutant (unknown origin) by high-throughput biochemical screening |
J Med Chem 57: 10176-91 (2014)
Article DOI: 10.1021/jm501578n
BindingDB Entry DOI: 10.7270/Q2XK8H5B |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of EGFR deletion (746 to 750 residues) mutant (unknown origin) |
Bioorg Med Chem Lett 26: 534-9 (2016)
Article DOI: 10.1016/j.bmcl.2015.11.078
BindingDB Entry DOI: 10.7270/Q2HH6MZQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of wild type EGFR (unknown origin) |
Bioorg Med Chem Lett 26: 534-9 (2016)
Article DOI: 10.1016/j.bmcl.2015.11.078
BindingDB Entry DOI: 10.7270/Q2HH6MZQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged EGFR catalytic domain (669 to 1210 residues) expressed in baculovirus expression system by mass... |
ACS Med Chem Lett 7: 100-4 (2016)
Article DOI: 10.1021/acsmedchemlett.5b00428
BindingDB Entry DOI: 10.7270/Q25T3NCC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech Inc.
Curated by ChEMBL
| Assay Description
Inhibition of wild-type EGFR (unknown origin) by high-throughput biochemical screening |
J Med Chem 57: 10176-91 (2014)
Article DOI: 10.1021/jm501578n
BindingDB Entry DOI: 10.7270/Q2XK8H5B |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17390
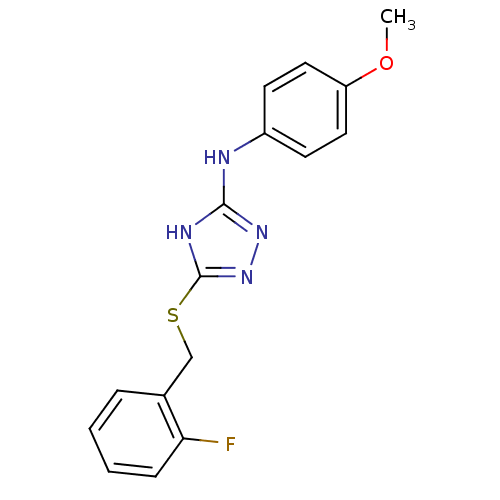
(1,2,4-Triazole Compound, 48 | 5-{[(2-fluorophenyl)...)Show InChI InChI=1S/C16H15FN4OS/c1-22-13-8-6-12(7-9-13)18-15-19-16(21-20-15)23-10-11-4-2-3-5-14(11)17/h2-9H,10H2,1H3,(H2,18,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17362

(1,2,4-Triazole Compound, 20 | 5-[(3-methylbut-2-en...)Show SMILES CC(C)=CCSc1nnc(Nc2ccccc2C)[nH]1 |(5.94,4.3,;4.59,5.03,;4.55,6.57,;3.28,4.22,;1.81,4.7,;.67,3.67,;-.8,4.15,;-1.27,5.61,;-2.81,5.61,;-3.29,4.15,;-4.75,3.67,;-5.9,4.7,;-5.13,6.03,;-5.9,7.37,;-7.44,7.37,;-8.21,6.03,;-7.44,4.7,;-8.21,3.37,;-2.04,3.24,)| Show InChI InChI=1S/C14H18N4S/c1-10(2)8-9-19-14-16-13(17-18-14)15-12-7-5-4-6-11(12)3/h4-8H,9H2,1-3H3,(H2,15,16,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.150 | -55.5 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17395
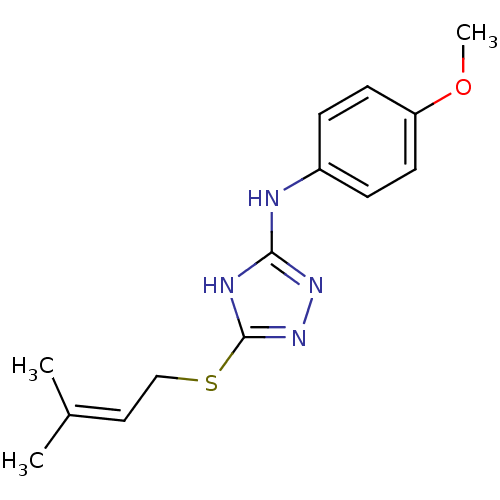
(1,2,4-Triazole Compound, 53 | N-(4-methoxyphenyl)-...)Show SMILES COc1ccc(Nc2nnc(SCC=C(C)C)[nH]2)cc1 |(-9.75,8.7,;-8.21,8.7,;-7.44,7.37,;-8.21,6.03,;-7.44,4.7,;-5.9,4.7,;-4.75,3.67,;-3.29,4.15,;-2.81,5.61,;-1.27,5.61,;-.8,4.15,;.67,3.67,;1.81,4.7,;3.28,4.22,;4.51,5.15,;5.93,4.55,;4.32,6.68,;-2.04,3.24,;-5.13,6.03,;-5.9,7.37,)| Show InChI InChI=1S/C14H18N4OS/c1-10(2)8-9-20-14-16-13(17-18-14)15-11-4-6-12(19-3)7-5-11/h4-8H,9H2,1-3H3,(H2,15,16,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Nuclear receptor ROR-gamma
(Homo sapiens (Human)) | BDBM504229

(N-[2-[(2R)-2-Fluoro-3-hydroxy- 3-methyl-butyl]-6-i...)Show SMILES CC(C)Oc1cc2C(=O)N(C[C@@H](F)C(C)(C)O)Cc2cc1NC(=O)c1cnn2cccnc12 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q24B34FX |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50141636
(CHEMBL3758502)Show SMILES CC1(C)OCc2c1nc(nc2Nc1n[nH]c2ccccc12)-c1cn[nH]c1 Show InChI InChI=1S/C18H17N7O/c1-18(2)14-12(9-26-18)16(22-15(21-14)10-7-19-20-8-10)23-17-11-5-3-4-6-13(11)24-25-17/h3-8H,9H2,1-2H3,(H,19,20)(H2,21,22,23,24,25) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of EGFR deletion (746 to 750 residues) mutant (unknown origin) |
Bioorg Med Chem Lett 26: 534-9 (2016)
Article DOI: 10.1016/j.bmcl.2015.11.078
BindingDB Entry DOI: 10.7270/Q2HH6MZQ |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17358
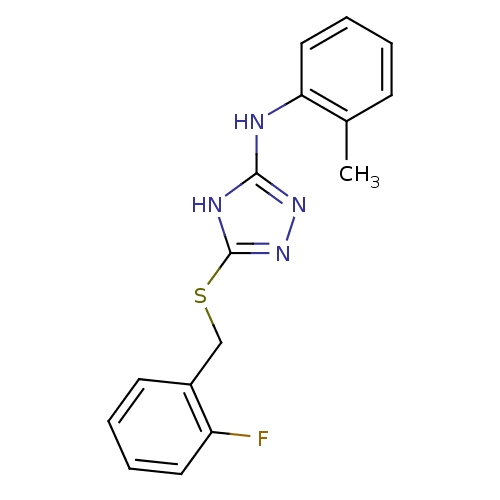
(1,2,4-Triazole Compound, 16 | 5-{[(2-fluorophenyl)...)Show InChI InChI=1S/C16H15FN4S/c1-11-6-2-5-9-14(11)18-15-19-16(21-20-15)22-10-12-7-3-4-8-13(12)17/h2-9H,10H2,1H3,(H2,18,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.230 | -54.5 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Nuclear receptor ROR-gamma
(Homo sapiens (Human)) | BDBM504219
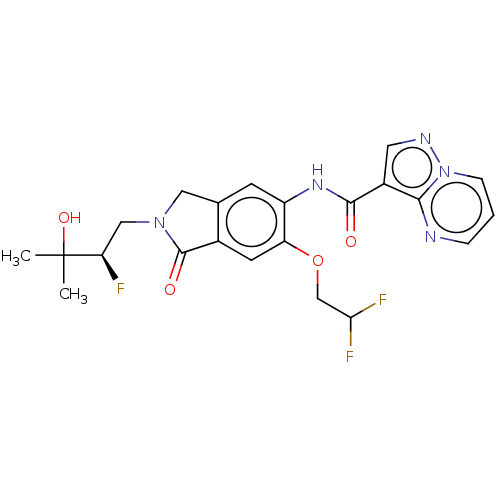
((R)-N-(6-(2,2-Difluoroethoxy)- 2-(2-fluoro-3-hydro...)Show SMILES CC(C)(O)[C@H](F)CN1Cc2cc(NC(=O)c3cnn4cccnc34)c(OCC(F)F)cc2C1=O |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q24B34FX |
More data for this
Ligand-Target Pair | |
Nuclear receptor ROR-gamma
(Homo sapiens (Human)) | BDBM504215

(N-[6-(3-Fluorocyclobutoxy)-2- [(2R)-2-fluoro-3-hyd...)Show SMILES CC(C)(O)[C@H](F)CN1Cc2cc(NC(=O)c3cnn4cccnc34)c(O[C@@H]3C[C@H](F)C3)cc2C1=O |r,wU:26.27,28.30,wD:4.4,(-7.2,.34,;-7.24,1.88,;-8.73,1.48,;-8.01,3.22,;-5.7,1.88,;-4.93,3.22,;-4.93,.55,;-3.39,.55,;-2.49,1.79,;-1.02,1.32,;.31,2.09,;1.64,1.32,;2.98,2.09,;2.98,3.63,;1.64,4.4,;4.31,4.4,;4.47,5.93,;5.98,6.25,;6.75,4.92,;8.25,4.6,;8.73,3.13,;7.7,1.99,;6.19,2.31,;5.72,3.77,;1.64,-.22,;2.98,-.99,;2.98,-2.53,;1.89,-3.62,;2.98,-4.71,;2.98,-6.25,;4.07,-3.62,;.31,-.99,;-1.02,-.22,;-2.49,-.7,;-2.96,-2.16,)| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q24B34FX |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17430
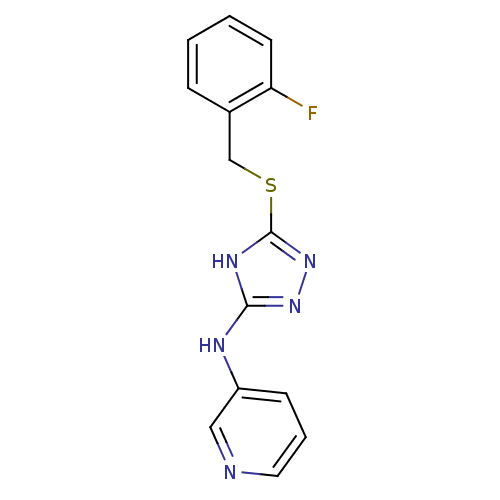
(1,2,4-Triazole Compound, 88 | N-(5-{[(2-fluorophen...)Show InChI InChI=1S/C14H12FN5S/c15-12-6-2-1-4-10(12)9-21-14-18-13(19-20-14)17-11-5-3-7-16-8-11/h1-8H,9H2,(H2,17,18,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50355063
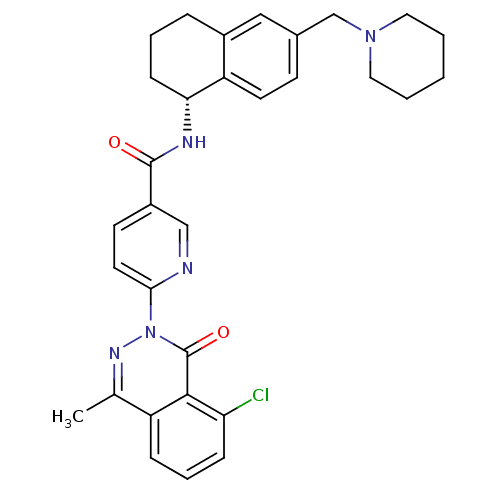
(CHEMBL1834619)Show SMILES Cc1nn(-c2ccc(cn2)C(=O)N[C@@H]2CCCc3cc(CN4CCCCC4)ccc23)c(=O)c2c(Cl)cccc12 |r| Show InChI InChI=1S/C31H32ClN5O2/c1-20-24-8-6-9-26(32)29(24)31(39)37(35-20)28-14-12-23(18-33-28)30(38)34-27-10-5-7-22-17-21(11-13-25(22)27)19-36-15-3-2-4-16-36/h6,8-9,11-14,17-18,27H,2-5,7,10,15-16,19H2,1H3,(H,34,38)/t27-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human B1 receptor |
J Med Chem 54: 7232-46 (2011)
Article DOI: 10.1021/jm200808v
BindingDB Entry DOI: 10.7270/Q28C9WP1 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of EGFR L858R mutant (unknown origin) |
Bioorg Med Chem Lett 26: 534-9 (2016)
Article DOI: 10.1016/j.bmcl.2015.11.078
BindingDB Entry DOI: 10.7270/Q2HH6MZQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM5446

(CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...)Show InChI InChI=1S/C22H23N3O4/c1-4-16-6-5-7-17(12-16)25-22-18-13-20(28-10-8-26-2)21(29-11-9-27-3)14-19(18)23-15-24-22/h1,5-7,12-15H,8-11H2,2-3H3,(H,23,24,25) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech Inc.
Curated by ChEMBL
| Assay Description
Inhibition of EGFR T790M/del746 to 750 mutant (unknown origin) by high-throughput biochemical screening |
J Med Chem 57: 10176-91 (2014)
Article DOI: 10.1021/jm501578n
BindingDB Entry DOI: 10.7270/Q2XK8H5B |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50613695
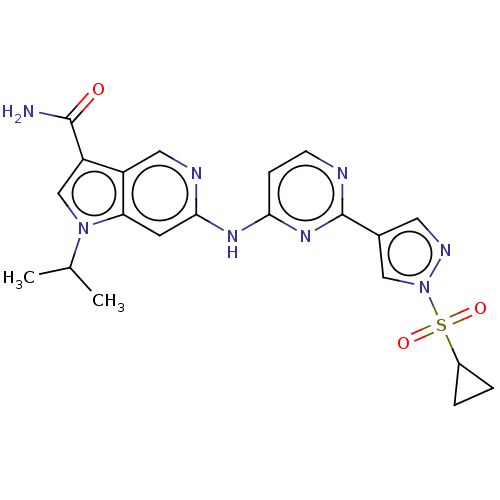
(CHEMBL5274166) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | 0.350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Nuclear receptor ROR-gamma
(Homo sapiens (Human)) | BDBM504233

((R)-N-(6-Ethoxy-2-(2-fluoro-3- hydroxy-3-methylbut...)Show SMILES CCOc1cc2C(=O)N(C[C@@H](F)C(C)(C)O)Cc2cc1NC(=O)c1cnn2cccnc12 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q24B34FX |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50141902

(CHEMBL3758602)Show SMILES CC1(C)OCc2c1nc(nc2Nc1n[nH]c2c(Cl)cccc12)-c1cn[nH]c1 Show InChI InChI=1S/C18H16ClN7O/c1-18(2)14-11(8-27-18)16(23-15(22-14)9-6-20-21-7-9)24-17-10-4-3-5-12(19)13(10)25-26-17/h3-7H,8H2,1-2H3,(H,20,21)(H2,22,23,24,25,26) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of EGFR deletion (746 to 750 residues) mutant (unknown origin) |
Bioorg Med Chem Lett 26: 534-9 (2016)
Article DOI: 10.1016/j.bmcl.2015.11.078
BindingDB Entry DOI: 10.7270/Q2HH6MZQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17411

(1,2,4-Triazole Compound, 69 | methyl 4-[(5-{[(2-fl...)Show InChI InChI=1S/C17H15FN4O2S/c1-24-15(23)11-6-8-13(9-7-11)19-16-20-17(22-21-16)25-10-12-4-2-3-5-14(12)18/h2-9H,10H2,1H3,(H2,19,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Nuclear receptor ROR-gamma
(Homo sapiens (Human)) | BDBM504232

((R)-N-(2-(2-Fluoro-3-hydroxy- 3-methylbutyl)-6-iso...)Show SMILES CC(C)Oc1cc2C(=O)N(C[C@@H](F)C(C)(C)O)Cc2cc1NC(=O)c1cnn2cc(C)cnc12 |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q24B34FX |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17377

(1,2,4-Triazole Compound, 35 | 5-(benzylsulfanyl)-N...)Show InChI InChI=1S/C15H13ClN4S/c16-12-6-8-13(9-7-12)17-14-18-15(20-19-14)21-10-11-4-2-1-3-5-11/h1-9H,10H2,(H2,17,18,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Nuclear receptor ROR-gamma
(Homo sapiens (Human)) | BDBM504234

(US11034698, Example 83 | US11034698, Example 84)Show SMILES CC(C)(O)[C@H](F)CN1Cc2cc(NC(=O)c3cnn4cccnc34)c(O[C@@H]3CCOC3)cc2C1=O |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q24B34FX |
More data for this
Ligand-Target Pair | |
Nuclear receptor ROR-gamma
(Homo sapiens (Human)) | BDBM504239
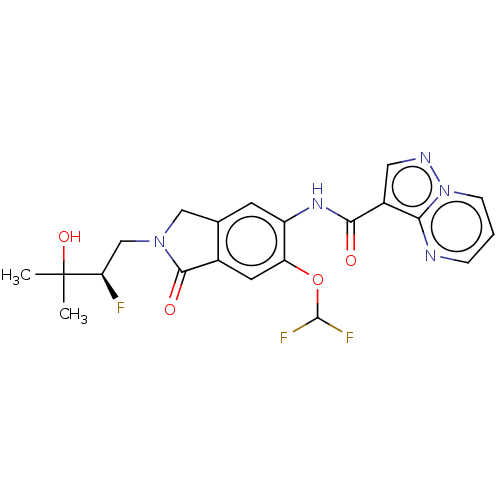
((R)-N-(6-(Difluoromethoxy)-2- (2-fluoro-3-hydroxy-...)Show SMILES CC(C)(O)[C@H](F)CN1Cc2cc(NC(=O)c3cnn4cccnc34)c(OC(F)F)cc2C1=O |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q24B34FX |
More data for this
Ligand-Target Pair | |
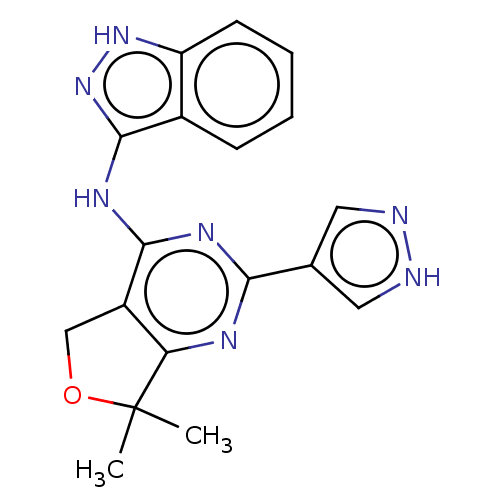
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50141636

(CHEMBL3758502)Show SMILES CC1(C)OCc2c1nc(nc2Nc1n[nH]c2ccccc12)-c1cn[nH]c1 Show InChI InChI=1S/C18H17N7O/c1-18(2)14-12(9-26-18)16(22-15(21-14)10-7-19-20-8-10)23-17-11-5-3-4-6-13(11)24-25-17/h3-8H,9H2,1-2H3,(H,19,20)(H2,21,22,23,24,25) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of EGFR L858R mutant (unknown origin) |
Bioorg Med Chem Lett 26: 534-9 (2016)
Article DOI: 10.1016/j.bmcl.2015.11.078
BindingDB Entry DOI: 10.7270/Q2HH6MZQ |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17352
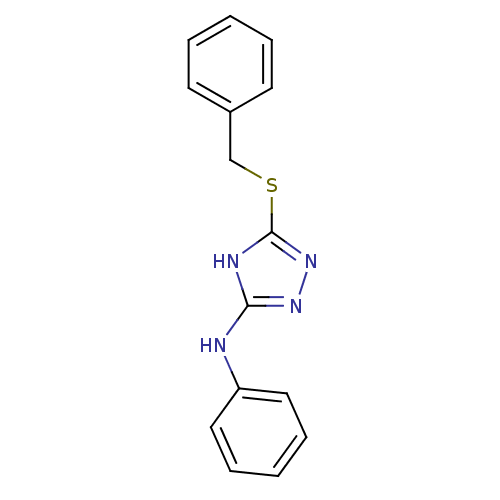
(1,2,4-Triazole Compound, 6 | 5-(benzylsulfanyl)-N-...)Show InChI InChI=1S/C15H14N4S/c1-3-7-12(8-4-1)11-20-15-17-14(18-19-15)16-13-9-5-2-6-10-13/h1-10H,11H2,(H2,16,17,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | -52.6 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17416
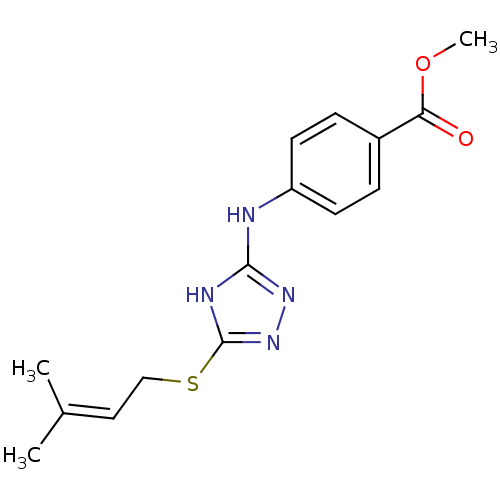
(1,2,4-Triazole Compound, 74 | methyl 4-({5-[(3-met...)Show SMILES COC(=O)c1ccc(Nc2nnc(SCC=C(C)C)[nH]2)cc1 |(-10.52,10.03,;-9.75,8.7,;-8.21,8.7,;-7.44,10.03,;-7.44,7.37,;-8.21,6.03,;-7.44,4.7,;-5.9,4.7,;-4.75,3.67,;-3.29,4.15,;-2.81,5.61,;-1.27,5.61,;-.8,4.15,;.67,3.67,;1.81,4.7,;3.28,4.22,;4.42,5.25,;5.89,4.78,;4.1,6.76,;-2.04,3.24,;-5.13,6.03,;-5.9,7.37,)| Show InChI InChI=1S/C15H18N4O2S/c1-10(2)8-9-22-15-17-14(18-19-15)16-12-6-4-11(5-7-12)13(20)21-3/h4-8H,9H2,1-3H3,(H2,16,17,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Interleukin-1 receptor-associated kinase 4
(Homo sapiens (Human)) | BDBM50514930

(CHEMBL4464832)Show SMILES CN(C)c1nc2cc(N3CCC(CO)CC3)c(NC(=O)c3cnn4cccnc34)cc2s1 Show InChI InChI=1S/C22H25N7O2S/c1-27(2)22-26-17-10-18(28-8-4-14(13-30)5-9-28)16(11-19(17)32-22)25-21(31)15-12-24-29-7-3-6-23-20(15)29/h3,6-7,10-12,14,30H,4-5,8-9,13H2,1-2H3,(H,25,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant full length His6-tagged IRAK4 expressed in baculovirus expression system using H-KKARFSRFAGSSPSQSSMVAR as substrate i... |
J Med Chem 62: 6223-6240 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00439
BindingDB Entry DOI: 10.7270/Q2CC1419 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50036257
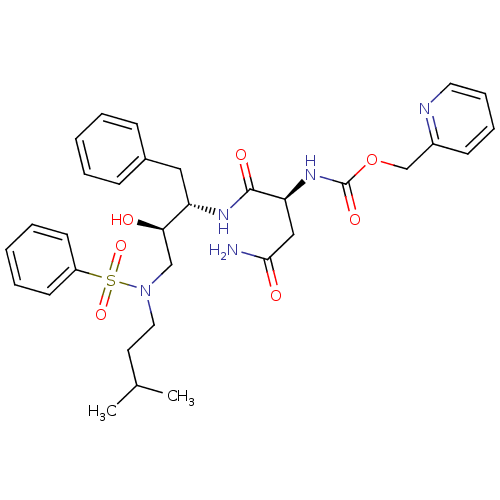
(((S)-1-{(1S,2R)-3-[Benzenesulfonyl-(3-methyl-butyl...)Show SMILES CC(C)CCN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)OCc1ccccn1)S(=O)(=O)c1ccccc1 Show InChI InChI=1S/C32H41N5O7S/c1-23(2)16-18-37(45(42,43)26-14-7-4-8-15-26)21-29(38)27(19-24-11-5-3-6-12-24)35-31(40)28(20-30(33)39)36-32(41)44-22-25-13-9-10-17-34-25/h3-15,17,23,27-29,38H,16,18-22H2,1-2H3,(H2,33,39)(H,35,40)(H,36,41)/t27-,28-,29+/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Discovery Research
Curated by ChEMBL
| Assay Description
Binding affinity of the compound for HIV-1 protease was determined |
J Med Chem 38: 581-4 (1995)
BindingDB Entry DOI: 10.7270/Q2SF2V61 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50141610
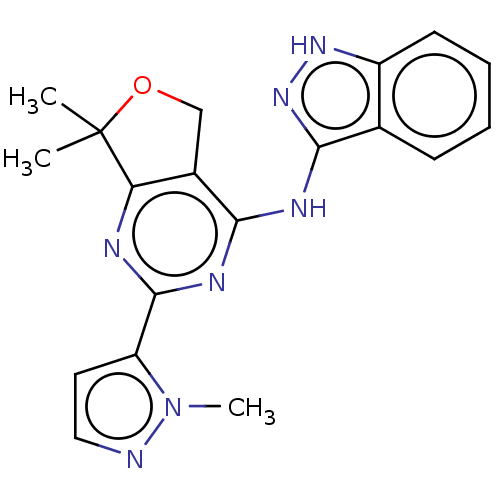
(CHEMBL3759096)Show SMILES Cn1nccc1-c1nc2c(COC2(C)C)c(Nc2n[nH]c3ccccc23)n1 Show InChI InChI=1S/C19H19N7O/c1-19(2)15-12(10-27-19)16(23-18(21-15)14-8-9-20-26(14)3)22-17-11-6-4-5-7-13(11)24-25-17/h4-9H,10H2,1-3H3,(H2,21,22,23,24,25) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| <0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of EGFR L858R mutant (unknown origin) |
Bioorg Med Chem Lett 26: 534-9 (2016)
Article DOI: 10.1016/j.bmcl.2015.11.078
BindingDB Entry DOI: 10.7270/Q2HH6MZQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50141610

(CHEMBL3759096)Show SMILES Cn1nccc1-c1nc2c(COC2(C)C)c(Nc2n[nH]c3ccccc23)n1 Show InChI InChI=1S/C19H19N7O/c1-19(2)15-12(10-27-19)16(23-18(21-15)14-8-9-20-26(14)3)22-17-11-6-4-5-7-13(11)24-25-17/h4-9H,10H2,1-3H3,(H2,21,22,23,24,25) | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| <0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of EGFR deletion (746 to 750 residues) mutant (unknown origin) |
Bioorg Med Chem Lett 26: 534-9 (2016)
Article DOI: 10.1016/j.bmcl.2015.11.078
BindingDB Entry DOI: 10.7270/Q2HH6MZQ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Interleukin-1 receptor-associated kinase 4
(Homo sapiens (Human)) | BDBM50514929
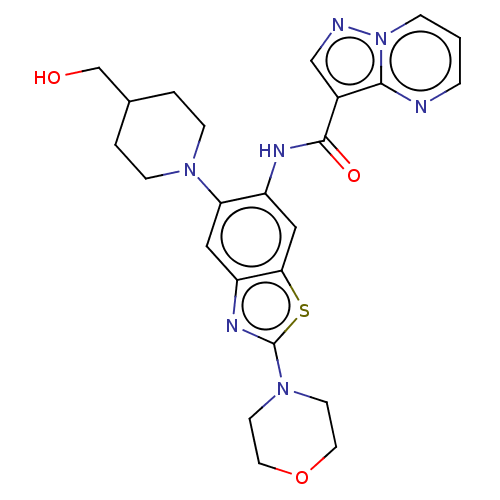
(CHEMBL4474636)Show SMILES OCC1CCN(CC1)c1cc2nc(sc2cc1NC(=O)c1cnn2cccnc12)N1CCOCC1 Show InChI InChI=1S/C24H27N7O3S/c32-15-16-2-6-29(7-3-16)20-12-19-21(35-24(28-19)30-8-10-34-11-9-30)13-18(20)27-23(33)17-14-26-31-5-1-4-25-22(17)31/h1,4-5,12-14,16,32H,2-3,6-11,15H2,(H,27,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant full length His6-tagged IRAK4 expressed in baculovirus expression system using H-KKARFSRFAGSSPSQSSMVAR as substrate i... |
J Med Chem 62: 6223-6240 (2019)
Article DOI: 10.1021/acs.jmedchem.9b00439
BindingDB Entry DOI: 10.7270/Q2CC1419 |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17431
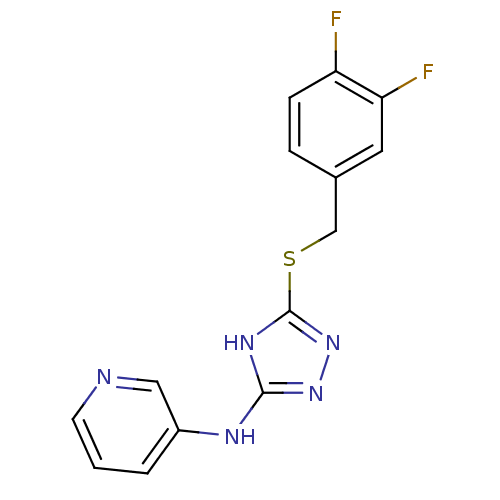
(1,2,4-Triazole Compound, 89 | N-(5-{[(3,4-difluoro...)Show InChI InChI=1S/C14H11F2N5S/c15-11-4-3-9(6-12(11)16)8-22-14-19-13(20-21-14)18-10-2-1-5-17-7-10/h1-7H,8H2,(H2,18,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Nuclear receptor ROR-gamma
(Homo sapiens (Human)) | BDBM504218

(N-[6-(3-Chlorocyclobutoxy)-2- [(2R)-2-fluoro-3-hyd...)Show SMILES CC(C)(O)[C@H](F)CN1Cc2cc(NC(=O)c3cnn4cccnc34)c(O[C@@H]3C[C@H](Cl)C3)cc2C1=O |r,wU:26.27,28.30,wD:4.4,(-7.2,.34,;-7.24,1.88,;-8.73,1.48,;-8.01,3.22,;-5.7,1.88,;-4.93,3.22,;-4.93,.55,;-3.39,.55,;-2.49,1.79,;-1.02,1.32,;.31,2.09,;1.64,1.32,;2.98,2.09,;2.98,3.63,;1.64,4.4,;4.31,4.4,;4.47,5.93,;5.98,6.25,;6.75,4.92,;8.25,4.6,;8.73,3.13,;7.7,1.99,;6.19,2.31,;5.72,3.77,;1.64,-.22,;2.98,-.99,;2.98,-2.53,;1.89,-3.62,;2.98,-4.71,;2.98,-6.25,;4.07,-3.62,;.31,-.99,;-1.02,-.22,;-2.49,-.7,;-2.96,-2.16,)| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q24B34FX |
More data for this
Ligand-Target Pair | |
Methionine aminopeptidase 2
(Homo sapiens (Human)) | BDBM17385

(1,2,4-Triazole Compound, 43 | N-(4-chlorophenyl)-5...)Show SMILES CC(C)=CCSc1nnc(Nc2ccc(Cl)cc2)[nH]1 |(5.94,4.35,;4.57,5.06,;4.5,6.59,;3.28,4.22,;1.81,4.7,;.67,3.67,;-.8,4.15,;-1.27,5.61,;-2.81,5.61,;-3.29,4.15,;-4.75,3.67,;-5.9,4.7,;-5.13,6.03,;-5.9,7.37,;-7.44,7.37,;-8.21,8.7,;-8.21,6.03,;-7.44,4.7,;-2.04,3.24,)| Show InChI InChI=1S/C13H15ClN4S/c1-9(2)7-8-19-13-16-12(17-18-13)15-11-5-3-10(14)4-6-11/h3-7H,8H2,1-2H3,(H2,15,16,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GSK
| Assay Description
MetAP2 activity was monitored by measuring the initial velocity of turnover of the artificial substrate Met-AMC. Assays were performed in 96-well mi... |
J Med Chem 50: 3777-85 (2007)
Article DOI: 10.1021/jm061182w
BindingDB Entry DOI: 10.7270/Q2B856D7 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50498973
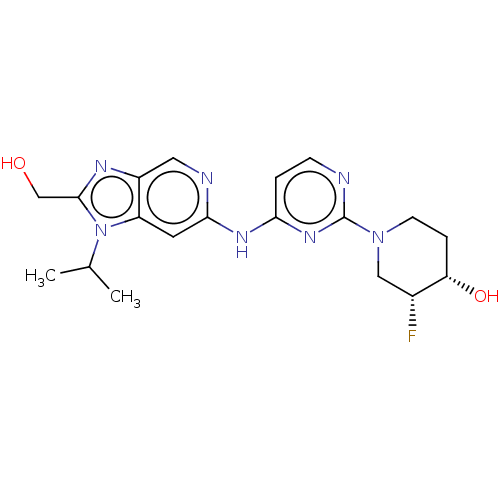
(CHEMBL3734934)Show SMILES CC(C)n1c(CO)nc2cnc(Nc3ccnc(n3)N3CC[C@H](O)[C@H](F)C3)cc12 |r| Show InChI InChI=1S/C19H24FN7O2/c1-11(2)27-14-7-17(22-8-13(14)23-18(27)10-28)24-16-3-5-21-19(25-16)26-6-4-15(29)12(20)9-26/h3,5,7-8,11-12,15,28-29H,4,6,9-10H2,1-2H3,(H,21,22,24,25)/t12-,15+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >0.530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of human wild type EGFR using Fl-EEPLYWSFPAKKK-CONH2 as substrate preincubated for 30 mins followed by addition of substrate measured afte... |
J Med Chem 58: 8877-95 (2015)
Article DOI: 10.1021/acs.jmedchem.5b01412
BindingDB Entry DOI: 10.7270/Q2833W1G |
More data for this
Ligand-Target Pair | |
Nuclear receptor ROR-gamma
(Homo sapiens (Human)) | BDBM504238

((R)-N-(2-(2-Fluoro-3-hydroxy- 3-methylbutyl)-6-(ox...)Show SMILES CC(C)(O)[C@H](F)CN1Cc2cc(NC(=O)c3cnn4cccnc34)c(OC3COC3)cc2C1=O |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 0.590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
The IRAK4 reaction conditions were optimized using an IRAK1-derived peptide (sequence H-KKARFSRFAGSSPSQSSMVAR) to provide a linear reaction rate over... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q24B34FX |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50613687
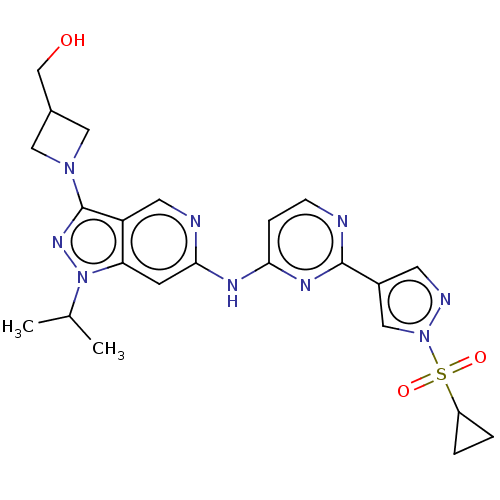
(CHEMBL5285503) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | <0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50613688

(CHEMBL5282716) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| UniChem
| | <0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50357674
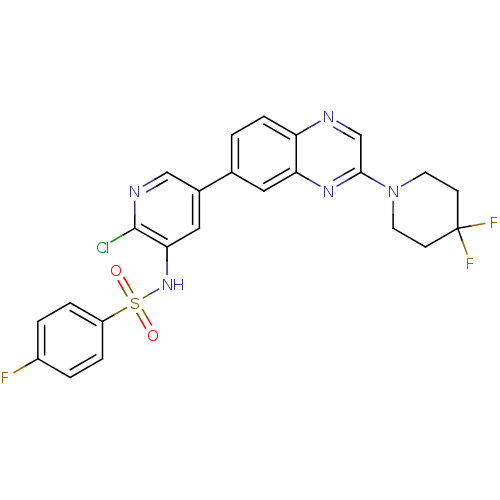
(CHEMBL1914739)Show SMILES Fc1ccc(cc1)S(=O)(=O)Nc1cc(cnc1Cl)-c1ccc2ncc(nc2c1)N1CCC(F)(F)CC1 Show InChI InChI=1S/C24H19ClF3N5O2S/c25-23-21(32-36(34,35)18-4-2-17(26)3-5-18)12-16(13-30-23)15-1-6-19-20(11-15)31-22(14-29-19)33-9-7-24(27,28)8-10-33/h1-6,11-14,32H,7-10H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of N-terminus polyHis-tagged human recombinant PI3Kalpha expressed in baculovirus infected insect Sf9 cells using phosphoinositol-4,5-bisp... |
J Med Chem 54: 4735-51 (2011)
Article DOI: 10.1021/jm200386s
BindingDB Entry DOI: 10.7270/Q2FB53C5 |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50355062
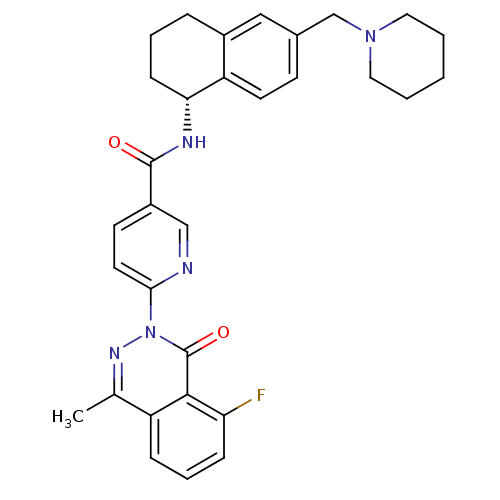
(CHEMBL1834752)Show SMILES Cc1nn(-c2ccc(cn2)C(=O)N[C@@H]2CCCc3cc(CN4CCCCC4)ccc23)c(=O)c2c(F)cccc12 |r| Show InChI InChI=1S/C31H32FN5O2/c1-20-24-8-6-9-26(32)29(24)31(39)37(35-20)28-14-12-23(18-33-28)30(38)34-27-10-5-7-22-17-21(11-13-25(22)27)19-36-15-3-2-4-16-36/h6,8-9,11-14,17-18,27H,2-5,7,10,15-16,19H2,1H3,(H,34,38)/t27-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human B1 receptor |
J Med Chem 54: 7232-46 (2011)
Article DOI: 10.1021/jm200808v
BindingDB Entry DOI: 10.7270/Q28C9WP1 |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50141636

(CHEMBL3758502)Show SMILES CC1(C)OCc2c1nc(nc2Nc1n[nH]c2ccccc12)-c1cn[nH]c1 Show InChI InChI=1S/C18H17N7O/c1-18(2)14-12(9-26-18)16(22-15(21-14)10-7-19-20-8-10)23-17-11-5-3-4-6-13(11)24-25-17/h3-8H,9H2,1-2H3,(H,19,20)(H2,21,22,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Genentech
Curated by ChEMBL
| Assay Description
Inhibition of wild type EGFR (unknown origin) |
Bioorg Med Chem Lett 26: 534-9 (2016)
Article DOI: 10.1016/j.bmcl.2015.11.078
BindingDB Entry DOI: 10.7270/Q2HH6MZQ |
More data for this
Ligand-Target Pair | |
B1 bradykinin receptor
(Homo sapiens (Human)) | BDBM50355057
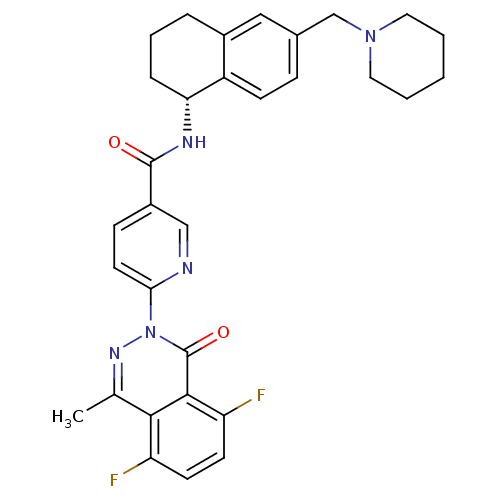
(CHEMBL1834751)Show SMILES Cc1nn(-c2ccc(cn2)C(=O)N[C@@H]2CCCc3cc(CN4CCCCC4)ccc23)c(=O)c2c(F)ccc(F)c12 |r| Show InChI InChI=1S/C31H31F2N5O2/c1-19-28-24(32)11-12-25(33)29(28)31(40)38(36-19)27-13-9-22(17-34-27)30(39)35-26-7-5-6-21-16-20(8-10-23(21)26)18-37-14-3-2-4-15-37/h8-13,16-17,26H,2-7,14-15,18H2,1H3,(H,35,39)/t26-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Antagonist activity at human B1 receptor |
J Med Chem 54: 7232-46 (2011)
Article DOI: 10.1021/jm200808v
BindingDB Entry DOI: 10.7270/Q28C9WP1 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50357645
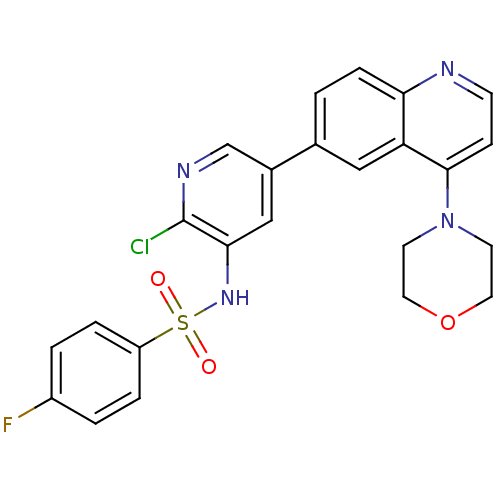
(CHEMBL1738719)Show SMILES Fc1ccc(cc1)S(=O)(=O)Nc1cc(cnc1Cl)-c1ccc2nccc(N3CCOCC3)c2c1 Show InChI InChI=1S/C24H20ClFN4O3S/c25-24-22(29-34(31,32)19-4-2-18(26)3-5-19)14-17(15-28-24)16-1-6-21-20(13-16)23(7-8-27-21)30-9-11-33-12-10-30/h1-8,13-15,29H,9-12H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of N-terminus polyHis-tagged human recombinant PI3Kalpha expressed in baculovirus infected insect Sf9 cells using phosphoinositol-4,5-bisp... |
J Med Chem 54: 4735-51 (2011)
Article DOI: 10.1021/jm200386s
BindingDB Entry DOI: 10.7270/Q2FB53C5 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data